Abstract
OBJECTIVE
In the Multi-Ethnic Study of Atherosclerosis (MESA), we evaluated associations of baseline levels of a lipoprotein-based insulin resistance (IR) index (LP-IR), IR-related lipoprotein particles, mean particle sizes, and lipids, with incident type 2 diabetes, independent of confounders, glucose, insulin, and HOMA-IR.
RESEARCH DESIGN AND METHODS
Among 5,314 adults aged 45–84 years without baseline diabetes or cardiovascular disease, 656 cases of diabetes were identified during a mean follow-up of 7.7 years. Lipoprotein particle concentrations, size, and LP-IR were determined by nuclear magnetic resonance spectroscopy of stored baseline plasma. Potential effect modification, by race/ethnicity, sex, baseline use of lipid-lowering medications or hormone therapy, or glucose strata (<90, 90–99, and ≥100 mg/dL), was also evaluated.
RESULTS
Higher levels of LP-IR, large VLDL particles (VLDL-P), small LDL particles, triglycerides (TG), and TG–to–HDL cholesterol (HDL-C) ratio and lower levels of large HDL particles, smaller HDL and LDL size, and larger VLDL size were significantly associated with incident diabetes adjusted for confounders and glucose or insulin. These also were similar by race/ethnicity, sex, and treatment group. Associations were similar for LP-IR, large VLDL-P, mean VLDL size, TG, and TG–to–HDL-C ratio; they persisted for LP-IR, large VLDL-P, or mean VLDL size adjusted for HOMA-IR or TG–to–HDL-C ratio and glucose but not for the TG–to–HDL-C ratio adjusted for LP-IR or for HOMA-IR or insulin if adjusted for LP-IR and glucose.
CONCLUSIONS
Among ethnically diverse men and women, LP-IR, large VLDL-P, large VLDL size, TG, and TG–to–HDL-C ratio were associated with incident diabetes independent of established risk factors, glucose, insulin, or HOMA-IR, as well as the use of lipid-lowering medications or hormone therapy.
Introduction
The identification of early predictors of type 2 diabetes is important given the potential to prevent or delay the onset of diabetes by lifestyle modification and/or medication (1). The higher triglyceride (TG) and lower HDL cholesterol (HDL-C) levels that characterize diabetes often precede it because the reduction in insulin-mediated suppression of VLDL particle (VLDL-P) secretion occurs independent of, and often before, the reduction in the suppression of hepatic glucose production by insulin (2). The resulting hypersecretion of large, triglyceride-rich VLDL-P causes hypertriglyceridemia; subsequent smaller, cholesterol-depleted HDL particles (HDL-P) and LDL particles (LDL-P); and lower levels of HDL-C and LDL cholesterol (2). Measured by nuclear magnetic resonance (NMR) spectroscopy, higher levels of large VLDL-P and small LDL-P, lower levels of large HDL-P, smaller mean LDL and HDL particle size, and larger mean VLDL particle size are associated with insulin resistance (3–5) and incident diabetes (6–8), often independent of established risk factors, lipids, glucose, insulin resistance or sensitivity, or HbA1c. A Lipoprotein Insulin Resistance (LP-IR; LipoScience) index has recently been developed; it combines these six lipoprotein parameters, weighted by strength of correlations with HOMA-insulin resistance (IR) (9,10). LP-IR is cross-sectionally correlated with insulin sensitivity (Si), HOMA-IR (11), and the glucose disposal rate from a hyperinsulinemic-euglycemic clamp (10), but the association of LP-IR with incident type 2 diabetes has not yet been reported.
In addition to reflecting IR, lipoproteins and lipids may also directly contribute to diabetes pathogenesis via effects on β-cell function, insulin production, and glucose homeostasis (12–14). Furthermore, statins are associated with a slightly increased risk of diabetes (15–19), and higher baseline levels of TG and glucose were independently related to incident type 2 diabetes in three large randomized statin trials (20). However, prior studies of lipoprotein-related diabetes risk have not explicitly evaluated potential effect modification by use of lipid-lowering medications or hormone therapy (HT), which also alters levels of lipid and lipoprotein particles (21,22). Finally, despite ethnic differences in diabetes risk (23,24), few studies (6) have evaluated sex or race/ethnicity variation in lipoprotein-related risk of diabetes. Therefore, we hypothesized that in a multiethnic cohort of men and women, higher baseline levels of LP-IR, large VLDL-P, small LDL-P, small HDL-P, and TG; lower levels of large HDL-P and HDL-C; and larger mean VLDL particle size and smaller mean LDL and HDL particle size would be associated with incident diabetes, independent of established risk factors and potential confounders. We further hypothesized that lipoprotein associations with diabetes would be partially independent of baseline levels of glucose or insulin and of baseline HOMA-IR; would be similar for individuals using lipid-lowering medications or HT at baseline compared with untreated participants; and, in some cases, would be modified by sex and race/ethnicity, as previously reported for mean VLDL size and small HDL-P (6).
Research Design and Methods
Study Population and Risk Factor Measurement
The design and objectives of the Multi-Ethnic Study of Atherosclerosis (MESA), sponsored by the National Heart, Lung, and Blood Institute, have been described elsewhere (25). Briefly, the multicenter prospective cohort study included 6,814 community-dwelling men and women, ages 45–84, of African American, Hispanic, white, and Chinese-American ethnicity. Exclusion criteria for MESA included self-reported cardiovascular disease (heart attack, angina, coronary revascularization, pacemaker or defibrillator implantation, valve replacement, heart failure, or cerebrovascular disease); pregnancy; cancer; cognitive impairment; or weight >136 kg. Participants provided informed consent at their field center, and the study was approved by the institutional review boards of the participating institutions and the University of Pittsburgh. Standardized questionnaires and procedures were used to determine age, sex, ethnicity, height, weight, waist circumference, blood pressure, medications, smoking, family history of diabetes, alcohol use, and physical activity at the baseline MESA examination in 2000–2002. Physical activity (moderate and vigorous) was defined as MET*min/week. Smoking was defined as never, ever (smoked ≥100 cigarettes in lifetime), or current (smoked cigarettes in the past 30 days). Alcohol use was defined as never, former, or current. For women, hormone replacement therapy was defined as ever or never. Hypertension was defined as systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg or self-reported hypertension and antihypertensive medication use. Insulin resistance was estimated using the HOMA-IR, which was calculated as insulin (µU/mL) × (glucose [mg/dL] × 0.055)/22.5 (26). For this report, participants also were categorized as “untreated,” that is, those reporting no lipid-lowering medications or HT use at baseline; “treated,” comprising those reporting use of lipid-lowering medications (primarily statins) at baseline; and “HT users,” who included women reporting use of HT but no lipid-lowering medications at baseline.
Incident Diabetes
Type 2 diabetes was defined as fasting glucose ≥126 mg/dL (7 mmol/L) or oral hypoglycemic medication or insulin use. Participants with diabetes at the baseline study visit were excluded. The date of incident diabetes was defined as the first of the four in-person follow-up MESA examinations at which a participant met the criteria for type 2 diabetes.
Laboratory Assessment
At the baseline visit, blood was drawn following a 12-h fast, and samples were stored at −70°C. Concentrations of lipoprotein particles were measured at LipoScience, Inc. (Raleigh, NC) using NMR spectroscopy (LipoProfile-3 algorithm) on plasma EDTA specimens. HDL, LDL, and VLDL subclasses were quantified using the amplitudes of their spectroscopically distinct lipid methyl group NMR signals (27). Total concentrations of HDL-P, LDL-P, and VLDL-P are the sums of the particle concentrations of the respective subclasses. Estimated ranges of particle diameter for the subclasses were as follows: large VLDL-P, 60 nm; medium VLDL-P, 42–60 nm; small VLDL-P, 29–42 nm; intermediate-density lipoprotein particles (IDL-P), 23–29 nm; large LDL-P, 20.5–23 nm; small LDL-P, 18–20.5 nm; large HDL-P, 9.4–14 nm; medium HDL-P, 8.2–9.4 nm; and small HDL-P, 7.3–8.2 nm. Mean particle sizes are weighted averages derived from the sum of the diameter of each subclass multiplied by its relative mass percentage. Mean VLDL size is not calculated for individuals with extremely low VLDL levels (VLDL-TG <30 mg/dL; n = 384 in this study.) A calculated LP-IR, which ranges from 0 (least) to 100 (most) insulin resistant, is also provided (9). As previously described in detail, the LP-IR is calculated by weighting large VLDL-P, small LDL-P, large HDL-P, and VLDL, LDL, and HDL size by the strength and independence of their association with HOMA-IR in a subset of MESA participants (10). Weights are capped at the lower and upper end for each lipoprotein particle concentration and size, with weight = 0 for individuals with VLDL size that was not calculated because of very low VLDL-TG (10). Interassay reproducibility (coefficient of variation [CV]), determined from 80 replicate analyses of 8 plasma pools over 20 days, was 6% for LP-IR; 8%, 3%, and 2% for total VLDL-P, LDL-P, and HDL-P; 0.7% for LDL and HDL size; 4% for VLDL size; 7%, 13%, and 22% for large, medium, and small VLDL-P; 43%, 12%, and 10% for IDL-P and large and small LDL-P; and 9%, 14%, and 6% for large, medium, and small HDL-P, respectively.
Lipids, insulin, and glucose in thawed baseline blood specimens were measured at a central laboratory (Collaborative Studies Clinical Laboratory at Fairview University Medical Center, Minneapolis, MN), usually within 2 weeks of sample collection, using Centers for Disease Control and Prevention/National Heart, Lung, and Blood Institute standards. Plasma EDTA HDL-C was measured by the cholesterol oxidase method (Roche Diagnostics) after precipitation of non-HDL-C with magnesium/dextran, with CV = 2.9%. Plasma EDTA triglycerides was measured by Triglyceride GB reagent (Roche Diagnostics, Indianapolis, IN) on the Roche COBAS FARA centrifugal analyzer with a coefficient of variation (CV) of 4.0%. For those with triglycerides ≤400 mg/dL, LDL cholesterol was calculated using the Friedewald equation (28). Serum glucose was measured by rate reflectance spectrophotometry using thin-film adaptation of the glucose oxidase method on a Vitros analyzer (Johnson & Johnson Clinical Diagnostics, Inc., Rochester, NY) with a CV of 1.1%. Serum insulin was measured by an immunoenzymatic sandwich assay using the Access Ultrasensitive Insulin Reagent on a Beckman Access (Beckman Instruments, Inc.), with a CV of 4.9%.
Statistical Analysis
SAS version 9.2 (SAS, Inc., Cary, NC) was used for analyses; a two-tailed P value <0.05 was considered statistically significant. Because of skewed distributions, insulin, HOMA-IR, TG, the ratio of TG to HDL-C, large VLDL-P, and high physical activity levels were log-transformed when used as continuous variables in regression analyses. Baseline participant characteristics were compared for those with and without incident diabetes using t tests, Wilcoxon tests for skewed variables previously noted, or χ2 tests for categorical variables. In this report, Cox proportional hazards regression was used to calculate hazard ratios (HRs) for incident diabetes for each predictor, both for quartiles and per SD, calculated from distributions in the entire sample. Follow-up time was calculated as days from baseline visit to the first follow-up visit with diagnosed diabetes or, for participants without incident diabetes, to the last visit attended. The exact method was used for tied event times, which were uncommon. The proportional hazards assumption was tested for each predictor using interaction terms with time and was met for all predictor variables. Glucose had a significant interaction with time, however, with stronger HRs for earlier than later diabetes cases. Therefore, the glucose–time interaction was included in any models that adjusted for glucose, although this had a minimal effect on results. Sensitivity analyses also evaluated results after excluding diabetes cases that occurred during the first 3 years of follow-up. Unless otherwise specified, all models were adjusted for a base set of covariates: age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, physical activity, and smoking. Subsequent models were sequentially adjusted for other IR-related measures. Potential differences in associations with incident diabetes by sex, race/ethnicity, or baseline use of lipid-lowering medications or HT were evaluated using multiplicative interaction terms and stratified models. For these sensitivity analyses, the number (cases/total) in each strata were as follows: for baseline treatment, 453/3,825 for untreated, 129/778 for lipid-lowering, and 74/711 for HT users; for glucose categories, 132/2,953, 188/1,546, and 336/815 for <90, 90–99, and 100–125 mg/dL, respectively; and by sex, 310/2,486 for men and 346/2,828 for women.
Results
After excluding participants with baseline diabetes (n = 859), with no follow-up (n = 293); with missing values for diabetes (n = 24); lipid-lowering medications, lipoproteins, alcohol use, or physical activity (n = 48); or family history of diabetes (n = 276), 5,314 MESA participants were available for analysis of incident diabetes. Over a mean of 7.7 years of follow-up, 656 diabetes cases were identified. Cases were more likely than noncases to be African American or Hispanic, to have a family history of diabetes, and to be using lipid-lowering medications (91% statins) at baseline (Table 1), but they were similar by sex and baseline HT use (data not shown). Cases also had higher mean levels of BMI, LP-IR, HOMA-IR; higher mean concentrations of glucose and insulin, TG, small HDL-P, total and small LDL-P, and large and medium VLDL-P; lower mean concentrations of HDL-C and total, large, and medium HDL-P; smaller mean HDL and LDL particle size; and larger mean VLDL particle size (Table 1). Except for total HDL-P and medium VLDL-P, all of these remained significantly associated with incident diabetes in Cox proportional hazards models adjusted for age, sex, and race/ethnicity (data not shown). Mean baseline glucose was substantially higher among cases than noncases (Table 1). Furthermore, incident diabetes increased across baseline glucose categories (data not shown) from 4.5% (132/2,953) of those with baseline glucose <90 mg/dL, 12.2% (188/1,546) for glucose 90–99 mg/dL, and 41.2% (336/815) of those with baseline glucose 100–125 mg/dL. Despite this strong association, however, 29% of incident diabetes cases had baseline glucose of 90–99 mg/dL, and 20% had baseline glucose <90 mg/dL.
Table 1.
Characteristics of MESA participants without baseline diabetes, by incident diabetes
| Characteristics | Total (N = 5,314) | Incident diabetes |
P value | |
|---|---|---|---|---|
| No (n = 4,658) | Yes (n = 656) | |||
| Age, years | 61.6 ± 10.2 | 61.7 ± 10.3 | 60.8 ± 9.5 | 0.08 |
| Race/ethnicity, % | ||||
| White | 42.5 | 44.2 | 30.5 | <0.0001 |
| Chinese American | 11.6 | 11.5 | 12.0 | |
| African American | 25.2 | 24.5 | 30.6 | |
| Hispanic American | 20.7 | 19.8 | 26.8 | |
| Lipid-lowering medication, % | 14.6 | 13.9 | 19.7 | <0.0001 |
| Family history of diabetes, % | 34.8 | 32.9 | 48.6 | <0.0001 |
| Physical activity, MET*min/week | 5,833 ± 5,832 | 5,836 ± 5,711 | 5,810 ± 6,637 | 0.24 |
| BMI, kg/m2 | 28.0 ± 5.3 | 27.6 ± 5.0 | 31.1 ± 5.9 | <0.0001 |
| LP-IR | 42.4 ± 23.4 | 40.7 ± 23.2 | 54.3 ± 21.4 | <0.0001 |
| Glucose, mg/dL | 89.4 ± 10.5 | 87.8 ± 9.0 | 100.8 ± 12.8 | <0.0001 |
| Insulin, μU/L | 6.4 ± 4.4 | 6.0 ± 4.0 | 9.5 ± 5.8 | <0.0001 |
| HOMA-IR | 1.4 ± 1.1 | 1.3 ± 1.0 | 2.4 ± 1.6 | <0.0001 |
| HDL-C, mg/dL | 51.7 ± 15.0 | 52.4 ± 15.2 | 46.7 ± 12.2 | <0.0001 |
| LDL-C, mg/dL | 118.0 ± 30.8 | 118.0 ± 30.6 | 117.6 ± 32.3 | 0.57 |
| TG, mg/dL | 127.0 ± 77.5 | 123.5 ± 75.2 | 152.0 ± 88.9 | <0.0001 |
| HDL-P, μmol/L | ||||
| Total | 34 ± 7 | 34 ± 7 | 33 ± 6 | 0.0001 |
| Large | 6.2 ± 3.5 | 6.3 ± 3.5 | 5.1 ± 2.9 | <0.0001 |
| Medium | 13.7 ± 6.9 | 13.9 ± 7.0 | 12.2 ± 6.2 | <0.0001 |
| Small | 14.5 ± 5.6 | 14.2 ± 5.5 | 16.1 ± 5.7 | <0.0001 |
| HDL size, nm | 9.27 ± 0.45 | 9.29 ± 0.46 | 9.13 ± 0.41 | <0.0001 |
| LDL-P, nmol/L | ||||
| Total | 1,246 ± 334 | 1,236 ± 331 | 1,322 ± 351 | <0.0001 |
| Large | 601 ± 257 | 609 ± 254 | 541 ± 270 | <0.0001 |
| Small | 518 ± 378 | 500 ± 375 | 653 ± 377 | <0.0001 |
| LDL size, nm | 20.77 ± 0.54 | 20.80 ± 0.54 | 20.58 ± 0.53 | <0.0001 |
| VLDL-P, nmol/L | ||||
| Large | 4.7 ± 6.2 | 4.4 ± 5.9 | 6.9 ± 7.3 | <0.0001 |
| Medium | 28.4 ± 21.6 | 28.1 ± 21.6 | 30.7 ± 21.4 | 0.0002 |
| Small | 34.0 ± 19.6 | 34.1 ± 19.5 | 33.4 ± 19.7 | 0.46 |
| VLDL size, nm* | 48.3 ± 7.9 | 47.7 ± 7.7 | 51.7 ± 8.8 | <0.0001 |
Results are mean ± SD, unless stated otherwise. LDL-C, LDL cholesterol.
VLDL size missing in n = 384 (21 cases).
LP-IR was strongly correlated (Spearman correlation ρ ≥ 0.5) with the six lipoprotein parameters from which it is calculated (i.e., concentrations of large VLDL-P, large HDL-P, and small LDL-P and mean size of VLDL, HDL, and LDL particles) and with concentrations of HOMA-IR, insulin, TG, HDL-C, and TG–to–HDL-C ratio, with very strong correlations with large VLDL-P and TG–to–HDL-C ratio (Supplementary Table 2). Correlations of BMI and glucose with insulin and HOMA-IR were stronger than with LP-IR, large VLDL-P, mean VLDL size, TG, HDL-C, or TG–to–HDL-C ratio.
Preliminary analyses evaluated potential differences in diabetes risk associations by sex, race/ethnicity, or baseline treatment status (untreated, lipid-lowering medications, or HT use). Among untreated participants, there were no significant interactions between race/ethnicity and any of the IR-related lipoproteins, lipids, insulin, or HOMA-IR in relation to incident diabetes. However, both total LDL-P and small HDL-P had significant interactions by sex (P = 0.03 for each). For total LDL-P, HRs (95% CI) per SD were 1.30 (1.15–1.47) for women and 1.06 (0.94–1.20) for men, and for small HDL-P, HRs (95% CI) were 1.11 (0.96–1.28) for women but 1.39 (1.19–1.61) for men. These results were consistent with HRs calculated across quartiles of total LDL-P and small HDL-P (Supplementary Table 2), which showed that when additionally adjusted for HOMA-IR, HRs for the fourth versus the first quartile (Q4 vs. Q1) remained significantly elevated for LDL-P among women and for small HDL-P among men. For baseline treatment status (lipid-lowering medication or HT use vs. none), no significant interactions were found for LP-IR, IR-related lipoproteins or lipids, or insulin in relation to diabetes risk based on interaction terms and consistent with stratified analyses (data not shown). For HOMA-IR, however, diabetes risk was lower for treated participants (HR [95% CI] in Q4 vs. Q1 4.96 [2.38–10.32) than for untreated participants (6.24 [4.10–9.50]) or HT users (5.85 [2.47–13.87]; P for interaction = 0.04). Therefore, primary analyses used the entire study sample (n = 5,314) adjusted for treatment group; sensitivity analyses for HOMA-IR were restricted to untreated participants.
Among the entire study sample adjusted for base covariates (age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, smoking, and physical activity) and treatment (none, lipid-lowering medication use, or HT use) (Table 2), risk of incident diabetes was increased twofold or more for high (Q4 vs. Q1) small LDL-P and low (Q1 vs. Q4) large HDL-P, HDL-C, and smaller LDL particle size; it was increased slightly less for smaller HDL particle size. HRs were attenuated with additional adjustment for glucose or insulin or HOMA-IR. None remained strongly associated with incident diabetes if adjusted for the ratio of TG to HDL-C, with similar results if adjusted for TG, HDL-C, or both (data not shown). Conversely, the association of low HDL-C with incident diabetes risk persisted after adjusting for large VLDL-P but not after adjusting for LP-IR (Table 2).
Table 2.
Risk (HR [95% CI]) of incident diabetes by quartiles of HDL and LDL indices, sequentially adjusted, among MESA participants without diabetes at baseline (N = 5,314)
| Predictors and covariates | Q1 | Q2 | Q3 | Q4 | P value for trend |
|---|---|---|---|---|---|
| HDL size, nm | ≤8.9 | 9.0–9.2 | 9.3–9.6 | ≥9.7 | |
| Base covariates* | 1.79 (1.37–2.35) | 1.52 (1.16–1.98) | 1.19 (0.91–1.57) | 1.0 (Reference) | <0.0001 |
| + Glucose | 1.36 (1.04–1.78) | 1.24 (0.95–1.62) | 1.06 (0.81–1.40) | 1.0 (Reference) | <0.0001 |
| + Insulin† | 1.34 (1.02–1.76) | 1.21 (0.93–1.58) | 1.05 (0.79–1.38) | 1.0 (Reference) | 0.01 |
| + HOMA-IR† | 1.17 (0.89–1.54) | 1.10 (0.84–1.44) | 0.98 (0.74–1.29) | 1.0 (Reference) | 0.12 |
| + TGtoHDL-C ratio† | 1.24 (0.94–1.64) | 1.12 (0.85–1.47) | 1.01 (0.76–1.33) | 1.0 (Reference) | 0.06 |
| Large HDL-P, µmol/L | ≤3.4 | 3.5–5.3 | 5.4–8.0 | ≥8.1 | |
| Base covariates* | 2.30 (1.75–3.03) | 1.80 (1.38–2.34) | 1.41 (1.08–1.85) | 1.0 (Reference) | <0.0001 |
| + Glucose | 1.67 (1.27–2.19) | 1.38 (1.06–1.81) | 1.39 (1.06–1.82) | 1.0 (Reference) | <0.0001 |
| + Insulin† | 1.63 (1.23–2.17) | 1.44 (1.10–1.88) | 1.24 (0.95–1.62) | 1.0 (Reference) | 0.0004 |
| + HOMA-IR† | 1.40 (1.06–1.85) | 1.30 (0.99–1.69) | 1.17 (0.89–1.53) | 1.0 (Reference) | 0.02 |
| + TGtoHDL-C ratio† | 1.36 (0.99–1.88) | 1.25 (0.94–1.67) | 1.16 (0.88–1.52) | 1.0 (Reference) | 0.06 |
| Medium HDL-P, µmol/L | ≤8.9 | 9.0–12.8 | 12.9–17.5 | ≥17.6 | |
| Base covariates* | 1.55 (1.22–1.98) | 1.39 (1.09–1.77) | 1.25 (0.98–1.61) | 1.0 (Reference) | <0.0001 |
| + Glucose | 1.40 (1.10–1.79) | 1.18 (0.92–1.51) | 1.24 (0.97–1.59) | 1.0 (Reference) | <0.0001 |
| + Insulin† | 1.32 (1.04–1.69) | 1.22 (0.96–1.56) | 1.16 (0.91–1.49) | 1.0 (Reference) | 0.03 |
| + HOMA-IR† | 1.24 (0.97–1.58) | 1.15 (0.90–1.47) | 1.12 (0.88–1.44) | 1.0 (Reference) | 0.10 |
| + TGtoHDL-C ratio† | 1.24 (0.97–1.59) | 1.22 (0.95–1.56) | 1.23 (0.96–1.57) | 1.0 (Reference) | 0.15 |
| HDL-C, mg/dL | ≤40 | 41–49 | 50–59 | ≥60 | |
| Base covariates* | 2.37 (1.80–3.12) | 1.87 (1.44–2.44) | 1.51 (1.15–1.98) | 1.0 (Reference) | <0.0001 |
| + Glucose | 1.64 (1.25–2.16) | 1.53 (1.18–1.99) | 1.36 (1.03–1.78) | 1.0 (Reference) | <0.0001 |
| + Insulin† | 1.68 (1.27–2.23) | 1.47 (1.13–1.92) | 1.31 (0.99–1.72) | 1.0 (Reference) | 0.0003 |
| + HOMA-IR† | 1.42 (1.07–1.89) | 1.32 (1.01–1.72) | 1.22 (0.93–1.60) | 1.0 (Reference) | 0.02 |
| + Large VLDL-P† | 1.78 (1.34–2.39) | 1.59 (1.22–2.08) | 1.41 (1.08–1.98) | 1.0 (Reference) | <0.0001 |
| + LP-IR | 1.15 (0.84–1.60) | 1.10 (0.82–1.47) | 1.15 (0.87–1.52) | 1.0 (Reference) | 0.54 |
| LDL size, nm | ≤20.3 | 20.4–20.8 | 20.9–21.1 | ≥21.2 | |
| Base covariates* | 2.41 (1.86–3.12) | 2.01 (1.56–2.58) | 1.43 (1.09–1.88) | 1.0 (Reference) | <0.0001 |
| + Glucose | 1.68 (1.30–2.18) | 1.65 (1.28–2.12) | 1.26 (0.96–1.66) | 1.0 (Reference) | <0.0001 |
| + Insulin† | 1.84 (1.41–2.48) | 1.68 (1.31–2.17) | 1.29 (0.98–1.70) | 1.0 (Reference) | <0.0001 |
| + HOMA-IR† | 1.62 (1.24–2.11) | 1.55 (1.20–2.00) | 1.24 (0.94–1.63) | 1.0 (Reference) | 0.0001 |
| + TGtoHDL-C ratio† | 1.30 (1.01–1.92) | 1.51 (1.15–1.98) | 1.24 (0.94–1.64) | 1.0 (Reference) | 0.03 |
| Small LDL-P, nmol/L | ≤108 | 109–501 | 502–780 | ≥781 | |
| Base covariates* | 1.0 (Reference) | 1.55 (1.17–2.04) | 2.11 (1.62–2.76) | 2.42 (1.86–3.16) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.46 (1.11–1.92) | 1.81 (1.38–2.36) | 1.80 (1.38–2.34) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.35 (1.02–1.78) | 1.71 (1.30–2.24) | 1.86 (1.42–2.44) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 1.27 (0.96–1.68) | 1.55 (1.18–2.03) | 1.65 (1.26–2.15) | 0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.34 (1.02–1.77) | 1.57 (1.18–2.08) | 1.48 (1.09–2.02) | 0.02 |
Statistically significant HRs are bolded.
Models adjusted for base covariates of age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, smoking, physical activity, and treatment group (none, lipid-lowering medications, or HT use). Subsequent models additionally adjust for covariate(s) listed.
Log transformed.
For LP-IR, VLDL-related parameters, insulin, and HOMA-IR adjusted for base covariates and treatment group, HRs (Q4 vs. Q1) ranged from ∼2.5 for TG and the TG–to–HDL-C ratio to 3.28 for LP-IR, 3.84 for insulin, and 5.93 for HOMA-IR. For LP-IR, large VLDL-P, and mean VLDL size, HRs (Q4 vs. Q1) remained significantly elevated (1.5- to 2-fold) when adjusted for glucose, insulin, HOMA-IR, or the TG–to–HDL-C ratio, as well as for glucose and HOMA-IR or glucose and the TG–to–HDL-C ratio. In contrast, TG and the TG–to–HDL-C ratio remained associated with incident diabetes adjusted for glucose, insulin, or HOMA-IR but not adjusted for LP-IR. Results adjusted for TG, HDL-C, or both (data not shown) were similar to results adjusted for the TG–to–HDL-C ratio. For insulin and HOMA-IR, HRs (Q4 vs. Q1) remained significantly associated with diabetes if adjusted for LP-IR, large VLDL-P, or the TG–to–HDL-C ratio, but not when simultaneously adjusted for LP-IR and glucose, which both added significantly to diabetes risk in all models.
To further evaluate associations with diabetes risk independent of glucose, models were rerun stratified by baseline glucose categories (<90, 90–99, and 100–125 mg/dL) and adjusted for base covariates and treatment status. Within all three categories of baseline glucose, HRs for diabetes were significantly increased (Q4 vs. Q1) for LP-IR, small LDL-P, the TG–to–HDL-C ratio, and TG. In contrast, HRs for HOMA-IR (Q4 vs. Q1) were significantly related to diabetes risk only for participants with baseline glucose <90 mg/dL, and insulin was not significantly associated with diabetes risk for participants with baseline glucose of 100–125 mg/dL. HRs (Q4 vs. Q1) also did not reach statistical significance for VLDL-P size among those with glucose 100–125 mg/dL and for large VLDL-P among those with glucose <90 mg/dL.
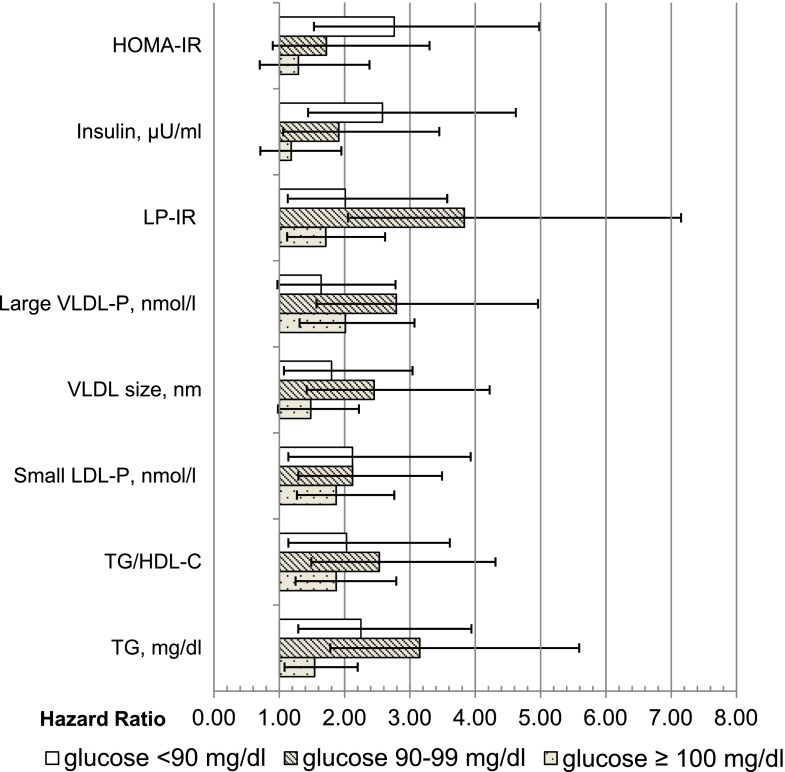
Finally, in sensitivity analyses, we excluded incident diabetes cases with ≤3 years of follow-up (n = 226), and, as shown in Tables 2 and 3, results were essentially unchanged for models adjusted for base covariates or base covariates and glucose. Given evidence of weaker associations among treated participants, HOMA-IR models restricted to untreated participants were also rerun. Overall, estimates (Fig. 1 and Table 3) were similar to those for the entire group, except that, as shown in Fig. 1, among those with glucose <90 mg/dL, the HR (95% CI) for HOMA-IR (Q4 vs. Q1) increased from 2.76 (1.53–4.98) to 3.53 (1.86–6.66), which did not alter overall conclusions.
Table 3.
Risk (HR [95% CI]) of incident diabetes by quartiles of VLDL- and insulin resistance–related indices, sequentially adjusted, among MESA participants without diabetes at baseline (N = 5,314)
| Predictors | Q1 | Q2 | Q3 | Q4 | P value for trend |
|---|---|---|---|---|---|
| LP-IR | 0–24 | 25–42 | 43–61 | 62–100 | |
| Base covariates* | 1.0 (Reference) | 1.47 (1.08–1.99) | 2.21 (1.66–2.94) | 3.28 (2.46–4.38) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.32 (0.97–1.79) | 1.77 (1.33–2.35) | 2.02 (1.52–2.70) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.25 (0.92–1.70) | 1.70 (1.27–2.28) | 2.15 (1.59–2.92) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 1.13 (0.83–1.54) | 1.47 (1.10–1.97) | 1.70 (1.25–2.30) | <0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.30 (0.95–1.78) | 1.77 (1.28–2.45) | 2.29 (1.55–3.38) | <0.0001 |
| + HOMA-IR† + glucose | 1.0 (Reference) | 1.22 (0.89–1.66) | 1.69 (1.26–2.25) | 1.93 (1.43–2.60) | <0.0001 |
| + TGtoHDL-C ratio† + glucose | 1.0 (Reference) | 1.21 (0.88–1.66) | 1.53 (1.11–2.12) | 1.59 (1.08–2.35) | 0.01 |
| VLDL size, nm‡ | ≤42.4 | 42.5–46.5 | 46.6–52.5 | ≥52.6 | |
| Base covariates* | 1.0 (Reference) | 1.35 (1.02–1.79) | 1.68 (1.28–2.20) | 2.63 (2.03–3.42) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.24 (0.94–1.65) | 1.25 (0.95–1.64) | 1.81 (1.39–2.37) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.22 (0.92–1.61) | 1.36 (1.04–1.79) | 1.96 (1.50–2.56) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 1.15 (0.86–1.52) | 1.21 (0.92–1.59) | 1.68 (1.28–2.19) | <0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.23 (0.92–1.64) | 1.41 (1.06–1.86) | 1.92 (1.43–2.59) | <0.0001 |
| + HOMA-IR† + glucose | 1.0 (Reference) | 1.21 (0.91–1.61) | 1.18 (0.90–1.56) | 1.69 (1.29–2.22) | <0.0001 |
| + TGtoHDL-C ratio† + glucose | 1.0 (Reference) | 1.18 (0.89–1.57) | 1.14 (0.86–1.51) | 1.54 (1.14–2.07) | 0.008 |
| Large VLDL-P, nmol/L | ≤0.70 | 0.8–2.30 | 2.4–6.2 | 6.3–55.9 | |
| Base covariates* | 1.0 (Reference) | 1.28 (0.96–1.71) | 2.02 (1.54–2.65) | 2.73 (2.08–3.57) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.30 (0.97–1.73) | 1.71 (1.30–2.24) | 1.97 (1.50–2.60) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.08 (0.80–1.44) | 1.55 (1.17–2.05) | 1.87 (1.41–2.48) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 0.99 (0.74–1.32) | 1.35 (1.03–1.79) | 1.55 (1.17–2.06) | <0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.17 (0.87–1.56) | 1.63 (1.22–2.18) | 1.75 (1.24–2.47) | 0.0003 |
| + HOMA-IR† + glucose | 1.0 (Reference) | 1.25 (0.93–1.68) | 1.49 (1.12–1.97) | 1.82 (1.36–2.43) | <0.0001 |
| + TGtoHDL-C ratio† + glucose | 1.0 (Reference) | 1.24 (0.93–1.66) | 1.53 (1.14–2.04) | 1.55 (1.09–2.20) | 0.005 |
| TG, mg/dL | ≤76 | 77–108 | 109–156 | ≥157 | |
| Base covariates* | 1.0 (Reference) | 1.63 (1.25–2.13) | 1.82 (1.40–2.38) | 2.56 (1.97–3.32) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.37 (1.05–1.79) | 1.59 (1.22–2.08) | 1.78 (1.37–2.31) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.47 (1.12–1.92) | 1.54 (1.18–2.01) | 1.93 (1.47–2.52) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 1.40 (1.07–1.83) | 1.43 (1.09–1.86) | 1.69 (1.29–2.21) | 0.0003 |
| + Large VLDL-P† | 1.0 (Reference) | 1.41 (1.07–1.86) | 1.34 (1.00–1.79) | 1.33 (0.92–1.93) | 0.21 |
| + LP-IR | 1.0 (Reference) | 1.21 (0.92–1.60) | 1.08 (0.80–1.45) | 1.11 (0.79–1.55) | 0.94 |
| TGtoHDL-C ratio | <1.384 | 1.384–2.169 | 2.170–3.576 | >3.577 | |
| Base covariates* | 1.0 (Reference) | 1.49 (1.13–1.98) | 2.29 (1.75–3.00) | 2.58 (1.96–3.39) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.30 (0.98–1.72) | 1.76 (1.35–2.31) | 1.82 (1.38–2.39) | <0.0001 |
| + Insulin† | 1.0 (Reference) | 1.31 (0.98–1.74) | 1.87 (1.43–2.46) | 1.87 (1.41–2.48) | <0.0001 |
| + HOMA-IR† | 1.0 (Reference) | 1.22 (0.92–1.63) | 1.70 (1.30–2.23) | 1.61 (1.22–2.14) | 0.0002 |
| + Large VLDL-P† | 1.0 (Reference) | 1.31 (0.98–1.75) | 1.76 (1.32–2.36) | 1.48 (1.04–2.10) | 0.01 |
| + LP-IR | 1.0 (Reference) | 1.04 (0.77–1.41) | 1.20 (0.87–1.66) | 1.00 (0.68–1.46) | 0.94 |
| Insulin, μU/L | ≤3.4 | 3.5–5.1 | 5.2–8.0 | ≥8.1 | |
| Base covariates* | 1.0 (Reference) | 1.35 (0.97–1.89) | 2.23 (1.64–3.04) | 3.84 (2.83–5.22) | <0.0001 |
| + Glucose | 1.0 (Reference) | 1.02 (0.73–1.43) | 1.30 (0.95–1.78) | 1.63 (1.19–2.23) | <0.0001 |
| + LP-IR | 1.0 (Reference) | 1.16 (0.83–1.63) | 1.70 (1.23–2.34) | 2.64 (1.91–3.64) | <0.0001 |
| + Large VLDL-P† | 1.0 (Reference) | 1.23 (0.88–1.72) | 1.87 (1.37–2.57) | 3.02 (2.20–4.14) | <0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.22 (0.87–1.71) | 1.89 (1.38–2.58) | 3.03 (2.21–4.15) | <0.0001 |
| +TGtoHDL-C ratio† + glucose | 1.0 (Reference) | 0.93 (0.66–1.31) | 1.14 (0.83–1.57) | 1.36 (0.98–1.88) | 0.005 |
| + LP-IR + glucose | 1.0 (Reference) | 0.90 (0.64–1.26) | 1.05 (0.76–1.45) | 1.20 (0.86–1.68) | 0.06 |
| HOMA-IR | < 0.712 | 0.712–1.114 | 1.115–1.815 | >1.815 | |
| Base covariates* | 1.0 (Reference) | 1.43 (0.99–2.07) | 2.62 (1.86–3.68) | 5.93 (4.26–8.26) | <0.0001 |
| + Glucose | 1.0 (Reference) | 0.99 (0.68–1.44) | 1.27 (0.90–1.80) | 1.73 (1.22–2.46) | <0.0001 |
| + LP-IR | 1.0 (Reference) | 1.27 (0.88–1.85) | 2.10 (1.48–2.98) | 4.37 (3.09–6.19) | <0.0001 |
| + Large VLDL-P† | 1.0 (Reference) | 1.33 (0.92–1.93) | 2.28 (1.61–3.22) | 4.88 (3.47–6.87) | <0.0001 |
| + TGtoHDL-C ratio† | 1.0 (Reference) | 1.33 (0.92–1.93) | 2.28 (1.61–3.22) | 4.88 (3.47–6.86) | <0.0001 |
| +TGtoHDL-C ratio† + glucose | 1.0 (Reference) | 0.94 (0.65–1.37) | 1.14 (0.80–1.62) | 1.46 (1.02–2.10) | 0.001 |
| + LP-IR + glucose | 1.0 (Reference) | 0.90 (0.62–1.31) | 1.05 (0.74–1.50) | 1.29 (0.89–1.87) | 0.02 |
Statistically significant HRs are bolded.
Models adjusted for base covariates of age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, smoking, physical activity, and treatment group (none, lipid-lowering medication, or HT use). Subsequent models are additionally adjust for the covariate(s) listed.
Log transformed.
VLDL size missing in n = 384 (21 cases).
Figure 1.
Adjusted HRs (95% CIs) of incident diabetes among the entire study sample, comparing quartile 4 vs. quartile 1, adjusted for age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, smoking, physical activity, and treatment group (none, lipid-lowering medication use, or HT use).
Conclusions
In a large, multiethnic cohort of men and women, higher levels of LP-IR, large VLDL-P, and small LDL-P and lower levels of large HDL-P, larger mean VLDL size, smaller mean HDL and LDL size, HDL-C, TG, TG–to–HDL-C ratio, insulin, and HOMA-IR were associated with incident diabetes over 7.7 years’ mean follow-up, adjusted for potential confounders including age, sex, race/ethnicity, BMI, family history of diabetes, alcohol use, smoking, and physical activity. Higher LP-IR, higher large VLDL-P concentration, and larger mean VLDL particle size remained associated with diabetes when additionally adjusted for HOMA-IR, the TG–to–HDL-C ratio, or for concentrations of glucose, insulin, HDL-C, TG and when simultaneously adjusted for glucose and HOMA-IR or glucose and the TG–to–HDL-C ratio. In contrast, associations with diabetes were not significant for HDL-C, TG, or TG–to–HDL-C ratio if adjusted for LP-IR. For HOMA-IR or insulin, diabetes associations persisted when adjusted for LP-IR but not both LP-IR and glucose. Furthermore, LP-IR, large VLDL-P, small LDL-P, TG, and TG–to–HDL-C ratio, but not insulin or HOMA-IR, were significantly associated with diabetes risk in each category of baseline glucose (<90, 90–99, and 100–125 mg/dL). Finally, associations of IR-related lipoproteins with diabetes were similar by race/ethnicity, by use of lipid-lowering medication or HT at baseline, and by sex (except total LDL-P and small HDL-P). These results suggest that IR-related lipoprotein and lipid indices, particularly LP-IR, large VLDL-P, mean VLDL size, TG, and TG–to–HDL-C ratio may robustly add to glucose, insulin, and HOMA-IR in identifying risk of incident diabetes.
Our results are generally consistent with several prior studies that did not include LP-IR (3,4,6–8). Compared with the large Women’s Health Study (WHS), including >26,000 women with 1,687 cases of incident diabetes over (a median of) 13.3 years, our study showed slightly stronger associations with diabetes for large VLDL-P than for HDL size. However, direct comparison is difficult because HRs in the WHS were calculated for quintiles, rather than quartiles, of lipoprotein parameters, and some covariates adjusted for differed from those in our study (e.g., HbA1c, which was not available in MESA at baseline). Relative to the WHS, our study also found weaker associations with total LDL-P and slightly stronger associations with small HDL-P among the entire sample (data not shown), consistent with our finding of significant interactions by sex; that is, diabetes was more strongly associated with total LDL-P in women and with small HDL-P in men. In the Insulin Resistance Atherosclerosis Study (IRAS) (n cases/total = 130/830 over a mean of 5.2 years), higher total LDL-P, small HDL-P, larger mean VLDL size, and smaller HDL size were associated with incident diabetes adjusted for age, sex, and race and small HDL-P and large VLDL size persisted when adjusted for both HDL-C and TG (6). In contrast to our results, IRAS found stronger associations of small HDL-P for women than men and for African American and Hispanic than white participants, and stronger associations of VLDL size for women than men, although only the last interaction reached statistical significance. Their stratified analyses showed no relationship of larger VLDL size with incident diabetes among men, suggesting that unadjusted confounders (e.g., fibrates or HT) may explain differences between their earlier, smaller study and our study. Additional studies should evaluate the potential effect modification of sex or race/ethnicity, but our results suggest that LP-IR, large VLDL-P, and mean VLDL size are robustly associated with risk of diabetes across sex and racial/ethnic groups.
This is the first study to report that IR-related lipoproteins are related to diabetes risk among those using lipid-lowering medications (primarily statins) and HT at baseline, as well as among those not using lipid-altering medications. This conclusion was based on the absence of statistically significant interactions with treatment group, as well as by review of stratified results (data not shown). Our results are consistent with prior reports of clinical trials of statins showing that high TG (≥150 mg/dL) and glucose (≥100 mg/dL) are independently associated with diabetes risk (20,29). Associations among treated participants and HT users likely reflect their metabolic risk before starting medications. In our study lipid-lowering medications were primarily statins, and results for other lipid-lowering medications may differ according to their effects on lipoproteins. For example, statins tend to increase small HDL-P more than large HDL-P, at least among those with metabolic syndrome (30). Cholesteryl ester transfer protein (CETP) inhibitors increase HDL and LDL particle size (31,32) and may improve glucose homeostasis (18). In contrast, niacin increases both LDL and HDL particle size (33) yet is associated with poorer glucose homeostasis (19). Other studies should evaluate these questions, particularly with clinical trial data.
Our results should be carefully interpreted in light of the strengths and limitations of our observational study design. Strengths include the large, multiethnic cohort with careful, standardized data collection. Diabetes is defined using measured glucose and medication use that was carefully assessed at baseline and each of the four follow-up visits. The timing of diabetes diagnosis is defined by clinic visit, which occurred approximately every 2 years; this is less precise than some events such as clinical myocardial infarction or death. The high prevalence of unrecognized diabetes in the community also makes it difficult to define the exact onset of diabetes, however, and defining it at regularly occurring clinic visits should minimize surveillance bias. The data were carefully analyzed, but the possibility of unmeasured confounding with this observational data remains. Use of lipid-lowering medications and HT at baseline was not randomized, there is no information on biomarker levels before treatment, and sample sizes of the two treatment groups were much smaller than for those not using lipid-altering medications at baseline. The sample size for those using lipid-lowering medications at baseline was similar to the entire sample evaluated in the IRAS report, however, and a lack of significant multiplicative interactions with treatment group were consistent with results from stratified analyses (data not shown). Finally, these analyses involve multiple comparisons that increase the possibility of type 1 error (false-positive results). However, interpretation of the current study’s results relied primarily on the magnitude of HRs across risk factor quartiles rather than on statistical significance; that is, statistically significant HRs were typically >1.40, and nonsignificant HRs were close to 1.0.
In conclusion, in a large, multiethnic cohort of men and women, lipoprotein particle concentrations and size, HDL-C, TG, and the TG–to–HDL-C ratio are robustly related to incident diabetes, when adjusted for multiple confounders, among those using as well as not using lipid-lowering medications or HT at baseline. A lipoprotein-based insulin resistance score had associations within incident diabetes similar to VLDL-related measures, including large VLDL-P, larger mean VLDL size, TG, and TG–to–HDL-C ratio, but independent of both the TG–to–HDL-C ratio and glucose or HOMA-IR and glucose. These results suggest that IR-related lipoproteins and lipids may provide information about diabetes risk beyond established risk factors, glucose, insulin, or HOMA-IR.
Supplementary Material
Article Information
Acknowledgments. The manuscript has been reviewed and approved for submission by the MESA Publications & Policy Committee and Steering Committee.
Funding. This research was supported by contracts N01-HC-95159 through N01-HC-95169 from the National Heart, Lung, and Blood Institute.
Duality of Interest. This study was funded by an investigator-initiated unrestricted research grant from LipoScience, Inc., to the University of Pittsburgh, which supported the efforts of R.H.M. in this study. R.H.M. has received speaker honoraria from the National Lipid Association for educational (nonpromotional) activities. S.M. has received research support from AstraZeneca, Atherotec Diagnostics, and the National Heart, Lung, and Blood Institute; served as a consultant to Pfizer, Genzyme, and Quest Diagnostics; received speaker honoraria from AstraZeneca, Abbott, and the National Lipid Association for educational (nonpromotional) activities; and received travel expense reimbursement from Pfizer. D.C.G. is a research consultant for a clinical trial of a glucose-lowering medication marketed by Merck and a Data and Safety Monitoring Board member for a clinical trial of a glucose-lowering medication marketed by Takeda. No other potential conflicts of interest relevant to this article were reported.
LipoScience exercised no control over and had no involvement in this study other than performing the blinded laboratory measurements for MESA.
Author Contributions. R.H.M. conceived the design of the study (analysis plan/article proposal), obtained funding, analyzed and interpreted the data, and wrote the first and subsequent drafts of the manuscript in collaboration with coauthors. S.M., A.G.B., M.R.C., C.T.S., and D.C.G. provided input on the study design/analysis plan/article proposal, critically reviewed all manuscript drafts, and approved the final manuscript. C.L.W. provided input on statistical analysis and interpretation of the results, critically reviewed all manuscript drafts, and approved the final manuscript. R.H.M. and C.L.W. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Prior Presentation. An abstract based on this work was presented as an oral presentation at the American Heart Association Epidemiology and Prevention 2014 Scientific Sessions, San Francisco, CA, 18–21 March 2014.
Footnotes
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc14-0645/-/DC1.
References
- 1.Diabetes Prevention Program Research Group, Knowler WC, Fowler SE, Hamman RF, et al. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet 2009;374:1677–1686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sparks JD, Sparks CE, Adeli K. Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol 2012;32:2104–2112 [DOI] [PubMed] [Google Scholar]
- 3.Garvey WT, Kwon S, Zheng D, et al. Effects of insulin resistance and type 2 diabetes on lipoprotein subclass particle size and concentration determined by nuclear magnetic resonance. Diabetes 2003;52:453–462 [DOI] [PubMed] [Google Scholar]
- 4.Goff DC Jr, D’Agostino RB Jr, Haffner SM, Otvos JD. Insulin resistance and adiposity influence lipoprotein size and subclass concentrations. Results from the Insulin Resistance Atherosclerosis Study. Metabolism 2005;54:264–270 [DOI] [PubMed] [Google Scholar]
- 5.Wang J, Stančáková A, Soininen P, et al. Lipoprotein subclass profiles in individuals with varying degrees of glucose tolerance: a population-based study of 9399 Finnish men. J Intern Med 2012;272:562–572 [DOI] [PubMed] [Google Scholar]
- 6.Festa A, Williams K, Hanley AJ, et al. Nuclear magnetic resonance lipoprotein abnormalities in prediabetic subjects in the Insulin Resistance Atherosclerosis Study. Circulation 2005;111:3465–3472 [DOI] [PubMed] [Google Scholar]
- 7.Mora S, Otvos JD, Rosenson RS, Pradhan A, Buring JE, Ridker PM. Lipoprotein particle size and concentration by nuclear magnetic resonance and incident type 2 diabetes in women. Diabetes 2010;59:1153–1160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hodge AM, Jenkins AJ, English DR, O’Dea K, Giles GG. NMR-determined lipoprotein subclass profile predicts type 2 diabetes. Diabetes Res Clin Pract 2009;83:132–139 [DOI] [PubMed] [Google Scholar]
- 9.Otvos JD, inventor; LipoScience, Inc, assignee. Lipoprotein insulin resistance indexes and related methods, systems and computer programs for generating same. U.S. patent 8,386,187. 26 February 2013
- 10.Shalaurova I, Connelly MA, Garvey WT, Otvos JD. Lipoprotein insulin resistance index: a lipoprotein particle-derived measure of insulin resistance. Metab Syndr Relat Disord 2014;12:422–429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mikus CR, Patel MJ, Slentz CA, Bateman LA, Willis LH, Kraus WE. Comparison of lipoprotein-derived insulin resistance score and standard measures of insulin sensitivity. Diabetes 2013;62(Suppl. 1):A167 [Google Scholar]
- 12.Fryirs MA, Barter PJ, Appavoo M, et al. Effects of high-density lipoproteins on pancreatic beta-cell insulin secretion. Arterioscler Thromb Vasc Biol 2010;30:1642–1648 [DOI] [PubMed] [Google Scholar]
- 13.Drew BG, Rye KA, Duffy SJ, Barter P, Kingwell BA. The emerging role of HDL in glucose metabolism. Nat Rev Endocrinol 2012;8:237–245 [DOI] [PubMed] [Google Scholar]
- 14.von Eckardstein A, Sibler RA. Possible contributions of lipoproteins and cholesterol to the pathogenesis of diabetes mellitus type 2. Curr Opin Lipidol 2011;22:26–32 [DOI] [PubMed] [Google Scholar]
- 15.Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet 2010;375:735–742 [DOI] [PubMed] [Google Scholar]
- 16.Preiss D, Seshasai SR, Welsh P, et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: a meta-analysis. JAMA 2011;305:2556–2564 [DOI] [PubMed] [Google Scholar]
- 17.Culver AL, Ockene IS, Balasubramanian R, et al. Statin use and risk of diabetes mellitus in postmenopausal women in the Women’s Health Initiative. Arch Intern Med 2012;172:144–152 [DOI] [PubMed] [Google Scholar]
- 18.Barter PJ, Rye KA, Tardif JC, et al. Effect of torcetrapib on glucose, insulin, and hemoglobin A1c in subjects in the Investigation of Lipid Level Management to Understand its Impact in Atherosclerotic Events (ILLUMINATE) trial. Circulation 2011;124:555–562 [DOI] [PubMed] [Google Scholar]
- 19.Goldberg RB, Jacobson TA. Effects of niacin on glucose control in patients with dyslipidemia. Mayo Clin Proc 2008;83:470–478 [DOI] [PubMed] [Google Scholar]
- 20.Waters DD, Ho JE, DeMicco DA, et al. Predictors of new-onset diabetes in patients treated with atorvastatin: results from 3 large randomized clinical trials. J Am Coll Cardiol 2011;57:1535–1545 [DOI] [PubMed] [Google Scholar]
- 21.Mackey RH, Kuller LH, Sutton-Tyrrell K, Evans RW, Holubkov R, Matthews KA. Lipoprotein subclasses and coronary artery calcium in postmenopausal women from the healthy women study. Am J Cardiol 2002;90(8A):71i–76i [DOI] [PubMed] [Google Scholar]
- 22.Hsia J, Otvos JD, Rossouw JE, et al.; Women’s Health Initiative Research Group . Lipoprotein particle concentrations may explain the absence of coronary protection in the Women's Health Initiative hormone trials. Arterioscler Thromb Vasc Biol 2008;28:1666–1671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bertoni AG, Burke GL, Owusu JA, et al. Inflammation and the incidence of type 2 diabetes: the Multi-Ethnic Study of Atherosclerosis (MESA). Diabetes Care 2010;33:804–810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lutsey PL, Pereira MA, Bertoni AG, Kandula NR, Jacobs DR Jr. Interactions between race/ethnicity and anthropometry in risk of incident diabetes: the Multi-Ethnic Study of Atherosclerosis. Am J Epidemiol 2010;172:197–204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bild DE, Bluemke DA, Burke GL, et al. Multi-Ethnic Study of Atherosclerosis: objectives and design. Am J Epidemiol 2002;156:871–881 [DOI] [PubMed] [Google Scholar]
- 26.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–419 [DOI] [PubMed] [Google Scholar]
- 27.Jeyarajah EJ, Cromwell WC, Otvos JD. Lipoprotein particle analysis by nuclear magnetic resonance spectroscopy. Clin Lab Med 2006;26:847–870 [DOI] [PubMed] [Google Scholar]
- 28.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499–502 [PubMed] [Google Scholar]
- 29.Waters DD, Ho JE, Boekholdt SM, et al. Cardiovascular event reduction versus new-onset diabetes during atorvastatin therapy: effect of baseline risk factors for diabetes. J Am Coll Cardiol 2013;61:148–152 [DOI] [PubMed] [Google Scholar]
- 30.Rosenson RS, Otvos JD, Hsia J. Effects of rosuvastatin and atorvastatin on LDL and HDL particle concentrations in patients with metabolic syndrome: a randomized, double-blind, controlled study. Diabetes Care 2009;32:1087–1091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rashedi N, Brennan D, Kastelein JJ, Nissen SE, Nicholls S. Impact of cholesteryl ester transfer protein inhibition on nuclear magnetic resonance derived lipoprotein particle parameters. Atheroscler Suppl 2011;12:48 [Google Scholar]
- 32.Ballantyne CM, Miller M, Niesor EJ, Burgess T, Kallend D, Stein EA. Effect of dalcetrapib plus pravastatin on lipoprotein metabolism and high-density lipoprotein composition and function in dyslipidemic patients: results of a phase IIb dose-ranging study. Am Heart J 2012;163:515–21, 521e1–3 [DOI] [PubMed] [Google Scholar]
- 33.Jafri H, Alsheikh-Ali AA, Mooney P, Kimmelstiel CD, Karas RH, Kuvin JT. Extended-release niacin reduces LDL particle number without changing total LDL cholesterol in patients with stable CAD. J Clin Lipidol 2009;3:45–50 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.