Abstract
The progression of autoimmune diseases is dictated by deviations in the fine balance between proinflammatory versus regulatory responses, and pathogen recognition receptors (PRRs) play a key role in maintaining this balance. Previously, we have reported that ligation of Toll-like receptor 2 (TLR2) and Dectin 1 on antigen-presenting cells by zymosan results in a regulatory immune response that prevents type 1 diabetes (T1D). Here, we show that TLR2 and Dectin 1 engagement by zymosan promotes regulatory T-cell (Treg) responses against the pancreatic β-cell–specific antigen (Ag). Unlike the TLR4 ligand, bacterial lipopolysaccharide, which induced proinflammatory cytokines and pathogenic T cells, zymosan induced a mixture of pro- and anti-inflammatory factors and Tregs, both in vitro and in vivo. Ag-specific T cells that are activated using zymosan-exposed dendritic cells (DCs) expressed Foxp3 and produced large amounts of IL-10, TGF-β1, and IL-17. NOD mice that received β-cell-Ag–loaded, zymosan-exposed DCs showed delayed hyperglycemia. Injection of NOD mice at the prediabetic age and early hyperglycemic stage with β-cell-Ag, along with zymosan, results in a superior protection of the NOD mice from diabetes as compared with mice that received zymosan alone. This therapeutic effect was associated with increased frequencies of IL-10–, IL-17–, IL-4–, and Foxp3-positive T cells, especially in the pancreatic lymph nodes. These results show that zymosan can be used as an immune regulatory adjuvant for modulating the T-cell response to pancreatic β-cell-Ag and reversing early-stage hyperglycemia in T1D.
Introduction
Innate immunity, initiated primarily by environmental factors such as microbes, plays a key role in initiating or preventing the T-cell response to pancreatic β-cell-Ag in type 1 diabetes (T1D). Although it has been suggested that the proinflammatory response mediated by pathogen recognition receptors (PRRs) facilitates β-cell-Ag presentation by activated antigen-presenting cells (APCs) (1), environmental factors such as bacterial and viral infections are also known to have a protective effect in T1D (2–5). Innate immune response is mediated by an array of PRRs such as Toll-like receptors (TLRs) and C-type lectin receptors (CLRs) that primarily recognize microbial products. In recent years, studies, including ours, have shown that innate immune responses induced through TLR2 and Dectin 1 using zymosan, a fungal cell wall component, are regulatory in nature and involve, in addition to proinflammatory factors, the expression of IL-2, IL-10, TGF-β1, and retinaldehyde dehydrogenase 1A2 (Raldh1A2) by one or other type of APCs (6–13). Importantly, the innate immune response induced by zymosan has the ability to prevent/delay disease in T1D and experimental autoimmune encephalomyelitis (EAE) models, even upon disease onset (6–11).
In this report, we show that zymosan-induced innate immune response facilitates regulatory T-cell (Treg) induction and/or expansion and Th1 to Th17 skewing of the T-cell response to pancreatic β-cell-Ag. Importantly, treatment with zymosan along with β-cell-Ag resulted in a significant delay in hyperglycemia in NOD mice even when the treatment was initiated at an early hyperglycemic stage as compared with treatment with zymosan alone. These observations show that zymosan has therapeutic values as a tolerogenic adjuvant and can be used for promoting β-cell-Ag–specific tolerance and to reverse early-stage hyperglycemia in T1D.
Research Design and Methods
Mice
Wild-type (WT) NOD/LtJ, NOD-BDC2.5-TCR transgenic (TCR-Tg), NOD-Scid, C57BL/6, and OT-II-TCR-Tg mice were purchased from The Jackson Laboratory (Bar Harbor, ME). OT-II-TCR-Tg-Foxp3-GFP-knockin, NOD-Foxp3-GFP, and NOD-BDC2.5-Foxp3-GFP mice were generated at our animal facilities. To detect hyperglycemia in NOD mice, glucose levels in blood collected from the tail vein of WT NOD-Ltj or NOD-Scid mice were monitored using the Ascensia Microfill blood glucose test strips (Bayer, Mishawaka, IN). All animal studies were approved by the animal care and use committee of University of Illinois at Chicago (UIC) and the Medical University of South Carolina (MUSC).
Peptide Ags, Cell Lines, and Abs
Immunodominant β-cell-Ag peptides, viz. 1) insulin B (9–23), 2) GAD65 (206–220), 3) GAD65 (524–543), 4) IA-2β (755–777), 5) IGRP (123–145), 6) BDC2.5 TCR reactive peptide (YVRPLWVRME; referred to as BDC peptide), and 7) OVA (323–339) peptides, were described in our earlier studies (14–16). Peptides 1–5 were pooled at an equal molar ratio and used as β-cell-Ag for in vitro and in vivo experiments.
Purified zymosan A of Saccharomyces cerevisiae was prepared as described previously (6,7). Bacterial lipopolysaccharide (LPS; Escherichia coli origin, ion-exchange purified), curdlan, phorbol myristic acid (PMA), ionomycin, brefeldin A, and monensin were purchased from Sigma-Aldrich, BD Biosciences, eBioscience, Invivogen, and Invitrogen. Normal rat serum, various fluorochrome-conjugated reagents and antibodies (Abs), and isotype control Abs (Invitrogen, BD Biosciences, eBioscience, R&D Systems, and Biolegend Laboratories) were used for FACS. Magnetic bead-based total and CD4+ T-cell and CD11c+ dendritic cell (DC) isolation kits (Miltenyi Biotec and Invitrogen) were used for enriching or depleting T cells and DCs. Paired Abs and standards for ELISA were purchased from R&D Systems, BD Biosciences, Invitrogen, and eBioscience.
Treating NOD Mice With Zymosan and β-Cell-Ag
Twelve-week-old euglycemic (glucose levels <110 mg/dL; prediabetic age) and 10–20-week-old early hyperglycemic (glucose levels between 140 and 250 mg/dL; early hyperglycemic stage) WT female NOD/Ltj mice were treated with zymosan and/or β-cell-Ag. Although insulitis in NOD mice is very heterogeneous at any given age, 12-week-old euglycemic mice represent the prediabetic stage because the hyperglycemia begins to appear at this stage. Mice were injected with zymosan (i.v. 25 μg/mouse/day on days 1, 3, 5, 16, 18, and 20) in PBS. Some groups of mice were injected intravenously with β-cell-Ag (0.5 μg/mouse/day on days 5 and 20) in PBS. Mice with glucose levels >250 mg/dL for two consecutive weeks were considered diabetic.
Experiments Using NOD-BDC2.5-TCR-Tg Mice and T Cells
Four-week-old NOD-BDC2.5-Foxp3-GFP mice were injected intravenously with bacterial LPS (5 μg/mouse/day) or zymosan (25 μg/mouse/day) for three consecutive days and BDC2.5 peptide (5 μg/mouse) on day 3. Spleen cells from these mice (2 × 106/mouse) were injected into 4-week-old WT NOD mice and examined for blood glucose levels every other day. In a separate experiment, 4-week-old WT NOD mice were treated with zymosan and/or β-cell-Ag as described above and injected with 2 × 106 ex vivo–activated BDC2.5-TCR-Tg T cells. The recipients were monitored for hyperglycemia every other day. In one set of experiments, 12-week-old euglycemic WT NOD mice were treated with zymosan and/or β-cell-Ag as described above, and CFSE-labeled BDC2.5-TCR-Tg T cells were injected intravenously (2 × 106/mouse). After 96 h, spleen and pancreatic lymph node (PnLN) cells from these mice were stained for Vβ4 and CD4 and examined for proliferating T cells by FACS.
DCs, T Cells, and In Vitro Assays
Splenic CD11c+ cells and total T cells were enriched using magnetic separation reagents. Bone marrow–derived DCs (BMDCs) were generated as described previously (6). Splenic CD11c+ DCs and BMDCs were incubated with zymosan (25 μg/mL) or LPS (2 μg/mL) for different time points. Pilot experiments using varying amounts of these agents were performed to determine the optimum culture conditions based on TNF-α production. cDNA prepared from DCs was subjected to qualitative and real-time quantitative PCR using SYBR Green PCR Master Mix (ABI Prism). DCs from 36-h cultures were examined for the levels of surface activation markers after staining with fluorochrome-labeled specific Abs. Spent media were tested for cytokine levels by ELISA.
Purified total T cells (1 × 105 cells/well) were incubated with zymosan-exposed or unexposed DCs (2 × 104 DCs/well) in the presence of anti-CD3 Ab (2 μg/mL). In some assays, DCs were incubated with β-cell-Ag (5 μg/mL) and zymosan (25 μg/mL) or LPS (2 μg/mL) agents overnight, washed, and cultured with CD4+ T cells. In some assays, spleen and PnLN cells (2 × 105 cells/well) from treated and control mice were stimulated with anti-CD3 Ab (2 µg/mL) or β-cell-Ag (5 μg/mL) for 48 h. Spent media from these cultures were tested for cytokines.
FACS Analysis
Freshly isolated and ex vivo–cultured cells were washed using PBS supplemented with 2% FBS and 10 mmol/L EDTA (pH 7.4) and blocked with anti-CD16/CD32 Fc block Ab or 5% rat serum on ice for 15 min. For surface staining, cells were incubated with fluorescein isothiocyanate–, phycoerythrin–, and phycoerythrin-cyanine 5– or phycoerythrin-Texas Red–labeled appropriate Abs, in different combinations, on ice for 45 min and washed three times before analysis. Cells were also stained using isotype-matched control Abs for determining the background.
DC and T-Cell Transfer Experiment
BMDCs were cultured in the presence of zymosan (25 μg/mL) or LPS (2 μg/mL) with β-cell-Ag (5 μg/mL) or Ag alone for 24 h, washed, and injected into 8-week-old prediabetic female NOD mice (i.v. 5 × 106 cells/mouse). In some experiments, total splenocytes (5 × 106 cells/mouse) or purified T cells (2 × 106 cells/mouse) from the spleens of control and treated mice (15 days posttreatment) were transferred into euglycemic female NOD mice and monitored as described above. In some experiments, freshly isolated T cells from BDC2.5-TCR-Tg mice were labeled with CFSE and injected into zymosan and/or β-cell-Ag–treated WT NOD mice. The recipients were killed 4 days post–T-cell transfer to determine the proliferation of donor T cells. In addition, BDC2.5-TCR-Tg T cells were activated using control and zymosan-exposed and BDC2.5 peptide–pulsed DCs, and T cells purified from these cultures were injected into 6-week-old WT mice (intravenously).
Histochemical Analysis and Examination of Inflammatory Response
Pancreata were fixed in 10% formaldehyde and 5-µm paraffin sections were made and stained with hematoxylin-eosin (H-E). Stained sections were analyzed using a grading system in which 0 = no evidence of infiltration, 1 = peri-islet infiltration (<5%), 2 = 5–25% islet infiltration, 3 = 25–50% islet infiltration, and 4 = greater than 50% islet infiltration, as described in our earlier studies (6,14–17). About 150 islets were examined for every group. In some experiments, pancreatic sections were stained using anti-insulin and antiglucagon Abs followed by Alexa Fluor 488– and 568–linked secondary Abs and DAPI and scored for insulitis based on DAPI-positive cells in islet areas and insulin expression. Insulitis was scored as described for H-E–stained sections, and insulin-positive and -negative islets were counted. Areas that appeared to have completely lost islets were considered as grade 5 and included in this grading approach. In some experiments, H-E–stained kidney and liver sections were examined for inflammation and tissue damage. Further, serum samples collected from zymosan-treated and control mice were tested for C-reactive protein (CRP) levels by ELISA (R&D Systems).
Statistical Analysis
Mean, SD, and statistical significance (P value) were calculated using Microsoft Excel, GraphPad online, and/or other online statistical applications. Two-tailed Student t test was used unless specified for values from in vitro and ex vivo assays. Log-rank analysis was performed to compare T1D incidence (hyperglycemia) of the test group with that of the control group. Fisher exact test was used for comparing the total number of infiltrated islets in test versus control groups. A P value of ≤0.05 was considered significant. In most experiments, unless specified, individual treated groups were compared with the nontreated group for calculating P values.
Results
TLR2 and Dectin 1 Engagement by Zymosan Modulates the Proinflammatory Response of DCs
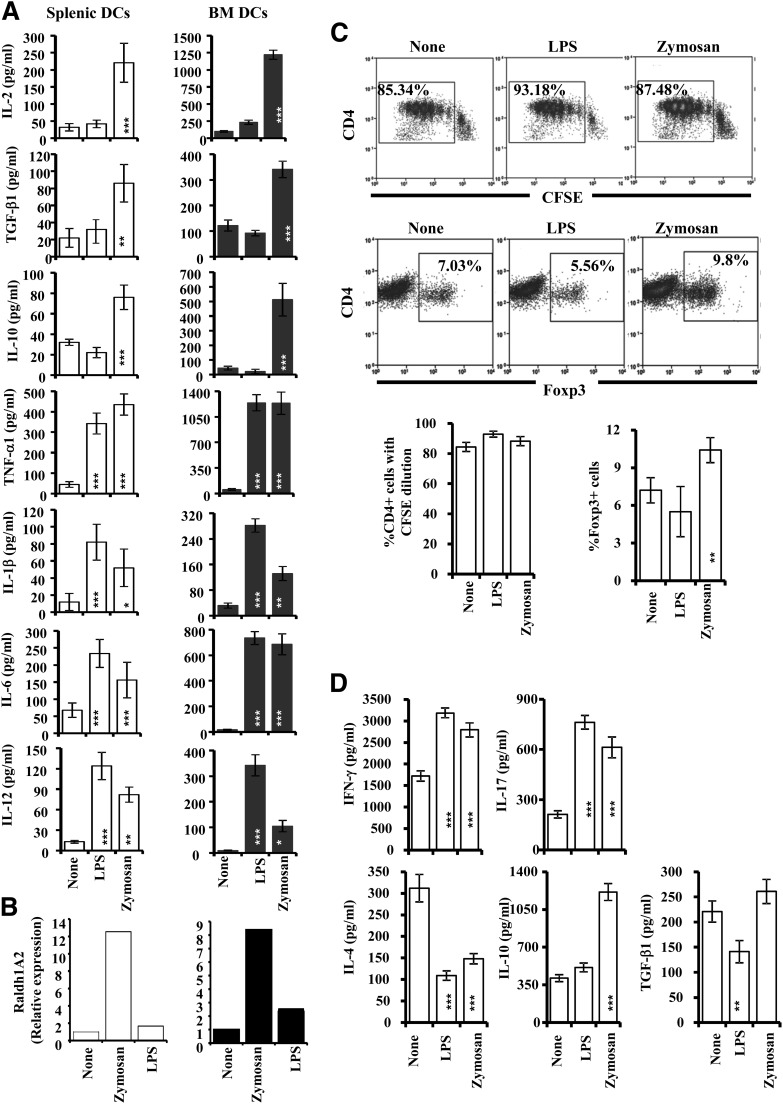
In this study, we compared the innate immune response induced by zymosan through TLR2 and Dectin 1 with that induced through TLR4 by bacterial LPS. BMDCs (generated using GM-CSF and IL-4, represent primarily myeloid DCs) and peripheral DCs were activated using LPS or zymosan and examined for the amount of cytokines secreted. As observed in Fig. 1A, whereas zymosan-exposed DCs produced large amounts of the immune regulatory cytokines IL-2, IL-10, and TGF-β1 along with proinflammatory cytokines (TNF-α, IL-6, IL-1β, and IL-12), LPS induced only proinflammatory cytokines in these DCs. We have also examined the ability of zymosan-exposed or unexposed DCs from NOD mice to express Raldh1A2 in comparison with DCs that are treated with LPS. As observed in Fig. 1B, exposure to zymosan, but not LPS, upregulated Raldh1A2 expression in both splenic and BMDCs, albeit at a much lower level (∼10-fold induction from basal level) than previously reported (∼200-fold induction from basal level) (10). However, expression of this enzyme, in combination with the observed immune regulatory cytokines, by DCs suggests that zymosan-exposed APCs may promote a Treg response upon Ag presentation.
Figure 1.
Zymosan-exposed DCs produce regulatory innate immune response and modulate T-cell response. Immature DCs, generated in vitro from bone marrow cells (BMDCs) using GM-CSF and IL-4 and freshly isolated splenic CD11c+ DCs, were left untreated or exposed to zymosan (25 μg/mL) or LPS (1 μg/mL) for different durations. A: Cytokine levels (as indicated) were measured by ELISA in supernatants obtained from the above cultures at the 48-h time point. Mean ± SD of values from three to four individual experiments carried out in duplicate or triplicate are shown. B: Cells harvested after 12 h were used in real-time quantitative PCR assays. Expression levels were calculated relative to housekeeping gene (actin) expression, and the values of zymosan- and LPS-treated samples were compared against the value of untreated (none) sample, which was considered as 1. This assay was repeated at least three times in duplicate and the mean values are shown. C: Splenic DCs were incubated with zymosan or LPS and OVA (323–339) peptide for 24 h, washed, and incubated with purified T cells (CFSE labeled or unlabeled) from OT-II-TCR-Tg mice for 96 h. CFSE-labeled cells were stained using fluorochrome-labeled anti-mouse CD4 Ab and examined for CFSE dilution by FACS. Cells, from wells where unlabeled T cells were used, were also stained for surface CD4 and intracellular Foxp3 and examined by FACS. Representative FACS graphs (upper panels) and mean ± SD of percent values (lower panels) of CD4+ T cells with CFSE dilution and Foxp3 expression are shown. This experiment was repeated at least four times with similar results. D: Cell-free supernatants from these T-cell cultures were tested for cytokine levels by ELISA. Mean ± SD of values from three individual experiments carried out in duplicate or triplicate are shown. Statistical significance was assessed by comparing the values of zymosan and LPS groups separately with that of control group by Student t test. *P < 0.05; **P < 0.01; ***P < 0.001.
Ag Presentation by Zymosan-Exposed DCs Induces Foxp3+ and IL-10–Secreting T Cells
Next, zymosan DCs were examined for their ability to modulate T-cell response using purified T cells from OT-II-TCR-Tg mice. As observed in Fig. 1C, DCs that were exposed to LPS and zymosan induced similar levels of proliferation in Ag-specific T cells. However, the zymosan-exposed DCs induced significantly higher frequencies of Foxp3+ cells in culture as compared with unexposed or LPS-exposed DCs. Examination of the cytokine profiles revealed that T cells activated using DCs that were exposed to zymosan produced significantly higher amounts of IFN-γ, IL-17, and IL-10 and reduced amounts of IL-4 as compared with untreated control DC-activated T cells (Fig. 1D). However, T cells that were activated using the TLR4 ligand produced high amounts of IFN-γ and IL-17 and lower amounts of TGF-β1, compared with control DC-activated T cells. These observations show that TLR2- and Dectin 1–engaged DCs and TLR4-engaged DCs modulate T-cell function differently.
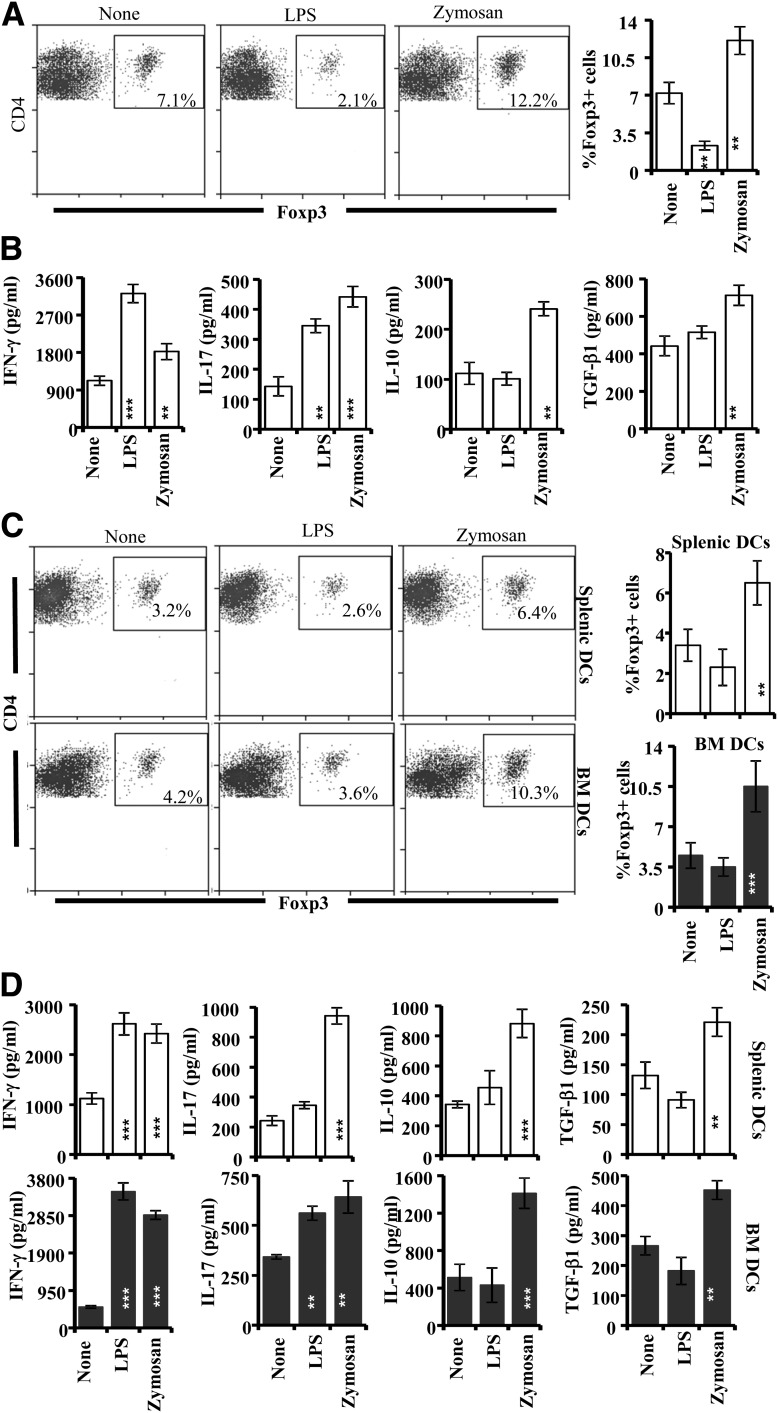
NOD Mouse T Cells That Were Activated in the Presence of Zymosan-Exposed APCs Show Regulatory Phenotype
Both BDC2.5-TCR-Tg cells and T cells from WT NOD mice demonstrate diabetogenic properties upon activation (14,17). Therefore, to further understand the effect of TLR2- and Dectin 1–mediated innate immune response on T cells in a T1D model, BDC2.5-TCR-Tg and WT NOD mouse T cells were activated in the presence of zymosan-exposed splenic DCs. The zymosan-exposed DCs induced significantly higher frequencies of Foxp3+ cells in the cultures of WT NOD mouse T cells as compared with unexposed or LPS-exposed DCs (Fig. 2A). On the other hand, when LPS-exposed DCs were used, considerably low frequencies of T cells in the cultures expressed Foxp3 as compared with controls (Fig. 2A). T cells from WT NOD mice produced higher IL-10 and TGF-β1 when activated in the presence of zymosan-exposed, but not LPS-exposed, DCs. (Fig. 2B). Similar to WT T cells, zymosan-exposed, but not LPS-exposed, DCs (both splenic and BMDCs) induced an increase in the frequency of Foxp3+ cells in BDC2.5-TCR-Tg T-cell cultures (Fig. 2C). BDC2.5-TCR-Tg T-cell cultures also showed significantly higher amounts of IL-10, TGF-β1, and IL-17 along with IFN-γ when zymosan-exposed DCs were used for Ag presentation (Fig. 2D). These observations show that innate immune response induced by zymosan, but not LPS, has the ability to modulate the properties of diabetogenic T cells and induce T cells with a regulatory phenotype.
Figure 2.
Zymosan-exposed, but not LPS-exposed, NOD mouse DCs induce Foxp3+ and IL-10+ T cells. A: Purified T cells from NOD-Foxp3-GFP mice were activated using anti-CD3 Ab in the presence of zymosan- or LPS-exposed splenic DCs for 96 h. Cells from these cultures were stained for surface CD4 and examined for GFP+ T cells. B: Supernatants from these NOD-Foxp3-GFP T-cell cultures were examined for the amounts of secreted cytokines by ELISA. C: Splenic and BMDCs were prepared, cultured without or with zymosan and LPS for 24 h and BDC2.5 peptide, washed, and incubated with purified T cells from NOD-BDC2.5-TCR-Tg or NOD-BDC2.5-Foxp3-GFP mice for 96 h. NOD-BDC2.5-Foxp3-GFP T cells were stained for CD4 and examined for CD4+GFP+ T cells. D: Supernatants from primary cultures of NOD-BDC2.5-TCR-Tg cells were examined for the amounts of secreted cytokines by ELISA. These experiments were repeated at least three times with similar results. Representative FACS graphs (left panels) and mean ± SD of percent values (right panels) of CD4+ T cells with GFP (Foxp3) are shown for panels A and C. Mean ± SD values are shown for panels B and D. Statistical significance was assessed by comparing the values of zymosan and LPS groups separately with that of control group by Student t test. **P < 0.01; ***P < 0.001.
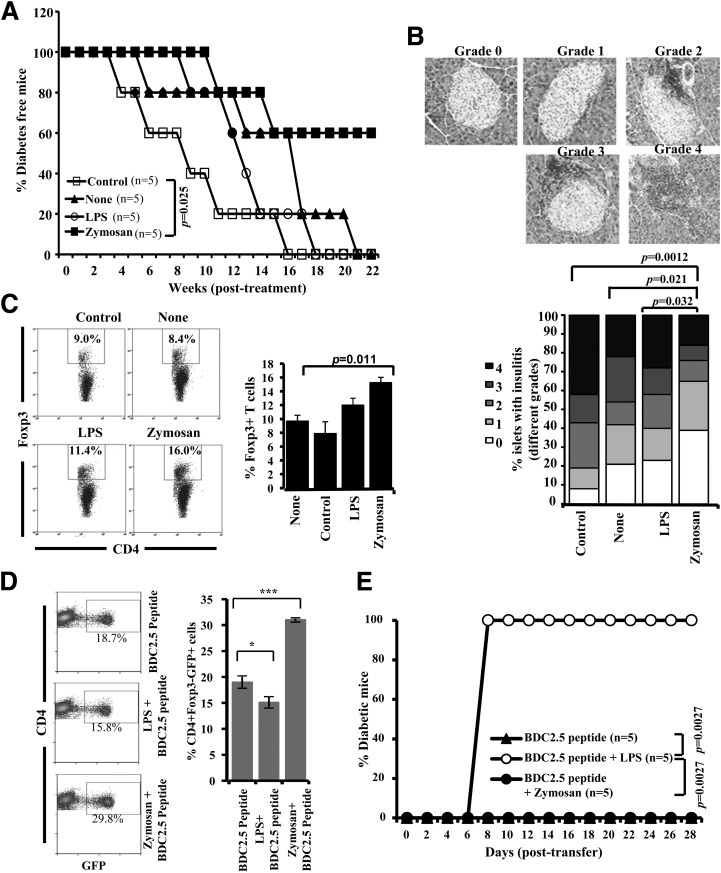
Zymosan-Exposed β-Cell-Ag–Pulsed DC Treatment Results in Suppressed Insulitis and Delayed Hyperglycemia in NOD Mice
To assess the immune regulatory nature of Ag presentation by zymosan-exposed DCs in vivo as compared with TLR4-engaged DCs, 8-week-old NOD mice were injected with zymosan- or LPS-exposed, β-cell-Ag–pulsed BMDCs and monitored for hyperglycemia. NOD mice that received zymosan-exposed, but not LPS-exposed, β-cell-Ag–pulsed DCs showed a significant delay in hyperglycemia as compared with β-cell-Ag–loaded control DC recipient mice (Fig. 3A). One set of euglycemic mice from a similar experiment was killed 30 days posttreatment and pancreatic tissues were examined for insulitis. As observed in Fig. 3B, mice that received zymosan-exposed and β-cell-Ag–pulsed DCs had significantly higher numbers of islets with less severe insulitis or no immune cell infiltration compared with untreated or control DC recipient mice. Spleen cells from one set of mice that were killed 15 days posttreatment were examined for the frequency of Foxp3+ T cells. As shown in Fig. 3C, the number of Foxp3-expressing CD4+ Tregs is significantly higher in mice that were injected with β-cell-Ag–pulsed, zymosan-exposed DCs as compared with β-cell-Ag–loaded (none) DC recipients or mice that received DCs that were not pulsed with β-cell-Ag (control). Although LPS-exposed, β-cell-Ag–loaded DC recipients showed a modest increase in Foxp3+ T cells compared with controls, they developed a more or less similar level of insulitis as the control DC recipients. These results suggest that β-cell-Ag presentation by the zymosan-exposed DCs has the ability to promote induction/expansion of Tregs in vivo.
Figure 3.
Zymosan does not have proinflammatory adjuvant activity like bacterial LPS. Eight-week-old euglycemic female NOD mice were injected intravenously with untreated (control group); β-cell-Ag–pulsed (none group); β-cell-Ag–pulsed, zymosan-exposed (zymosan group); or β-cell-Ag–pulsed, LPS-exposed (LPS group) BMDCs (2 × 106 cells/mouse). A: Mice were bled every week postinjection to examine blood glucose levels. The group that received zymosan- and/or LPS-exposed DCs was compared in a log-rank test to the control group for calculating the P value. B: One set of treated and control mice from parallel experiments were killed 30 days after the last injection. Pancreatic tissues were processed for H-E staining to evaluate insulitis as described in research design and methods. Islets with representative insulitis grade (upper panel) and the percentages of islets with different grades of lymphocyte infiltration plotted as bar diagram (lower panel) are shown. Sections of pancreatic tissues from three mice per group were examined for insulitis, and the insulitis score of at least 150 islets/group was plotted as a bar diagram. C: One set of mice was killed 15 days posttreatment and examined for CD4+Foxp3+ T-cell frequency in the spleen by FACS. Representative FACS graphs (left panel) and mean ± SD of percent values from three mice per group tested independently (right panel) are shown. Statistical significance was assessed by Student t test. D: Four-week-old NOD-BDC2.5-Foxp3-GFP mice were injected intravenously with zymosan (25 μg/mouse/day) and bacterial LPS (5 μg/mouse/day) for three consecutive days or left untreated. All three groups of mice received BDC2.5 peptide (5 μg/mouse) on day 3. Spleen cells from these mice were tested for GFP expression in CD4+ T cells by FACS. Representative FACS graphs (left panel) and mean ± SD of percent values (right panel) of CD4+ T cells with GFP (Foxp3) expression are shown. *P < 0.05; ***P < 0.001. E: T cells from the groups of mice described for panel C were injected into 4-week-old WT NOD mice (2 × 106 cells/mouse) and tested for blood glucose levels every other day. Mice with a blood glucose level of 250 mg/dL for two consecutive tests were considered diabetic. Log-rank test was used to calculate P values.
T Cells From LPS-Treated, but Not Zymosan-Treated, BDC2.5-TCR-Tg Mice Induce Hyperglycemia in Young NOD Mice
To assess whether zymosan and LPS induce proinflammatory adjuvant effects in vivo and promote the generation of diabetogenic T cells, young BDC2.5-Foxp3-GFP mice were injected with bacterial LPS or zymosan along with the BDC2.5 peptide. Zymosan-injected NOD-BDC2.5-Foxp3-GFP mice showed a profound increase in the CD4+GFP+ T-cell numbers, compared with the control group of mice (Fig. 3D). On the other hand, LPS recipients showed lower CD4+GFP+ T-cell frequency compared with control mice. Importantly, T cells from only LPS-recipient, but not zymosan-recipient, mice could induce hyperglycemia in young NOD mice (Fig. 3E). T cells from mice that received BDC2.5 peptide alone did not induce hyperglycemia, perhaps due to tolerogenic Ag presentation by steady-state APCs in the absence of adjuvant treatment in these peptide-recipient mice. Whether intravenous injection with zymosan can lead to a strong inflammatory response was also assessed by determining the serum levels of CRP in zymosan-treated mice. As observed in Supplementary Fig. 1, a marginal increase in the serum CRP levels was detected 48 h after zymosan treatment. On the other hand, intravenous injection with LPS induced a fivefold increase in the serum CRP levels as compared with control. These results indicate that zymosan does not have a strong proinflammatory adjuvant property, as opposed to that of LPS.
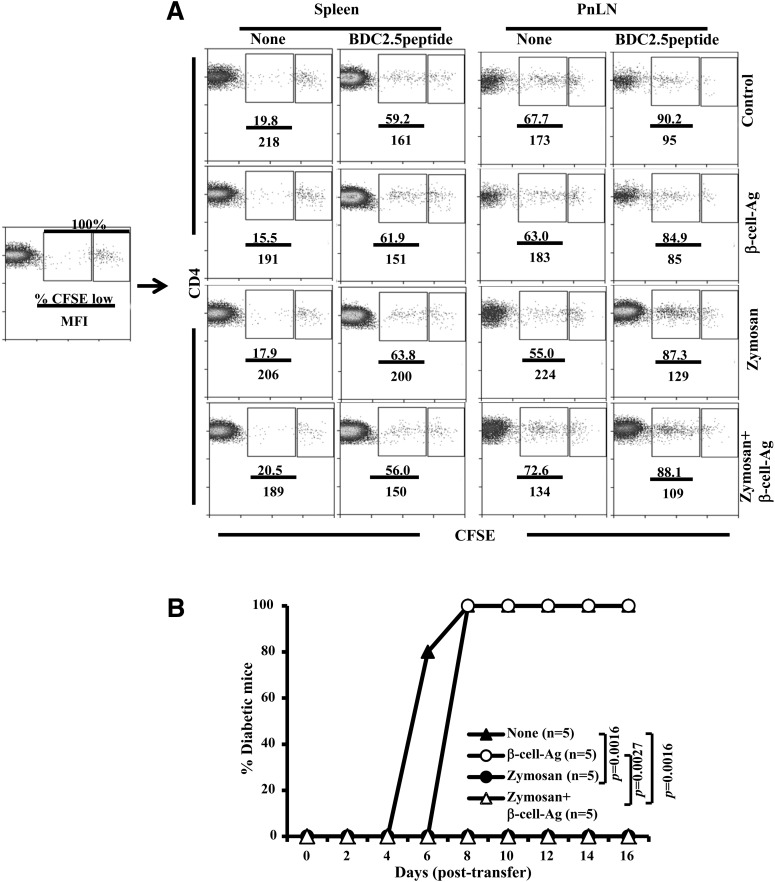
Zymosan-Induced Innate Immune Response Does Not Suppress β-Cell-Ag–Specific T-Cell Proliferation but Modulates Their Diabetogenic Function
Next, we examined whether zymosan treatment–induced modulation of immune response to β-cell-Ag affects T-cell proliferation in vivo. Twelve-week-old (prediabetic age) NOD mice were treated with zymosan and/or β-cell-Ag, injected with CFSE-labeled BDC2.5-TCR-Tg T cells alone or along with BDC2.5 peptide, and examined for CFSE dilution in donor cells by FACS. As anticipated, PnLN, but not the spleen, of control mice showed profound proliferation of BDC2.5 T cells in the absence of BDC2.5 peptide injection (Fig. 4A). Importantly, PnLN of all groups of mice that were treated with zymosan and/or β-cell-Ag also showed a significant number of proliferating BDC2.5 T cells. PnLN from all BDC2.5 peptide–injected mice showed a more or less similar extent of BDC2.5 T-cell proliferation (Fig. 4A). In addition, upon peptide injection, spleen cells from zymosan-treated and untreated mice showed a similar extent of BDC2.5 T-cell proliferation. These results, in association with the observations of Figs. 2 and 3, suggest that although the zymosan-induced innate immune response may not have a significant suppressive effect on T-cell proliferation, it may influence the functionality of these T cells.
Figure 4.
Zymosan-induced innate immune response does not suppress β-cell-Ag–specific T-cell proliferation but modulates their diabetogenic function in vivo. Twelve-week-old prediabetic age WT NOD mice were injected intravenously with zymosan (25 μg/mouse/day) for three consecutive days or left untreated. One set of treated and untreated mice received β-cell-Ag (0.5 μg/mouse) on day 3. A: On day 5, these mice were injected intravenously with CFSE-labeled T cells from NOD-BDC2.5-TCR-Tg mice (2 × 106 cells/mouse) and killed on day 9; CFSE dilution in CD4+Vβ4+ T cells was examined by FACS. Vβ4+ cells were gated for the graphs shown. Please note that 100% of CD4+ T cells from donor BDC2.5 mice are Vβ4+; ∼7.0% WT NOD (recipient) mice T cells are also Vβ4+. Therefore, the indicated approach for analysis has been used to calculate percent CFSE low cells and mean fluorescence intensity (MFI) values for each graph, as shown in the far left panel. Representative graphs from two independent experiments are shown. B: Zymosan- and β-cell-Ag–treated and control mice were injected with ex vivo BDC2.5 peptide–activated T cells from NOD-BDC2.5-TCR-Tg mice (2 × 106 cells/mouse) on day 5, and the recipient mice were monitored for hyperglycemia as described for Fig. 3. P values were calculated by log-rank test.
Our previous report (14) has shown that young WT NOD mice injected with in vitro–activated TCR-Tg T cells can serve as an effective adoptive transfer model for T1D to assess if the Ag-specific T cells are pathogenic. In this model, if the TCR-Tg T cells are pathogenic, then 100% of recipient mice develop overt hyperglycemia within 6–8 days. If the T cells are not pathogenic in nature, then the recipients fail to develop T1D for months. Therefore, to further assess the regulatory nature of TLR2- and Dectin 1–dependent innate immune response, young WT NOD mice were treated with zymosan and/or β-cell-Ag, followed by injection of in vitro–activated diabetogenic BDC2.5-TCR-Tg T cells. As observed in Fig. 4B, diabetogenic BDC2.5-TCR-Tg induced overt hyperglycemia in control and β-cell-Ag–treated mice within 8 days. However, these T cells failed to induce hyperglycemia in zymosan- and zymosan plus β-cell-Ag–treated groups of mice. These results suggest that zymosan-induced innate immune response alters the function of diabetogenic T cells in vivo.
Zymosan Treatment Does Not Produce Side Effects Such as Global Immune Suppression but Causes Increased Cell Death in PnLN
Various effects of TLR2- and Dectin 1–dependent innate immune response on secondary lymphoid organs as well as nonlymphoid organs were examined to further understand the potential side effects of zymosan treatment. Twelve-week-old (prediabetic age) mice were treated with zymosan, and various tissues were examined for cell death. Cells from the pancreas, PnLN, and spleen were examined for dead/dying (both apoptotic and necrotic) cells by Annexin V and 7-AAD staining. While spleen cells from zymosan- and zymosan plus β-cell-Ag–recipient mice did not show a difference in cell death, PnLN of these treated mice had a higher number of Annexin V– and/or 7-AAD–positive cells compared with control mice (Supplementary Fig. 2A). Importantly, CD4+ T cells from pancreas and PnLN of only zymosan plus β-cell-Ag–recipient mice, but not zymosan alone or the control group of mice, showed considerable cell death (Supplementary Fig. 2B). These results suggest that zymosan treatment causes immune cell death at sites of high inflammation, and in addition to the regulatory cytokine–mediated effect, apoptotic elimination of activated immune cells, including T cells, could be a feature of zymosan-induced immune modulation.
To assess whether the zymosan treatment affects DC numbers and their functionality, leading to T-cell death, pancreatic, PnLN, and spleen cells were examined for the frequencies of DCs. As observed in Supplementary Fig. 3A, treatment with zymosan at the dose selected for this study did not induce a considerable increase in the number of CD11c+ DCs in the pancreas, PnLN, or spleen. Further, the expression levels of apoptotic ligands FAS-L and TRAIL were comparable in the pancreas, PnLN, or spleen DCs of control and zymosan-treated groups of mice. Further analysis revealed that myeloid (CD8a−), lymphoid (CD8a−), and plasmacytoid (PDCA1) DC frequencies were not noticeably different in the spleen and PnLN of control and zymosan-treated mice (Supplementary Fig. 4). Although the Ag-presenting function of DCs from zymosan-treated and control mice may be different in vivo, these observations indicate that the frequency of DCs is not affected by zymosan treatment and they may not have a direct role in causing T-cell death in the pancreatic microenvironment of zymosan-treated mice.
We have also examined whether zymosan treatment induces side effects such as inflammation in nonlymphoid organs like kidney and liver. As observed in Supplementary Fig. 5, intravenous injection with low-dose zymosan does not cause noticeable inflammation in the nonlymphoid organs such as kidney and liver. To assess whether zymosan and β-cell-Ag treatment induces general immune suppression, the ability of T cells from treated NOD mice to proliferate against alloantigen in a mixed lymphocyte reaction assay was tested. As observed in Supplementary Fig. 6, T cells from zymosan- and zymosan plus β-cell-Ag–treated mice showed a more pronounced proliferative response than T cells from untreated or β-cell-Ag–treated NOD mice. While this enhanced proliferative ability of T cells from zymosan-treated mice, compared with control T cells, could be an effect of the cytokines IL-2 and TNF-α produced by zymosan-exposed APCs, this result suggests that zymosan treatment does not produce general immune suppression.
β-Cell-Ag Delivery Along With Zymosan Results in the Prevention of T1D
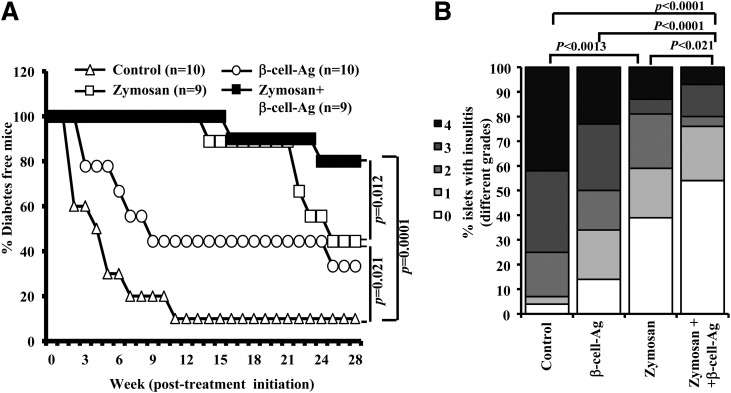
As shown in our previous report (6), the innate immune response induced in NOD mice by injecting a higher dose of zymosan (100 μg/injection, i.p.) could achieve long-term protection from T1D. Results from experiments using zymosan-exposed DCs presented in Fig. 3 suggest that a relatively short-term treatment, with smaller doses of zymosan, may be sufficient to achieve a long-lasting therapeutic effect in conjunction with β-cell-Ag. Twelve-week-old prediabetic age NOD mice were coadministrated with zymosan and β-cell-Ag and monitored for hyperglycemia. As observed in Fig. 5A, mice that were treated with zymosan and β-cell-Ag remained diabetes free for a significantly longer duration than the mice that received zymosan alone. In addition, as observed in our previous study (13), mice that received zymosan also showed significant protection from T1D as compared with untreated mice.
Figure 5.
Treatment using zymosan and β-cell-Ag resulted in better protection of NOD mice from hyperglycemia as compared with treatment with zymosan alone. Twelve-week-old euglycemic female NOD mice were pooled, randomly picked, and left untreated (control) or treated with zymosan (on days 1, 3, 5, 13, 15, 17, 25, 27, and 29 with 25 μg/mouse/day). A group of animals received β-cell-Ag on days 5, 17, and 29. A: Mice were checked every week for hyperglycemia, and blood glucose level of 250 mg/dL for two consecutive weeks was considered diabetic. Log-rank test was performed to compare different groups of mice, and the P value is shown on each graph. The group that received β-cell-Ag was also compared with control mice. B: One set of treated and control mice from parallel experiments was killed 4 weeks after the last injection, and pancreatic tissues were processed for H-E staining to evaluate insulitis as described in research design and methods. The percentages of islets with different grades of insulitis plotted as a bar diagram are shown. Sections of pancreatic tissues from at least four mice per group were examined for insulitis, and the insulitis score of at least 150 islets/group were plotted as a bar diagram.
The pancreatic islets of prediabetic mice that received zymosan and β-cell-Ag showed significantly less severe immune cell infiltration and insulitis compared with peptide-treated and untreated control mice within 30 days posttreatment (Fig. 5B). Whereas the mice that received low-dose zymosan alone also showed significantly less severe insulitis compared with untreated mice, the difference in insulitis observed between pancreatic islets of mice that received β-cell-Ag alone and untreated controls was not statistically significant, at 30 days posttreatment. These results suggest that exposure of APCs to zymosan in vivo leads to the skewing of T-cell response from pathogenic to protective type.
Early Hyperglycemic Mice Show Prolonged Protection From Diabetes When β-Cell-Ag Is Delivered Along With Zymosan
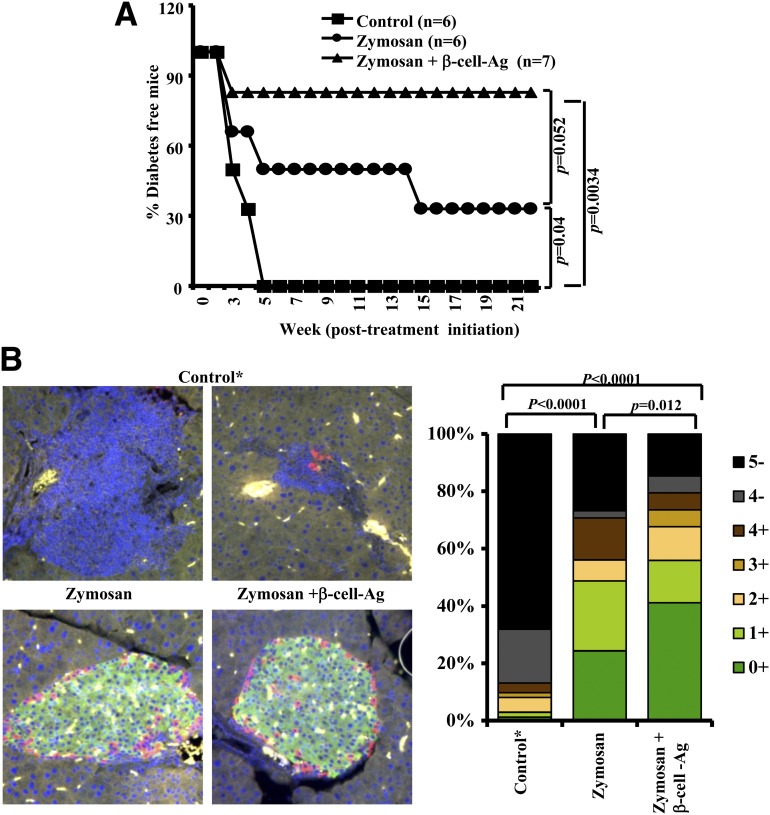
Encouraged by the results of Fig. 5, we examined whether modulating the immune response against β-cell-Ag, with the help of zymosan-induced innate immune response, can control autoimmunity in early hyperglycemic mice and restore euglycemia. Early hyperglycemic (glucose 140–250 mg/dL) mice were treated with zymosan alone or along with β-cell-Ag and examined for blood glucose levels every 3rd day. One hundred percent of noninjected control mice were diabetic within 6 weeks posttreatment. However, the majority of the mice that received zymosan along with β-cell-Ag showed normal glucose levels and remained diabetes free for >20 weeks posttreatment (Fig. 6A and Supplementary Fig. 7). Although less profound, mice that received zymosan alone also showed significant protection from diabetes compared with control mice.
Figure 6.
Treatment using zymosan and β-cell-Ag resulted in better protection of early hyperglycemic mice from diabetes as compared with mice that were treated with zymosan. A: Early hyperglycemic mice (glucose levels between 140 and 250 mg/dL; screened from 12–20-week-old mice, pooled, and randomly picked) were left untreated or treated intravenously with zymosan (on days 1, 3, 5, 13, 15, 17, 25, 27, and 29 with 25 μg/mouse/day). Some animals received β-cell-Ag on days 5, 17, and 29. Mice were bled every 3 days for glucose levels. Mice with glucose levels 250 mg/dL for two consecutive weeks were considered diabetic. Log-rank test was performed to compare the hyperglycemia incidence in treated and control groups of mice, and the significant P values are shown. Glucose levels of each group are shown in Supplementary Fig. 5. B: Pancreatic tissue obtained from a parallel set of euglycemic-treated and control mice, 4 weeks after the final injection, were sectioned and stained using anti-insulin Ab followed by anti-rat IgG Alexa Fluor 488 reagent and antiglucagon Ab followed by anti-rabbit IgG Alexa Fluor 568 reagent and mounted using antifade containing DAPI. Insulitis was scored based on DAPI staining as described in research design and methods. Representative islet areas (left panel) and the percentages of insulin-positive and -negative islets with different grades of insulitis plotted as a bar diagram (right panel) are shown. At least 60 islets from a total of three to four mice were examined for each group. Statistical significance was calculated by comparing insulin-positive islets with <50% infiltration; grade ≤3 insulitis. *Since 100% of control mice developed overt hyperglycemia within 5 weeks posttreatment initiation (no islet structures were left at this stage), a fresh batch of mice with glucose levels between 140 and 250 mg/dL at the time of testing were included as a surrogate control. Insulin- and glucagon-positive islet areas were not detected in pancreatic tissues from overt hyperglycemic mice (not shown). Yellow fluorescence appears to be nonspecific staining and/or autofluorescence.
Examination of the pancreatic tissue from one set of these mice, 30 days posttreatment, revealed that zymosan- and β-cell-Ag–treated mice have a significantly higher number of infiltration-free islets or islets with less severe insulitis compared with other groups after 30 days posttreatment (Supplementary Fig. 8A). Another set of mice from a similar experiment were killed on day 30, and the pancreatic tissue sections were stained for insulin and glucagon to assess islet function. Although differences in the overall insulitis severities between zymosan and zymosan plus β-cell-Ag groups were not statistically significant in Supplementary Fig. 6, the latter group showed a profound increase in the number of insulin-positive islets compared with zymosan recipients (Fig. 6C). Overall, the percentage of insulin-positive islets was significantly low in control and β-cell-Ag–treated mice compared with zymosan and zymosan plus β-cell-Ag recipients. These results suggest that the zymosan-induced innate immune response can significantly affect the infiltration of lymphocytes into the pancreatic islets, resulting in protection of insulin-producing islets from autoimmune destruction.
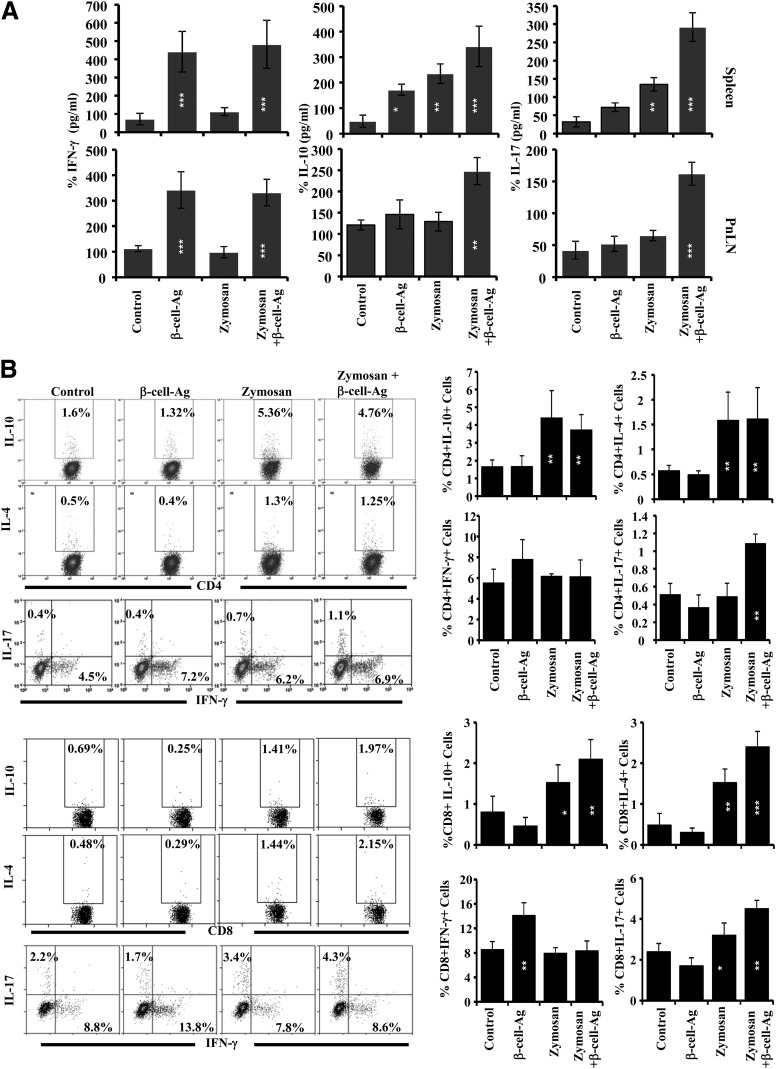
T Cells From Zymosan and β-Cell-Ag–Treated Mice Proliferate Against β-Cell-Ag and Produce IL-17 and IL-10
Spleen and PnLN cells obtained from mice that were killed 30 days posttreatment were examined for their ability to respond to an ex vivo challenge with β-cell-Ag. Both CD4+ and CD8+ T cells from mice that received β-cell-Ag or zymosan plus β-cell-Ag showed a comparable extent of CFSE dilution upon ex vivo challenge, which was significantly higher than that of their counterparts from untreated control mice (Supplementary Fig. 8B). However, the cytokine profiles of spleen and PnLN cells from zymosan- and β-cell-Ag–recipient groups were significantly different when challenged with β-cell-Ag ex vivo (Fig. 7A). Whereas all β-cell-Ag recipient groups produced comparable levels of IFN-γ, which is significantly higher than mice that have not received β-cell-Ag, zymosan plus β-cell-Ag recipients produced significantly higher amounts of IL-10 and IL-17 as compared with mice that received β-cell-Ag alone. Further, as observed in Fig. 7B, whereas the CD4+IFN-γ+ T-cell frequencies were comparable in all groups upon nonspecific activation using PMA and ionomycin, similar to the observations of Fig. 7A, all zymosan-recipient groups had significantly higher frequencies of IL-10– and IL-4–expressing cells. Importantly, CD8+ T cells not only showed a similar trend but also the difference in frequencies of IL-10– and IL-4–producing cells was more pronounced among these T cells in zymosan plus β-cell-Ag–treated mice, compared with untreated controls. Moreover, zymosan β-cell-Ag–recipient mice showed a higher number of IL-17–producing CD4+ and CD8+ T cells as compared with control groups. On the other hand, higher numbers of CD8+IFN-γ+ T cells were detected in β-cell-Ag–treated, but not zymosan plus β-cell-Ag–treated, mice. These observations suggest that zymosan-exposed APCs skew the T-cell response against β-cell-Ag from IFN-γ production to IL-17, IL-10, and/or IL-4 production (from pathogenic Th1 and TC1 to less pathogenic or T1D-protective Th17 and TC17, and Th2 and TC2) in vivo.
Figure 7.
Treatment using zymosan and β-cell-Ag modulates the T-cell response in NOD mice. Twelve-week-old NOD mice were treated with zymosan and/or β-cell-Ag as described for Fig. 5, killed 30 days posttreatment, and spleen and PnLN cells were examined for proliferative and cytokine responses. A: Spleen and PnLN cells were cultured with β-cell-Ag for 72 h, and cell-free supernatants were tested for cytokine levels by ELISA. Mean ± SD of values from at least three mice per group tested in triplicate are shown. B: Freshly isolated cells were stimulated with PMA and ionomycin in the presence of brefeldin A for 4 h and stained for CD4+ (upper panels) and CD8+ (lower panels) cells and intracellular cytokines for FACS analysis. Representative FACS graphs of splenic CD4+ and CD8+ cells with specific cytokine expression (left panels) and mean ± SD of percent values of cells from four to five mice tested independently (right panels) are shown. Statistical significance was assessed by comparing the values of treated groups with that of the untreated control group by Student t test. *P < 0.05; **P < 0.01; ***P < 0.001.
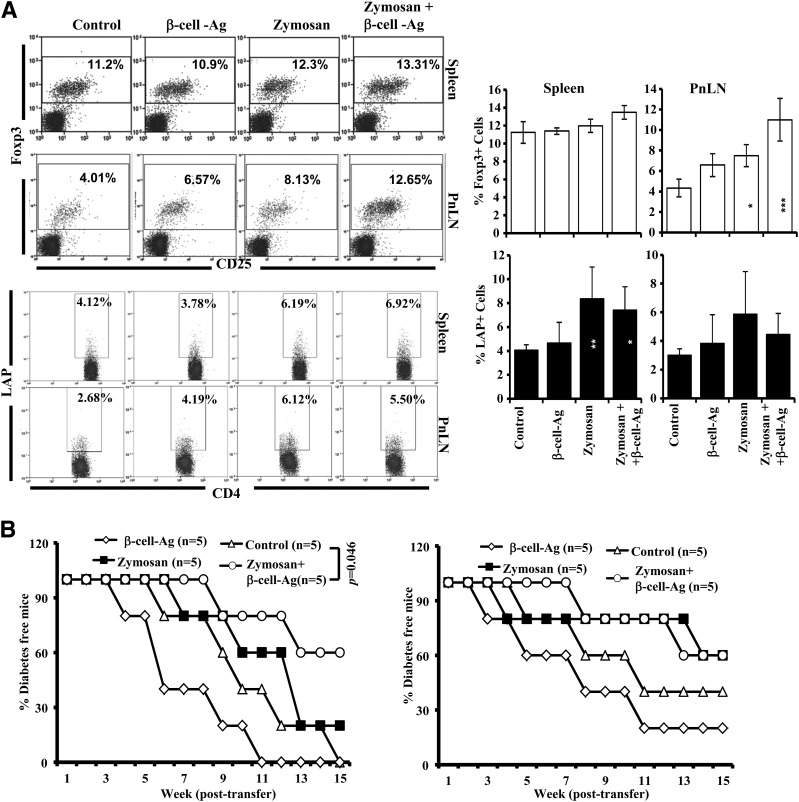
Treatment With Zymosan and β-Cell-Ag Leads to Increase in T Cells With Regulatory Phenotype
To realize whether zymosan and/or β-cell-Ag treatment can induce and /or expand T cells with regulatory properties, spleen and PnLN cells from zymosan- and/or β-cell-Ag–treated mice were examined for Foxp3+ and LAP+ T cells by FACS. As observed in Fig. 8A, significantly higher numbers of Foxp3+CD4+ T cells were found in the PnLN of mice treated with zymosan and β-cell-Ag as compared with control mice. However, the splenic Foxp3+CD4+ T-cell frequencies in zymosan- and β-cell-Ag–treated mice were not different. Significantly higher numbers of splenic CD4+ T cells from mice that were treated with zymosan alone or along with β-cell-Ag were positive for surface LAP, and most of these LAP-positive cells were found to be CD25− (not shown). Splenic Tregs from the mice that were killed 15 days posttreatment were adoptively transferred into 8- and 10-week-old female NOD mice to determine their potential to prevent hyperglycemia. CD4+CD25+ and LAP+ T cells isolated from the spleen of treated and untreated mice did not show significant differences in their ability to suppress autoimmunity, when transferred into young WT female mice (not shown), suggesting that the per cell suppressive abilities of these purified populations from treated mice are not superior to their control counterparts. On the other hand, as observed in Fig. 8B, prediabetic (8-week-old) mice that received total T cells from the spleen of zymosan plus β-cell-Ag–treated mice showed higher protection from T1D compared with mice that received T cells from control groups of mice. Ten-week-old mice that received T cells from zymosan alone or along with β-cell-Ag also developed hyperglycemia relatively slowly compared with mice that received T cells from control mice. Moreover, WT NOD mice that received total spleen cells from treated and untreated mice (Supplementary Fig. 9) and NOD-Scid mice that received purified T cells also showed similar disease incidence trend as that of WT NOD mice (not shown). These observations, in association with Fig. 7, not only suggest that T cells play a key role in zymosan plus β-cell-Ag treatment–induced protection from T1D but also indicate that the overall increase in multiple populations of T cells with regulatory phenotype, and not the superior ability of specific subsets, contributes to this disease protection.
Figure 8.
Treatment using zymosan and β-cell-Ag results in the induction of Foxp3+ and LAP+ T cells. Twelve-week-old NOD mice were treated with zymosan and/or β-cell-Ag as described for Fig. 5. A: One set of mice was killed 30 days posttreatment, and spleen and PnLN cells were examined for surface CD4 and intracellular Foxp3 and analyzed by FACS (upper panels). Cells were also stained for surface CD4 and LAP for FACS analysis (lower panels). CD4+ population was gated for all panels shown. Representative FACS graphs (left panels) and mean ± SD of percent values of cells from at least five mice per group tested independently (right panels) are shown. Statistical significance was assessed by comparing the values of treated groups with that of untreated control group by Student t test. *P < 0.05; **P < 0.01; ***P < 0.001. B: Splenic T cells were purified from treated and untreated groups of mice and injected intravenously into 8-week-old (left panel) and 10-week-old (right panel) female NOD mice (2 × 106 cells/mouse; five mice per group). These T-cell recipients were tested for blood glucose levels every week, and the mice that showed glucose levels >250 mg/dL for two consecutive weeks were considered diabetic. Log-rank test was used to assess statistical significance.
Discussion
Previously, we and others have reported that inducing innate immune response through PRRs like TLR2 and Dectin 1 using the fungal cell wall agent zymosan could suppress autoimmunity and prevent T1D in NOD mice (6,8). Here, we show that coadministration of β-cell-Ag and zymosan leads to prolonged protection of NOD mice from diabetes, and this effect appears to be dependent on a combination of zymosan-induced regulatory innate immune response involving IL-10, TGF-β1, IL-2, and Raldh1A2 and modulation of T-cell response by zymosan-exposed APCs.
It has been shown that engagement of TLR2 by its ligands induces both pro- and anti-inflammatory cytokine responses by APCs and IL-10 in T cells (18–22). However, our observations show that the TLR2 ligands Pam3Cys and Pam2Cys do not induce IL-2 and TGF-β1 in APCs independently of Dectin 1 engagement (Supplementary Fig. 10). The synergistic signaling through TLR2 and Dectin 1 (23–26), although it does not induce a significant increase in the expression of coinhibitory ligands such as PD-L1 and PD-L2, triggers the production of high levels of immune regulatory factors IL-10, TGF-β1, IL-2, and Raldh1A2 (6,7,10). Considering the involvement of these factors in immune regulation (10,27–30), we hypothesized that Ag presentation by zymosan-exposed APCs that produce these factors can skew the T-cell response from proinflammatory to anti-inflammatory/regulatory type. Our observations that Ag presentation by zymosan-exposed DCs can promote IL-10 and Foxp3 expression in T cells support this notion.
Since T1D is the result of gradual expansion of autoreactive T cells and an increase in inflammation, exposure of APCs to the immunodominant self β-cell-Ag–specific peptides in early hyperglycemic NOD mice can result in the induction and/or expansion of pathogenic T cells, leading to early hyperglycemia (14,15). Our observation that treatment with β-cell-Ag and zymosan induces protection from T1D in prediabetic NOD mice, more significantly, as compared with treatment with zymosan or β-cell-Ag alone, suggests that the zymosan-exposed APCs modulate T-cell response against β-cell-Ag in vivo, and this modulated response is protective in nature. This notion is substantiated by the observation that zymosan-exposed, β-cell-Ag–loaded DCs could induce better Treg response and prevent T1D compared with β-cell-Ag–loaded, nontreated DCs in NOD mice. Importantly, although the treatment of NOD mice with low-dose zymosan does not cause an increase in the frequencies of DCs or their subsets in the spleen and pancreatic microenvironment, the Ag-presenting ability of these cells may profoundly be altered by exposure to zymosan in vivo.
The protective nature of T cells from zymosan- and β-cell-Ag–treated mice was evident from higher frequencies of T cells with the regulatory phenotype, in PnLNs in particular, and their ability to produce IL-10 and IL-4 upon challenge/exposure to β-cell-Ag. Interestingly, T cells from mice that were treated with zymosan and β-cell-Ag also produce relatively higher amounts of IL-17. These observations indicate that zymosan-induced innate immune response contributes to the skewing of the T-cell response against β-cell-Ag from IFN-γ production (pathogenic Th1 and TC1) to IL-17, IL-10, and/or IL4 production (less pathogenic or T1D protective Th17 and TC17, and Th2 and TC2) in vivo. Although T1D is generally considered to be a Th1-mediated autoimmune disease, skewing immune response to Th17 and Th2 type results in protection of NOD mice from the disease (31,32). Recent studies, including ours, have shown that a shift in the immune response in NOD mice from Th1 to Th17 under various conditions can lead to prevention of the disease or delayed hyperglycemia (31,32). On the other hand, the potential contribution of a shift in the T-cell response toward IL-4 production when peptide vaccine, oral tolerance, and adjuvant therapy approaches are used has been well recognized for many years (33–37). Interestingly, we observed that the percentages of IL-10– and IL-4–positive T cells were higher in the spleen of both zymosan alone and zymosan plus β-cell-Ag groups, indicating that these cells may be the result of zymosan-induced innate immune response. On the other hand, an increase in the percentage of IL-17–producing cells was observed primarily in zymosan plus β-cell-Ag–treated mice, indicating a T-cell response specific to this Ag under conditions of zymosan-induced innate immune response. In addition to the difference in cytokine profiles of T cells from zymosan- and β-cell-Ag–treated and control mice, these treated mice showed higher Foxp3+ T-cell frequencies in the PnLN, but not spleen, compared with zymosan-treated mice. This suggests selective trafficking of Foxp3+ cells to the pancreatic microenvironment.
Treatment with proinflammatory agents such as bacterial LPS (TLR4 ligand) and poly I:C (TLR3 ligand) can delay hyperglycemia in NOD mice when the treatment is initiated at early stages (38–44). Although the potential mechanism is not known, a previous study has shown that the prolonged treatment of NOD mice at different stages of disease progression with LPS from Salmonella enterica results in an increase in the Treg numbers and protection from the T1D (39). Our observations show that treatment with E. coli LPS-exposed DCs can also result in a modest increase in Foxp3+ cells in vivo, but they fail to produce protection of the NOD mice from T1D. A role for TNF-α produced by APCs upon exposure to these TLR ligands and other microbial agents and a better immune regulation achieved through immune potentiation and activation of Tregs, especially at early stages of insulitis, have been suggested (45). In fact, previous reports have shown that TNF-α has contradictory effects at different stages of disease development in NOD mice (46,47). On the other hand, we and others have reported the unique ability of zymosan to induce the expression of IL-10, TGF-β1, IL-2, PD-L1, and/or Raldh1A2 in APCs (6–10,48) and promote protection from autoimmune diseases, even at prediabetic age and early hyperglycemic stage. Increased cell death in the PnLN, but not spleen, of zymosan-treated mice suggests an additional mechanism to eliminate inflammatory immune cells and suppress autoimmunity. Lack of significant difference in the expression levels of FAS-L and TRAIL on DCs from zymosan-treated mice suggests that cell death in the pancreatic microenvironment could be DC independent. On the other hand, our current observations that only zymosan, but not LPS, could induce immune regulatory factors such as IL-2, IL-10, and TGF-β1 in DCs, suppress the diabetogenic T cell function, and promote protection from T1D further demonstrate the regulatory nature of the zymosan-associated innate immune response.
In spite of the disease-promoting effect of zymosan in a genetically susceptible mouse model of arthritis (49), the ability of zymosan to ameliorate and/or prevent disease progression has been demonstrated in T1D and EAE models (6–11). Whereas low-dose zymosan protected mice from EAE, treatment with a high dose caused initiation of EAE through Th17 cells (11,50). Th17 cells, unlike in the T1D model where they are protective/less pathogenic (31,32), play a pathogenic role in EAE, arthritis, and many other autoimmune conditions (51–53). However, in association with previous reports, including ours (31,32), we demonstrate that skewing the T-cell response to IL-17 production instead of IFN-γ, in combination with other factors such as Tregs, IL-10, and IL-4, results in suppressed autoimmunity and delayed hyperglycemia in NOD mice.
In conclusion, our observations show that the TLR2- and Dectin 1–dependent innate immune response induced by zymosan is significantly different from that induced by a TLR4 ligand E. coli LPS. Whereas cooperative signaling though TLR2 and Dectin 1 induced by zymosan (23–26) involves both pro- and anti-inflammatory responses, signaling through TLR4 induces primarily proinflammatory responses in DCs. Although the role of zymosan-induced cell death at the site of inflammation in reduced insulitis and protection of NOD mice from T1D requires further investigation, our current study shows that the synergistic innate immune response mediated by TLR2 and Dectin 1 has the unique ability to not only produce regulatory innate immunity but also modulate T-cell response to β-cell-Ag and protect NOD mice from T1D. Importantly, our observations show that an overall increase in T cells with regulatory properties and a skewing in the immune response (cells that express Foxp3, LAP, IL-10, IL-17, and IL-4), rather than the superior suppressive ability of an individual population, is responsible for the protection of NOD mice from T1D. We conclude that zymosan has the potential to be used as a tolerogenic/immune regulatory adjuvant for promoting β-cell-Ag–specific immune modulation and reverse hyperglycemia, at least at early stages.
Supplementary Material
Article Information
Acknowledgments. The authors thank Biorepository & Tissue Analysis Shared Resource, Hollings Cancer Center, MUSC, for the histology service.
Funding. This work was supported by internal funds from MUSC and UIC, National Institutes of Health grant R01-AI-073858, American Diabetes Association grant ADA-1-13-IN-57, and JDRF regular grant JDRF-32-2008-343 to C.V.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. S.K.-M. designed experiments and researched and analyzed data. M.H.S. and N.P. researched data. R.G. researched and analyzed data and reviewed and edited the manuscript. B.M.J. researched data and reviewed and edited the manuscript. C.V. designed experiments; analyzed data; and wrote, reviewed, and edited the manuscript. C.V. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db14-1145/-/DC1.
References
- 1.Roy CR, Mocarski ES. Pathogen subversion of cell-intrinsic innate immunity. Nat Immunol 2007;8:1179–1187 [DOI] [PubMed] [Google Scholar]
- 2.Dunne DW, Cooke A. A worm’s eye view of the immune system: consequences for evolution of human autoimmune disease. Nat Rev Immunol 2005;5:420–426 [DOI] [PubMed] [Google Scholar]
- 3.Rook GA. Hygiene hypothesis and autoimmune diseases. Clin Rev Allergy Immunol 2012;42:5–15 [DOI] [PubMed] [Google Scholar]
- 4.Bach JF. Infections and autoimmune diseases. J Autoimmun 2005;25(Suppl.):74–80 [DOI] [PubMed] [Google Scholar]
- 5.Boettler T, von Herrath M. Protection against or triggering of type 1 diabetes? Different roles for viral infections. Expert Rev Clin Immunol 2011;7:45–53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Karumuthil-Melethil S, Perez N, Li R, Vasu C. Induction of innate immune response through TLR2 and dectin 1 prevents type 1 diabetes. J Immunol 2008;181:8323–8334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dillon S, Agrawal S, Banerjee K, et al. Yeast zymosan, a stimulus for TLR2 and dectin-1, induces regulatory antigen-presenting cells and immunological tolerance. J Clin Invest 2006;116:916–928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Burton OT, Zaccone P, Phillips JM, et al. Roles for TGF-beta and programmed cell death 1 ligand 1 in regulatory T cell expansion and diabetes suppression by zymosan in nonobese diabetic mice. J Immunol 2010;185:2754–2762 [DOI] [PubMed] [Google Scholar]
- 9.Sun X, Zhang M, El-Zataari M, et al. TLR2 mediates Helicobacter pylori-induced tolerogenic immune response in mice. PLoS ONE 2013;8:e74595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Manicassamy S, Ravindran R, Deng J, et al. Toll-like receptor 2-dependent induction of vitamin A-metabolizing enzymes in dendritic cells promotes T regulatory responses and inhibits autoimmunity. Nat Med 2009;15:401–409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li H, Gonnella P, Safavi F, et al. Low dose zymosan ameliorates both chronic and relapsing experimental autoimmune encephalomyelitis. J Neuroimmunol 2013;254:28–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sukhithasri V, Nisha N, Biswas L, Anil Kumar V, Biswas R. Innate immune recognition of microbial cell wall components and microbial strategies to evade such recognitions. Microbiol Res 2013;168:396–406 [DOI] [PubMed] [Google Scholar]
- 13.Kemper C, Köhl J. Novel roles for complement receptors in T cell regulation and beyond. Mol Immunol 2013;56:181–190 [DOI] [PubMed] [Google Scholar]
- 14.Karumuthil-Melethil S, Perez N, Li R, Prabhakar BS, Holterman MJ, Vasu C. Dendritic cell-directed CTLA-4 engagement during pancreatic beta cell antigen presentation delays type 1 diabetes. J Immunol 2010;184:6695–6708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Perez N, Karumuthil-Melethil S, Li R, Prabhakar BS, Holterman MJ, Vasu C. Preferential costimulation by CD80 results in IL-10-dependent TGF-beta1(+) -adaptive regulatory T cell generation. J Immunol 2008;180:6566–6576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sofi MH, Gudi RR, Karumuthil-Melethil S, Perez N, Johnson BM, Vasu C. pH of drinking water influences the composition of gut microbiome and type 1 diabetes incidence. Diabetes 2014;63:632–644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li R, Perez N, Karumuthil-Melethil S, Vasu C. Bone marrow is a preferential homing site for autoreactive T-cells in type 1 diabetes. Diabetes 2007;56:2251–2259 [DOI] [PubMed] [Google Scholar]
- 18.Wang Q, McLoughlin RM, Cobb BA, et al. A bacterial carbohydrate links innate and adaptive responses through Toll-like receptor 2. J Exp Med 2006;203:2853–2863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zanin-Zhorov A, Cahalon L, Tal G, Margalit R, Lider O, Cohen IR. Heat shock protein 60 enhances CD4+ CD25+ regulatory T cell function via innate TLR2 signaling. J Clin Invest 2006;116:2022–2032 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 20.Liu H, Komai-Koma M, Xu D, Liew FY. Toll-like receptor 2 signaling modulates the functions of CD4+ CD25+ regulatory T cells. Proc Natl Acad Sci U S A 2006;103:7048–7053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Round JL, Lee SM, Li J, et al. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 2011;332:974–977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shen Y, Giardino Torchia ML, Lawson GW, Karp CL, Ashwell JD, Mazmanian SK. Outer membrane vesicles of a human commensal mediate immune regulation and disease protection. Cell Host Microbe 2012;12:509–520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Goodridge HS, Underhill DM. Fungal recognition by TLR2 and Dectin-1. Handbook Exp Pharmacol 2008;183:87–109 [DOI] [PubMed] [Google Scholar]
- 24.Underhill DM. Collaboration between the innate immune receptors dectin-1, TLRs, and Nods. Immunol Rev 2007;219:75–87 [DOI] [PubMed] [Google Scholar]
- 25.Dennehy KM, Ferwerda G, Faro-Trindade I, et al. Syk kinase is required for collaborative cytokine production induced through Dectin-1 and Toll-like receptors. Eur J Immunol 2008;38:500–506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ferwerda G, Meyer-Wentrup F, Kullberg BJ, Netea MG, Adema GJ. Dectin-1 synergizes with TLR2 and TLR4 for cytokine production in human primary monocytes and macrophages. Cell Microbiol 2008;10:2058–2066 [DOI] [PubMed] [Google Scholar]
- 27.Marie JC, Letterio JJ, Gavin M, Rudensky AY. TGF-beta1 maintains suppressor function and Foxp3 expression in CD4+CD25+ regulatory T cells. J Exp Med 2005;201:1061–1067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zheng XX, Steele AW, Hancock WW, et al. A noncytolytic IL-10/Fc fusion protein prevents diabetes, blocks autoimmunity, and promotes suppressor phenomena in NOD mice. J Immunol 1997;158:4507–4513 [PubMed] [Google Scholar]
- 29.Huang X, Zhu J, Yang Y. Protection against autoimmunity in nonlymphopenic hosts by CD4+ CD25+ regulatory T cells is antigen-specific and requires IL-10 and TGF-beta. J Immunol 2005;175:4283–4291 [DOI] [PubMed] [Google Scholar]
- 30.Cao W, Manicassamy S, Tang H, et al. Toll-like receptor-mediated induction of type I interferon in plasmacytoid dendritic cells requires the rapamycin-sensitive PI(3)K-mTOR-p70S6K pathway. Nat Immunol 2008;9:1157–1164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nikoopour E, Schwartz JA, Huszarik K, et al. Th17 polarized cells from nonobese diabetic mice following mycobacterial adjuvant immunotherapy delay type 1 diabetes. J Immunol 2010;184:4779–4788 [DOI] [PubMed] [Google Scholar]
- 32.Kriegel MA, Sefik E, Hill JA, Wu HJ, Benoist C, Mathis D. Naturally transmitted segmented filamentous bacteria segregate with diabetes protection in nonobese diabetic mice. Proc Natl Acad Sci U S A 2011;108:11548–11553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mi QS, Ly D, Zucker P, McGarry M, Delovitch TL. Interleukin-4 but not interleukin-10 protects against spontaneous and recurrent type 1 diabetes by activated CD1d-restricted invariant natural killer T-cells. Diabetes 2004;53:1303–1310 [DOI] [PubMed] [Google Scholar]
- 34.Calcinaro F, Gambelunghe G, Lafferty KJ. Protection from autoimmune diabetes by adjuvant therapy in the non-obese diabetic mouse: the role of interleukin-4 and interleukin-10. Immunol Cell Biol 1997;75:467–471 [DOI] [PubMed] [Google Scholar]
- 35.Ploix C, Bergerot I, Fabien N, Perche S, Moulin V, Thivolet C. Protection against autoimmune diabetes with oral insulin is associated with the presence of IL-4 type 2 T-cells in the pancreas and pancreatic lymph nodes. Diabetes 1998;47:39–44 [DOI] [PubMed] [Google Scholar]
- 36.Lin MS, Tse HM, Delmastro MM, et al. A multivalent vaccine for type 1 diabetes skews T cell subsets to Th2 phenotype in NOD mice. Immunol Res 2011;50:213–220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vizler C, Bercovici N, Heurtier A, et al. Relative diabetogenic properties of islet-specific Tc1 and Tc2 cells in immunocompetent hosts. J Immunol 2000;165:6314–6321 [DOI] [PubMed] [Google Scholar]
- 38.Harada M, Kishimoto Y, Makino S. Prevention of overt diabetes and insulitis in NOD mice by a single BCG vaccination. Diabetes Res Clin Pract 1990;8:85–89 [DOI] [PubMed] [Google Scholar]
- 39.Caramalho I, Rodrigues-Duarte L, Perez A, Zelenay S, Penha-Gonçalves C, Demengeot J. Regulatory T cells contribute to diabetes protection in lipopolysaccharide-treated non-obese diabetic mice. Scand J Immunol 2011;74:585–595 [DOI] [PubMed] [Google Scholar]
- 40.Brás A, Aguas AP. Diabetes-prone NOD mice are resistant to Mycobacterium avium and the infection prevents autoimmune disease. Immunology 1996;89:20–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Toyota T, Satoh J, Oya K, Shintani S, Okano T. Streptococcal preparation (OK-432) inhibits development of type I diabetes in NOD mice. Diabetes 1986;35:496–499 [DOI] [PubMed] [Google Scholar]
- 42.Fukushima K, Abiru N, Nagayama Y, et al. Combined insulin B:9-23 self-peptide and polyinosinic-polycytidylic acid accelerate insulitis but inhibit development of diabetes by increasing the proportion of CD4+Foxp3+ regulatory T cells in the islets in non-obese diabetic mice. Biochem Biophys Res Commun 2008;367:719–724 [DOI] [PubMed] [Google Scholar]
- 43.Quintana FJ, Rotem A, Carmi P, Cohen IR. Vaccination with empty plasmid DNA or CpG oligonucleotide inhibits diabetes in nonobese diabetic mice: modulation of spontaneous 60-kDa heat shock protein autoimmunity. J Immunol 2000;165:6148–6155 [DOI] [PubMed] [Google Scholar]
- 44.Montandon R, Korniotis S, Layseca-Espinosa E, et al. Innate pro-B-cell progenitors protect against type 1 diabetes by regulating autoimmune effector T cells. Proc Natl Acad Sci U S A 2013;110:E2199–E2208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wu AJ, Hua H, Munson SH, McDevitt HO. Tumor necrosis factor-alpha regulation of CD4+CD25+ T cell levels in NOD mice. Proc Natl Acad Sci U S A 2002;99:12287–12292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Christen U, Wolfe T, Möhrle U, et al. A dual role for TNF-alpha in type 1 diabetes: islet-specific expression abrogates the ongoing autoimmune process when induced late but not early during pathogenesis. J Immunol 2001;166:7023–7032 [DOI] [PubMed] [Google Scholar]
- 47.Grewal IS, Grewal KD, Wong FS, Picarella DE, Janeway CA Jr, Flavell RA. Local expression of transgene encoded TNF alpha in islets prevents autoimmune diabetes in nonobese diabetic (NOD) mice by preventing the development of auto-reactive islet-specific T cells. J Exp Med 1996;184:1963–1974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Slack EC, Robinson MJ, Hernanz-Falcón P, et al. Syk-dependent ERK activation regulates IL-2 and IL-10 production by DC stimulated with zymosan. Eur J Immunol 2007;37:1600–1612 [DOI] [PubMed] [Google Scholar]
- 49.Frasnelli ME, Tarussio D, Chobaz-Péclat V, Busso N, So A. TLR2 modulates inflammation in zymosan-induced arthritis in mice. Arthritis Res Ther 2005;7:R370–R379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Veldhoen M, Hocking RJ, Flavell RA, Stockinger B. Signals mediated by transforming growth factor-beta initiate autoimmune encephalomyelitis, but chronic inflammation is needed to sustain disease. Nat Immunol 2006;7:1151–1156 [DOI] [PubMed] [Google Scholar]
- 51.Korn T, Mitsdoerffer M, Croxford AL, et al. IL-6 controls Th17 immunity in vivo by inhibiting the conversion of conventional T cells into Foxp3+ regulatory T cells. Proc Natl Acad Sci U S A 2008;105:18460–18465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wu HJ, Ivanov II, Darce J, et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 2010;32:815–827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Noack M, Miossec P. Th17 and regulatory T cell balance in autoimmune and inflammatory diseases. Autoimmun Rev 2014;13:668–677 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.