Abstract
In this chapter we discuss the complex mixing behavior of plasma membrane lipids. To do so, we first introduce the plasma membrane and membrane mixtures often used to model its complexity. We then discuss the nature of lipid phase behavior in bilayers and the distinction between these phases and other manifestations of nonrandom mixing found in one-phase mixtures, such as clusters, micelles, and microemulsions. Finally, we demonstrate the applicability of Gibbs phase diagrams to the study of increasingly complex model membrane systems, with a focus on phase coexistence, morphology and their implications for the cell plasma membrane.
Keywords: lipid bilayer; plasma membrane (PM); model membranes; high-Tm lipid; low-Tm lipid; non-ideal mixing; phase separation; macroscopic domains; nanoscopic domains; cholesterol (chol); phosphatidylethanolamine (PE); phosphatidylserine (PS); phosphatidylcholine (PC); distearoyl-PC (DSPC); dioleoyl-PC (DOPC); palmitoyl,oleoyl-PC (POPC); sphingomyelin (SM)
INTRODUCTION: MODELING THE PLASMA MEMBRANE
The cellular plasma membrane (PM) is a highly complex and heterogeneous environment. It is approximately half lipid and half protein by volume, but only ~15% of the large volume fraction of membrane protein actually lies within the membrane (Figure 1) [1]. The PM bilayer itself is “asymmetric,” meaning its inner and outer leaflets differ in both lipid and protein compositions. At the macroscopic level, a cell’s PM can exhibit large scale lipid and protein compositional variation such as the distinct environments of the basolateral and apical regions of epithelial cells [2]. At the nanoscopic level, membrane proteins and lipids are further confined and sorted on a size scale of tens of nanometers as determined by diffusion [3,4], electron microscopy [5], Fluorescence Resonance Energy Transfer (FRET) [6] and super resolution microscopy [7]. Nanoscale heterogeneities, whatever their basis of formation, are often called “lipid rafts”, a term originally reserved for SM- and cholesterol-rich domains. These rafts seem to serve as sites for a variety of key events in the life of a cell, such as protein sorting, signal transduction, and virus budding [8]. What mechanisms control this rich variety of membrane heterogeneities? Do membrane proteins establish their lipid environment, or do lipid structures and their mixing behaviors control membrane heterogeneity, or is the cytoskeleton involved, or is there some combination of interactions [9]? To address these questions, simple lipid mixtures can be used to model the compositions of the cell PM. Unlike the hundreds of components of a cell PM, three- or four-component model mixtures enable all possible combinations of components to be explored and phase behavior established. Helpful for the researcher, particular lipids can be chosen for these mixtures in order to create the macroscopic phase domains that greatly facilitate solving the phase diagrams. With these model mixtures, we can ascertain which aspects of the observed phases or other heterogeneities arise from lipid components of the mixture, and which from proteins.
Figure 1.
The plasma membrane (maroon) is comprised of ~ 50% protein (yellow) by volume. Approximately 15% of the protein (helix) is actually within the membrane itself, with most in the aqueous space outside or inside the cell.
In this chapter, we focus only on symmetric bilayers with compositions that might be useful models for the outer leaflet of animal cell plasma membranes. We default to this simple symmetric membrane model for two reasons: (1) preparing asymmetric bilayers in vitro is challenging, and only recently have robust preparations of asymmetric systems been achieved [10]; and (2) mixtures that model the outer leaflet give rise to different coexisting lipid phases [11]. These include the liquid disordered (Ld) phase characterized by fast translational diffusion and low acyl chain order, the liquid ordered (Lo) phase with fast diffusion and high chain order, and the solid gel (Lβ) phase having slow diffusion and high chain order. Of biological significance in these outer leaflet models is the coexistence of Lo and Ld phases, which might mimic raft behavior in the PM. In contrast, the PM inner leaflet, modeled as mixtures of PE + PS + cholesterol, has not been shown to separate into Ld and Lo phases, or any other type of phase coexistence: optical microscopy reveals no micron-scale domains in these mixtures and no “nanodomains” are detected by methods such as FRET or Electron Spin Resonance (ESR) that are sensitive to the nanometer size scale [12]. In fact, whereas the outer leaflet lipid components spontaneously form bilayers, the polyunsaturated PEs abundant in the inner leaflet of animal cell plasma membranes tend to form hexagonal or cubic phases. It is therefore worth noting how remarkable it is that plasma membranes are bilayers, and that perhaps the asymmetric compositions are crucial for this structure.
To help understand the importance of lipid mixtures and phases as they pertain to the PM, we now provide a brief introduction to lipid bilayers and the distinction between phase separation and nonrandom mixing in one-phase lipid mixtures.
THE LIPID BILAYER AS A STATE OF MATTER
For many scientists, the lipid bilayer is at the same time both familiar in overall structure, yet also strange as a state of matter: it is a two-dimensional physical mixture of lipids and proteins. The lipid mixture is both the milieu of membrane proteins and also the substrate of membrane-bound and other enzymes. Unlike water that provides the rather uniform solvent for the majority of a cell’s proteins, for the one third of the cell’s proteins that reside in membranes, the lipid bilayer can provide several distinctly different states of density and viscosity, as well as an interface between distinct states. Given these complexities, how should we frame our most pressing questions about cell membranes? We start with some fundamentals.
Lipids comprise one of the four main categories of biomolecules, along with proteins, nucleotide polymers RNA and DNA, and carbohydrates. But unlike these polymers, biological lipids exist as physical mixtures. Thus the thermodynamics of mixing, and in turn composition, have much to say about lipid behaviors.
When the compositions of the various membranes of a eukaryotic cell are compared, the PM stands out [13]. First of all, its content of cholesterol can be ten-fold higher than that of any other cell membrane, except for the trans-Golgi, which is close. Second, although the PM is predominantly or entirely liquid, it contains a remarkably high fraction of lipids that are solid in their pure, hydrated state at physiological temperatures, namely SM and gangliosides. As we show below, this notable content of both high melting temperature (“high-Tm”) lipids and cholesterol has a dramatic influence on the membrane properties.
THE NATURE OF SMALL CLUSTERS AND PHASE SEPARATION
Mixtures of molecules — any unlike molecules — are never randomly mixed, but instead show compositionally distinct clusters, with either like neighbors being favored, or in the case of cholesterol, unlike neighbors being favored [14]. Whereas entropy works toward random mixing, interaction energies between different neighboring molecules work in the opposite way, favoring nonrandom distributions of molecules. When differences of interaction energies are large enough to overcome entropic mixing, distinct phases can coexist. Distinct phases are defined to be stable domains of molecules, having phase properties such as chain order parameter and density that are uniform throughout the phase domain.
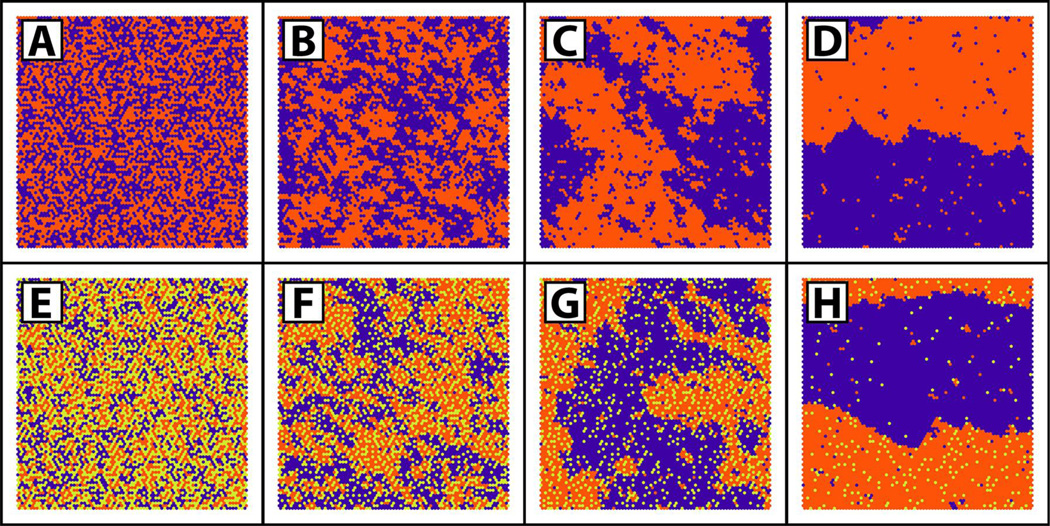
A simple case that illustrates these principles is that of a binary lipid mixture (Figure 2 A–D) [15]. For a given composition, only one parameter is needed to model the nonrandom mixing [16]: ΔEm, defined here to be ΔEm = Eab − (Eaa + Ebb)/2, which is the energetic cost of unlike molecular neighbors, with “a” and “b” referring to two different molecules, e.g. two different phospholipids. For ΔEm = 0 kT, mixing is random. Even so, numerous “clusters” of 3–5 like-molecules occur [17]. When ΔEm = +0.4 kT, like-molecule neighbors are more energetically favorable and numerous clusters of 10–20 like-molecules are found. This mixture is still a single phase, and these clusters do not have separate phase properties, but instead merely reflect the slight preference of like-molecules to be neighbors. Based strictly on composition, one might be tempted to term these clusters “domains”, but this is generally not done, because “domains” implies distinct properties, which is not the case: ΔEm is not large enough to overcome mixing entropy, and these clusters do not show cooperative behaviors. However, when ΔEm > +0.55 kT, interaction energies do dominate and distinct phase separation occurs [16], with lipids in a given phase having the same order and cooperative behavior.
Figure 2.
Monte Carlo simulations provide pictures of molecule distributions in mixtures of three components representing e.g. a low-Tm lipid (purple), a high-TM lipid (orange) and cholesterol (yellow-green) [15]. Two-component (A–D) mixtures containing equal amounts of the low-Tm lipid and cholesterol show random mixing at ΔEm = 0 kT (A), non-ideal mixing at ΔEm = +0.4 kT (B), large-scale clusters at ΔEm = +0.55 kT (C), and phase separation at ΔEm = 0.7 kT (D). Three-component mixtures (E–H) containing equal amounts of the low-Tm and high-Tm lipids show increasing levels of clustering and eventual phase separation as cholesterol content is decreased from 45% (E), to 30% (F), to 20% (G), to 5% (H). For (E–H), the ΔEm for interactions between low-Tm – high-Tm, low-Tm – chol, and high-Tm – chol are respectively +0.8 kT, −0.8 kT, and −1.2 kT.
For a three-component mixture representing e.g. a lipid with a high temperature solid-to-liquid transition (high-Tm), a low-Tm lipid and cholesterol (Figure 2 E–H), more interaction energies come into play and more complex phase behavior is observed [15]. In addition to interaction energies, phase behavior in three-component mixtures is also highly dependent on composition. For instance, traveling from composition E to H in Figure 2 shows that simply decreasing the cholesterol concentration in a three-component mixture where cholesterol preferentially favors the high-Tm lipid can drive a system from one-phase non-random mixing into the two-phase region.
Experimentally, the distinction between mixing and phase separation is clear from fast methods that provide a snapshot of all individual compositional environments rather than an average. ESR, with a timescale of ~ 10−9 sec [18], shows that an ESR-labeled lipid experiences two distinct environments within a region of 2-phase coexistence, with each phase having characteristic phase properties, including order parameter. But with a small change in composition into a single phase region, even within an extremely nonrandom, highly clustered single phase, the clusters have the same order parameter as the surroundings, i.e. there is no separate ESR signal from lipid clusters [19,20].
DISTINCT DOMAINS THAT ARE NOT SEPARATE PHASES
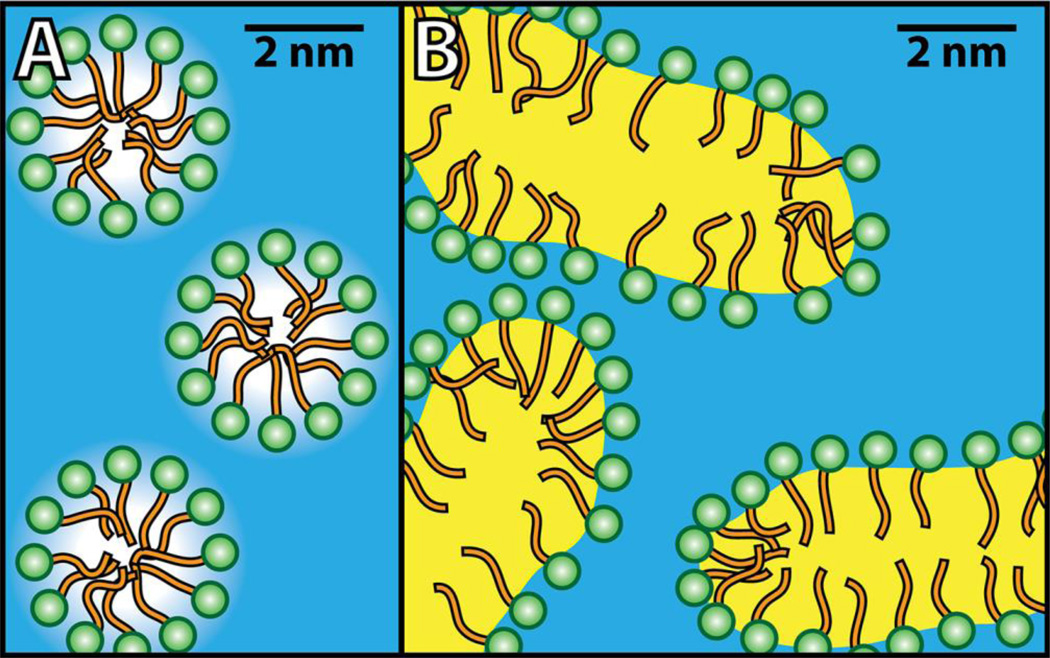
Nonrandom mixing within a single phase is not always as simple as described above. For example, when detergent micelles form in water, the size of the clusters is limited (Figure 3A). Rather than being a separate phase, micelles are better described as a high-order aggregation of amphiphiles characterized by an aggregation number and a well-defined concentration beyond which the micelles form [21]. A solution with micelles is a “structured one-phase mixture”, with micelles behaving as a chemical and physical environment that is distinct from the surrounding medium, but not a separate phase. For example, fluorescent or ESR-labeled probes provide distinctly different signals from within the micelle compared to the surroundings. Nonetheless, aqueous micelles behave as a single phase [22]. Although no convincing data show the 2D equivalent in bilayers of the familiar 3D micelle formation, this does not preclude 2D micelles within a bilayer.
Figure 3.
Schematic representations of one-phase systems with micelles (A) and microemulsions (B). In (A), amphiphilic molecules coalesce into small spherical clusters on the order of ~ 5 nm called micelles, with their hydrophilic moeity (green) exposed to water (blue) and their hydrophobic moeity (orange) largely shielded from water. Similar to micelles are microemulsions (B), where water-insoluble molecules generically termed “oil” (yellow) are surrounded by amphiphiles forming large continuous structures in water.
Related to micelles, but less familiar, is the case of the structured one-phase microemulsion (Figure 3B) [21]. In some cases, these are related to ordinary micelles, with the micelle interior swollen with “oil” (water-insoluble components), or the reverse, or with no discontinuous environment (bicontinuous). The size scale of its smaller dimension is 3 – 50 nm.
MODELING BILAYER PHASE BEHAVIOR
The phase separation of simple lipid membrane mixtures is a candidate mechanism by which a cell could influence raft formation, since at whatever temperature or other external conditions it exists, a cell is in control of the lipid composition of its membranes. For a cell membrane where composition can vary, it would be illuminating to understand the effects of composition on the types and amounts of coexisting phases. A great difficulty here is that cell membranes have hundreds of different lipid components, making impossible the complete description of the mixture in terms of each component as a variable. But multiple-component phase diagrams are of such great practical use that this problem was solved many years ago — by geologists [23]. Their solution was to group similar components, with sometimes many different minerals represented using a single “pseudocomponent”.
In the case of the hundreds of plasma membrane lipids, grouping lipids into just three categories of pseudocomponents enables meaningful comparisons of lipid bilayer mixtures by use of Gibbs Triangle phase diagrams. A particularly useful plasma membrane outer leaflet model has at least three lipid components: one high-Tm lipid, one low Tm-lipid, and cholesterol [11]. These three-component mixtures have rich phase behavior, and several such three-component phase diagrams have been solved. A notable finding is that many different mixtures of high-Tm lipid/low-Tm lipid/chol, whatever the particulars of the lipids examined, have the same type of phase diagram.
PHASE DIAGRAMS APPLICABLE TO CELL PLASMA MEMBRANES
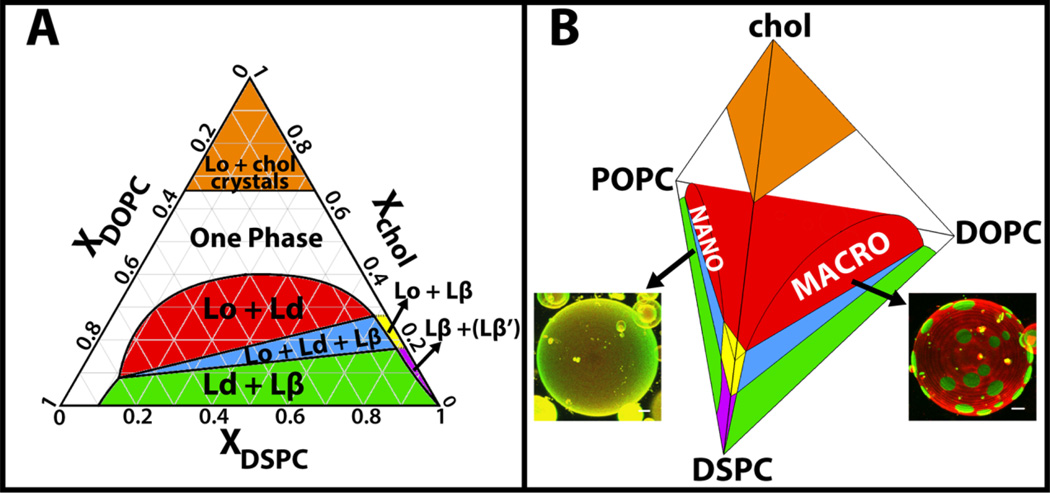
A typical case of such a solved mixture is DSPC/DOPC/chol (Figure 4A) [19,24]. Here is what we learn from this phase diagram:
Above Χchol ~ 0.4 mole fraction (and below Χchol ~ 0.67 mole fraction where cholesterol monohydrate crystals precipitate) a single phase exists. Highly nonrandom mixing occurs in this single phase, i.e. extensive clustering of some components. Such clusters within this single phase, while compositionally distinct, do not behave like small regions of different properties. Rather, clusters within bilayer single phases are the hallmark of nonrandom mixing, and have the phase properties of that phase. This one phase region is neither Ld nor Lo, but instead changes continuously from one to the other as composition is varied.
The Lo phase precipitates from the solid Lβ phase when Χchol > 0.16. Huang has shown that this special concentration corresponds to each cholesterol molecule in the solid phase being surrounded by a single layer of ~ 6 nearest-neighbor lipids that are shielding the largely hydrophobic cholesterol from unfavorable contact with bulk water [14]. But a higher cholesterol concentration than 0.16 cannot be accommodated in the Lβ phase, so with more cholesterol, the Lo phase separates, having the higher Χchol of ~ 0.25. This phase is not limited to this particular cholesterol concentration, and accommodates up to Χchol = 0.67.
The 2-phase region is noteworthy in modeling the Ld + Lo coexistence that could occur in cell plasma membranes. Bilayers prepared within this wide compositional region can have a range of different Lo compositions, for example from DSPC/DOPC/chol = 0.3/0.3/0.4 to 0.7/0.02/0.28. Similarly, the Ld phase within this same region can range from DSPC/DOPC/chol = 0.1/0.8/0.1 to 0.2/0.4/0.4. In general, the Lo phase is richer in the high-Tm lipid and cholesterol, whereas the Ld phase is rich in the low-Tm lipid. The higher the Xchol within this region, the closer are the Ld and Lo phases in both composition and also phase properties such as density and order.
Notice the region of three-phase coexistence. It would be hard to argue from such a phase diagram that cells could never have the solid Lβ phase in their plasma membrane. Might there be a membrane protein that would prefer such an environment, or perhaps the interface of Lβ/Lo or Lβ/Ld?
Figure 4.
Three- and four-component phase diagrams reveal behaviors of lipid mixtures [24]. The three-component phase diagram of DSPC/DOPC/chol is shown in (A) with regions of phase coexistence colored and labeled. Other three-component bilayer mixtures containing a high-Tm lipid, a low-Tm lipid and cholesterol have similar phase behavior, but their Lo + Ld coexistence region can be either macroscopic (large patches under optical microscopy, shown for a vesicle of DSPC/DOPC/chol [25]) or nanoscopic (uniform under optical microscopy, shown for a vesicle of DSPC/POPC/chol [26]) as shown in (B); (B) Four-component mixture of DSPC/DOPC/POPC/chol, colored as in (A). A size transition occurs from nanoscopic Lo + Ld coexistence at high POPC concentrations to macroscopic at high DOPC concentrations. Scale bars for GUV images, 5 microns.
By mapping out composition-dependent phase regions, three-component phase diagrams enable simple but quantitative thinking about complex mixtures. These phase diagrams imply that plasma membranes in cells can have a wide range of phase properties, and in turn, a wide range of differences in properties of coexisting phases. For example, protein partitioning between Ld and Lo domains might vary considerably depending upon details of composition, especially fraction of cholesterol. If we seek to find the rules that describe how proteins select their location to be Ld over Lo or the reverse, having the phase diagram in front of us makes clear which compositions should be examined in order to explore the possible range of partitioning behaviors in cell plasma membranes.
Useful as they are for describing the phase behaviors over all possible compositions, three-component phase diagrams do not capture the full range of interesting phenomena. This deficiency is revealed by comparing images of DSPC/DOPC/chol vesicles [25] with those of DSPC/POPC/chol [26] (Figure 4B), or SM/DOPC/chol vesicles with those of SM/POPC/chol. Whereas the phase diagrams have similar boundaries and the same phase coexistence regions, the liquid-liquid coexistence region is macroscopic in the DOPC mixtures, and nanoscopic in the POPC mixtures. Given the abundance of POPC in animal cell plasma membranes and the near absence of DOPC (http://lipidlibrary.aocs.org/), mixtures that mimic the outer leaflet of the plasma membrane are better described by the POPC-containing mixtures than by the DOPC-containing ones. Indeed, the size scale of the POPC system is more comparable to the size scale of rafts detected in cell plasma membranes. This naturally leads to the question: what governs the transition between macroscopic and nanoscopic phase separation in model membranes, and can a cell exert such control over domain size?
FOUR COMPONENT MODEL MEMBRANES
Understanding the striking difference between macroscopic and nanoscopic phase domains is informed by the four-component phase diagram determined for DSPC/DOPC/POPC/chol (Figure 4B) [24]. The phase boundaries shift smoothly as POPC is replaced by DOPC, giving no hint that domain size changes from nanometers in POPC-rich mixtures, to micron-size as DOPC fraction is increased. This nano ↔ macro transition is not a phase transition! Instead, it is a morphology transition: domain size is changing, not the nature of the coexisting phases. These equilibrium observations confirm that lipid bilayers can have phase organization on the size scale of tens of nanometers. In fact, the sizes of these nanodomains have been directly measured by SANS [27]. Nanodomain dimensions are in the range of 10 – 20 nm. Apparently, a higher interfacial energy penalty — line tension — occurs for Lo and Ld coexistence when the Ld phase is rich in DOPC compared to POPC, leading to less boundary and larger domains [27]. Though it has not yet been experimentally confirmed, models show that the domain size transition from nanoscopic to macroscopic can occur at a well-defined value of line-tension; domains are nanoscopic below this value and macroscopic above it [28]. It is possible that similar line-tension driven morphology switches are important in the behavior of a cell’s PM.
CONCLUSION
The animal cell plasma membrane contains hundreds of different lipid components, far more than are feasible to study systematically. But membrane properties are of such importance to cellular functions that useful models and techniques have been developed to study mixtures of fewer components. Simplifying the plasma membrane complexity can be done by selecting representative high-Tm lipids and low-Tm lipids along with cholesterol, which act as stand-ins for the many lipids of the cell membrane. Complex mixing behavior is observed with just three or four of these lipid types. In three- and four-component mixtures containing a high-Tm lipid, cholesterol and one or two low-Tm lipids, phases with different compositions and properties can coexist. In these mixtures, a small change in cholesterol content can switch a membrane between two coexisting phases or just one phase, or between two and three coexisting phases. Besides cholesterol, altering the relative amounts of the other lipids can affect domain size and physical properties, and the relative amounts of any coexisting domains. Even in mixtures where no phase separation occurs, a highly-clustered single phase can exist. Could these non-trivial mixing behaviors of lipids in model membranes explain the heterogeneities detected in the cell plasma membrane? In order to more closely reflect the plasma membrane phase behaviors, future model membrane work should focus on the influence of protein components and the presence of asymmetry of the bilayer leaflets on phase behavior.
SUMMARY.
Membrane mixtures of three or four representative components are used to provide good models for the complex cell plasma membrane
Phase diagrams help guide studies involving model membranes by showing how phase behavior depends on composition
Lipid mixtures can exhibit one-phase non-ideal mixing, as well as two- or three-phase coexistence
Three-component lipid mixtures exhibit complex phase behavior, and four-component mixtures can show transitions in domain size from nanoscopic to macroscopic in the two phase region
Phase coexistence in chemically simple model mixtures might reflect the heterogeneities detected in cell plasma membranes
ACKNOWLEDGEMENTS
This work was supported in part by funding from the U.S. National Science Foundation (MCB-0842839) and the U.S. National Institutes of Health (GM105684) to GWF. DGA was supported by the U.S. National Science Foundation Graduate Research Fellowship (DGE-114153). We are grateful for help with figures from F.A. Heberle (Figure 2), and T. Konyakhina, J. Zhao, and J. Wu (Figure 4).
Biographies
David Ackerman received his B.A. in Physics, Math and Integrated Sciences from Northwestern University in 2010, where he studied exoplanets under Fred Rasio. He is currently a Ph.D. candidate in the field of Biophysics at Cornell University, where he works in Jerry Feigenson’s membrane physical chemistry lab. His thesis work focuses on using both coarse-grained and atomistic molecular dynamics to model phase separation in multiple-component membrane mixtures.
Jerry Feigenson is a Professor in the Cornell Department of Molecular and Cell Biology and Director of Graduate Studies for the Cornell Field of Biophysics. His 1968 B.S. in Chemistry is from the Rensselaer Polytechnic Institute, where he studied surface chemistry in the laboratory of Sydney Ross; his 1974 Ph.D. in Chemistry is from the California Institute of Technology, where he studied NMR with Sunney Chan. He joined the Cornell faculty in 1974, after brief postdoctoral study at Oxford University in the fluorescence laboratory of George Radda. He has been a Visiting Professor of Physics at Keio University in Japan with Kazuhiko Kinosita, and a Visiting Scholar at Scripps Institution of Oceanography with Vic Vacquier.
References
- 1.Sheetz MP. Glycoprotein motility and dynamic domains in fluid plasma membranes. Annual Review of Biophysics and Biomolecular Structure. 1993;22:417–431. doi: 10.1146/annurev.bb.22.060193.002221. [DOI] [PubMed] [Google Scholar]
- 2.van Meer G, Simons K. Viruses budding from either the apical or the basolateral plasma membrane domain of MDCK cells have unique phospholipid compositions. The EMBO Journal. 1982;1:847–852. doi: 10.1002/j.1460-2075.1982.tb01258.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dietrich C, Yang B, Fujiwara T, Kusumi A, Jacobson K. Relationship of lipid rafts to transient confinement zones detected by single particle tracking. Biophysical Journal. 2002;82:274–284. doi: 10.1016/S0006-3495(02)75393-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Meder D, Moreno MJ, Verkade P, Vaz WLC, Simons K. Phase coexistence and connectivity in the apical membrane of polarized epithelial cells. Proceedings of the National Academy of Sciences USA. 2006;103:329–334. doi: 10.1073/pnas.0509885103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Plowman SJ, Muncke C, Parton RG, Hancock JF. H-ras, K-ras, and inner plasma membrane raft proteins operate in nanoclusters with differential dependence on the actin cytoskeleton. Proceedings of the National Academy of Sciences USA. 2005;102:15500–15505. doi: 10.1073/pnas.0504114102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goswami D, Gowrishankar K, Bilgrami S, Ghosh S, Raghupathy R, Chadda R, Vishwakarma R, Rao M, Mayor S. Nanoclusters of GPI-anchored proteins are formed by cortical actin-driven activity. Cell. 2008;135:1085–1097. doi: 10.1016/j.cell.2008.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eggeling C, Ringemann C, Medda R, Schwarzmann G, Sandhoff K, Polyakova S, Belov VN, Hein B, von Middendorff C, Schönle A, Hell SW. Direct observation of the nanoscale dynamics of membrane lipids in a living cell. Nature. 2009;457:1159–1162. doi: 10.1038/nature07596. [DOI] [PubMed] [Google Scholar]
- 8.Rajendran L, Simons K. Lipid rafts and membrane dynamics. Journal of Cell Science. 2005;118:1099–1102. doi: 10.1242/jcs.01681. [DOI] [PubMed] [Google Scholar]
- 9.Dinic J, Ashrafzadeh P, Parmryd I. Actin filaments attachment at the plasma membrane in live cells cause the formation of ordered lipid domains. Biochimica et Biophysica Acta. 2013;1828:1102–1111. doi: 10.1016/j.bbamem.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 10.Son M, London E. The dependence of lipid asymmetry upon phosphatidylcholine acyl chain structure. Journal of Lipid Research. 2013;54:223–231. doi: 10.1194/jlr.M032722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Feigenson GW. Phase boundaries and biological membranes. Annual Review of Biophysics and Biomolecular Structure. 2007;36:63–77. doi: 10.1146/annurev.biophys.36.040306.132721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang TY, Silvius JR. Cholesterol does not induce segregation of liquid-ordered domains in bilayers modeling the inner leaflet of the plasma membrane. Biophysical Journal. 2001;81:2762–2773. doi: 10.1016/S0006-3495(01)75919-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van Meer G, Voelker DR, Feigenson GW. Membrane lipids: where they are and how they behave. Nature Reviews Molecular Cell Biology. 2008;9:112–124. doi: 10.1038/nrm2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang J, Feigenson GW. A microscopic interaction model of maximum solubility of cholesterol in lipid bilayers. Biophysical Journal. 1999;76:2142–2157. doi: 10.1016/S0006-3495(99)77369-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heberle FA, Feigenson GW. Phase separation in lipid membranes. Cold Spring Harbor Perspectives in Biology. 2011;3:1–14. doi: 10.1101/cshperspect.a004630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guggenheim EA. Mixtures: the theory of the equilibrium properties of some simple classes of mixtures, solutions and alloys. Oxford, United Kingdom: Clarendon Press; 1952. [Google Scholar]
- 17.Huang J, Feigenson GW. Monte Carlo simulation of lipid mixtures: finding phase separation. Biophysical Journal. 1993;65:1788–1794. doi: 10.1016/S0006-3495(93)81234-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sezer D, Freed JH, Roux B. Simulating electron spin resonance spectra of nitroxide spin labels from molecular dynamics and stochastic trajectories. The Journal of Chemical Physics. 2008;128:165106. doi: 10.1063/1.2908075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Heberle FA, Wu J, Goh SL, Petruzielo RS, Feigenson GW. Comparison of three ternary lipid bilayer mixtures: FRET and ESR reveal nanodomains. Biophysical Journal. 2010;99:3309–3318. doi: 10.1016/j.bpj.2010.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Petruzielo RS. Compositional heterogeneity in biologically relevant membrane models [dissertation] Ithaca: Cornell Unviersity; 2013. p. 171. [Google Scholar]
- 21.Wennerström H. Micelles. Physical chemistry of surfactant association. Physics Reports. 1979;52:1–86. [Google Scholar]
- 22.Langevin D. Micelles and microemulsions. Annual Review of Physical Chemistry. 1992;43:341–369. [Google Scholar]
- 23.Morse SA. Basalts and Phase Diagrams: An Introduction to the Quantitative Use of Phase Diagrams in Igneous Petrology. New York: Springer-Verlag; 1980. [Google Scholar]
- 24.Konyakhina TM, Wu J, Mastroianni JD, Heberle FA, Feigenson GW. Phase diagram of a 4-component lipid mixture: DSPC/DOPC/POPC/chol. Biochimica et Biophysica Acta. 2013;1828:2204–2214. doi: 10.1016/j.bbamem.2013.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhao J, Wu J, Heberle FA, Mills TT, Klawitter P, Huang G, Costanza G, Feigenson GW. Phase studies of model biomembranes: complex behavior of DSPC/DOPC/cholesterol. Biochimica et Biophysica Acta. 2007;1768:2764–2776. doi: 10.1016/j.bbamem.2007.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhao J, Wu J, Shao H, Kong F, Jain N, Hunt G, Feigenson GW. Phase studies of model biomembranes: macroscopic coexistence of Lα+Lβ, with light-induced coexistence of Lα+Lo Phases. Biochimica et Biophysica Acta. 2007;1768:2777–2786. doi: 10.1016/j.bbamem.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Heberle FA, Petruzielo RS, Pan J, Drazba P, Kučerka N, Standaert RF, Feigenson GW, Katsaras J. Bilayer thickness mismatch controls domain size in model membranes. Journal of the American Chemical Society. 2013;135:6853–6859. doi: 10.1021/ja3113615. [DOI] [PubMed] [Google Scholar]
- 28.Amazon J, Goh SL, Feigenson GW. Competition between line tension and curvature stabilizes modulated phase patterns on the surface of giant unilamellar vesicles: a simulation study. Physical Review E. 2013;87:022708. doi: 10.1103/PhysRevE.87.022708. [DOI] [PubMed] [Google Scholar]