Abstract
Background
The effectiveness of ultraviolet (UV) radiation, visible light, or infrared light therapy for the treatment of acne is the subject of ongoing scientific debate.
Objective
This study was conducted to investigate changes in sebum production and the expression of inflammatory cytokines, matrix metalloproteinases (MMPs), and antimicrobial peptides (AMPs), following exposure of cultured human sebocytes to UVA radiation and light at wavelengths of 650 nm and 830 nm.
Methods
Reverse transcription polymerase chain reaction assays were performed to measure the gene expression levels of inflammatory cytokines (interleukin [IL]-1β, IL-6, IL-8, and tumor necrosis factor-α), MMPs (MMP-1, MMP-3, and MMP-9), and AMPs (psoriasin, hBD-2, hBD-3, and LL-37) in cultured sebocytes after exposure to UVA radiation (2 J/cm2, 3 J/cm2, and 5 J/cm2) and light at wavelengths of 650 nm (14 J/cm2, 29 J/cm2, and 87 J/cm2) and 830 nm (5 J/cm2, 10 J/cm2, and 30 J/cm2). Expression of inflammatory cytokine proteins and sebum production were measured using enzyme-linked immunoassays and a lipid analysis kit, respectively.
Results
Exposure of cultured sebocytes to UVA radiation and light at wavelengths of 650 nm and 830 nm did not show a significant increase in the expression of inflammatory cytokines, MMPs, or AMPs. Sebum production was not significantly decreased after exposure to UVA radiation and light at both wavelengths.
Conclusion
We propose that UVA radiation, visible light, and infrared light can be used to target Propionibacterium acnes for the treatment of acne, without an increase in the expression of inflammatory biomarkers and sebum production.
Keywords: Inflammatory cytokines, Infrared light, Sebocytes, Ultraviolet A, Visible light
INTRODUCTION
Acne vulgaris is a common skin disorder involving the sebaceous glands that are associated with hair follicles. It has been debated whether treatment with ultraviolet (UV) radiation, visible light or infrared light worsens, improves, or has no effect on acne vulgaris. UV radiation has been found to enhance the comedogenicity of sebum1. Exposure to UV radiation, primarily UVA, can cause acneiform eruptions, which consist of multiple uniform red papular lesions. UVB, UVA, blue, blue/red, infrared light therapy, or photodynamic therapy with topical aminolevulinic acid for acne may provide some therapeutic benefits2,3,4,5,6,7,8. However, a systematic review found little evidence for or against the hypothesis that exposure to natural sunlight improves acne9. Overall, definitive evidence for the beneficial effects of sunlight exposure on acne is lacking.
Abnormal keratinization of keratinocytes, proliferation of Propionibacterium acnes, and perturbation of sebaceous gland function are all known to play important roles in the pathophysiology of acne10. We conducted this study to evaluate the effects of exposure to UVA radiation, visible light, and infrared light on cultured human sebocytes. The expression of inflammatory cytokines, matrix metalloproteinases (MMPs), and antimicrobial peptides (AMPs) was measured after exposure of cultured human sebocytes to UVA radiation and light at wavelengths of 650 nm (visible) and 830 nm (infrared). In addition, sebum production was measured in cultured human sebocytes after exposure to UVA radiation and light at wavelengths of 650 nm and 830 nm.
MATERIALS AND METHODS
Culture of human sebocytes
Primary cultures of sebocytes were performed according to the method described previously11. They were established from occipital hair follicle sebaceous glands. Sebaceous glands were dissected from hair follicles under a binocular microscope and transferred to tissue culture dishes. Sebaceous gland explants were maintained in Dulbecco's modified Eagle's medium (DMEM; HyClone Laboratories, Logan, UT, USA), supplemented with penicillin (100 U/ml), streptomycin (100 µg/ml), and 20% heat-inactivated fetal calf serum (HyClone Laboratories, Logan, UT, USA), at 37℃ in a humidified atmosphere containing 5% CO2. Explants were maintained for an initial period of 3 days in DMEM; the medium was then changed to EpiLife (MEPI500CA; Gibco BRL, Grand Island, NY, USA), supplemented with penicillin (100 U/ml), streptomycin (100 µg/ml), and fungizone (250 µg/ml). Thereafter, the medium was changed every 3 days. When the cultures became subconfluent, cells were harvested using 0.25% trypsin and 10 mM ethylenediaminetetraacetic acid in Hank's balanced salt solution and subcultured using a split ratio of 1 : 3. Only cells obtained after the second passage were used for the experiments in this study. Sebocytes in these cell populations were identified by staining with hematoxylin and eosin (Muto Pure Chemicals Co., Tokyo, Japan), Oil red O, and Nile red (Sigma, St. Louis, MO, USA), and by immunofluorescence detection of cytokeratin 1 and 7 (Chemicon, Billerica, MA, USA).
Treatment of cultured sebocytes with UVA radiation and light at wavelengths of 650 nm and 830 nm
Cultured human sebocytes were treated with increasing doses of UVA (2 J/cm2, 3 J/cm2, and 5 J/cm2) by using a UV phototherapy unit (Choongwae Machinery Co., Seoul, Korea), and with light at wavelengths of 650 nm (14 J/cm2, 29 J/cm2, and 87 J/cm2) and 830 nm (5 J/cm2, 10 J/cm2, and 30 J/cm2) by using portable light source (AinA, Daegu, Korea), before being harvested for assays evaluating the expression of genes and proteins and the production of sebum.
Reverse transcription polymerase chain reaction
Polymerase chain reaction (PCR) amplifications were performed in triplicate by using the First Strand cDNA Synthesis kit (Promega, Madison, WI, USA) and oligonucleotide primers (Genotech, Daejeon, Korea) for interleukin (IL)-1β (5'-GGG CCT CAA GGA AAA GAA TC-3', 5'-TTC TGC TTG AGA GGT GCT GA-3'), IL-6 (5'-TAC CCC CAG GAG AAG ATT CC-3', 5'-GAG GTG CCC ATG CTA CAT TT-3'), IL-8 (5'-AGA TAT TGC ACG GGA GAA-3', 5'-GAA ATA AAG GAG AAA CCA-3'), tumor necrosis factor (TNF)-α (5'-TCC TTC AGA CAC CCT CAA CC-3', 5'-GGC TAC ATG GGA ACA GCC TA-3'), MMP-1 (5'-CTG AAG GTG ATG AAG CAG CC-3', 5'-AGT CCA AGA GAA TGG CCG AG-3'), MMP-3 (5'-CTC ACA GAC CTG ACT CGG TT-3', 5'-CAC GCC TGA AGG AAG AGA TG-3'), MMP-9 (5'-CAC TGT CCA CCC CTC AGA GC-3', 5'-GCC ACT TGT CGG CGA TAA GG-3'), psoriasin (5'-TGC TGA CGA TGA TGA AGG AG-3', 5'-ATG TCT CCC AGC AAG GAC AG-3'), human β-defensin (hBD)-2 (5'-ATC AGC CAT GAG GGT CTT GT-3', 5'-GAG ACC ACA GGT GCC AAT TT-3'), hBD-3 (5'-AGC CTA GCA GCT ATG AGG ATC-3', 5'-CTT CGG CAG CAT TTT CGGCCA-3') and LL-37 (5'-GGG TAG GGC ACA CAC TAG GA-3', 5'-GGA CAG TGA CCC TCA ACC AG-3'). Image J (NIH Image, Bethesda, MD, USA) was used for quantitative analysis of band intensity in gel images. Target gene messenger RNA (mRNA) levels were normalized to that of β-actin and presented as relative ratios. The Student's t-test was used for the statistical analysis of all data. A p-value of <0.05 was considered statistically significant.
1) Extraction of total RNA
A modified acid phenol method using the TRIzol reagent (Invitrogen, Grand Island, NY, USA) was used to isolate total cellular RNA.
2) Isolation of mRNA
The Dynabeads mRNA purification kit (Dynal, Carlsbad, CA, USA) was used to isolate mRNA from total cellular RNA.
3) Reverse transcription-PCR
Synthesis of cDNA from 5 µg total RNA was performed using a cDNA synthesis kit containing ImProm-IITM reverse transcriptase and random primers (Promega). GoTaq Flexi DNA Polymerase was used for all PCR assays. The following amplification program was used, with slight modifications for specific genes as indicated: 35 cycles of denaturation at 95℃ for 1 min, followed by annealing at 56℃ for 1 min, and extension at 72℃ for 1 min. Modified conditions used for specific genes were as follows: IL-1β, 25 cycles, annealing at 56℃; psoriasin, 30 cycles, annealing at 59℃; hBD-2, 28 cycles, annealing at 57℃; hBD-3, 28 cycles, annealing at 61℃; and LL-37, 28 cycles, annealing at 61℃.
Enzyme-linked immunosorbent assay
Production of IL-1β, IL-6, IL-8, and TNF-α by cultured sebocytes was assayed using an Enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Shanghai, China), according to the manufacturer's instructions. Samples were tested in duplicate. Briefly, 50 µl of sample was mixed with 50 µl of biotinylated antibody reagent, 100 µl of prepared streptavidin-horseradish peroxidase, and 100 µl of premixed tetramethylbenzidine substrate solution, in that order. Samples were incubated in the dark at room temperature for 30 min, after which reactions were stopped by adding 100 µl of stop solution per sample. Absorbance was measured using a VersaMax Microplate Reader (Molecular Devices, Sunnyvale, CA, USA).
Lipid assay
Cultured sebocytes were exposed to UVA radiation and light at wavelengths of 650 nm and 830 nm and were seeded in 100-mm dishes. After 5 days, when cultures reached 70% confluence, the culture medium was removed and phosphate-buffered saline (PBS) was added. Cells were resuspended in PBS and collected by centrifugation (100 g, 5 min). The supernatant was removed, and lipid extraction buffer (0.9% NaCl and 1% Triton X-100) was added to the cell pellet. After homogenization by vortexing, the mixture was centrifuged (15,700 g, 15 min), and the resulting supernatant was retained for lipid analysis. Lipid levels were measured in duplicate, using an enzymatic assay kit (ASAN Co., Seoul, Korea). Lipid measurements were normalized to sample protein levels (as determined using the Bradford method).
RESULTS
Treatment of cultured sebocytes with UVA radiation did not cause a significant increase in the expression of inflammatory cytokines, MMPs, or AMPs
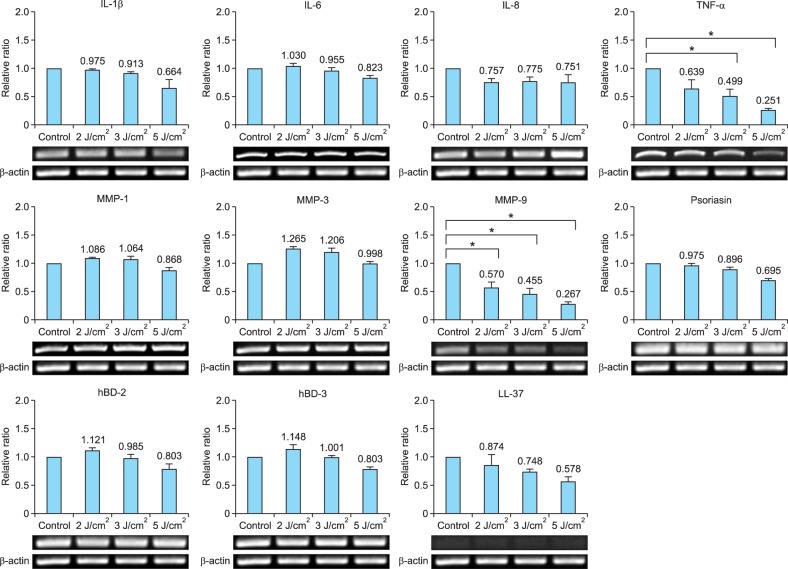
The gene and protein expression of IL-1β, IL-6, and IL-8 in cultured sebocytes treated with UVA radiation (2 J/cm2, 3 J/cm2, and 5 J/cm2) did not show a significant increase compared to that observed in the control (Fig. 1, 2). However, TNF-α gene expression levels in sebocytes treated with 3 J/cm2 and 5 J/cm2 of UVA showed a significant decrease compared to that in the control (p<0.05) (Fig. 1). This was not reflected in the levels of TNF-α protein expression, which did not show a significant decrease compared to that in the control (Fig. 2). The gene expression levels of MMP-1 and MMP-3 in UVA-treated cells (2 J/cm2, 3 J/cm2, and 5 J/cm2) did not show a significant increase compared to those in the control (Fig. 1). In contrast, MMP-9 gene expression showed a significant decrease in UVA-treated cells (2 J/cm2, 3 J/cm2, and 5 J/cm2) compared to that in the control (p<0.05) (Fig. 1). The expression levels of AMPs, including psoriasin, hBD-2, hBD-3, and LL-37, in UVA-treated sebocytes (2 J/cm2, 3 J/cm2, and 5 J/cm2) did not show a significant change compared to those in the control (Fig. 1).
Fig. 1. The gene expression levels of inflammatory biomarkers in cultured human sebocytes treated with ultraviolet A (UVA) radiation (2 J/cm2, 3 J/cm2, and 5 J/cm2) did not show significant changes compared to those in the controls. However, gene expression of tumor necrosis factor (TNF)-α was significantly decreased in UVA-treated sebocytes (3 J/cm2 or 5 J/cm2) compared to the control (*p<0.05). In addition, matrix metalloproteinase (MMP)-9 gene expression was significantly decreased in UVA-treated cells (2 J/cm2, 3 J/cm2, and 5 J/cm2) compared to the control (*p<0.05). The expression level of each gene was normalized to that of β-actin and presented as a relative ratio. Error bars represent standard errors calculated based on three independent experiments. IL: interleukin, hBD: human β-defensin.
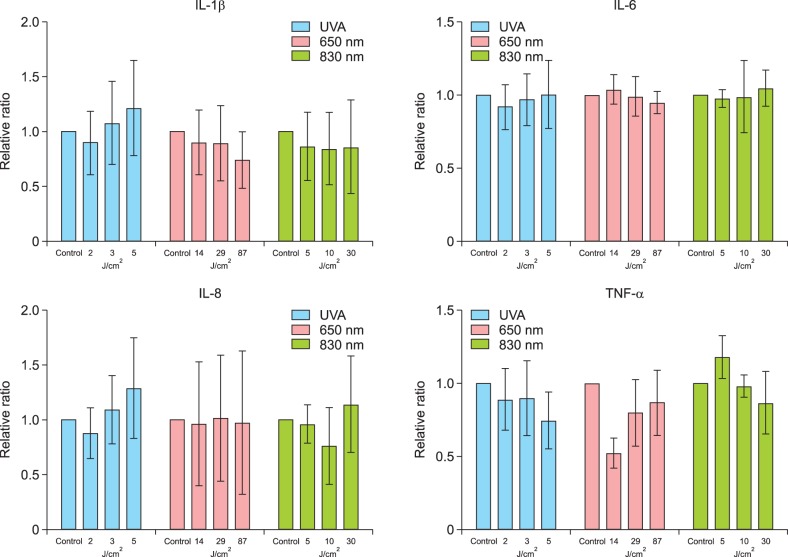
Fig. 2. The protein expression levels of interleukin (IL)-1β, IL-6, and IL-8 in cultured human sebocytes treated with ultraviolet A (UVA) radiation and light at wavelengths of 650 nm and 830 nm did not show significant change compared to those in the controls. The expression of tumor necrosis factor (TNF)-α protein was insignificantly decreased in sebocytes treated with UVA radiation and light at a wavelength of 650 nm compared to the control. Expression levels are presented as relative ratios. Error bars represent standard errors calculated based on three independent experiments.
Treatment of cultured sebocytes with light at a wavelength of 650 nm did not cause a significant increase in the expression of inflammatory cytokines, MMPs, or AMPs
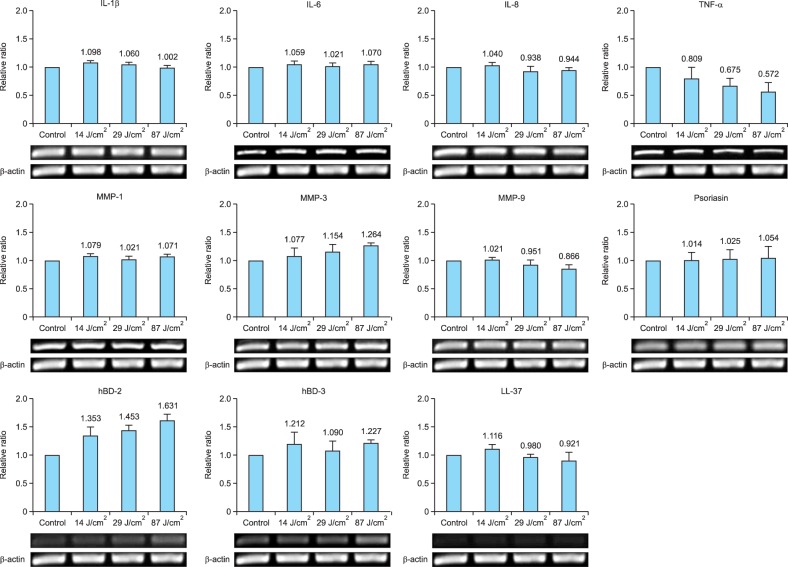
The gene and protein expression levels of inflammatory cytokines, including IL-1β, IL-6, IL-8, and TNF-α, in cultured sebocytes treated with light at a wavelength of 650 nm (14 J/cm2, 29 J/cm2, or 87 J/cm2) did not show a significant increase compared to those observed in the control (Fig. 2, 3). In addition, the gene expression levels of MMPs such as MMP-1, MMP-3, and MMP-9 in cultured sebocytes treated with light at a wavelength of 650 nm (14 J/cm2, 29 J/cm2, or 87 J/cm2) did not show a significant increase compared to those observed in the control (Fig. 3). Furthermore, the gene expression levels of AMPs such as psoriasin, hBD-2, hBD-3, and LL-37 in sebocytes treated with light at a wavelength of 650 nm (14 J/cm2, 29 J/cm2, or 87 J/cm2) did not show a significant change compared to those observed in the control (Fig. 3).
Fig. 3. The gene expression of inflammatory biomarkers in cultured sebocytes treated with light at a wavelength of 650 nm did not show a significant change compared to that in the control. The expression level of each gene was normalized to that of β-actin and presented as a relative ratio. Error bars represent standard errors calculated based on three independent experiments. IL: interleukin, TNF-α: tumor necrosis factor-α, MMP: matrix metalloproteinase, hBD: human β-defensin.
Treatment of cultured sebocytes with light at a wavelength of 830 nm did not cause a significant increase in the expression of inflammatory cytokines, MMPs, or AMPs
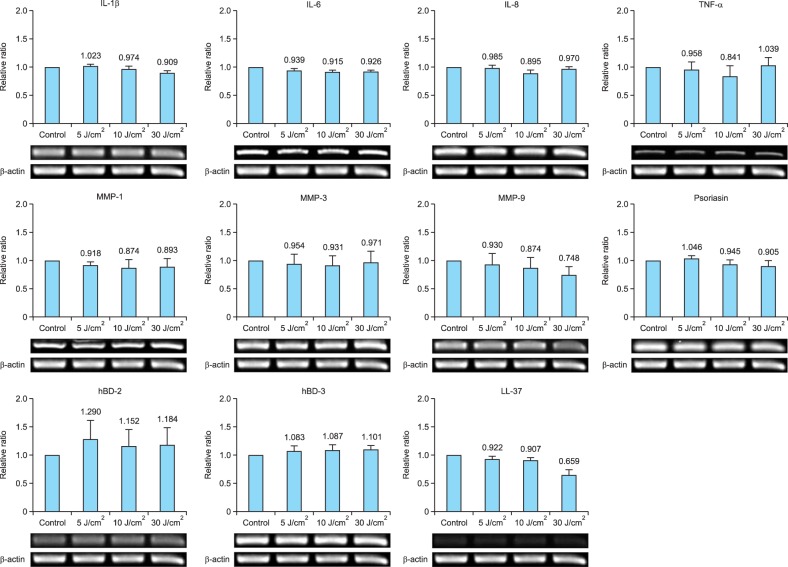
The gene and protein expression levels of inflammatory cytokines, including IL-1β, IL-6, IL-8, and TNF-α, in cultured sebocytes treated with light at a wavelength of 830 nm (5 J/cm2, 10 J/cm2, or 30 J/cm2) did not show a significant increase compared to those observed in the control (Fig. 2, 4). The gene expression levels of MMPs such as MMP-1, MMP-3, and MMP-9 and AMPs such as psoriasin, hBD-2, hBD-3, and LL-37 in cells treated with light at a wavelength of 830 nm (5 J/cm2, 10 J/cm2, or 30 J/cm2) did not show a significant change compared to those observed in the control (Fig. 4).
Fig. 4. The gene expression levels of inflammatory biomarkers in cultured human sebocytes treated with light at a wavelength of 830 nm did not show a significant change compared to those observed in the control. The expression level of each gene was normalized to that of β-actin and presented as a relative ratio. Error bars represent standard errors calculated based on three independent experiments. IL: interleukin, TNF-α: tumor necrosis factor-α, MMP: matrix metalloproteinase, hBD: human β-defensin.
Treatment of cultured sebocytes with UVA radiation and light at wavelengths of 650 nm and 830 nm did not cause a significant decrease in sebum production
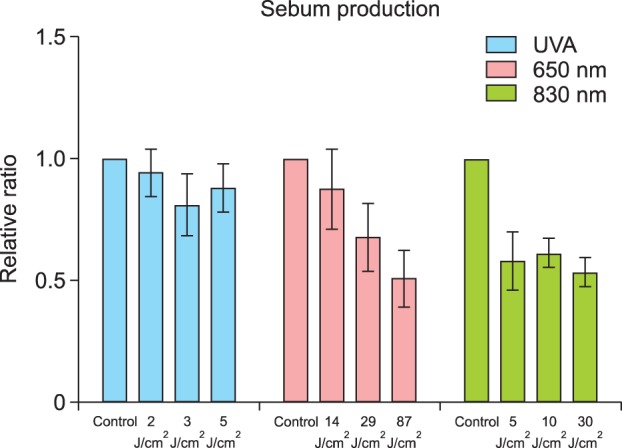
Sebum production in cultured sebocytes treated with UVA radiation (2 J/cm2, 3 J/cm2, or 5 J/cm2) did not show a significant decrease compared to that in the control (Fig. 5). Sebum production in cultured sebocytes treated with light at a wavelength of 650 nm (14 J/cm2, 29 J/cm2, or 87 J/cm2) showed a decreasing trend, as the dosage increased. An insignificant decrease in sebum production was observed in cultured sebocytes treated with light at a wavelength of 830 nm (5 J/cm2, 10 J/cm2, or 30 J/cm2) compared with that in the control (Fig. 5).
Fig. 5. Sebum production in cultured sebocytes treated with ultraviolet A (UVA) radiation and light at wavelengths of 650 nm and 830 nm showed an insignificant decrease compared to that in the control. Sebum production levels are presented as relative ratios. Error bars represent standard errors calculated based on three independent experiments.
DISCUSSION
Excessive sebum production and abnormal lipid secretion from sebaceous glands contribute to the formation of primary acne lesions12. It was reported that UV (UVA and UVB) irradiation of human back skin caused increased sebum production during the first 3 days after treatment13. The effects of UVB irradiation on sebaceous gland function have been widely studied. Hyperplasia of sebaceous glands, following UVB irradiation, was observed in biopsy-based studies in a hairless murine model14. Another study reported that UVB irradiation markedly increased both cell proliferation and sebum production in cultured hamster sebocytes15. In contrast, our previous study demonstrated that UVB treatment did not increase sebum production in cultured human sebocytes16. In the present study, we examined the effects of treating cultured human sebocytes with UVA radiation, visible light, and infrared light. Similar to our previous observations with UVB irradiation, we found that these treatments did not lead to a significant decrease in sebum production.
It was also reported that UV irradiation induced the expression of inflammatory cytokines, including IL-1α, IL-6, IL-1ra, and IL-10, in human back skin, at approximately 5 days after treatment12. Interestingly, time-dependent patterns of gene induction (involving TNF-α, IL-1β, and matrilysin) have also been observed in cultured HaCaT cells irradiated with UVA/UVB17. Consistent with such reports, studies involving UVB irradiation of whole mouse skin or human skin have also reported increased expression of inflammatory cytokines such as IL-1, IL-6, and TNF-α at the mRNA level18,19. Production of inflammatory cytokines, MMPs, or AMPs by sebocytes can significantly contribute to the formation and aggravation of acne lesions20. In our previous study, UVB irradiation of sebocytes resulted in an increase in the expression of inflammatory cytokines16. In contrast, the present study found that exposure to UVA, visible light, or infrared light did not cause a significant increase in the expression of inflammatory cytokines, MMPs, or AMPs.
It has been proposed that exposure to sunlight can stimulate the production of sebum and secretion of inflammatory cytokines, thereby promoting the development or aggravation of acne lesions. In contrast, sunlight exposure has been reported to induce anti-inflammatory effects in keratinocytes, by inhibiting IL-1α and ICAM-1 (intercellular adhesion molecule 1) production21. According to Charakida et al.22, up to 70% of patients report improvement in their acne after sunlight exposure. However, since sunlight is a mixture of UV, visible, and infrared wavelengths, it is unclear which of these components might be responsible for the favorable effects on acne. Blue light appears to be the most effective, despite poor depth of skin penetration23. Red light is able to penetrate deeper, reaching the skin layer that contains the sebaceous glands24. Although the various light-based treatments appear to target P. acnes directly or indirectly, red light exposure may produce additional anti-inflammatory effects, e.g., by influencing cytokine release from macrophages25. This study examined the effects of exposure to UVA, visible light, and infrared light on cultured human sebocytes, focusing on sebum production and expression of inflammatory biomarkers.
In summary, exposure of cultured human sebocytes to UVA radiation, visible (650 nm) light, and infrared (830 nm) light did not lead to a significant increase in the expression of the inflammatory biomarkers tested or in sebum production. Light therapy for acne vulgaris is primarily aimed at targeting P. acnes. This study, therefore, provides evidence that UVA radiation, visible light, and infrared light may be used for treatment of acne, without a significant increase in the expression of inflammatory biomarkers and sebum production in sebocytes.
ACKNOWLEDGMENT
This research was supported by Daekyoung Global Leader through the Korean Institute for Advancement of Technology; the Daegugyeong Institute for Regional Program Evaluation.
References
- 1.Mills OH, Porte M, Kligman AM. Enhancement of comedogenic substances by ultraviolet radiation. Br J Dermatol. 1978;98:145–150. doi: 10.1111/j.1365-2133.1978.tb01615.x. [DOI] [PubMed] [Google Scholar]
- 2.Mills OH, Kligman AM. Ultraviolet phototherapy and photochemotherapy of acne vulgaris. Arch Dermatol. 1978;114:221–223. [PubMed] [Google Scholar]
- 3.Papageorgiou P, Katsambas A, Chu A. Phototherapy with blue (415 nm) and red (660 nm) light in the treatment of acne vulgaris. Br J Dermatol. 2000;142:973–978. doi: 10.1046/j.1365-2133.2000.03481.x. [DOI] [PubMed] [Google Scholar]
- 4.Ammad S, Edwards C, Gonzalez M, Mills CM. The effect of blue light phototherapy on mild to moderate acne. Br J Dermatol. 2002;147(Suppl 62):95. [Google Scholar]
- 5.Kawada A, Aragane Y, Kameyama H, Sangen Y, Tezuka T. Acne phototherapy with a high-intensity, enhanced, narrow-band, blue light source: an open study and in vitro investigation. J Dermatol Sci. 2002;30:129–135. doi: 10.1016/s0923-1811(02)00068-3. [DOI] [PubMed] [Google Scholar]
- 6.Sigurdsson V, Knulst AC, van Weelden H. Phototherapy of acne vulgaris with visible light. Dermatology. 1997;194:256–260. doi: 10.1159/000246114. [DOI] [PubMed] [Google Scholar]
- 7.Hamilton FL, Car J, Lyons C, Car M, Layton A, Majeed A. Laser and other light therapies for the treatment of acne vulgaris: systematic review. Br J Dermatol. 2009;160:1273–1285. doi: 10.1111/j.1365-2133.2009.09047.x. [DOI] [PubMed] [Google Scholar]
- 8.Jeong E, Hong JW, Min JA, Lee DW, Sohn MY, Lee WJ, et al. Topical ALA-Photodynamic therapy for acne can induce apoptosis of sebocytes and down-regulate their TLR-2 and TLR-4 expression. Ann Dermatol. 2011;23:23–32. doi: 10.5021/ad.2011.23.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Magin P, Pond D, Smith W, Watson A. A systematic review of the evidence for 'myths and misconceptions' in acne management: diet, face-washing and sunlight. Fam Pract. 2005;22:62–70. doi: 10.1093/fampra/cmh715. [DOI] [PubMed] [Google Scholar]
- 10.Winston MH, Shalita AR. Acne vulgaris. Pathogenesis and treatment. Pediatr Clin North Am. 1991;38:889–903. doi: 10.1016/s0031-3955(16)38158-5. [DOI] [PubMed] [Google Scholar]
- 11.Fujie T, Shikiji T, Uchida N, Urano Y, Nagae H, Arase S. Culture of cells derived from the human sebaceous gland under serum-free conditions without a biological feeder layer or specific matrices. Arch Dermatol Res. 1996;288:703–708. doi: 10.1007/BF02505281. [DOI] [PubMed] [Google Scholar]
- 12.Zouboulis CC. Acne and sebaceous gland function. Clin Dermatol. 2004;22:360–366. doi: 10.1016/j.clindermatol.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 13.Suh DH, Kwon TE, Youn JI. Changes of comedonal cytokines and sebum secretion after UV irradiation in acne patients. Eur J Dermatol. 2002;12:139–144. [PubMed] [Google Scholar]
- 14.Lesnik RH, Kligman LH, Kligman AM. Agents that cause enlargement of sebaceous glands in hairless mice. II. Ultraviolet radiation. Arch Dermatol Res. 1992;284:106–108. doi: 10.1007/BF00373379. [DOI] [PubMed] [Google Scholar]
- 15.Akitomo Y, Akamatsu H, Okano Y, Masaki H, Horio T. Effects of UV irradiation on the sebaceous gland and sebum secretion in hamsters. J Dermatol Sci. 2003;31:151–159. doi: 10.1016/s0923-1811(03)00003-3. [DOI] [PubMed] [Google Scholar]
- 16.Lee WJ, Park KH, Sohn MY, Lee WC, Lee SJ, Kim DW. Ultraviolet B irradiation increases the expression of inflammatory cytokines in cultured sebocytes. J Dermatol. 2013;40:993–997. doi: 10.1111/1346-8138.12330. [DOI] [PubMed] [Google Scholar]
- 17.Skiba B, Neill B, Piva TJ. Gene expression profiles of TNF-alpha, TACE, furin, IL-1beta and matrilysin in UVA- and UVB-irradiated HaCat cells. Photodermatol Photoimmunol Photomed. 2005;21:173–182. doi: 10.1111/j.1600-0781.2005.00162.x. [DOI] [PubMed] [Google Scholar]
- 18.Scordi IA, Vincek V. Timecourse study of UVB-induced cytokine induction in whole mouse skin. Photodermatol Photoimmunol Photomed. 2000;16:67–73. doi: 10.1034/j.1600-0781.2000.d01-6.x. [DOI] [PubMed] [Google Scholar]
- 19.Murphy GM, Dowd PM, Hudspith BN, Brostoff J, Greaves MW. Local increase in interleukin-1-like activity following UVB irradiation of human skin in vivo. Photodermatol. 1989;6:268–274. [PubMed] [Google Scholar]
- 20.Zouboulis CC, Adjaye J, Akamatsu H, Moe-Behrens G, Niemann C. Human skin stem cells and the ageing process. Exp Gerontol. 2008;43:986–997. doi: 10.1016/j.exger.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 21.Shnitkind E, Yaping E, Geen S, Shalita AR, Lee WL. Anti-inflammatory properties of narrow-band blue light. J Drugs Dermatol. 2006;5:605–610. [PubMed] [Google Scholar]
- 22.Charakida A, Seaton ED, Charakida M, Mouser P, Avgerinos A, Chu AC. Phototherapy in the treatment of acne vulgaris: what is its role? Am J Clin Dermatol. 2004;5:211–216. doi: 10.2165/00128071-200405040-00001. [DOI] [PubMed] [Google Scholar]
- 23.Elman M, Lebzelter J. Light therapy in the treatment of acne vulgaris. Dermatol Surg. 2004;30:139–146. doi: 10.1111/j.1524-4725.2004.30053.x. [DOI] [PubMed] [Google Scholar]
- 24.Ross EV. Optical treatments for acne. Dermatol Ther. 2005;18:253–266. doi: 10.1111/j.1529-8019.2005.05024.x. [DOI] [PubMed] [Google Scholar]
- 25.Young S, Bolton P, Dyson M, Harvey W, Diamantopoulos C. Macrophage responsiveness to light therapy. Lasers Surg Med. 1989;9:497–505. doi: 10.1002/lsm.1900090513. [DOI] [PubMed] [Google Scholar]