Abstract
Oncocytic lipoadenoma is an exceedingly uncommon neoplasm of the salivary gland composed of oncocytic epithelium and adipose tissue. Retrospective. Seven cases of oncocytic lipoadenoma were analyzed in order to further characterize the clinical and pathologic features of this rare tumor. The patients included six males and one female who ranged from 40 to 83 years of age (mean 62 years) at presentation. All tumors arose in the parotid gland. Grossly, the tumors were solitary, well circumscribed and had light brown to yellow cut surfaces. Histologically, the tumors were composed of an admixed population of oncocytes and adipocytes in varying proportions, with the lipomatous component ranging from 5 to 70 %. Other common features included the presence of serous acini, ductal elements, sebaceous glands, and a patchy chronic inflammation. Clinical follow up information, available in all cases, with a duration of 3–148 months (mean 57 months), showed no evidence of tumor recurrence. Due to its rarity, oncocytic lipoadenoma can pose problems in diagnosis, although the distinctive morphologic features of this neoplasm allow for separation from more commonly recognized oncocytic neoplasms of the salivary glands.
Keywords: Oncocytic lipoadenoma, Parotid gland, Salivary gland, Sialolipoma, Adenolipoma
Introduction
Oncocytic neoplasms of the salivary glands consist of a wide array of morphologically diverse tumors, all of which are characterized histologically by a predominance of oncocytes. With the exception of papillary cystadenoma lymphomatosum (Warthin tumor), salivary gland tumors with prominent oncocytic features are uncommon, and include oncocytoma, oncocytic carcinoma, and oncocytic cystadenoma, as well as oncocytic variants of pleomorphic adenoma, myoepithelioma, mucoepidermoid carcinoma, and epithelial-myoepithelial carcinoma [1–11].
Another recently characterized oncocytic neoplasm arising in the salivary gland has been termed oncocytic lipoadenoma. First described in 1998, oncocytic lipoadenoma is a histologically distinctive tumor composed of oncocytic cells and mature adipocytes [12]. The tumor is decidedly rare; with only a few examples in the literature, mostly as case reports [13–24]. With scant published data, we analyzed seven new cases of oncocytic lipoadenoma in order to further delineate the clinicopathologic features of this unusual salivary gland neoplasm, combined with a thorough review of the literature.
Materials and Methods
Seven cases of oncocytic lipoadenoma were identified from the files of the Departments of Pathology within Southern California Permanente Medical Group. Hematoxylin and eosin stained slides from all cases were reviewed, with a range of 1–9 slides (mean 4 slides) per case available for analysis. Clinical data, treatment, and follow-up information were obtained from electronic medical records. This clinical investigation was conducted in accordance and compliance with all statutes, directives, and guidelines of an Internal Review Board authorization (#5968) performed under the direction of Southern California Permanente Medical Group and the Code of Federal Regulations, Title 45, Part 46.
Results
Clinical Features
Clinical data are summarized in Table 1. The patients included six men and one woman. Their ages at presentation ranged from 40 to 83 years of age, with a mean age of 62 years (median 65 years). A description of clinical symptoms was available for all patients. Six patients presented with a painless, slow growing neck mass, while one patient complained of headache and ear pain. The duration of symptoms ranged from 3 to 180 months (mean 41 months). All tumors were located in the parotid gland (4 right, 3 left). Preoperative fine needle aspiration of the tumor was performed in six patients, with diagnoses of pleomorphic adenoma (n = 2), Warthin tumor (n = 1), oncocytic lesion (n = 1), and nondiagnostic (n = 2).
Table 1.
Clinicopathologic features of seven cases of oncocytic lipoadenoma
| Case no. | Age (years) | Sex | Symptom duration | Anatomic site and side | Size (cm) | % Fat component | Treatment | Follow-up (months) |
|---|---|---|---|---|---|---|---|---|
| 1 | 61 | Male | 15 years | Left parotid gland | 2.0 | 20 | Superficial parotidectomy | ANED (42) |
| 2 | 83 | Male | 3 months | Right parotid gland | 2.5 | 15 | Superficial parotidectomy | ANED (12) |
| 3 | 67 | Male | 6 months | Right parotid gland | 4.0 | 5 | Superficial parotidectomy | ANED (3) |
| 4 | 40 | Female | 9 months | Right parotid gland | 4.0 | 50 | Superficial parotidectomy | ANED (148) |
| 5 | 56 | Male | “Years” | Left parotid gland | 3.5 | 70 | Superficial parotidectomy | ANED (110) |
| 6 | 65 | Male | 24 months | Left parotid gland | 1.9 | 50 | Superficial parotidectomy | ANED (57) |
| 7 | 65 | Male | 24 months | Right parotid gland | 3.5 | 50 | Superficial parotidectomy | ANED (26) |
ANED alive with no evidence of disease
Pathologic Features
On gross examination, the tumors were well circumscribed and limited to the parotid gland, with solid homogeneous, light brown or yellow cut surfaces (Fig. 1). The tumors ranged from 1.9 to 4.0 cm in greatest dimension (mean 3.1 cm).
Fig. 1.
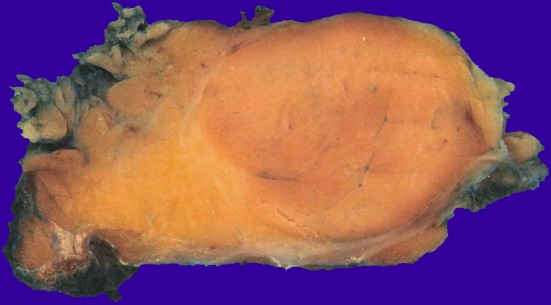
Macroscopically oncocytic lipoadenoma is represented by a well circumscribed nodule with a light brown cut surface
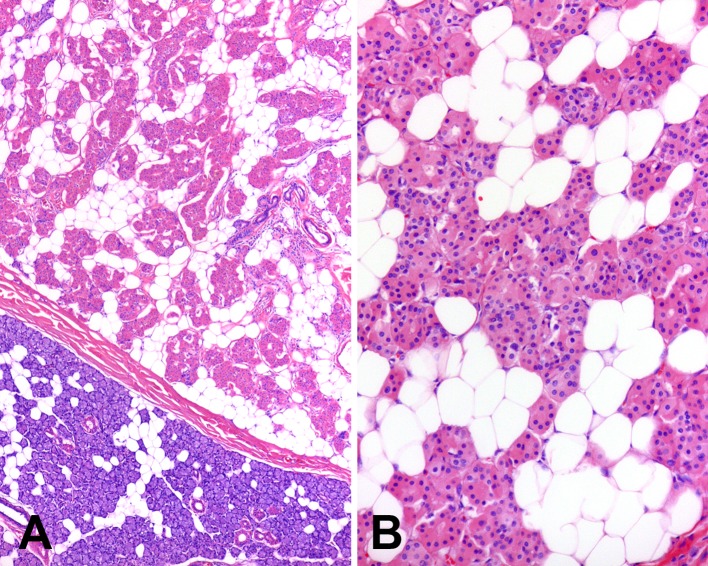
Microscopically all tumors were surrounded by a thin, fibrous capsule (Fig. 2a). In three cases, the capsule was poorly formed and incomplete, with areas of uninvolved salivary gland parenchyma directly adjacent to tumor. The tumors were composed of a mixed population of oncocytes and adipocytes in varying proportions (Fig. 2b). The oncocytes were round to polygonal with distinct cell borders and abundant granular, eosinophilic cytoplasm. Nuclei were centrally located, with a vesicular to pyknotic appearance, and contained small, variably prominent nucleoli. The oncocytes were arranged in compact nests and small acini. The adipocytic component of the tumors ranged from 5 to 70 %. The adipocytes had the appearance of mature fat, with mild variability in size and peripherally located hyperchromatic, small nuclei. In some tumors, the adipocytes were relatively evenly dispersed between the nests of oncocytes, while in others they were irregularly distributed, resulting in areas dominated by adipose tissue with scant oncocytes or a prominent oncocytic component with a paucity of fat cells (Fig. 3). Neither the oncocytic nor the lipomatous components of the tumors showed any cellular pleomorphism or mitoses.
Fig. 2.
Microscopically oncocytic lipoadenomas are (a) surrounded by a fibrous capsule and (b) composed of oncocytes and adipocytes
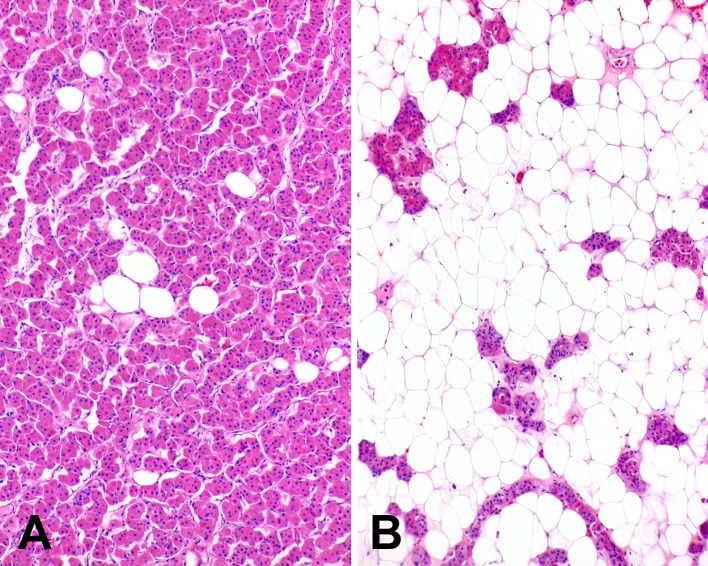
Fig. 3.
Oncocytic lipoadenomas may have a variable appearance with either the (a) oncocytic or (b) lipomatous components of the tumor predominating
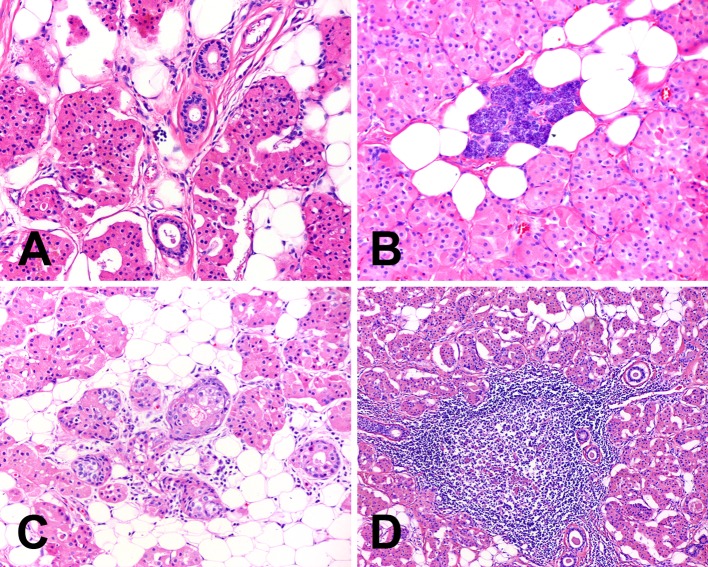
Scattered ductal elements were observed as a minor feature in all cases (Fig. 4a). These consisted of collections of striated ducts as well as larger interlobular ducts, often with periductal fibrosis. Conspicuous serous acini were present in four tumors (Fig. 4b) and rare sebaceous glands were identified in six tumors (Fig. 4c). Six tumors displayed a patchy inflammatory infiltrate composed predominantly of small lymphocytes and occasional plasma cells. The lymphocytic infiltrate was particularly prominent in one case with well formed germinal centers (Fig. 4d). Stromal alteration characterized by dense fibrocollagenous tissue was seen in one case.
Fig. 4.
Other microscopic findings often seen in oncocytic lipoadenomas include (a) ductal elements, (b) serous acini, (c) sebaceous glands, and (d) chronic inflammation
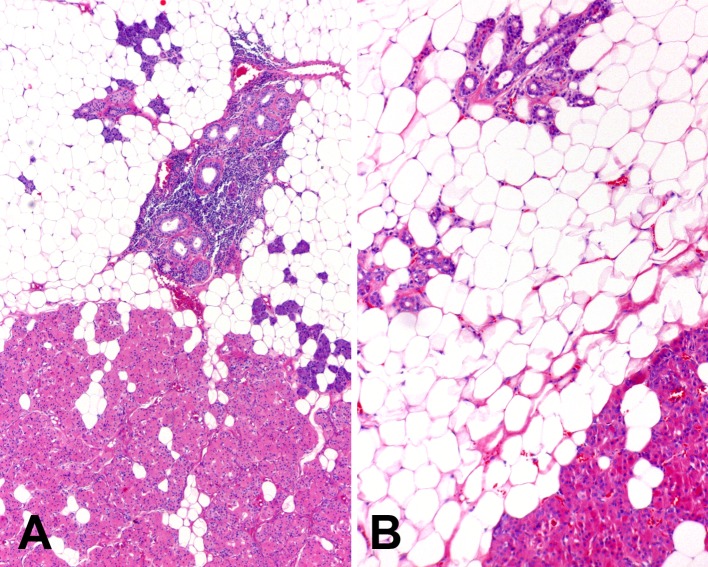
One unusual tumor, in addition to admixed oncocytic and lipomatous components, contained a well delineated area composed of predominantly adipose tissue with islands of salivary gland duct-acinar units resembling sialolipoma (Fig. 5). None of the tumors exhibited necrosis, vascular invasion, perineural invasion, capsular invasion, or extraglandular extension.
Fig. 5.
(a) Low and (b) intermediate magnification images of an example of oncocytic lipoadenoma with an area resembling sialolipoma. The lower aspects of the images show the typical microscopic appearance of oncocytic lipoadenoma while the upper aspects show a predominance of mature adipose tissue with scattered normal salivary gland ducts and acini similar to that observed in sialolipoma
Treatment and Follow-up
Treatment consisted of superficial parotidectomy in all cases. Clinical follow up information was available for all patients with a mean duration of 57 months (range 3–148 months). None of the patients developed local recurrence and all are alive with no evidence of disease.
Discussion
Lipoadenoma and adenolipoma are designations used to refer to neoplasms composed of epithelial and lipomatous elements. They have been described in many anatomic sites, including the skin, breast, parathyroid, thyroid, and salivary gland [24–31]. Mixed lipomatous and epithelial tumors where the latter component is comprised of oncocytes have been termed oncocytic lipoadenoma, which appear to be unique to salivary glands. Oncocytic lipoadenoma is rare; to date twenty cases have been reported in the literature (Table 2) [12–24]. The “adenoma” designation highlights the predominance of the epithelial component, rather than sialolipoma which emphasizes the fatty component. Combining the cases from the present clinical series with those previously described shows a wide age range at presentation (7–89 years) with a mean age of 57.7 years. The tumor occurs slightly more frequently in men than women, with a male to female ratio of 1.5:1. Patients typically present with a painless, slowly growing mass or swelling that has not uncommonly been present for an extended duration of time (mean 34.5 months; range 2–180 months) [12, 14, 15, 17–21, 23]. The vast majority of oncocytic lipoadenomas have originated in the parotid gland, with only few reports of submandibular gland involvement [12, 16, 22]. The tumor has thus far not been documented in minor salivary glands. Based on present data and other reports, oncocytic lipoadenoma appears to be a benign tumor with no risk or recurrence or aggressive behavior (mean follow up 40 months) [12, 14, 18, 19, 22–24].
Table 2.
Previously reported cases of oncocytic lipoadenoma of the salivary glands
| References | Age (years) | Sex | Anatomic site | Size (cm) | % Fat component | Follow-up (months) |
|---|---|---|---|---|---|---|
| Hirokawa et al. [12] | 66 | F | Submandibular gland | 11.0 | 50 | ANED (30) |
| Kato and Korie [13] | 57 | F | Parotid gland | 4.4 | 40 | NR |
| Klieb and Perez-Ordoñez [14] | 46 | F | Parotid gland | 3.0 | 30–40 | ANED (6) |
| Aouad et al. [15] | 38 | M | Parotid gland | 4.0 | NR | NR |
| Pusiol et al. [16] | 73 | M | Submandibular gland | 9.0 | 80 | ANED (NR) |
| Chahwala et al. [17] | 50 | F | Parotid gland | 13.5 | NR | NR |
| Ilie et al. [18] | 64 | M | Parotid gland | 5.0 | 70 | ANED (24) |
| Tokyol et al. [19] | 56 | M | Parotid gland | 7.0 | 40 | ANED (6) |
| McNeil et al. [20] | 70 | M | Parotid gland | 4.2 | 40 | NR |
| Mitsimponas et al. [21] | 55 | F | Parotid gland | 2.4 | 10 | ANED (12) |
| Casadei et al. [22] | NR | M | NR | NR | 30 | ANED (NR) |
| Casadei et al. [22] | NR | F | Submandibular gland | NR | 30 | ANED (NR) |
| Casadei et al. [22] | NR | F | Parotid gland | NR | 30 | ANED (NR) |
| Devadoss et al. [23] | 50 | F | Parotid gland | 13.5 | 50–60 | ANED (24) |
| Agaimy et al. [24] | 63 | M | Parotid gland | 4.5 | 10 | ANED (6) |
| Agaimy et al. [24] | 29 | M | Parotid gland | 4.5 | 20 | ANED (141) |
| Agaimy et al. [24] | 54 | F | Parotid gland | 2.9 | 10 | ANED (18) |
| Agaimy et al. [24] | 55 | M | Parotid gland | 2.7 | 70 | ANED (13) |
| Agaimy et al. [24] | 7 | F | Parotid gland | NR | 50 | NR |
| Agaimy et al. [24] | 89 | M | Parotid gland | 4.2 | 80 | NR |
M male, F female, NR not reported, ANED alive with no evidence of disease
On gross examination, oncocytic lipoadenomas are well circumscribed neoplasms with pushing borders. The cut surface may vary from tan to brown to yellow, depending on the amount of adipose tissue present. Histologically there is typically evidence of a thin fibrous capsule separating the tumor from the surrounding salivary gland parenchyma. The neoplasm has a morphologically distinctive appearance characterized by the presence of two distinct cell populations: oncocytes and adipocytes. The oncocytes are usually arranged in nests and acini with a microscopic appearance resembling a conventional oncocytoma. The lipomatous component of the tumor has the appearance of mature adipose tissue and can comprise as little as 5 % to as much as 80 % of the tumor volume [12–24]. The adipocytes may be relatively evenly dispersed among the oncocytes, or may be irregularly distributed with either the oncocytic or lipomatous component predominating in a given area of the tumor. This should be taken into consideration when rendering a diagnosis on core needle biopsy or fine needle aspiration material, where sampling is limited.
Although not typically emphasized, foci of normal salivary gland elements including ducts, serous acini, and sebaceous glands, are a frequent finding in oncocytic lipoadenomas [14, 16, 18–21, 24]. A mild tumor associated lymphocytic inflammatory infiltrate, seen in the majority of the cases in the present series and similarly observed by others, is also a common feature [12, 14, 18–21, 24].
The histologic appearance of oncocytic lipoadenoma is sufficiently distinctive to be recognizable on routine hematoxylin and eosin stained sections. Immunohistochemical studies are not usually required for diagnosis, though the immunophenotype of oncocytic lipoadenoma has been previously elucidated. The lesional oncocytes express AE1/AE3, CK7, and EMA [13, 14, 18–20, 23], with p63, CK5/6, CK14, CK19, and high molecular weight keratin positivity observed in a basal cell distribution [14, 18, 20]. The tumor cells are negative for muscle specific actin, smooth muscle actin, and calponin [14, 18–20, 23].
The principle differential diagnostic considerations of oncocytic lipoadenoma include other oncocytic neoplasms of the salivary gland. Among the various salivary gland tumors exhibiting a prominent oncocytic component, oncocytic lipoadenoma is most likely to be mistaken for oncocytoma. Both are well circumscribed tumors comprised of classical eosinophilic oncocytic cells. Oncocytomas, however, are composed exclusively of oncocytes and lack the lipomatous component characteristic of oncocytic lipoadenoma. The presence of adipose tissue similarly allows for separation of oncocytic lipoadenoma from stroma poor Warthin tumor, Warthin tumor with nodular oncocytic proliferation, and oncocytic pleomorphic adenoma. Nodular oncocytic hyperplasia shows multifocal nests of oncocytes intermixed with adipocytes and normal acinar cells, perhaps simulating the appearance of oncocytic lipoadenoma. Oncocytic lipoadenoma, however, is at least partially surrounded by a fibrous connective tissue capsule, while nodular oncocytic hyperplasia represents an unencapsulated proliferation of oncocytes frequently with multifocal distribution.
The differential diagnosis also includes sialolipoma. Sialolipoma is characteristically composed predominantly of adipose tissue with admixed islands of salivary gland acini, ductal, myoepithelial and basal cells [32]. The epithelial/myoepithelial elements may show focal oncocytic change in some cases [32–35], but unlike oncocytic lipoadenoma, sialolipoma in general lacks a prominent oncocytic component. The tumor tends to show lobulation with evenly distributed salivary gland elements within the fat.
In the present series we did observe one case that showed a discrete area resembling sialolipoma within an otherwise typical oncocytic lipoadenoma. Similar salivary gland tumors exhibiting features of both oncocytic lipoadenoma and sialolipoma have been illustrated by other investigators, one of which was described using the term “oncocytic sialolipoma” [16, 24]. Whether to classify such lesions as oncocytic lipoadenoma, oncocytic sialolipoma, or as a hybrid lipoepithelial salivary gland tumor remains to be determined. Nonetheless, the existence of these phenotypically mixed cases suggests a possible histogenetic relationship between oncocytic lipoadenoma and sialolipoma.
The pathogenetic mechanisms underlying the development of oncocytic lipoadenoma remain elusive. It could be argued that the neoplasm represents an oncocytoma with lipomatous metaplasia. Adipocytic differentiation in salivary gland tumors such as pleomorphic adenoma has been attributed to transdifferentiation of constituent myoepithelial cells resulting in metaplastic transition to adipocytes [36, 37]. However, ultrastructural and immunohistochemical studies have not demonstrated a definite myoepithelial cell population in oncocytic lipoadenoma [13, 14, 18–20, 23]. A subset of cells immunoreactive for p63 and basal type keratins have been observed in oncocytic lipoadenomas in a peripheral basal cell type distribution, though the exact nature of this particular cell population is unclear [14, 18, 20]. These could represent pluripotent basal or stem cells capable of lipomatous metaplastic change, though this remains speculative.
From a molecular genetics perspective, cytogenetic analysis has been performed on a single case of oncocytic lipoadenoma which demonstrated a t(12;14) translocation involving the high mobility group AT-hook 2 (HMGA2) gene on chromosome 12 [18]. HMGA2 encodes a protein belonging to the non-histone high mobility group protein family. The protein influences an array of cellular processes through regulation of gene transcription [38]. In addition to oncocytic lipoadenoma, HMGA2 gene rearrangements have also been recognized in a number of other neoplasms, including soft tissue lipomas and a subset of salivary gland pleomorphic adenomas [39, 40]. Further molecular analyses may yield more definitive evidence as to the significance of HMGA2 gene disruption in the development of oncocytic lipoadenoma.
Oncocytic lipoadenoma is a unique salivary gland neoplasm. The current 7 cases serves to further characterize the clinical and pathologic features of this rare tumor. Oncocytic lipoadenomas have a distinctive morphologic appearance that permits accurate diagnosis and separation from other salivary gland neoplasms with a prominent oncocytic component. Clinically, the tumor behaves in a benign fashion, with no risk of recurrence following surgical excision.
Acknowledgments
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of Southern California Permanente Medical Group.
References
- 1.Blanck C, Eneroth CM, Jakobsson PA. Oncocytoma of the parotid gland: neoplasm or nodular hyperplasia? Cancer. 1970;25:919–925. doi: 10.1002/1097-0142(197004)25:4<919::AID-CNCR2820250427>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 2.Johns ME, Batsakis JG, Short CD. Oncocytic and oncocytoid tumors of the salivary glands. Laryngoscope. 1973;83:1940–1952. doi: 10.1288/00005537-197312000-00005. [DOI] [PubMed] [Google Scholar]
- 3.Gray SR, Cornog JL, Jr, Seo IS. Oncocytic neoplasms of salivary glands: a report of fifteen cases including two malignant oncocytomas. Cancer. 1976;38:1306–1317. doi: 10.1002/1097-0142(197609)38:3<1306::AID-CNCR2820380333>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 4.Palmer TJ, Gleeson MJ, Eveson JW, Cawson RA. Oncocytic adenomas and oncocytic hyperplasia of salivary glands: a clinicopathological study of 26 cases. Histopathology. 1990;16:487–493. doi: 10.1111/j.1365-2559.1990.tb01549.x. [DOI] [PubMed] [Google Scholar]
- 5.Brandwein MS, Huvos AG. Oncocytic tumors of major salivary glands. A study of 68 cases with follow-up of 44 patients. Am J Surg Pathol. 1991;15:514–528. doi: 10.1097/00000478-199106000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Thompson LD, Wenig BM, Ellis GL. Oncocytomas of the submandibular gland. A series of 22 cases and a review of the literature. Cancer. 1996;78:2281–2287. doi: 10.1002/(SICI)1097-0142(19961201)78:11<2281::AID-CNCR3>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 7.Paulino AF, Huvos AG. Oncocytic and oncocytoid tumors of the salivary glands. Semin Diagn Pathol. 1999;16:98–104. [PubMed] [Google Scholar]
- 8.Skálová A, Michal M, Ryska A, Simpson RH, Kinkor Z, Walter J, Leivo I. Oncocytic myoepithelioma and pleomorphic adenoma of the salivary glands. Virchows Arch. 1999;434:537–546. doi: 10.1007/s004280050381. [DOI] [PubMed] [Google Scholar]
- 9.Weinreb I, Seethala RR, Perez-Ordoñez B, Chetty R, Hoschar AP, Hunt JL. Oncocytic mucoepidermoid carcinoma: clinicopathologic description in a series of 12 cases. Am J Surg Pathol. 2009;33:409–416. doi: 10.1097/PAS.0b013e318184b36d. [DOI] [PubMed] [Google Scholar]
- 10.Zhou CX, Shi DY, Ma DQ, Zhang JG, Yu GY, Gao Y. Primary oncocytic carcinoma of the salivary glands: a clinicopathologic and immunohistochemical study of 12 cases. Oral Oncol. 2010;46:773–778. doi: 10.1016/j.oraloncology.2010.07.014. [DOI] [PubMed] [Google Scholar]
- 11.Seethala RR. Oncocytic and apocrine epithelial myoepithelial carcinoma: novel variants of a challenging tumor. Head Neck Pathol. 2013;7(Suppl 1):S77–S84. doi: 10.1007/s12105-013-0461-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hirokawa M, Shimizu M, Manabe T, Ito J, Ogawa S. Oncocytic lipoadenoma of the submandibular gland. Hum Pathol. 1998;29:410–412. doi: 10.1016/S0046-8177(98)90125-3. [DOI] [PubMed] [Google Scholar]
- 13.Kato M, Horie Y. Oncocytic lipoadenoma of the parotid gland. Histopathology. 2000;36:285–286. doi: 10.1046/j.1365-2559.2000.0872e.x. [DOI] [PubMed] [Google Scholar]
- 14.Klieb HB, Perez-Ordoñez B. Oncocytic lipoadenoma of the parotid gland with sebaceous differentiation. Study of its keratin profile. Virchows Arch. 2006;449:722–725. doi: 10.1007/s00428-006-0317-z. [DOI] [PubMed] [Google Scholar]
- 15.Aouad R, Matar N, Sader-Ghorra C, Haddad A. Pathology quiz case 1. Oncocytic lipoadenoma of the parotid gland. Arch Otolaryngol Head Neck Surg. 2008;134(446):448. doi: 10.1001/archotol.134.4.446. [DOI] [PubMed] [Google Scholar]
- 16.Pusiol T, Franceschetti I, Scialpi M, Piscioli I. Oncocytic sialolipoma of the submandibular gland with sebaceous differentiation: a new pathological entity. Indian J Pathol Microbiol. 2009;52:379–382. doi: 10.4103/0377-4929.55000. [DOI] [PubMed] [Google Scholar]
- 17.Chahwala Q, Siddaraju N, Singh N, Goneppanavar M, Basu D. Fine needle aspiration cytology of oncocytic lipoadenoma of the parotid gland: report of a rare case. Acta Cytol. 2009;53:437–439. doi: 10.1159/000325348. [DOI] [PubMed] [Google Scholar]
- 18.Ilie M, Hofman V, Pedeutour F, Attias R, Santini J, Hofman P. Oncocytic lipoadenoma of the parotid gland: immunohistochemical and cytogenetic analysis. Pathol Res Pract. 2010;206:66–72. doi: 10.1016/j.prp.2009.02.008. [DOI] [PubMed] [Google Scholar]
- 19.Tokyol C, Dilek FH, Aktepe F, Ayçiçek A, Altuntaş A. Oncocytic lipoadenoma of the parotid gland: a case report with fine needle aspiration cytology findings. Kulak Burun Bogaz Ihtis Derg. 2010;20:146–149. [PubMed] [Google Scholar]
- 20.McNeil ML, Bullock MJ, Trites JR, Hart RD, Taylor SM. Oncocytic lipoadenoma of the parotid gland with sebaceous differentiation in a 73-year-old male. J Otolaryngol Head Neck Surg. 2010;39:E48–E50. [PubMed] [Google Scholar]
- 21.Mitsimponas KT, Agaimy A, Schlittenbauer T, Nkenke E, Neukam FW. Oncocytic lipoadenoma of the parotid gland: a report of a new case and review of the literature. Int J Clin Exp Pathol. 2012;5:1000–1006. [PMC free article] [PubMed] [Google Scholar]
- 22.Casadei GP, Collina G, Salerno A, Canani FB, Dall’olio D, Lega S. Oncocytic lipoadenomatous tumours of salivary glands. Histopathology. 2012;61:1225–1227. doi: 10.1111/j.1365-2559.2012.04312.x. [DOI] [PubMed] [Google Scholar]
- 23.Devadoss CW, Murugan P, Basu D, Jagdish S. Oncocytic lipoadenoma of the parotid gland: report of a rare case. J Clin Diagn Res. 2012;6:1076–1078. [Google Scholar]
- 24.Agaimy A, Ihrler S, Märkl B, Lell M, Zenk J, Hartmann A, Michal M, Skalova A. Lipomatous salivary gland tumors: a series of 31 cases spanning their morphologic spectrum with emphasis on sialolipoma and oncocytic lipoadenoma. Am J Surg Pathol. 2013;37:128–137. doi: 10.1097/PAS.0b013e31826731e0. [DOI] [PubMed] [Google Scholar]
- 25.Schröder S, Böcker W, Hüsselmann H, Dralle H. Adenolipoma (thyrolipoma) of the thyroid gland report of two cases and review of literature. Virchows Arch A Pathol Anat Histopathol. 1984;404:99–103. doi: 10.1007/BF00704254. [DOI] [PubMed] [Google Scholar]
- 26.Hitchcock MG, Hurt MA, Santa Cruz DJ. Adenolipoma of the skin: a report of nine cases. J Am Acad Dermatol. 1993;29:82–85. doi: 10.1016/0190-9622(93)70157-O. [DOI] [PubMed] [Google Scholar]
- 27.Dietrich CU, Pandis N, Andersen JA, Heim S. Chromosome abnormalities in adenolipomas of the breast: karyotypic evidence that the mesenchymal component constitutes the neoplastic parenchyma. Cancer Genet Cytogenet. 1994;72:146–150. doi: 10.1016/0165-4608(94)90131-7. [DOI] [PubMed] [Google Scholar]
- 28.Yau KC, Tsang WY, Chan JK. Lipoadenoma of the parotid gland with probable striated duct differentiation. Mod Pathol. 1997;10:242–246. [PubMed] [Google Scholar]
- 29.Hirokawa M, Bando Y, Tashiro T, Kuma S, Kawata I, Sano T. Parotid lipoadenoma with sclerotic and polycystic changes. Virchows Arch. 2002;440:549–550. doi: 10.1007/s00428-002-0600-6. [DOI] [PubMed] [Google Scholar]
- 30.Daboin KP, Ochoa-Perez V, Luna MA. Adenolipomas of the head and neck: analysis of 6 cases. Ann Diagn Pathol. 2006;10:72–76. doi: 10.1016/j.anndiagpath.2005.07.012. [DOI] [PubMed] [Google Scholar]
- 31.Seethala RR, Ogilvie JB, Carty SE, Barnes EL, Yim JH. Parathyroid lipoadenomas and lipohyperplasias: clinicopathologic correlations. Am J Surg Pathol. 2008;32:1854–1867. doi: 10.1097/PAS.0b013e31817a8ef7. [DOI] [PubMed] [Google Scholar]
- 32.Nagao T, Sugano I, Ishida Y, Asoh A, Munakata S, Yamazaki K, Konno A, Kondo Y, Nagao K. Sialolipoma: a report of seven cases of a new variant of salivary gland lipoma. Histopathology. 2001;38:30–36. doi: 10.1046/j.1365-2559.2001.01054.x. [DOI] [PubMed] [Google Scholar]
- 33.Parente P, Longobardi G, Bigotti G. Hamartomatous sialolipoma of the submandibular gland: case report. Br J Oral Maxillofac Surg. 2008;46:599–600. doi: 10.1016/j.bjoms.2008.02.006. [DOI] [PubMed] [Google Scholar]
- 34.Akrish S, Leiser Y, Shamira D, Peled M. Sialolipoma of the salivary gland: two new cases, literature review, and histogenetic hypothesis. J Oral Maxillofac Surg. 2011;69:1380–1384. doi: 10.1016/j.joms.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 35.Nonaka CF, Pereira KM, de Andrade Santos PP, de Almeida Freitas R, da Costa Miguel MC. Sialolipoma of minor salivary glands. Ann Diagn Pathol. 2011;15:6–11. doi: 10.1016/j.anndiagpath.2009.12.009. [DOI] [PubMed] [Google Scholar]
- 36.Ng WK, Ma L. Pleomorphic adenoma with extensive lipometaplasia. Histopathology. 1995;27:285–288. doi: 10.1111/j.1365-2559.1995.tb00223.x. [DOI] [PubMed] [Google Scholar]
- 37.Skálová A, Stárek I, Simpson RH, Kucerová V, Dvorácková J, Curík R, Dusková M. Spindle cell myoepithelial tumours of the parotid gland with extensive lipomatous metaplasia. A report of four cases with immunohistochemical and ultrastructural findings. Virchows Arch. 2001;439:762–767. doi: 10.1007/s004280100469. [DOI] [PubMed] [Google Scholar]
- 38.Cleynen I, Van de Ven WJ. The HMGA proteins: a myriad of functions (Review) Int J Oncol. 2008;32:289–305. [PubMed] [Google Scholar]
- 39.Hatano H, Morita T, Ogose A, Hotta T, Kobayashi H, Segawa H, Uchiyama T, Takenouchi T, Sato T. Clinicopathological features of lipomas with gene fusions involving HMGA2. Anticancer Res. 2008;28(1B):535–538. [PubMed] [Google Scholar]
- 40.Stenman G. Fusion oncogenes in salivary gland tumors: molecular and clinical consequences. Head Neck Pathol. 2013;7(Suppl 1):S12–S19. doi: 10.1007/s12105-013-0462-z. [DOI] [PMC free article] [PubMed] [Google Scholar]