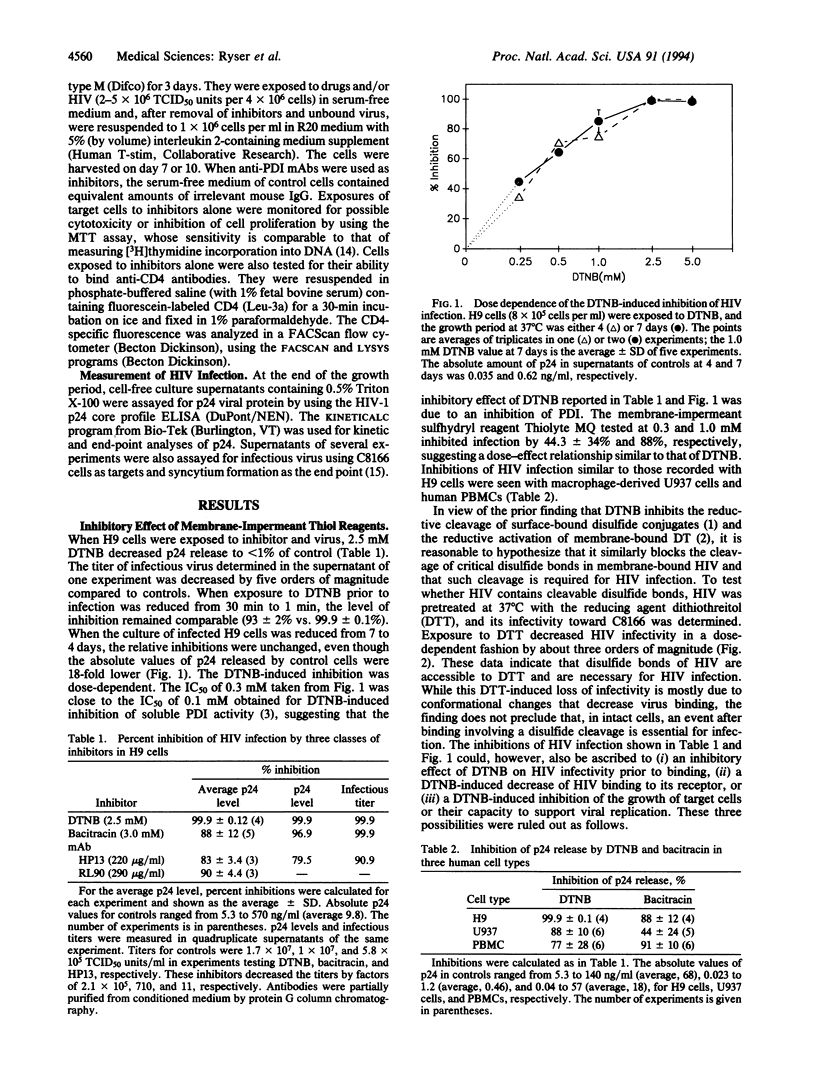
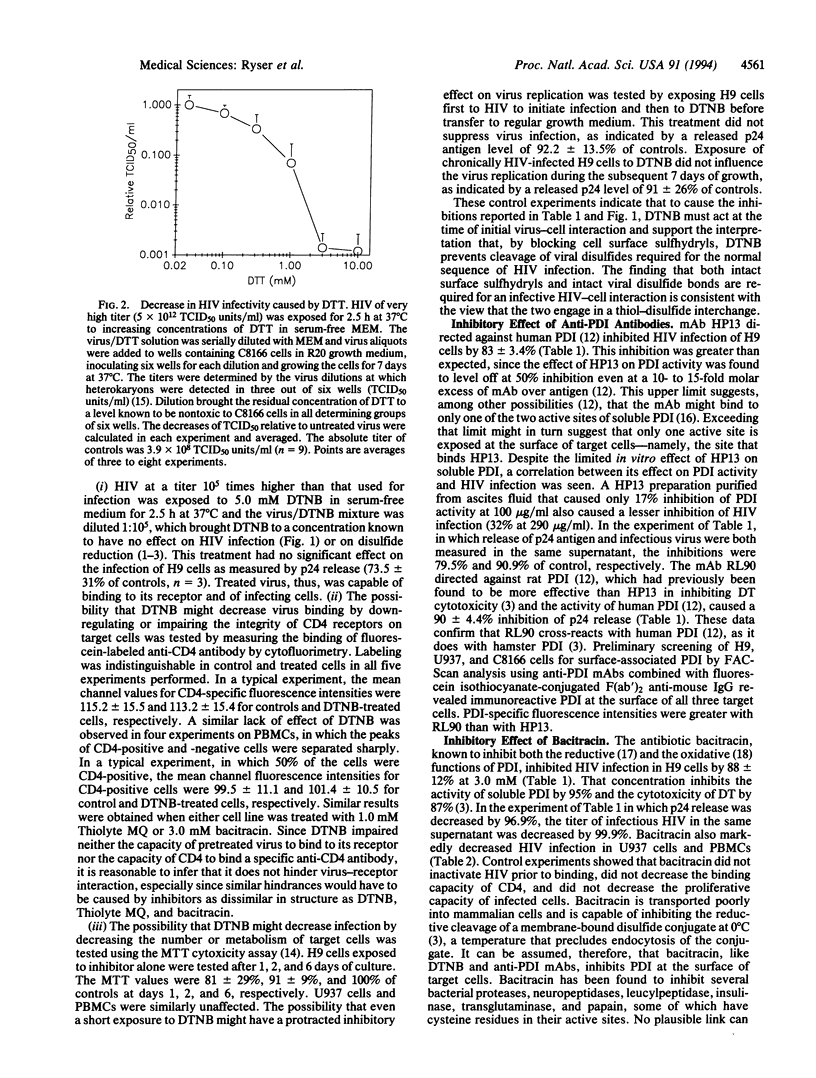
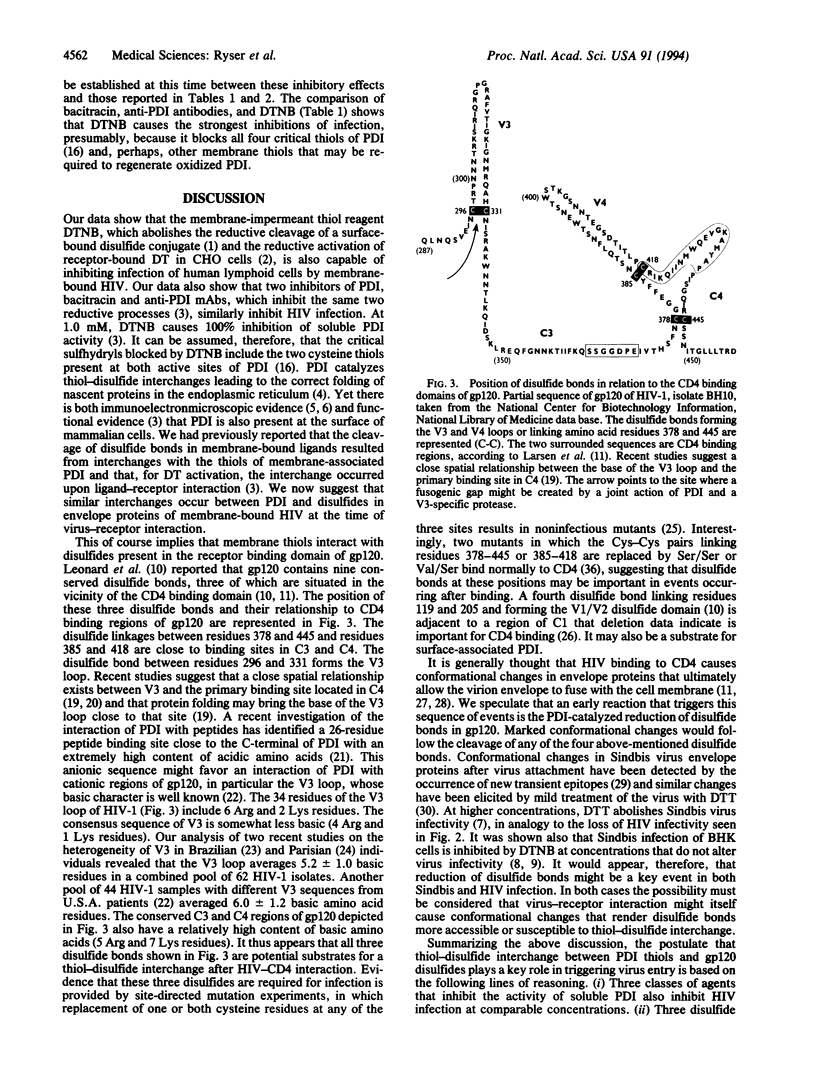
Abstract
The cell surface of mammalian cells is capable of reductively cleaving disulfide bonds of exogenous membrane-bound macromolecules (for instance, the interchain disulfide of diphtheria toxin), and inhibiting this process with membrane-impermeant sulfhydryl reagents prevents diphtheria toxin cytotoxicity. More recently it was found that the same membrane function can be inhibited by bacitracin, an inhibitor of protein disulfide-isomerase (PDI), and by monoclonal antibodies against PDI, suggesting that PDI catalyzes a thiol-disulfide interchange between its thiols and the disulfides of membrane-bound macromolecules. We provide evidence that the same reductive process plays a role in the penetration of membrane-bound human immunodeficiency virus (HIV) and show that HIV infection of human lymphoid cells is markedly inhibited by the membrane-impermeant sulfhydryl blocker 5,5'-dithiobis(2-nitrobenzoic acid), by bacitracin, and by anti-PDI antibodies. The results imply that HIV and its target cell engage in a thiol-disulfide interchange mediated by PDI and that the reduction of critical disulfides in viral envelope glycoproteins may be the initial event that triggers conformational changes required for HIV entry and cell infection. These findings suggest additional approaches to impede cell infection by HIV.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abell B. A., Brown D. T. Sindbis virus membrane fusion is mediated by reduction of glycoprotein disulfide bridges at the cell surface. J Virol. 1993 Sep;67(9):5496–5501. doi: 10.1128/jvi.67.9.5496-5501.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anthony R. P., Paredes A. M., Brown D. T. Disulfide bonds are essential for the stability of the Sindbis virus envelope. Virology. 1992 Sep;190(1):330–336. doi: 10.1016/0042-6822(92)91219-k. [DOI] [PubMed] [Google Scholar]
- Callebaut C., Krust B., Jacotot E., Hovanessian A. G. T cell activation antigen, CD26, as a cofactor for entry of HIV in CD4+ cells. Science. 1993 Dec 24;262(5142):2045–2050. doi: 10.1126/science.7903479. [DOI] [PubMed] [Google Scholar]
- Capon D. J., Ward R. H. The CD4-gp120 interaction and AIDS pathogenesis. Annu Rev Immunol. 1991;9:649–678. doi: 10.1146/annurev.iy.09.040191.003245. [DOI] [PubMed] [Google Scholar]
- Chaix M. L., Chappey C., Couillin I., Rozenbaum W., Levy J. P., Saragosti S. Diversity of the V3 region of HIV in Paris, France. AIDS. 1993 Sep;7(9):1199–1204. doi: 10.1097/00002030-199309000-00008. [DOI] [PubMed] [Google Scholar]
- Clements G. J., Price-Jones M. J., Stephens P. E., Sutton C., Schulz T. F., Clapham P. R., McKeating J. A., McClure M. O., Thomson S., Marsh M. The V3 loops of the HIV-1 and HIV-2 surface glycoproteins contain proteolytic cleavage sites: a possible function in viral fusion? AIDS Res Hum Retroviruses. 1991 Jan;7(1):3–16. doi: 10.1089/aid.1991.7.3. [DOI] [PubMed] [Google Scholar]
- Edman J. C., Ellis L., Blacher R. W., Roth R. A., Rutter W. J. Sequence of protein disulphide isomerase and implications of its relationship to thioredoxin. Nature. 1985 Sep 19;317(6034):267–270. doi: 10.1038/317267a0. [DOI] [PubMed] [Google Scholar]
- Eiden L. E., Lifson J. D. HIV interactions with CD4: a continuum of conformations and consequences. Immunol Today. 1992 Jun;13(6):201–206. doi: 10.1016/0167-5699(92)90154-Y. [DOI] [PubMed] [Google Scholar]
- Feener E. P., Shen W. C., Ryser H. J. Cleavage of disulfide bonds in endocytosed macromolecules. A processing not associated with lysosomes or endosomes. J Biol Chem. 1990 Nov 5;265(31):18780–18785. [PubMed] [Google Scholar]
- Flynn D. C., Meyer W. J., Mackenzie J. M., Jr, Johnston R. E. A conformational change in Sindbis virus glycoproteins E1 and E2 is detected at the plasma membrane as a consequence of early virus-cell interaction. J Virol. 1990 Aug;64(8):3643–3653. doi: 10.1128/jvi.64.8.3643-3653.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedman R. B. Protein disulfide isomerase: multiple roles in the modification of nascent secretory proteins. Cell. 1989 Jun 30;57(7):1069–1072. doi: 10.1016/0092-8674(89)90043-3. [DOI] [PubMed] [Google Scholar]
- Hartshorn K. L., Vogt M. W., Chou T. C., Blumberg R. S., Byington R., Schooley R. T., Hirsch M. S. Synergistic inhibition of human immunodeficiency virus in vitro by azidothymidine and recombinant alpha A interferon. Antimicrob Agents Chemother. 1987 Feb;31(2):168–172. doi: 10.1128/aac.31.2.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson V. A., Barlow M. A., Merrill D. P., Chou T. C., Hirsch M. S. Three-drug synergistic inhibition of HIV-1 replication in vitro by zidovudine, recombinant soluble CD4, and recombinant interferon-alpha A. J Infect Dis. 1990 Jun;161(6):1059–1067. doi: 10.1093/infdis/161.6.1059. [DOI] [PubMed] [Google Scholar]
- Kaetzel C. S., Rao C. K., Lamm M. E. Protein disulphide-isomerase from human placenta and rat liver. Purification and immunological characterization with monoclonal antibodies. Biochem J. 1987 Jan 1;241(1):39–47. doi: 10.1042/bj2410039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kido H., Fukutomi A., Katunuma N. A novel membrane-bound serine esterase in human T4+ lymphocytes immunologically reactive with antibody inhibiting syncytia induced by HIV-1. Purification and characterization. J Biol Chem. 1990 Dec 15;265(35):21979–21985. [PubMed] [Google Scholar]
- Lekutis C., Olshevsky U., Furman C., Thali M., Sodroski J. Contribution of disulfide bonds in the carboxyl terminus of the human immunodeficiency virus type I gp120 glycoprotein to CD4 binding. J Acquir Immune Defic Syndr. 1992;5(1):78–81. [PubMed] [Google Scholar]
- Leonard C. K., Spellman M. W., Riddle L., Harris R. J., Thomas J. N., Gregory T. J. Assignment of intrachain disulfide bonds and characterization of potential glycosylation sites of the type 1 recombinant human immunodeficiency virus envelope glycoprotein (gp120) expressed in Chinese hamster ovary cells. J Biol Chem. 1990 Jun 25;265(18):10373–10382. [PubMed] [Google Scholar]
- Mandel R., Ryser H. J., Ghani F., Wu M., Peak D. Inhibition of a reductive function of the plasma membrane by bacitracin and antibodies against protein disulfide-isomerase. Proc Natl Acad Sci U S A. 1993 May 1;90(9):4112–4116. doi: 10.1073/pnas.90.9.4112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer W. J., Gidwitz S., Ayers V. K., Schoepp R. J., Johnston R. E. Conformational alteration of Sindbis virion glycoproteins induced by heat, reducing agents, or low pH. J Virol. 1992 Jun;66(6):3504–3513. doi: 10.1128/jvi.66.6.3504-3513.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milich L., Margolin B., Swanstrom R. V3 loop of the human immunodeficiency virus type 1 Env protein: interpreting sequence variability. J Virol. 1993 Sep;67(9):5623–5634. doi: 10.1128/jvi.67.9.5623-5634.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizunaga T., Katakura Y., Miura T., Maruyama Y. Purification and characterization of yeast protein disulfide isomerase. J Biochem. 1990 Nov;108(5):846–851. doi: 10.1093/oxfordjournals.jbchem.a123291. [DOI] [PubMed] [Google Scholar]
- Moore J. P., Thali M., Jameson B. A., Vignaux F., Lewis G. K., Poon S. W., Charles M., Fung M. S., Sun B., Durda P. J. Immunochemical analysis of the gp120 surface glycoprotein of human immunodeficiency virus type 1: probing the structure of the C4 and V4 domains and the interaction of the C4 domain with the V3 loop. J Virol. 1993 Aug;67(8):4785–4796. doi: 10.1128/jvi.67.8.4785-4796.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noiva R., Freedman R. B., Lennarz W. J. Peptide binding to protein disulfide isomerase occurs at a site distinct from the active sites. J Biol Chem. 1993 Sep 15;268(26):19210–19217. [PubMed] [Google Scholar]
- Potts K. E., Kalish M. L., Lott T., Orloff G., Luo C. C., Bernard M. A., Alves C. B., Badaro R., Suleiman J., Ferreira O. Genetic heterogeneity of the V3 region of the HIV-1 envelope glycoprotein in Brazil. Brazilian Collaborative AIDS Research Group. AIDS. 1993 Sep;7(9):1191–1197. doi: 10.1097/00002030-199309000-00007. [DOI] [PubMed] [Google Scholar]
- Roth R. A. Bacitracin: an inhibitor of the insulin degrading activity of glutathione-insulin transhydrogenase. Biochem Biophys Res Commun. 1981 Jan 30;98(2):431–438. doi: 10.1016/0006-291x(81)90858-5. [DOI] [PubMed] [Google Scholar]
- Ryser H. J., Mandel R., Ghani F. Cell surface sulfhydryls are required for the cytotoxicity of diphtheria toxin but not of ricin in Chinese hamster ovary cells. J Biol Chem. 1991 Oct 5;266(28):18439–18442. [PubMed] [Google Scholar]
- Sattentau Q. J., Moore J. P. Conformational changes induced in the human immunodeficiency virus envelope glycoprotein by soluble CD4 binding. J Exp Med. 1991 Aug 1;174(2):407–415. doi: 10.1084/jem.174.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Syu W. J., Huang J. H., Essex M., Lee T. H. The N-terminal region of the human immunodeficiency virus envelope glycoprotein gp120 contains potential binding sites for CD4. Proc Natl Acad Sci U S A. 1990 May;87(10):3695–3699. doi: 10.1073/pnas.87.10.3695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tschachler E., Buchow H., Gallo R. C., Reitz M. S., Jr Functional contribution of cysteine residues to the human immunodeficiency virus type 1 envelope. J Virol. 1990 May;64(5):2250–2259. doi: 10.1128/jvi.64.5.2250-2259.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varandani P. T., Raveed D., Nafz M. A. Insulin degradation. XXIII. Distribution of glutathione-insulin transhydrogenase in isolated rat hepatocytes as studied by immuno-ferritin and electron microscopy. Biochim Biophys Acta. 1978 Jan 18;538(2):343–353. doi: 10.1016/0304-4165(78)90362-8. [DOI] [PubMed] [Google Scholar]
- Werner A., Levy J. A. Human immunodeficiency virus type 1 envelope gp120 is cleaved after incubation with recombinant soluble CD4. J Virol. 1993 May;67(5):2566–2574. doi: 10.1128/jvi.67.5.2566-2574.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyatt R., Thali M., Tilley S., Pinter A., Posner M., Ho D., Robinson J., Sodroski J. Relationship of the human immunodeficiency virus type 1 gp120 third variable loop to a component of the CD4 binding site in the fourth conserved region. J Virol. 1992 Dec;66(12):6997–7004. doi: 10.1128/jvi.66.12.6997-7004.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimori T., Semba T., Takemoto H., Akagi S., Yamamoto A., Tashiro Y. Protein disulfide-isomerase in rat exocrine pancreatic cells is exported from the endoplasmic reticulum despite possessing the retention signal. J Biol Chem. 1990 Sep 15;265(26):15984–15990. [PubMed] [Google Scholar]



