Abstract
Background and objectives
Steroid-resistant nephrotic syndrome is a rare kidney disease involving either immune-mediated or genetic alterations of podocyte structure and function. The rare nature, heterogeneity, and slow evolution of the disorder are major obstacles to systematic genotype-phenotype, intervention, and outcome studies, hampering the development of evidence-based diagnostic and therapeutic concepts. To overcome these limitations, the PodoNet Consortium has created an international registry for congenital nephrotic syndrome and childhood-onset steroid-resistant nephrotic syndrome.
Design, setting, participants, & measurements
Since August of 2009, clinical, biochemical, genetic, and histopathologic information was collected both retrospectively and prospectively from 1655 patients with childhood-onset steroid-resistant nephrotic syndrome, congenital nephrotic syndrome, or persistent subnephrotic proteinuria of likely genetic origin at 67 centers in 21 countries through an online portal.
Results
Steroid-resistant nephrotic syndrome manifested in the first 5 years of life in 64% of the patients. Congenital nephrotic syndrome accounted for 6% of all patients. Extrarenal abnormalities were reported in 17% of patients. The most common histopathologic diagnoses were FSGS (56%), minimal change nephropathy (21%), and mesangioproliferative GN (12%). Mutation screening was performed in 1174 patients, and a genetic disease cause was identified in 23.6% of the screened patients. Among 14 genes with reported mutations, abnormalities in NPHS2 (n=138), WT1 (n=48), and NPHS1 (n=41) were most commonly identified. The proportion of patients with a genetic disease cause decreased with increasing manifestation age: from 66% in congenital nephrotic syndrome to 15%–16% in schoolchildren and adolescents. Among various intensified immunosuppressive therapy protocols, calcineurin inhibitors and rituximab yielded consistently high response rates, with 40%–45% of patients achieving complete remission. Confirmation of a genetic diagnosis but not the histopathologic disease type was strongly predictive of intensified immunosuppressive therapy responsiveness. Post-transplant disease recurrence was noted in 25.8% of patients without compared with 4.5% (n=4) of patients with a genetic diagnosis.
Conclusions
The PodoNet cohort may serve as a source of reference for future clinical and genetic research in this rare but significant kidney disease.
Keywords: children, podocytopathies, nephrin, podocin, WT1
Introduction
Approximately 12%–15% of children with idiopathic nephrotic syndrome do not respond to oral steroid therapy (1,2). Steroid-resistant nephrotic syndrome (SRNS) is a challenging clinical condition with highly variable outcomes, and 50% of children progress to ESRD within 15 years (3,4). Although in some patients, temporary or persistent remission is achieved by intensified immunosuppressive therapies (IITs), others exhibit a multidrug-resistant phenotype. Historically, diagnostic evaluation and prognostic classification relied largely on histopathologic assessment. In recent years, abnormalities in a growing number of genes essential for podocyte development, structure, and function have been identified in patients with congenital nephrotic syndrome (CNS) and SRNS (5). The ongoing discovery of genetic podocytopathies is about to redefine the physiopathologic understanding, diagnostic assessment, prognostic judgment, and therapeutic approaches in childhood-onset SRNS. However, the development of evidence-based management algorithms has been hampered by the low incidence of SRNS, which is estimated at 2–4 per million person-years (1).
To overcome the limitations imposed by the rarity of the disorder, the PodoNet Consortium has created an international registry for CNS and childhood-onset SRNS. Over the past 5 years, comprehensive clinical, medication-related, histopathologic, and genetic information from >1600 children with the disorder has been collected in 21 countries. The detailed description of the PodoNet cohort presented here aims to provide a source of reference for future clinical and genetic research in CNS and SRNS.
Materials and Methods
Registry Description
The PodoNet Consortium was formed in 2008 by research groups from Heidelberg, Paris, Rome, Bergamo, Genova, and Ankara. The PodoNet project (www.podonet.org) encompasses clinical, genetic, and experimental research into hereditary podocyte disorders. The clinical activities encompass a web-based international clinical registry and a central biobank for SRNS. To date, investigators from 67 clinical units in 21 countries of Europe, the Middle East, and Latin America have joined the PodoNet Registry Consortium. The registry study protocol was approved by the local institutional review boards/ethics committees, and written informed consent is obtained from the families. Data protection is ensured by pseudonymized data input.
The registry accepts patients under current medical care with childhood-onset (age ≤20 years old) SRNS, CNS, or persistent subnephrotic proteinuria with likely genetic disease.
The items collected by the registry are given in Supplemental Appendix. Retrospective and prospective information is collected. The regular data entries are discontinued when RRT is initiated, with a follow-up entry providing information about kidney transplantation and occurrence, treatment, and outcome of post-transplant disease recurrence.
For incident patients, a uniform set of diagnostic and therapeutic algorithms is proposed (www.podonet.org/opencms/opencms/podonet/podonet_en/Algorithms/index.html), but adherence to the protocols is not mandatory for inclusion of patients in the registry.
In the prospective part of the registry, serum and urine samples are collected at 6-month intervals and stored in a central biorepository. Also, DNA samples are collected from all available and consenting patients and any affected relatives. Central genetic screening has been offered for patients with lacking or incomplete screening results.
Definitions
SRNS was defined by persistent proteinuria after 4 weeks of oral steroid therapy (prednisone at 60 mg/m2 per day). Subnephrotic proteinuria was defined as 24-hour protein excretion >100 mg but <1 g/m2 per day, urine protein-to-creatinine ratio (UPCr) of 0.2 (0.6 if age <2 years old) to 2 mg/mg, or dipstick proteinuria of 1+. Nephrotic-range proteinuria was defined by 24-hour protein excretion ≥1 g/m2 per day, UPCr>2 mg/mg, or dipstick ≥2+. Complete remission of proteinuria was defined by the attainment of the following criteria during therapy: 24-hour protein excretion <100 mg/m2 per d, UPCr<0.2 mg/mg, dipstick of 0 or (+), or in absence of proteinuria information, serum albumin >3.5 g/dl. Partial remission was defined by 24-hour protein excretion of 0.1–1 g/m2/d, UPCr=0.2–2 mg/mg, dipstick of 1+, and if available, serum albumin >3.0 g/dl or if no proteinuria information was available, serum albumin of 3.0–3.5 g/dl.
Throughout the manuscript, percentages are given relative to all patients with available information regarding a particular topic.
Results
Cohort Structure
From August of 2009 to August of 2014, 1655 patients were registered who were ages 0–20 years old at disease onset. Among these, 261 (15.8%) patients were living in Italy, 227 (13.7%) patients were living in Germany, 226 (13.7%) patients were living in Poland, 189 (11.4%) patients were living in other European countries, 491 (29.7%) patients were living in Turkey or Georgia, 153 (9.2%) patients were living in the Middle East (Syria, Lebanon, Iran, and the United Arab Emirates), and 108 (6.5%) patients were living in Latin America (Chile and Colombia). Parental ethnicity (reported in 1274 families) was Caucasian in 1151 (90.3%) patients, Hispanic in 93 (7.3%) patients, Indian in nine (0.7%) patients, east Asian in five (0.4%) patients, African in three (0.2%) patients, Native American in two (0.2%) patients, and mixed in 11 (0.9%) patients. Among the patients with available respective information, parental consanguinity was reported in 28.6%, and familial disease occurrence was reported in 25.6%. Consanguinity and familial disease occurrence were most common in the Middle Eastern countries (Supplemental Table 1).
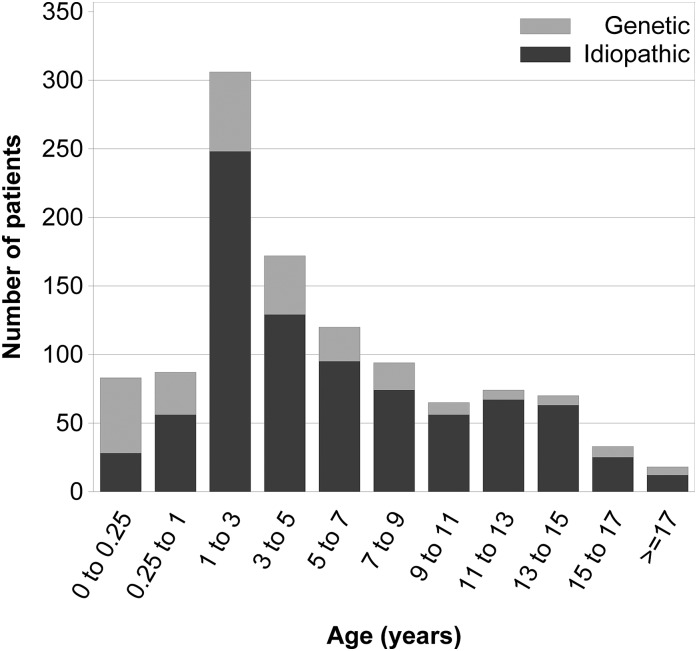
In 1544 patients, the age at first disease manifestation was reported (Figure 1). Among these, 207 (13.4%) were incident patients with first disease manifestation within 6 months before enrolment. Of the prevalent patients, 329 (24.6%) patients had been followed for 0.5–2 years, 345 (25.8%) patients had been followed for 2–5 years, 320 (23.9%) patients had been followed for 5–10 years, and 343 (25.6%) patients had been followed for >10 years at the time of enrolment.
Figure 1.
Age at first disease manifestation in children with and without an identified genetic cause of steroid-resistant nephrotic syndrome.
Information Content
Clinical and biochemical information from the time of first disease manifestation was available in 1310 (79.2%) patients. In total, 11,934 clinical follow-up entries were made for 1525 (92.1%) patients. Medication data were entered for 1283 (77.5%) patients and comprised 26,406 individual start, discontinuation, or dose change entries. Renal biopsy information was available from 1368 (82.7%) patients; the results of two sequential biopsies were recorded in 145 patients, and the results of more than two biopsies were recorded in 26 patients. Results of genetic screening were reported for 1174 (70.9%) patients, and information on family medical history was available from 1014 study participants (61.3%). The PodoNet biobank contains DNA samples from 950 patients, one or several serum samples from 472 patients, and urine samples from 450 patients.
First Disease Manifestation
The patient characteristics according to age at first disease manifestation are given in Table 1. Almost three quarters of the cohort presented between 1–11 years of age, whereas 6% were categorized as congenital, 7% were categorized as early infantile (onset age of 3–12 months), and 16% were categorized as adolescent-onset nephrotic syndrome (onset age ≥12 years old). Among the children who underwent mutation screening, genetic disease detection rate was markedly higher (66%) in patients with CNS than in children first presenting at age 6 years old and older (16%). Regarding clinical and biochemical characteristics, hypoalbuminemia at time of diagnosis was more common in CNS, whereas hypertension was more prevalent in adolescents. Nephrotic-range proteinuria was not yet present in 8.7% of the children at the time of diagnosis, but most of these patients progressed to full nephrotic syndrome during follow-up.
Table 1.
Key characteristics at time of diagnosis and response to intensified immunosuppressive therapy stratified by age at disease onset
| Age at Disease Onset | No. of Patients (%) | Genetic Cause Identified N (%) | Nephrotic-Range Proteinuria (%) | Hypertension (%) | Serum Albumin (g/dl) | eGFRa (ml/min per 1.73 m2) | Response to IIT | ESRD at Last Observation (%) | Post-Tx Recurrence N/NTx (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Partial Remission N/Ninfo (%) | Complete Remission N/Ninfo (%) | |||||||||
| <3 mo | 98 (6.3) | 55 (66.3) | 92.7 | 10.2 | 1.7 (0.9) | 62 (41–80) | 2/11 (18.2) | 3/11 (27.3) | 60.9 | 2/34 (5.9) |
| 3–12 mo | 106 (6.9) | 31 (35.6) | 89.7 | 17.0 | 2.1 (1.0) | 89 (53–132) | 3/33 (9.1) | 16/33 (48.5) | 37.8 | 0/21 (0.0) |
| 1–5 yr | 786 (50.9) | 119 (21.8) | 93.3 | 14.3 | 2.0 (0.9) | 105 (78–147) | 97/505 (19.2) | 231/505 (45.7) | 21.5 | 16/86 (18.6) |
| 6–11 yr | 350 (22.7) | 40 (15.6) | 88.1 | 16.6 | 2.6 (1.0) | 105 (76–145) | 49/208 (23.6) | 73/208 (35.1) | 27.2 | 11/36 (30.6) |
| ≥12 yr | 204 (13.2) | 24 (16.1) | 89.8 | 27.9 | 2.6 (1.0) | 93 (64–129) | 29/128 (22.7) | 42/128 (32.8) | 28.6 | 3/26 (11.5) |
Data are given as number (%), mean (SD), or median (interquartile range). IIT, immunosuppressive therapy; Ninfo, number of patients with reported information; Tx, transplant; NTx, number of patients who received a kidney transplant.
Median (interquartile range)
Follow-Up Information
The median (interquartile range [IQR]) duration of follow-up from first manifestation was 3.7 (1.4–7.2) years. At the most recent observation, 61.3% of patients were still without RRT (CKD stage 1: 411 [60.2%], stage 2: 178 [26.1%], stage 3: 55 [8.1%], stage 4: 22 [3.2%], and stage 5: 17 [2.5%]), 11.7% of patients required dialysis, 14.2% of patients had received kidney allografts, 2.3% of patients were deceased, and 10.6% of patients were lost of follow-up. Among the patients without RRT, 33% were in full remission and 22.5% were in partial remission at last observation. Among the patients with full or partial remission at last observation, 66.9% had normal kidney function, 26.4% were in CKD stage 2, 5.1% in CKD stage 3, and 1.6% in CKD stages 4 or 5.
Histopathologic Findings
First kidney biopsies were performed in 1368 patients at a median (IQR) of 2 (1–8) months after first disease manifestation. The most common histopathologic diagnosis was FSGS (56.0%) followed by minimal change nephropathy (MCN; 21.1%), mesangioproliferative GN (MesPGN; 12.4%), and diffuse mesangial sclerosis (DMS; 2.9%). Information on average age, eGFR, and prevalence of hypertension at the time of diagnosis as well as prevalence of genetic disease according to histopathologic diagnosis is given in Table 2. Repeat biopsies were performed in 171 (12.5%) patients at a median (IQR) of 31 (17–54) months after the initial biopsy. The diagnosis changed in 54% of patients (in most cases from MCN to FSGS [26 of 47] and from MesPGN to FSGS [16 of 33]).
Table 2.
Key characteristics according to histopathologic diagnosis sorted by patient age at time of biopsy
| Histopathologic Diagnosis | No. of Patients N (%) | Genetic cause N/Ninfo (%) | At First Biopsy | IIT Responsiveness | ESRD at Last Observation (%) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Agea (yr) | Serum Albumin (g/dl) | Hypertension (%) | eGFRa (ml/min per 1.73 m2) | Partial Remission N/Ninfo (%) | Complete Remission N/Ninfo (%) | ||||
| CNS | 8 (0.6) | 3/7 (43.0) | 0.2 (0.2–0.7) | 1.4 (0.4) | 0.0 | 56 (29–98) | 0/1 (0.0) | 0/1 (0.0) | 57.1 |
| DMS | 39 (2.9) | 22/35 (62.9) | 1.3 (0.6–2.3) | 2.0 (0.8) | 26.3 | 59 (37–97) | 0/10 (0.0) | 1/10 (10.0) | 72.2 |
| MCN | 283 (21.1) | 24/195 (12.3) | 4.2 (2.4–7.6) | 2.5 (1.0) | 15.8 | 108 (86–155) | 41/202 (20.3) | 103/202 (51.0) | 12.9 |
| MesPGN | 166 (12.4) | 23/123 (18.7) | 4.4 (2.4–8.4) | 2.4 (1.0) | 15.1 | 118 (84–162) | 26/101 (25.7) | 41/101 (40.6) | 18.9 |
| FSGS | 752 (56.0) | 122/555 (22.0) | 6.2 (3.0–11.0) | 2.7 (1.0) | 18.9 | 97 (68–142) | 93/469 (19.8) | 183/469 (39.0) | 30.9 |
| GGS | 17 (1.3) | 4/12 (33.3) | 6.6 (3.4–9.1) | 2.8 (0.8) | 17.7 | 17 (10–85) | 0/3 (0.0) | 1/3 (33.3) | 68.8 |
| MN | 11 (0.8) | 0/11 (0.0) | 7.0 (2.7–13.6) | 2.4 (0.9) | 27.3 | 116 (103–138) | 3/10 (30.0) | 5/10 (50.0) | 9.1 |
| MPGN | 35 (2.6) | 4/25 (16.0) | 10.5 (5.8–11.9) | 2.9 (0.8) | 22.9 | 112 (61–148) | 4/16 (25.0) | 6/16 (37.5) | 23.5 |
| Other | 31 (2.3) | 9/27 (33.3) | 8.8 (3.2–11.7) | 2.7 (1.1) | 6.7 | 45 (29–98) | 4/16 (25.0) | 6/16 (37.5) | 24.1 |
Information on genetic disease, features at first biopsy, IIT responsiveness, and fraction of ESRD at last observations refers to diagnosis by initial biopsy. Data are given as number (%), mean (SD), or median (interquartile range). CNS, Finnish-type congenital nephrotic syndrome; DMS, diffuse mesangial sclerosis; MCN, minimal change nephropathy; MesPGN, mesangioproliferative GN; GGS, global glomerulosclerosis; MN, membranous nephropathy; MPGN, membranoproliferative GN; Ninfo, number of patients with reported information.
Median (interquartile range).
Extrarenal Symptoms
In 17.3% of the patients, one or several extrarenal abnormalities were reported (Table 3). Alterations of the morphology and/or function of the central nervous system (brain anomaly, microcephaly, and/or mental retardation) were the most common extrarenal abnormalities, affecting 5.3% of all patients. Other features included symptoms suggestive of WT1 disease (sex reversal/urogenital abnormalities and cancer), impaired mitochondrial energy metabolism (myopathy, cardiomyopathy, and impaired hearing), Pierson syndrome (impaired vision), and Schimke syndrome (osteodysplasia).
Table 3.
Extrarenal abnormalities
| Type of Extrarenal Abnormality | N | % |
|---|---|---|
| Patients without reported extrarenal abnormalities | 1368 | 82.7 |
| Mental retardation | 65 | 3.9 |
| Anomalies of centralnervous system | 42 | 2.5 |
| Microcephaly | 17 | 1.0 |
| Visual impairment | 32 | 1.9 |
| Hearing disorder | 25 | 1.5 |
| Anomalies of peripheral nervous system | 7 | 0.4 |
| Myopathy | 13 | 0.8 |
| Cardiomyopathy | 6 | 0.4 |
| Urogenital abnormalities | 33 | 2.0 |
| Impaired sex differentiation | 16 | 1.0 |
| Short stature | 84 | 5.1 |
| Facial dysmorphism | 37 | 2.2 |
| Spondyloepiphyseal dysplasia | 8 | 0.5 |
| Polydactyly | 5 | 0.3 |
| Nail patella syndrome | 2 | 0.1 |
| Cardiac structural disorder | 36 | 2.2 |
| Malignant disorder | 21 | 1.3 |
| Hematologic disorders | 7 | 0.4 |
| Diabetes mellitus | 7 | 0.4 |
| Other endocrine abnormalities | 23 | 1.4 |
| Skin abnormalities | 13 | 0.8 |
| Abnormalities of the gastrointestinal tract | 5 | 0.3 |
| Autoimmune disorder | 9 | 0.5 |
| Connatal cytomegaly virus | 9 | 0.5 |
| Hepatitis B | 6 | 0.4 |
| Hepatitis C | 10 | 0.6 |
Mutation Screening Results
In total, 3037 individual gene screens were performed in 1174 of 1655 study participants (70.9%). The extent of genetic screening varied among countries. Among 11 countries contributing at least 30 patients to the registry, screening prevalence was highest in Chile (91% of patients), Syria (84%), and Germany (80%) and lowest in Colombia (26%) and Iran (21%).
The most commonly screened genes were NPHS2 and WT1 (performed in 93% and 77% of screened patients, respectively), whereas other podocyte genes were screened more selectively guided by age, histopathology, and/or syndromic features or as part of joint research projects. In addition, 27 patients recently underwent comprehensive screening using a next generation sequencing panel of 31 podocyte genes. Genetic diagnoses were established in 277 (23.6%) patients; among these, mutations in NPHS2 accounted for 49.8% of patients, mutations in WT1 accounted for 17.3% of patients, mutations in NPHS1 accounted for 14.8% of patients, mutations in SMARCAL1 accounted for 4.3% of patients, and mutations in PLCE1 accounted for 3.6% of patients. The remaining 10% of patients were attributable to variants in nine different genes (Table 4).
Table 4.
Results of genetic screening studies
| Gene | Patients Screened | Patients with Causative Mutation | Screened Positive (%) |
|---|---|---|---|
| NPHS2 | 1088 | 138 | 12.7 |
| WT1 | 902 | 48 | 5.3 |
| NPHS1 | 208 | 41 | 19.7 |
| SMARCAL1 | 68 | 12 | 17.6 |
| PLCE1 | 75 | 10 | 13.3 |
| PTPRO | 45 | 6 | 13.3 |
| LAMB2 | 84 | 5 | 6.0 |
| INF2 | 112 | 4 | 3.6 |
| COQ6 | 30 | 3 | 10.0 |
| MYO1E | 48 | 2 | 4.2 |
| TRPC6 | 96 | 1 | 1.0 |
| COQ2 | 56 | 1 | 1.8 |
| LMX1B | 27 | 1 | 3.7 |
| ADCK4 | 27 | 1 | 3.7 |
| PDSS2 | 56 | 0 | 0.0 |
| ACTN4 | 59 | 0 | 0.0 |
| CD2AP | 56 | 0 | 0.0 |
| All | 1174 | 277 | 23.6 |
Pharmacologic Therapy
Among 1283 patients with available information on pharmacologic therapy, 42.6% received steroid therapy, 3.5% received cyclophosphamide pulse therapy, 21.2% received oral cyclophosphamide, 66.3% received ciclosporin A, 12.2% received tacrolimus, 24.9% received mycophenolate-mofetil (MMF), and 6.6% received rituximab at any time during the observation period. Renin angiotensin system (RAS) antagonists were applied in 76.1% of patients (angiotensin-converting enzyme inhibitor [72.3%] and/or angiotensin type 1 receptor blocker [33.7%]). In 1234 patients, 5240 drug treatment periods applied for at least 3 weeks (excluding intravenous therapies) were documented.
The distribution of treatment protocols, duration of administration, and treatment responses are summarized in Table 5. Calcineurin inhibitor (CNI)–based therapy showed the highest response rates, with approximately two thirds of patients achieving partial or complete proteinuria remission. Steroid pulse therapy and cyclophosphamid-based protocols generally showed poor efficacy and did not achieve better results than antiproteinuric treatment with RAS inhibitors, which were frequently coadministered with IIT.
Table 5.
Medication protocols applied in 1234 patients
| Medication Protocol | No. of Patients | No. of Periods | Cotreated with RAS Antagonists (%) | Cotreated with Oral Steroids (%) | Treatment Duration (mo) | Response to Therapy | |
|---|---|---|---|---|---|---|---|
| Partial Remission N/Ninfo (%) | Complete Remission N/Ninfo (%) | ||||||
| RAS antagonists only | 623 | 950 | — | 63.3 | 5.2 (1.9–17.1) | 109/531 (20.5) | 141/531 (26.6) |
| Oral steroids only | 722 | 1119 | — | — | 2.0 (1.4–4.7) | 53/567 (9.3) | 62/567 (10.9) |
| Steroid pulses | 541 | 1114 | 51.0 | 72.4 | 0.1 (0.1–0.9) | 42/279 (15.1) | 25/279 (9.0) |
| CPH (oral or pulse) | 273 | 336 | 39.0 | 92.0 | 2.5 (1.6–3.0) | 11/131 (8.4) | 22/131 (16.8) |
| CNI | 806 | 1210 | 70.7 | 89.5 | 8.3 (3.0–23.0) | 153/707 (21.6) | 311/707 (44.0) |
| MMF | 177 | 219 | 80.4 | 71.7 | 6.7 (3.0–14.9) | 19/148 (12.8) | 57/148 (38.5) |
| CNI + MMF | 137 | 167 | 81.4 | 76.0 | 7.7 (3.0–23.9) | 26/119 (21.8) | 57/119 (47.9) |
| Rituximab | 79 | 91 | 87.9 | 76.9 | 0.0 (0.0–4.6) | 10/66 (15.2) | 29/66 (43.9) |
Treatment duration is given as median and interquartile range. Renin angiotensin system (RAS) antagonists are angiotensin-converting enzyme inhibitor and/or angiotensin type 1 receptor blocker. CPH, cyclophosphamide; CNI, calcineurin inhibitor (ciclosporin A or tacrolimus); MMF, mycophenolate-mofetil.
The use of steroid therapy and nonsteroidal IIT increased with age at first manifestation but was not limited to patients beyond a certain threshold age. Steroid therapy (oral and/or intravenous pulses) was reportedly administered in 28.6% of the children with CNS and 59.4% of those with disease manifestation at age 3–12 months old; 24.5% of the children with CNS and 42.5% of those with onset at 3–12 months also received nonsteroidal IIT (CNI, MMF, cyclophosphamide, or rituximab). IIT response rates according to age at first manifestation are given in Table 1.
The initial histopathologic diagnosis did not predict IIT responsiveness in this SRNS cohort. Remission rates differed only marginally between MCN, MesPGN, and FSGS (Table 2).
Among 156 incident patients since the start of the registry in 2009 with disease onset at >3 months, oral steroid resistance, and documented follow-up pharmacotherapy, pulse steroids were initially administered to ascertain steroid resistance in 63 (40.4%) patients, and 97 (62.2%) patients received CNI therapy as first nonsteroidal therapy (39 with and 58 without prior steroid pulses) as recommended in the PodoNet consensus treatment algorithm; seven (4.5%) patients received other nonsteroidal IITs, 21 (13.5%) patients received RAS only, and seven (4.5%) patients were continued on oral steroids only.
In 115 (42%) of the patients in whom a genetic diagnosis was established at some time during follow-up, IIT protocols were applied; in 12 patients, complete proteinuria remission was observed during IIT.
Late Outcomes
Children with CNS and early infantile nephrotic syndrome were more likely to have ESRD at the last documented observation than older children (Table 1). With respect to histopathologic diagnosis, patients with DMS and global glomerulosclerosis had the highest likelihood of ESRD (Table 2). Patients with FSGS were more likely to progress than those with MCN.
In total, 217 patients underwent kidney transplantation. Proteinuria recurrence was reported in 33 (15.2%) of these patients. The risk of disease recurrence increased with age at first disease manifestation (Table 1).
The histopathologic diagnoses at last (native) kidney biopsy in patients with post–transplant disease recurrence were FSGS in 22 (68.8%) patients, MesPGN in four (12.5%) patients, global glomerulosclerosis in two (6.3%) patients, MCN in two (6.3%) patients, membranoproliferative GN in one (3.1%) patient, DMS in one (3.1%) patient, and unknown in one patient.
Although post–transplant disease recurrence occurred in 28.5% of patients without a genetic diagnosis, recurrence was noted in four (4.5%) patients of those diagnosed with genetic disease. In all of these patients, NPHS2 mutations had been identified.
Discussion
In this work, we present essential reference information for pediatric SRNS. With >1600 patients enrolled in 67 centers on three continents, the PodoNet Registry cohort represents, by far, the largest collection of clinicopathologic and genetic information assembled to date.
The registry provides important information regarding the distribution of clinical and histopathologic phenotypes in childhood SRNS. We also compiled valuable information about extrarenal disease manifestations. Neurologic abnormalities were most commonly reported followed by a wide range of anomalies concerning various organs and tissues. Altogether, syndromic phenotypes accounted only for a small fraction of patients, and SRNS occurred as an isolated kidney disease in 83% of the patients in the cohort.
Historically, renal histopathology has been used as a key criterion for diagnostic and prognostic categorization in children with SRNS. Here, we provide demographic, clinical, and genetic information on the individual histopathologic entities in the pediatric SRNS population. The three major histologic diagnoses (FSGS, MCN, and MesPGN) presented with similar degrees of hypoalbuminemia and comparable prevalence of hypertension. Patients with FSGS were slightly older and more often had reduced eGFR at the time of diagnosis.
FSGS was observed almost two times as often as MCN and MesPGN together. The predominance of FSGS contrasts slightly with previous single-center surveys in south Asian and African populations, where MCN was equally or even more common than FSGS (6–8). This variation might be related to differences in ethnic composition, biopsy indication policies, or periods of observation analyzed. A trend toward an increasing incidence of FSGS has been noted in several studies (9–11).
The other, much rarer histopathologic entities DMS, membranoproliferative GN, and membranous nephropathy typically presented with milder hypoalbuminemia and a slightly higher prevalence of hypertension than MCN, MesPGN, and FSGS. Patients with DMS usually manifested at earlier age with established renal failure.
Abnormalities in podocyte genes are considered likely in children with multidrug-resistant nephrotic syndrome (12,13). In 21 countries participating in PodoNet, genetic screening was performed in more than two thirds of the affected children. Of these, almost one quarter of the patients tested were screened positive for one of the monogenic disorders known to cause SRNS. In agreement with published numbers (14), the detection rate was highest in children with CNS and significantly lower with increasing age at first disease manifestation during the first 6 years of life. However, even among patients first presenting at adolescent age, a genetic disease cause could be established in more than one of seven patients. A detailed analysis of the genetic findings in the adolescent segment of the cohort has recently been published (15). The observed gene detection rate is most likely an underestimate, because until recently, the high cost and processing times of conventional Sanger sequencing allowed for screening of only a few genes selected according to age at disease onset, histopathology findings, and in familial cases, mode of inheritance. Also, within the lifetime of the registry, several new genes have been identified (5)—some as part of the activities of the PodoNet consortium (16,17). Our findings indicate that SRNS may be explained by rare abnormalities in a large number of genes. The advent of next generation sequencing will soon allow us to routinely screen all genes associated with SRNS. The detection rates per gene and per patient published in this report, which were obtained mainly by classic screening strategies and conventional technologies, may serve as a benchmark for future efforts to establish genetic diagnoses in patients with SRNS.
Another noteworthy aspect of this study is the association of genetic disease causes with histopathologic diagnoses. Our findings underline the limited value of kidney biopsies in distinguishing genetic from nongenetic disease etiologies. Genetic abnormalities were found in 22% of patients with FSGS, 19% of patients with MesPGN, and 12% of patients with MCN. Close associations with specific genetic disorders were limited to DMS (WT1 and PLCE1 nephropathies) and CNS (NPHS1 disease).
Detailed information on pharmacotherapy is available from >1200 children in the registry. The wide range of reported monotherapies and combined therapies reflects the complexity of SRNS management with frequent medication changes because of lack of efficiency or—observed or anticipated—adverse effects. Moreover, significant variation was noted regarding first- and second-line drug choices across the participating centers and countries, and treatment preferences seemed to change with time. Among the patients diagnosed with SRNS since the start of the PodoNet Registry, 40% adhered to the internal consensus recommendation to ascertain steroid resistance by intravenous steroid pulse therapy, and 62% used CNI as first-choice second-line IIT. Remarkably, steroid therapy and even IIT protocols were applied in some 25% of children presenting with CNS. These insights illustrate the large current variability of global clinical practice patterns.
Our preliminary analysis of proteinuria response patterns with different IIT protocols allows several tentative conclusions. First, steroid pulses and cyclophosphamide are of very limited, if any, efficacy in children with SRNS. Second, 40%–50% of patients with SRNS seem to respond to CNI by achieving complete remission of proteinuria. Third, rituximab may be an equally effective therapeutic option as CNI. Fourth, MMF may be less efficacious than CNI and rituximab in SRNS. Fifth, nonspecific antiproteinuric therapy by RAS inhibition is associated with complete remission of proteinuria in some 25% and partial remission in another 20% of patients with SRNS, an effect to be aware of in the evaluation of treatment effects and account for in the design of future pharmacotherapeutic trials in this condition.
These main findings are well in line with the existing body of evidence derived from observational studies and clinical trials (18–26) and provide a valuable reference framework for future clinical research. More detailed analyses of intervention-response patterns, relapse rates during and after discontinuation of treatment, adverse effect patterns, etc. are beyond the scope of this report but will be performed.
Whereas proteinuria was multidrug resistant in the vast majority of genetic cases, a few patients with genetic diagnoses apparently exhibited responsiveness to IIT. Although beyond the scope of this study, these peculiar cases deserve a detailed analysis of the nature of the genetic abnormalities diagnosed and the pharmacotherapies applied.
A diagnostically and prognostically relevant feature of SRNS is post-transplant disease recurrence, which was reported in 15% of >200 allograft recipients in the registry. Proteinuria recurrence occurred almost exclusively in patients without a genetic diagnosis; four patients with apparent recurrence despite genetic disease likewise will require additional evaluation.
Finally, our study underlines the clinical usefulness of genetic testing in children with SRNS. Establishing a genetic diagnosis is far superior to histopathologic disease classification in predicting IIT responsiveness and post-transplant disease recurrence in patients with SRNS. However, because hereditary forms of SRNS encompass a large number of genes, it will take the routine use of the emerging next-generation sequencing technologies for simultaneous assessment of all known podocytopathy genes to materialize the full potential of genetic screening.
Disclosures
None.
Supplementary Material
Acknowledgments
We acknowledge the continued support of patient families and medical staff at all participating centers.
The PodoNet project has received financial support from E-Rare (German Ministry of Education and Research), European Union 7th Framework Programme (EURenOmics) Grant 2012-305608, Chilean Research Agency (Fondecyt) Grant 11090045, Polish Ministry of Science and Education Grant N402631840, and German Research Foundation Grant Scha 477/11-1. F.O. was supported by Scientific and Technological Research Council of Turkey Grant 108S417 and Hacettepe University Infrastructure Project Grants 06A101008 and 011A101003.
PodoNet Collaborators: Chile: Marta Azocar, Hospital Luis Calvo Mackenna-Facultad de Chile, Santiago; Lily Quiroz, Roberto del Rio Childrens Hospital, Santiago; Colombia: Lina Maria Serna Higuita, Hospital Pablo Toban Uriba, Medellín; Czech Republic: Jiří Dušek, University Hospital Motol, Prague; France: Bruno Ranchin, Centre de Reference des Maladies Renales Rare/University Hospital, Lyon; Michel Fischbach, University Children's Hospital, Strasbourg; Georgia: Tinatin Davitaia, M. Iashvili Children's Central Hospital, Tbilisi; Germany: Jutta Gellermann, Charité University Hospital, Berlin; Jun Oh, University Children's Hospital, Hamburg; Anette Melk, Medical University Children's Hospital, Hannover; Franz Schaefer, Heidelberg University Center for Pediatrics and Adolescent Medicine, Heidelberg; Marianne Wigger, University Children's Hospital, Rostock; Greece: Fotios Papachristou, Hippokration General Hospital, Aristotle University, Thessaloniki; Hungary: Peter Sallay, Semmelweis University Hospital, Budapest; Iran: Alaleh Gheissari, St. Al Zahra Hospital, Isfahan University of Medical Science, Isfahan; Italy: Guiseppe Remuzzi, Clinical Research Centre of Rare Diseases “Aldo & Cele Daccò,” Istituto di Richerche Farmacologiche “Mario Negri,” Bergamo; Andrea Pasini, S.Orsola-Malpighi Hospital, Bologna; Gian Marco Ghiggeri, Instituto G. Gaslini, Genova; Gianluigi Ardissino, IRCCS Fondazione Ospedale Maggiore Policlinico, Milano; Elisa Benetti, Ascenda Ospedaliera University, Padua; Francesco Emma, Ospedale Pediatrico Bambino, Rome; Lebanon: Bilal Aoun, Rafic Hariri University Hospital, Beirut; Pauline Abou-Jaoudé, Notre Dame de Secours University Hospital, Byblos; Lithuania: Augustina Jankauskiene, Vilnius University Children's Hospital, Vilnius; Poland: Anna Wasilewska, Unversity Hospital, Bialystok; Lidia Hyla-Klekot, Pediatrics and Oncology Center, Chorzow; Aleksandra Zurowska, Medical University, Gdansk; Dorota Drozdz, Jagiellonian University Nedical College, Krakow; Marcin Tkaczyk, Polish Mothers Memorial Hospital Research Institute, Lodz; Halina Borzecka, Medical University, Lublin; Magdalena Silska, Poznan University of Medical Sciences, Poznan; Tomasz Jarmolinski, Pommeranian Academy of Medicine, Szczecin; Agnieszka Firszt-Adamczyk, Wojewodzki Hospital, Torun; Joanna Ksiazek, Centrum Zdrowia Dziecka, Warsaw; Elzbieta Kuzma-Mroczkowska, University Hospital, Warsaw; Anna Medynska, Medical University, Wroclaw; Maria Szczepanska, Medical University of Silesia in Katowice, Zabrze; Portugal: Alberto Caldas Afonso, Hospital S.Joao Pediatrics, Porto; Helena Jardim, Centre Hospitalar, Porto; Serbia: Amira Peco-Antic, University Children's Hospital, Belgrade; Radovan Bogdanovic, Institue of Mother and Healthcare of Serbia, Belgrade; Sweden: Rafael T. Krmar, Karolinska University Children's Hospital, Stockholm; Switzerland: Giacomo D. Simonetti, University Children's Hospital, Bern; Syria: Bassam Saeed, Kidney Hospital, Damascus; Turkey: Ali Anarat, Cukurova University Children's Hospital, Adana; Aysin Bakkaloglu, Hacettepe University Medical Faculty, Ankara; Ayse Balat, Gaziantep University Medical Faculty, Gaziantep; Z. Esra Baskin, Baskent University Hospital, Ankara; Nilgun Cakar, Diskapi Children's Hospital, Ankara; Ozlem Erdogan, Sami Ulus Children's Hospital, Ankara; Birsin Özcakar, Ankara University Medical School, Ankara; Fatih Ozaltin, Hacettepe University, Ankara; Onur Sakallioglu, Gulhane Military Academy of Medicine, Ankara; Oguz Soylemezoglu, Gazi University Hospital, Ankara; Sema Akman, Akdeniz University, Antalya; Faysal Gok, Gulhane Military Academy of Medicine, Gulhane; Salim Caliskan, Cerrahpasa Medical Faculty, Istanbul University, Istanbul; Cengiz Candan, Goztepe Hospital, Istanbul; Sevinc Emre, Istanbul Medical Faculty, Istanbul; Sevgi Mir, Ege University Medical Faculty, Izmir; Ipek Akil, Celal Bayar University, Manisa; Pelin Ertan, Celal Bayar University, Manisa; Ozan Özkaya, Ondokuz Mayis University, Samsun; Mukaddes Kalyoncu, Karadeniz University, Trabzon; United Arab Emirates: Eva Simkova, Dubai Hospital, Dubai; Entesar Alhammadi, Al Qassimi Hospital, Sharjah; Ukraine: Roman Sobko, Western Ukrainian Specialised Medical Centre, Lviv.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.06260614/-/DCSupplemental.
References
- 1.McKinney PA, Feltbower RG, Brocklebank JT, Fitzpatrick MM: Time trends and ethnic patterns of childhood nephrotic syndrome in Yorkshire, UK. Pediatr Nephrol 16: 1040–1044, 2001 [DOI] [PubMed] [Google Scholar]
- 2.Kim JS, Bellew CA, Silverstein DM, Aviles DH, Boineau FG, Vehaskari VM: High incidence of initial and late steroid resistance in childhood nephrotic syndrome. Kidney Int 68: 1275–1281, 2005 [DOI] [PubMed] [Google Scholar]
- 3.Mekahli D, Liutkus A, Ranchin B, Yu A, Bessenay L, Girardin E, Van Damme-Lombaerts R, Palcoux JB, Cachat F, Lavocat MP, Bourdat-Michel G, Nobili F, Cochat P: Long-term outcome of idiopathic steroid-resistant nephrotic syndrome: A multicenter study. Pediatr Nephrol 24: 1525–1532, 2009 [DOI] [PubMed] [Google Scholar]
- 4.Zagury A, Oliveira AL, Montalvão JA, Novaes RH, Sá VM, Moraes CA, Tavares MS: Steroid-resistant idiopathic nephrotic syndrome in children: Long-term follow-up and risk factors for end-stage renal disease. J Bras Neurol 35: 191–199, 2013 [DOI] [PubMed] [Google Scholar]
- 5.Saleem MA: New developments in steroid-resistant nephrotic syndrome. Pediatr Nephrol 28: 699–709, 2013 [DOI] [PubMed] [Google Scholar]
- 6.Mubarak M, Lanewala A, Kazi JI,Akhter F, Sher A, Fayyaz A, Bhatti S: Histopathological spectrum of childhood nephrotic syndrome in Pakistan. Clin Exp Nephrol 13: 589–593, 2009 [DOI] [PubMed] [Google Scholar]
- 7.Nammalwar BR, Vijayakumar M, Prahlad N: Experience of renal biopsy in children with nephrotic syndrome. Pediatr Nephrol 21: 286–288, 2006 [DOI] [PubMed] [Google Scholar]
- 8.Bhimma R, Adhikari M, Asharam K: Steroid-resistant nephrotic syndrome: The influence of race on cyclophosphamide sensitivity. Pediatr Nephrol 21: 1847–1853, 2006 [DOI] [PubMed] [Google Scholar]
- 9.Srivastava T, Simon SD, Alon US: High incidence of focal segmental glomerulosclerosis in nephrotic syndrome of childhood. Pediatr Nephrol 13: 13–18, 1999 [DOI] [PubMed] [Google Scholar]
- 10.Bonilla-Felix M, Parra C, Dajani T, Ferris M, Swinford RD, Portman RJ, Verani R: Changing patterns in the histopathology of idiopathic nephrotic syndrome in children. Kidney Int 55: 1885–1890, 1999 [DOI] [PubMed] [Google Scholar]
- 11.Banaszak B, Banaszak P: The increasing incidence of initial steroid resistance in childhood nephrotic syndrome. Pediatr Nephrol 27: 927–932, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ruf RG, Lichtenberger A, Karle SM, Haas JP, Anacleto FE, Schultheiss M, Zalewski I, Imm A, Ruf EM, Mucha B, Bagga A, Neuhaus T, Fuchshuber A, Bakkaloglu A, Hildebrandt F, Arbeitsgemeinschaft Für Pädiatrische Nephrologie Study Group : Patients with mutations in NPHS2 (podocin) do not respond to standard steroid treatment of nephrotic syndrome. J Am Soc Nephrol 15: 722–732, 2004 [DOI] [PubMed] [Google Scholar]
- 13.Büscher AK, Kranz B, Büscher R, Hildebrandt F, Dworniczak B, Pennekamp P, Kuwertz-Bröking E, Wingen AM, John U, Kemper M, Monnens L, Hoyer PF, Weber S, Konrad M: Immunosuppression and renal outcome in congenital and pediatric steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol 5: 2075–2084, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hinkes BG, Mucha B, Vlangos CN, Gbadegesin R, Liu J, Hasselbacher K, Hangan D, Ozaltin F, Zenker M, Hildebrandt F, Arbeitsgemeinschaft für Paediatrische Nephrologie Study Group : Nephrotic syndrome in the first year of life: Two thirds of cases are caused by mutations in 4 genes (NPHS1, NPHS2, WT1, and LAMB2). Pediatrics 119: e907–e919, 2007 [DOI] [PubMed] [Google Scholar]
- 15.Lipska BS, Iatropoulos P, Maranta R, Caridi G, Ozaltin F, Anarat A, Balat A, Gellermann J, Trautmann A, Erdogan O, Saeed B, Emre S, Bogdanovic R, Azocar M, Balasz-Chmielewska I, Benetti E, Caliskan S, Mir S, Melk A, Ertan P, Baskin E, Jardim H, Davitaia T, Wasilewska A, Drozdz D, Szczepanska M, Jankauskiene A, Higuita LM, Ardissino G, Ozkaya O, Kuzma-Mroczkowska E, Soylemezoglu O, Ranchin B, Medynska A, Tkaczyk M, Peco-Antic A, Akil I, Jarmolinski T, Firszt-Adamczyk A, Dusek J, Simonetti GD, Gok F, Gheissari A, Emma F, Krmar RT, Fischbach M, Printza N, Simkova E, Mele C, Ghiggeri GM, Schaefer F, PodoNet Consortium : Genetic screening in adolescents with steroid-resistant nephrotic syndrome. Kidney Int 84: 206–213, 2013 [DOI] [PubMed] [Google Scholar]
- 16.Mele C, Iatropoulos P, Donadelli R, Calabria A, Maranta R, Cassis P, Buelli S, Tomasoni S, Piras R, Krendel M, Bettoni S, Morigi M, Delledonne M, Pecoraro C, Abbate I, Capobianchi MR, Hildebrandt F, Otto E, Schaefer F, Macciardi F, Ozaltin F, Emre S, Ibsirlioglu T, Benigni A, Remuzzi G, Noris M, PodoNet Consortium : MYO1E mutations and childhood familial focal segmental glomerulosclerosis. N Engl J Med 365: 295–306, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ozaltin F, Ibsirlioglu T, Taskiran EZ, Baydar DE, Kaymaz F, Buyukcelik M, Kilic BD, Balat A, Iatropoulos P, Asan E, Akarsu NA, Schaefer F, Yilmaz E, Bakkaloglu A, PodoNet Consortium : Disruption of PTPRO causes childhood-onset nephrotic syndrome. Am J Hum Genet 89: 139–147, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hodson EM, Willis NS, Craig JC: Interventions for idiopathic steroid-resistant nephrotic syndrome in children. Cochrane Database Syst Rev 11: CD003594, 2010 [DOI] [PubMed] [Google Scholar]
- 19.Lombel RM, Gipson DS, Hodson EM, Kidney Disease: Improving Global Outcomes : Treatment of steroid-sensitive nephrotic syndrome: New guidelines from KDIGO. Pediatr Nephrol 28: 415–426, 2013 [DOI] [PubMed] [Google Scholar]
- 20.Ehrich JH, Geerlings C, Zivicnjak M, Franke D, Geerlings H, Gellermann J: Steroid-resistant idiopathic childhood nephrosis: Overdiagnosed and undertreated. Nephrol Dial Transplant 22: 2183–2193, 2007 [DOI] [PubMed] [Google Scholar]
- 21.Gellermann J, Ehrich JH, Querfeld U: Sequential maintenance therapy with cyclosporin A and mycophenolate mofetil for sustained remission of childhood steroid-resistant nephrotic syndrome. Nephrol Dial Transplant 27: 1970–1978, 2012 [DOI] [PubMed] [Google Scholar]
- 22.Gulati A, Sinha A, Gupta A, Kanitkar M, Sreenivas V, Sharma J, Mantan M, Agarwal I, Dinda AK, Hari P, Bagga A: Treatment with tacrolimus and prednisolone is preferable to intravenous cyclophosphamide as the initial therapy for children with steroid-resistant nephrotic syndrome. Kidney Int 82: 1130–1135, 2012 [DOI] [PubMed] [Google Scholar]
- 23.Gulati A, Sinha A, Jordan SC, Hari P, Dinda AK, Sharma S, Srivastava RN, Moudgil A, Bagga A: Efficacy and safety of treatment with rituximab for difficult steroid-resistant and -dependent nephrotic syndrome: Multicentric report. Clin J Am Soc Nephrol 5: 2207–2212, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hamasaki Y, Yoshikawa N, Nakazato H, Sasaki S, Iijima K, Nakanishi K, Matsuyama T, Ishikura K, Ito S, Kaneko T, Honda M, for Japanese Study Group of Renal Disease in Children : Prospective 5-year follow-up of cyclosporine treatment in children with steroid-resistant nephrosis. Pediatr Nephrol 28: 765–771, 2013 [DOI] [PubMed] [Google Scholar]
- 25.Roberti I, Vyas S: Long-term outcome of children with steroid-resistant nephrotic syndrome treated with tacrolimus. Pediatr Nephrol 25: 1117–1124, 2010 [DOI] [PubMed] [Google Scholar]
- 26.Aizawa-Yashiro T, Tsuruga K, Watanabe S, Oki E, Ito E, Tanaka H: Novel multidrug therapy for children with cyclosporine-resistant or -intolerant nephrotic syndrome. Pediatr Nephrol 26: 1255–1261, 2011 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.