Abstract
Background
Butyrophilin-like 2 (BTNL2) rs2076530 gene polymorphism has been implicated in susceptibility to sarcoidosis. However, results from previous studies are not consistent. To assess the association of BTNL2 polymorphism and sarcoidosis susceptibility, a meta-analysis was performed.
Methods
PubMed, Embase were searched for eligible case-control studies. Data were extracted and pooled odds ratios (OR) with 95% confidence intervals (CI) were calculated.
Results
Ten studies involving a total of 3303 cases and 2514 controls were included in this meta-analysis. Combined data indicated that BTNL2 rs2076530 polymorphism was associated with sarcoidosis susceptibility in allelic model (A vs. G, OR = 1.59, 95%CI: 1.47–1.72), dominant model (AA + AG vs. GG, OR = 2.10, 95%CI: 1.67–2.65), and recessive model (AA vs. AG + GG, OR = 1.93, 95%CI: 1.49–2.50).
Conclusions
This meta-analysis indicates that BTNL2 rs2076530 polymorphism contributes to the risk of sarcoidosis.
Introduction
Sarcoidosis is a multisystemic inflammatory disease with unknown aetiology, characterized by the formation of noncaseating granulomas and an exaggerated cellular immune response due to increased inflammatory activity of macrophages and CD4 helper T cells[1,2]. Many studies show that genetic susceptibility and environmental factors contribute to disease development. Among multiple potential factors contributing to sarcoidosis, one of them is the polymorphism of butyrophilin-like 2 (BTNL2) gene.
BTNL2, a member of the immunoglobulin superfamily, reduces proliferation and cytokine production from activated T cells, suggesting a role of functional BTNL2 as a negative costimulatory molecule involved in T-cell activation[3]. The G→A transition constituting rs2076530 (G16071A) leads to a premature truncation of the protein, thereby disrupting insertion in the cell membrane, which is a necessary process for downregulating activated T cells[4,5]. So the truncated BTNL2 seems to be involved in sarcoidosis. Several studies have been undertaken to evaluate this potential relationship between BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility. However, results from different reports of different geographic areas are not consistent. Besides, many studies were small in size and, as such, may on their own lack sufficient statistical power to address this issue adequately. In an effort to clarify the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility, we performed a meta-analysis on all the studies identified by systematic review of literatures.
Methods
Literature sources and search strategy
This meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) criteria[6]. Two investigators (Y.Lin, J.Wei) searched PubMed, Embase databases up to 4 August 2014 independently. Disagreements were resolved through discussion or adjudicated by a third author (D.Cheng). All eligible articles were screened, and their references were checked for other relevant studies. The search strategy was as follows: (“sarcoidosis” or “Schaumann Syndrome” or “Boeck Disease” or “Besnier Boeck Schaumann Syndrome”) and (“SNP” or “variant” or “polymorphism” or “mutation”) in combination with (“BTNL2” or “butyrophilin-like 2” or “rs2076530”). There was no restriction on languages.
Reports that fulfilled the following criteria were included: (1) evaluating the BTNL2 rs2076530 polymorphism and sarcoidosis risks; (2) case-control designs on unrelated individuals; (3) odds ratios (OR) with 95% confidence intervals (CI) or sufficient data for their calculation were available; (4) the control subjects satisfied the Hardy-Weinberg equilibrium (HWE); (5) the diagnoses of sarcoidosis were consistent with the International Consensus Statement on Sarcoidosis[7,8] or verified by histology. And reports were excluded if any of the following conditions existing: (1) reviews, abstracts and studies with overlapping or repeated data, (2) data about allelic frequencies could not be obtained. In case of overlapping or repeated studies, the one with most subjects was chosen.
Quality assessment
Two authors evaluated the study quality independently according to the Newcastle-Ottawa scale (NOS)[9], which assesses the quality of non-randomised studies on the basis of selection of participants, comparability of groups, and exposure assessment. Disagreement was settled as described above.
Data extraction
The data were extracted by two investigators (Y.Lin, J.Wei) independently and a consensus was reached on all items. Any disagreement was resolved as described above. The following data was collected: first author, year, ethnicity, sample size, mean age, gender ratio, sarcoidosis criteria, source of controls, and genotype or allele distribution in cases and controls.
Statistical analysis
The data were analyzed using Review Manager 5.2 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark) and STATA 12.0 software (Stata Corp LP, College Station, TX, USA). Pearson’s χ2 test was used to determine whether the observed frequencies of genotypes in control groups conformed to HWE. The strength of the associations between BTNL2 rs2076530 polymorphism and sarcoidosis risk was evaluated using OR with 95% CI. The pooled ORs were calculated for allelic model (A vs. G), dominant model (AA + AG vs. GG) and recessive model (AA vs. AG + GG) respectively. Heterogeneity was calculated using the Q-test and I 2-statistics[10]. If the P-value of the Q-test was>0.10, the pooled OR was assessed in a fixed-effects model; otherwise, the random-effects model was applied[11]. I 2 values were used to quantify heterogeneity[12]. Z-test were used to determine the significance of the pooled OR, in which a P-value of <0.05 was considered statistically significant. Subgroup analyses were performed to evaluate the ethnic-specific effects. For the subgroup analysis by ethnicity, the study populations were stratified into three groups: Caucasians, Asians, and mixed population. We also performed sensitivity analysis by extracting a single study each time to check the stability of the results. Publication bias was assessed using Egger’s test, Begg’s test and funnel plot.
Results
Study inclusion and characteristics
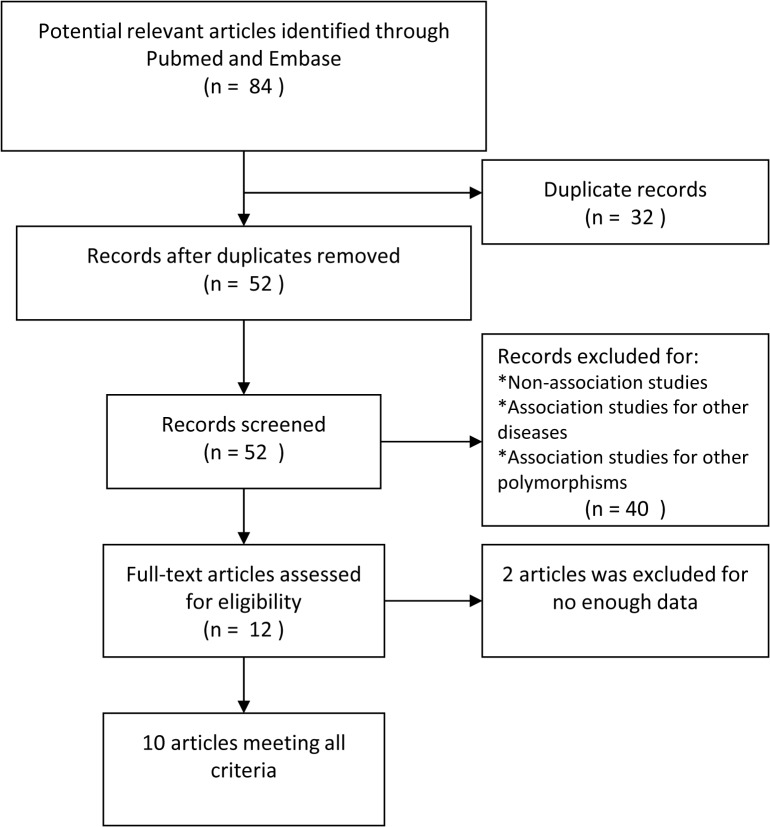
The procedure for including eligible studies is shown in Fig 1. A total of 84 articles were retrieved after initial search. Among them, thirty-two duplicate records were removed. After evaluating titles and abstracts, forty results were excluded for not relating to original study about association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility. After reading the full text, two articles were excluded for not enough data available. Finally, ten articles were included (the subjects of eight studies were Caucasion population[4,13,14,15,16,17,18,19], one study included Asian population[20] and one study involved mixed population[21]), containing 3303 cases and 2514 controls. In seven studies[4,13,15,17,18,19,20], the diagnoses of sarcoidosis were consistent with the International Consensus Statement on Sarcoidosis[7,8]. In the other three studies[14,16,–21], the diagnoses of sarcoidosis were verified by histology. Of the ten included studies, five articles[4,14,16,19,21] provided genotype distributions of BTNL2 gene rs2076530 in sarcoidosis group (containing 1629 subjects) and control group (containing 1065 subjects). In the other five articles[13,15,17,18,19], only allele distributions could be obtained. It is noteworthy that the study by Rybicki et al[13] was performed in two ethnic populations (Caucasion and African American), but only the data of Caucasion population could be obtained through the article. We tried to contact the authors via email to get more information, but did not get any reply. The NOS scores of the including studies ranged from 7 to 9, indicating that the methodological quality was generally good. The general characteristics of all the including studies are listed in Table 1.
Fig 1. Flow of study indentification, inclusion and exclusion.
Table 1. The baseline characteristics of all study populations in the meta-analysis.
| First author | Year | Country | Ethnicity | Score | Total sample size | Allele distributions | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cases | Controls | Cases | Controls | |||||||
| A | G | A | G | |||||||
| Rybicki BA | 2005 | USA | Caucasion | 8 | 366 | 366 | 483 | 249 | 403 | 329 |
| Valentonyte R | 2005 | German | Caucasion | 8 | 904 | 427 | 1241 | 567 | 489 | 365 |
| Li Y | 2006 | German | Caucasion | 7 | 210 | 202 | 291 | 129 | 250 | 154 |
| Spagnolo P | 2007 | UK and Netherlands | Caucasion | 8 | 288 | 446 | 386 | 190 | 512 | 380 |
| Milman N | 2011 | Denmark | Caucasion | 9 | 87 | 113 | 129 | 45 | 126 | 100 |
| Wijnen PA | 2011 | Netherlands | Caucasion | 9 | 632 | 200 | 836 | 428 | 240 | 160 |
| Morais A | 2012 | Portugal | Caucasion | 9 | 151 | 150 | 197 | 105 | 167 | 133 |
| Suzuku H | 2012 | Japan | Asian | 8 | 237 | 287 | 355 | 119 | 355 | 219 |
| Ozdemir M | 2014 | Turkey | Caucasion | 7 | 375 | 271 | 59 | 47 | 48 | 56 |
| Gaillot-Drevon M | 2014 | France | Mixed | 7 | 53 | 52 | 512 | 238 | 295 | 247 |
Quantitative analysis
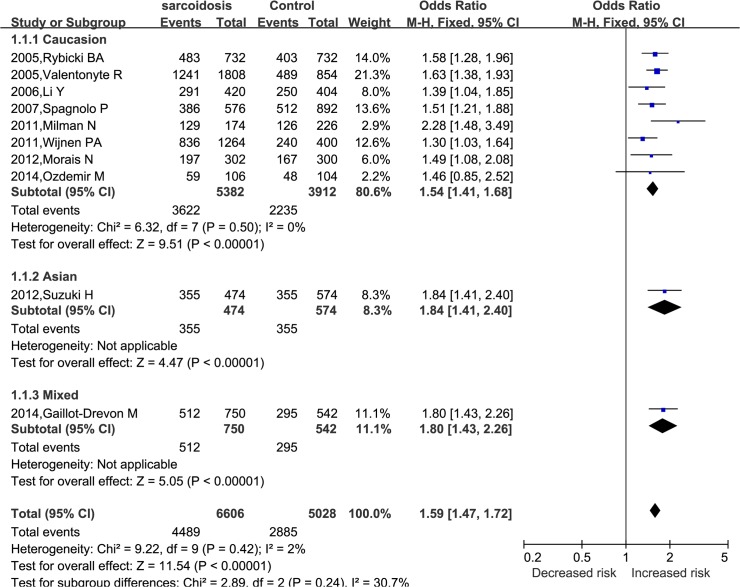
We found that the A allele of BTNL2 rs2076530 was associated with an increased risk of sarcoidosis in allelic model (A vs. G, P<0.00001). The pooled OR was 1.59 (95%CI: 1.47–1.72) under the fixed effect model, without between-study heterogeneity (I 2 = 2%, P = 0.42) (Fig 2). In subgroup analyses by ethnicity, there were similar results in Caucasion (OR = 1.54, 95%CI: 1.41–1.68, P<0.00001), Asian (OR = 1.84, 95%CI: 1.41–2.40, P<0.00001), and mixed population (OR = 1.80, 95%CI: 1.43–2.26, P<0.00001) (Fig 2).
Fig 2. Forest plots of OR with 95% CI for the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility subanalyzed by enthnicity in A vs G.
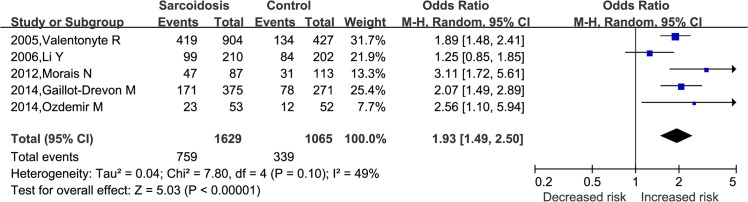
Combined data from the five studies with available genotype data [4, 14,16, 19,21] showed similar results under dominant model (AA + AG vs. GG, OR = 2.10, 95%CI: 1.67–2.65, P<0.00001, Fig 3), and recessive model (AA vs. AG + GG, OR = 1.93, 95%CI: 1.49–2.50, P <0.00001, Fig 4). As most of the subjects in the five studies were Caucasion, no subgroup analyses were performed under dominant model and recessive model.
Fig 3. Forest plots of OR with 95% CI for the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility in AA+AG vs GG.
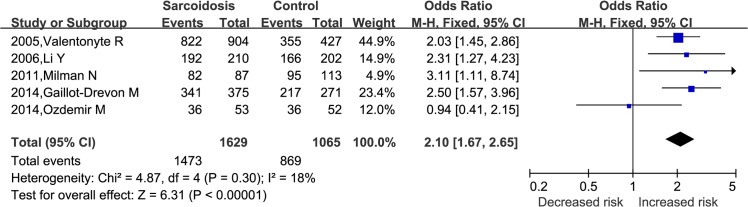
Fig 4. Forest plots of OR with 95% CI for the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility in AA vs AG+GG.
Sensitivity analysis
We performed sensitivity analysis for statistically significant result. For the association of the BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility among the overall populations, the observed significant result was not materially altered after sequentially excluding each study.
Publication bias
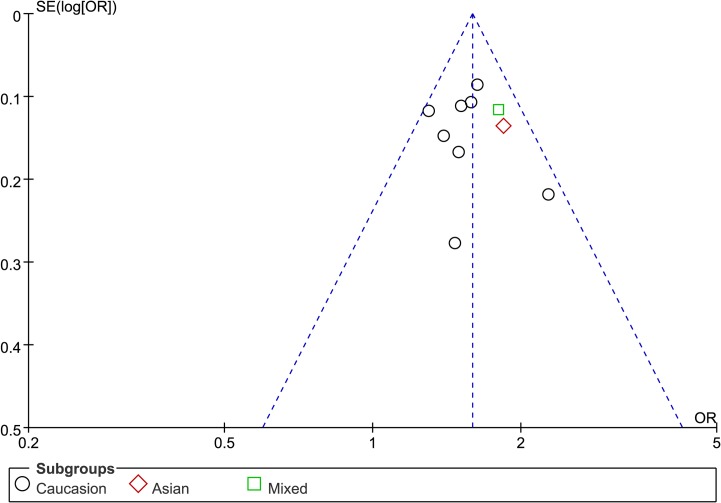
Neither Egger’s test nor Begg’s test indicated significant publication bias (P = 0.956 and P = 0.592, respectively) in allelic model. The shape of the funnel plot was symmetrical (Fig 5).
Fig 5. Funnel plot for evaluation of publication bias in the included studies on the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility in A vs G.
Discussion
Sarcoidosis has gained much attention from physicians since 1877 when it was first described. Although much progress has been made, much confusion still remains about this disease[7,8], with its cause being an improtant one.
It is assumed that genes contribute to the development of sarcoidosis since its trend of family clustering [22]and the different morbidities between ethnic groups, which have been described by several observational studies. Two previous Genome-wide association studies (GWASs) conducted in European and American populations [23,24]implicate multiple shared loci in sarcoidosis succeptibility, one of which, the single nucleotide polymorphism rs2076530 in BTNL2 gene has been studied in many other ethnic groups. The aim of this study was to investigate the association of the BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility by conducting a meta-analysis in ten case–control studies including 5817 subjects. We provided convincing evidence on the contributory effect of BTNL2 rs2076530 polymorphism in the development of sarcoidosis. The results of this meta-analysis are consistent with the findings of the two previous GWASs concerning sarcoidosis[23,24]. The significant increased A allele of BTNL2 rs2076530 observed in sarcoidosis group, implys the role of this truncating single nucleotide polymorphism in sarcoidosis susceptibility.
Our study had some advantages in several key aspects. Firstly, there was no between-study heterogeneity. Secondly, no publication bias was indicated. Thirdly, there was little modification after sequentially excluding individual study. These characters of our meta-analysis indicated a reliable and stable result about the association between BTNL2 rs2076530 polymorphism and sarcoidosis risk.
In interpreting the results of this study, the limitations of the meta-analysis should be mentioned. Firstly, sarcoidosis is a complicated and multi-factor disease and potential interactions between gene-gene and gene-enviroment should be considered. However, this meta-analysis only assessed the association of BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility. Secondly, nearly all the including studies were from the Caucasians, so the results may only be applicable to this ethnic population. In particular, it is worthy to note that BTNL2 rs2076530 polymorphism was not strongly associated with sarcoidosis in African Americans in American GWAS [24]. Therefore further studies in other ethnic groups such as Asians, Africans and Latinos are required. Thirdly, the Asian subgroup only included one study. The small number may result in low statistical power. Fourthly, due to insufficient data reported by the original studies, further stratifications by gender, clinical course or other clinical variables were not possible.
In conclusion, this is the first meta-analysis concerning BTNL2 rs2076530 polymorphism and sarcoidosis susceptibility up to date. This meta-analysis extended previous findings on the association between the BTNL2 rs2076530 polymorphism and sarcoidosis, by showing that the A allele of BTNL2 rs2076530 was associated with an increased risk of sarcoidosis susceptibility. Additional well designed large studies were needed to validate our findings. Further systematic studies should be encouraged to explore the underlying mechanisms of this association.
Supporting Information
(DOC)
(DOC)
(DOCX)
(DOCX)
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
The authors have no support or funding to report.
References
- 1. Newman LS, Rose CS, Maier LA (1997) Sarcoidosis. N Engl J Med 336: 1224–1234. [DOI] [PubMed] [Google Scholar]
- 2. Ziegenhagen MW, Muller-Quernheim J (2003) The cytokine network in sarcoidosis and its clinical relevance. J Intern Med 253: 18–30. [DOI] [PubMed] [Google Scholar]
- 3. Arnett HA, Escobar SS, Gonzalez-Suarez E, Budelsky AL, Steffen LA, Boiani N, et al. (2007) BTNL2, a butyrophilin/B7-like molecule, is a negative costimulatory molecule modulated in intestinal inflammation. J Immunol 178: 1523–1533. [DOI] [PubMed] [Google Scholar]
- 4. Valentonyte R, Hampe J, Huse K, Rosenstiel P, Albrecht M, Stenzel A, et al. (2005) Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet 37: 357–364. [DOI] [PubMed] [Google Scholar]
- 5. Nguyen T, Liu XK, Zhang Y, Dong C (2006) BTNL2, a butyrophilin-like molecule that functions to inhibit T cell activation. J Immunol 176: 7354–7360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Moher D, Liberati A, Tetzlaff J, Altman DG, Grp P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Bmj-British Medical Journal 339. [PMC free article] [PubMed] [Google Scholar]
- 7.(1999) Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med 160: 736–755. [DOI] [PubMed]
- 8. Costabel U, Hunninghake GW. (1999) ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J 14: 735–737. [DOI] [PubMed] [Google Scholar]
- 9. Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. (2000) The newcastle-ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses The Ottawa Health Research Institute; Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. [Google Scholar]
- 10. Higgins J, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Statistics in medicine 21: 1539–1558. [DOI] [PubMed] [Google Scholar]
- 11. DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Controlled clinical trials 7: 177–188. [DOI] [PubMed] [Google Scholar]
- 12. JPT CHH, Green S (2011) Cochrane handbook for systematic reviews of interventions version 5.1. 0 [updated March 2011] The Cochrane Collaboration. [Google Scholar]
- 13. Rybicki BA, Walewski JL, Maliarik MJ, Kian H, Iannuzzi MC (2005) The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet 77: 491–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Li Y, Wollnik B, Pabst S, Lennarz M, Rohmann E, Gillissen A, et al. (2006) BTNL2 gene variant and sarcoidosis. Thorax 61: 273–274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Spagnolo P, Sato H, Grutters JC, Renzoni EA, Marshall SE, Ruven HJ, et al. (2007) Analysis of BTNL2 genetic polymorphisms in British and Dutch patients with sarcoidosis. Tissue Antigens 70: 219–227. [DOI] [PubMed] [Google Scholar]
- 16. Milman N, Svendsen CB, Nielsen FC, van Overeem Hansen T (2011) The BTNL2 A allele variant is frequent in Danish patients with sarcoidosis. Clin Respir J 5: 105–111. 10.1111/j.1752-699X.2010.00206.x [DOI] [PubMed] [Google Scholar]
- 17. Wijnen PA, Voorter CE, Nelemans PJ, Verschakelen JA, Bekers O, Drent M (2011) Butyrophilin-like 2 in pulmonary sarcoidosis: a factor for susceptibility and progression? Hum Immunol 72: 342–347. 10.1016/j.humimm.2011.01.011 [DOI] [PubMed] [Google Scholar]
- 18. Morais A, Lima B, Peixoto MJ, Alves H, Marques A, Delgado L (2012) BTNL2 gene polymorphism associations with susceptibility and phenotype expression in sarcoidosis. Respir Med 106: 1771–1777. 10.1016/j.rmed.2012.08.009 [DOI] [PubMed] [Google Scholar]
- 19. Ozdemir M, Saydam F, Kurt E, Degirmenci I, Tuncel T, Cilingir O, et al. (2014) Is there a genetic predisposition for Turkish patients with sarcoidosis in the 329-bp region containing the BTNL2 rs2076530 polymorphism? Turkish Journal of Medical Sciences 44: 590–594. [DOI] [PubMed] [Google Scholar]
- 20. Suzuki H, Ota M, Meguro A, Katsuyama Y, Kawagoe T, Ishihara M, et al. (2012) Genetic characterization and susceptibility for sarcoidosis in Japanese patients: risk factors of BTNL2 gene polymorphisms and HLA class II alleles. Invest Ophthalmol Vis Sci 53: 7109–7115. 10.1167/iovs.12-10491 [DOI] [PubMed] [Google Scholar]
- 21. Gaillot-Drevon M, Calender A, Blay JY, Valeyre D, Israel-Biet D, Roy P, et al. (2014) Lack of correlation of BTNL2 polymorphism and cancer risk in sarcoidosis. BTNL2 and cancer risk in sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 31: 136–141. [PubMed] [Google Scholar]
- 22. Rybicki BA, Iannuzzi MC, Frederick MM, Thompson BW, Rossman MD, Bresnitz EA, et al. (2001) Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Respir Crit Care Med 164: 2085–2091. [DOI] [PubMed] [Google Scholar]
- 23. Hofmann S, Franke A, Fischer A, Jacobs G, Nothnagel M, Gaede KI, et al. (2008) Genome-wide association study identifies ANXA11 as a new susceptibility locus for sarcoidosis. Nat Genet 40: 1103–1106. 10.1038/ng.198 [DOI] [PubMed] [Google Scholar]
- 24. Adrianto I, Lin CP, Hale JJ, Levin AM, Datta I, Parker R, et al. (2012) Genome-wide association study of African and European Americans implicates multiple shared and ethnic specific loci in sarcoidosis susceptibility. PLoS One 7: e43907 10.1371/journal.pone.0043907 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOC)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.