Abstract
Objective
To investigate whether postnatal weight at first detection of retinopathy of prematurity (ROP) can predict preterm infants who will develop severe ROP warranting treatment.
Design
This modern, population-based cohort included 147 infants born at gestational age (GA) <32 weeks in the Gothenburg region during 2011–2012 and screened for ROP at Sahlgrenska University hospital. GA, birth weight (BW), and weekly postnatal weight from birth until postmenstrual age (PMA) 40 weeks data were retrospectively retrieved. Birth weight SD scores (BWSDS) were calculated. ROP data, including first detected ROP stage, maximal ROP stage, ROP treatment, and PMA at first detected sign of ROP were also retrieved. Weight SDS (WSDS) at first ROP detection was calculated.
Results
Stepwise multivariate logistic regression analysis revealed that the best fit-model of risk factors for developing severe ROP warranting treatment included; GA (OR=0.28, CI 95% 0.12 to 0.66, p<0.01) and WSDS at first ROP detection (OR=0.22, CI 95% 0.05 to 0.89, p<0.05).
Conclusions
Low weight and low WSDS at first ROP detection can be useful predictors for ROP warranting treatment.
INTRODUCTION
Retinopathy of prematurity (ROP) is a condition that threatens the sight of very preterm infants. Survival rates for the most preterm infants have increased in the last decades1; thus, improved identification of infants at greatest risk for severe ROP is important.
The pathogenesis of ROP is multifactorial. Major risk factors for ROP reflect the infant’s immaturity and include low gestational age (GA)2 and low birth weight (BW).3 Other established risk factors are comorbidities reflecting the infant’s general state of health, such as need for prolonged mechanical ventilation,4 persistent ductus arteriosus,5 sepsis,6 necrotising enterocolitis5 and intraventricular haemorrhage.7
Studies have found that the course of ROP can predict its severity. Low postmenstrual age (PMA), nasal retina site at first detection of ROP, and speed of ROP progression have been reported as risk factors for developing severe ROP.8–11
During recent years, postnatal weight development has emerged as an important predictor of severe ROP.12–16 Research by Smith, Hellström, and coworkers elucidated the association between low insulin-like growth factor 1 (IGF-1) and poor post-natal weight gain in the development of ROP.13,17 These findings have resulted in several surveillance models that use poor postnatal weight gain as a predictor for developing severe ROP.16,18–20 In our study population, the online surveillance system Weight, Insulin-like growth factor I, Neonatal, Retinopathy of Prematurity (WINROP) was used as a supplement to established screening criteria.
With the intention of finding improved risk predictors for developing ROP warranting treatment, we analysed the longitudinal postnatal weight gain of preterm infants. Our most important finding is that low weight SD score (WSDS) and low weight at first detection of ROP can be useful predictors for ROP warranting treatment.
METHODS
Study population
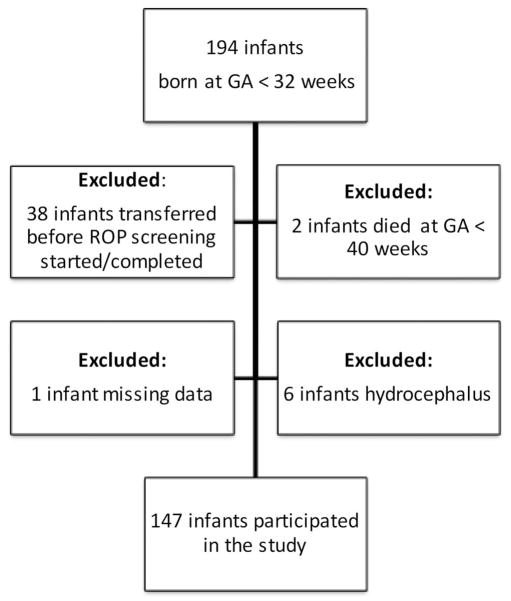
For this study, all infants born at GA <32 weeks in the Gothenburg region in 2011–2012, and screened for ROP at Sahlgrenska University hospital, were enrolled (n=194). GA at birth was calculated using ultrasound examinations performed before 20 weeks of gestation. Neonatal data, such as GA, BW and gender, were collected retrospectively from the infants’ hospital records. Weekly postnatal weights, from birth until PMA 40 weeks, were retrospectively collected for every infant. Weight data were retrieved from the infants’ hospital records and, when discharged, from the infants’ records at their child health care centre. Infants transferred to other hospitals before ROP screening began or was completed were excluded (n=38), as were infants who died before PMA 40 weeks, since ROP screening was incomplete (n=2). Infants with incomplete weekly weight measurements were also excluded (n=1), as well as those with hydrocephalus, since their weight gain did not reflect physiological post-natal growth (n=6) (figure 1).
Figure 1.
Flowchart of the study population.
ROP examination and treatment
ROP screening began at postnatal age 5–6 weeks and continued until the retina was fully vascularised, or until regression of ROP. Eye examinations were performed according to a routine protocol and consisted of dilated ocular fundus examinations.
For classification of ROP, the International Classification of ROP revisited was used.21 The recommendations of the Early Treatment for ROP Cooperative Group were followed for treatment.22
Data regarding maximum ROP in the worse eye, date of diagnosis of ROP stages and eventual treatment were retrieved from the Swedish national register for retinopathy of prematurity (SWEDROP). Time of first detection of ROP was defined as the PMA when ROP was first diagnosed, regardless of stage.
Statistical analysis
Birth weight SD scores (BWSDS) and WSDS were calculated with a Swedish gender-specific reference that is considered to reflect undisturbed intrauterine growth.23 When calculating weight and WSDS at first detection of ROP, we used the weight measured during the PMA week when ROP was first detected. IBM SPSS Statistics 20 for Microsoft Windows (IBM Corporation, Armonk, New York, USA) and SAS V.9.3 (SAS, Cary, North Carolina, USA) were used for statistical analysis. Mann–Whitney U test was used for calculating differences in medians of variables at first detection of ROP. Univariate and multivariate logistic regression analysis were applied to evaluate the impact of different risk factors for ROP warranting treatment. ORs and 95% CIs were calculated for each risk factor. Pearson’s correlation test was used to estimate correlations between risk factors. The fit of the models was checked with the Hosmer–Lemeshow goodness-of-fit test. Receiver operating characteristic (ROC) curve and area under the curve (AUC) analyses were performed to estimate the predictive capacity of the models.
RESULTS
Birth characteristics and ROP frequency
For this study, 147 infants were eligible and 51.7% (76/147) were male. The median BW was 1185 g (range 440–2840 g), the median BWSDS was −1.04 SDS (range −4.55–5.20 SDS), and the median GA was 29 weeks and 1 day (range 22 weeks 6 days to 31 weeks 6 days). In this cohort, 10.2% (15/147) received ROP treatment (table 1).
Table 1.
Infant characteristics at birth and at first detection of ROP correlated to the severity of ROP.
| Characteristics | No ROP | ROP not treated | ROP treated | All infants |
|---|---|---|---|---|
| Birth characteristics, median (range) | ||||
| Number of infants, (%) | 99 (67.3) | 33 (22.4) | 15 (10.2) | 147 |
| GA, (week+day) | 30+1 (25+5 to 31+6) | 26+4 (24+1 to 29+4) | 24+0 (22+6 to 28+0) | 29+1 (22+6 to 31+6) |
| BW, (g) | 1375 (725–2840) | 875 (580–1430) | 600 (440–1000) | 1185 (440–2840) |
| Gender, male (%) | 59 (60) | 11 (33) | 6 (40) | 76 (51.7) |
| BWSDS (SDS) | −1.08 (−4.55 to 5.20) | −0.78 (−3.74 to 1.25) | −0.91 (−2.94 to 0.75) | −1.04 (−4.55 to 5.20) |
| Infant characteristics, median (range) | ||||
| PMA at first detection of ROP (week+day) | 34+0 (31+3 to 38+1) | 32+3 (30+6 to 35+6) | 33+4 (30+6 to 38+1) | |
| Weight at first detection of ROP (g) | 1995 (1500–3130) | 1540 (1265–2000) | 1908 (1265–3130) | |
| WSDS at first detection of ROP (SDS) | −1.18 (−2.90 to 0.44) | −2.19 (−3.12 to −1.13) | −1.42 (−3.12 to 0.44) | |
BW, birth weight; BWSDS, birth weight SD score; GA, gestational age; ROP, retinopathy of prematurity; SDS, SD score; WSDS, weight SD score.
Postnatal weight development
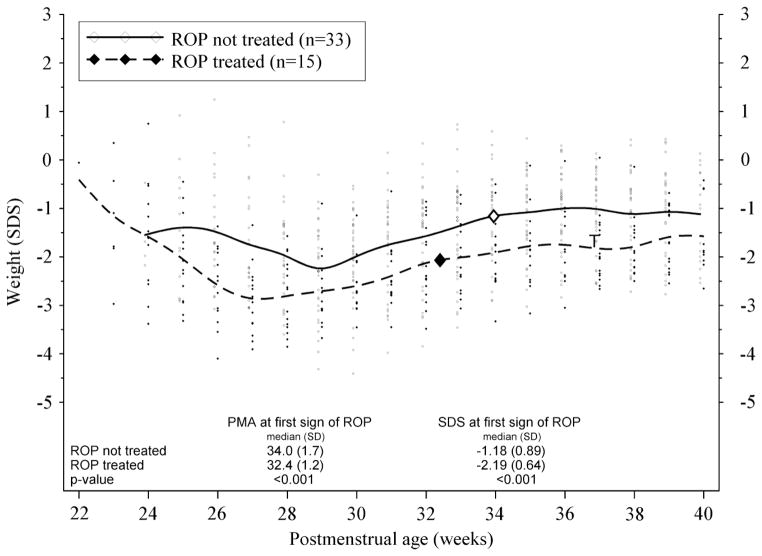
Infants who developed ROP warranting treatment had poorer postnatal weight development compared to those who developed less severe ROP not warranting treatment (figure 2).
Figure 2.
Postnatal weight development (weight SDS vs PMA (week)) of preterm infants who developed and were treated for retinopathy of prematurity (ROP) compared with those who developed ROP but did not warrant treatment. Large squares indicate median PMA at first detection of ROP. ‘T’ on graph indicates median PMA (36 weeks +6 days) at time of ROP treatment. PMA, postmenstrual age.
First detection of ROP
Of the 48 infants who developed ROP, 50% (24/48) had ROP stage 1 at first detection of ROP, and 50% (24/48) had ROP stage 2. In 10 infants, ROP was detected at their first ROP screening examination; of these, four infants developed ROP warranting treatment and six infants developed maximum ROP stages 1–2.
Significant differences in variables at first detection of ROP
When comparing variables at first detection of ROP, we found that the median PMA at first detection of ROP was significantly lower in infants who later developed ROP warranting treatment compared to those who did not warrant treatment (34 weeks +0 day vs 32 weeks+3 days; p<0.001). Median weight (1540 vs 1995 g; p<0.001) and median WSDS (−1.18 vs −2.19 SDS; p<0.001) at first detection of ROP were also lower in infants who later warranted ROP treatment (figure 2).
Logistic regression analysis
Univariate logistic regression analysis to calculate risk factors for developing ROP warranting treatment revealed that the natal risk factors GA at birth (p<0.001) and BW (50 g increment) (p<0.001) were significant risk factors, but BWSDS and gender were not. Significant risk factors at first detection of ROP were weight (50 g increment) (p=0.001) and WSDS (p=0.002), as well as PMA (p=0.003).
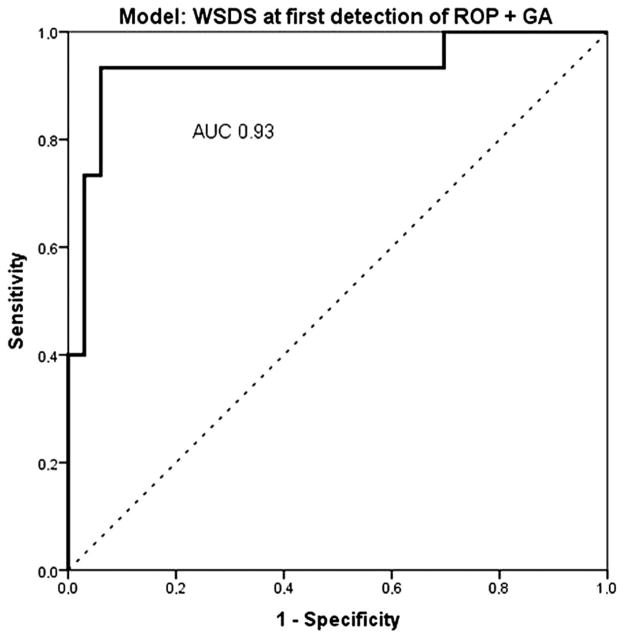
Multivariate logistic regression analysis was performed to assess the risk factors available in the dataset: GA at birth, BW (50 g increment), PMA at first detection of ROP, weight at first detection of ROP (50 g increment), and WSDS at first detection of ROP. The best-fit model, created by stepwise backward multivariate logistic regression analysis, included GA and WSDS at first detection of ROP (table 2). This combination of variables explained between 50% (Cox & Snell R2) and 70% (Nagelkerke R2) of the infants’ risk for developing ROP warranting treatment, and correctly classified 69% of the cases (Hosmer–Lemeshow goodness-of-fit 0.5). The ROC curves for the model comprising of the risk factor GA and WSDS at first detection of ROP resulted in an AUC of 0.93 (95% CI 0.84 to 1.0, p<0.001) (figure 3).
Table 2.
Association between infant characteristics and ROP treatment according to univariate and multivariate logistic regression analyses
| Univariate logistic regression | p Value | OR | 95% CI |
|---|---|---|---|
| GA (dec) | <0.001 | 0.18 | 0.08 to 0.42 |
| BW (50 g increments) | <0.001 | 0.54 | 0.40 to 0.74 |
| Male gender | 0.34 | 0.59 | 0.20 to 1.75 |
| BWSDS (SDS) | 0.92 | 1.02 | 0.70 to 1.47 |
| PMA at first detection of ROP | 0.003 | 0.35 | 0.18 to 0.70 |
| Weight at first detection of ROP (50 g increment) | 0.001 | 0.73 | 0.60 to 0.78 |
| WSDS at first detection of ROP (SDS) | 0.002 | 0.21 | 0.08 to 0.57 |
| Multivariate logistic regression | |||
| GA | 0.003 | 0.28 | 0.12 to 0.66 |
| WSDS at first detection of ROP (SDS) | 0.034 | 0.22 | 0.05 to 0.89 |
BW, birth weight; BWSDS, birth weight SD score; GA, gestational age; PMA, postmenstrual age; ROP, retinopathy of prematurity; SDS, SD score; WSDS, weight SD score.
Figure 3.
Receiver operator characteristic (ROC) curve comprising the risk factors gestational age (GA) (dec) and weight standard deviation score (WSDS) at first detection of retinopathy of prematurity (ROP). Area under the curve (AUC) 0.93 (95% CI 0.84 to 1.0, p<0.001). AUC reference line is 0.5.
DISCUSSION
In this study, we established a correlation between weight and WSDS at first detection of ROP, and infants’ risk of developing ROP warranting treatment. Based on our results, weight and WSDS at first detection of ROP are potential predictors for severe ROP, which could have important implications when planning ROP screening of infants born preterm. In univariate logistic regression analysis we found that all included variables at first detection of ROP were significant as risk factors; low weight (50 g increment) at first detection of ROP (OR=0.73, 95% CI 0.60 to 0.78, p=0.001), low WSDS at first detection of ROP (OR=0.21, 95% CI 0.08 to 0.57, p=0.002) as well as PMA at first detection of ROP (OR=0.35, CI 95% 0.18 to 0.70, p=0.003). In this study, no infants with a weight >2000 g or a WSDS above −1.13 SDS at first detection of ROP developed ROP warranting treatment. Whether these weight limits at first detection of ROP are applicable to other cohorts in predicting ROP warranting treatment requires further investigation.
Our findings are consistent with previous studies that identified poor postnatal weight gain as a major risk factor for developing severe ROP.12–16 Previous studies have shown that low PMA at first detection of ROP is a predictor for developing severe ROP.2 When we performed multivariate logistic regression analysis, we found that WSDS at first detection of ROP remained independently significant in relation to PMA at first detection of ROP, while weight (50 g increment) at first detection of ROP was found to have a high correlation with PMA at first detection of ROP (Pearson’s correlations test). Multivariate logistic regression analysis, including WSDS at first detection of ROP and GA was the best-fit model of risk factors, and correctly classified 70% of the infants who developed ROP warranting treatment. When we estimated the predictive value of these risk factors by ROC curve analysis, we found an AUC of 0.93 for the combination GA and WSDS at first detection of ROP. This is a high AUC predictive value, and could be explained by the combination of risk factors in the model. The risk factors reflect both the infants’ immaturity (low GA) as well as poor postnatal growth (low WSDS at first detection of ROP). GA and BW are strong risk factors for severe ROP but blunt. GA and BW as screening criteria results in numerous and unnecessary stressful ROP screening examinations to find the 5–10% of infants warranting ROP treatment. When we identify new risk factors, in this case, factors related to postnatal growth (eg, WINROP, Children’s Hospital of Philadelphia, and as here the weight at first detection of ROP) and combine these risk factors with the well-established degree of immaturity (low GA) we have an improved possibility to identify and select the right infants for ROP screening. In clinical settings where WINROP analysis as a complementary screening tool is not practical (such as cases with incomplete weight measurements), the simplified variable ‘the infants weight at first detection of ROP’ could be used as a supplement to traditional screening for improved prediction of disease severity.
A strength of this study is that it includes a modern, population-based cohort of infants born in the Gothenburg region and screened at the Sahlgrenska University hospital during 2010–2011. Another strength is that the weight data were completely collected weekly up to 40 weeks PMA.
A major limitation of this study is the small number of infants in the cohort: only 15 infants warranted ROP treatment. Although we obtained clear statistical significance with the Mann-Whitney U test calculation of means and medians, our results need to be validated in a larger cohort of infants. Another limitation of this study is that in 21% (10/48) of the infants, ROP was detected at their first ROP screening examination. Consequently, the date of onset of ROP in these infants is unknown since they were not screened previously. When we compared differences in characteristics between infants with ROP detected at their first ROP screening examination or not, we found no significant differences in BW, GA, maximum stage of ROP, and ROP treatment. When we exclude the infants with ROP detected at their first screening examination, the results persist: that there are significant differences in PMA, weight and WSDS at first detection of ROP between infants warranting ROP treatment or not.
We used the weight measured in the same PMA week as the first detection of ROP to represent the weight of the infant at first detection of ROP, and the two may differ because the date of measurement may be different from the day of ROP examination. We do not think that these differences affect the results, since when we compare infants who develop ROP warranting treatment with those who do not, we find no significant difference in the number of days between the actual day of weight measurement and the actual day of first detection of ROP.
The eye examination used for ROP screening is known to be stressful and painful for preterm infants.24,25 Thus, it is important to find new risk factors and predictors of which infants are at greatest risk of developing severe ROP. Improved identification of those infants at greatest risk for ROP warranting treatment will allow ophthalmological interventions to focus on these infants at the highest risk and will spare infants at minor risk from at least some stressful eye examinations.
To the best of our knowledge, this study is the first to have established that weight and WSDS at first detection of ROP are risk factor for ROP warranting treatment. If ophthalmologists screening for ROP have access to the weight of a preterm infant when ROP is first detected, it could help them to predict or estimate that infant’s risk of developing ROP warranting treatment; this would be a noteworthy advancement in the procedure for ROP screening. We intend to further investigate, in a larger cohort of preterm infants, whether weight/WSDS at first detection of ROP is a useful risk predictor in clinical ROP screening praxis.
Acknowledgments
Special thanks to Johan Stockenberg for his assistance regarding the statistical analysis.
Funding This study was supported by the Herman Svensson Foundation, the Swedish Medical Research Council (grant numbers 2011–2432 and 2008-2842), government grants (grant numbers ALFGB-137491 and ALFGB-21611), VINNOVA (grant numbers 2009-01152 and 2009-00221), and the Torsten and Ragnar Söderberg Foundation.
Footnotes
Contributors PL analysed the data and takes responsibility for the integrity of the data and the accuracy of the data analysis. PL, CL, LEHS, A-LH and AH wrote the manuscript. ÅW was responsible for the collection of data. ÅW reviewed the manuscript.
Competing interests None.
Ethics approval Regional ethical board in Gothenburg.
Provenance and peer review Not commissioned; externally peer reviewed.
References
- 1.Hack M, Fanaroff AA. Outcomes of children of extremely low birthweight and gestational age in the 1990’s. Early Hum Dev. 1999;53:193–218. doi: 10.1016/s0378-3782(98)00052-8. [DOI] [PubMed] [Google Scholar]
- 2.Austeng D, Kallen KB, Ewald UW, et al. Incidence of retinopathy of prematurity in infants born before 27 weeks’ gestation in Sweden. Arch Ophthalmol. 2009;127:1315–19. doi: 10.1001/archophthalmol.2009.244. [DOI] [PubMed] [Google Scholar]
- 3.Darlow BA, Hutchinson JL, Henderson-Smart DJ, et al. Prenatal risk factors for severe retinopathy of prematurity among very preterm infants of the Australian and New Zealand Neonatal Network. Pediatrics. 2005;115:990–6. doi: 10.1542/peds.2004-1309. [DOI] [PubMed] [Google Scholar]
- 4.Sears JE, Pietz J, Sonnie C, et al. A change in oxygen supplementation can decrease the incidence of retinopathy of prematurity. Ophthalmology. 2009;116:513–18. doi: 10.1016/j.ophtha.2008.09.051. [DOI] [PubMed] [Google Scholar]
- 5.Arroe M, Peitersen B. Retinopathy of prematurity: review of a seven-year period in a Danish neonatal intensive care unit. Acta Paediatr. 1994;83:501–5. doi: 10.1111/j.1651-2227.1994.tb13067.x. [DOI] [PubMed] [Google Scholar]
- 6.Klinger G, Levy I, Sirota L, et al. Outcome of early-onset sepsis in a national cohort of very low birth weight infants. Pediatrics. 2010;125:e736–40. doi: 10.1542/peds.2009-2017. [DOI] [PubMed] [Google Scholar]
- 7.Fortes Filho JB, Eckert GU, Procianoy L, et al. Incidence and risk factors for retinopathy of prematurity in very low and in extremely low birth weight infants in a unit-based approach in southern Brazil. Eye (London, England) 2009;23:25–30. doi: 10.1038/sj.eye.6702924. [DOI] [PubMed] [Google Scholar]
- 8.Austeng D, Kallen KBM, Hellstrom A, et al. Natural History of retinopathy of prematurity in infants born before 27 weeks’ Gestation in Sweden. Arch Ophthalmol. 2010;128:1289–94. doi: 10.1001/archophthalmol.2010.234. [DOI] [PubMed] [Google Scholar]
- 9.Hardy RJ, Palmer EA, Dobson V, et al. Risk analysis of prethreshold retinopathy of prematurity. Arch Ophthalmol. 2003;121:1697–701. doi: 10.1001/archopht.121.12.1697. [DOI] [PubMed] [Google Scholar]
- 10.Hagadorn JI, Richardson DK, Schmid CH, et al. Cumulative illness severity and progression from moderate to severe retinopathy of prematurity. J Perinatol. 2007;27:502–9. doi: 10.1038/sj.jp.7211780. [DOI] [PubMed] [Google Scholar]
- 11.Yang MB, Donovan EF, Wagge JR. Race, gender, and clinical risk index for babies (CRIB) score as predictors of severe retinopathy of prematurity. J AAPOS. 2006;10:253–61. doi: 10.1016/j.jaapos.2006.01.004. [DOI] [PubMed] [Google Scholar]
- 12.Allegaert K, Vanhole C, Casteels I, et al. Perinatal growth characteristics and associated risk of developing threshold retinopathy of prematurity. J AAPOS. 2003;7:34–7. doi: 10.1067/mpa.2003.S1091853102420150. [DOI] [PubMed] [Google Scholar]
- 13.Lofqvist C, Andersson E, Sigurdsson J, et al. Longitudinal postnatal weight and insulin-like growth factor I measurements in the prediction of retinopathy of prematurity. Arch Ophthalmol. 2006;124:1711–18. doi: 10.1001/archopht.124.12.1711. [DOI] [PubMed] [Google Scholar]
- 14.Fortes Filho JB, Bonomo PP, Maia M, et al. Weight gain measured at 6 weeks after birth as a predictor for severe retinopathy of prematurity: study with 317 very low birth weight preterm babies. Graefes Arch Clin Exp Ophthalmol. 2009;247:831–6. doi: 10.1007/s00417-008-1012-3. [DOI] [PubMed] [Google Scholar]
- 15.Binenbaum G, Ying GS, Quinn GE, et al. A clinical prediction model to stratify retinopathy of prematurity risk using postnatal weight gain. Pediatrics. 2011;127:e607–14. doi: 10.1542/peds.2010-2240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hellstrom A, Hard AL, Engstrom E, et al. Early weight gain predicts retinopathy in preterm infants: new, simple, efficient approach to screening. Pediatrics. 2009;123:E638–45. doi: 10.1542/peds.2008-2697. [DOI] [PubMed] [Google Scholar]
- 17.Hellstrom A, Engstrom E, Hard AL, et al. Postnatal serum insulin-like growth factor I deficiency is associated with retinopathy of prematurity and other complications of premature birth. Pediatrics. 2003;112:1016–20. doi: 10.1542/peds.112.5.1016. [DOI] [PubMed] [Google Scholar]
- 18.Loqvist C, Hansen-Pupp I, Andersson E, et al. Validation of a new Retinopathy of prematurity screening method monitoring longitudinal postnatal weight and Insulinlike growth factor I. Arch Ophthalmol. 2009;127:622–7. doi: 10.1001/archophthalmol.2009.69. [DOI] [PubMed] [Google Scholar]
- 19.Binenbaum G, Ying GS, Quinn GE, et al. The CHOP postnatal weight gain, birth weight, and gestational age retinopathy of prematurity risk model. Arch Ophthalmol. 2012;130:1560–5. doi: 10.1001/archophthalmol.2012.2524. [DOI] [PubMed] [Google Scholar]
- 20.Eckert GU, Fortes Filho JB, Maia M, et al. A predictive score for retinopathy of prematurity in very low birth weight preterm infants. Eye (London, England) 2012;26:400–6. doi: 10.1038/eye.2011.334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gole GA, Ells AL, Katz X, et al. The international classification of retinopathy of prematurity revisited. Arch Ophthalmol. 2005;123:991–9. doi: 10.1001/archopht.123.7.991. [DOI] [PubMed] [Google Scholar]
- 22.Good WV, Hardy RJ, Dobson V, et al. Revised indications for the treatment of retinopathy of prematurity—Results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121:1684–96. doi: 10.1001/archopht.121.12.1684. [DOI] [PubMed] [Google Scholar]
- 23.Marsal K, Persson PH, Larsen T, et al. Intrauterine growth curves based on ultrasonically estimated foetal weights. Acta Paediatr. 1996;85:843–8. doi: 10.1111/j.1651-2227.1996.tb14164.x. [DOI] [PubMed] [Google Scholar]
- 24.Kleberg A, Warren I, Norman E, et al. Lower stress responses after newborn individualized developmental care and assessment program care during eye screening examinations for retinopathy of prematurity: A randomized study. Pediatrics. 2008;121:E1267–E78. doi: 10.1542/peds.2006-2510. [DOI] [PubMed] [Google Scholar]
- 25.Stevens B, Johnston C, Petryshen P, et al. Premature infant pain profile: Development and initial validation. Clin J Pain. 1996;12:13–22. doi: 10.1097/00002508-199603000-00004. [DOI] [PubMed] [Google Scholar]