Abstract
Degeneration of the intervertebral disc is characterized by changes in proteoglycan status, loss of bound water molecules, decreased tissue osmotic pressure and a resulting mechanical failure of the disc. A similar spectrum of changes is evident in osteoarthritic articular cartilage. When healthy, resident cells in these skeletal tissues respond to applied mechanical loads by regulating their own osmotic state and the hydration of the extracellular matrix. The transcription factor Tonicity-Responsive Enhancer Binding Protein (TonEBP or NFAT5) is known to mediate the osmoadaptive response in these and other tissues. While the molecular basis of how osmotic loading controls matrix homeostasis is not completely understood, TonEBP regulates the expression of aggrecan and β1,3-glucoronosyltransferase in nucleus pulposus cells, in addition to targets that allow for survival under hypertonic stress. Moreover, in chondrocytes, TonEBP controls expression of several collagen subtypes and Sox9, a master regulator of aggrecan and collagen II expression. Thus, TonEBP-mediated regulation of the matrix composition allows disc cells and chondrocytes to modify the extracellular osmotic state itself. On the other hand, TonEBP in immune cells induces expression of TNF-α, IL-6 and MCP-1, pro-inflammatory molecules closely linked to matrix catabolism and pathogenesis of both disc degeneration and osteoarthritis, warranting investigations of this aspect of TonEBP function in skeletal cells. In summary, the TonEBP system, through its effects on extracellular matrix and osmoregulatory genes can be viewed primarily as a protective or homeostatic response to physiological loading.
Keywords: Intervertebral disc, TonEBP, Osmoregulation, Proteoglycan-rich matrix, Cartilage, Disc degeneration
1. Introduction
Both back pain and osteoarthritis (OA) are common, costly, and painful conditions (Katz, 2006; Lee et al., 2013). As the second most frequent neurological ailment in the United States, back pain causes Americans to spend upwards of $100 billion annually on treatment (Katz, 2006). This type of chronic pain is closely linked to degenerative changes that impair function of tissues of the intervertebral disc, the soft tissue between vertebrae. Intervertebral disc degeneration is characterized by changes in proteoglycan status, loss of bound water molecules, decreased tissue osmotic pressure and a resulting decline in the ability of the tissues to absorb biomechanical forces (Haefeli et al., 2006; Le Maitre et al., 2007). Over 96% of people age 60 and above exhibit signs of disc degeneration on autopsy, making it an almost ubiquitous feature of aging (Lawrence, 1969; Miller et al., 1988). Not surprisingly, the same spectrum of changes is evident in degeneration of another proteoglycan-rich tissue, the articular cartilage. Indeed, in OA, disease progression is closely linked to the loss of aggrecan and decrease in tissue osmolarity1 (Wheaton et al., 2004; Hani et al., 2014). In the disc and articular cartilage, resident cells are able to respond to applied mechanical loads by regulating their own osmotic state and the hydration status of the extracellular matrix.
While the components of matrices are similar in disc and cartilage, the proportion of the individual matrix components is different, resulting in tissues with differing architecture and properties. The intervertebral disc is composed of three tissue types: the inner nucleus pulposus (NP), a fibrocartilagenous annulus fibrosus (AF), and cartilage endplates (CEP) that cover the contiguous vertebrae. In terms of matrix composition, there are more similarities between NP, CEP and articular cartilage than any one of them with the AF. The disc provides mobility to the spine during flexion, extension and rotation. In the adult disc, NP cells are sparse and reside within a gelatinous network of proteoglycans and collagens. The AF comprises concentric loops of collagen I fibers in the outer aspect of the tissue, transitioning to a mixture of collagen I and II in the inner AF and the NP (Roberts and Urban, 2011). While the most abundant proteoglycan is aggrecan, smaller amounts of versican, biglycan, decorin, and fibromodulin are also present (Cs-Szabo et al., 2002). On the other hand, the articular cartilage provides an almost frictionless articulating diarthrodial joint surface. Long-lived chondrocytes are sparsely distributed within an abundant extracellular matrix, which is composed mainly of aggrecan and collagen II along with decorin, biglycan, and fibromodulin (Roughley and Lee, 1994).
The high proteoglycan content of the disc and the articular cartilage accounts for elevated water content in these tissues; the percentage water of the NP and AF is approximately 77% and 70%, respectively (Roberts and Urban, 2011). The term solid–fluid phase biphasic mixture was used by Q. Wang et al. (2013) to describe the composite structure of cartilage and a similar term could be used to describe the fluid phase of the disc (Q. Wang et al., 2013).Water molecules exist in both tissues in free and bound forms: the bound water reflects the charge density distribution mediated by both proteoglycans and to a lesser extent the fibrous proteins, while the free water status is dependent on both the hydrostatic as well as the osmotic pressure. Within this hydrostatically loaded environment, cells of both the disc and the synovial joint have evolved mechanisms to regulate water transport and the external osmotic pressure. This mini-review describes the molecular mechanisms by which cells of the disc and articular cartilage respond to osmotic changes, with special focus on the role of the osmo-responsive transcription factor TonEBP or nuclear factor of activated T cells 5 (NFAT5) in regulating cell function and tonicity2.
2. Hyperosmolarity promotes matrix synthesis in the intervertebral disc and articular cartilage
2.1. The intervertebral disc
Reports from a number of laboratories indicate that the intervertebral disc is hyperosmolar when compared to other tissues. Values reported vary from 430 to 496 mOsm (Ishihara et al., 1997; van Dijk et al., 2011). This unusually high extracellular osmotic pressure affects both cell function and matrix synthesis. Ishihara and colleagues were the first to demonstrate this relationship in disc tissue using bovine NP explants (Ishihara et al., 1997). Upon addition of NaCl to the culture medium to increase its osmolarity, an observed increase in [35S]sulfate incorporation rate suggested an overall elevation in proteoglycan synthesis. Importantly, since sucrose produced a similar result, it was inferred that the stimulatory effect on proteoglycan synthesis was related to the medium tonicity rather than the presence of Na+ or Cl− ions. Subsequent work, using isolated NP cells, indicated that glycosaminoglycan (GAG) production and sulfate incorporation rates were highest in medium at 370 mOsm(Takeno et al., 2007). Later, van Dijk et al. showed that when polyethylene glycol (PEG) was used to increase medium tonicity to 570 mOsm, preservation of NP cell phenotype (as measured by aggrecan and collagen expression) was better than when sucrose was the osmolyte (van Dijk et al., 2011, 2013).
How extracellular osmolarity regulates the expression levels of specific matrix molecules has received intense study in recent years. In human NP and AF cells, expression levels of aggrecan and collagen II were increased in cells under hyperosmotic conditions (500 mOsm compared to 400 mOsm control), while collagen I expression was downregulated (Wuertz et al., 2007). Similarly, in bovine NP cells, an increase in medium osmolarity from 300 to 500 mOsm increased aggrecan expression, while decreasing levels of MMP-3 mRNA (Neidlinger-Wilke et al., 2012). To relate osmolarity to loading, rabbit discs were cultured under hyper-osmotic condition (485 mOsm 8 h/day) and before measuring proteoglycan synthesis (Haschtmann et al., 2006).While loading did not influence proteoglycan content or disc cell viability, after 28 days in culture, the raised osmotic pressure prevented aberrant overexpression of collagen I and appeared to influence aggrecan expression. More recently, Spillekom et al. found that expression of brachyury, a phenotypic indicator of the NP, as well as aggrecan and GAG synthesis was at optimal levels when cells were cultured in medium adjusted to 400 mOsm/L (Spillekom et al., 2014). Although there are few studies of the osmotic responsiveness of the AF, Chen et al. showed that in a hyperosmotic environment (450 mOsm), there were decreased levels of aggrecan and collagen II in the transition zone of the disc. Incubation in either hypoosmotic or hyperosmotic media increased mRNA levels of the small proteoglycans bigycan and decorin in transition zone cells, while both conditions decreased mRNA levels of these transcripts in the NP (Chen et al., 2002). The above studies provide strong evidence that cells of the intervertebral disc, especially those of the NP, respond to environmental osmotic changes by altering their matrix production. The 1997 study by Ishihara et al. demonstrated that maximum rates of sulfated GAG production resulted from bringing medium osmolarity to an approximately physiological level (430 mOsm), suggesting that osmotic changes accompanying tissue degeneration may affect matrix production rate.
2.2. Articular cartilage
In a 1986 study, Schneiderman et al. evaluated the effects of mechanical and osmotic loading on [35S]sulfate incorporation into the GAGs of the femoral cartilage (Schneiderman et al., 1986). These workers demonstrated that both types of pressure decreased incorporation and that this was not due to changes in solute transport. A more detailed study was performed by Bayliss and Urban, who questioned how increases in extracellular osmolarity impacted proteoglycan synthesis in both load-bearing (human articular cartilage) and non-loadbearing (bovine nasal cartilage) tissues (Urban and Bayliss, 1989). In contrast to the earlier work (above), they showed that, in both tissue types, ionic strength positively influenced [35S]sulfate and 3H-proline incorporation rates, subsequently reporting that incorporation rates were maximized between 350 and 400 mOsm; values that are similar to that of native cartilage (Urban et al., 1993).
The relationship between osmotic loading and the impact of biomechanical forces on articular cartilage has also received attention. Under hyperosmotic conditions (560–680 mOsm), there was a decrease in aggrecan promoter activity and mRNA expression following loading for 24 h. Since the decrease in promoter activity was relieved when osmotic loading was performed in a cyclic manner, the authors suggested that both the osmotic environment and the applied force regulated the transcriptional response (Palmer et al., 2001). Finally, there is evidence linking osmotic pressure to expression of the chondrocyte phenotype. Tew et al. revealed that hyperosmotic treatment increased the mRNA stability of SOX9 and other genes important to chondrocyte function as well as the activity of a COL2A1 enhancer-driven reporter (Tew et al., 2009, 2011). In an equine model of OA, it was shown that the osmotically driven increase in SOX9 mRNA stability was disease dependent (Peffers et al., 2010).
Thus, hyper-osmolarity regulates expression of the most critical matrix genes in both the NP and articular cartilage: aggrecan and collagen II. While the molecular basis of this response has not been completely characterized, within the last decade increasing attention has been focused on the role of transcription factor TonEBP in mediating the osmotic response of these connective tissues.
3. The osmotic response of both chondrocytes and NP cells is mediated by TonEBP
3.1. General osmotic response of TonEBP
Shifts in the extracellular osmotic status of a tissue are common and elicit a robust and coordinated molecular response. A hypertonic challenge activates membrane electrolyte transporters, which, by balancing intercellular and extracellular solute concentrations prevent large volume changes. This is a short-term fix, however, as the resulting change in osmotic pressure can influence or even damage protein and DNA structure and function and trigger autophagy, senescence or apoptosis (Cheung and Ko, 2013). To prevent these potentially damaging events, the transcription factor TonEBP drives the expression of a suite of genes, which facilitate the exchange of accumulated charged ions for small organic non-ionic osmolytes. These well-described TonEBP targets include aldose reductase (AR), betaine-γ-amino butyric acid transporter (BGT1), sodium myo-inositol transporter (SMIT), and taurine transporter (TauT), which regulate the transport of sorbitol, betaine, myo-inositol, and taurine across the plasma membrane, respectively (Yancey et al., 1982; Garcia-Perez and Burg, 1991).While signaling events that immediately follow osmotic shifts and converge at TonEBP have been studied (Tsai et al., 2007; Gallazzini et al., 2011; Zhou et al., 2013), the identity of upstream a cell surface osmosensor/s complex in mammals remains elusive. The TRPV4/AQP4 complex is proposed as an osmosensor in astrocytes, but, it responds only to hypotonic shifts (Benfenati et al., 2011). More relevant to this discussion are the findings that integrin α1β1, α6β4 (Jauliac et al., 2002; Moeckel et al., 2006) as well as stretching(Scherer et al., 2014) activate TonEBP signaling; suggesting that mechanical deformation can directly influence TonEBP. Regardless of the nature of upstream osmosensor, following hypertonic stress, TonEBP is activated via increased mRNA expression and stabilization (Cai et al., 2005) accompanied by a rise in overall protein abundance, nuclear shuttling (Woo et al., 2000), and C-terminal (AD1, AD2 and AD3) transactivation domain activity (Lee et al., 2003). In the nucleus, TonEBP homodimers are formed which preferentially bind to the tonicity responsive enhancer (TonE) consensus sequence in target gene promoters (López-Rodríguez et al., 2001; Stroud et al., 2002). The osmolarity of the environment determines TonEBP nuclear to cytoplasmic abundance, and in this way regulates the status of its activity and target gene expression (Woo et al., 2000; Tong et al., 2006).
Loading of the hydrostatically pressurized articular cartilage and the intervertebral disc results in minute-to-minute fluctuations in extracellular osmolarity (Urban, 1994; Roberts et al., 1998). From this perspective, it is not surprising that TonEBP is robustly expressed in both NP and AF tissues, as was first reported by our group (Tsai et al., 2006). In that study, following silencing of TonEBP, we demonstrated a decreased survival of NP cells exposed to hypertonic medium. This observation lent strong support to the hypothesis that this transcription factor is necessary for NP cell viability in the hypertonic milieu of the disc. Furthermore, hyperosmotic challenge led to increases in mRNA levels of some of the most well studied TonEBP target genes – SMIT, BGT1, and TauT. In this and later studies, our group defined critical matrix-related targets of TonEBP in the NP (discussed below) as well as the water channel protein AQP2 (Gajghate et al., 2009). Moreover, TonEBP has been shown to be expressed in both human articular chondrocytes (van der Windt et al., 2010) and the chondrogenic cell line ATDC5 (Caron et al., 2013). Thus, TonEBP is a critical factor in the maintenance of cell health in the osmotically demanding environments of the NP and articular cartilage.
3.2. TonEBP regulates matrix genes in the intervertebral disc and articular cartilage
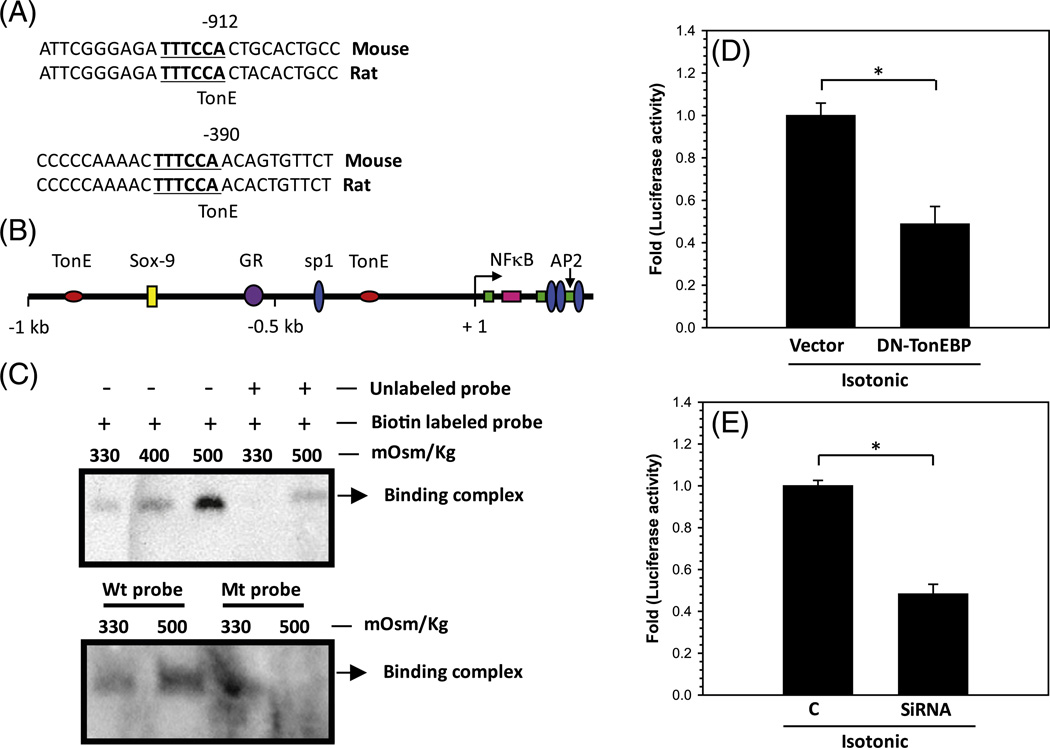
Importantly, aside from regulating intracellular osmolarity, TonEBP is a key regulator of the content and, therefore, osmotic status of the extracellular matrix. Tsai et al. identified a consensus TonE element in the promoter of the aggrecan gene that functionally interacted with TonEBP (Fig. 1) (Tsai et al., 2006, 2007). Suppression of TonEBP activity by dominant negative protein or siRNA dramatically decreased aggrecan promoter activity. This was the first evidence in NP cells that TonEBP transcriptionally regulates matrix genes. Later investigations by our group showed that TonEBP also regulated the expression of β1,3-glucoronosyltransferase (GlcAT-I), an enzyme required for the synthesis of chondroitin sulfate chains of aggrecan (Hiyama et al., 2009). A follow-up study revealed that BMP-2 and TGF-β-mediated induction of GlcAT-I was also dependent on TonEBP activity (Hiyama et al., 2010). Based on these findings, it is clear that by controlling the expression and synthesis of aggrecan, TonEBP permits disc cells to autoregulate and adapt to their hyperosmotic state (Fig. 2).
Fig. 1.
Regulation of aggrecan gene promoter activity by TonEBP. A,DNA sequence of the promoter region of rat and mouse aggrecan gene. TonE consensus sequence is marked in bold and underlined. B, promoter organization of the rat aggrecan gene. The transcription start site is marked as + 1. TonE sites are shown as ovals on either side of a conserved Sox-9 binding site. C, electromobility shift assay to examine functional binding of TonEBP to TonE motif in the rat aggrecan gene promoter. An oligonucleotide probe containing the TonE motif (−912 b) in the rat aggrecan promoter was incubated with nuclear extracts from rat nucleus pulposus cells cultured under isotonic and hypertonic (400 and 500 mosmol/kg) conditions, and binding was detected using chemiluminescence. Specificity was confirmed by inclusion of excess unlabeled wild type probe or a probe containing mutation in the TonE site (Mt probe) in the binding reaction. The binding signal is significantly diminished when either a wild type competitor probe or a mutant probe is used. D, nucleus pulposus cells were co-transfected with DN-TonEBP and aggrecan reporter plasmids. Twenty-four hours after transfection, cells were cultured in isotonic medium for 24 h and luciferase activity measured. Expression of DN-TonEBP results in decreased aggrecan promoter activity compared with control cells that receive empty backbone vector. E, aggrecan promoter construct was transiently transfected into siRNA expressing and control cells (C) and reporter activity measured in isotonic media. Compared with control cells, the silenced nucleus pulposus cells elicit a marked reduction in aggrecan reporter activity. Data represent mean ± S.D. from three independent experiments, performed in triplicate (n= 3); *, p b 0.05. This research was originally published in The Journal of Biological Chemistry. Tsai TT, Danielson KG, Guttapalli A, Oguz E, Albert TJ, Shapiro IM, Risbud MV. TonEBP/OREBP is a regulator of nucleus pulposus cell function and survival in the intervertebral disc. J Biol Chem. 2006; 1;281(35):25416-24. © the American Society for Biochemistry and Molecular Biology.
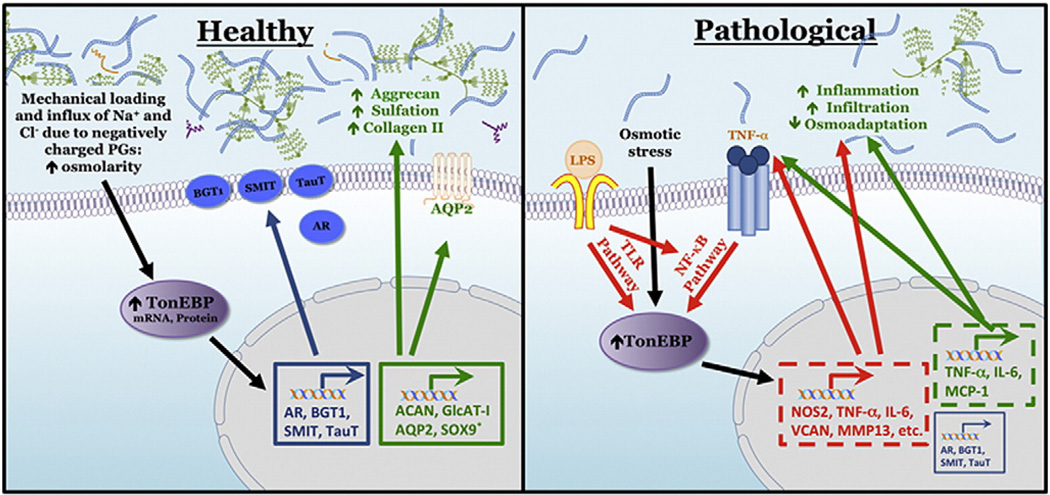
Fig. 2.
Schematic representation of the osmotic response in healthy (left panel) and degenerative (right panel) disc or articular cartilage tissues. In healthy tissue, hyperosmolarity results from high negative charge of proteoglycans resulting in influx of NaCl. Hyperosmolarity results in robust increase in TonEBP mRNA, protein, and nuclear shuttling. TonEBP binds to TonE sites in target promoters to drive expression of osmotic response genes (blue) (AR, BGT1, SMIT, TauT), protecting against cellular damage during hypertonic stress. Similarly, TonEBP induces transcription of genes involved in matrix homeostasis (green) (ACAN, GlcAT-I, AQP2, Sox9) to autoregulate the extracellular osmotic environment. In the pathological state, activation of TLR or NF-κB pathways induce TonEBP to act on a specific set of targets (red). Activity on osmoadaptation targets (blue) is decreased with this type of activation. Osmotic stress may also induce TonEBP activation of pro-inflammatory targets (green). It should be noted that in disc and cartilage relationship between pathological stimuli and TonEBP has not been studied yet. *Studies have not addressed whether TonEBP binds TonE in Sox9. AR, aldose reductase; BGT1, betaine-g-amino butyric acid transporter; SMIT, sodium myo-inositol transporter; TauT, taurine transporter (TauT); ACAN, Aggrecan; GlcAT-I, β1,3-glucuronosyl transferase 1; AQP2, Aquaporin 2; TLR, Toll-like receptor; TNF-a, tumor necrosis factor-a; IL-6, interleukin 6;NOS2, nitric oxide synthase 2; VCAN, versican; MMP-13, matrix metallopeptidase 13. Targets in dashed border indicate results that have not been verified in NP cells or chondrocytes.
To date, there have been a limited number of investigations concerning the function of TonEBP in chondrocytes and the role of this transcription factor in the maintenance of cartilage structure. Van der Windt et al. examined the effect of osmolarity on human articular chondrocytes showing that physiological osmolarity (380 mOsm) improved the expression of multiple markers including aggrecan, sox9 and collagen II and suppressed the expression of collagen I (van der Windt et al., 2010). While the presence of TonE elements in these genes was not addressed, induction was found to be TonEBP-dependent. Similarly, in ATDC5 cells hyperosmotic conditions promoted the expression of sox9, collagen IIa1, collagen Xa1, Runx2, and aggrecan along with increased GAG content (Caron et al., 2013). Noteworthy, knockdown of TonEBP significantly decreased expression of these markers, indicating that an osmo-dependent TonEBP pathway was involved. Since Sox9 is a critical regulator of chondrocyte differentiation and transcriptionally controls aggrecan and collagen expression (Lefebvre and Smits, 2005), this study pointed to a role for TonEBP in development. Caron et al. (2013) noted that when TonEBP was silenced, the osmotically-induced increase in Sox9 expression was blunted (Caron et al., 2013).While this study further implied a linkage between osmolarity and cartilage development, the details of that relationship require further clarification.
In summary, although it is clear that activity of TonEBP is linked to both cartilage and disc function, much remains to be learned concerning its regulation and matrix-related targets. It should be noted that two TonEBP null mice have previously been developed; however, perinatal lethality has mitigated their use for studies mentioned above (López-Rodríguez et al., 2004; Mak et al., 2011). A TonEBPfl/fl mouse has recently been developed which should add significantly to current knowledge of the function of this critical transcription factor in cartilage and the intervertebral disc (Wiig et al., 2013).
4. Degeneration, inflammation, and the TonEBP-mediated response
Both degenerative disc disease and OA are characterized by decreased levels of aggrecan and collagen II along with increased expression of the pro-catabolic MMP and ADAMTS enzymes (Im et al., 2007; Le Maitre et al., 2007; Verma and Dalal, 2011; Wei et al., 2014). In the context of intervertebral disc degeneration, it is likely that the increase in activity of these enzymes is triggered by the pro-inflammatory cytokines IL-1, IL-6, TNF-α, and IL-17 (Risbud and Shapiro, 2014); in OA, a similar increase in cytokines has been observed (Lee et al., 2013). As discussed earlier, a decline in water-binding aggrecan during degeneration causes NP tissue dehydration leading to functional deficits and, often, pain (Nguyen et al., 2008; Fenty et al., 2013). Loss of water is also identified as an early feature of OA (Hani et al., 2014; Souza et al., 2014). Not surprisingly, a number of workers have explored the relationship between inflammation, environmental osmolarity, and activity of TonEBP (Fig. 2).
Esensten et al. (2005) demonstrated that TonEBP drove the expression of a key pro-inflammatory cytokine in hypertonic culture, binding to the TNF-α promoter via a TonE element (Esensten et al., 2005). In addition to TNF-α, hypertonicity-dependent induction of IL-6 has been shown (Ueno et al., 2013). It is important to note that since increased osmolarity influences expression of IL-6 in a post-transcriptional manner (Tew et al., 2011), it is likely that micro RNAs are involved along with TonEBP. Importantly, since these inflammatory cytokines and others promote degeneration by enhancing the chemotaxis of immune cells into the disc (J. Wang et al., 2013), it was not surprising to find that a functional TonE element was also present in the 5′ flanking region of the MCP-1 (CCL2) gene and that the expression was increased under hypertonic conditions (Kojima et al., 2010). Expression of MCP-1 is closely tied to the pathogenesis of both disc degeneration (Phillips et al., 2013) and OA (Chou et al., 2013; Liu et al., 2013). This hypertonicity-dependent increase in MCP-1 promoter activity was ablated when the TonE was deleted (Kojima et al., 2010). Küper et al. (2012) provided further support for MCP-1 as TonEBP target by demonstrating that an increase in glucose or mannitol concentration increased MCP-1 expression (Küper et al., 2012). This discovery, that TonEBP drives expression of pro-inflammatory targets in addition to osmoregulatory genes, may have important implications in tissues such as NP and articular cartilage, which experience large daily swings in both osmotic pressure due to changes in applied forces resulting from locomotion (Boos et al., 1993; Wuertz et al., 2007).
Of equal importance in the context of both degenerative diseases is the discovery that TonEBP can be activated by conditions other than osmolarity (Hiyama et al., 2009; Gogate et al., 2012; Halterman et al., 2012) (Fig. 2). For example, TonEBP mediates the effects of Toll-like receptors (TLRs) in macrophages by up-regulating target genes including Nos2, Tnf and Il-6, essential for antimicrobial immunity (Buxadé et al., 2012). TLR is a family of receptors that mediate cellular response toward pathogenic molecules distinct from host molecules that are commonly referred to as pathogen-associated molecular patterns (PAMPs) e.g. LPS and endogenous host molecules arising from cell death and damage termed as danger associated molecular patterns (DAMPs) e.g. HMGB1. It is important to recognize that NP cells and chondrocytes express multiple TLRs of which TLR2 and TLR4 are studied the most (Liu-Bryan and Terkeltaub, 2010; Rajan et al., 2013). In addition, TonEBP influences a number of targets relevant to matrix homeostasis in response to LPS-mediated TLR activation: Vcan, Has1, Fn1, Timp1, Tnc, and Mmp13 (Buxadé et al., 2012). TonEBP protein was shown to be up-regulated and localized to the nucleus in fibroblast like synoviocytes from rheumatoid arthritis patients and this expression was further enhanced by TNF-α or IL1-β (Yoon et al., 2011). Furthermore, TonEBP directly enhances the activity of the NF-κB pathway in response to LPS treatment and directly interacts with p65 under hypertonic conditions (Roth et al., 2010). These findings beg the question: Does TonEBP play a role in disease pathogenesis? Indeed, for OA cartilage, a connection between LPS and induction of MMPs and ADAMTSs has been well established (Kim et al., 2006; Lee et al., 2013); likewise, in the intervertebral disc, the relationship between a broad range of pro-inflammatory cytokines, LPS and matrix degeneration has been elucidated (Wang et al., 2011; Fujita et al., 2012; Rajan et al., 2013; Tian et al., 2013; Maidhof et al., 2014). Based on these observations and studies of immune cell function, it is likely that TonEBP can be activated by at least two distinct mechanisms as follows: Firstly, the canonical pathway, which responds to changes in the osmotic status of the extracellular milieu. Secondly, TonEBP can be activated in response to receptor (TLR) activation. The possible pathways for activation are shown in Fig. 2. While the noncanonical pathways need to be verified for cartilage and the intervertebral disc, Kimet al. found that when macrophages were pretreated with LPS, the response of osmotic-response genes (AR, BGT1, SMIT) was mitigated. On the other hand, cells pretreated with hyperosmotic medium exhibited a blunted response to TLR-stimuli as measured by IL-6 levels (Kim et al., 2013). Should these findings be extended to the previously described osmo-dependent TonEBP targets in disc and cartilage, such as Aggrecan, Sox9, and GlcAT-I, it follows that their regulation may be disturbed in an inflammatory microenvironment, such as that present during degeneration.
Finally, it should be noted that unlike the tonicity-mediated shifts in electrolytes regulated by rate-limiting sodium transporters, TonEBP responds to hypertonic environments by driving the accumulation of small polyhydric alcohols (myoinositol and sorbitol), methylamines (betaine) and amino acids (taurine) through controlling expression of target genes that are involved in either the synthesis or transport of these molecules (Handler and Kwon, 2001). The advantage posed by this response is that these compounds do not perturb the activities of enzymes and macromolecules in the crowded cytosol. Indeed, the TonEBP system generates chaperons that serve to structurally protect osmotically-sensitive proteins (Woo et al., 2002; Tsai et al., 2006). From this perspective, the TonEBP system through its effects on extracellular matrix and osmoregulatory genes can be viewed primarily as a protective or homeostatic response to counteract the effects of chronic stress. Stress, either as a result of extended changes in tonicity or as an inflammatory response, would be expected to trigger pathogenic events that lead to system failure and tissue degradation. From this perspective elucidating the mechanism by which TonEBP influences tissue function will provide new insights into treatment of the commonest skeletal diseases.
Acknowledgments
This work was supported by grants from the National Institutes of Health AR055655, AR064733 and AR050087. Zariel I. Johnson is supported by T32 AR052273.
Footnotes
Osmolarity refers to the total concentration of both cell-penetrating and non-cell-penetrating solutes in extracellular environment.
Tonicity refers to only the concentration of non-cell-penetrating solutes
References
- Benfenati V, Caprini M, Dovizio M, Mylonakou MN, Ferroni S, Ottersen OP, Amiry-Moghaddam M. An aquaporin-4/transient receptor potential vanilloid 4 (AQP4/TRPV4) complex is essential for cell-volume control in astrocytes. Proc. Natl. Acad. Sci. 2011 Feb 8;108(6):2563–2568. doi: 10.1073/pnas.1012867108. http://dx.doi.org/10.1073/pnas.1012867108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boos N, Wallin A, Gbedegbegnon T, Aebi M, Boesch C. Quantitative MR imaging of lumbar intervertebral disks and vertebral bodies: influence of diurnal water content variations. Radiology. 1993;188:351–354. doi: 10.1148/radiology.188.2.8327677. http://dx.doi.org/10.1148/radiology.188.2.8327677. [DOI] [PubMed] [Google Scholar]
- Buxadé M, Lunazzi G, Minguillón J, Iborra S, Berga-Bolaños R, Del Val M, Aramburu J, López-Rodríguez C. Gene expression induced by Toll-like receptors in macrophages requires the transcription factor NFAT5. J. Exp. Med. 2012;209:379–393. doi: 10.1084/jem.20111569. http://dx.doi.org/10.1084/jem.20111569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Q, Ferraris JD, Burg MB. High NaCl increases TonEBP/OREBP mRNA and protein by stabilizing its mRNA. Am. J. Physiol. Ren. Physiol. 2005;289:F803–F807. doi: 10.1152/ajprenal.00448.2004. http://dx.doi.org/10.1152/ajprenal.00448.2004. [DOI] [PubMed] [Google Scholar]
- Caron MMJ, van der Windt AE, Emans PJ, van Rhijn LW, Jahr H, Welting TJM. Osmolarity determines the in vitro chondrogenic differentiation capacity of progenitor cells via nuclear factor of activated T-cells 5. Bone. 2013;53:94–102. doi: 10.1016/j.bone.2012.11.032. http://dx.doi.org/10.1016/j.bone.2012.11.032. [DOI] [PubMed] [Google Scholar]
- Chen J, Baer AE, Paik PY, Yan W, Setton LA. Matrix protein gene expression in intervertebral disc cells subjected to altered osmolarity. Biochem. Biophys. Res. Commun. 2002;293:932–938. doi: 10.1016/S0006-291X(02)00314-5. http://dx.doi.org/10.1016/S0006-291X(02)00314-5. [DOI] [PubMed] [Google Scholar]
- Cheung CY, Ko BC. NFAT5 in cellular adaptation to hypertonic stress — regulations and functional significance. J. Mol. Signal. 2013;8:5. doi: 10.1186/1750-2187-8-5. http://dx.doi.org/10.1186/1750-2187-8-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou CH, Lee CH, Lu LS, Song IW, Chuang HP, Kuo SY, Wu JY, Chen YT, Kraus VB, Wu CC, Lee MTM. Direct assessment of articular cartilage and underlying subchondral bone reveals a progressive gene expression change in human osteoarthritic knees. Osteoarthr. Cartil. 2013;21:450–461. doi: 10.1016/j.joca.2012.11.016. http://dx.doi.org/10.1016/j.joca.2012.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cs-Szabo G, Ragasa-San Juan D, Turumella V, Masuda K, Thonar EJ, An HS. Changes in mRNA and protein levels of proteoglycans of the anulus fibrosus and nucleus pulposus during intervertebral disc degeneration. Spine (Phila Pa 1976) 2002;27:2212–2219. doi: 10.1097/00007632-200210150-00006. http://dx.doi.org/10.1097/01.BRS.0000029249.02864.5B. [DOI] [PubMed] [Google Scholar]
- Esensten JH, Tsytsykova AV, Lopez-Rodriguez C, Ligeiro FA, Rao A, Goldfeld AE. NFAT5 binds to the TNF promoter distinctly from NFATp, c, 3 and 4, and activates TNF transcription during hypertonic stress alone. Nucleic Acids Res. 2005;33:3845–3854. doi: 10.1093/nar/gki701. http://dx.doi.org/10.1093/nar/gki701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenty M, Crescenzi R, Fry B, Squillante D, Turk D, Maurer PM, Borthakur A. Novel imaging of the intervertebral disk and pain. Glob. Spine. J. 2013;3:127–132. doi: 10.1055/s-0033-1347930. http://dx.doi.org/10.1055/s-0033-1347930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita N, Gogate SS, Chiba K, Toyama Y, Shapiro IM, Risbud MV. Prolyl hydroxylase 3 (PHD3)modulates catabolic effects of tumor necrosis factor-α (TNF-α) on cells of the nucleus pulposus through co-activation of nuclear factor κB (NF-κB)/p65 signaling. J. Biol. Chem. 2012;287:39942–39953. doi: 10.1074/jbc.M112.375964. http://dx.doi.org/10.1074/jbc.M112.375964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gajghate S, Hiyama A, Shah M, Sakai D, Anderson DG, Shapiro IM, Risbud MV. Osmolarity and intracellular calcium regulate aquaporin2 expression through TonEBP in nucleus pulposus cells of the intervertebral disc. J. Bone Miner. Res. 2009;24:992–1001. doi: 10.1359/JBMR.090103. http://dx.doi.org/10.1359/jbmr.090103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallazzini M, Heussler GE, Kunin M, Izumi Y, Burg MB, Ferraris JD. High NaCl-induced activation of CDK5 increases phosphorylation of the osmoprotective transcription factor TonEBP/OREBP at threonine 135, which contributes to its rapid nuclear localization. Mol. Biol. Cell. 2011 Mar 1;22(5):703–714. doi: 10.1091/mbc.E10-08-0681. http://dx.doi.org/10.1091/mbc.E10-08-0681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Perez A, Burg MB. Renal medullary organic osmolytes. Physiol. Rev. 1991;71:1081–1115. doi: 10.1152/physrev.1991.71.4.1081. [DOI] [PubMed] [Google Scholar]
- Gogate SS, Fujita N, Skubutyte R, Shapiro IM, Risbud MV. Tonicity enhancer binding protein (TonEBP) and hypoxia-inducible factor (HIF) coordinate heat shock protein 70 (Hsp70) expression in hypoxic nucleus pulposus cells: role of Hsp70 inHIF-1α degradation. J. Bone. Miner. Res. 2012;27:1106–1117. doi: 10.1002/jbmr.1571. http://dx.doi.org/10.1002/jbmr.1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haefeli M, Kalberer F, Saegesser D, Nerlich AG, Boos N, Paesold G. The course of macroscopic degeneration in the human lumbar intervertebral disc. Spine (Phila Pa 1976) 2006;31:1522–1531. doi: 10.1097/01.brs.0000222032.52336.8e. http://dx.doi.org/10.1097/01.brs.0000222032.52336.8e. [DOI] [PubMed] [Google Scholar]
- Halterman JA, Kwon HM, Wamhoff BR. Tonicity-independent regulation of the osmosensitive transcription factor TonEBP (NFAT5) Am. J. Physiol. Cell Physiol. 2012;302:C1–C8. doi: 10.1152/ajpcell.00327.2011. http://dx.doi.org/10.1152/ajpcell.00327.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Handler JS, Kwon HM. Transcriptional regulation by changes in tonicity. Kidney Int. 2001 Aug;60(2):408–411. doi: 10.1046/j.1523-1755.2001.060002408.x. [DOI] [PubMed] [Google Scholar]
- Hani AFM, Kumar D, Malik AS, Ahmad RMKR, Razak R, Kiflie A. Noninvasive and in vivo assessment of osteoarthritic articular cartilage: a review on MRI investigations. Rheumatol. Int. 2014 doi: 10.1007/s00296-014-3052-9. http://dx.doi.org/10.1007/s00296-014-3052-9. [DOI] [PubMed] [Google Scholar]
- Haschtmann D, Stoyanov JV, Ferguson SJ. Influence of diurnal hyperosmotic loading on the metabolism and matrix gene expression of a whole-organ intervertebral disc model. J. Orthop. Res. 2006;24:1957–1966. doi: 10.1002/jor.20243. http://dx.doi.org/10.1002/jor.20243. [DOI] [PubMed] [Google Scholar]
- Hiyama A, Gajghate S, Sakai D, Mochida J, Shapiro IM, Risbud MV. Activation of TonEBP by calcium controls {beta}1,3-glucuronosyltransferase-I expression, a key regulator of glycosaminoglycan synthesis in cells of the intervertebral disc. J. Biol. Chem. 2009;284:9824–9834. doi: 10.1074/jbc.M807081200. http://dx.doi.org/10.1074/jbc.M807081200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiyama A, Gogate SS, Gajghate S, Mochida J, Shapiro IM, Risbud MV. BMP-2 and TGF-beta stimulate expression of beta1,3-glucuronosyl transferase 1 (GlcAT-1) in nucleus pulposus cells through AP1, TonEBP, and Sp1: role of MAPKs. J. Bone Miner. Res. 2010;25:1179–1190. doi: 10.1359/jbmr.091202. http://dx.doi.org/10.1359/jbmr.091202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Im HJ, Muddasani P, Natarajan V, Schmid TM, Block JA, Davis F, van Wijnen AJ, Loeser RF. Basic fibroblast growth factor stimulates matrix metalloproteinase- 13 via the molecular cross-talk between the mitogen-activated protein kinases and protein kinase Cdelta pathways in human adult articular chondrocytes. J. Biol. Chem. 2007;282:11110–11121. doi: 10.1074/jbc.M609040200. http://dx.doi.org/10.1074/jbc.M609040200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishihara H, Warensjo K, Roberts S, Urban JP. Proteoglycan synthesis in the intervertebral disk nucleus: the role of extracellular osmolality. Am. J. Physiol. 1997;272:C1499–C1506. doi: 10.1152/ajpcell.1997.272.5.C1499. [DOI] [PubMed] [Google Scholar]
- Jauliac S, López-Rodriguez C, Shaw LM, Brown LF, Rao A, Toker A. The role of NFAT transcription factors in integrin-mediated carcinoma invasion. Nat. Cell Biol. 2002 Jul;4(7):540–544. doi: 10.1038/ncb816. [DOI] [PubMed] [Google Scholar]
- Katz JN. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J. Bone Joint Surg. Am. 2006;88(Suppl. 2):21–24. doi: 10.2106/JBJS.E.01273. http://dx.doi.org/10.2106/JBJS.E.01273. [DOI] [PubMed] [Google Scholar]
- Kim HA, Cho ML, Choi HY, Yoon CS, Jhun JY, Oh HJ, Kim HY. The catabolic pathway mediated by Toll-like receptors in human osteoarthritic chondrocytes. Arthritis Rheum. 2006;54:2152–2163. doi: 10.1002/art.21951. http://dx.doi.org/10.1002/art.21951. [DOI] [PubMed] [Google Scholar]
- Kim NH, Hong BK, Choi SY, Moo Kwon H, Cho CS, Yi EC, Kim WU. Reactive oxygen species regulate context-dependent inhibition of NFAT5 target genes. Exp. Mol. Med. 2013;45:e32. doi: 10.1038/emm.2013.61. http://dx.doi.org/10.1038/emm.2013.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima R, Taniguchi H, Tsuzuki A, Nakamura K, Sakakura Y, Ito M. Hypertonicity-induced expression of monocyte chemoattractant protein-1 through a novel cis-acting element and MAPK signaling pathways. J. Immunol. 2010;184:5253–5262. doi: 10.4049/jimmunol.0901298. http://dx.doi.org/10.1155/2012/513015. [DOI] [PubMed] [Google Scholar]
- Küper C, Beck FX, Neuhofer W. NFAT5 contributes to osmolality-induced MCP-1 expression in mesothelial cells. Mediat. Inflamm. 2012;2012:513015. doi: 10.1155/2012/513015. http://dx.doi.org/10.1155/2012/513015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawrence JS. Disc degeneration. Its frequency and relationship to symptoms. Ann. Rheum. Dis. 1969;28:121–138. doi: 10.1136/ard.28.2.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Maitre CL, Pockert A, Buttle DJ, Freemont AJ, Hoyland JA. Matrix synthesis and degradation in human intervertebral disc degeneration. Biochem. Soc. Trans. 2007;35:652–655. doi: 10.1042/BST0350652. http://dx.doi.org/10.1042/BST0350652. [DOI] [PubMed] [Google Scholar]
- Lee S, Do, Colla E, Sheen MR, Na KY, Kwon HM. Multiple domains of TonEBP cooperate to stimulate transcription in response to hypertonicity. J. Biol. Chem. 2003;278:47571–47577. doi: 10.1074/jbc.M308795200. http://dx.doi.org/10.1074/jbc.M308795200. [DOI] [PubMed] [Google Scholar]
- Lee AS, Ellman MB, Yan D, Kroin JS, Cole BJ, van Wijnen AJ, Im H-J. A current review of molecular mechanisms regarding osteoarthritis and pain. Gene. 2013;527:440–447. doi: 10.1016/j.gene.2013.05.069. http://dx.doi.org/10.1016/j.gene.2013.05.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lefebvre V, Smits P. Transcriptional control of chondrocyte fate and differentiation. Birth Defects Res. C Embryo Today. 2005;75:200–212. doi: 10.1002/bdrc.20048. http://dx.doi.org/10.1002/bdrc.20048. [DOI] [PubMed] [Google Scholar]
- Liu SC, Hsu CJ, Fong YC, Chuang SM, Tang CH. CTGF induces monocyte chemoattractant protein-1 expression to enhance monocyte migration in human synovial fibroblasts. Biochim. Biophys. Acta. 2013;1833:1114–1124. doi: 10.1016/j.bbamcr.2012.12.014. http://dx.doi.org/10.1016/j.bbamcr.2012.12.014. [DOI] [PubMed] [Google Scholar]
- Liu-Bryan R, Terkeltaub R. Chondrocyte innate immune myeloid differentiation factor 88-dependent signaling drives procatabolic effects of the endogenous Tolllike receptor 2/Toll-like receptor 4 ligands low molecular weight hyaluronan and high mobility group box chromosomal protein 1 in mice. Arthritis Rheum. 2010 Jul;62(7):2004–2012. doi: 10.1002/art.27475. http://dx.doi.org/10.1002/art.27475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Rodríguez C, Aramburu J, Jin L, Rakeman AS, Michino M, Rao A. Bridging the NFAT and NF-kappaB families: NFAT5 dimerization regulates cytokine gene transcription in response to osmotic stress. Immunity. 2001;15:47–58. doi: 10.1016/s1074-7613(01)00165-0. [DOI] [PubMed] [Google Scholar]
- López-Rodríguez C, Antos CL, Shelton JM, Richardson JA, Lin F, Novobrantseva TI, Bronson RT, Igarashi P, Rao A, Olson EN. Loss of NFAT5 results in renal atrophy and lack of tonicity-responsive gene expression. Proc. Natl. Acad. Sci. U. S. A. 2004;101:2392–2397. doi: 10.1073/pnas.0308703100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maidhof R, Jacobsen T, Papatheodorou A, Chahine NO. Inflammation induces irreversible biophysical changes in isolated nucleus pulposus cells. PLoS One. 2014;9:e99621. doi: 10.1371/journal.pone.0099621. http://dx.doi.org/10.1371/journal.pone.0099621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mak MC, Lam KM, Chan PK, Lau YB, Tang WH, Yeung PKK, Ko BCB, Chung SMS, Chung SK. Embryonic lethality in mice lacking the nuclear factor of activated T cells 5 protein due to impaired cardiac development and function. PLoS One. 2011;6:e19186. doi: 10.1371/journal.pone.0019186. http://dx.doi.org/10.1371/journal.pone.0019186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine (Phila Pa 1976) 1988;13:173–178. [PubMed] [Google Scholar]
- Moeckel GW, Zhang L, Chen X, Rossini M, Zent R, Pozzi A. Role of integrin alpha1beta1 in the regulation of renal medullary osmolyte concentration. Am. J. Physiol. Ren. Physiol. 2006 Jan;290(1):F223–F231. doi: 10.1152/ajprenal.00371.2004. [DOI] [PubMed] [Google Scholar]
- Neidlinger-Wilke C, Mietsch A, Rinkler C, Wilke H-J, Ignatius A, Urban J. Interactions of environmental conditions and mechanical loads have influence on matrix turnover by nucleus pulposus cells. J. Orthop. Res. 2012;30:112–121. doi: 10.1002/jor.21481. http://dx.doi.org/10.1002/jor.21481. [DOI] [PubMed] [Google Scholar]
- Nguyen AM, Johannessen W, Yoder JH, Wheaton AJ, Vresilovic EJ, Borthakur A, Elliott DM. Noninvasive quantification of human nucleus pulposus pressure with use of T1rho-weighted magnetic resonance imaging. J. Bone Joint Surg. Am. 2008;90:796–802. doi: 10.2106/JBJS.G.00667. http://dx.doi.org/10.2106/JBJS.G.00667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer GD, Chao Ph PH, Raia F, Mauck RL, Valhmu WB, Hung CT. Time-dependent aggrecan gene expression of articular chondrocytes in response to hyperosmotic loading. Osteoarthr. Cartil. 2001;9:761–770. doi: 10.1053/joca.2001.0473. http://dx.doi.org/10.1053/joca.2001.0473. [DOI] [PubMed] [Google Scholar]
- Peffers MJ, Milner PI, Tew SR, Clegg PD. Regulation of SOX9 in normal and osteoarthritic equine articular chondrocytes by hyperosmotic loading. Osteoarthr. Cartil. 2010;18:1502–1508. doi: 10.1016/j.joca.2010.08.011. http://dx.doi.org/10.1016/j.joca.2010.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips KLE, Chiverton N, Michael ALR, Cole AA, Breakwell LM, Haddock G, Bunning RAD, Cross AK, LeMaitre CL. The cytokine and chemokine expression profile of nucleus pulposus cells: implications for degeneration and regeneration of the intervertebral disc. Arthritis Res. Ther. 2013;15:R213. doi: 10.1186/ar4408. http://dx.doi.org/10.1186/ar4408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajan NE, Bloom O, Maidhof R, Stetson N, Sherry B, Levine M, Chahine NO. Toll-like receptor 4 (TLR4) expression and stimulation in a model of intervertebral disc inflammation and degeneration. Spine (Phila Pa 1976) 2013;38:1343–1351. doi: 10.1097/BRS.0b013e31826b71f4. http://dx.doi.org/10.1097/BRS.0b013e31826b71f4. [DOI] [PubMed] [Google Scholar]
- Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat. Rev. Rheumatol. 2014;10:44–56. doi: 10.1038/nrrheum.2013.160. http://dx.doi.org/10.1038/nrrheum.2013.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts S, Urban JPG. Intervertebral discs. In: Riihimäki H, Viikari-Juntura E, editors. Encyclopedia of Occupational Health and Safety. Geneva: International Labor Organization; 2011. [Google Scholar]
- Roberts N, Hogg D, Whitehouse GH, Dangerfield P. Quantitative analysis of diurnal variation in volume and water content of lumbar intervertebral discs. Clin. Anat. 1998;11:1–8. doi: 10.1002/(SICI)1098-2353(1998)11:1<1::AID-CA1>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Roth I, Kwon HM, Martin P, Fe E. Osmoprotective transcription factor NFAT5/TonEBP modulates nuclear factor-B activity. Physiol. C. 2010;21:3459–3474. doi: 10.1091/mbc.E10-02-0133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roughley PJ, Lee ER. Cartilage proteoglycans: structure and potential functions. Microsc. Res. Tech. 1994;28:385–397. doi: 10.1002/jemt.1070280505. http://dx.doi.org/10.1002/jemt.1070280505. [DOI] [PubMed] [Google Scholar]
- Scherer C, Pfisterer L, Wagner A, Hödebeck M, Cattaruzza M, Hecker M, Korff T. Arterial wall stress controls NFAT5 activity in vascular smooth muscle cells. J. Am. Heart Assoc. 2014;3:e000626. doi: 10.1161/JAHA.113.000626. http://dx.doi.org/10.1161/JAHA.113.000626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneiderman R, Keret D, Maroudas A. Effects of mechanical and osmotic pressure on the rate of glycosaminoglycan synthesis in the human adult femoral head cartilage: an in vitro study. J. Orthop. Res. 1986;4:393–408. doi: 10.1002/jor.1100040402. http://dx.doi.org/10.1002/jor.1100040402. [DOI] [PubMed] [Google Scholar]
- Souza RB, Kumar D, Calixto N, Singh J, Schooler J, Subburaj K, Li X, Link TM, Majumdar S. Response of knee cartilage T1rho and T2 relaxation times to in vivo mechanical loading in individuals with and without knee osteoarthritis. Osteoarthr. Cartil. 2014 doi: 10.1016/j.joca.2014.04.017. http://dx.doi.org/10.1016/j.joca.2014.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spillekom S, Smolders LA, Grinwis GCM, Arkesteijn ITM, Ito K, Meij BP, Tryfonidou MA. Increased osmolarity and cell clustering preserve canine notochordal cell phenotype in culture. Tissue Eng. C. Methods. 2014 doi: 10.1089/ten.TEC.2013.0479. http://dx.doi.org/10.1089/ten.TEC.2013.0479. [DOI] [PubMed] [Google Scholar]
- Stroud JC, Lopez-Rodriguez C, Rao A, Chen L. Structure of a TonEBP–DNA complex reveals DNA encircled by a transcription factor. Nat. Struct. Biol. 2002;9:90–94. doi: 10.1038/nsb749. http://dx.doi.org/10.1038/nsb749. [DOI] [PubMed] [Google Scholar]
- Takeno K, Kobayashi S, Negoro K, Uchida K, Miyazaki T, Yayama T, Shimada S, Baba H. Physical limitations to tissue engineering of intervertebral disc cells: effect of extracellular osmotic change on glycosaminoglycan production and cell metabolism. Laboratory investigation. J. Neurosurg. Spine. 2007;7:637–644. doi: 10.3171/SPI-07/12/637. http://dx.doi.org/10.3171/SPI-07/12/637. [DOI] [PubMed] [Google Scholar]
- Tew SR, Peffers MJ, McKay TR, Lowe ET, Khan WS, Hardingham TE, Clegg PD. Hyperosmolarity regulates SOX9 mRNA posttranscriptionally in human articular chondrocytes. Am. J. Physiol. Cell Physiol. 2009;297:C898–C906. doi: 10.1152/ajpcell.00571.2008. http://dx.doi.org/10.1152/ajpcell.00571.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tew SR, Vasieva O, Peffers MJ, Clegg PD. Post-transcriptional gene regulation following exposure of osteoarthritic human articular chondrocytes to hyperosmotic conditions. Osteoarthr. Cartil. 2011;19:1036–1046. doi: 10.1016/j.joca.2011.04.015. http://dx.doi.org/10.1016/j.joca.2011.04.015. [DOI] [PubMed] [Google Scholar]
- Tian Y, Yuan W, Fujita N, Wang J, Wang H, Shapiro IM, Risbud MV. Inflammatory cytokines associated with degenerative disc disease control aggrecanase-1 (ADAMTS-4) expression in nucleus pulposus cells through MAPK and NF-κB. Am. J. Pathol. 2013;182:2310–2321. doi: 10.1016/j.ajpath.2013.02.037. http://dx.doi.org/10.1016/j.ajpath.2013.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong EHY, Guo JJ, Huang AL, Liu H, Hu CD, Chung SSM, Ko BCB. Regulation of nucleocytoplasmic trafficking of transcription factor OREBP/TonEBP/NFAT5. J. Biol. Chem. 2006;281:23870–23879. doi: 10.1074/jbc.M602556200. http://dx.doi.org/10.1074/jbc.M602556200. [DOI] [PubMed] [Google Scholar]
- Tsai TT, Danielson KG, Guttapalli A, Oguz E, Albert TJ, Shapiro IM, Risbud MV. TonEBP/OREBP is a regulator of nucleus pulposus cell function and survival in the intervertebral disc. J. Biol. Chem. 2006;281:25416–25424. doi: 10.1074/jbc.M601969200. http://dx.doi.org/10.1074/jbc.M601969200. [DOI] [PubMed] [Google Scholar]
- Tsai TT, Guttapalli A, Agrawal A, Albert TJ, Shapiro IM, Risbud MV. MEK/ERK signaling controls osmoregulation of nucleus pulposus cells of the intervertebral disc by transactivation of TonEBP/OREBP. J. Bone Miner. Res. 2007;22:965–974. doi: 10.1359/jbmr.070322. http://dx.doi.org/10.1359/jbmr.070322. [DOI] [PubMed] [Google Scholar]
- Ueno M, Shen WJ, Patel S, Greenberg AS, Azhar S, Kraemer FB. Fat-specific protein 27 modulates nuclear factor of activated T cells 5 and the cellular response to stress. J. Lipid Res. 2013;54:734–743. doi: 10.1194/jlr.M033365. http://dx.doi.org/10.1194/jlr.M033365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urban JP. The chondrocyte: a cell under pressure. Br. J. Rheumatol. 1994;33:901–908. doi: 10.1093/rheumatology/33.10.901. [DOI] [PubMed] [Google Scholar]
- Urban JP, Bayliss MT. Regulation of proteoglycan synthesis rate in cartilage in vitro: influence of extracellular ionic composition. Biochim. Biophys. Acta. 1989;992:59–65. doi: 10.1016/0304-4165(89)90050-0. [DOI] [PubMed] [Google Scholar]
- Urban JP, Hall AC, Gehl KA. Regulation of matrix synthesis rates by the ionic and osmotic environment of articular chondrocytes. J. Cell. Physiol. 1993;154:262–270. doi: 10.1002/jcp.1041540208. http://dx.doi.org/10.1002/jcp.1041540208. [DOI] [PubMed] [Google Scholar]
- Van der Windt AE, Haak E, Das RHJ, Kops N, Welting TJM, Caron MMJ, van Til NP, Verhaar JAN, Weinans H, Jahr H. Physiological tonicity improves human chondrogenic marker expression through nuclear factor of activated T-cells 5 in vitro. Arthritis Res. Ther. 2010;12:R100. doi: 10.1186/ar3031. http://dx.doi.org/10.1186/ar3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Dijk B, Potier E, Ito K. Culturing bovine nucleus pulposus explants by balancing medium osmolarity. Tissue Eng. C Methods. 2011;17:1089–1096. doi: 10.1089/ten.TEC.2011.0215. http://dx.doi.org/10.1089/ten.TEC.2011.0215. [DOI] [PubMed] [Google Scholar]
- Van Dijk BGM, Potier E, Ito K. Long-term culture of bovine nucleus pulposus explants in a native environment. Spine. J. 2013;13:454–463. doi: 10.1016/j.spinee.2012.12.006. http://dx.doi.org/10.1016/j.spinee.2012.12.006. [DOI] [PubMed] [Google Scholar]
- Verma P, Dalal K. ADAMTS-4 and ADAMTS-5: key enzymes in osteoarthritis. J. Cell. Biochem. 2011;112:3507–3514. doi: 10.1002/jcb.23298. http://dx.doi.org/10.1002/jcb.23298. [DOI] [PubMed] [Google Scholar]
- Wang J, Markova D, Anderson DG, Zheng Z, Shapiro IM, Risbud MV. TNF-α and IL-1β promote a disintegrin-like and metalloprotease with thrombospondin type I motif-5-mediated aggrecan degradation through syndecan-4 in intervertebral disc. J. Biol. Chem. 2011;286:39738–39749. doi: 10.1074/jbc.M111.264549. http://dx.doi.org/10.1074/jbc.M111.264549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Tian Y, Phillips KLE, Chiverton N, Haddock G, Bunning RA, Cross AK, Shapiro IM, Le Maitre CL, Risbud MV. Tumor necrosis factor α- and interleukin-1β-dependent induction of CCL3 expression by nucleus pulposus cells promotes macrophage migration through CCR1. Arthritis Rheum. 2013a;65:832–842. doi: 10.1002/art.37819. http://dx.doi.org/10.1002/art.37819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Yang YY, Niu HJ, Zhang WJ, Feng QJ, Chen W-F. An ultrasound study of altered hydration behaviour of proteoglycan-degraded articular cartilage. BMC Musculoskelet. Disord. 2013b;14:289. doi: 10.1186/1471-2474-14-289. http://dx.doi.org/10.1186/1471-2474-14-289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei J, Richbourgh B, Jia T, Liu C. ADAMTS-12: a multifaced metalloproteinase in arthritis and inflammation. Mediat. Inflamm. 2014;2014:649718. doi: 10.1155/2014/649718. http://dx.doi.org/10.1155/2014/649718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheaton AJ, Borthakur A, Shapiro EM, Regatte RR, Akella SVS, Kneeland JB, Reddy R. Proteoglycan loss in human knee cartilage: quantitation with sodium MR imaging—feasibility study. Radiology. 2004;231:900–905. doi: 10.1148/radiol.2313030521. http://dx.doi.org/10.1148/radiol.2313030521. [DOI] [PubMed] [Google Scholar]
- Wiig H, Schröder A, Neuhofer W, Jantsch J, Kopp C, Karlsen TV, Boschmann M, Goss J, Bry M, Rakova N, Dahlmann A, Brenner S, Tenstad O, Nurmi H, Mervaala E, Wagner H, Beck FX, Müller DN, Kerjaschki D, Luft FC, Harrison DG, Alitalo K, Titze J. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J. Clin. Invest. 2013;123:2803–2815. doi: 10.1172/JCI60113. http://dx.doi.org/10.1172/JCI60113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo SK, Dahl SC, Handler JS, Kwon HM. Bidirectional regulation of tonicity-responsive enhancer binding protein in response to changes in tonicity. Am. J. Physiol. Ren. Physiol. 2000;278:F1006–F1012. doi: 10.1152/ajprenal.2000.278.6.F1006. [DOI] [PubMed] [Google Scholar]
- Woo SK, Lee S, Do, Na KY, Park WK, Kwon HM. TonEBP/NFAT5 stimulates transcription of HSP70 in response to hypertonicity. Mol. Cell. Biol. 2002;22:5753–5760. doi: 10.1128/MCB.22.16.5753-5760.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuertz K, Urban JPG, Klasen J, Ignatius A, Wilke HJ, Claes L, Neidlinger-Wilke C. Influence of extracellular osmolarity and mechanical stimulation on gene expression of intervertebral disc cells. J. Orthop. Res. 2007;25:1513–1522. doi: 10.1002/jor.20436. http://dx.doi.org/10.1002/jor.20436. [DOI] [PubMed] [Google Scholar]
- Yancey PH, Clark ME, Hand SC, Bowlus RD, Somero GN. Living with water stress: evolution of osmolyte systems. Science. 1982;217:1214–1222. doi: 10.1126/science.7112124. [DOI] [PubMed] [Google Scholar]
- Yoon HJ, You S, Yoo SA, Kim NH, Kwon HM, Yoon CH, Cho CS, Hwang D, Kim WU. NF-AT5 is a critical regulator of inflammatory arthritis. Arthritis Rheum. 2011;63:1843–1852. doi: 10.1002/art.30229. http://dx.doi.org/10.1002/art.30229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X, Wang H, Burg MB, Ferraris JD. Inhibitory phosphorylation of GSK-3β by AKT, PKA, and PI3K contributes to high NaCl-induced activation of the transcription factor NFAT5 (TonEBP/OREBP) Am. J. Physiol. Ren. Physiol. 2013 Apr 1;304(7):F908–F917. doi: 10.1152/ajprenal.00591.2012. http://dx.doi.org/10.1152/ajprenal.00591.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]