Abstract
Sphingomonas koreensis is an aerobic Gram-negative rod originally described in 2001 following isolation from natural mineral water in Korea. Here, we report a case study with Sphingomonas koreensis as the causative agent of meningitis. To our knowledge, this is the first documented case of Sphingomonas koreensis as a human pathogen.
CASE REPORT
In 1997, a 14-year-old female patient underwent surgery due to a syrinx in the cervical portion of her medulla. A cystoperitoneal shunt system was inserted, and the column was stabilized with pedicle screws and metal plates corresponding to T1-T2 and T11-T12.
In 2003, she was diagnosed with secondary amenorrhea due to a pituitary adenoma. A transsphenoidal resection of the tumor was attempted in 2005, but bleeding resulted in the operation being unsuccessful.
In November 2012, the patient was diagnosed with bacterial meningitis. The cerebrospinal fluid (CSF) showed the following: elevated leukocytes of 290 nucleated cells/μl with 80% neutrophils, protein concentration at 0.7 g/liter, and CSF glucose concentration of 1.1 mmol/liter. No bacteria were observed in a concentrated Gram stain of the CSF, but Staphylococcus epidermidis and Staphylococcus warneri were isolated. After susceptibility testing of the two isolates, the patient received a 2-week course of intravenous ceftriaxone (4 g once daily). Twelve days after stopping the ceftriaxone treatment, the symptoms of meningitis returned. The CSF showed elevated leukocytes of 853 nucleated cells/μl with 85% neutrophils, protein concentration at 0.6 g/liter, and CSF glucose concentration of 0.6 mmol/liter. Again, no bacteria were observed in a concentrated Gram stain, but S. epidermidis was cultured from the CSF and intravenous ceftriaxone was reinstituted. Initially, the clinical response was satisfactory; however, the symptoms recurred in January 2013. Following this recurrence, the decision was made to switch the treatment plan to intravenous meropenem (2 g three times a day) and intravenous vancomycin (1 g twice daily). Treatment with intravenous meropenem and vancomycin continued until the removal of the shunt system in February 2013. It should be noted that the peritoneal part of the shunt system could not be removed. The remaining part had no interaction with the central nervous system.
Two weeks after surgical removal of the shunt, the patient was admitted to the Department of Infectious Diseases with a headache, stiff neck, and confusion. The CSF showed the following: elevated leukocytes of 335 nucleated cells/μl with 70% neutrophils, protein concentration of 1.2 g/liter, and CSF glucose concentration of 0.6 mmol/liter. Treatment with intravenous meropenem (2 g three times a day) and vancomycin (1 g twice daily) was initiated, and temporary external lumbar drainage was performed. A magnetic resonance imaging scan showed a cranionasal fistula at the point of the previous transsphenoidal resection from 2005. Subsequently, a brain computed tomography showed two osseous defects in the sphenoid sinus. Blood cultures taken at the time of admission were without growth, but Sphingomonas koreensis was cultured from the CSF. The CSF was taken before initiation of antibiotic treatment, and no bacteria were observed in a concentrated Gram stain. Additional CSF cultures taken from the lumbar drain over a 3-day period in late February and March did not result in growth. However, 5 days following hospitalization, S. koreensis was cultured again from the CSF. Treatment with intrathecal gentamicin (8 mg once daily) was added to the intravenous meropenem and vancomycin treatment, and the lumbar drain was replaced. Cultivation of the tip of the drain was without growth. After antimicrobial susceptibility testing, the antibiotic treatment was changed to oral trimethoprim-sulfamethoxazole (80/400 mg twice daily). The two osseous defects in the sphenoid sinus were surgically repaired during the hospitalization period. Slowly, the lumbar drainage was reduced, which concluded with removal of the drain and discharge of the patient in late March 2013. On discharge, she had received a total of 3 weeks of treatment with oral trimethoprim-sulfamethoxazole.
In September 2013, the patient was readmitted to the Department of Infectious Diseases presenting with fatigue, headache, and neck pain that had been increasing in severity over 3 to 4 days. The patient described the symptoms as being similar to those of the previous episode of meningitis. The clinical examination revealed no neck stiffness, and she was fully conscious. Her temperature was 37.5°C. The CSF showed 45 nucleated cells/μl, of which 27 were neutrophils. The protein concentration was normal at 0.34 g/liter, but the glucose concentration was low at 1.6 mmol/liter compared to a serum glucose of 6.3 mmol/liter. Laboratory results showed a C-reactive protein concentration of <3.0 mg/liter (normal range of <6 mg/liter), leukocytes of 10.4 × 109/liter (normal range of 3.5 × 109 to 8.8 × 109/liter), and a neutrophil count of 8.96 × 109/liter (normal range of 1.5 × 109 to 7.5 × 109/liter). Treatment with intravenous ceftriaxone (4 g once daily) and intravenous vancomycin (1 g twice daily) was initiated. No bacteria were observed in a concentrated Gram stain of the CSF taken at the time of admission, but again, S. koreensis was cultured. Blood and urine cultures were without growth.
It was decided to treat the second episode of S. koreensis meningitis with higher doses of oral trimethoprim-sulfamethoxazole (240/1,200 mg four times daily). The patient weighed 50 kg, and it was suspected that the dosage used for the previous episode had been inadequate. However, the dosage had to be reduced to 160/800 mg four times daily after 12 days of treatment, due to increasing side effects (fatigue and nausea). After a total of 3 weeks of oral trimethoprim-sulfamethoxazole treatment, she received another 2 weeks of treatment with oral moxifloxacin (400 mg once daily). To date, which is more than a year, the patient has had no recurrent symptoms of meningitis.
The two isolates of S. koreensis from February and September 2013 were isolated at the same department of clinical microbiology. For both episodes, growth (yellow colonies) was observed from the two spinal fluid samples on 5% horse blood agar after 72 h of incubation (Fig. 1). Colonies were examined with matrix-assisted laser desorption ionization–time of flight mass spectrometry (MALDI-TOF MS), using the Shimadzu/SARAMIS system (Shimadzu Corporation, Kyoto, Japan, and Anagnos-Tec GmbH, Potsdam-Golm, Germany). The system comprises an Axima Assurance mass spectrometer system (Shimadzu Corporation) and the Shimadzu Biotech Launchpad software program and the SARAMIS database application. The system was operated with a matrix consisting of α-cyano-4-hydroxycinnamic acid in acetonitrile, ethanol, and water from Anagnos-Tec GmbH. Both isolates obtained an unambiguous diagnosis of Sphingomonas koreensis with a score of 99.9.
FIG 1.
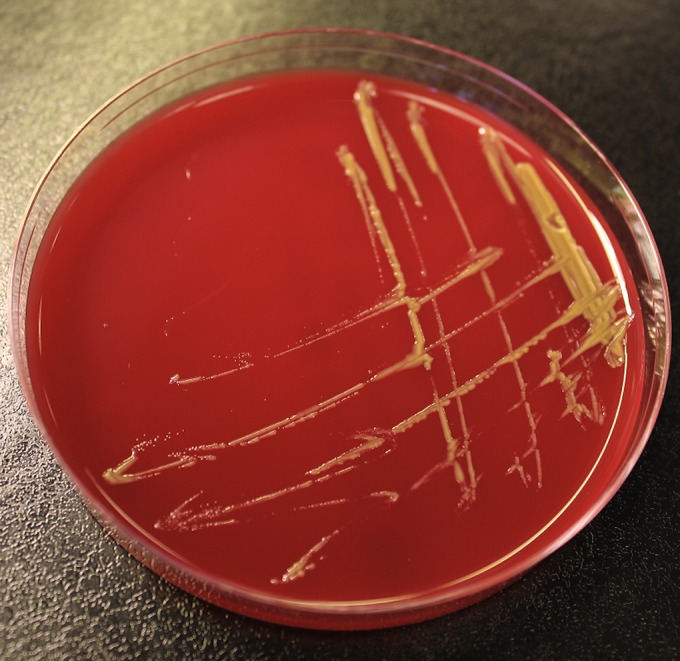
Sphingomonas koreensis on 5% horse blood agar.
The second isolate was also subjected to partial 16S rRNA gene sequencing (MicroSeq 500 system; Perkin-Elmer, Applied Biosystems Division, Foster City, CA) to confirm the MALDI-TOF diagnosis. The isolate consensus sequence (433 bp) had a 100% match to the Sphingomonas koreensis type strain JSS-26 (T) (GenBank accession no. AF131296). There was good separation from the second best match, Sphingomonas soli type strain T5-04 (T) (98.61%) (GenBank accession no. AB166883), and from Sphingomonas paucimobilis type strain ATCC 29837 (T) (94.23%) (GenBank accession no. U37337). Primary antimicrobial susceptibility testing was performed using the standard EUCAST disk diffusion method on Mueller-Hinton agar, although no zone diameter breakpoints are available. For the following antimicrobial agents, no zones were present: ampicillin, piperacillin-tazobactam, ceftriaxone, aztreonam, meropenem, and gentamicin. However, zones were present for ciprofloxacin, moxifloxacin, and trimethoprim-sulfamethoxazole. Antimicrobial susceptibility testing for these three antibiotics was performed with a gradient MIC method (Etest; bioMérieux, Lyon, France) on Mueller-Hinton agar. The following MICs were obtained: 0.75 mg/liter for ciprofloxacin, 0.19 mg/liter for moxifloxacin, and 0.047 mg/liter for trimethoprim-sulfamethoxazole.
The genus Sphingomonas was first proposed in 1990. It is characterized by the fact that the cellular lipids contain sphingoglycolipids, and the major respiratory quinone is ubiquinone 10 (1). It is a Gram-negative, strictly aerobic and nonfermentative rod (1). The genus has since been divided into 4 genera: Sphingomonas sensu stricto, Sphingobium, Novosphingobium, and Sphingopyxis (2). S. koreensis was originally described in 2001, after it was discovered in mineral water in Korea, hence the name “koreensis” (3). It is motile with a single polar flagellum and catalase, oxidase, and beta-galactosidase positive. Colonies are opaque and yellow (3). It is possible that S. koreensis could have been misidentified as another Sphingomonas species based on phenotypic characteristics. However, the biochemical profile differs significantly from other Sphingomonas species, and, with, e.g., the API ID system, it should be possible to separate the different species (3).
Little is known about the S. koreensis susceptibility pattern. In one study, the antimicrobial susceptibilities of 27 different strains of Sphingomonas sensu stricto were tested using the ATB PSE5 strips (bioMérieux) (4). The isolates were tested at one or two concentrations. At a colistin concentration of 2 mg/liter, 92.6% of the strains showed growth, 29.6% at a meropenem concentration of 8 mg/liter, 25.9% at a ciprofloxacin concentration of 2 mg/liter, 3.7% at a gentamicin concentration of 8 mg/liter, and 55.6% at a piperacillin-tazobactam concentration of 16 mg/liter (4). The strain isolated from our patient had low ciprofloxacin, moxifloxacin, and trimethoprim-sulfamethoxazole MICs, which was the reason why trimethoprim-sulfamethoxazole and subsequently moxifloxacin were chosen for treatment.
To our knowledge, this is the first report of S. koreensis as a human pathogen. Another member of the Sphingomonas genus, S. paucimobilis, is an opportunistic pathogen associated with both nosocomial and community-acquired infections (5–8). S. paucimobilis is considered to be a low-virulence organism, and mortality rates associated with S. paucimobilis have been reported to be close to zero in three reviews, though it has been associated with septic shock (5–7). A mortality rate of 5.5% (3 of 55 patients) has been reported; however, mortality was believed to be associated with patient comorbidity in the study (8). S. paucimobilis has been associated with a wide range of infections, such as pneumonia, intravascular catheter-related infections, skin and soft tissue infections, urinary tract infections, and meningitis (5–8). Another Sphingomonas sp., Sphingomonas mucosissima, has been described to cause bacteremia (9).
In our case, it is likely that S. koreensis was present in the central nervous system in the time period between first isolation (February 2013) and final occurrence and hospitalization (September 2013). S. koreensis has been isolated only on the two occasions described in this report at our Department of Clinical Microbiology. Accordingly, it seems very unlikely that the patient was reinfected after the first attempt of antibiotic treatment. It is more likely that treatment failure caused by insufficient doses of trimethoprim-sulfamethoxazole was responsible for the reoccurrence. There are two possible explanations for the introduction of S. koreensis into the central nervous system. It could have been introduced during the removal surgery of the cystoperitoneal shunt system in February 2013 or through the two osseous defects in the sphenoid sinus. S. koreensis must be considered to be a low-virulence organism, and this could explain why the patient was asymptomatic for several months between February and September 2013. Furthermore, when she presented at our department in September 2013, it was with relatively mild symptoms, and she had no fever. The diagnosis of meningitis was made because the patient was known to have recurrent meningitis and because the patient described the symptoms as being similar to those of the previous episode. The exact origin of the S. koreensis is unknown. As a commensal of the environment, including natural mineral water, there could be multiple sources.
This case demonstrates the importance of species identification, even if isolates are considered or suspected to be contaminants. In this case, the finding of S. koreensis at the second episode of meningitis clearly indicates that this was a true pathogen and the cause of meningitis in the patient. In conclusion, the unique case we report here indicates that antibiotic therapy should be directed against the specific organism, S. koreensis.
ACKNOWLEDGMENTS
We thank Amanda G. Vang for proofreading the manuscript and Olav D. Larsen for assistance with the work.
REFERENCES
- 1.Yabuuchi E, Yano I, Oyaizu H, Hashimoto Y, Ezaki T, Yamamoto H. 1990. Proposals of Sphingomonas paucimobilis gen. nov. and comb. nov., Sphingomonas parapaucimobilis sp. nov., Sphingomonas yanoikuyae sp. nov., Sphingomonas adhaesiva sp. nov., Sphingomonas capsulata comb. nov., and two genospecies of the genus Sphingomonas. Microbiol Immunol 34:99–119. [DOI] [PubMed] [Google Scholar]
- 2.Takeuchi M, Hamana K, Hiraishi A. 2001. Proposal of the genus Sphingomonas sensu stricto and three new genera, Sphingobium, Novosphingobium and Sphingopyxis, on the basis of phylogenetic and chemotaxonomic analyses. Int J Syst Evol Microbiol 51:1405–1417. doi: 10.1099/00207713-51-4-1405. [DOI] [PubMed] [Google Scholar]
- 3.Lee JS, Shin YK, Yoon JH, Takeuchi M, Pyun YR, Park YH. 2001. Sphingomonas aquatilis sp. nov., Sphingomonas koreensis sp. nov, and Sphingomonas taejonensis sp. nov., yellow-pigmented bacteria isolated from natural mineral water. Int J Syst Evol Microbiol 51:1491–1498. doi: 10.1099/00207713-51-4-1491. [DOI] [PubMed] [Google Scholar]
- 4.Vaz-Moreira I, Nunes OC, Manaia CM. 2011. Diversity and antibiotic resistance patterns of Sphingomonadaceae isolates from drinking water. Appl Environ Microbiol 77:5697–5706. doi: 10.1128/AEM.00579-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bayram N, Devrim I, Apa H, Gulfidan G, Turkyilmaz HN, Gunay I. 2013. Sphingomonas paucimobilis infections in children: 24 case reports. Mediterr J Hematol Infect Dis 5:e2013040. doi: 10.4084/MJHID.2013.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lin JN, Lai CH, Chen YH, Lin HL, Huang CK, Chen WF, Wang JL, Chung HC, Liang SH, Lin HH. 2010. Sphingomonas paucimobilis bacteremia in humans: 16 case reports and a literature review. J Microbiol Immunol Infect 43:35–42. doi: 10.1016/S1684-1182(10)60005-9. [DOI] [PubMed] [Google Scholar]
- 7.Ryan MP, Adley CC. 2010. Sphingomonas paucimobilis: a persistent Gram-negative nosocomial infectious organism. J Hosp Infect 75:153–157. doi: 10.1016/j.jhin.2010.03.007. [DOI] [PubMed] [Google Scholar]
- 8.Toh HS, Tay HT, Kuar WK, Weng TC, Tang HJ, Tan CK. 2011. Risk factors associated with Sphingomonas paucimobilis infection. J Microbiol Immunol Infect 44:289–295. doi: 10.1016/j.jmii.2010.08.007. [DOI] [PubMed] [Google Scholar]
- 9.Angelakis E, Roux V, Raoult D. 2009. Sphingomonas mucosissima bacteremia in patient with sickle cell disease. Emerg Infect Dis 15:133–134. doi: 10.3201/eid1501.080465. [DOI] [PMC free article] [PubMed] [Google Scholar]


