Abstract
Objective
Determine whether HIV and cART affect resting state functional connectivity (rs-fc) between the striatum and cortical regions.
Methods
49 HIV uninfected (HIV−) and 132 HIV infected (HIV+) (65% receiving combination anti-retroviral treatment [cART]) had laboratory studies (current and nadir CD4 T-cell counts, and plasma HIV viral load), neuropsychological performance (NP) testing, and neuroimaging. Rs-fc, which examines the coordination of neural activity in distant brain regions, was used to investigate cortico-striatal functional connections. The effect of cART was assessed comparing HIV+ individuals on cART (HIV+/cART+), and HIV+ individuals not currently receiving cART (HIV+/cART−). Relationships between laboratory tests, cognitive performance, and cART on subcortical-cortical rs-fc were assessed by an analysis of variance.
Results
HIV+ individuals had lower cortico-striatal functional connectivity than HIV− controls, specifically between the striatum and default mode network (DMN; p <0.001) and ventral attention network (VATT; p <0.001). HIV+/cART+ individuals had higher functional connectivity between the striatum and DMN (p=0.02) and VATT (p = 0.01) compared to HIV+/cART− subjects. Laboratory (current and nadir CD4 T-cell counts, plasma viral load) and NP were not correlated with cortico-striatal rs-fc.
Conclusions
HIV was associated with disrupted cortico-striatal networks, consistent with HIV’s known impact on subcortical areas. Interestingly, within certain networks HIV+/cART+ individuals had similar rs-fc compared to HIV− controls, suggesting possible improvements in HIV related neural dysfunction due to medications. Rs-fc may be a sensitive biomarker of neural insult and its recovery following cART. Additional studies may show rs-fc has utility in measuring acute inflammation caused by HIV.
Keywords: HIV, resting state functional connectivity, combination anti-retroviral therapy (cART), HIV associated neurocognitive disorders (HAND), striatum, caudate, putamen
Introduction
Human immune-deficiency virus (HIV) enters the human brain soon after seroconversion. While HIV does not directly infect neurons, it does cause prominent neurological symptoms including HIV associated neurocognitive disorder (HAND) [1,2]. Combination anti-retroviral therapy (cART) has greatly extended the lives of HIV infected (HIV+) individuals and diminished the number of opportunistic infections. However, the prevalence of HAND has remained relatively unchanged due to cART, [3] with milder forms now predominating.
Neuropsychological performance (NP) testing remains crucial for detecting neural dysfunction due to HIV. However, cognitive normality does not imply the absence of neural dysfunction. Indeed, pathophysiologic changes occur prior to changes in NP [4,5]. Additionally, not all HIV+ individuals develop HAND justifying the need to use other modalities to study neurobiological changes that can occur in HIV+ individuals [6]. Neuroimaging may provide an additional non-invasive method to assess the effects of HIV on brain function [7,8].
Blood oxygen level dependent (BOLD) functional magnetic resonance imaging (fMRI) is a measure of neural activity. Resting-state functional connectivity (rs-fc) quantifies the temporal correlation in spontaneous BOLD fluctuations [9,10]. Sets of mutually correlated brain regions define resting state networks (RSNs). These RSNs are topographically similar to task based co-activations [11–14]. A benefit of rs-fc is that multiple networks can be delineated without requiring an individual to perform multiple tasks to assess each brain area [15]. HIV has been shown to cause disruptions in functional connectivity within multiple cortical RSNs: including the default mode (DMN), control (CON), ventral (VATT), and sensory-motor (SMN) networks [16,17]. Several of these RSNs correspond to cognitive domains often affected by HIV [3,18,19]. However, previous rs-fc studies in HIV+ individuals have focused on cortical and not subcortical changes.
Pathological studies have shown that HIV has a strong predilection for subcortical areas [e.g. the striatum (caudate and putamen)]. The highest concentrations of HIV and greatest atrophy due to the virus are often seen within subcortical regions [20–23]. In the pre-cART era, HIV was often considered a subcortical dementia due to motor deficits, and striatal neurodegeneration. However in the cART era, cortical cognitive deficiencies have been observed in addition to subcortical deficits [24–26]. Furthermore, task based fMRI has shown deficits in cortico-striatal task activations [27,28]. The effects of cART on cortico-striatal functional connections remain unknown.
Here we cross-sectionally studied the effects of HIV on cortico-striatal rs-fc. Four a priori striatal regions of interest (ROIs), previously defined by membership to canonical RSNs, were used. Functional connections from cortical regions to these ROIs are associated with somato-motor function (SMN), executive control (CON), attention to stimuli (VATT), and organization of tasks and memory (DMN). Many of these aforementioned cognitive functions and RSNs have been previously shown to be affected by HIV [3,29]. We assessed relationships between rs-fc and typical laboratory (plasma viral load, current CD4 T-cell count, nadir CD4 T-cell count) and NP measures in HIV+ individuals to determine whether this imaging modality could be used as a brain biomarker of disease. In summary, the purpose of this study is to determine whether HIV affects functional connectivity in cortico-striatal networks and the impact of cART on cortico-striatal networks.
Methods
Subject Demographics
Individuals were selected from ongoing studies conducted at the Washington University in Saint Louis (WUSTL) Infectious Disease Clinic. Participants were excluded if they had a history of neurological illness (other than HIV) or infection, stroke, or major psychiatric disorders [22]. Participants were also excluded for active substance abuse if they were positive for cocaine, barbiturates, heroin, and phencyclidine (PCP). In addition, we obtained self-reported past substance use (6-month) for these same drugs of abuse. All participants (n=181) provided consent using forms approved by the WUSTL institutional review board. Each individual had a neuro-medical examination, NP testing, and a MRI scan on the same day. A total of 4 HIV− and 1 HIV+ subject were excluded as they could not successfully complete all NP and neuroimaging testing. HIV− controls (n=45) were similar to HIV+ (n=131) individuals in both gender and education. HIV+ individuals were either entirely naïve or currently not taking cART (HIV+/cART−) (n=49) or had been receiving stable cART for at least 3 months (HIV+/cART+) (n=82) prior to evaluation (Table 1). HIV serostatus was confirmed with immunoassay western blot or through polymerase chain reaction. Clinical laboratory evaluations (current plasma CD4 cell count and HIV viral load) were performed within 3 months of testing. Of the 49 HIV+/cART− individuals, 43 individuals were cART naïve (never taken cART). A small number of HIV+/cART− (n=6) were not receiving therapy at the time of scanning but had received medications in the past. Nadir CD4 cell counts, estimated duration of infection (DOI), and current and previous cART history were identified from medical history or self-report. Within the HIV+ cohort, only two individuals had prior Hepatitis-C virus (HCV) infection. These HIV+ subjects had received treatment and had undetectable HCV viral loads.
Table 1. Demographics of participants.
SD = standard deviation, IQR = Inter-quartile range, and NA= not available. The neuropsychological z- score (NPZ-4) was created by standardizing and combining a common test battery consisting of trails A–B, digit-symbol, and the Hopkins verbal learning test.
| HIV− n=45 |
HIV+/CART− n=49 |
HIV+/CART+ n=82 |
|
|---|---|---|---|
| Demographics | |||
| Age (years old) [SD] | 31.7 [10.9] | 34.9 [15.4] | 41.5 a [14.5] |
| Gender (% Male) | 58 | 86 b | 63 |
| Education (years) [SD] | 13.4 [2.7] | 13.4 [2.3] | 12.9 [3.1] |
| Duration of Infection (months) [SD] | NA | 64.4 [83.7] | 112.1 c [85.6] |
| Laboratory Values | |||
| Current plasma CD4 (cells/mL) [IQR] | NA | 388.0 [300.5, 583.0] | 649.2 c [447.8, 812.8] |
| Nadir plasma CD4 (cells/mL) [IQR] | NA | 336.5 [236.0, 419.0] | 247.5[44.25, 348.2] |
| Log10 plasma viral load (copies/mL) [SD] | NA | 3.38 [2.95] | 1.50 c [1.47] |
| % Individuals with Virological suppression (<50 copies/mL) | 23 | 77 c | |
| NPZ-4 (z-score) [IQR] | −0.14 [−0.8,0.5] | −0.47 [−1.0, 0.11] | −0.35 [−0.67, 0.15] |
= p < 0.01, for HIV+/cART+ compared to HIV−
= p < 0.05, for HIV+/cART− compared to HIV+/cART+
= p < 0.001, for HIV+/cART− compared to HIV+/cART+
NP Testing and Neuromedical Examination
A common battery of NP tests (including Trail-Making Tests A & B, the Hopkins Verbal Learning Test, and Digit-Symbol Modalities Test) was administered to all participants. These NP tests examine multiple cognitive domains and have previously been used for diagnosing neurocognitive impairment [30]. Raw scores were normalized using demographic-adjusted normative means for age, sex, education and race. A standardized Z-score was calculated by subtracting the appropriate normative mean from the raw score and then dividing by the normative standard deviation for each test. A global summary score (NPZ-4) was created by averaging z-scores from each test [31,32]. All participants had a detailed neurological examination performed by a board certified neurologist (BMA).
Image Acquisition
All neuroimaging was performed on a 3.0-Tesla Tim-Trio scanner (Siemens, Erlangen, Germany) equipped with the standard 12-channel head coil. A high-resolution, 3-dimensional, sagittal, T1-weighted, magnetization-prepared rapid gradient echo scan (MPRAGE) was acquired (echo time [TE] = 16 milliseconds, repetition time [TR] = 2,400 milliseconds, inversion time = 1,000 milliseconds, flip angle = 8°, 256 × 256 acquisition matrix, 1 mm3 voxels). Rs-fc scans were collected using a gradient spin-echo sequence (TE = 27 milliseconds, TR = 2.2 seconds, 64 × 64 acquisition matrix, flip angle = 90°). A total of 36 contiguous, 4.0-mm-thick slices were acquired parallel to the anterior commissure/posterior commissure plane (total of 164 frames). Two rs-fc magnetic resonance imaging (rs-fcMRI) scans (each ~ 6 minutes in duration) were performed with participants instructed to fixate on a cross-hair and remain awake (eyes open).
Structural Magnetic Resonance Imaging (MRI) Preprocessing
Structural data preprocessing and intra-subject image alignment were completed with FreeSurfer and the FreeSurfer functional analysis stream (FS-FAST) [33]. In the first step, MPRAGE structural scans were processed using automated algorithms [FreeSurfer version 5.1 software package (http://surfer.nmr.mgh.harvard.edu)]. This software constructs a surface mesh representation of the cerebral cortex based on each participant’s MPRAGE scan and non-linearly registers each brain to a parcellated spherical atlas [34–38]. These aligned subject scans were used to create subcortical ROIs.
Rs-fc Preprocessing
Rs-fc scans were preprocessed using FS-FAST (http://surfer.nmr.mgh.harvard.edu). Initial rs-fc preprocessing included several steps to prepare the data and remove noise artifacts. The first four frames of each rs-fc scan were omitted in order to remove initial magnetization gradient artifact. Next, systematic odd-even slice intensity differences from the inter-leaved image acquisition were removed. Head motion was corrected utilizing affine transformations both within and across scans for each participant. Additional in-house software was used for motion characterization to exclude subjects with excessive movement [33,39,40]. Participants were included if the standard deviation (SD) of the mean rs-fc signal was < 2.2% (after nuisance regression) or frame displacement was < 0.2 mm. A total of 6 HIV− and 9 HIV+ individuals were removed from subsequent neuroimaging analyses due to excessive head movement and/or poor image registration [40]. Following correction for motion, data underwent further preprocessing steps, including: 1) whole brain signal regression, 2) ventricular (CSF) and white matter signal regression based on individual subject anatomy [41], 3) movement time-series regression, and 4) low-pass temporal filtering to remove frequencies below 0.08Hz. These filters and regressions reduced the overall potential contribution of noise arising from non-neuronal origin. The order of preprocessing was completed in this sequence to diminish the potential re-introduction of spurious noise [42].
rs-fc and structural MRI alignment
The structural MPRAGE and preprocessed rs-fc scans were cross-aligned using a robust boundary-based registration [43]. Rs-fc scans were then transformed to common spherical coordinates after sampling from the middle of the cortical ribbon using a single non-linear interpolation. This procedure reduces potential contribution of partial volume effects, brain atrophy, and blurring across a two dimensional grey matter sheet [44]. A 4-mm full-width half-maximum (FWHM) smoothing kernel was used in the surface space, reducing contamination from adjacent white matter voxels, and neighboring sulci.
rs-fc Post-processing
Four a priori striatal ROIs representing common RSNs [the default mode (DMN), control (CON), ventral attention (VATT), and sensorimotor (SMN)] were selected according to the extant literature [39]. These areas correspond to areas often affected by HIV [20–22,45]. The ROIs were based on individual subject morphology from Freesurfer in order to minimize signal loss due to misalignment, individual variation, and atrophy [39].
Striatal ROIs served as seeds for assessing functional connectivity between subcortical and cortical surface areas. The DMN, composed of the precuneus, inferior frontal gyrus (parsopercularis), lateral temporal, and middle prefrontal cortex, was maximally correlated with the anterior caudate and inferior putamen areas adjacent to the nucleus accumbens (see figure 1). The VATT network (purple), which comprised the anterior cingulate and pre-supplementary motor areas, was maximally correlated with the putamen (anterior and midline). This VATT putamen ROI also correlated to pre-somatomotor areas, insular and lateral parietal cerebral areas of the aforementioned VATT network.[46]. The CON network (orange) was comprised of the lateral frontal, cingulate, and supramarginal gyrus and was primarily correlated with the superior/posterior caudate and lateral putamen. Finally, the SMN network (blue), composed of the precentral and postcentral gyri, and the posterior insula, was maximally correlated with the posterior putamen. All subcortical seeds were defined using previous literature [33,39]. Many of these cortical networks derived from striatal seeds spatially overlapped with previous RSNs visualized in HIV+ patients [16].
Figure 1.
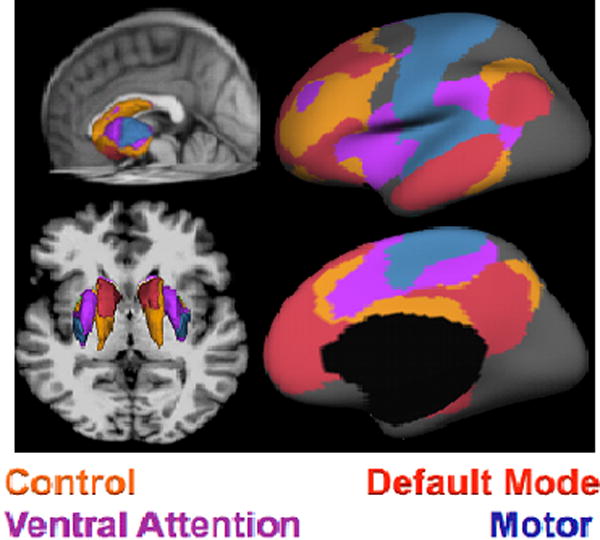
The seeded regions (center panel) were used in this analysis to determine striatum to whole-brain correlations. These striatal seeds were sourced from the previous literature [39]. The default mode (DMN) (red) seed correlates with the medial caudate (red). The ventral attention (VATT) network (violet), which was composed of the supramarginal, insula and supplementary motor areas on the cortex, was functionally connected to anterior aspects of the putamen (violet). The striatal control network (CON) seed (orange), which was composed of the lateral frontal lobe, isthmus of cingulate and parietal areas, was functionally correlated with the tail of the caudate (orange). Finally, the striatal motor network (SMN) (blue) was composed of the pre and post-central gyri, and insula and had strongest functional correlations to the posterior putamen (blue).
For each striatal seed, a cortical surface correlation map was obtained by computing the Pearson’s product moment correlation between the seed ROI’s time course and the time courses of all vertices on the cortical surface. Next, group-level average cortical maps were created by averaging Fisher’s r-to-z transformed correlation matrices for the HIV− and HIV+ cohorts. A random effects analysis on these images created statistical maps for each group. Finally, hierarchical clustering was used to determine the spatially contiguous cortical regions (clusters) that share statistically significant BOLD time courses with the striatal seed region between groups. Rs-fc clusters were deemed significant at the cluster wise threshold of p <0.001 following Monte Carlo null-z simulation [47]. Results from these tests were corrected for multiple comparisons using false discovery rate (FDR) correction method for p <0.05 [48].
Statistical Methods
Demographic differences among the groups were compared using chi-square tests or Kruskal-Wallis tests for dichotomous or continuous variables. Pearson’s product-moment correlations were used to assess for relationships between functional correlations and clinical laboratory variables (e.g. current plasma CD4 T-cell count, nadir plasma CD4 T-cell count, current plasma viral load) at the ROI level. Statistical differences between seed to whole brain correlation maps between the HIV− and HIV+ groups were assessed using an analysis of co-variance (ANCOVA) with gender and age included as co-factors. Tukey’s honest significant difference (HSD) tests were used for the 3-way group ANCOVA comparison. The R package suite (http://r-project.org) was used for all statistical analyses.
Results
Demographic and laboratory variables were mostly similar between HIV− and HIV+ groups
A total of 176 out of 181 possible participants had clinical laboratory values, NP testing, and neuroimaging (Table 1). The three groups were demographically similar for education, however, a significant difference existed for the mean age of the HIV+ and HIV− groups (p<0.05), thus age was used as a covariate in for all subsequent analyses. In the three-way analysis, the HIV+/cART+ individuals were significantly older than HIV− individuals (p <0.05). In regards to gender, the HIV+/cART− group had more males (28% more than the HIV− group (p <0.05). Predictably, the HIV+/cART+ group had higher current CD4 T-cell counts and lower current plasma viral load than HIV+/cART− individuals (p < 0.001). A large portion of the HIV+/cART+ group was virologically suppressed (p < 0.001). The two HIV+ groups did not differ according to nadir CD4 T-cell counts. All three groups had similar mean NPZ-4 test scores, with mean z-scores of −0.14, −0.35, and −0.47 for HIV−, HIV+/cART−, and HIV+/cART+ respectively.
Cortico-striatal functional connectivity was reduced in HIV+ compared to HIV−individuals
Mean striatal ROI correlation maps in the HIV− group were similar to those previously seen in the literature [33]. Compared to HIV− individuals, HIV+ individuals had reduced functional connectivity between striatal ROI seeds and focal areas of the cortex. In particular, striatal rs-fc projections to the DMN and VATT RSNs were significantly diminished for HIV+ individuals compared to HIV− participants. Within the DMN (red in Figure 1) significantly reduced functional connectivity was observed within five cortical clusters (posterior cingulate, parsopercularis, superior frontal, middle temporal, and inferior parietal) [49] (all p values <0.001) (Supplementary Table 1). Additionally, rs-fc between the striatum and the VATT were reduced (violet in Figure 1) in HIV+ compared to HIV− individuals with significant cortical clusters seen within the pre-supplementary motor association (pSMA) clusters (p <0.001) (Supplementary Table 1). No significant differences were observed for HIV+ and HIV− participants between the striatum and other cortical RSN’s (e.g. CON and SMN).
Rs-fc did not correlate with typical laboratory or NP measurements
We assessed whether significant relationships existed between functional connectivity and typical laboratory markers of HIV infection or NP. For the each of the cortico-striatal functional connectivity measures, no significant relationships were observed for laboratory measurements, including plasma CD4 T-cell count, plasma viral load, or nadir plasma CD4 T-cell count. In addition, no significant correlation was observed between NPZ-4 score and rs-fc.
cART mitigated rs-fc disruptions associated with HIV
We further split the HIV+ cohort into those receiving medications (HIV+/cART+) or not (HIV+/cART−). In clusters demonstrating a significant effect of HIV (see Figure 2, supplementary table 1), we extracted the mean functional connectivity value for each subject and compared the mean values for the HIV−, HIV+/cART+ and HIV+/cART− by ANCOVA. Overall, HIV− individuals had higher mean rs-fc values compared to HIV+ individuals. Three ROI-cluster pairs with topographical membership to DMN and VATT networks had significant group differences. Within striato-preSMA functional connections (VATT), a stair case effect was observed. HIV− individuals had higher functional connections than HIV+/cART+ or HIV+/cART− individuals. Of note, HIV+/cART+ individuals had higher functional connections than HIV+/cART− individuals (Figure 3 left panel). For striato-superior frontal functional connections (DMN), a somewhat different picture was seen (Figure 3, center panel). HIV− and HIV+/cART+ had similar functional connections. Both groups had greater functional connections than HIV+/cART− individuals. Finally, for striato-inferior frontal connections (Figure 3, right panel), HIV− individuals had significantly greater functional connections than either HIV+/cART+ or HIV+/cART− groups. No significant differences were observed between the two HIV+ groups. These results suggest that cART may have differential effects on functional connectivity with regional variability present in response to medications.
Figure 2.
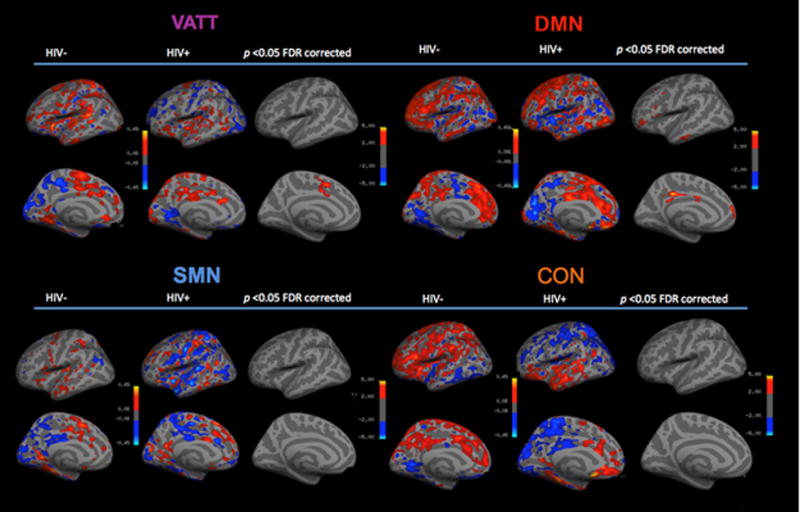
Significant differences in cortico-striatal resting state functional connectivity between HIV+ and HIV− individuals. For each of the networks, the left most panel reflects the average group correlations (partial correlations, r) for HIV− individuals. For each of the networks, the center column depicts the cortico-striatal connectivity for the HIV+ cohort. For each of the networks, the right column depicts the difference between HIV− and HIV+ individuals. Clusters that were significantly different between the two groups were observed only within the VATT and DMN resting state networks. For the figure -log10 p-values are displayed on color bars, peak values and clusters are provided in Supplementary Table 1. HIV+ individuals did not have significantly higher correlations than HIV− individuals for all networks after correction for multiple comparisons.
Figure 3.
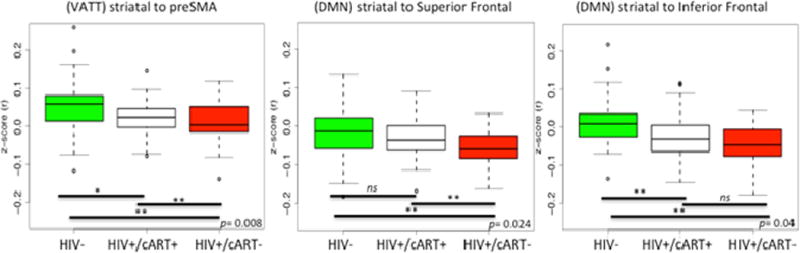
Combination antiretroviral therapy (cART) had a beneficial effect on rs-fc for only specific regions of the VATT and DMN. These plots show average correlations for the ROI to cluster connectivity for HIV− (green), HIV+/cART− (red) and HIV+/cART+ (white) groups. ROI-cluster pairs within the DMN and VATT networks were significantly different amongst the groups. Within striato-preSMA functional connections (VATT), HIV− individuals had higher rs-fc than HIV+/cART+ or HIV+/cART− individuals. HIV+/cART+ individuals had higher functional connections than HIV+/cART− individuals (left panel). For striato-superior frontal functional connections (DMN), HIV− and HIV+/cART+ had similar rs-fc. Both groups had greater functional connections than HIV+/cART− individuals (center panel). For striato-inferior frontal connections, HIV− individuals had significantly greater functional connections than either HIV+/cART+ or HIV+/cART− individuals. No significant differences were observed between the two HIV+ groups (right panel).
Discussion
We investigated the effects of HIV on cortico-striatal functional connectivity, and whether cART had significant effects on functional connectivity within defined RSNs [39]. HIV+ individuals had decreased functional connectivity with significant decreases seen in select RSNs (DMN and VATT). The cortical areas involved are similar to those previously observed to be affected by HIV and other disorders [11,16,17]. Observed differences in functional connectivity were not correlated to typical clinical laboratory measurements (i.e., current CD4, CD4 nadir, HIV viral load) or cognitive impairment (i.e., NPZ-4 scores). In addition, HIV+ individuals on stable cART had greater cortical-striatal functional connectivity within certain nodes of the DMN and VATT compared to HIV+/cART− individuals. In one instance, HIV+/cART+ had rs-fc similar to HIV− controls.
The exact mechanisms of HIV-related neuro-degeneration are still unknown, yet some parts of the puzzle are becoming clearer. HIV is known to primarily infect T-cells and activated macrophages that can enter the CNS. Over the course of the disease, this inflammatory processes can affect supporting cells (microglia and astrocytes) leading to synapto-dendritic dysfunction and alterations in neuronal function [50,51]. Observed losses in cortico-striatal connectivity could reflect these changes. Pathology studies have shown damage to striatal neurons, which may cause a loss of coherence between neuronal communications with the cortex and vice versa [27,28,52].
Rs-fc is sensitive to HIV pathology within the DMN, CON, and other cortical networks [16]. We build upon our previous results by demonstrating subcortical involvement is also present using rs-fc. Observed decreases in functional connectivity converges with previous literature that has demonstrated subcortical atrophy [3,18,53]. The striatal ganglion are critical to higher cognitive function [54–56], fine motor control [21], attention to salient stimuli [57], and processing associations between memory and attention [39,58]. The effects of HIV appeared to be specific for certain cortico-striatal networks (DMN and VATT) and not others (SMN and CON) affected. Our results complement previous studies by our group that have observed a decrease in cortical rs-fc primarily within nodes of the DMN and VATT [16]. With our current whole brain clustering approach, we observed that HIV had the greatest impact on the DMN. This may reflect that the DMN has the highest overall intra-network connectivity, largest size, and/or greatest susceptibility to neurodegenerative disease [59]. Although cortical volumetric neurodegeneration has been observed in the SMN with more advanced disease, we did not see a decrease in connectivity in these areas [60]. Our current rs-fc results are in agreement with previous studies by our group that did not observe cortical functional connectivity changes in the SMN due to HIV [16,61]. Of note, recent magnetoencephalography (MEG) studies have showed both structural and functional connectivity loss in the SMN [62,63]. Additional studies are necessary to investigate these discrepancies amongst various imaging modalities. Observed dissimilarities amongst observations with regards to the SMN may reflect differences in modalities (MRI vs. MEG), methods (structural vs. functional), age of individuals (younger vs. older), and/or degree of cognitive impairment. Larger longitudinal studies that include HIV+ individuals over a wide age range and degree of cognitive impairment are needed to further resolve these discrepancies. A resting state signature of HIV pathology may exist but further pathology/imaging correlates are needed.
The Department of Health and Human Services now recommends immediate initiation of cART with HIV positive diagnosis [64]. With these guidelines in place, it is important to determine the effects of cART on brain functional connectivity. In general, most research groups have found cART to have beneficial effects on cognitive status and neurobiology [28,63–66]. While cART may have detrimental effects [65], these do not outweigh the tremendous benefit in restoring overall health and lifestyle [66]. The effects of cART using NP testing results have shown mostly restorative effects, however inconsistencies in NP testing were noted (for review see Joska et al. 2010) [68]. In this study, the HIV+ individuals on cART therapy had functional connectivity closer to HIV− levels, but with similar NP scores (see figure 3). At the neurovascular level, chronic inflammation alters the hemodynamic response, a phenomenon that has been previously reported in advanced HIV infection [69–71]. cART reduces both glial activation and inflammation caused by HIV [72,73]. cART also decreases HIV viral load in the CNS [3] and reduces synapto-dendritic injury that occurs directly and indirectly by HIV [50,74]. We speculate that cART reduces inflammation and improves synaptic communication. A reduction in neuronal synaptic injury and inflammation could lead to increases in neuronal synchronous activity that is reflected within specific canonical resting state networks. Further, our results suggest that at least some of the neural damage due to HIV within certain brain regions may be reversible with appropriate treatment. However, longitudinal rs-fc studies of HIV+ individuals before and after initiation of cART are needed.
Interestingly, we also found that traditional markers of HIV disease status (e.g., nadir CD4 cell count, viral load) were not correlated with either cognitive performance or functional connectivity. This finding is consistent with previous studies that failed to demonstrate a consistent relationship between laboratory values and neuroimaging [16,75,76] and NP. However dissimilarities exist in the literature [77]. With the introduction of cART, traditional markers of HIV disease status have become convolved with the effects of medication status. Individuals with HIV may continue to experience neurologic dysfunction due to the virus despite good virologic control and nominal laboratory values. In fact, recent studies have found that plasma based biomarkers can fluctuate over the course of disease, and lose correlational relationships with degree of impairment [78–80]. Therefore it is important to combine metrics such as rs-fc that are sensitive to differences not seen by other typical tests used in evaluating HIV+ patients (e.g. NP testing).
The principle motivation for the investigation of cortico-striatal functional connectivity in HIV+ subjects was that subcortical regions show the highest viral burden in the brain. However, these same regions have an additional unique feature that may explain observed results. One possible explanation for our findings of reduced connectivity in the striatum can be related to the high amounts of dopaminergic (D2) receptors that are diminished in HIV+ individuals [81–83]. In chronically infected HIV+ subjects, fronto-striatal motor functions are impaired by losses of dopamine activity in the striatum [81]. Additionally, studies have shown that rs-fc is diminished between VATT areas and DMN during interventional dopamine depletion [84]. While we did not test for dopamine levels through CSF or radiolabeled tracers, it is possible that observed loss of cortico-striatal rs-fc are related to the loss of the dopamine receptor rich areas of the caudate and putamen [85,86].
While we attempted to design this study with similar cohorts for demographic variables, our study has limitations. First, we relied on self-report to describe duration of infection (DOI). These measurements may be prone to over or under estimation. Second, while chronic substance abuse was not a measure of this study, HIV− and HIV+ cohorts had similar prevalence of cocaine, barbiturates, heroin, methamphetamine, and phencyclidine (PCP) according to self-reported 6 month history. Future studies are needed to specifically address the potential deleterious effects of substance abuse on functional connectivity in HIV+ individuals. Finally, it is important to note that the lack of significant correlational relationships between rs-fc metrics and clinical laboratory values (e.g. Nadir CD4) are confounded by effects of aging and metabolic fluctuations that are not linear, therefore additional longitudinal studies are needed.
In conclusion, this study integrates previous findings of a loss of rs-fc in HIV and shows that this technique may identify the effects of both HIV and cART on brain networks. Observed changes in the cortico-striatal networks were not correlated with cognitive performance and typical laboratory markers. Finally, we found that HIV+/cART+ individuals had greater rs-fc in certain networks than HIV+/cART− individuals.
Supplementary Material
Supplementary Table 1: The size, magnitude, and coordinates of significant differences in rs-fc between HIV− and HIV+ individuals. Freesurfer based labels are provided with the peak −log10 p-value, corrected for false discovery rate (FDR). The Montreal Neurological Institute (MNI) coordinates (x,y,z) are provided for the centroid of the cluster.
Acknowledgments
Funding
The authors thank Laurie Baker M.A, Jodi Heaps PhD, and Elizabeth Westerhaus M.A. for their assistance in collecting imaging data and NP testing. This work was supported by grants from the National Institutes of Health (K23MH081786, R01NR12657, R01NR012907, R01NR014449, and R21MH0999979) (BMA), Grossman Chancellor’s Fellowship (MO), National Science Foundation (IGERT 0548890) (MO) The content is solely the responsibility of the authors and does not necessarily represent the official view of the NIH.
Footnotes
Author Contributions: M. Ortega proposed the project, analyzed the data, performed statistical analyses, drafted, and revised the manuscript. M.B. Brier drafted and revised the manuscript, and provided statistical support. B.M. Ances proposed the project, supervised the project, performed neuromedical examinations, drafted and revised the manuscript.
References
- 1.Kaul M, Garden GA, Lipton SA. Pathways to neuronal injury and apoptosis in HIV-associated dementia. Nature. 2001;410:988–994. doi: 10.1038/35073667. [DOI] [PubMed] [Google Scholar]
- 2.Anthony IC, Bell JE. The Neuropathology of HIV/AIDS. Int Rev Psychiatry. 2008;20:15–24. doi: 10.1080/09540260701862037. [DOI] [PubMed] [Google Scholar]
- 3.Heaton RK, Franklin DR, Ellis RJ, McCutchan JA, Letendre SL, Leblanc S, et al. HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol. 2011;17:3–16. doi: 10.1007/s13365-010-0006-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jessen Krut J, Mellberg T, Price RW, Hagberg L, Fuchs D, Rosengren L, et al. Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS One. 2014;9:e88591. doi: 10.1371/journal.pone.0088591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Garvey LJ, Pavese N, Politis M, Ramlackhansingh A, Brooks DJ, Taylor-Robinson SD, et al. Increased microglia activation in neurologically asymptomatic HIV-infected patients receiving effective ART. AIDS. 2014;28:67–72. doi: 10.1097/01.aids.0000432467.54003.f7. [DOI] [PubMed] [Google Scholar]
- 6.Grant I, Franklin DR, Deutsch R, Woods SP, Vaida F, Ellis RJ, et al. Asymptomatic HIV-associated neurocognitive impairment increases risk for symptomatic decline. Neurology. doi: 10.1212/WNL.0000000000000492. Published Online First: 9 May 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Clifford DB, Ances BM. HIV-associated neurocognitive disorder. Lancet Infect Dis. 2013;13:976–86. doi: 10.1016/S1473-3099(13)70269-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Holt JL, Kraft-Terry SD, Chang L. Neuroimaging studies of the aging HIV-1-infected brain. J Neurovirol. 2012;18:291–302. doi: 10.1007/s13365-012-0114-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Biswal B, Yetkin FZ, Haughton VM, Hyde JS. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med. 34 doi: 10.1002/mrm.1910340409. http://f1000.com/714597885. [DOI] [PubMed] [Google Scholar]
- 10.Fox MD, Snyder AZ, Vincent JL, Corbetta M, Essen V, Dc, et al. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A. 2005;102:9673–9678. doi: 10.1073/pnas.0504136102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD. Neurodegenerative diseases target large-scale human brain networks. Neuron. 2009;62:42–52. doi: 10.1016/j.neuron.2009.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Smith SM, Fox PT, Miller KL, Glahn DC, Fox PM, Mackay CE, et al. Correspondence of the brain’s functional architecture during activation and rest. Proc Natl Acad Sci U S A. 2009;106:13040–5. doi: 10.1073/pnas.0905267106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Damoiseaux JS, Rombouts SaRB, Barkhof F, Scheltens P, Stam CJ, Smith SM, et al. Consistent resting-state networks across healthy subjects. Proc Natl Acad Sci U S A. 2006;103:13848–53. doi: 10.1073/pnas.0601417103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Van den Heuvel MP, Mandl RCW, Kahn RS, Hulshoff Pol HE. Functionally linked resting-state networks reflect the underlying structural connectivity architecture of the human brain. Hum Brain Mapp. 2009;30:3127–41. doi: 10.1002/hbm.20737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hampson M, Peterson BS, Skudlarski P, Gatenby JC, Gore JC. Detection of functional connectivity using temporal correlations in MR images. Hum Brain Mapp. 2002;15:247–62. doi: 10.1002/hbm.10022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thomas JB, Brier MR, Snyder AZ, Vaida FF, Ances BM. Pathways to neurodegeneration: effects of HIV and aging on resting-state functional connectivity. Neurology. 2013;80:1186–93. doi: 10.1212/WNL.0b013e318288792b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang X, Foryt P, Ochs R, Chung J-H, Wu Y, Parrish T, et al. Abnormalities in resting-state functional connectivity in early human immunodeficiency virus infection. Brain Connect. 2011;1:207–17. doi: 10.1089/brain.2011.0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Woods SP, Moore DJ, Weber E, Grant I. Cognitive neuropsychology of HIV-associated neurocognitive disorders. Neuropsychol Rev. 2009;19:152–68. doi: 10.1007/s11065-009-9102-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Malaspina L, Woods SP, Moore DJ, Depp C, Letendre SL, Jeste D, et al. Successful cognitive aging in persons living with HIV infection. J Neurovirol. 2011;17:110–9. doi: 10.1007/s13365-010-0008-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Stout JC, Ellis RJ, Jernigan TL, Archibald SL, Abramson I, Wolfson T, et al. Progressive cerebral volume loss in human immunodeficiency virus infection: a longitudinal volumetric magnetic resonance imaging study. HIV Neurobehavioral Research Center Group. Arch Neurol. 1998;55:161–8. doi: 10.1001/archneur.55.2.161. [DOI] [PubMed] [Google Scholar]
- 21.Berger JR, Arendt G. HIV dementia: the role of the basal ganglia and dopaminergic systems. J Psychopharmacol. doi: 10.1177/026988110001400304. Published Online First: 2010. [DOI] [PubMed] [Google Scholar]
- 22.Ances BM, Ortega M, Vaida F, Heaps J, Paul R. Independent effects of HIV, aging, and HAART on brain volumetric measures. J Acquir Immune Defic Syndr. 2012;59:469–77. doi: 10.1097/QAI.0b013e318249db17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Becker JT, Sanders J, Madsen SK, Ragin A, Kingsley L, Maruca V, et al. Subcortical brain atrophy persists even in HAART-regulated HIV disease. Brain Imaging Behav. 2011;5:77–85. doi: 10.1007/s11682-011-9113-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hardy DJ, Vance DE. The neuropsychology of HIV/AIDS in older adults. Neuropsychol Rev. 2009;19:263–72. doi: 10.1007/s11065-009-9087-0. [DOI] [PubMed] [Google Scholar]
- 25.Tozzi V. Cognitive function in treated HIV patients. Neurobehav HIV Med. 2010;95 [Google Scholar]
- 26.Scott JC, Woods SP, Carey CL, Weber E, Bondi MW, Grant I. Neurocognitive consequences of HIV infection in older adults: an evaluation of the “cortical” hypothesis. AIDS Behav. 2011;15:1187–96. doi: 10.1007/s10461-010-9815-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Melrose RJ, Tinaz S, Castelo JMB, Courtney MG, Stern CE. Compromised fronto-striatal functioning in HIV: an fMRI investigation of semantic event sequencing. Behav Brain Res. 2008;188:337–47. doi: 10.1016/j.bbr.2007.11.021. [DOI] [PubMed] [Google Scholar]
- 28.Ernst T, Yakupov R, Nakama H, Crocket G, Cole M, Watters M, et al. Declined neural efficiency in cognitively stable human immunodeficiency virus patients. Ann Neurol. 2009;65:316–25. doi: 10.1002/ana.21594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Barkhof F, Haller S, Rombouts SaRB. Resting-state functional MR imaging: a new window to the brain. Radiology. 2014;272:29–49. doi: 10.1148/radiol.14132388. [DOI] [PubMed] [Google Scholar]
- 30.Van Gorp WG, Miller EN, Satz P, Visscher B. Neuropsychological performance in HIV-1 immunocompromised patients: a preliminary report. J Clin Exp Neuropsychol. 1989;11:763–73. doi: 10.1080/01688638908400930. [DOI] [PubMed] [Google Scholar]
- 31.Robertson KR, Smurzynski M, Parsons TD, Wu K, Bosch RJ, Wu J, et al. The prevalence and incidence of neurocognitive impairment in the HAART era. AIDS. 2007;21:1915–21. doi: 10.1097/QAD.0b013e32828e4e27. [DOI] [PubMed] [Google Scholar]
- 32.Tozzi V, Balestra P, Bellagamba R, Corpolongo A, Salvatori MF, Visco-Comandini U, et al. Persistence of neuropsychologic deficits despite long-term highly active antiretroviral therapy in patients with HIV-related neurocognitive impairment: prevalence and risk factors. J Acquir Immune Defic Syndr. 2007;45:174–82. doi: 10.1097/QAI.0b013e318042e1ee. [DOI] [PubMed] [Google Scholar]
- 33.Yeo BTT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65. doi: 10.1152/jn.00338.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fischl B, Sereno MI, Dale AM. Cortical surface-based analysis. II: Inflation, flattening, and a surface-based coordinate system. Neuroimage. 1999;9:195–207. doi: 10.1006/nimg.1998.0396. [DOI] [PubMed] [Google Scholar]
- 35.Fischl B. Automatically Parcellating the Human Cerebral Cortex. Cereb Cortex. 2004;14:11–22. doi: 10.1093/cercor/bhg087. [DOI] [PubMed] [Google Scholar]
- 36.Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999;9:179–94. doi: 10.1006/nimg.1998.0395. [DOI] [PubMed] [Google Scholar]
- 37.Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80. doi: 10.1016/j.neuroimage.2006.01.021. [DOI] [PubMed] [Google Scholar]
- 38.Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, et al. Whole Brain Segmentation: Neurotechnique Automated Labeling of Neuroanatomical Structures in the Human Brain. Neuron. 2002;33:341–355. doi: 10.1016/s0896-6273(02)00569-x. [DOI] [PubMed] [Google Scholar]
- 39.Choi EY, Yeo BTT, Buckner RL. The organization of the human striatum estimated by intrinsic functional connectivity. J Neurophysiol. 2012;108:2242–63. doi: 10.1152/jn.00270.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Power JD, Barnes Ka, Snyder AZ, Schlaggar BL, Petersen SE. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage. 2012;59:2142–54. doi: 10.1016/j.neuroimage.2011.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Van Dijk KRa, Hedden T, Venkataraman A, Evans KC, Lazar SW, Buckner RL. Intrinsic functional connectivity as a tool for human connectomics: theory, properties, and optimization. J Neurophysiol. 2010;103:297–321. doi: 10.1152/jn.00783.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hallquist MN, Hwang K, Luna B. The nuisance of nuisance regression: spectral misspecification in a common approach to resting-state fMRI preprocessing reintroduces noise and obscures functional connectivity. Neuroimage. 2013;82:208–25. doi: 10.1016/j.neuroimage.2013.05.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Greve DN, Fischl B. Accurate and robust brain image alignment using boundary-based registration. Neuroimage. 2009;48:63–72. doi: 10.1016/j.neuroimage.2009.06.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Polimeni JR, Fischl B, Greve DN, Wald LL. Laminar analysis of 7T BOLD using an imposed spatial activation pattern in human V1. Neuroimage. 2010;52:1334–46. doi: 10.1016/j.neuroimage.2010.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Arendt G, Hefter H, Elsing C, Strohmeyer GHJ. Motor dysfunction in HIV-infected patients without clinically detectable central-nervous deficit. J. doi: 10.1007/BF00315660. Published Online First: 1990. http://www.springerlink.com/index/K76465858137QW16.pdf. [DOI] [PubMed]
- 46.Picard N, Strick P. Imaging the premotor areas. Curr Opin Neurobiol. 2001;11:663–672. doi: 10.1016/s0959-4388(01)00266-5. [DOI] [PubMed] [Google Scholar]
- 47.Field AP. Meta-analysis of correlation coefficients: a Monte Carlo comparison of fixed- and random-effects methods. Psychol Methods. 2001;6:161–80. doi: 10.1037/1082-989x.6.2.161. [DOI] [PubMed] [Google Scholar]
- 48.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. 1995:289–300. [Google Scholar]
- 49.Raichle ME, Macleod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL. A default mode of brain function. 2001 doi: 10.1073/pnas.98.2.676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Crews L, Patrick C, Achim CL, Everall IP, Masliah E. Molecular pathology of neuro-AIDS (CNS-HIV) Int J Mol Sci. 2009;10:1045–63. doi: 10.3390/ijms10031045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lu S-M, Tremblay M-È, King IL, Qi J, Reynolds HM, Marker DF, et al. HIV-1 Tat-induced microgliosis and synaptic damage via interactions between peripheral and central myeloid cells. PLoS One. 2011;6:e23915. doi: 10.1371/journal.pone.0023915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Du Plessis S, Vink M, Joska Ja, Koutsilieri E, Stein DJ, Emsley R. HIV infection and the fronto-striatal system: a systematic review and meta-analysis of fMRI studies. AIDS. 2013:1–9. doi: 10.1097/QAD.0000000000000151. [DOI] [PubMed] [Google Scholar]
- 53.Fama R, Rosenbloom MJ, Sassoon Sa, Rohlfing T, Pfefferbaum A, Sullivan EV. Thalamic volume deficit contributes to procedural and explicit memory impairment in HIV infection with primary alcoholism comorbidity. Brain Imaging Behav. 2014;d doi: 10.1007/s11682-013-9286-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Van den Heuvel MP, Sporns O. Rich-club organization of the human connectome. J Neurosci. 2011;31:15775–86. doi: 10.1523/JNEUROSCI.3539-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tomasi D, Volkow ND, Wang R, Telang F, Wang G, Chang L, et al. Dopamine transporters in striatum correlate with deactivation in the default mode network during visuospatial attention. PLoS One. 2009;4:e6102. doi: 10.1371/journal.pone.0006102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jahfari S, Waldorp L, van den Wildenberg WPM, Scholte HS, Ridderinkhof KR, Forstmann BU. Effective connectivity reveals important roles for both the hyperdirect (fronto-subthalamic) and the indirect (fronto-striatal-pallidal) fronto-basal ganglia pathways during response inhibition. J Neurosci. 2011;31:6891–9. doi: 10.1523/JNEUROSCI.5253-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zink CF, Pagnoni G, Martin ME, Dhamala M, Berns GS. Human striatal response to salient nonrewarding stimuli. J Neurosci. 2003;23:8092–7. doi: 10.1523/JNEUROSCI.23-22-08092.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Dang L, Donde A, Madison C. Striatal dopamine influences the default mode network to affect shifting between object features. J Cogn …. 2012;24:1960–1970. doi: 10.1162/jocn_a_00252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Buckner RL, Andrews-Hanna JR, Schacter DL. The brain’s default network: anatomy, function, and relevance to disease. Ann N Y Acad Sci. 2008;1124:1–38. doi: 10.1196/annals.1440.011. [DOI] [PubMed] [Google Scholar]
- 60.Thompson PM, Dutton RA, Hayashi KM, Toga AW, Lopez OL, Aizenstein HJ, et al. Thinning of the cerebral cortex visualized in HIV/AIDS reflects CD4+ T lymphocyte decline. Proc Natl Acad Sci USA. 2005;102:15647–15652. doi: 10.1073/pnas.0502548102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Thomas JB, Brier MR, Ortega M, Benzinger TL, Ances BM. Weighted brain networks in disease: centrality and entropy in human immunodeficiency virus and aging. Neurobiol Aging. doi: 10.1016/j.neurobiolaging.2014.06.019. Published Online First: 21 June 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Becker KM, Heinrichs-Graham E, Fox HS, Robertson KR, Sandkovsky U, O’Neill J, et al. Decreased MEG beta oscillations in HIV-infected older adults during the resting state. J Neurovirol. 2013;19:586–94. doi: 10.1007/s13365-013-0220-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wilson TW, Heinrichs-Graham E, Becker KM, Aloi J, Robertson KR, Sandkovsky U, et al. Multimodal neuroimaging evidence of alterations in cortical structure and function in HIV-infected older adults. Hum Brain Mapp. 2014;00 doi: 10.1002/hbm.22674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.DHHS Panel on Antiretroviral Guidelines for Adults. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. Afr J Med Pract. 2009;5:79–104. [PubMed] [Google Scholar]
- 65.Caniglia EC, Cain LE, Justice A, Tate J, Logan R, Sabin C, et al. Antiretroviral penetration into the CNS and incidence of AIDS-defining neurologic conditions. Neurology. 2014;83:134–41. doi: 10.1212/WNL.0000000000000564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ellis R, Langford D, Masliah E. HIV and antiretroviral therapy in the brain: neuronal injury and repair. Nat Rev Neurosci. 2007;8:33–44. doi: 10.1038/nrn2040. [DOI] [PubMed] [Google Scholar]
- 67.Joska Ja, Gouse H, Paul RH, Stein DJ, Flisher AJ. Does highly active antiretroviral therapy improve neurocognitive function? A systematic review. J Neurovirol. 2010;16:101–14. doi: 10.3109/13550281003682513. [DOI] [PubMed] [Google Scholar]
- 68.Joska Ja, Gouse H, Paul RH, Stein DJ, Flisher AJ. Does highly active antiretroviral therapy improve neurocognitive function? A systematic review. J Neurovirol. 2010;16:101–14. doi: 10.3109/13550281003682513. [DOI] [PubMed] [Google Scholar]
- 69.Juengst SB, Aizenstein HJ, Figurski J, Lopez OL, Becker JT. Alterations in the hemodynamic response function in cognitively impaired HIV/AIDS subjects. J Neurosci Methods. 2007;163:208–12. doi: 10.1016/j.jneumeth.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 70.Ge Y, Law M, Johnson G, Herbert J, Babb JS, Mannon LJ, et al. Dynamic Susceptibility Contrast Perfusion MR Imaging of Multiple Sclerosis Lesions: Characterizing Hemodynamic Impairment and Inflammatory Activity. 2005:1539–1547. [PMC free article] [PubMed] [Google Scholar]
- 71.Brydon L, Harrison NA, Walker C, Steptoe A, Critchley HD. Peripheral inflammation is associated with altered substantia nigra activity and psychomotor slowing in humans. Biol Psychiatry. 2008;63:1022–9. doi: 10.1016/j.biopsych.2007.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Young AC, Yiannoutsos CT, Hegde M, Lee E, Peterson J, Walter R, et al. Cerebral metabolite changes prior to and after antiretroviral therapy in primary HIV infection. Neurology. doi: 10.1212/WNL.0000000000000932. Published Online First: 26 September 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sailasuta N, Ross W, Ananworanich J, Chalermchai T, DeGruttola V, Lerdlum S, et al. Change in brain magnetic resonance spectroscopy after treatment during acute HIV infection. PLoS One. 2012;7:e49272. doi: 10.1371/journal.pone.0049272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Masliah E, Heaton RK, Marcotte TD, Ellis RJ, Wiley CA, Mallory M, et al. Dendritic injury is a pathological substrate for human immunodeficiency virus-related cognitive disorders. HNRC Group. The HIV Neurobehavioral Research Center. Ann Neurol. 1997;42:963–72. doi: 10.1002/ana.410420618. [DOI] [PubMed] [Google Scholar]
- 75.Chang L, Andres M, Sadino J, Jiang CS, Nakama H, Miller E, et al. Impact of apolipoprotein E ɛ4 and HIV on cognition and brain atrophy: antagonistic pleiotropy and premature brain aging. Neuroimage. 2011;58:1017–27. doi: 10.1016/j.neuroimage.2011.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ragin AB, Du H, Ochs R, Wu Y, Sammet CL, Shoukry A, et al. Structural brain alterations can be detected early in HIV infection. Neurology. 2012;79:2328–34. doi: 10.1212/WNL.0b013e318278b5b4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gannon P, Khan MZ, Kolson DL. Current understanding of HIV-associated neurocognitive disorders pathogenesis. Curr Opin Neurol. 2011;24:275–83. doi: 10.1097/WCO.0b013e32834695fb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Barreiro P, Labarga P, Fernández-Montero JV, Poveda E, de Mendoza C, Sánchez C, et al. Longitudinal changes in viral RNA concentration in patients with chronic hepatitis C and/or HIV infection in the absence of antiviral therapy. J Clin Virol. 2013;58:391–5. doi: 10.1016/j.jcv.2013.06.031. [DOI] [PubMed] [Google Scholar]
- 79.Luz PM, Grinsztejn B, Velasque L, Pacheco AG, Veloso VG, Moore RD, et al. Long-Term CD4+ Cell Count in Response to Combination Antiretroviral Therapy. PLoS One. 2014;9:e93039. doi: 10.1371/journal.pone.0093039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Joska Ja, Westgarth-Taylor J, Myer L, Hoare J, Thomas KGF, Combrinck M, et al. Characterization of HIV-Associated Neurocognitive Disorders among individuals starting antiretroviral therapy in South Africa. AIDS Behav. 2011;15:1197–203. doi: 10.1007/s10461-010-9744-6. [DOI] [PubMed] [Google Scholar]
- 81.Chang L, Wang G-J, Volkow ND, Ernst T, Telang F, Logan J, et al. Decreased brain dopamine transporters are related to cognitive deficits in HIV patients with or without cocaine abuse. Neuroimage. 2008;42:869–78. doi: 10.1016/j.neuroimage.2008.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wang G-J, Chang L, Volkow ND, Telang F, Logan J, Ernst T, et al. Decreased brain dopaminergic transporters in HIV-associated dementia patients. Brain. 2004;127:2452–8. doi: 10.1093/brain/awh269. [DOI] [PubMed] [Google Scholar]
- 83.Obermann M, Küper M, Kastrup O, Yaldizli O, Esser S, Thiermann J, et al. Substantia nigra hyperechogenicity and CSF dopamine depletion in HIV. J Neurol. 2009;256:948–53. doi: 10.1007/s00415-009-5052-3. [DOI] [PubMed] [Google Scholar]
- 84.Cole DM, Beckmann CF, Oei NYL, Both S, van Gerven JMa, Rombouts SaRB. Differential and distributed effects of dopamine neuromodulations on resting-state network connectivity. Neuroimage. 2013;78:59–67. doi: 10.1016/j.neuroimage.2013.04.034. [DOI] [PubMed] [Google Scholar]
- 85.Cools R. Dopaminergic control of the striatum for high-level cognition. Curr Opin Neurobiol. 2011;21:402–7. doi: 10.1016/j.conb.2011.04.002. [DOI] [PubMed] [Google Scholar]
- 86.Smith Y, Kieval JZ. Anatomy of the dopamine system in the basal ganglia. Trends Neurosci. 2000;23:S28–33. doi: 10.1016/s1471-1931(00)00023-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Table 1: The size, magnitude, and coordinates of significant differences in rs-fc between HIV− and HIV+ individuals. Freesurfer based labels are provided with the peak −log10 p-value, corrected for false discovery rate (FDR). The Montreal Neurological Institute (MNI) coordinates (x,y,z) are provided for the centroid of the cluster.


