Abstract
Background
POTEE (POTE ankyrin domain family, member E) is a newly identified cancer-testis antigen that has been found to be expressed in a wide variety of human cancers including cancers of the colon, prostate, lung, breast, ovary, and pancreas.
Aim
To measure the serum levels of POTEE in patients with non-small-cell lung cancer (NSCLC) and to explore the clinical significance of POTEE in NSCLC.
Patients and Methods
104 NSCLC patients, 66 benign lung disease patients and 80 healthy volunteers were enrolled in this study from May 2013 to February 2014. Serum POTEE levels were measured using enzyme-linked immunosorbent assay (ELISA). Numerical variables were recorded as means ± standard deviation (SD) and analyzed by independent t tests. Categorical variables were calculated as rates and were analyzed using a χ2 test or Fisher’s exact test. Survival curves were estimated and compared using the Kaplan-Meier method and log-rank tests.
Results
Serum POTEE levels were significantly higher in NSCLC patients than in benign lung disease patients and healthy controls (mean ± SD [pg/ml], 324.38± 13.84 vs. 156.93 ± 17.38 and 139.09 ± 15.80, P<0.001) and were significantly correlated with TNM stage. Survival analysis revealed that patients with low serum POTEE had longer progression-free survival (PFS) than those with high serum POTEE (P=0.021). Cox multivariate analysis indicated that POTEE was an independent prognostic factor of progression-free survival (P =0.009, hazard ratio, 2.440).
Conclusions
Serum POTEE level in NSCLC patients is associated with TNM stage and is a potential prognostic factor.
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide, with non-small-cell lung cancer (NSCLC) representing approximately 80% of these cases [1, 2]. Most patients are diagnosed at an advanced stage of disease [3]; they have little prospect of effective and curative treatment. Five-year survival rates are less than 15% [4]. A promising breakthrough with the potential to improve outcomes for NSCLC patients involves the introduction of validated biomarkers into clinical management. These biomarkers may be crucial not only for early diagnosis but also to inform treatment decisions to achieve optimal therapeutic interventions [5, 6].
Cancer testis (CT) antigens are a type of protein that is expressed in normal adult tissue only in the testis as well as a variety of tumors of different histological origins. In view of their pattern of expression and high immunogenicity, CT antigenss are considered very attractive candidates as cancer biomarkers and vaccines [7–9]. POTEE (POTE ankyrin domain family, member E) is a newly identified cancer testis antigen expressed in normal testis, ovary, and placenta, and in many cancers, including those of the prostate, colon, lung, breast, ovary, and pancreas [10–12]. In humans, POTE gene family consists of 13 highly homologous members located on 8 different chromosomes. Previous studies have shown that POTE molecules are localized in the cytoplasm towards the inner aspect of cellular membrane [13]. POTEE, a dominant subtype expressed in many cancers and cancer cell lines [11, 14]. Our previous study revealed the differential expression of POTEE between cisplatin-resistant A549/DDP and its parental A549 cells by proteomics analysis [15].
However, the functions and roles of POTEE in lung cancer remain unclear. In this study, we preliminarily explored the relationship between serum POTEE level and clinical characteristics and survival in NSCLC patients.
Materials and Methods
Patient, Healthy Controls and Serum Samples
Peripheral blood was drawn from patients and healthy donors at Shanghai Pulmonary Hospital from May 2013 to February 2014. Sera were obtained from a study cohort of 250 participants as follows: 104 patients with NSCLCs, including adenocarcinoma, squamous cell carcinoma, and large-cell anaplastic carcinoma; 66 benign lung disease patients; and 80 healthy donors. The inclusion criteria for the NSCLC patients were as follows: (1) pathologically confirmed NSCLC; (2) age ≥18 years old; (3) had not received any anti-tumor therapy. Tumor staging was determined according to the 2009 TNM staging classification system. Demographic and pathological data, including age, gender, and smoking history, were collected. The inclusion criteria for benign lung disease patients were as follows: (1) no history of cancer; (2) benign lung disease patients confirmed by pathogen or pathology detection; (3) the diagnosis of benign lung disease patients were bronchiectasis, chronic obstructive pulmonary disease, tuberculosis, pneumonia, chronic bronchitis and lung abscess, the characteristics and demographics of these patients are included in S1 Table; (4) age ≥18 years old. Patients that did not meet aforementioned criteria were excluded. This study was approved by the Ethics Committee of Shanghai Pulmonary Hospital. Written informed consent was obtained from each study participant.
Enzyme-Linked Immunosorbent Assay
Serum POTEE levels were determined by enzyme-linked immunosorbent assay (ELISA) using an immunoassay kit (Wuhan Huamei Biotech, China; Cat. no.CSB-EL740863 HU) according to the manufacturer’s protocol. The optical density (OD) at 450 nm was measured, and the standard curves were established with OD 450 as the Y axis; these curves were used to determine protein expression levels. Results are reported as the concentration of POTEE (pg/ml) in the serum sample.
Measurement of common serum tumor markers
Serum tumor markers, including carcinoeyonmbric antigen (CEA), cytokeratin (CYFRA21-1) and carbohydrate antigen 19–9 (CA19-9) were determined by radioimmunoassay (RIA) using CEA, CYFRA21-1(CIS Bio International) and CA19-9 (China Institute of Atomic Energy) RIA kits according to the manufacturer’s protocol. Results were gained by 1470 WIZA RDTM counter instruments.
Statistical Analysis
Statistical analyses were performed using SPSS 17.0 software (Chicago, IL, USA) and GraphPad Prism 5 (GraphPad Software, San Diego, CA, USA). Numerical variables were recorded as means ± SD and analyzed using independent t tests. Categorical variables are presented as rates and were analyzed using χ2 test or Fisher’s exact test. To evaluate the diagnostic potential of POTEE, the receiver operating characteristics (ROC) curve was plotted using data produced by logistic regression analysis, and the area under the curve (AUC) was calculated. Survival curves were estimated and compared using the Kaplan-Meier method and log-rank tests. Univariate Cox regression was performed on each clinical covariate to examine its influence on patient survival. Multivariate models were based on step-wise addition. A P value <0.05 was considered statistically significant.
Results
Demographic Characteristics of All Study Participants
Our study cohort of 250 participants comprised 104 NSCLC patients, 66 benign lung disease patients, and 80 healthy subjects. Mean age was 61.3 ± 9.5 yr in the NSCLC patients group, 58.5 ± 11.5 yr in the benign lung disease patients group, and 57.9 ± 13.6 yr in the healthy control group (P>0.05). The basic characteristics of 104 patients with NSCLC, 66 benign lung disease patients, and 80 healthy control subjects are summarized in Table 1.
Table 1. Basic Characteristics of Study Participants.
| Variables | NSCLC (n, %) | Benign (n, %) | Controls (n, %) | P value |
|---|---|---|---|---|
| Age | 0.097 | |||
| ≥65 yr | 33 (31.7) | 21 (31.8) | 26 (32.5) | |
| <65 yr | 71 (68.3) | 45 (68.2) | 54 (67.5) | |
| Sex | 0.410 | |||
| Male | 59 (56.7) | 43 (65.2) | 52 (65.0) | |
| Female | 45 (43.3) | 23 (34.8) | 28 (35.0) | |
| Smoking status | 0.232 | |||
| Yes | 66 (63.5) | 34 (51.5) | 43 (53.7) | |
| No | 38 (36.5) | 32 (48.5) | 37 (46.3) |
Serum POTEE levels in NSCLC Patients, Benign Lung Disease Patients, and Healthy Control Subjects
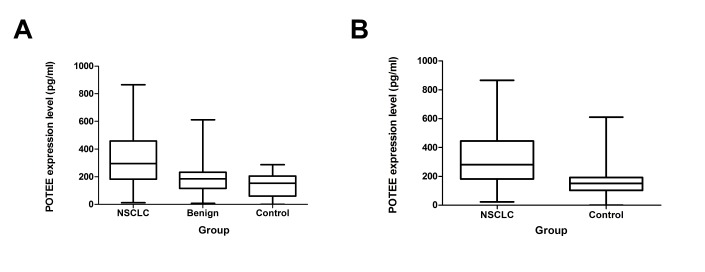
Serum POTEE levels were significantly higher in NSCLC patients than in benign lung disease patients and healthy control subjects. The mean serum POTEE level was 324.38 ± 13.84 pg/ml for the NSCLC patient group, 156.93 ± 17.38 pg/ml for the benign lung disease group, and 139.09 ± 15.80 pg/ml for the healthy control group (P<0.001) (Fig 1A). The mean serum POTEE level in NSCLC patients was also significantly higher than the mean of 147.15 ± 6.58 pg/ml for all non-NSCLC participants (P<0.001) (Fig 1B).
Fig 1.
1A. Serum POTEE levels in NSCLC patients, benign disease and healthy controls. Mean POTEE level was 324.38 ± 13.84 pg/ml in the NSCLC group, 156.93 ± 17.38 pg/ml in benign lung disease group, and 139.09 ± 15.8 in control group (P<0.001). 1B. Serum POTEE levels in patients with NSCLC and controls. Mean POTEE level was 324.38 ± 13.84 pg/ml in the NSCLC group and 139.09 ± 15.8 pg/ml in control group (P<0.001).
The Potential of POTEE as a Serologic Tumor Biomarker in NSCLC
A ROC curve analysis was carried out to assess the value of POTEE in NSCLC diagnosis. ROC curve is to reflect the sensitivity and specificity of continuous variable comprehensive index, the area under the curve the greater the diagnosis accuracy is higher. The AUC was 0.793 (95% confidence interval [CI], 0.7–0.9). We chose the point with highest sensitivity and specificity, 205.27 pg/ml, as the cutoff value. With the cutoff value, which was defined as the normal value based on the mean value (plus two standard deviations) obtained from healthy controls, serum POTEE achieved a diagnostic sensitivity of 68.3% and a specificity of 82.9% (Fig 2A). We also detected the serum levels of commonly used serologic tumor markers (TM), including carcinoeyonmbric antigen (CEA), carbohydrate antigen 19–9 (CA19-9), and cytokeratin (CYFRA21-1) in the NSCLC group. Their sensitivities were 51.0% (CEA), 56.3% (CA19-9), and 60.4% (CYFRA21-1), the specificity was 95.2% (CEA), 94.5% (CA19-9), and 93.8% (CYFRA21-1), and the accuracy was 77.7% (CEA), 79.3% (CA19-9), and 80.6% (CYFRA21-1) (Fig 2B). Among the 104 NSCLC patients, the combined detection sensitivity of CEA, CA19-9, CYFRA21-1, and POTEE was 89.3%, which was obviously higher than the sensitivity of any single detection (P<0.05).
Fig 2.
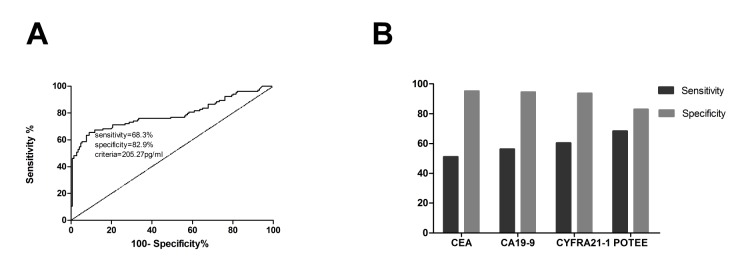
2A. ROC curve of POTEE as a serologic tumor marker in NSCLC. Using a cut off value 205.27 pg/ml, ROC analysis revealed an area under curve (AUC) of 0.793 (P<0.01), indicating a high sensitivity and specificity to differentiate benign lung disease group and healthy controls from NSCLC patients. Fig 2B. Diagnostic values of CEA, CYFRA 21–1, CA199 and POTEE in N SCLC. CEA, CYEFRA 21–1, CA199 and POTEE sensitivity were 51.0%, 56.3%, 60.4% and 68.3%, the specificity were 95.2%, 94.5%, 93.8% and 82.9%.
Relation of Serum POTEE to Clinicopathological Characteristics
As shown in Table 2, serum POTEE level was significantly correlated with TNM stage. Patients in advanced TNM stages had higher POTEE levels than those with early stages of lung cancer (P = 0.036). However, no significant correlation was observed between serum POTEE level and other clinicopathologic parameters, including age, gender, smoking history, pathology, or family history.
Table 2. Relation of Serum POTEE to Clinicopathological Characteristics of 104 Patients with NSCLC.
| Group | Categories | n (%) | POTEE levels (pg/ml) | P value |
|---|---|---|---|---|
| Gender | Male | 59 (56.7) | 327.30 ± 25.83 | 0.864 |
| Female | 45 (43.3) | 320.55 ± 29.58 | ||
| Age, yr | ≥65 | 33 (31.7) | 311.64 ± 34.51 | 0.656 |
| <65 | 71 (68.3) | 330.30 ± 23.53 | ||
| Histological type | Adenocarcinoma | 64 (61.5) | 294.03 ± 25.02 | 0.907 |
| Squamous | 29 (27.9) | 312.62 ± 37.17 | ||
| Adenosquamous | 11 (10.6) | 309.67 ± 60.35 | ||
| TNM stage # | I–IIIA | 25 (24.0) | 252.60 ± 38.85 | 0.036* |
| IIIB–IV | 79 (76.0) | 347.09 ± 21.85 | ||
| PS score | 0–1 | 94 (90.4) | 313.13 ± 20.54 | 0.228 |
| 2 | 10 (9.6) | 229.85 ± 59.75 | ||
| Family history | Yes | 11 (10.6) | 262.06 ± 80.08 | 0.495 |
| No | 93 (89.4) | 305.45 ± 19.73 | ||
| Smoking history | Yes | 66 (63.5) | 338.63 ± 28.19 | 0.142 |
| Never | 38 (36.5) | 279.12 ± 25.78 |
PS, performance status;
#, 2 patients in stage I, 5 patients in stage II, 18 patients in stage IIIA, 34 patients in stage IIIB, 45 patients in stage IV;
*, serum POTEE level was associated with TNM stage.
Association Between Serum POTEE Level and Efficacy of Platinum-Based Chemotherapy
We analyzed whether serum POTEE level is related to tumor response to chemotherapy. Among the 104 patients with NSCLC, 76 patients were at TNM stage IIIb–IV and received doublet chemotherapy. Among 76 patients, 33 patients had been treated with gemcitabine plus cisplatin, 23 had been treated with vinorelbine plus cisplatin, 11 had been treated with paclitaxel plus cisplatin and 9 had been treated with vinorelbine plus carboplatin. The POTEE serum level cutoff value of 205.27 pg/ml with highest sensitivity and specificity was selected to categorize patients as high POTEE (n = 43) and low POTEE (n = 33) groups. We analyzed patient responses to platinum-based chemotherapy (Table 3). For patients with high POTEE expression (≥205.27 pg/ml), objective response rate (ORR) and disease control rate (DCR) were significantly lower than for those with low POTEE expression (<205.27 pg/ml) (P = 0.023; P = 0.043). Serum POTEE expression was strongly negatively correlated with the response of patients to platinum-based chemotherapy.
Table 3. Association Between Serum POTEE Level and Efficacy.
| High POTEE expression (≥205.27 pg/ml) | Low POTEE expression (<205.27 pg/ml) | P value | |
|---|---|---|---|
| PR, n | 7 | 13 | |
| SD, n | 12 | 9 | |
| PD, n | 24 | 11 | |
| ORR, % | 16.3 | 39.3 | 0.023 |
| DCR, % | 44.2 | 66.6 | 0.043 |
ORR, objective response rate; DCR, disease control rate; PR, partial response; PD, progression disease; SD, stable disease.
Association between Serum POTEE Level and Survival
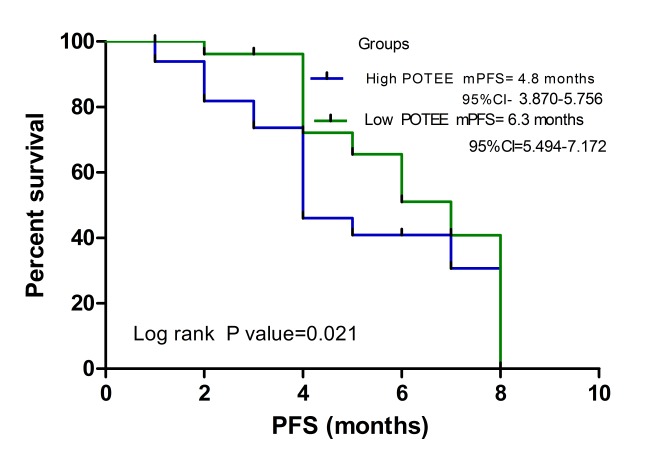
In the Kaplan-Meier survival curve analysis, the group of patients with low POTEE expression had a significantly longer median progression-free survival (PFS; 6.3 months; 95% CI, 5.494–7.172) than the group with high POTEE expression (PFS, 4.8 months; 95% CI, 3.870–5.756) (P = 0.021) (Fig 3). As shown in Table 4, a Cox proportional hazards analysis was used to further evaluate the potential of serum POTEE expression level as a prognostic biomarker. Univariate analysis showed that serum POTEE expression level was associated with prognosis (HR, 1.913; P = 0.040). In the multivariate Cox proportional hazards analysis, low POTEE expression level was significantly associated with longer PFS (HR, 2.440; P = 0.009). As expected, disease stage was strongly associated with shorter PFS in both univariate and multivariate analyses (P<0.05).
Fig 3. Kaplan-Meier survival curves in relation to serum POTEE level in patients with NSCLC.
Survival curves were analyzed by Kaplan-Meier method and log-rank test. Patients with high POTEE levels had a significantly poorer survival than those with low POTEE levels (P = 0.021).
Table 4. Univariate and Multivariate Analyses of POTEE Status with Regard to PFS.
| Univariate analysis | Multivariate analysis | |||||
|---|---|---|---|---|---|---|
| Variables | HR | 95% CI | P value | HR | 95% CI | P value |
| POTEE level (low vs. high) | 1.913 | 1.030–3.551 | 0.040* | 2.440 | 1.252–4.757 | 0.009* |
| Age (≥60 vs. <60, yr) | 1.171 | 0.653–2.097 | 0.597 | 1.168 | 0.633–2.154 | 0.620 |
| Sex (male vs. female) | 0.728 | 0.399–1.328 | 0.301 | 0.591 | 0.314–1.113 | 0.103 |
| Histology | 1.115 | 0.753–1.652 | 0.586 | 1.140 | 0.722–1.801 | 0.573 |
| TNM stage (I-IIIA vs. IIIB-IV) | 4.089 | 1.818–9.199 | 0.001* | 4.789 | 2.082–11.014 | 0.001* |
| Smoking history | 1.321 | 0.720–2.424 | 0.369 | 0.807 | 0.366–2.776 | 0.593 |
| Family history | 0.557 | 0.172–1.798 | 0.328 | 1.927 | 0.504–7.376 | 0.338 |
CI, confidence interval; HR, hazard ratio;
*, PFS was associated with POTEE level and TNM stage.
Discussion
To our knowledge, this study is the first to assess POTEE expression in serum by ELISA to examine its clinical significance in NSCLC. We observed that the expression of POTEE in sera was significantly higher in NSCLC patients compared with benign lung disease patients and healthy volunteers. Furthermore, we preliminarily analyzed the clinical significance of POTEE in NSCLC patients. Levels of serum POTEE were significantly correlated with TNM stage. Survival analysis revealed that patients with low serum POTEE levels had longer progression-free survival (PFS) than those with high POTEE levels. High POTEE expression levels were associated with poor response to chemotherapy in NSCLC patients. Moreover, multivariate analysis also showed that high POTEE expression was an independent factor for poor survival in NSCLC patients. These results suggest that POTEE expression may be associated with prognosis of NSCLC patients and could serve as a potential biomarker.
Up to date, few previous studies reported an association between POTEE and cancer prognosis because POTEE is a newly identified CT antigen. The gene family POTE is classified into three groups: group 1 includes only POTE 8; group 2 includes POTE15, -18, and -21; and group 3 includes POTE2, -14, and -22 [16, 17]. In the initial studies, POTE proteins were thought to be located at the inner plasma membrane and were classified as CT antigens because they are expressed in many cancers and some normal reproductive tissues [16, 17]. Several previous studies have reported POTE roles in both physiologic and pathologic conditions, including the cytoskeletal structure [8], apoptotic pathways [11], epigenetic regulation of tumor progression [18], and lineage-specific differentiation of embryonic cells [19]. However, the function of POTE in cancer remains unknown. Refield et al. studied the subcellular distribution of all POTE proteins and their association with the progression and metastasis of malignancies. They found that POTE proteins correlated well with malignant progression and metastasis in a variety of tissues and that POTEG and /or POTEH are pivotal for cancer cells to grow and survive [20]. Liu et al. found that transient expression of POTEE or POTEF induces apoptosis in Hela cells. These findings indicate that the POTE gene family encodes a family of pro-apoptotic proteins [21]. Yi-Hsiang Huang et al. determined peptide modification can enhance the immunogenicity of POTE epitopes to induce T cells that kill human cancer cells, this result suggest that POTE may also be a molecular target for cancer immunotherapy [22].
In the last few years the researches for CT antigen have made some progress. The unique expression pattern of CT antigen makes them become candidates for use in early diagnosis and as a prognostic marker. The CT antigen MAGE has been found in 85% of NSCLC cases, 75% of early multiple myeloma cases, and 35% early breast cancer cases [23]. The CT antigen NY-ESO-1 has also been identified in 32% of early NSCLC cases, 29% of esophageal cancer cases, and 23% of early transitional cell carcinoma case [23]. These CT antigens can be used as biomarkers for early diagnosis of tumors. Importantly, MAGE can be detected in the peripheral blood of hepotocellular carcinoma patients, and their expression levels are closely related to metastasis status and prognosis in hepatocellular carcinoma patients [24–26]. In addition, MAGE, BAGE and GAGE CT antigen can be detected in the ascites of patients with ovarian cancer and in the cerebrospinal fluid of patients with brain metastases from melanoma. Thus, CT antigens can be used as biomarkers of tumor metastasis and prognosis [27, 28]. Because of their pattern of expression, CT antigens are very attractive candidates for immune-based therapies of cancer, and many clinical trials are testing their use in cancer patients [9, 29, 30].
In the present study, there had some limits. Our study cohort size was small; to establish the clinical utility of POTEE in serum, further studies should be performed with larger cohorts. In order to improve the specificity of POTEE ROC curve, we consider the following several aspect work needs to be done: (1) to enlarge the sample size to further optimize the cutoff value and confirm our results; (2) further, the perspective study should be performed; (3) to try the available combination of serum tumor biomarkers. In addition, we also need to investigate the functional mechanisms of POTEE in NSCLC.
In conclusion, our results show that POTEE is over-expressed in NSCLC and that POTEE level in serum is associated with TNM stage and prognosis in NSCLC patients.
Supporting Information
(DOC)
Acknowledgments
We thank our volunteers for donating their blood and our collaborators for collecting blood samples and participant data.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This study was supported by grants from the National Natural Science Foundation of China (no. 81401890 and no. 81372392) and the Key Project of the Science and Technology Commission of Shanghai Municipality (no. 11JC1411300). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Chen Z, Wang T, Cai L, Su C, Zhong B, Lei Y, et al. Clinicopathological significance of non-small cell lung cancer with high prevalence of Oct-4 tumor cells. Journal of experimental & clinical cancer research: CR. 2012;31: 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Goldstraw P, Ball D, Jett JR, Le Chevalier T, Lim E, Nicholson AG, et al. Non-small-cell lung cancer. Lancet. 2011;378: 1727–1740. 10.1016/S0140-6736(10)62101-0 [DOI] [PubMed] [Google Scholar]
- 3. Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clinic proceedings. 2008;83: 584–594. 10.4065/83.5.584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA: a cancer journal for clinicians. 2014;64: 9–29. 10.3322/caac.21208 [DOI] [PubMed] [Google Scholar]
- 5. Kalia M. Biomarkers for personalized oncology: recent advances and future challenges. Metabolism: clinical and experimental. 2015;64: S16–21. 10.1016/j.metabol.2014.10.027 [DOI] [PubMed] [Google Scholar]
- 6. Tufman A, Huber RM. Biological markers in lung cancer: A clinician's perspective. Cancer biomarkers: section A of Disease markers. 2010;6: 123–135. 10.3233/CBM-2009-0124 [DOI] [PubMed] [Google Scholar]
- 7. Ghafouri-Fard S, Modarressi MH. Cancer-testis antigens: potential targets for cancer immunotherapy. Archives of Iranian medicine. 2009;12: 395–404. [PubMed] [Google Scholar]
- 8. Caballero OL, Chen YT. Cancer/testis (CT) antigens: potential targets for immunotherapy. Cancer science. 2009;100: 2014–2021. 10.1111/j.1349-7006.2009.01303.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Scanlan MJ, Gure AO, Jungbluth AA, Old LJ, Chen YT. Cancer/testis antigens: an expanding family of targets for cancer immunotherapy. Immunological reviews. 2002;188: 22–32. [DOI] [PubMed] [Google Scholar]
- 10. Bera TK, Zimonjic DB, Popescu NC, Sathyanarayana BK, Kumar V, Lee B, et al. POTE, a highly homologous gene family located on numerous chromosomes and expressed in prostate, ovary, testis, placenta, and prostate cancer. Proceedings of the National Academy of Sciences of the United States of America. 2002;99: 16975–16980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bera TK, Huynh N, Maeda H, Sathyanarayana BK, Lee B, Pastan I. Five POTE paralogs and their splice variants are expressed in human prostate and encode proteins of different lengths. Gene. 2004;337: 45–53. [DOI] [PubMed] [Google Scholar]
- 12. Bera TK, Saint Fleur A, Lee Y, Kydd A, Hahn Y, Popescu NC, et al. POTE paralogs are induced and differentially expressed in many cancers. Cancer research. 2006;66: 52–56. [DOI] [PubMed] [Google Scholar]
- 13. Das S, Ise T, Nagata S, Maeda H, Bera TK, Pastan I. Palmitoylation of POTE family proteins for plasma membrane targeting. Biochemical and biophysical research communications. 2007;363: 751–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Bera TK, Walker DA, Sherins RJ, Pastan I. POTE protein, a cancer-testis antigen, is highly expressed in spermatids in human testis and is associated with apoptotic cells. Biochemical and biophysical research communications. 2012;417: 1271–1274. 10.1016/j.bbrc.2011.12.125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kuang P, Zhou C, Li X, Ren S, Li B, Wang Y, et al. Proteomics-based identification of secreted protein dihydrodiol dehydrogenase 2 as a potential biomarker for predicting cisplatin efficacy in advanced NSCLC patients. Lung cancer. 2012;77: 427–432. 10.1016/j.lungcan.2012.03.016 [DOI] [PubMed] [Google Scholar]
- 16. Hahn Y, Bera TK, Pastan IH, Lee B. Duplication and extensive remodeling shaped POTE family genes encoding proteins containing ankyrin repeat and coiled coil domains. Gene. 2006;366: 238–245. [DOI] [PubMed] [Google Scholar]
- 17. Lee Y, Ise T, Ha D, Saint Fleur A, Hahn Y, Liu XF, et al. Evolution and expression of chimeric POTE-actin genes in the human genome. Proceedings of the National Academy of Sciences of the United States of America. 2006;103: 17885–17890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Liu X, Tang H, Zhang Z, Li W, Wang Z, Zheng Y, et al. POTEH hypomethylation, a new epigenetic biomarker for glioma prognosis. Brain research. 2011;1391: 125–131. 10.1016/j.brainres.2011.03.042 [DOI] [PubMed] [Google Scholar]
- 19. Bera TK, Saint Fleur A, Ha D, Yamada M, Lee Y, Lee B, et al. Selective POTE paralogs on chromosome 2 are expressed in human embryonic stem cells. Stem cells and development. 2008;17: 325–332. 10.1089/scd.2007.0079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Redfield SM, Mao J, Zhu H, He Z, Zhang X, Bigler SA, et al. The C-terminal common to group 3 POTES (CtG3P): a newly discovered nucleolar marker associated with malignant progression and metastasis. American journal of cancer research. 2013;3: 278–289. [PMC free article] [PubMed] [Google Scholar]
- 21. Liu XF, Bera TK, Liu LJ, Pastan I. A primate-specific POTE-actin fusion protein plays a role in apoptosis. Apoptosis: an international journal on programmed cell death. 2009;14: 1237–1244. 10.1007/s10495-009-0392-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Huang YH, Terabe M, Pendleton CD, Stewart Khursigara D, Bera TK, Pastan I, et al. Identification and enhancement of HLA-A2.1-restricted CTL epitopes in a new human cancer antigen-POTE. PloS one. 2013;8: e64365 10.1371/journal.pone.0064365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Suri A. Cancer testis antigens—their importance in immunotherapy and in the early detection of cancer. Expert opinion on biological therapy. 2006;6: 379–389. [DOI] [PubMed] [Google Scholar]
- 24. Mou DC, Cai SL, Peng JR, Wang Y, Chen HS, Pang XW, et al. Evaluation of MAGE-1 and MAGE-3 as tumour-specific markers to detect blood dissemination of hepatocellular carcinoma cells. British journal of cancer. 2002;86: 110–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hoon DS, Kuo CT, Wascher RA, Fournier P, Wang HJ, O'Day SJ. Molecular detection of metastatic melanoma cells in cerebrospinal fluid in melanoma patients. The Journal of investigative dermatology. 2001;117: 375–378. [DOI] [PubMed] [Google Scholar]
- 26. Hofmann M. Ruschenburg I mRNA detection of tumor-rejection genes BAGE, GAGE, and MAGE in peritoneal fluid from patients with ovarian carcinoma as a potential diagnostic tool. Cancer. 2002;96: 187–193. [DOI] [PubMed] [Google Scholar]
- 27. Aruga A, Takeshita N, Kotera Y, Okuyama R, Matsushita N, Ohta T, et al. Phase I clinical trial of multiple-peptide vaccination for patients with advanced biliary tract cancer. Journal of translational medicine. 2014;12: 61 10.1186/1479-5876-12-61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Obara W, Ohsawa R, Kanehira M, Takata R, Tsunoda T, Yoshida K, et al. Cancer peptide vaccine therapy developed from oncoantigens identified through genome-wide expression profile analysis for bladder cancer. Japanese journal of clinical oncology. 2012;42: 591–600. 10.1093/jjco/hys069 [DOI] [PubMed] [Google Scholar]
- 29. Rosenberg SA. A new era of cancer immunotherapy: converting theory to performance. CA: a cancer journal for clinicians. 1999;49: 70–73, 65 [DOI] [PubMed] [Google Scholar]
- 30. Old LJ. Cancer vaccines: an overview. Cancer immunity. 2008;8 Suppl 1: 1 [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.