Abstract
Campylobacter jejuni is an important pathogen of foodborne illness. Transmigration across the intestinal epithelial barrier and invasion are considered as primary reasons for tissue damage triggered by C. jejuni. Using knockout mutants, it was shown that the serine protease HtrA may be important for stress tolerance and physiology of C. jejuni. HtrA is also secreted in the extracellular environment, where it can cleave junctional host cell proteins such as E-cadherin. Aim of the present study was to establish a genetic complementation system in two C. jejuni strains in order to introduce the wild-type htrA gene in trans, test known htrA phenotypes, and provide the basis to perform further mutagenesis. We confirm that reexpression of the htrA wild-type gene in ΔhtrA mutants restored the following phenotypes: 1) C. jejuni growth at high temperature (44 °C), 2) growth under high oxygen stress conditions, 3) expression of proteolytically active HtrA oligomers, 4) secretion of HtrA into the supernatant, 5) cell attachment and invasion, and 6) transmigration across polarized epithelial cells. These results establish a genetic complementation system for htrA in C. jejuni, exclude polar effects in the ΔhtrA mutants, confirm important HtrA properties, and permit the discovery and dissection of new functions.
Keywords: cellular invasion, chaperone, E-cadherin, flagellum, HtrA, molecular pathogenesis, paracellular, secretion, signaling, stress response, transwell, virulence
Introduction
Foodborne and waterborne diseases are responsible for very high rates of morbidity and mortality in humans worldwide. The World Health Organization reported that these diseases kill an estimated number of 2.2 million people annually, most of whom are children [1]. The Gram-negative pathogen Campylobacter jejuni is the most common cause of bacterial foodborne infections in developed countries [2, 3]. C. jejuni colonizes the intestinal tract of a wide range of wild and domestic birds as well as agriculturally important mammals. Thus, handling and consumption of contaminated poultry meat products or raw milk have been established as the most frequent sources of campylobacteriosis in humans [4]. An infectious dose of a few hundred bacteria is sufficient to establish colonization in the human host. The clinical symptoms of Campylobacter infections include diarrhea, abdominal pain, and fever, which in rare cases can lead to the development of more serious complications such as reactive arthritis and Guillain–Barrè and Miller–Fisher syndromes [5, 6]. In contrast to the disease-causing infections in humans, no apparent symptoms are found in colonized poultry or other avian species, indicating a well-adapted commensalism in the avian cecum [2, 7]. In line with these observations, C. jejuni exhibits a microaerophilic lifestyle with optimal growth temperature at 37–42 °C.
During transmission, C. jejuni is exposed to multiple stress conditions, such as osmotic stress, pH changes, atmospheric oxygen exposure, and temperature fluctuation, requiring a resilient stress response system [7–9]. C. jejuni is a typical facultative intracellular pathogen due to its capabilities to adhere to and invade into human gut epithelial cells [2]. However, a main difference between C. jejuni infection of humans and poultry is the apparently elevated number of bacteria entering epithelial cells in the human gut [2]. This led to the common view that C. jejuni adhesion to and entrance of human intestinal epithelial cells may represent critical steps, which are essential for disease development [10].
The C. jejuni flagellar genes and motility are required for invasion of intestinal epithelial cells, and increased mucosal viscosity resulted in enhanced bacterial motility, adherence and invasion [11]. The C. jejuni flagellum also serves as a type-III secretion system for the extracellular release of factors that may control bacteria–host interactions [12–15]. These secreted effector proteins include the so-called Campylobacter invasion antigens (CiaA-H) and flagellar coexpressed determinants (FedA-D) [13, 16–18]. Moreover, C. jejuni binding to host target cells has been reported to include a series of outer membrane adhesins such as PEB1, MOMP, JlpA, CadF, and FlpA [10, 16, 19–21]. In addition, the chaperone domain of serine protease HtrA (high temperature requirement A) contributes to cell binding and invasion, probably by assisting in properly folding of one or more of the above adhesins [22, 23]. A prerequisite of C. jejuni host cell invasion is the induction of cytoskeletal rearrangements driven by the small Rho GTPases Cdc42 and Rac1 [24]. These GTPases are activated upon binding and have significant impact during the course of invasion [16, 24]. The signaling cascades stimulating Cdc42 and Rac1 are complex and require CadF, certain receptors (fibronectin, integrin-β1, PDGF- and EGF-receptor tyrosine kinases), cytosolic kinases (Src, FAK, and PI3-kinase), and guanine exchange factors (Tiam-1, Vav-2, and DOCK180), which mediate cytoskeletal remodelling in host cells followed by bacterial uptake [25, 26].
Recent studies have demonstrated that HtrA is a novel secreted virulence factor of C. jejuni [22, 23, 26]. HtrA proteins are widely conserved in many bacteria and represent a group of heat shock induced serine proteases with supplementary chaperone function [27–30]. HtrA proteases commonly consist of a signal peptide, a trypsin-like serine protease module, and 1–2 PDZ domains to facilitate protein–protein interactions [31]. HtrA proteins have significant impact on the survival and virulence properties of multiple pathogens [28, 30, 31]. For example, as compared to wild-type (wt) C. jejuni, it was shown that growth of ΔhtrA mutants is severely diminished at 44 °C and tolerance of the ΔhtrA mutant against oxygen stress is also significantly reduced [22]. These proteases have been commonly found to act in the periplasm, where they assemble proteolytic active oligomers with crucial function in controlling protein quality [32, 33]. For a long time, it was believed that HtrA family members are strictly operating only intracellularly within the bacteria. However, we have recently unraveled a new feature of HtrA during infection. In C. jejuni and its close relative Helicobacter pylori, HtrA proteins are actively secreted in the extracellular space, where they can hijack host proteins [26, 34, 35]. Infection experiments in vitro indicated that HtrA can open the cell-to-cell junctions in cell monolayers by cleaving-off the extracellular domain of the surface adhesion protein E-cadherin followed by paracellular transmigration of the bacteria [26, 35]. Deletion of the htrA gene leads to a defect in E-cadherin shedding and impaired transmigration of C. jejuni across monolayers of polarized epithelial cells in vitro [26]. An insect infection model has shown that the outcome of a C. jejuni infection is also affected by HtrA, because fewer Galleria mellonella larvae are killed by an htrA mutant as compared to the wt strain [36]. In addition, using C. jejuni ΔhtrA deletion mutants, we have shown that HtrA plays a pivotal role in inducing host cell apoptosis and immunopathology in the gut of mice [37, 38]. Goal of the present study was to establish a genetic complementation system in C. jejuni to confirm the importance of HtrA in heat tolerance, oxygen stress resistance, epithelial cell adhesion, invasion, and transmigration of the bacteria.
Results
Complementation confirms the importance of HtrA in heat tolerance and oxygen stress resistance by C. jejuni
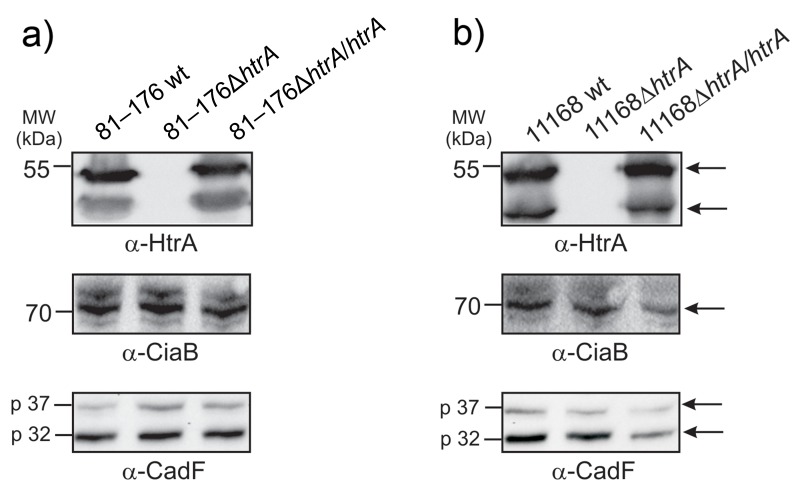
We have generated several ΔhtrA deletion mutants in the well-known C. jejuni strains 81–176 and NCTC11168 [29, 39]. Next aim was to develop a fast and reliable genetic complementation system of the wt htrA gene for further investigation of the importance of this factor both during conventional growth and infection processes. For this purpose, the htrA gene of C. jejuni NCTC11168 including its own promoter was ligated to a kanamycin resistance gene, and this cassette was inserted in the pseudogene downstream of Cj0208, a region which is present in both strains. Correct integration of htrA in the C. jejuni chromosomes of 81–176 and NCTC11168 was confirmed by PCR and standard sequencing (data not shown). Reexpression of HtrA proteins was verified by Western blotting (Fig. 1a/b). The α-CiaB and α-CadF blots served as loading controls and confirmed that equal amounts of protein were present in each lane.
Fig. 1.
Western blotting confirms genetic complementation of wild-type (wt) htrA gene in C. jejuni ΔhtrA deletion mutants. The wt htrA gene with its own promoter was complemented in the ΔhtrA deletion mutants of C. jejuni strains 81–176 (a) and NCTC11168 (b). Bands show presence of HtrA protein expression. As loading controls, the α-CiaB and α-CadF blots confirmed that equal amounts of protein were present in each sample
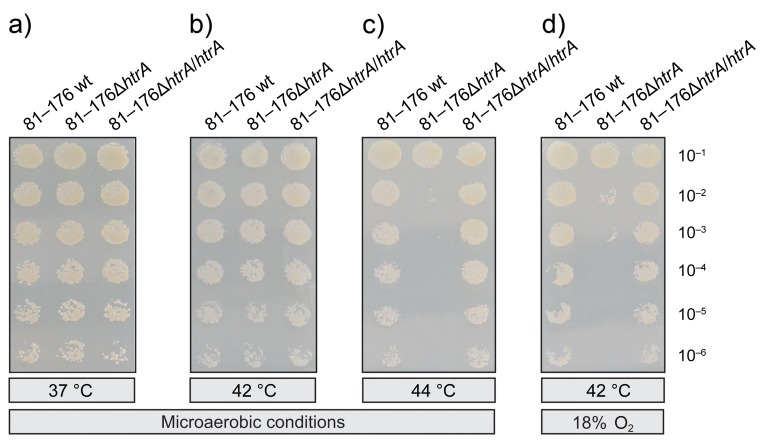
Next, we investigated the growth of C. jejuni strains on Mueller Hinton (MH) agar plates at 37 °C, 42 °C, and 44 °C under microaerobic conditions (~5% O2) or at 42 °C in the presence of ~18% O2 (Fig. 2). All strains were growing equally well at 37 °C and 42 °C under microaerobic conditions, but growth of the 81–176ΔhtrA mutant was severely impaired at 44 °C and tolerance of the mutant to an atmosphere containing 18% O2 was strongly reduced as compared to the isogenic wt strain (Fig. 2a–d). Genetic complementation of the wt htrA gene in ΔhtrA mutant strain restored C. jejuni 81–176 growth in the presence of 5% O2 at 44 °C (Fig. 2c) and growth in the presence of 18% O2 at 42 °C (Fig. 2d). The same experiments were performed with wt, ΔhtrA mutant, and complemented htrA in strain NCTC11168 with similar results compared to strain 81–176 (data not shown). These experiments demonstrate that C. jejuni HtrA plays a crucial role in heat tolerance and oxygen stress resistance of these bacteria.
Fig. 2.
Genetic complementation confirms the importance of HtrA in heat tolerance and oxygen stress resistance by C. jejuni. Serial dilutions of C. jejuni strains [81–176 wild-type (wt), 81–176ΔhtrA, and complemented 81–176ΔhtrA/htrA] were spotted onto MH agar plates as indicated. The plates were incubated for 3 days in jars under microaerobic conditions at 37 °C (a), 42 °C (b), and 44 °C (c) or at 42 °C in the presence of 17–18% O2 (d). Representative sections of the agar plates from three independent experiments are presented. The results show that reintroduction of wt htrA gene in the ΔhtrA mutant strain restored C. jejuni growth under the indicated conditions. Experiments were done in triplicates
Secretion of HtrA in the extracellular space is restored by the complemented C. jejuni strains
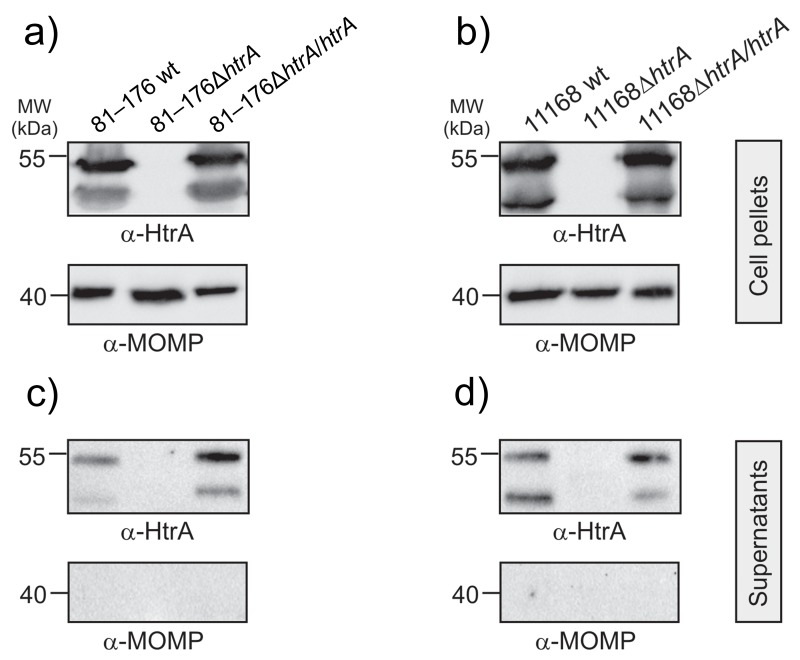
To test if HtrA is secreted by the complemented C. jejuni variants, all above described strains were grown for 12 h in brain heart infusion (BHI) liquid broth medium containing 10% fetal calf serum (FCS). Bacterial supernatants and cell pellets were prepared, and the presence of secreted HtrA proteins in the supernatant was investigated by immunoblotting using α-HtrA antibodies (Fig. 3a–d). The results show that all wt C. jejuni and htrA complemented strains exhibit similarly strong HtrA signals in the supernatant fraction while the corresponding ΔhtrA deletion mutants did not (Fig. 3b/d). As control, the bacterial pellets and supernatants were probed with α-MOMP antibodies. The α-MOMP blots of bacterial cell pellets show strong bands of similar intensity as expected, confirming that equal amounts of protein were present in each sample (Fig. 3a/c), while the supernatants are devoid of MOMP, indicating absence of lysed bacteria and cell debris (Fig. 3b/d). Taken together, these experiments indicate that the complemented strains restored their ability to deliver HtrA proteins in the extracellular environment.
Fig. 3.
Western blotting showing presence of HtrA protein in bacterial pellets (top) and culture supernatants (bottom). Wild-type (wt), isogenic ΔhtrA deletion mutants, and complemented ΔhtrA/htrA strains of C. jejuni 81–176 (a, b) and NCTC11168 (c, d) were grown in BHI medium with 10% FCS for 12 h at 37 °C, and then fractionated. Equal amounts of protein in each bacterial pellet and absence of bacterial lysis in the supernatants were confirmed by probing with α-MOMP antibody
HtrA proteins of the complemented C. jejuni strains form proteolytically active oligomers
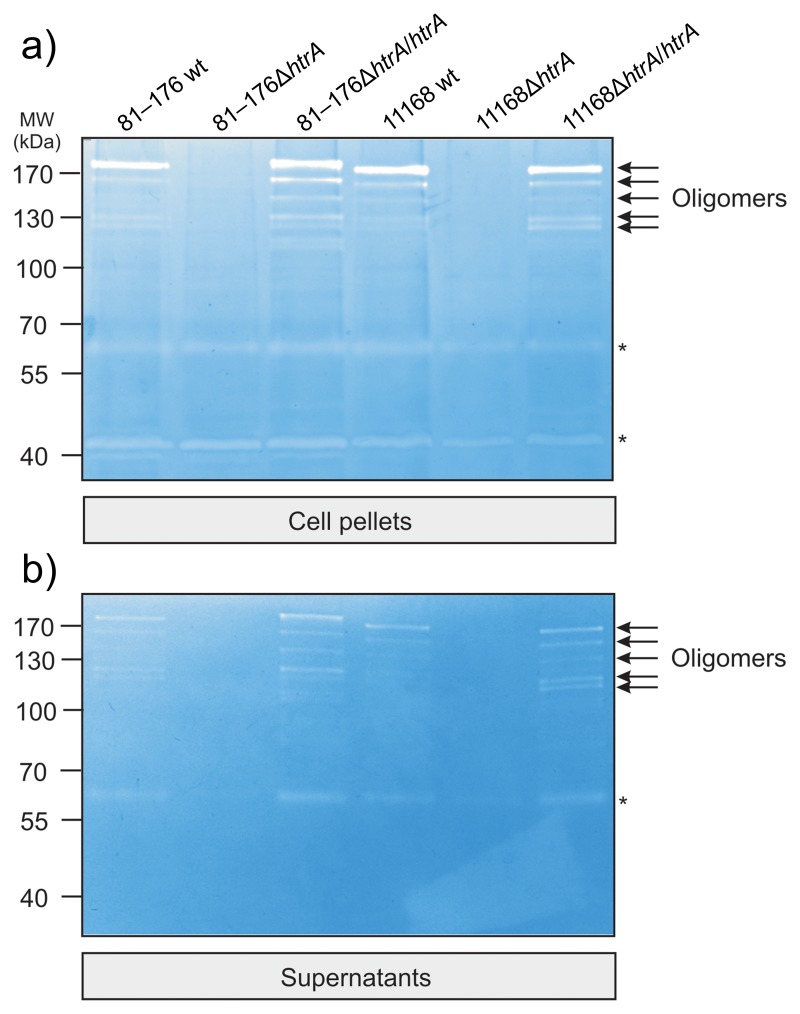
Next, we aimed to analyze if complemented HtrA can form proteolytically active oligomers. For this purpose, the fractionated samples generated for Fig. 3 were subjected to casein zymography. Bacterial pellets and sterile filtered culture supernatants were loaded onto a 0.1% casein containing gel and separated under nondenaturating conditions [35]. The results show that HtrA from both wt C. jejuni and complemented strains gave rise to active HtrA oligomers with a molecular weight ranging from ~120 kDa to more than 200 kDa in the pellet (Fig. 4a, arrows) and supernatant (Fig. 4b, arrows). As control, corresponding signals for proteolytically active HtrA were completely absent in both ΔhtrA deletion mutants as expected (Fig. 4a/b).
Fig. 4.
Secreted and cell-associated HtrA of the complemented C. jejuni strains generate proteolytically active oligomers. Wild-type (wt), isogenic ΔhtrA deletion mutants, and complemented ΔhtrA/htrA strains of C. jejuni 81–176 and NCTC11168 were grown in BHI medium with 10% FCS for 12 h at 37 °C. Bacterial pellets (a) and culture supernatants (b) were prepared and subjected to investigation of protease activity by casein zymography. The position of proteolytically active oligomeric HtrA proteins is indicated with arrows. Asterisks label the position of two other proteolytically active protein bands at ~65 kDa and ~42 kDa, respectively
Complementation of htrA restores cell adhesion and invasion properties of C. jejuni
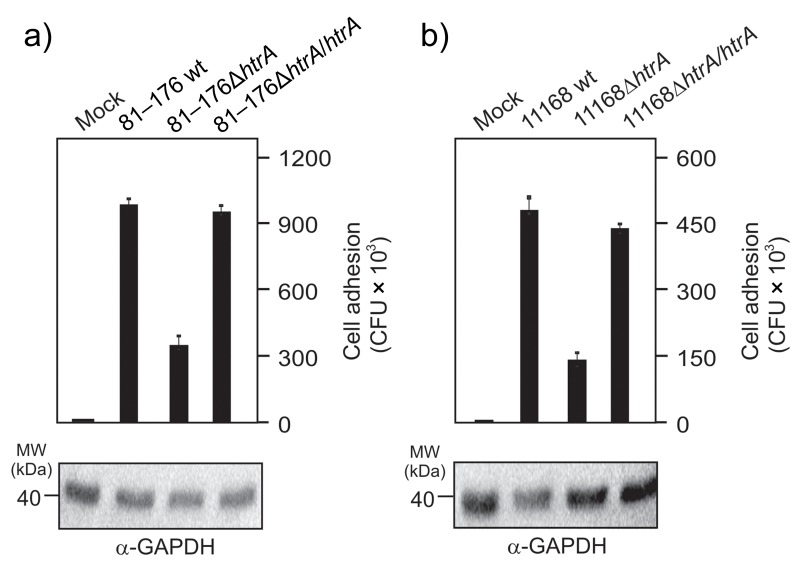
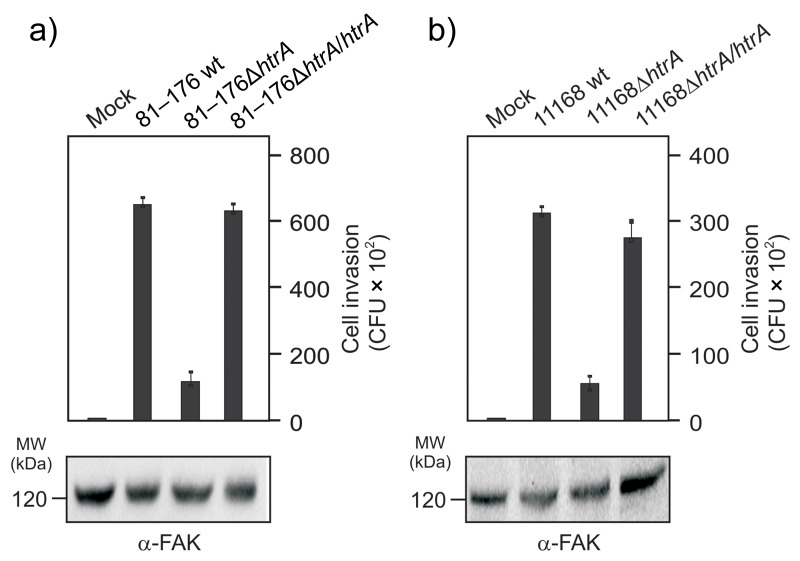
Recent work has shown that the chaperone domain of HtrA contributes to binding and invasion of epithelial cells by C. jejuni NCTC11168 [23]. We therefore aimed to test if our htrA complemented strains can restore this phenotype. For this purpose, nonpolarized INT-407 intestinal epithelial cells were infected for 6 h with the above strains, followed by gentamicin protection assay and determination of the colony forming units (CFU). The results show that both C. jejuni wt and htrA complemented strains are able to adhere to INT-407 cells with similar extent, while both ΔhtrA deletion mutants exhibit a strong reduction in the number of cell-associated bacteria (Fig. 5a/b). In control experiments, 250 μg/mL gentamicin killed all extracellular bacteria (data not shown). In agreement with the cell binding properties of described strains, we found that both wt C. jejuni and htrA complemented strains are able to invade INT-407 cells, while the two ΔhtrA deletion mutants show strong deficiency for invasion (Fig. 6a/b). These results further support the view that HtrA is involved in C. jejuni adhesion to and invasion of intestinal epithelial cells.
Fig. 5.
Genetic complementation of htrA restores binding of C. jejuni to human intestinal epithelial cells. INT-407 cells infected for 6 h with wild-type (wt), isogenic ΔhtrA deletion mutants, and complemented ΔhtrA/htrA strains of C. jejuni 81–176 (a) or NCTC11168 (b). Cell adhesion of the C. jejuni strains was analyzed by gentamicin protection assay. The α-GAPDH blot served as loading control and confirmed that equal amounts of protein are present in each sample. Bars represent averages and standard deviations of three independent experiments
Fig. 6.
Genetic complementation of htrA restores invasive properties of C. jejuni in human intestinal epithelial cells. INT-407 cells were infected for 6 h with wild-type (wt), isogenic ΔhtrA deletion mutants, and complemented ΔhtrA/htrA strains of C. jejuni 81–176 (a) or NCTC11168 (b). Intracellular C. jejuni were quantified by gentamicin protection assay. The α-FAK blot served as loading control and confirmed that equal amounts of protein are present in each sample. Bars represent averages and standard deviations of three independent experiments
Transmigration of C. jejuni across polarized epithelial cells is restored by genetic complementation of HtrA
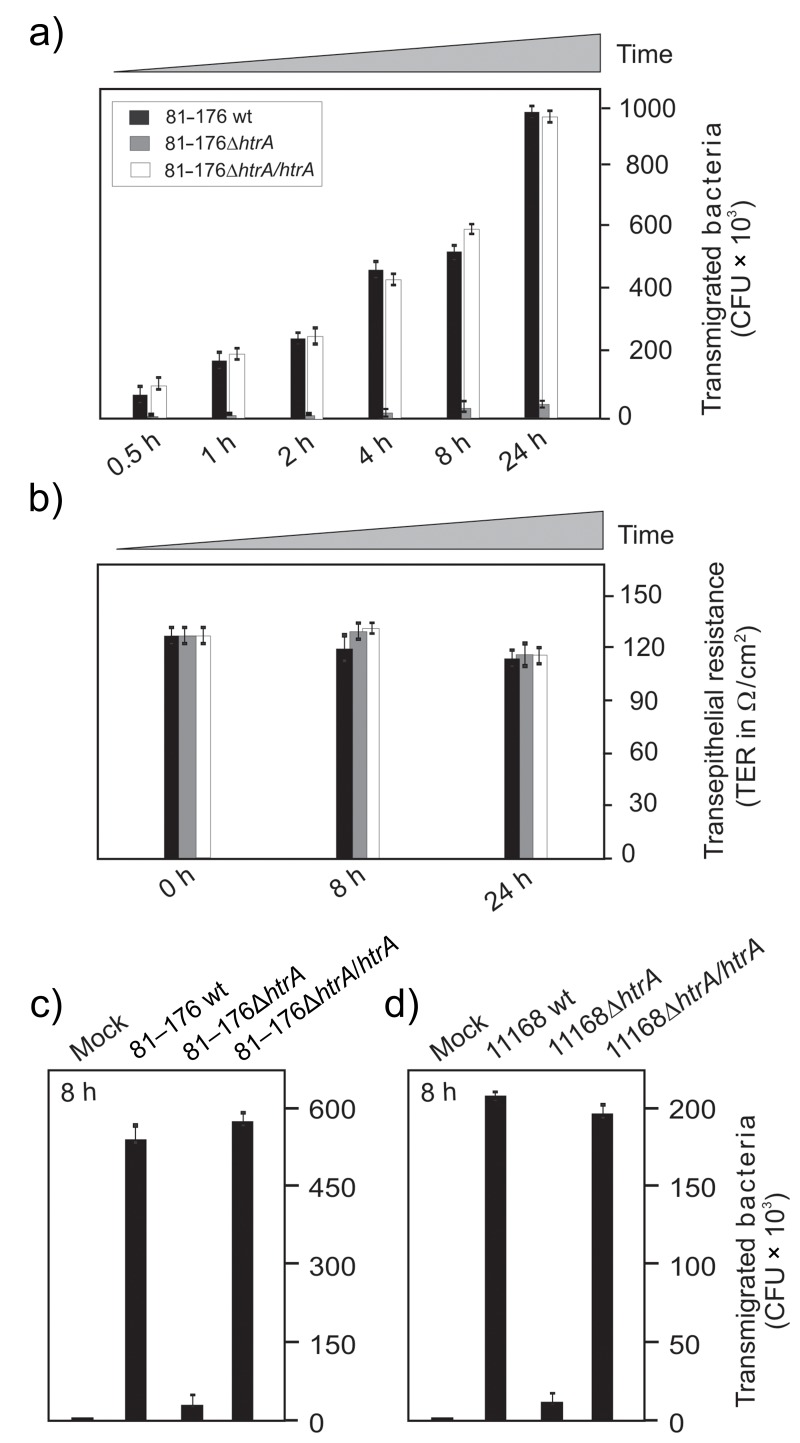
Finally, we aimed to analyze if the complemented C. jejuni strains can restore the transmigration defect of ΔhtrA mutants across polarized epithelial cells reported recently [26, 39]. To test this important function of HtrA, polarized MKN-28 cells were seeded and differentiated over 14 days in a transwell filter system. The transepithelial electrical resistance (TER) was determined over time and reached values between 140 and 150 Ω/cm2 in the confluent monolayer, similar to previously reported data [40]. MKN-28 cells were then infected with the C. jejuni strains, followed by determination of CFU in the bottom chambers during the time course of 0.5 to 24 h. The results indicate that C. jejuni 81–176 wt and the htrA complemented strains transmigrated quickly during the first 30 min and increased up to 900,000 CFU over time, while the isogenic ΔhtrA deletion mutant did not (Fig. 7a). We also measured the TER values and did not detect significant differences before and after infection (Fig. 7b). Similar transmigration rates were obtained when we compared the various strains of 81–176 (Fig. 7c) and NTCT111638 (Fig. 7d) in parallel experiments. As control, nonpathogenic Escherichia coli Top-10 did not transmigrate under the same conditions as expected (data not shown). These observations further support the view that C. jejuni HtrA plays an important role during transmigration of these bacteria across polarized epithelial cells.
Fig. 7.
Transmigration of C. jejuni across polarized epithelial cells is restored by genetic complementation of htrA. Differentiated MKN-28 epithelial cells were grown in a transwell filter system for 14 days to reach a confluent monolayer. The cells were infected in the apical chamber. A time course of infection with wild-type (wt), isogenic ΔhtrA deletion mutants, and complemented ΔhtrA/htrA strains of C. jejuni 81–176 (a–c) or NCTC11168 (d) is shown. Transmigrated bacteria were harvested from the bottom chambers, grown on MH agar plates, and CFUs were determined in triplicates (a, c, d). The transepithelial resistance (TER) was measured before and after infection and did not change significantly during the indicated time course (b)
Discussion
HtrA is a conserved periplasmic protein that exhibits both protease and chaperone activities with important function in protein quality control in many bacteria [31–33]. In addition, it has been reported that HtrA is crucial for the virulence of numerous bacterial pathogens such as Yersinia enterocolitica [41], Salmonella enterica [42], Klebsiella pneumonia [43], Shigella flexneri [44], Listeria monocytogenes [45], and Chlamydia trachomatis [46]. It is well-known that HtrA is critical for stress tolerance and survival of most bacteria, because it degrades and prevents aggregation of periplasmic proteins that can misfold under stress conditions [31–33], but only a few reports have studied the discrete functions of HtrA protease and chaperone activities during infection [27–30].
Our previous reports suggested that HtrA of two gastrointestinal pathogens, C. jejuni and H. pylori, can be secreted into the cell culture supernatant, where HtrA plays a major role in crossing polarized epithelial monolayers by these bacteria in vitro [26, 34, 35]. In addition, using C. jejuni ΔhtrA deletion mutants, we have shown that HtrA plays a pivotal role in inducing host cell apoptosis and immunopathology during campylobacteriosis in the gut of two mouse models in vivo [37, 38]. However, knockout mutagenesis of individual genes in the bacteria can generate polar effects; thus, genetic complementation is necessary. In the present study, we complemented the wt htrA gene in two C. jejuni ΔhtrA deletion mutants in order to 1) confirm its important role in heat tolerance, oxygen stress resistance, epithelial cell adhesion, invasion, and transmigration of the bacteria and to 2) establish a tool for further mutagenesis of htrA across the entire gene.
During transmission, C. jejuni requires significant capabilities to adapt to and survive in various natural environments. In the present study, we first investigated the importance of HtrA in the stress tolerance of C. jejuni and demonstrate that it is required both for heat and oxygen tolerance, which may be important for successful survival of the bacteria during the contamination cycle. In agreement with previous studies, we show in the present work that HtrA is required for C. jejuni growth at high temperatures. Brøndsted and coworkers proposed that C. jejuni HtrA is a functional ortholog of HtrA in E. coli, which is involved in the degradation of nonnative proteins in the periplasm [22]. Thus, the temperature sensitivity of the C. jejuni ΔhtrA mutants could be a result of the accumulation of misfolded proteins in this subcellular compartment, similar to observations made in E. coli [31–33]. This conclusion is supported by the complementation of the temperature-sensitive phenotype of an E. coli htrA mutant with C. jejuni wt htrA [22]. Remarkably, it was found that deletion of htrA in C. jejuni leads to overexpression of two cytoplasmic chaperones, DnaK and ClpB [22], that are well-known factors involved in the refolding of heat-denatured proteins and solubilization of larger protein aggregates in E. coli by the expenditure of ATP [47, 48]. Questions arise as to whether the accumulation of misfolded proteins in the periplasm increases the level of cytoplasmic chaperones and how exactly the signal is transmitted from periplasm to cytoplasm, which should be investigated in future studies.
In contrast to the growth and survival of bacterial htrA mutants of other pathogenic Gram-negative bacteria [41, 49, 50], the C. jejuni ΔhtrA mutants showed reduced oxygen tolerance [22]. Even though C. jejuni is a microaerobic organism, it is able to perform aerobic respiration [51], and an increase in oxygen tension could lead to the accumulation of oxidatively damaged proteins in the periplasm that require HtrA for removal [22]. Our experiments confirmed these earlier observations and showed that this phenotype in the C. jejuni ΔhtrA mutant can be restored by wt htrA complementation.
In addition, we tested if the C. jejuni strains can generate proteolytic active HtrA oligomers. Using casein zymography, we could demonstrate that wt and htrA complemented C. jejuni formed caseinolytic active oligomers with expected sizes of trimers and higher. These observations are in agreement with reports on HtrA in other bacteria such as E. coli, where the HtrA oligomers are highly proteolytic active rather than the monomer [52]. These oligomers were found both in total cell lysates and culture supernatants of C. jejuni; however, it remained unknown how HtrA can cross the bacterial outer membrane. HtrAs in Gram-negative bacteria such as C. jejuni contain a signal peptide important for Sec-dependent cleavage and transport of the protease across the inner membrane into the periplasm [26, 34, 35]. Commonly, HtrAs exhibit no sequence homology to typical autotransporters, which process themselves by autoproteolysis. This is in line with our observation that the HtrA protease activity is not required for secretion [39]. Thus, HtrA very likely requires the assistance of other bacterial factors for delivery. Candidates are secretion systems called type I–VII (T1SS–T7SS), but various available full genome sequences of C. jejuni do not encode orthologs of classical T1SS–T7SS transporters [53–55]. For example, a putative conjugative T4SS has been only found on the pVir plasmid, but pVir is not present in the used strain NCTC11168, and is therefore not involved in HtrA secretion. A few putative T2SS components (encoded by cj1470 and cj1474c genes) have been noted in NCTC11168, which could be involved in protein secretion [53]. However, the genes cj1343c, cj1470c, cj1471c, and cj1474c were later described to be involved in natural competence and DNA import into C. jejuni [56]. Whether these genes may play a role in HtrA secretion remains to be elucidated. Forthcoming experiments should study in detail how HtrA is secreted by C. jejuni.
Even though HtrA has been described traditionally as a stress response protein, other findings indicate that HtrA has also specific functions during infection that may be stress-independent. Previous studies have shown that binding of C. jejuni to epithelial host cells is highly dependent on HtrA expression [22]. In addition, an htrA mutant was detected in a C. jejuni transposon library screened for reduced invasion capability [57]. Both studies concluded that particularly the chaperone activity of HtrA may have a significant impact on the interaction of C. jejuni and host cells. Lack of HtrA reduced bacterial binding to epithelial cells 5–10 times more [22], as compared to any other known adhesin [16, 25, 58–60], suggesting a pleiotropic effect. Impaired adherence and invasion of the htrA mutant cannot be explained by reduced C. jejuni motility, since the htrA mutation does not affect these parameters [26]. Instead, Brøndsted and coworkers suggested that the reduced adherence of the ΔhtrA mutants to epithelial cells can be caused by misfolding or improper localization of one or more adhesion factors [22]. One such option is the CadF adhesin for the basolateral receptor fibronectin, which could explain why apical colonization is similarly high between ΔhtrA mutants and wt C. jejuni in the mouse gut in vivo [37, 38]. Alternatively, HtrA may influence other processes in the periplasm such as N-linked glycosylation of C. jejuni surface proteins like PEB3 and thereby could impact bacterial adherence to epithelial cells [61]. These various options should be investigated in future.
Finally, deletion of the htrA gene in two strains resulted in strong deficiency of C. jejuni to travel across polarized MKN-28 epithelial cells cultivated in a transwell chamber [26, 35]. We demonstrate here that genetic complementation of htrA can also restore the deficiency of ΔhtrA mutants to transmigrate across polarized epithelial cells. Thus, HtrA is a pivotal factor that may allow C. jejuni to cross the intestinal barrier in order to access deeper tissues in vivo. In fact, live C. jejuni were recovered from other organs in infected animals such as the spleen [62–64], liver [63–65], mesenteric lymph nodes [63], and blood [66]. This suggests that C. jejuni exhibits the capability not only to adhere to and enter into enterocytes, but can also travel within the host, pass the intestinal epithelial barrier, enter the lamina propria, and even access other organs of various infected hosts. Thus, the contribution of HtrA in these processes should be studied in more detail.
Taken together, our results demonstrate that various described phenotypic defects of C. jejuni ΔhtrA mutants can be restored by genetic complementation of the wt htrA gene. This suggests that we have established a reliable genetic system serving as a tool for further mutagenesis of htrA across the entire gene and more in-depth functional studies.
Materials and Methods
E. coli and C. jejuni strains
The E. coli strain Top-10 was purchased from Invitrogen (Darmstadt/Germany) and grown in conventional LB broth medium (10 g/l tryptone, 5 g/l yeast extract, 10 g/l NaCl). The two C. jejuni wt isolates 81–176 and NCTC11168 were used because their genome sequences are available. The corresponding isogenic knockout mutants 11168ΔhtrA and 81–176ΔhtrA were previously described [22, 23, 26]. The C. jejuni strains were commonly cultured at 37 °C for 48 h on Campylobacter blood-free selective agar base containing selective supplement or on MH agar plates (Oxoid, Wesel/Germany). In case of the mutant C. jejuni strains, all agar plates were supplemented with 30 µg/ml of kanamycin or 30 µg/ml of chloramphenicol, respectively [67]. C. jejuni was grown under microaerobic conditions generated by CampyGen packs in 3.5 l anaerobic jars of Oxoid [68].
Genetic complementation of the htrA gene in C. jejuni
The plasmid pCam-148 was kindly provided by Dr. Dennis Linton (University Manchester/UK) and used for genetic complementation of the htrA gene in the C. jejuni chromosome. In brief, pCam-148 harbors a 2178-bp fragment of C. jejuni NCTC11168 chromosomal DNA (position 205,297 to 207,475 containing a pseudogene downstream of Cj0208) cloned into the SmaI restriction site of plasmid pUC18. pCam-148 contains a unique SpeI restriction site in the C. jejuni sequence. This SpeI site was used to introduce three additional restriction sites (NotI, MluI, and NruI) by the primer annealing approach. Afterwards, a 1694-bp fragment of the htrA gene of C. jejuni NCTC11168 was amplified by PCR, including the 200-bp upstream and 75-bp downstream sequences (with promotor and terminator). The resulting PCR product (with flanking NotI and MluI sites, respectively) was introduced into the pSB-249 vector, and then further subcloned into the NotI and MluI sites of pCam-148. As a selection marker, we used the 795-bp aph gene with its own promoter permitting kanamycin resistance (from plasmid pRYSK12, kindly provided by Dr. Sabine Kienesberger, University Graz/Austria). This kanamycin resistance cassette was cloned into the MluI and NruI sites of pCam-148 adjacent to the htrA gene, resulting in the final htrA complementation construct. This plasmid was then transformed into the C. jejuni NCTC11168ΔhtrA and 81–176ΔhtrA deletion mutants, respectively (named NCTC11168ΔhtrA/htrA or 81–176ΔhtrA/htrA). Correct integration of htrA in the C. jejuni chromosomes was approved by PCR, and expression of HtrA proteins was confirmed by Western blotting.
Growth of C. jejuni under stress conditions on MH agar plates
Wt C. jejuni, ΔhtrA mutants, and corresponding ΔhtrA/htrA complemented strains were grown for 48 h on MH agar plates at 37 °C under microaerobic conditions as described above. All bacterial strains were suspended in BHI medium. The optical density was determined by measuring the suspension at λ = 600 nm (OD600) in a UV/Vis spectrometer Lambda 2 (Perkin Elmer, Waltham/USA) and adjusted to 0.1. Serial dilutions (10−1 through 10−6) were prepared, and 10 μL volumes were spotted onto MH agar plates. These plates were incubated for 3 days either under microaerobic conditions at 37 °C, 42 °C or 44 °C, or at 42 °C in atmosphere containing ~18% O2 produced by the candle jar procedure as described [22]. Experiments were repeated at least three times.
HtrA secretion assays
Wt C. jejuni and mutant strains were suspended in BHI medium supplemented with 10% FCS. The optical density was determined and adjusted to OD600 = 0.2. To allow HtrA secretion in the culture supernatant, the bacteria were incubated for 12 h shaking at 160 rpm. The cell pellets and the supernatants were separated by centrifugation at 4000 rpm. The supernatants were then passed through 0.21 μm sterile filters (Sigma-Aldrich, Taufkirchen/Germany) to remove remnant bacterial cells. Absence of live bacteria in the supernatant was verified by the lack of CFU after 3 days of incubation on MH agar plates. The obtained bacterial pellets and supernatants were then analyzed by Western blotting and casein zymography as described below.
Infection of INT-407 cells
INT-407 human embryonic intestinal epithelial cells, a typical nonpolarized cell line, was obtained from the American Type Culture Collection (ATCC CCL-6). The cells were grown at 37 °C in 5% CO2 using six-well plates with MEM medium containing Earle’s salts and l-Glutamine (Gibco, Darmstadt/Germany) and Pen/Strep antibiotics cocktail (Invitrogen). After reaching confluency of ~70%, cells were washed twice with Dulbecco’s phosphate buffered saline (DPBS, Sigma-Aldrich) and replaced by fresh antibiotics-free medium 12 h before infection. For infection, C. jejuni was harvested from MH agar plates, suspended in BHI medium, and the number of bacteria was determined through optical density measurement at OD600. The INT-407 cells were infected using a multiplicity of infection (MOI) of 100.
Gentamicin protection assay
After infection, the INT-407 cells were washed three times with 1 ml of prewarmed MEM medium per well to remove nonbound C. jejuni. To determine the CFU corresponding to intracellular bacteria, the cells were treated for 2 h with 250 μg/ml gentamicin (Sigma-Aldrich), washed three times with medium, and then incubated with 1 ml of 0.1% (w/v) saponin (Sigma-Aldrich) in DPBS at 37 °C for 15 min to lyse the cells. Afterwards, the treated cells were suspended thoroughly, diluted, and plated on MH agar plates. To determine the total CFU of cell-associated bacteria, the infected cell monolayers were incubated with 1 ml of 0.1% (w/v) saponin in DPBS for 15 min at 37 °C without prior treatment with gentamicin. The resulting suspensions were diluted and plated on MH agar plates as described above. For each strain, the level of bacterial adhesion and uptake was determined by calculating the number of CFU as described [69]. All experiments were performed in triplicates.
Infection of MKN-28 cells in a transwell filter system
The human polarized epithelial cell line MKN-28 was obtained from the Japanese Collection of Research Biosources (JCRB0253). MKN-28 cells are cultured at 37 °C in 5% CO2 using RPMI-1640 medium containing l-Glutamine, 25 mM HEPES, and Pen/Strep antibiotics cocktail (Invitrogen). To study bacterial transmigration, the cells were grown for 14 days in a transwell filter system with pore size of 3.0 µm (Corning B.V. Lifescience, Schiphol/Netherlands) placed in 12-well plates. As control, the TER values were measured every 2 days followed by medium change as described [26]. Twelve hours before infection, the growth medium was removed, cells were washed with DPBS, and fresh medium without antibiotics was added. Infections were carried out with the different C. jejuni strains using a MOI of 50 for the indicated periods of time (30 min to 24 h). At each given time point, 10 μL aliquots from the basolateral chamber of the transwell were taken and incubated on MH agar plates for 3 days under microaerobic conditions. The number of transmigrated bacteria was determined by counting the CFU in triplicates.
Casein zymography
Bacterial lysates, culture supernatants, or recombinant HtrA were separated under nonreducing conditions in gels containing casein. Subsequently, gels were renatured in 2.5% Triton-X-100 and equilibrated in developing buffer [26–35]. Caseinolytic activity was visualized by staining with 0.5% Coomassie Blue R250.
Antibodies and Western blotting
Two polyclonal antibodies, α-FAK and α-GAPDH, were purchased from Santa Cruz (Heidelberg/Germany). In addition, we generated polyclonal antibodies raised against conserved peptides corresponding to the following amino acid (aa) residues in four C. jejuni proteins: HtrA (aa 288–301: QGDTKKAYKNQEGA), CadF (aa 293–306: QDNPRSSNDTKEGR), CiaB (aa 597–610: C-EIDNSGEFERYKKK), and MOMP (aa 400–413: C-NLDQGVNTNESADH). For this purpose, all four peptides were synthesized with an additional amino-terminal cysteine residue (Biosynthan, Berlin/Germany), used for covalent conjugation to the Limulus polyphemus hemocyanin carrier protein [70]. Two rabbits each were immunized using standard protocols of Biogenes GmbH (Berlin/Germany). The resulting antisera were affinity-purified, and the specificity against the HtrA, CadF, CiaB, and MOMP proteins was confirmed by SDS–PAGE and Western blotting. Horseradish peroxidase-conjugated antirabbit polyvalent sheep immunoglobulin was used as secondary antibody (DAKO, Glostrup/Denmark). Blots were developed with ECL Plus Western blot reagents (GE Healthcare, Munich/Germany) as described [71, 72].
Statistics
All data were evaluated using Student t-test with SigmaStat statistical software (version 2.0). Statistical significance was defined by P ≤ 0.05 and P ≤ 0.005. All error bars shown in figures and those quoted following the +/− signs represent standard deviation.
Acknowledgements
The work of S.B. is supported through a grant by the German Science Foundation (project B10 of CRC-796). We thank Dr. Dennis Linton (University Manchester, UK) for providing the plasmid pCam-148, and Dr. Sabine Kienesberger (University Graz/Austria) for plasmid pRYSK12. We also thank Dr. Silja Wessler (University Salzburg, Austria) and Dr. Omar A. Oyarzabal (University of Vermont, VT, USA) for critical discussion of the data.
Abbreviations
- BHI
brain heart infusion
- CFU
colony forming
- FCS
fetal calf serum
- MH
Mueller-Hinton
- MOI
multi-plicity of infection
- NaCl
sodium chloride
- DPBS
Dulbecco’s phosphate buffered saline
- TER
transepithelial electrical resistance
- OD
optical density
- HtrA
high temperature resistant protein A
Funding Statement
Financial disclosure, grant support: The work of S.B. was supported by a grant of the German Science Foundation (project B10 in CRC-796). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the article.
Footnotes
Competing interests: The authors have declared that no competing interests exist.
Author contributions: Conceived and designed the experiments: M.B., N.T. Performed the experiments: M.B., N.T., J.L. Analyzed the data: M.B., N.T., J.L., S.B. Wrote the paper: M.B., S.B., N.T.
Contributor Information
Manja Boehm, Department of Biology, Division of Microbiology, Friedrich Alexander University Erlangen/Nuremberg, Staudtstr. 5, D-91058 Erlangen, Germany.
Judith Lind, Department of Biology, Division of Microbiology, Friedrich Alexander University Erlangen/Nuremberg, Staudtstr. 5, D-91058 Erlangen, Germany.
Steffen Backert, Department of Biology, Division of Microbiology, Friedrich Alexander University Erlangen/Nuremberg, Staudtstr. 5, D-91058 Erlangen, Germany.
Nicole Tegtmeyer, Department of Biology, Division of Microbiology, Friedrich Alexander University Erlangen/Nuremberg, Staudtstr. 5, D-91058 Erlangen, Germany.
References
- 1.World Health Organization. Foodborne Diseases. Geneva, Switzerland: WHO; http://www.who.int/foodsafety/areas_work/foodborne-diseases/en/; 2014. [Google Scholar]
- 2.Young KT, Davis LM, DiRita VJ. Campylobacter jejuni: molecular biology and pathogenesis. Nat Rev Microbiol. 2007;5:665–679. doi: 10.1038/nrmicro1718. [DOI] [PubMed] [Google Scholar]
- 3.Nachamkin I, Szymanski CM, Blaser MJ. Campylobacter. Washington, DC: ASM Press; 2008. [Google Scholar]
- 4.Oyarzabal OA, Backert S. Microbial Food Safety. New York: Springer; 2011. [Google Scholar]
- 5.Talukder RK, Sutradhar SR, Rahman KM, Uddin MJ, Akhter H. Guillian–Barre syndrome. Mymensingh Med J. 2011;20:748–756. [PubMed] [Google Scholar]
- 6.Wakerley BR, Uncini A, Yuki N GBS Classification Group. Guillain–Barré and Miller Fisher syndromes—new diagnostic classification. Nat Rev Neurol. 2014;10:537–544. doi: 10.1038/nrneurol.2014.138. [DOI] [PubMed] [Google Scholar]
- 7.Hofreuter D. Defining the metabolic requirements for the growth and colonization capacity of Campylobacter jejuni. Front Cell Infect Microbiol. 2014;4:137. doi: 10.3389/fcimb.2014.00137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wassenaar TM, Blaser MJ. Pathophysiology of Campylobacter jejuni infections of humans. Microbes Infect. 1999;1:1023–1033. doi: 10.1016/s1286-4579(99)80520-6. [DOI] [PubMed] [Google Scholar]
- 9.Gölz G, Sharbati S, Backert S, Alter T. Quorum sensing dependent phenotypes and their molecular mechanisms in Campylobacterales. Eur J Microbiol Immunol (Bp) 2012;2:50–60. doi: 10.1556/EuJMI.2.2012.1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ó Cróinín T, Backert S. Host epithelial cell invasion by Campylobacter jejuni: trigger or zipper mechanism? Front Cell Infect Microbiol. 2012;2:25. doi: 10.3389/fcimb.2012.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Szymanski CM, King M, Haardt M, Armstrong GD. Campylobacter jejuni motility and invasion of Caco-2 cells. Infect Immun. 1995;63:4295–4300. doi: 10.1128/iai.63.11.4295-4300.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Young GM, Schmiel DH, Miller VL. A new pathway for the secretion of virulence factors by bacteria: the flagellar export apparatus functions as a protein-secretion system. Proc Natl Acad Sci USA. 1999;96:6456–6461. doi: 10.1073/pnas.96.11.6456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Konkel ME, Gray SA, Kim BJ, Garvis SG, Yoon JJ. Identification of the enteropathogens Campylobacter jejuni and Campylobacter coli based on the cadF virulence gene and its product. J Clin Microbiol. 1999;37:510–517. doi: 10.1128/jcm.37.3.510-517.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Christensen JE, Pacheco SA, Konkel ME. Identification of a Campylobacter jejuni — secreted protein required for maximal invasion of host cells. Mol Microbiol. 2009;73:650–662. doi: 10.1111/j.1365-2958.2009.06797.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Neal-McKinney JM, Christensen JE, Konkel ME. Aminoterminal residues dictate the export efficiency of the Campylobacter jejuni filament proteins via the flagellum. Mol Microbiol. 2010;76:918–931. doi: 10.1111/j.1365-2958.2010.07144.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Euker TP, Konkel ME. The cooperative action of bacterial fibronectin-binding proteins and secreted proteins promote maximal Campylobacter jejuni invasion of host cells by stimulating membrane ruffling. Cell Microbiol. 2012;14:226–238. doi: 10.1111/j.1462-5822.2011.01714.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barrero-Tobon AM, Hendrixson DR. Identification and analysis of flagellar coexpressed determinants (Feds) of Campylobacter jejuni involved in colonization. Mol Microbiol. 2012;84:352–369. doi: 10.1111/j.1365-2958.2012.08027.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Barrero-Tobon AM, Hendrixson DR. Flagellar biosynthesis exerts temporal regulation of secretion of specific Campylobacter jejuni colonization and virulence determinants. Mol Microbiol. 2014;93:957–974. doi: 10.1111/mmi.12711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pei Z, Burucoa C, Grignon B, Baqar S, Huang XZ, Kopecko DJ, Bourgeois AL, Fauchere JL, Blaser MJ. Mutation in the peb1A locus of Campylobacter jejuni reduces interactions with epithelial cells and intestinal colonization of mice. Infect Immun. 1998;66:938–943. doi: 10.1128/iai.66.3.938-943.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Konkel ME, Monteville MR, Rivera-Amill V, Joens LA. The pathogenesis of Campylobacter jejuni-mediated enteritis. Curr Issues Intest Microbiol. 2001;2:55–71. [PubMed] [Google Scholar]
- 21.Poly F, Guerry P. Pathogenesis of Campylobacter. Curr Opin Gastroenterol. 2008;24:27–31. doi: 10.1097/MOG.0b013e3282f1dcb1. [DOI] [PubMed] [Google Scholar]
- 22.Brøndsted L, Andersen MT, Parker M, Jørgensen K, Ingmer H. The HtrA protease of Campylobacter jejuni is required for heat and oxygen tolerance and for optimal interaction with human epithelial cells. Appl Environ Micro biol. 2005;71:3205–3212. doi: 10.1128/AEM.71.6.3205-3212.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bæk KT, Vegge CS, Brøndsted L. HtrA chaperone activity contributes to host cell binding in Campylobacter jejuni. Gut Pathog. 2011;3:13. doi: 10.1186/1757-4749-3-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Krause-Gruszczynska M, Rohde M, Hartig R, Genth H, Schmidt G, Keo T, Koenig W, Miller WG, Konkel ME, Backert S. Role of small Rho GTPases Rac1 and Cdc42 in host cell invasion of Campylobacter jejuni. Cell Microbiol. 2007;9:2431–2444. doi: 10.1111/j.1462-5822.2007.00971.x. [DOI] [PubMed] [Google Scholar]
- 25.Krause-Gruszczynska M, Boehm M, Rohde M, Tegtmeyer N, Takahashi S, Buday L, Oyarzabal OA, Backert S. The signaling pathway of Campylobacter jejuni-induced Cdc42 activation: Role of fibronectin, integrin beta1, tyrosine kinases and guanine exchange factor Vav2. Cell Commun Signal. 2011;9:32. doi: 10.1186/1478-811X-9-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Boehm M, Hoy B, Rohde M, Tegtmeyer N, Bæk KT, Oyarzabal OA, Brøndsted L, Wessler S, Backert S. Rapid paracellular transmigration of Campylobacter jejuni across polarized epithelial cells without affecting TER: role of proteolytic-active HtrA cleaving E-cadherin but not fibronectin. Gut Pathog. 2012;4:3. doi: 10.1186/1757-4749-4-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ingmer H, Brøndsted L. Proteases in bacterial pathogenesis. Res Microbiol. 2009;160:704–710. doi: 10.1016/j.resmic.2009.08.017. [DOI] [PubMed] [Google Scholar]
- 28.Frees D, Brøndsted L, Ingmer H. Bacterial proteases and virulence. Subcell Biochem. 2013;66:161–192. doi: 10.1007/978-94-007-5940-4_7. [DOI] [PubMed] [Google Scholar]
- 29.Backert S, Boehm M, Wessler S, Tegtmeyer N. Transmigration route of Campylobacter jejuni across polarized intestinal epithelial cells: paracellular, transcellular or both? Cell Commun Signal. 2013;11:72. doi: 10.1186/1478-811X-11-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Skorko-Glonek J, Zurawa-Janicka D, Koper T, Jarzab M, Figaj D, Glaza P, Lipinska B. HtrA protease family as therapeutic targets. Curr Pharm Des. 2013;19:977–1009. doi: 10.2174/1381612811319060003. [DOI] [PubMed] [Google Scholar]
- 31.Kim DY, Kim KK. Structure and function of HtrA family proteins, the key players in protein quality control. J Biochem Mol Biol. 2005;38:266–274. doi: 10.5483/bmbrep.2005.38.3.266. [DOI] [PubMed] [Google Scholar]
- 32.Clausen T, Southan C, Ehrmann M. The HtrA family of proteases: implications for protein composition and cell fate. Mol Cell. 2002;10:443–455. doi: 10.1016/s1097-2765(02)00658-5. [DOI] [PubMed] [Google Scholar]
- 33.Clausen T, Kaiser M, Huber R, Ehrmann M. HTRA proteases: regulated proteolysis in protein quality control. Nat Rev Mol Cell Biol. 2011;12:152–162. doi: 10.1038/nrm3065. [DOI] [PubMed] [Google Scholar]
- 34.Hoy B, Löwer M, Weydig C, Carra G, Tegtmeyer N, Geppert T, Schröder P, Sewald N, Backert S, Schneider G, Wessler S. Helicobacter pylori HtrA is a new secreted virulence factor that cleaves E-cadherin to disrupt intercellular adhesion. EMBO Rep. 2010;11:798–804. doi: 10.1038/embor.2010.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hoy B, Geppert T, Boehm M, Reisen F, Plattner P, Gadermaier G, Sewald N, Ferreira F, Briza P, Schneider G, Backert S, Wessler S. Distinct roles of secreted HtrA proteases from gram-negative pathogens in cleaving the junctional protein and tumor suppressor E-cadherin. J Biol Chem. 2012;287:10115–10120. doi: 10.1074/jbc.C111.333419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Champion OL, Karlyshev AV, Senior NJ, Woodward M, La Ragione R, Howard SL, Wren BW, Titball RW. Insect infection model for Campylobacter jejuni reveals that Omethyl phosphoramidate has insecticidal activity. J Infect Dis. 2010;201:776–782. doi: 10.1086/650494. [DOI] [PubMed] [Google Scholar]
- 37.Heimesaat MM, Alutis M, Grundmann U, Fischer A, Tegtmeyer N, Böhm M, Kühl AA, Göbel UB, Backert S, Bereswill S. The role of serine protease HtrA in acute ulcerative enterocolitis and extra-intestinal immune responses during Campylobacter jejuni infection of gnotobiotic IL-10 deficient mice. Front Cell Infect Microbiol. 2014;4:77. doi: 10.3389/fcimb.2014.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Heimesaat MM, Fischer A, Alutis M, Grundmann U, Boehm M, Tegtmeyer N, Göbel UB, Kühl AA, Bereswill S, Backert S. The impact of serine protease HtrA in apoptosis, intestinal immune responses and extra-intestinal histopathology during Campylobacter jejuni infection of infant mice. Gut Pathog. 2014;6:16. doi: 10.1186/1757-4749-6-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Boehm M, Haenel I, Hoy B, Brøndsted L, Smith TG, Hoover T, Wessler S, Tegtmeyer N. Extracellular secretion of protease HtrA from Campylobacter jejuni is highly efficient and independent of its protease activity and flagellum. Eur J Microbiol Immunol (Bp) 2013;3:163–173. doi: 10.1556/EuJMI.3.2013.3.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wroblewski LE, Shen L, Ogden S, Romero-Gallo J, Lapierre LA, Israel DA, Turner JR, Peek RM., Jr. Helicobacter pylori dysregulation of gastric epithelial tight junctions by urease-mediated myosin II activation. Gastroenterology. 2009;136:236–246. doi: 10.1053/j.gastro.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li SR, Dorrell N, Everest PH, Dougan G, Wren BW. Construction and characterization of a Yersinia enterocolitica O:8 high-temperature requirement (htrA) isogenic mutant. Infect Immun. 1996;64:2088–2094. doi: 10.1128/iai.64.6.2088-2094.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Humphreys S, Stevenson A, Bacon A, Weinhardt AB, Roberts M. The alternative sigma factor, sigmaE, is critically important for the virulence of Salmonella typhimurium. Infect Immun. 1999;67:1560–1568. doi: 10.1128/iai.67.4.1560-1568.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cortes G, de Astorza B, Benedi VJ, Alberti S. Role of the htrA gene in Klebsiella pneumoniae virulence. Infect Immun. 2002;70:4772–4776. doi: 10.1128/IAI.70.9.4772-4776.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Purdy GE, Hong M, Payne SM. Shigella flexneri DegP facilitates IcsA surface expression and is required for efficient intercellular spread. Infect Immun. 2002;70:6355–6364. doi: 10.1128/IAI.70.11.6355-6364.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wilson RL, Brown LL, Kirkwood-watts D, Warren TK, Lund SA, King DS, Jones KF, Hruby DE. Listeria monocytogenes 10403S HtrA is necessary for resistance to cellular stress and virulence. Infect Immun. 2006;74:765–768. doi: 10.1128/IAI.74.1.765-768.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gloeckl S, Ong VA, Patel P, Tyndall JD, Timms P, Beagley KW, Allan JA, Armitage CW, Turnbull L, Whitchurch CB, Merdanovic M, Ehrmann M, Powers JC, Oleksyszyn J, Verdoes M, Bogyo M, Huston WM. Identification of a serine protease inhibitor which causes inclusion vacuole reduction and is lethal to Chlamydia trachomatis. Mol Microbiol. 2013;89:676–689. doi: 10.1111/mmi.12306. [DOI] [PubMed] [Google Scholar]
- 47.Weibezahn J, Schlieker C, Tessarz P, Mogk A, Bukau B. Novel insights into the mechanism of chaperone-assisted protein disaggregation. Biol Chem. 2005;386:739–744. doi: 10.1515/BC.2005.086. [DOI] [PubMed] [Google Scholar]
- 48.Doyle SM, Wickner S. Hsp104 and ClpB: protein disaggregating machines. Trends Biochem Sci. 2009;34:40–48. doi: 10.1016/j.tibs.2008.09.010. [DOI] [PubMed] [Google Scholar]
- 49.Johnson K, Charles I, Dougan G, Pickard D, O’Gaora P, Costa G, Ali T, Miller I. The role of a stress-response protein in Salmonella typhimurium virulence. Mol Microbiol. 1991;5:401–407. doi: 10.1111/j.1365-2958.1991.tb02122.x. [DOI] [PubMed] [Google Scholar]
- 50.Elzer PH, Phillips RW, Robertson GT, Roop RM. The HtrA stress response protease contributes to resistance of Brucella abortus to killing by murine phagocytes. Infect Immun. 1996;64:4838–4841. doi: 10.1128/iai.64.11.4838-4841.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Smith MA, Finel M, Korolik V, Mendz GL. Characteristics of the aerobic respiratory chains of the microaerophiles Campylobacter jejuni and Helicobacter pylori. Arch Microbiol. 2000;174:1–10. doi: 10.1007/s002030000174. [DOI] [PubMed] [Google Scholar]
- 52.Krojer T, Sawa J, Schäfer E, Saibil HR, Ehrmann M, Clausen T. Structural basis for the regulated protease and chaperone function of DegP. Nature. 2008;453:885–890. doi: 10.1038/nature07004. [DOI] [PubMed] [Google Scholar]
- 53.Parkhill J, Wren BW, Mungall K, Ketley JM, Churcher C, Basham D, Chillingworth T, Davies RM, Feltwell T, Holroyd S, Jagels K, Karlyshev AV, Moule S, Pallen MJ, Penn CW, Quail MA, Rajandream MA, Rutherford KM, van Vliet AH, Whitehead S, Barrell BG. The genome sequence of the food-borne pathogen Campylobacter jejuni reveals hypervariable sequences. Nature. 2000;403:665–668. doi: 10.1038/35001088. [DOI] [PubMed] [Google Scholar]
- 54.Fouts DE, Mongodin EF, Mandrell RE, Miller WG, Rasko DA, Ravel J, Brinkac LM, DeBoy RT, Parker CT, Daugherty SC, Dodson RJ, Durkin AS, Madupu R, Sullivan SA, Shetty JU, Ayodeji MA, Shvartsbeyn A, Schatz MC, Badger JH, Fraser CM, Nelson KE. Major structural differences and novel potential virulence mechanisms from the genomes of multiple Campylobacter species. PLoS Biol. 2005;3:e15. doi: 10.1371/journal.pbio.0030015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hofreuter D, Tsai J, Watson RO, Novik V, Altman B, Benitez M, Clark C, Perbost C, Jarvie T, Du L, Galán JE. Unique features of a highly pathogenic Campylobacter jejuni strain. Infect Immun. 2006;74:4694–4707. doi: 10.1128/IAI.00210-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wiesner RS, Hendrixson DR, DiRita VJ. Natural transformation of Campylobacter jejuni requires components of a type II secretion system. J Bacteriol. 2003;185:5408–5418. doi: 10.1128/JB.185.18.5408-5418.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Novik V, Hofreuter D, Galan JE. Identification of Campylobacter jejuni genes involved in its interaction with epithelial cells. Infect Immun. 2010;78:3540–3553. doi: 10.1128/IAI.00109-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Boehm M, Krause-Gruszczynska M, Rohde M, Tegtmeyer N, Takahashi S, Oyarzabal OA, Backert S. Major host factors involved in epithelial cell invasion of Campylobacter jejuni: Role of fibronectin, integrin beta1, FAK, Tiam-1 and DOCK180 in activating Rho GTPase Rac1. Front Cell Infect Microbiol. 2011;1:17. doi: 10.3389/fcimb.2011.00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ashgar SS, Oldfield NJ, Wooldridge KG, Jones MA, Irving GJ, Turner DP, Ala’Aldeen DA. CapA, an autotransporter protein of Campylobacter jejuni, mediates association with human epithelial cells and colonization of the chicken gut. J Bacteriol. 2007;189:1856–1865. doi: 10.1128/JB.01427-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jin S, Joe A, Lynett J, Hani EK, Sherman P, Chan VL. JlpA, a novel surface exposed lipoprotein specific to Campylobacter jejuni, mediates adherence to host epithelial cells. Mol Microbiol. 2001;39:1225–1236. doi: 10.1111/j.1365-2958.2001.02294.x. [DOI] [PubMed] [Google Scholar]
- 61.Szymanski CM, Burr DH, Guerry P. Campylobacter protein glycosylation affects host cell interactions. Infect Immun. 2002;70:2242–2244. doi: 10.1128/IAI.70.4.2242-2244.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vuckovic D, Abram M, Doric M. Primary Campylobacter jejuni infection in different mice strains. Microb Pathog. 1998;24:263–268. doi: 10.1006/mpat.1997.0194. [DOI] [PubMed] [Google Scholar]
- 63.Watson RO, Novik V, Hofreuter D, Lara-Tejero M, Galan JE. A MyD88-deficient mouse model reveals a role for Nramp1 in Campylobacter jejuni infection. Infect Immun. 2007;75:1994–2003. doi: 10.1128/IAI.01216-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lamb-Rosteski JM, Kalischuk LD, Inglis GD, Buret AG. Epidermal growth factor inhibits Campylobacter jejuni-induced claudin-4 disruption, loss of epithelial barrier function, and Escherichia coli translocation. Infect Immun. 2008;76:3390–3398. doi: 10.1128/IAI.01698-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Nemelka KW, Brown AW, Wallace SM, Jones E, Asher LV, Pattarini D, Applebee L, Gilliland TC, Jr., Guerry P, Baqar S. Immune response to and histopathology of Campylobacter jejuni infection in ferrets (Mustela putorius furo) Comp Med. 2009;59:363–371. [PMC free article] [PubMed] [Google Scholar]
- 66.Everest PH, Goossens H, Sibbons P, Lloyd DR, Knutton S, Leece R, Ketley JM, Williams PH. Pathological changes in the rabbit ileal loop model caused by Campylobacter jejuni from human colitis. J Med Microbiol. 1993;38:316–321. doi: 10.1099/00222615-38-5-316. [DOI] [PubMed] [Google Scholar]
- 67.Zhou P, Hussain SK, Liles MR, Arias CR, Backert S, Kieninger J, Oyarzabal OA. A simplified and cost-effective enrichment protocol for the isolation of Campylobacter spp. from retail broiler meat without microaerobic incubation. BMC Microbiol. 2011;11:175. doi: 10.1186/1471-2180-11-175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Speegle L, Miller ME, Backert S, Oyarzabal OA. Use of cellulose filters to isolate Campylobacter spp. from naturally contaminated retail broiler meat. J Food Prot. 2009;72:2592–2596. doi: 10.4315/0362-028x-72.12.2592. [DOI] [PubMed] [Google Scholar]
- 69.Backert S, Hofreuter D. Molecular methods to investigate adhesion, transmigration, invasion and intracellular survival of the foodborne pathogen Campylobacter jejuni. J Microbiol Methods. 2013;95:8–23. doi: 10.1016/j.mimet.2013.06.031. [DOI] [PubMed] [Google Scholar]
- 70.Tegtmeyer N, Rivas Traverso F, Rohde M, Oyarzabal OA, Lehn N, Schneider-Brachert W, Ferrero RL, Fox JG, Berg DE, Backert S. Electron microscopic, genetic and protein expression analyses of Helicobacter acinonychis strains from a Bengal tiger. PLoS One. 2013;8:e71220. doi: 10.1371/journal.pone.0071220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tegtmeyer N, Wittelsberger R, Hartig R, Wessler S, Martinez-Quiles B S. Serine phosphorylation of cortactin controls focal adhesion kinase activity and cell scattering induced by Helicobacter pylori. Cell Host Microbe. 2011;9:520–531. doi: 10.1016/j.chom.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 72.Mueller D, Tegtmeyer N, Brandt S, Yamaoka Y, De Poire E, Sgouras D, Wessler S, Torres J, Smolka A, Backert S. c-Src and c-Abl kinases control hierarchic phosphorylation and function of the CagA effector protein in Western and East Asian Helicobacter pylori strains. J Clin Invest. 2012;122:1553–1566. doi: 10.1172/JCI61143. [DOI] [PMC free article] [PubMed] [Google Scholar]