Abstract
AIM: To investigate the effect and significance of selenium in early experimental gastric carcinogenesis.
METHODS: Weaning male Wistar rats were divided randomly into normal control group, experiment control group, low selenium (2 mg/L) group and high selenium (4 mg/L) group. Wistar rat gastric carcinogenesis was induced by N-methyl-N-nitro-N-nitroso guanidine (MNNG) (20 mg/kg) gavage daily for 10 d. Na2SeO3 was given by piped drinking 1 wk prior to MNNG gavage. The rats were killed at the 43rd wk. The surface characteristics of gastric mucosa were observed with naked eyes. Histopathologic changes of rat gastric mucosa were observed by HE staining and AB-PAS methods. The changes of cellular ultrastructure were observed under transmission electron microscope. Statistical analysis was carried out by SPSS.
RESULTS: The incidence rate of gastric mucosa erosion, hemorrhage and intestinal metaplasia was 0, 45.5%, 66.7%, and 92.9%, respectively (92.9% vs 45.5%, P<0.05) in the normal control group, experiment control group, low selenium group, and high selenium group. Leiomyoma formed in the process of inducement of rat gastric carcinoma. Dietary Na2SeO3 (2 and 4 mg/L) slightly increased the incidence rate of leiomyoma (0, 23%, 46.6%, and 46.6%). gastric mucosa did not change in the course of rat gastric carcinogenesis. Dietary Na2SeO3 by pipe drinking could expand the intracellular secretory canaliculus of parietal cells and increase the number of endocrine cells and lysosomes.
CONCLUSION: Dietary Na2SeO3 by pipe drinking aggravates gastric erosion, hemorrhage and promotes intestinal metaplasia of gastric mucosa. The mechanism may be related with the function of parietal cells.
Keywords: Selenium, Ultrastructure, Experimental gastric carcinogenesis
INTRODUCTION
Gastric cancer results from chronic superficial gastritis, atrophic gastritis, intestinal metaplasia, and heterotypic hyperplasia. Previous studies showed that selenium (Se) supplementation or selenium deficiency causes changes in the cellular ultrastructure[1-4]. Selenium is an essential trace element in mammals. The results of laboratory investigations and cohort studies suggest that selenium exhibits a bivalent effect in cancer, either increasing or decreasing the risk of cancer[5,6]. We conducted this study to investigate the effect and significance of selenium in experimental gastric carcinogenesis.
MATERIALS AND METHODS
Materials
A total of 62 weaned male Wistar rats, weighing 31-56 g, were purchased from Department of Experimental Animals, Health Science Center of Peking University. The rats were housed in wire-bottomed metal cages at controlled temperature (21-22 °C) with a humidity of 30-50%, and in light-dark cycle. The rats were divided into the normal control group (group 1) which was given dimethyl sulfoxide (DMSO)-0.9% NaCl vehicle gavage with drinking normal tap water, experiment control group (group 2) which was given N-methyl-N-nitro-N-nitroso guanidine (MNNG), 20 mg/kg (presented by Professor Deng-DJ) gavage with drinking normal tap water, low selenium (2 mg/L) group (group 3) which was given MNNG (20 mg/kg) gavage with drinking water containing 2 mg selenium/L as sodium selenite before 1 wk administration of MNNG, or at the beginning of the experiment, and high selenium (4 mg/L) group (group 4) which was given MNNG (20 mg/kg) gavage with drinking water containing 4 mg selenium/L as sodium selenite before 1 wk administration of MNNG (Table 1).
Table 1.
Groups of Wistar rats
| Group | Number | Dose of MNNG (mg/kg) | Dose of selenium in water (mg/L) | Time of selenium given |
| Normal control | 10 | 0 | 0 | / |
| Experimental control | 22 | 20 | 0 | / |
| Low selenium | 15 | 20 | 2 | 1 wk before MNNG gavage |
| High selenium | 15 | 20 | 4 | 1 wk before MNNG gavage |
Methods
The rats in groups 2-4 were given MNNG (20 mg/kg) gavage daily for 10 d, 1 wk after the experiment. MNNG was dissolved in DMSO at the concentration of 100 g/L and kept in a cool, dark place. The stock solution was diluted to 1 g/L with 0.9% physiological saline before use. The rats in normal group were given DMSO-0.9% NaCl vehicle gavage. Selenium containing water was prepared. The low and high selenium groups were given selenium containing water at the beginning of experiment, 1 wk before administration of MNNG. The experiment rats had free access to diet and water. The animals were examined and weighed twice a week.
The animals were killed at the 43rd wk. The rats were starved for 12 h and anesthetized with 2.5% pentobarbitone intraperitoneal injection before they were killed. The stomach and other organs of the rats were carefully examined. Tissues from the stomach were fixed in 10% neutral Phosphate-buffered formalin solution and embedded in paraffin. Serial sections (5-6 μm thick) were obtained from the stomach and stained with hematoxylin and eosin (HE), or Alcian blue-Periodic acid-Schiff (AB-PAS). Metaplasia of gastric mucosa was found as blue or purple blue granules in gastric mucosa cells after AB-PAS staining. For electron microscopy, the tissues from stomach were fixed for 2 h at 4 °C in 2.5 glutaraldehyde solution in 0.1 mol/L phosphate buffer, postfixed in 1.0% osmium tetroxide in the same buffer at 4 °C for 1 h, and then embedded in Epoxy resin. Ultrathin sections were stained with uranium acetate and lead acetate according to double stained method. The results were examined under a JEM-JEOL 100 electron microscope.
Statistical analysis
The clinicopathological results were statistically analyzed by SPSS software and χ2 test.
RESULTS
Clinicopathological results
The results are shown in Table 2. The stomachs in the normal control group were smooth on the surface of serosa. Their inner surfaces clearly presented longitudinal branching folds and gastric pits. All the surface cells of rat gastric mucosa in group 1 had violet red granules (Figure 2A). Erosion and hemorrhage of gastric mucosa were found in 10 of 22 rats (45.5%) in the experimental control group (Figure 1A). There were many blue or purple blue granules in the erosive gastric mucosa of group 2 (Figure 2B). Similar erosive, hemorrhagic and intestinal metaplasia of gastric mucosa was found in 10 of 15 rats (66.7%) of low selenium group, 13 of 14 rats (92.9%) of high selenium group. The incidence rate of intestinal metaplasia was significantly higher in low and high selenium groups than in experiment control group (P<0.05). The incidence of intestinal metaplasia was not significantly different between the high and low selenium groups. There were no significant changes by HE staining, but the glandular cavity of fundic gland was slightly larger in high selenium group than in the other groups. In experimental control group, a single neoplasm was found on the surface of serosa breaching the serosa (Figure 1B). The diameter was about 0.2-1 cm. A similar neoplasm was also found in low and high selenium groups. The incidence of neoplasm was slightly higher in these two groups than in experimental control group. The neoplasm was diagnosed as leiomyoma by HE staining.
Table 2.
Results of pathologic diagnosis
| Number of rats | Incidence rate of leiomyoma (%) | Incidence rate of intestinal metaplasia (%) | |
| Normal control | 10 | 0 | 0 |
| Experimental control | 22 | 23 | 45.5 |
| Low selenium | 15 | 46.6 | 66.7a |
| High selenium | 14 | 42.8 | 92.9a |
aP<0.05 vs experiment control group.
Figure 2.
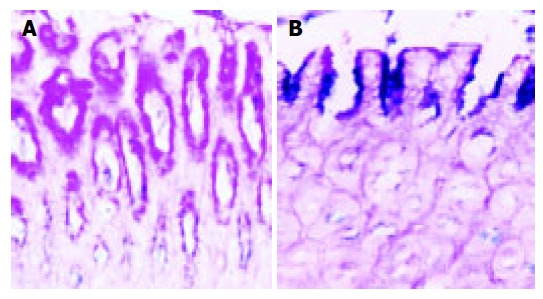
Normal mucosa (A) and erosive mucosa (B) of rat stomach, AB-PAS, ×200.
Figure 1.
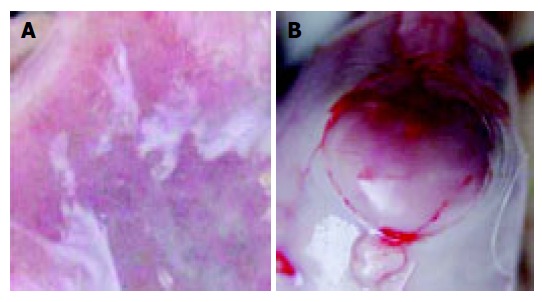
Erosive mucosa of rat stomach (A) and leiomyoma under serosa (B).
Ultrastructural changes of gastric mucosa
In normal control group, the most distinctive feature in the ultrastructure of gastric mucosa was the presence of a deep invagination of the apical membrane forming a secretory canaliculus. The membrane lining the canaliculus had numerous microvilli projecting into its lumen. About 40% of the cytoplasmic volume was occupied by large mitochondria (Figure 3). Fewer endocrine cells were found in the normal gastric mucosa. Pinosomes distributed in the capillary endothelium of normal control group were large in diameter. Compared to normal control group, the secretory canaliculi in parietal cells were scarce, microvilli projecting into canaliculus lumen were sparse, the pinosomes were more in number and smaller in diameter than in experimental control group. Compared to the normal and experimental control groups, the following characteristics were observed in selenium supplement group: (1) the secretory canaliculus expanded and the microvilli projecting into canaliculus were scarce (Figure 4); (2) endocrine cells scattered in gastric mucosa contained granules with a different diameter and high electron density, there were a few secondary lysosomes in cytoplasm of some endocrine cells (Figure 5); (3) more eosinophils were found in the erosive gastric mucosa (Figure 6); (4) the chief cells had abundant rough endoplasmic reticulum, mitochondria were more in number and larger in volume; (5) the pinosomes in capillary endothelium were slightly less. These characteristics were significant in high selenium group.
Figure 3.
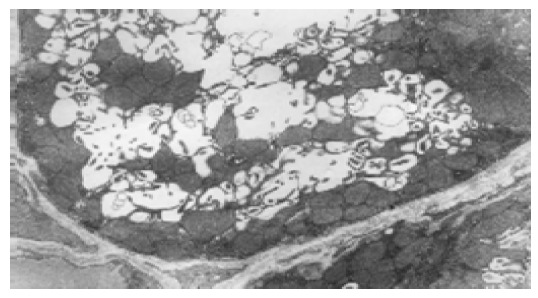
Intracellular secretory canaliculi of parietal cells in normal control group, TEM 5.8×103.
Figure 4.
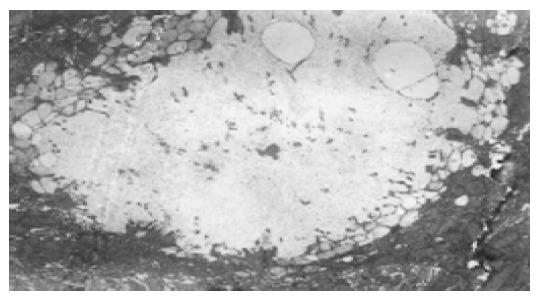
Expansion of intracellular secretory canaliculi of parietal cells in high selenium group, TEM 5.8×103.
Figure 5.
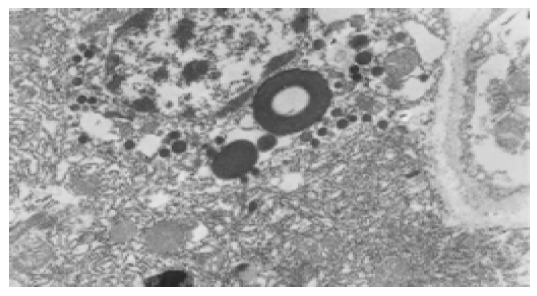
Endocrine cells and secondary lysosomes in high selenium group, TEM 10×103.
Figure 6.
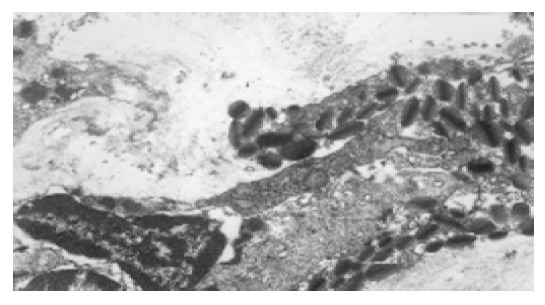
Infiltration of eosinophils in gastric mucosa of high selenium group, TEM 10×103.
DISCUSSION
Selenium, a constituent of antioxidant enzyme, has been proposed as a chemopreventive agent for many kinds of cancer[7-10]. Selenium supplement to diet or drinking water can prevent tumorigenesis and decrease the incidence rate of cancer[11-14]. The effect of selenium on carcinoma is still controversial[5,6]. It was reported that selenium promotes carcinogenesis and tumor development[15]. Gastric cancer is the second most frequent cause of cancer death in the world and the leading cause of cancer death in China. Studies have been focused on randomized nutritional intervention trial to prevent gastric cancer[16-20] and epidemiology in serum selenium concentrations and esophageal and gastric cancer[21]. Wu et al[21], reported that gastric cancer tissues have significantly higher concentrations of Fe, K, Mg, Na, Rb, Se, and Zn than normal gastric mucosal tissues. At present, the information on the effect of selenium in early experimental gastric carcinogenesis is scarcely available. This experiment was conducted to investigate the effect and significance of selenium on experimental gastric carcinogenesis by morphologic methods. The findings in this study showed that 2.0 and 4.0 g/L selenium in drinking water for 43 wk aggravated gastric mucosa erosion and hemorrhage and promoted intestinal metaplasia of gastric mucosa. Because gastric cancer results from chronic superficial gastritis, atrophic gastritis, intestinal metaplasia, heterotypic hyperplasia, supplement of selenium as sodium selenite does not decrease the risk of gastric cancer induced by MNNG. Furthermore, leiomyoma formed in the process of gastric carcinogenesis induced by MNNG. Dietary Na2SeO3 (2 and 4 mg/L) increased the incidence of leiomyoma. Since the general dose of selenium is 0.5-6 g/mL (or /g)[22] supplemented to diet and drinking water, the selenium dose in this study did not reach the toxic limits. Consequently the effects of sodium selenite maybe related to the kind of selenium, type of cancer, and different carcinogens.
Selenite induces oxidation and cross-linking of protein thiol groups, mitochondrial permeability transition, decrease in mitochondrial membrane potential, and release of cytochrome C in mitochondria isolated from rat liver, thus possibly inducing tumor cell apoptosis[1]. Zhong and Oberley[2] reported that oxidative stress is induced by sodium selenite at high concentrations, but outcomes are different. After acute exposure to selenite, cells display mitochondrial injury and cell death. After chronic exposure to selenite, cells show growth inhibition due to cell cycle arrest, increase the number of mitochondria and the levels of mitochondrial enzymes. Lysosomes in the liver and kidneys of selenium-deficient rats are enlarged and contain electron-dense areas[3]. Demirel-Yilmaz et al[4], reported that low or high doses of sodium selenite and vitamin E in diet can alter the microvascular permeability of different organs. Our findings in this study indicate that selenium supplement can lead to the expansion of secretory canaliculus, increase in the number of endocrine cells in gastric mucosa, alterations in the mitochondria and changes in the number and shape of pinosomes. Furthermore, our results also indicate that selenium supplement as sodium selenite can alter the ultrastructure of gastric mucosa. Though we did not find gastric cancer and gastric precancerosis, the results indicate that selenium supplement cannot prevent gastric carcinogenesis in early period of MNNG-induced gastric cancer. These mechanisms should be studied by future experiments.
Footnotes
Supported by the Shandong Educational Office, No. J00K67
Science Editor Wang XL and Guo SY Language Editor Elsevier HK
References
- 1.Kim TS, Jeong DW, Yun BY, Kim IY. Dysfunction of rat liver mitochondria by selenite: induction of mitochondrial permeability transition through thiol-oxidation. Biochem Biophys Res Commun. 2002;294:1130–1137. doi: 10.1016/S0006-291X(02)00612-5. [DOI] [PubMed] [Google Scholar]
- 2.Zhong W, Oberley TD. Redox-mediated effects of selenium on apoptosis and cell cycle in the LNCaP human prostate cancer cell line. Cancer Res. 2001;61:7071–7078. [PubMed] [Google Scholar]
- 3.Chareonpong-Kawamoto N, Higasa T, Yasumoto K. Histological study of iron deposits in selenium-deficient rats. Biosci Biotechnol Biochem. 1995;59:1913–1920. doi: 10.1271/bbb.59.1913. [DOI] [PubMed] [Google Scholar]
- 4.Demirel-Yilmaz E, Dinçer D, Yilmaz G, Turan B. The effect of selenium and vitamin E on microvascular permeability of rat organs. Biol Trace Elem Res. 1998;64:161–168. doi: 10.1007/BF02783332. [DOI] [PubMed] [Google Scholar]
- 5.Vinceti M, Wei ET, Malagoli C, Bergomi M, Vivoli G. Adverse health effects of selenium in humans. Rev Environ Health. 2001;16:233–251. doi: 10.1515/reveh.2001.16.4.233. [DOI] [PubMed] [Google Scholar]
- 6.Vinceti M, Nacci G, Rocchi E, Cassinadri T, Vivoli R, Marchesi C, Bergomi M. Mortality in a population with long-term exposure to inorganic selenium via drinking water. J Clin Epidemiol. 2000;53:1062–1068. doi: 10.1016/s0895-4356(00)00233-x. [DOI] [PubMed] [Google Scholar]
- 7.Klein EA. Selenium: epidemiology and basic science. J Urol. 2004;171:S50–S3; discussion S53. doi: 10.1097/01.ju.0000107837.66277.e9. [DOI] [PubMed] [Google Scholar]
- 8.Klein EA, Thompson IM. Update on chemoprevention of prostate cancer. Curr Opin Urol. 2004;14:143–149. doi: 10.1097/00042307-200405000-00002. [DOI] [PubMed] [Google Scholar]
- 9.Sinha R, El-Bayoumy K. Apoptosis is a critical cellular event in cancer chemoprevention and chemotherapy by selenium compounds. Curr Cancer Drug Targets. 2004;4:13–28. doi: 10.2174/1568009043481614. [DOI] [PubMed] [Google Scholar]
- 10.Prokopczyk B, Rosa JG, Desai D, Amin S, Sohn OS, Fiala ES, El-Bayoumy K. American Health Foundation, Valhalla, NY 10595, USA. Chemoprevention of lung tumorigenesis induced by a mixture of benzo(a)pyrene and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone by the organoselenium compound 1,4-phenylenebis(methylene)selenocyanate. Cancer Lett. 2000;161:35–46. doi: 10.1016/s0304-3835(00)00590-5. [DOI] [PubMed] [Google Scholar]
- 11.Nakaji S, Fukuda S, Sakamoto J, Sugawara K, Shimoyama T, Umeda T, Baxter D. Relationship between mineral and trace element concentrations in drinking water and gastric cancer mortality in Japan. Nutr Cancer. 2001;40:99–102. doi: 10.1207/S15327914NC402_4. [DOI] [PubMed] [Google Scholar]
- 12.La Vecchia C, Franceschi S. Nutrition and gastric cancer. Can J Gastroenterol. 2000;14 Suppl D:51D–54D. doi: 10.1155/2000/869862. [DOI] [PubMed] [Google Scholar]
- 13.Beno I, Klvanová J, Magálová T, Brtková A. Blood levels of natural antioxidants in gastric and colorectal precancerous lesions and cancers in Slovakia. Neoplasma. 2000;47:37–40. [PubMed] [Google Scholar]
- 14.Yang CS. Vitamin nutrition and gastroesophageal cancer. J Nutr. 2000;130:338S–339S. doi: 10.1093/jn/130.2.338S. [DOI] [PubMed] [Google Scholar]
- 15.Thompson HJ, Becci PJ. Effect of graded dietary levels of selenium on tracheal carcinomas induced by 1-methyl-1-nitrosourea. Cancer Lett. 1979;7:215–219. doi: 10.1016/s0304-3835(79)80083-x. [DOI] [PubMed] [Google Scholar]
- 16.Li H, Li HQ, Wang Y, Xu HX, Fan WT, Wang ML, Sun PH, Xie XY. An intervention study to prevent gastric cancer by micro-selenium and large dose of allitridum. Chin Med J (Engl) 2004;117:1155–1160. [PubMed] [Google Scholar]
- 17.Taylor PR, Qiao YL, Abnet CC, Dawsey SM, Yang CS, Gunter EW, Wang W, Blot WJ, Dong ZW, Mark SD. Prospective study of serum vitamin E levels and esophageal and gastric cancers. J Natl Cancer Inst. 2003;95:1414–1416. doi: 10.1093/jnci/djg044. [DOI] [PubMed] [Google Scholar]
- 18.You WC, Chang YS, Heinrich J, Ma JL, Liu WD, Zhang L, Brown LM, Yang CS, Gail MH, Fraumeni JF, et al. An intervention trial to inhibit the progression of precancerous gastric lesions: compliance, serum micronutrients and S-allyl cysteine levels, and toxicity. Eur J Cancer Prev. 2001;10:257–263. doi: 10.1097/00008469-200106000-00009. [DOI] [PubMed] [Google Scholar]
- 19.Mark SD, Qiao YL, Dawsey SM, Wu YP, Katki H, Gunter EW, Fraumeni JF, Blot WJ, Dong ZW, Taylor PR. Prospective study of serum selenium levels and incident esophageal and gastric cancers. J Natl Cancer Inst. 2000;92:1753–1763. doi: 10.1093/jnci/92.21.1753. [DOI] [PubMed] [Google Scholar]
- 20.Wei WQ, Abnet CC, Qiao YL, Dawsey SM, Dong ZW, Sun XD, Fan JH, Gunter EW, Taylor PR, Mark SD. Prospective study of serum selenium concentrations and esophageal and gastric cardia cancer, heart disease, stroke, and total death. Am J Clin Nutr. 2004;79:80–85. doi: 10.1093/ajcn/79.1.80. [DOI] [PubMed] [Google Scholar]
- 21.Wu CW, Wei YY, Chi CW, Lui WY, P'Eng FK, Chung C. Tissue potassium, selenium, and iron levels associated with gastric cancer progression. Dig Dis Sci. 1996;41:119–125. doi: 10.1007/BF02208592. [DOI] [PubMed] [Google Scholar]
- 22.Jao SW, Shen KL, Lee W, Ho YS. Effect of selenium on 1,2-dimethylhydrazine-induced intestinal cancer in rats. Dis Colon Rectum. 1996;39:628–631. doi: 10.1007/BF02056940. [DOI] [PubMed] [Google Scholar]


