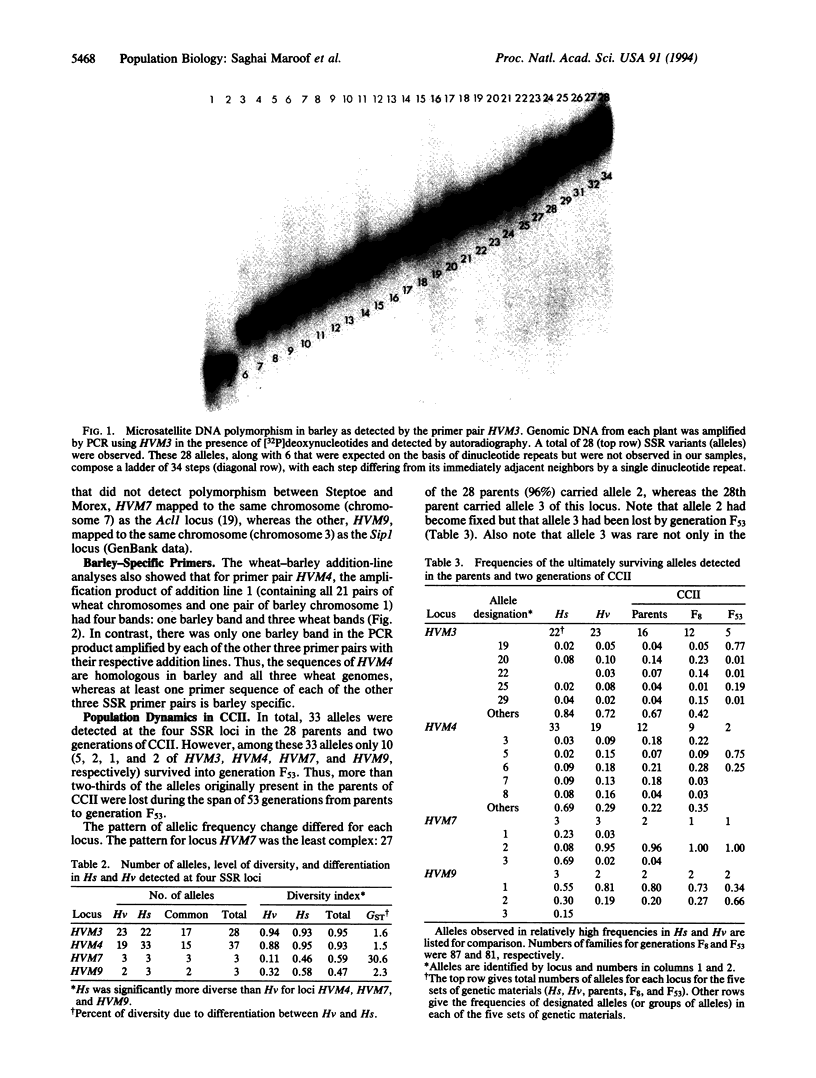
Abstract
This study was undertaken to assess the extent of genetic variation in barley simple sequence repeats (SSRs) and to study the evolutionary dynamics of SSR alleles. SSR polymorphisms were resolved by the polymerase chain reaction with four pairs of primers. In total, 71 variants were observed in a sample of 207 accessions of wild and cultivated barley. Analyses of wheat-barley addition lines and barley doubled haploids identified these variants (alleles) with four loci, each located on a different chromosome. The numbers of alleles detected at a locus corresponded to the number of nucleotide repeats in the microsatellite sequences. The numbers of alleles at two loci were 28 and 37; to our knowledge these are the largest numbers of alleles for single Mendelian loci reported in plants. Three alleles were resolved by each of the other two loci. Allelic diversity was greater in wild than in cultivated barley and surveys of two generations (F8 and F53) of Composite Cross II, an experimental population of cultivated barley, showed that few of the alleles present in the 28 parents survived into generation F53, whereas some infrequent alleles reached high frequencies. Such changes in frequency indicate that the chromosomal segments marked by the SSR alleles are under the influence of natural selection. The SSR variants allow specific DNA sequences to be followed through generations. Thus, the great resolving power of SSR assays may provide clues regarding the precise targets of natural and man-directed selection.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akkaya M. S., Bhagwat A. A., Cregan P. B. Length polymorphisms of simple sequence repeat DNA in soybean. Genetics. 1992 Dec;132(4):1131–1139. doi: 10.1093/genetics/132.4.1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allard R. W. The Wilhelmine E. Key 1987 invitational lecture. Genetic changes associated with the evolution of adaptedness in cultivated plants and their wild progenitors. J Hered. 1988 Jul-Aug;79(4):225–238. doi: 10.1093/oxfordjournals.jhered.a110503. [DOI] [PubMed] [Google Scholar]
- Allard R. W. The genetics of host-pathogen coevolution: implications for genetic resource conservation. J Hered. 1990 Jan-Feb;81(1):1–6. doi: 10.1093/oxfordjournals.jhered.a110918. [DOI] [PubMed] [Google Scholar]
- Condit R., Hubbell S. P. Abundance and DNA sequence of two-base repeat regions in tropical tree genomes. Genome. 1991 Feb;34(1):66–71. doi: 10.1139/g91-011. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards A., Civitello A., Hammond H. A., Caskey C. T. DNA typing and genetic mapping with trimeric and tetrameric tandem repeats. Am J Hum Genet. 1991 Oct;49(4):746–756. [PMC free article] [PubMed] [Google Scholar]
- Hansen L., von Wettstein-Knowles P. The barley genes Acl1 and Acl3 encoding acyl carrier proteins I and III are located on different chromosomes. Mol Gen Genet. 1991 Oct;229(3):467–478. doi: 10.1007/BF00267471. [DOI] [PubMed] [Google Scholar]
- Litt M., Luty J. A. A hypervariable microsatellite revealed by in vitro amplification of a dinucleotide repeat within the cardiac muscle actin gene. Am J Hum Genet. 1989 Mar;44(3):397–401. [PMC free article] [PubMed] [Google Scholar]
- Morgante M., Olivieri A. M. PCR-amplified microsatellites as markers in plant genetics. Plant J. 1993 Jan;3(1):175–182. [PubMed] [Google Scholar]
- Neale D. B., Saghai-Maroof M. A., Allard R. W., Zhang Q., Jorgensen R. A. Chloroplast DNA diversity in populations of wild and cultivated barley. Genetics. 1988 Dec;120(4):1105–1110. doi: 10.1093/genetics/120.4.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohde W., Becker D., Salamini F. Structural analysis of the waxy locus from Hordeum vulgare. Nucleic Acids Res. 1988 Jul 25;16(14B):7185–7186. doi: 10.1093/nar/16.14.7185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rundle S. J., Zielinski R. E. Organization and expression of two tandemly oriented genes encoding ribulosebisphosphate carboxylase/oxygenase activase in barley. J Biol Chem. 1991 Mar 15;266(8):4677–4685. [PubMed] [Google Scholar]
- Saghai Maroof M. A., Allard R. W., Zhang Q. F. Genetic diversity and ecogeographical differentiation among ribosomal DNA alleles in wild and cultivated barley. Proc Natl Acad Sci U S A. 1990 Nov;87(21):8486–8490. doi: 10.1073/pnas.87.21.8486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saghai-Maroof M. A., Soliman K. M., Jorgensen R. A., Allard R. W. Ribosomal DNA spacer-length polymorphisms in barley: mendelian inheritance, chromosomal location, and population dynamics. Proc Natl Acad Sci U S A. 1984 Dec;81(24):8014–8018. doi: 10.1073/pnas.81.24.8014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stallings R. L., Ford A. F., Nelson D., Torney D. C., Hildebrand C. E., Moyzis R. K. Evolution and distribution of (GT)n repetitive sequences in mammalian genomes. Genomics. 1991 Jul;10(3):807–815. doi: 10.1016/0888-7543(91)90467-s. [DOI] [PubMed] [Google Scholar]
- Tautz D. Hypervariability of simple sequences as a general source for polymorphic DNA markers. Nucleic Acids Res. 1989 Aug 25;17(16):6463–6471. doi: 10.1093/nar/17.16.6463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber J. L. Informativeness of human (dC-dA)n.(dG-dT)n polymorphisms. Genomics. 1990 Aug;7(4):524–530. doi: 10.1016/0888-7543(90)90195-z. [DOI] [PubMed] [Google Scholar]
- Weissenbach J., Gyapay G., Dib C., Vignal A., Morissette J., Millasseau P., Vaysseix G., Lathrop M. A second-generation linkage map of the human genome. Nature. 1992 Oct 29;359(6398):794–801. doi: 10.1038/359794a0. [DOI] [PubMed] [Google Scholar]
- Wu K. S., Tanksley S. D. Abundance, polymorphism and genetic mapping of microsatellites in rice. Mol Gen Genet. 1993 Oct;241(1-2):225–235. doi: 10.1007/BF00280220. [DOI] [PubMed] [Google Scholar]
- Zhang Q., Maroof M. A., Kleinhofs A. Comparative diversity analysis of RFLPs and isozymes within and among populations of Hordeum vulgare ssp. spontaneum. Genetics. 1993 Jul;134(3):909–916. doi: 10.1093/genetics/134.3.909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X., Kochert G. Phylogenetic distribution and genetic mapping of a (GGC)n microsatellite from rice (Oryza sativa L.). Plant Mol Biol. 1993 Feb;21(4):607–614. doi: 10.1007/BF00014544. [DOI] [PubMed] [Google Scholar]