Abstract
To date, no compounds or interventions exist that treat or prevent sarcomeric cardiomyopathies. Established therapies currently improve the outcome, but novel therapies may be able to more fundamentally affect the disease process and course. Investigations of the pathomechanisms are generating molecular insights that can be useful for the design of novel specific drugs suitable for clinical use. As perturbations in the heart are stage-specific, proper timing of drug treatment is essential to prevent initiation and progression of cardiac disease in mutation carrier individuals. In this review, we emphasize potential novel therapies which may prevent, delay, or even reverse hypertrophic cardiomyopathy caused by sarcomeric gene mutations. These include corrections of genetic defects, altered sarcomere function, perturbations in intracellular ion homeostasis, and impaired myocardial energetics.
Keywords: Hypertrophic cardiomyopathy, Dilated cardiomyopathy, Gene therapy, Ion channels, Energetics and microvasculature
1. Current state of therapeutic approaches in sarcomeric cardiomyopathies
Prior to the landmark discoveries that hypertrophic cardiomyopathy [HCM; and later dilated cardiomyopathy (DCM)] was caused by mutations in components of the cardiac sarcomere, clinical management was entirely symptom-based. In fact, the current AHA/ACC HCM guidelines released in 2011 still clearly state that direct therapeutic intervention should be focused on symptom relief, not the diagnosis per se.1 Likewise, the recently published 2014 ESC guidelines on HCM management describe drug therapies to manage symptoms and complications.2 Although this is a well-supported approach that decreases patient morbidity, it is, by nature, palliative in that it does not alter the natural history of the progressive cardiovascular remodelling that defines HCM/DCM. Beta-blockers, calcium channel blockers, and disopyramide are used to optimize haemodynamics by modulating the effects of existing left ventricular (LV) dysfunction in HCM, whereas patients with inherited DCM are managed with standard heart failure regimens. Prior to the ‘genetic era’, this was a reasonable approach because patients were not identified before the onset of symptoms, symptoms that were directly caused by significant pathogenic LV remodelling. Due to the widespread use of modern DNA sequencing techniques to perform mutational screening among the relatives of cardiomyopathy patients, the cohorts of genotype-positive individuals (mutation carriers) without disease expression have dramatically increased in recent years.3 The growing cohorts of mutation carriers who do not yet show cardiac remodelling have changed our view of the clinical syndrome from a disease of ‘thick or thin hearts’ to one of a complex longitudinal process that is often defined by both a distinct preclinical phase and a later stage of remodelling. This modern view of sarcomeric cardiomyopathies coupled with our advanced understanding of the molecular and cellular mechanisms that underlie disease pathogenesis sets the stage for advanced, targeted therapeutics to alter the natural history of sarcomeric cardiomyopathies, before end-stage irreversible remodelling occurs. Thus, in the following section, novel and specific approaches are addressed to the management of sarcomeric cardiomyopathies, with an emphasis on HCM (Table 1). Novel therapies to prevent onset and progression of sarcomeric cardiomyopathies may target the disease-causing gene directly or intervene with cellular pathomechanims that play a role in the (early) progression of cardiomyopathy, such as alterations of myofilament Ca2+ sensitivity, ion channel remodelling, perturbations in energetics, and microvascular dysfunction.
Table 1.
Therapies investigated in preclinical and clinical studies
| Defect | Potential target | Potential therapies |
|---|---|---|
| Sarcomeric proteins | ||
| Myofilament Ca2+ sensitivity |
Thin filament (troponin C) | Ca2+ desensitizers Blebbistatin6 Calmodulin antagonists7–9 Green tea10 β-blocker (nebivolol)a17 |
| Actin–myosin interaction | Actin–myosin interaction inhibitors Blebbistatin, 2,3-butanedione monoxime6 |
|
| Myosin activity | Myosin heavy chain | Small molecules Myosin inhibitors Myosin activators (omecamtiv mecarbil)28 |
| Gene mutation | Mutant genes | Gene therapy Trans-splicing54 mRNA silencing29,30 Gene replacement31 |
| Ion channels | ||
| LVOT obstruction Cardiac remodelling |
Ca2+/CaMKII | CaMKII inhibition Small molecules—CaMKII inhibitors88 L-type Ca2+ channel blocker (diltiazem),91,103 ongoing clinical trial in preclinical HCM104 INaL inhibition (ranolazine) |
| Na+ channel | Sodium channel blocker (disopyramide)a70–74 | |
| Ca2+ channel | Ca2+ channel blockers (verapamil and diltiazem)a75–77 | |
| Arrhythmias | K+ channel Late Na+ current |
Class III antiarrhythmic agent (amiodarone)a1,79 INaL inhibitor (ranolazine),60 ongoing clinical trial: ESTYLE-HCM; EUDRA-CT 2011-004507-20 |
| Diastolic dysfunction | Late Na+ current | INaL inhibitor (ranolazine)60 |
| Energy deficiency | ||
| Vascular dysfunction | LVOT obstruction and regional perfusion defects | β-Blockers and Ca2+ channel blocker (verapamil)a148 Ca2+ desensitizers (see above) |
| Disturbed energetics | Metabolic substrate modification | Metabolic therapy (perhexiline)151 |
Already used in clinic.
2. Targeting sarcomeric proteins
2.1. Modulation of myofilament Ca2+ sensitivity via the thin filament
HCM-associated mutations in myofilament proteins have been associated with increased myofilament Ca2+ sensitivity, whereas DCM-associated mutations have been proposed to underlie reduced sensitivity of myofilaments to Ca2+.4,5 Although the direct effect of sarcomeric mutations on myofilament Ca2+ sensitivity changes is under discussion, increased Ca2+ sensitivity seems to be a common factor in HCM due to secondary myofilament remodelling (e.g. due to altered disease-related phosphorylation patterns). The increases in the myofilament response to Ca2+ may contribute to impaired relaxation and diastolic dysfunction. Although the mechanisms responsible for increased myofilament Ca2+ sensitivity remain unclear, the use of Ca2+-desensitizing interventions (Table 1) may be an attractive alternative for the treatment of sarcomeric cardiomyopathies and alleviation of the disease-related symptoms (Figure 1). Ideally, Ca2+ desensitizers, by targeting specifically myofilament molecules involved in muscle contraction rather than the membrane-bound Ca2+-handling molecules, would avoid altering cytosolic Ca2+ homeostasis which would perturb the regulation of other Ca2+-based signalling pathways. Ca2+ desensitizers may also have the potential ability to prevent arrhythmias in HCM patients. This therapeutic advantage of compounds that target sarcomere Ca2+ sensitivity was first demonstrated in mouse models characterized by myofilament hypersensitivity to Ca2+ caused by troponin mutations or by the Ca2+-sensitizing agent EMD 57033.6 In vitro cardiac muscle from these animal models exhibited significant arrhythmia susceptibility that was prevented by the myosin inhibitor blebbistatin.6 The protective effect of blebbistatin provided the first direct evidence that myofilament Ca2+ desensitization is antiarrhythmic and may be beneficial in the treatment of HCM. The use of Ca2+-desensitizing compounds for the treatment of diastolic dysfunction is practically a novel idea. So far, the number of Ca2+-desensitizing interventions available for research, medical trials, or therapeutic use is very limited. Most of them are at present unsuitable for therapeutic use and can be only tested in animal models and in in vitro experiments as ‘proofs of concept’. Investigations of the mechanisms of Ca2+-desensitizing interventions are generating molecular insights into structural features that can be useful for the design of novel specific Ca2+-desensitizing drugs.
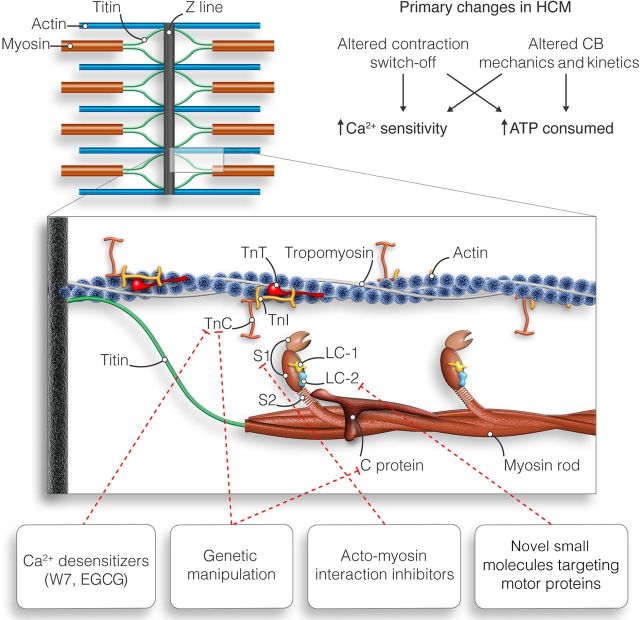
Figure 1.
Several myofilament proteins may be target of treatment to reverse primary functional changes of the sarcomeres. CB, cross-bridge; TnC, troponin C; TnI, troponin I; TnT, troponin T; LC, light chain; C protein, myosin-binding protein C. Modified from Ferrantini et al.154
Due to its central role as the Ca2+ sensor for cardiac muscle contraction, cardiac troponin C (cTnC) stands out as the obvious target to modulate cardiac muscle Ca2+ sensitivity (Figure 1). Unfortunately, there are only a few compounds that only target cTnC to decrease sarcomere Ca2+ sensitivity. An alternative approach is to use cTnC as a target of genetic manipulation. The intrinsic Ca2+-binding properties of cTnC can be finely or grossly tuned to design cTnC mutant constructs, which behave as Ca2+ desensitizers in solution systems and in isolated muscle models. Compounds have been identified that elicit their activity through binding either the N-terminal regulatory domain or the C-terminal structural domain of cTnC. Both groups of compounds likely interfere with the Ca2+-dependent interaction between cTnC and cardiac troponin I (cTnI) that is crucial in the signalling of muscle contraction. Because of the structural homology between cTnC and calmodulin (CaM), CaM-binding compounds originally developed as inhibitors of CaM function may also interact with cTnC and be candidates as modulators of cardiac myofilament Ca2+ sensitivity (Table 1). Early studies have shown that some hydrophobic CaM antagonists (calmidazolium, bepridil, trifluoperazine, chlorpromazine, and pimozide) stimulate myofibrillar ATPase activity, whereas others (W7, haloperidol, and mastoparan) inhibit ATPase activity.7 Among the compounds of the latter group, W7 has been studied more extensively as a potential Ca2+ desensitizer of striated muscle myofilaments. W7 [N-(6-aminohexyl)-5-chloro-1-naphthalenesulfinamide] was designed as a specific inhibitor of CaM function.8 It binds to both the N- and C-terminal hydrophobic substrate-binding sites of CaM, inhibiting binding of CaM to its myosin light-chain kinase target protein.9 W7 has been used to explore a wide range of physiological processes involving Ca2+ signalling in cardiomyocytes. Previous studies in rabbit skeletal and mouse cardiac muscles established that W7 inhibits force and ATPase during Ca2+ activation in both muscle types by reducing the ability of Ca2+ to activate thin filaments.10 The W7 inhibition is most likely mediated via specific interactions between W7 and cTnC. This notion is supported by the observation that W7 binds specifically to cTnC and not to tropomyosin, actin, or myosin.8 In addition, the possibility that W7 interferes directly with the actin–myosin interaction is unlikely as W7 has no effect on in vitro skeletal acto-myosin ATPase activity over the range of [W7] required for Ca2+-activated ATPase and force inhibition (M. Regnier, personal communication).
A number of studies suggest that consumption of green tea decreases the risk of several pathological conditions. Green tea (Camellia sinensis) contains catechins as biologically active polyphenols. Major catechins in green tea are (−)-epicatechin (EC), (−)-epigallocatechin (EGC), (−)-epicatechin-3-gallate (ECG), and (−)-epigallocatechin-3-gallate (EGCG). ECG and EGCG have been shown to be particularly effective against cardiovascular diseases.11 Epicatechin derivatives were found to significantly decrease pCa50 of force–pCa relationships in skinned ventricular trabeculae from rabbit hearts in a concentration-dependent manner.12 EC and EGC had no significant effects on cardiac myofilament Ca2+ sensitivity, indicating that the galloyl group in ECG and EGCG has a critical role in the Ca2+-desensitizing effects. NMR spectroscopy studies provided strong evidence that cTnC is one of the primary targets for EGCG in the myofilaments.13 The therapeutic effect of EGCG as a Ca2+ desensitizer was analysed in a transgenic mouse model of HCM expressing the TNNT2 ΔE160 deletion mutation.12 The mouse ΔE160 cTnT myocardium exhibited increased myofilament Ca2+-sensitivity. EGCG (30 µM) fully reversed the effects of increased myofilament Ca2+ sensitivity of the isolated HCM myocardium. Lower concentrations of EGCG were enough to improve the diastolic function of working hearts of ΔE160cTnT-Tg mice and increase their cardiac output. EGCG also restored the Ca2+ transient parameters without changing myocardial contractility and improved the diastolic dysfunction without changing the cardiomyocyte resting Ca2+ level. These results suggest that EGCG restores the impaired cardiac pump function due to diastolic dysfunction by reversing the increased myofilament Ca2+ sensitivity. EGCG is the first chemical compound that could ameliorate diastolic dysfunction of HCM, at least partially, through its direct Ca2+-desensitizing effects on cardiac myofilament. The use of EGCG as a therapeutic alternative for cardiac dysfunction is particularly interesting because it is attributed to have several benefits including anti-oxidative,11 anti-inflammatory,14 and vasorelaxant effects15 on the cardiovascular system. Its cardioprotective effects against ischaemia/reperfusion injury have been demonstrated as well.16 The use of transgenic animals will allow determining its relevance for the treatment of HCM and the overall effects of Ca2+ desensitization on diastolic dysfunction.
Among β-blockers that are commonly used in clinical pharmacotherapy of cardiovascular diseases, nebivolol has been reported to desensitize cardiac myofilaments.17 In both rabbit and human skinned cardiac trabeculae, nebivolol depressed maximal tension and displaced the Ca2+-tension relation to the right, whereas neither propranolol nor carvedilol had an effect. Experiments with intact trabeculae confirmed depressed contractility: when all β-adrenoceptors were blocked by propranolol, subsequent addition of nebivolol reduced developed twitch force. The Ca2+-desensitizing effect of nebivolol was related to the beneficial effects on myocardial function reported in situations of oxidative stress associated with intracellular Ca2+ overload. This preservation of contractile function by nebivolol might be due to compensation of the intracellular calcium overload through a shift of the force–Ca2+ relationship into a range where contraction is maintained. The mechanism of the Ca2+-desensitizing effect of nebivolol, however, remains unaddressed.
2.2. Modulation of thick filament function
The actin–myosin interface is also a potential site of action for Ca2+-desensitizing drugs (Figure 1 and Table 1). Myosin ATPase inhibitors such as blebbistatin18 and 2,3-butanedione monoxime have been used as desensitizing compounds in vitro and as excitation–contraction uncouplers for electrophysiological and mechanical studies both in vitro and ex vivo due to their ability to inhibit acto-myosin force-generating cross-bridge formation.6,19,20 As stated above, first evidence of the protective effect of Ca2+ desensitization on arrhythmia susceptibility associated with increased Ca2+ sensitivity has been given with actin–myosin interaction inhibitors.6 However, these compounds characterized by strong negative inotropic effects and cardiac toxicities are at present unsuitable for use in intact animals. There are also accessory proteins in the thick filaments that modify the actin–myosin interaction, but have not been explicitly investigated as targets of Ca2+-desensitizing agents. These include the essential and regulatory light chains of myosin and cardiac myosin-binding protein C. Both groups of proteins likely regulate cross-bridge kinetics, and modifications of their protein–protein interactions may be an additional route to alter Ca2+ sensitivity of force generation.
Studies of systems containing some HCM-mutant myosins imply that the mutant proteins have increased mechanical performance.21–24 Although the precise impact of specific HCM mutations on the maximal force-generating capacity of human cardiac sarcomeres in vivo remains somewhat controversial,25–27 advances in screening methods have enhanced the development of small molecules acting like cardiac myosin inhibitors that could become a resource for developing treatments for diseases involving myosin overactivity. These compounds, as well as other mutation-specific sarcomeric allosteric modulators, could rebalance contractility in HCM, therefore potentially reversing the course of disease.
Myosin activators, on the other hand, are small molecule drugs that bind to the myosin head, and stimulate its activity without increasing the cytosolic Ca2+ concentration. As a result, the systolic ejection time is lengthened. The first molecule omecamtiv mecarbil has been shown to ameliorate cardiac function by increasing the duration of ejection without changing the rates of contraction. Therefore, cardiac myosin activation may provide a new therapeutic approach for sarcomeric cardiomyopathies leading to DCM with systolic dysfunction.11
2.3. Targeting the genetic cause of sarcomeric cardiomyopathy by gene therapy
Another obvious therapy for inherited sarcomeric cardiomyopathy would be to target directly the cause of the disease by gene therapy (Figure 1 and Table 1). In theory, this could be done by removing the mutation at the mRNA level or by replacing endogenous mutant proteins by functional ones. Gene therapy to prevent or rescue the disease phenotype in cardiomyopathy mouse models has emerged in the recent years,29–33 and therefore paved the way for a causal therapy of sarcomeric cardiomyopathy in patients. Gene therapy is of particular interest for the severe forms of the disease which result in systolic heart failure and premature death, for which no treatments except heart transplantation are available. A growing body of evidence indicates that severe cardiomyopathies are due to the presence of double heterozygous, compound heterozygous, or homozygous mutations in sarcomeric genes.34–47 Specifically, all infants with truncating bi-allelic MYBPC3 mutations (expected to result in low level or the absence of cMyBP-C in the cardiac sarcomere) present at birth with neonatal cardiomyopathy (HCM, DCM, or LV non-compaction), which rapidly evolves into systolic heart failure and death within the first year of life.34–41,43,48
Gene therapy, a concept introduced as early as 1947,49 has been rediscovered in the last decade with the development of adeno-associated viral vectors (AAVs) exhibiting high-efficiency and long-lasting cardiac gene expression following a single administration. This has accelerated the field of gene therapy for heart failure, targeting proteins involved in calcium handling such as phospholamban50 and S100A1.51 The successful completion of Phase II trials of SERCA2a gene therapy demonstrated the feasibility and safety of AAV1-mediated gene transfer as well as the improvement of the symptoms and exercise capacity of patients with advanced heart failure.52 Using a combination of cardiac AAV serotype and cardiomyocyte-specific promoter, it is now possible to specifically target the heart after systemic administration.53
Different approaches have been evaluated in the context of HCM. The first strategies target the endogenous mutant sarcomeric pre-mRNA or RNA such as exon skipping, spliceosome-mediated RNA trans-splicing (SMaRT), or RNA silencing.29–31,54 In SMaRT, two independently transcribed RNA molecules, a target mutant pre-mRNA and a therapeutic pre-trans-splicing molecule (delivered by AAV), are spliced together (for a detailed review, see Wally et al.55). As a result, a full-length repaired mRNA is formed. Trans-splicing has the potential to treat autosomal-dominant diseases by repairing a mutant pre-mRNA. A recent proof-of-concept study demonstrated that 5′-trans-splicing repaired Mybpc3 mRNA in cardiac myocytes and in vivo in homozygous Mybpc3-targeted knock-in (Mybpc3-KI) mice, even if the efficiency was not sufficient to prevent the disease phenotype.54 An alternative approach is the in-frame skipping of mutated exons by antisense oligonucleotides (AONs), which mask exonic splicing enhancer motifs and therefore prevent the binding of regulatory splicing proteins that mediate exons inclusion into the mature mRNA.56,57 The resulting proteins, which are internally deleted of a small part, should remain functional. This strategy has been recently evaluated in Mybpc3-KI mice, using two AONs that were expected to produce an in-frame deletion of two exons. AONs were introduced in tandem into U7 small nuclear RNA and packaged in AAV9. A single systemic administration of AAV9 in newborn Mybpc3-KI mice produced a stable functional protein and transiently prevented the cardiac disease phenotype.30 More recently, allele-specific silencing of mutant Myh6 mRNA by AAV-mediated RNAi delivery delayed the expression of the disease phenotype (induced by cyclosporine) in heterozygous Myh6-KI mice.29
The alternative strategy consists of adding a functional sarcomeric protein in place in the sarcomere. Although this has been widely used to create transgenic animals,58 only two groups have envisioned this approach for sarcomeric cardiomyopathies in mouse models.31,32 The sarcomere is indeed a tightly regulated system in which the stoichiometry of all components is preserved. Therefore, additional expression of any sarcomeric protein is expected to replace in part or completely the endogenous protein level in the sarcomere. This approach is particularly attractive for MYBPC3 mutations that result in low level or the absence of mutant proteins. A very recent study has evaluated whether replacement of endogenous mutant cMyBP-C by exogenous wild-type cMyBP-C could prevent the cardiac phenotype in homozygous Mybpc3-KI mice, which genetically mimic the situation of the severe forms in patients.31 A single systemic injection of AAV9 encoding cMyBP-C under the control of human cardiac troponin T promoter in neonatal mice, which do not exhibit a disease phenotype at this stage, was sufficient to enable a long-term prevention (until 34 weeks) of the disease phenotype, including LV hypertrophy, diastolic, and systolic dysfunction. Importantly, these data show that endogenous mutant cMyBP-C proteins are almost fully replaced by exogenous functional cMyBP-C. These data paved the way for a causal therapy of severe neonatal sarcomeric cardiomyopathy due to bi-allelic MYBPC3 mutations.
3. Targeting ion channels in HCM
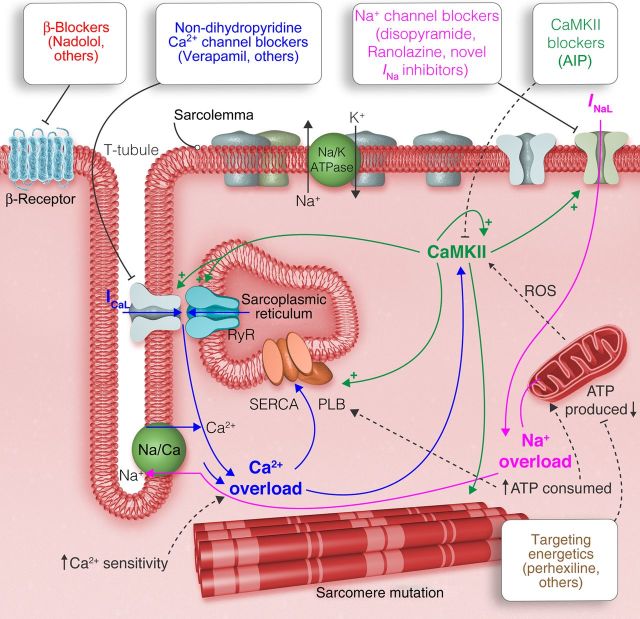
Drugs targeting ion channels are an important component of pharmacological therapy for many HCM patients (Table 1).59 To better understand how drugs affect the pathophysiological determinants of symptoms and arrhythmias in HCM patients, it is useful to know how this disease alters excitation–contraction coupling (ECC) in adult ventricular cardiomyocytes. A recent study characterized the abnormalities of ECC occurring in the human HCM myocardium as concomitant determinants of diastolic dysfunction and arrhythmias in this disease.60 Myocardial specimens from the interventricular septum of obstructive patients undergoing surgical myectomy were compared with non-failing non-hypertrophic surgical patients. Action potential duration (APD) was markedly prolonged in HCM cardiomyocytes compared with controls and was associated with prolonged QTc in patients from the HCM group, a common feature in patients with this disease.61 APD prolongation in HCM cardiomyocytes led to the increased risk of arrhythmogenic early after depolarizations (EADs) and was caused by an imbalance between inward and outward currents: repolarizing K+ currents were reduced and depolarizing L-type Ca2+ current and late Na+ current (INaL) were increased. HCM cardiomyocytes also displayed several abnormalities of intracellular Ca2+ cycling: while the amplitude of Ca2+ transients was preserved, the kinetics of Ca2+ rise and decay were markedly slower and diastolic Ca2+ concentration was increased, resulting in a higher rate of Ca2+ waves and delayed after depolarizations (DADs). Among the determinants of intracellular Ca2+ overload, a reduced Ca2+ extrusion through the Na+/Ca2+ exchanger (NCX) played a major role. NCX functional abnormalities were attributed to intracellular Na+ accumulation due to excessive Na+ entry via an increase in the late Na+ current. The abnormalities of ECC observed in single cells resulted in slower relaxation and increased diastolic tension in intact trabeculae isolated from the same samples, suggesting that alterations of Ca2+ cycling contribute to diastolic dysfunction in HCM myocardium. On the other hand, systolic function was preserved, with a slower rate of force development and prolonged twitches. Increased activity of Ca2+/CaM-dependent kinase II (CaMKII) appeared to underlie several of the observed functional abnormalities: increased phosphorylation of Na+ channels and enhanced Na+ current (INaL),62,63 increased ryanodine receptor phosphorylation caused Ca2+ waves and DADs,64 and increased phosphorylation of Ca2+ channels slowed Ca2+ current inactivation. Notably, all those changes ultimately lead to increased intracellular Ca2+ concentration, which is a main driver for CaMKII activation, thus creating a vicious cycle. Indeed, this type of positive feedback from Na–Ca–CaMKII back to higher [Na]i and more spontaneous sarcoplasmic reticulum (SR) Ca2+ release may be important in this pathological effect.65 Pharmacological intervention should therefore be aimed at impeding intracellular Ca2+ overload, by inhibiting either CaMKII or one of the other pathways of Ca2+ entry and cytosolic accumulation. The features of maladaptive remodelling in HCM cardiomyocytes, including electrical anomalies, Ca2+ handling abnormalities, and alterations of CaMKII signalling, are similar to other forms of pathological hypertrophy,66,67 and thus represent a potential common pathway of disease, eventually driving the common pathophysiological features occurring in patients with cardiac hypertrophy, namely arrhythmias and diastolic dysfunction. Alterations of ion channels are central in this process, thus drugs targeting ion channels directly (or indirectly via CaMKII inhibition) may play a crucial role in treating hypertrophic remodelling and, in particular, HCM (Figure 2).
Figure 2.
Treatment of sarcomeric cardiomyopathies may be directed at non-sarcomeric targets such as ion channels, β-adrenergic receptors, and Ca2+/CaMKII. In addition, metabolic therapy, such as perhexiline, may be targeted at the mitochondria to improve the energetic status of the heart. Modified from Coppini et al.60
3.1. Targeting ion channels to prevent detrimental effect of LV outflow tract obstruction
A principal determinant of symptoms in overt HCM is obstruction of the LV outflow tract (LVOT), which occurs at rest in approximately one-third of all patients (rest obstruction). The number of patients with LVOT increases during exercise (inducible obstruction) to two-thirds of all patients.68 The generation of a pathological gradient in the LVOT during systole (>30 mmHg) is determined by the increased systolic thickening of the upper septum and systolic motion of the (often elongated) anterior mitral valve leaflet towards the septum. When obstruction is present at rest, LVOT gradients lead to a reduction of cardiac output and congestive symptoms (e.g. dyspnoea). In patients with inducible obstruction, the generation of gradients during exertion limits exercise capacity. In symptomatic HCM individuals without obstruction, diastolic dysfunction and myocardial perfusion abnormalities determine reduced exercise capacity and angina.69 Besides β-blockers, disopyramide, a Class Ia sodium channel blocker, has been used largely as a first-line therapy for obstruction for more than 30 years70 and received a Class IIa recommendation in the latest HCM guidelines.1 Disopyramide was shown to reduce LVOT gradients and ameliorate obstructive symptoms in two-thirds of treated patients, with a significant benefit on survival and no apparent pro-arrhythmic effects when used properly.71 However, a significant number of patients cannot tolerate the drug due to its anti-cholinergic effects and the significant reduction of cardiac contractility, leading to a decreased ejection fraction (10% on average).72 Cellular mechanisms underlying the negative inotropic effect of disopyramide are poorly understood at present. At therapeutic concentrations (5–10 µM), disopyramide is known to reduce the upstroke velocity and the amplitude of action potentials,73 and may thus diminish or delay ECC and slowing septal contraction. Moreover, the reduction of intracellular Na+ is likely to limit Ca2+ levels and contractility via enhancement of Ca2+ extrusion and reduction of Ca2+ entry through the NCX. In HCM patients, these effects may result in a slower and reduced force generation by the septum, lower blood flow acceleration in the LVOT, diminished hydrodynamic pull on the mitral leaflet, and reduced mitral–septal contact, eventually leading to reduced outflow gradients.74
Non-dihydropyridine Ca2+ channel blockers such as verapamil and diltiazem are also commonly employed in symptomatic patients with non-obstructive HCM. On the contrary, HCM guidelines stand against the use of Ca2+ channel blockers in patients with LVOT obstruction and high gradients at rest,1 due to the risk of severe hypotension, bradycardia, and syncope mediated by the mild systemic vasodilation. Nonetheless, these drugs may be indicated in combination with β-blockers for patients with inducible obstruction and mild gradients. Verapamil is the most studied agent in HCM patients, despite a lack of definitive evidence that Ca2+ channel blockers ameliorate exercise capacity in non-obstructive symptomatic patients.1 Verapamil and diltiazem exert their beneficial effects on HCM-related symptoms in part through their negative inotropic and chronotropic effects and in part via improvement of myocardial diastolic function. Reduction of heart rate is mediated by a direct effect on Ca2+ current in sinoatrial cells and leads to prolongation of relaxation time. Reduced Ca2+ entry into ventricular myocytes determines a reduction in peak force with no effect on the speed of force generation, eventually causing a negative inotropic effect, that may be effective against obstruction.75 In addition, relaxation time of ventricular myocardium is reduced by Ca2+ channel blockers with a significant increase of LV early filling rates.76 The latter is likely to be a consequence of the decreased intracellular diastolic Ca2+, leading to reduced diastolic tension. Since coronary perfusion occurs predominantly during diastole, the reduction of diastolic tension after Ca2+ channel blocker administration leads to increased myocardial blood flow,77 preventing exercise-induced myocardial ischaemia, a common occurrence in HCM patients.
3.2. Targeting ion channels to prevent arrhythmias
While the presence of LVOT strongly affects the symptomatic state of HCM patients with a largely ‘stable’ phenotype, atrial and ventricular arrhythmias are the main determinant of the outcome and need to be addressed by aggressive preventive strategies, which to date are both insufficient and difficult to administer.78 Amiodarone is the most used antiarrhythmic agent to prevent ventricular tachycardia and fibrillation in high-risk HCM patients.1,79 As a Class III antiarrhythmic agent, the principal mode of action is the inhibition of rapid and slow delayed rectifier potassium channels (IKr), leading to delayed repolarization. This could represent a potential risk in HCM patients, who already suffer from a prolonged QT interval due to APD prolongation.60,61 However, amiodarone exerts a number of additional effects, including block of L-type Ca2+ current, peak and late Na+ currents; counteracting the AP prolonging effect of the compound and effectively antagonizing the pathological arrhythmogenic changes occurring in HCM myocytes, making it a potentially effective agent to prevent arrhythmias in this disorder. Nonetheless, clinical evidence suggests that amiodarone used for primary prevention does not exert a significant benefit in terms of survival,80 and is to be used only in patients with frequent symptomatic ventricular arrhythmias or to decrease the frequency of effective shocks in patients with implantable cardioverter/defibrillators.1 On the other hand, amiodarone is the most commonly used antiarrhythmic drug to maintain sinus rhythm in HCM patients with paroxysmal atrial fibrillation.
The previous observations emphasize the lack of effective pharmacological agents capable of ameliorating diastolic dysfunction and reducing ventricular arrhythmogenesis in HCM. The inhibitors of INaL may fill this gap. The first drug of this category, ranolazine, is the only commercially available INaL inhibitor, currently in use for the treatment of angina. The molecular effects of ranolazine were recently evaluated in surgical myocardial samples from HCM patients.60 Consistent with inhibition of the depolarizing INaL, ranolazine significantly reduced the prolonged APD in HCM myocytes, thus nearly abolishing the occurrence of EADs. This effect was paralleled by a marked acceleration of Ca2+ transients and a reduction of diastolic Ca2+ concentration, the latter more evident at high frequency of stimulation. Reduced intracellular Na+ accumulation leading to increased Ca2+ extrusion through the NCX underlie the beneficial effects of ranolazine on Ca2+ handling in HCM cardiomyocytes. In addition, normalization of Ca2+ cycling properties with ranolazine causes a mild reduction of systolic force, an acceleration of twitch force generation and relaxation, as well as a reduction of diastolic tension in HCM trabeculae, supporting a potential improvement of diastolic function. Taken together, these observations suggest that inhibition of INaL may exert a number of significant beneficial effects in HCM patients, by reducing both the arrhythmogenic potential and the intrinsic impairment of diastolic function at the cellular level. These features strongly support the use of INaL inhibitors to treat symptomatic HCM patients without obstruction. Of note, a clinical trial is ongoing, evaluating the effect of ranolazine on exercise capacity in this class of patients (RESTYLE-HCM; EUDRA-CT 2011-004507-20). A small proof-of-concept placebo-controlled clinical trial in patients with severe diastolic heart failure (RALI-DHF) recently suggested that filling pressures (LVEDP), pulmonary artery pressures, and wedge pressures could be reduced by ranolazine.81 Additionally, the slight negative inotropic effect of ranolazine, far from representing a concern for clinical use, may provide a safe option aimed at reducing septal hypercontractility and thus may relieve obstruction in HCM patients. Finally, by improving diastolic function, INaL inhibition has the potential to increase myocardial perfusion, thus addressing myocardial ischaemia in HCM. Recently, novel highly selective INaL inhibitors82,83 are in early clinical stages of development and may represent a valid alternative for HCM treatment in the near future.
3.3. Targeting ion channels to prevent adverse remodelling in manifest HCM
Studies on several models of hypertrophy highlighted alterations of intracellular Ca2+ handling, leading to intracellular Ca2+ overload as central determinants of pathological cardiomyocyte remodelling, acting via a number of signalling pathways, among which the CaMKII-dependent cascade plays a central role.84 CaMKII hyperactivation during disease is associated with activation of hypertrophic gene expression programme, changes in ion channel or SR protein levels, and may play a role in enhancing fibroblast growth and extracellular matrix expansion.85,86 With these mechanisms, CaMKII hyperactivation is likely to play a crucial role in driving progression of cardiac hypertrophy to heart failure.87 Sustained activation of CaMKII-dependent pathways play a central role in determining electro-mechanical myocardial dysfunction in human HCM,60 and may therefore be a highly relevant target for progression. Direct inhibition of cardiac CaMKII with small molecules is still in early preclinical development;88 therefore, the best option to date is to indirectly reduce CaMKII activity by lowering intracellular Ca2+ levels. Inhibition of Ca2+ current appears to be the most straightforward way of reducing intracellular Ca2+, as the amount of Ca2+ entering the cytosol via the L-type Ca2+ channels directly modulates CaMKII activity.89 Early reports on the use of L-type Ca2+ channel blockers in HCM patients showed a reduction of cardiac mass upon long-term administration.90 Treatment with diltiazem prevented worsening of diastolic dysfunction and limited progression to diastolic heart failure in HCM Tnnt2 mutant transgenic mice.91 At present, there is no evidence on whether treatment with L-type Ca2+ channel blockers is able to reduce CaMKII activation and alter cardiomyocyte remodelling in HCM.
A clinically relevant therapeutic option to address intracellular Ca2+ overload and reduce CaMKII activity in HCM is pharmacological inhibition of INaL. In pathological settings, there is continuous interplay among CaMKII, intracellular Ca2+, and INaL. On the one hand, enhanced CaMKII activity due to increased intracellular Ca2+ or oxidative stress increases INaL via specific phosphorylation of cardiac Na+ channel Nav1.5;63,92,93 on the other hand, increased INaL determines elevated intracellular Ca2+ and thus activates CaMKII.94 Such complex interplay is relevant for the progression of diastolic dysfunction in cardiac disease and may play a role in favouring decompensation of stable hypertrophy.95 INaL inhibition may therefore be a viable option for interrupting the CaMKII-dependent remodelling pathway in HCM. In support of this hypothesis, acute treatment with ranolazine led to a reduction of diastolic Ca2+ levels in human HCM cardiomyocytes.60 Over time, this effect may lead to overall lower CaMKII activity, eventually affecting the functional and structural remodelling of HCM myocardium, with possible implications for disease progression.96 In keeping with this observation, ranolazine administration has been shown to reduce the degree of myocyte hypertrophy and interstitial fibrosis in experimental models with moderate heart failure.97 In principle, INaL inhibition is a promising therapeutic strategy for HCM patients, with a wide range of potentially positive actions, which may critically impact on acute symptoms as well as on the natural history of the disease.
3.4. Targeting ion channels to prevent disease progression in mutation carriers
During the so-called pre-hypertrophic phase of HCM, periodic non-invasive cardiac screening is performed to identify early markers of disease, such as mitral valve abnormalities or a mild impairment of diastolic function.98,99 However, no clinical strategy exists to prevent disease progression in mutation carriers. While the primary disease cause is the gene mutation of a sarcomeric protein, alterations in intracellular Ca2+ handling are among the earliest secondary changes occurring in HCM myocardium, as confirmed by studies in transgenic or targeted mouse models.100–102 Intracellular Ca2+ overload may be present in the pre-hypertrophic phase of the disease. Thus, reducing intracellular Ca2+ in this critical phase is likely to affect phenotype presentation. This hypothesis has been tested by treating transgenic mice carrying the R403Q myosin heavy-chain mutation with diltiazem since birth.103 Ca2+ channel block, by diminishing intracellular Ca2+ overload, was able to reduce the development of hypertrophy, intramyocardial fibrosis, and myocyte disarray, to prevent pathological changes of SR protein expression, and to limit the extent of diastolic and systolic dysfunction in the adult mice. Such changes may well be mediated by alteration of CaMKII-mediated signalling. Following this intriguing preclinical evidence, an ongoing study is testing the hypothesis that diltiazem may prevent the development of the HCM phenotype in mutation carriers.104
In the presence of increased CaMKII activity, INaL is increased by CaMKII-dependent phosphorylation. An increase of INaL may also be an early change in the preclinical phase. Similar to diltiazem, INaL inhibitors such as ranolazine may lead to a sustained reduction of intracellular Ca2+, thus impacting on the signalling pathway leading to hypertrophy, tissue remodelling, and, eventually, electrical and mechanical dysfunction of affected myocardium. Whether INaL inhibition is able to prevent or significantly delay the onset of phenotype in mutation carriers remains to be assessed and deserves preclinical investigation. This is of particular interest since ranolazine has an improved profile when compared with diltiazem and may therefore become an important therapeutic option to prevent disease development in young individuals carrying high-risk mutations.
4. Targeting vascular dysfunction and energetics
Cellular ‘energy deficiency’ is a prominent feature of HCM,105 but how sarcomeric mutations cause this deficiency at the whole organ level is not clearly understood. Increased energy demand and decreased energy supply both likely contribute to the energy deprivation. Many HCM mutations increase ‘tension cost’,106,107 meaning the amount of ATP necessary for a unit of work is elevated, thus directly increasing energy demand. There is also evidence that energy supply may be compromised due to changes in substrate utilization or mitochondrial dysfunction.108 At the cardiac level, insufficient coronary perfusion, whether related to epicardial or microvascular abnormalities (i.e. vessels of <400 μm), will limit oxygen supply and exacerbate the primary cellular energy privation related to sarcomeric mutations.
4.1. Causes and consequences of primary energy deficiency in HCM
Energy deficiency, specifically resulting from inefficient generation of contractile force (e.g. requiring more ATP per pN of force generated), remains an enduring primary biophysical consequence of HCM sarcomeric mutations.5 Sophisticated techniques interrogating human HCM samples have confirmed this observation for the R403Q mutation in MYH7.106 Moreover, many forms of LV hypertrophy (LVH), whether inherited or acquired, are increasingly recognized to exhibit impaired myocardial perfusion reserve and oxygenation that are likely to contribute to the derangement in myocardial energetics.109 In HCM, the biophysically driven cellular energy deficiency is a primary feature of disease rather than the consequence of hypertrophy,110 as deduced from the observation that energy deficiency is manifest very early in the course of disease.111 The cumulative consequence of adaptations, ranging from those at the cellular level through the level of the myocardium (e.g. myocardial remodelling including the vasculature) to the level of systemic response (e.g. the autonomic response), is likely to extenuate this primary energy deficiency.
The immediate consequences of increased ATP demand are likely to be on intermediary metabolism and Ca2+ kinetics. Increased energy demand has a direct influence on metabolic pathway fluxes. Normally, energy supply and its influence on intermediary metabolism is exquisitely spatially regulated via the creatinine kinase (CK) system, reducing the temporal delays resulting from compartmentalization of cellular activities, and globally via AMP-activated protein kinase (AMPK). Threatened energy deficiency (i.e. increased ADP) is transmitted by the CK system, activating AMPK, the cellular energy gauge, increasing ATP production, and mitigating energy-consuming activities. These systems are sufficiently robust to ensure that even in advanced heart failure, myocardial ATP levels rarely fall below 75% of the levels seen in normal hearts. As a corollary, mutations in the γ2 subunit of AMPK, which mimic an energy deficiency signal, are a cause for HCM.112 Energy deprivation triggers cellular hypertrophy, cell death, and replacement fibrosis, which likely contributes to the microvascular remodelling described below. Delineating the contribution of these processes on overall energy deficiency and HCM progression remains an active research goal.113 The mechanisms culminating in LVH in HCM remain obscure. While it seems intuitive to attribute LVH to the consequences of energy deficiency on Ca2+ kinetics, perhaps via the calcineurin pathway, there is little to support this hypothesis.
Hypertrophy per se is characterized by changes in myocardial metabolism including a switch from fatty acid to carbohydrate utilization. Although this substrate shift is generally considered to be beneficial by virtue of the oxygen sparing effects of carbohydrate metabolism, there is an increasing recognition that increased carbohydrate metabolism is accompanied by a shift towards anaplerotic flux which contributes to a less energy-efficient myocardium. This substrate shift is driven by activation of the HIF1α–PPARγ axis.114 This intrinsic feature of hypertrophy is compounded by vascular consequences of HCM, including chronic HIF1α activation, perpetuating substrate shifts, and driving changes in Ca2+ handling that are likely to be detrimental.115,116
4.2. Evidence for vascular dysfunction and ischaemia in HCM patients
The occurrence of ischaemia in HCM is a well-recognized clinical phenomenon.117 HCM patients often experience chest pain and dyspnoea, and have an elevated arrhythmia risk, all typical of myocardial ischaemia. Circulating levels of markers of acute ischaemic damage were found to be elevated in some studies.118,119 Histological examinations report infarct-like areas in different stages of healing. Unlike in patients with isolated coronary artery stenosis, the spatial arrangement of scarring is not consistent with perfusion territories of larger coronary arteries, but is patchy with principal involvement of the mid-myocardium.120,121 Several functional studies have observed a diminished coronary flow reserve despite normal epicardial coronary arteries, proposing microvascular dysfunction as an explanation.122,123 The vascular dysfunction was found independent of the structural HCM subtype and occurs in asymmetrical sepal as well as apical hypertrophy.124 Further evidence for ischaemia arises from studies demonstrating hypoperfused myocardial regions during exercise in >50% of patients.125,126 Stress-induced reversible perfusion defects are found most frequently in hypertrophic regions in patients with normal or enhanced LV function.125 However, decreased perfusion is pathological only if tissue oxygenation is compromised; a blunted tissue oxygenation response was recently directly measured during vasodilator stress in HCM mutation carriers.127 The timing of the onset of vascular dysfunction during the disease process is currently unclear. The prevailing notion is that cardiac structural remodelling precedes vascular dysfunction, although no study has demonstrated a clear sequence. Microvascular dysfunction is often spatially associated with tissue remodelling, but it is also found adjacent to patchy fibrosis.47,127 Repeated bouts of ischaemia can induce hypertrophy,128 and fibrosis replaces terminally damaged cardiomyocytes. Thus, it should not be ruled out that vascular dysfunction is an early consequence of the genetic defect, precedes or even promotes hypertrophy and fibrosis, and contributes to the eventual development of ventricular dysfunction.
4.3. Causes for vascular dysfunction in HCM
Although some DCM gene mutations may affect vascular structure and function directly,129 in case of HCM the mutant proteins are almost exclusively expressed in cardiomyocyte. In HCM patients, the capillary density is decreased,130 at least in hypertrophic parts, and the arteriolar lumen of the intramural coronary arteries normalized to wall area is lower,131 partially due to thickening of the medial and intimal layers.132 What promotes this remodelling is currently unclear. In pulmonary arterial hypertension, endothelial cell dysfunction leads to similar remodelling of the pulmonary vasculature,133,134 but there is no obvious link yet found between HCM causing mutations and endothelial cell function.
Diastolic dysfunction during stress is a frequent observation in HCM and is a potentially early consequence of the genetic defect in patient and mouse models.91,102,104 Impaired LV relaxation and the resulting increased filling pressure impinge on diastolic coronary perfusion particularly at faster heart rates as a result of increased extravascular compression.135 Myocardial filling pressures may be further elevated in the presence of LVOT, aggravating extravascular compression particularly in the subendocardial layer. Consistent with this notion, surgical correction of LVOT improves myocardial blood flow reserve.136 Impaired relaxation of the ventricle may also diminish a critical ‘suction wave’ that is generated during normal relaxation, which is a critical driver of diastolic coronary blood flow.137 Other causes for vascular dysfunction in HCM are less frequently discussed, but additional mechanisms likely exist. Sarcomere mutation carriers are characterized by more severe microvascular dysfunction and increased prevalence of myocardial fibrosis when compared with sarcomere non-carriers, suggesting a direct link between sarcomere dysfunction and vascular dysfunction that is not yet understood.138 Anatomical causes that may contribute to ischaemia and angina symptoms include myocardial bridging, which has an increased incidence in HCM139 and coronary vasospasm, which also has been reported in HCM patients. It is conceivable that the metabolic and energetic changes in HCM disturb metabolic coronary flow regulation and manifest in regional vascular dysfunction, which is largely unexplored. It is intriguing that the mechanism whereby the matching of local oxygen demand with supply is continuously maintained is poorly understood.140
To fully understand all factors that contribute to vascular dysfunction in HCM, it will be critical to longitudinally evaluate the time of onset and progression of vascular dysfunction in relation to other manifestations starting in young HCM mutation carriers. It would be also valuable to generate or identify animal models that reproduce the vascular dysfunction and display, e.g. regional perfusion defects. Myosin heavy-chain mutant HCM mice develop focal replacement fibrosis141 and we have recently shown that Tnnt2 mutant HCM mice develop focal energy deprivation during stress, which was linked to increased myofilament Ca2+ sensitivity mediated by the mutation.142 Investigating the underlying causes would be informative.
4.4. Vascular dysfunction and energy deprivation in HCM and prognosis
Vascular dysfunction or the diminished ability to respond to hypoxia appears to contribute to a worse prognosis in HCM mutation carriers. Patients who have concomitant HCM and significant coronary artery disease are at a substantially increased risk of death than comparable patients without HCM.116 A substantial number of HCM patients have increased high-sensitivity cardiac troponin T marker serum concentrations, a circulating marker for ischaemic damage, and the level may predict the adverse outcome.143 HCM patients with certain common variants in hypoxia response genes, which affect the expression of hypoxia-inducible signalling molecules, had more severe hypertrophy and diastolic dysfunction.144 Vascular dysfunction is currently not utilized as a factor to determine risk for lethal arrhythmias, with the exception of an inappropriate drop of systemic blood pressure during exercise.145
To date, there is no evidence relating the degree of energy deficiency to prognosis. Notwithstanding the most parsimonious explanation that the degree of energy deficiency, as a continuous variable, does not contribute to outcomes, a number of other explanations may contribute to this lack of prognostic value. The most compelling is the relative lack of granularity afforded by the most readily available measure of energy deficiency, the PCr/ATP ratio. On an individual patient basis, this parameter, typically measured at rest, is a relatively noisy and insensitive measure even in the best MRI laboratories. The PCr/ATP ratio can be refined either through dynamic measurement146 and/or through the measurement of flux.147 Nevertheless, the confounding effect of cellular adaptation, maintaining the cellular energy charge to maintain the Gibbs free energy of ATP hydrolysis that is so critical to general cellular function, reduces the dynamic range and hence discriminating the value of energy parameters.
4.5. Vascular dysfunction and disturbed energetics as therapeutic targets in HCM
Vascular dysfunction represents a promising potential target for novel HCM therapies. Current pharmacological treatments to relieve LVOT and angina symptoms, β-blockers and Ca2+ channel antagonists, are thought to be largely effective because of the negative chronotropic and inotropic effects, increasing diastolic perfusion time, and reducing extravascular compression. In addition, verapamil has also been demonstrated to prevent regional perfusion defects in a considerable fraction of asymptomatic HCM patients.148 Despite this, current treatment has not been shown to alter the natural history of the disease.149 While this will need to be confirmed in clinical trials, this is intriguing and indicates that either the drugs are given too late and disease has progressed irreversibly, or we still lack fundamental understanding about the disease process. Other strategies to enhance relaxation, e.g. myofilament Ca2+ desensitization (discussed above), may exhibit a more attractive therapeutic profile.150 We know that the coronary microvascular dysfunction importantly contributes, as it is inversely related to death from cardiovascular causes in HCM patients as well as adverse LV remodelling and systolic dysfunction.69 Efforts are underway to test drugs that have benefits in patients with diastolic dysfunction unrelated to HCM. Typically, these drugs also influence vascular causes and consequences, e.g. ranolazine, ACE inhibitors etc., and it is quite possible that these studies will correct the underlying defect or at least point us into the direction we need to investigate.
An alternative strategy might be to target metabolic substrate modification with a goal of altering the state of myocardial oxygenation to augment any primary vascular therapeutic strategies. The advantage of a metabolic approach is that it addresses a common yet proximal cause of myocardial remodelling in HCM. The primary effect of shifting fatty acid to carbohydrate metabolism is the benefit related to oxygen sparing. This is especially germane to a myocardium with compromised perfusion. The benefits of this strategy have been exemplified by success of perhexiline (Figure 2), a partial fatty oxidation inhibitor, in the treatment of symptomatic HCM as well as patients with angina.151 A more nuanced perspective of re-balancing cardiac metabolism (i.e. other than fatty acid vs. carbohydrate) would confer additional benefits. It has been observed that, in LVH, glucose oxidation-derived carbon flux is shifted away from pyruvate dehydrogenase (PDH)-derived acetyl coenzyme A production towards anaplerosis (i.e. the use of glucose-derived carbons to contribute to the backbones of metabolic intermediates). Increased carboxylation of pyruvate via cytosolic malic enzyme generates malate, which ultimately feeds into the citric acid cycle (TCA). It has been argued that this shift away from direct glucose oxidation by PDH is profligate and may represent an excellent therapeutic target.152 While from a stoichiometric perspective increased anaplerosis may appear profligate, increased TCA intermediates may have additional benefits that have hitherto not been considered. We have recently demonstrated that, by stabilizing the transcriptional regulator Nrf2, the TCA intermediate fumarate up-regulates protective antioxidant response element genes.153 These additional consequences of metabolic modification may have profound influences beyond intermediary metabolism pertinent to the consequences of vascular privation.
Finally, it is worth bearing in mind that as well as pharmacological therapies, diverse cardiac interventions such as device therapy (e.g. cardiac resynchronization therapy) may also have an influence on HCM through their impact on intermediary metabolism. Importantly, as with other aspects of HCM as an archetypal cardiac disease, insights derived from metabolic modulation in HCM may, when applied with care, provide valuable lessons for the management of other forms of LVH.
5. Conclusions and future directions
For a not uncommon disorder for which the pharmacological treatment has not appreciably changed in 50 years, the preceding sections represent an exciting look into the future of the management of sarcomeric cardiomyopathies. The ability to identify patients at risk of developing a cardiomyopathy via genetic testing and eventually develop a tailored, mechanistic approach to altering the natural history of the disease process at the molecular level holds profound promise. For example, it is important to note that some subsets of patients eventually require cardiac transplantation, often at a young age. Even the ability to delay the need for transplant for 10–20 years by initiation of targeted therapies in the preclinical stage would represent a significant advance. Moreover, the ability to directly reduce the risk of sudden cardiac death or the onset of atrial fibrillation by mitigating ion channel remodelling would alter the burden of these complex cardiomypathies. While the eventual goal of developing genotype-specific risk assessments and fully targeted therapies still requires a more advanced understanding of early (preclinical) disease pathogenesis, it is clear that our growing ability to subdivide the process of LV remodelling into separate molecular and cellular bins will provide a framework for developing specific treatment regimens that can eventually be applied to specific genetic cohorts and finally alter the natural history of these common disorders as opposed to simply mitigating symptoms.
Conflict of interest: L.S.M. receives speaker's honoraria from Berlin-Chemie.
Funding
L.S.M. acknowledges research grants and funding from CVT, GILEAD, and MENARINI/Berlin-Chemie. L.S.M. is funded by Deutsche Forschungsgemeinschaft (DFG) GRK 1816 RP3, by the DZHK (Deutsches Zentrum für Herz-Kreislauf-Forschung), and the Leducq Foundation. L.C. is supported by grants from the DZHK (German Centre for Cardiovascular Research) and the German Ministry of Research and Education (BMBF), the Leducq Foundation (research grant no. 11, CVD 04), and Association Institut de Myologie (Paris). C.P. is supported by Telethon Italy grant GGP13162. H.A. is supported by British Heart Foundation, including the BHF centre of research excellence and the MRC. J.v.d.V. is supported by the Netherlands organization for Scientific Research (NWO; VIDI grant 91711344) and by a CVON grant (Cardiovasculair Onderzoek Nederland; ARENA consortium). We further acknowledge support from the 7th Framework Program of the European Union (‘BIG-HEART’, grant agreement 241577). J.C.T. is supported by the National Institutes of Health (grants HL075619 and HL107046).
References
- 1.Gersh BJ, Maron BJ, Bonow RO, Dearani JA, Fifer MA, Link MS, Naidu SS, Nishimura RA, Ommen SR, Rakowski H, Seidman CE, Towbin JA, Udelson JE, Yancy CW American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, American Association for Thoracic Surgery, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography, Interventions, Society of Thoracic Surgeons. 2011 ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2011;124:e783–e831. doi: 10.1161/CIR.0b013e318223e2bd. [DOI] [PubMed] [Google Scholar]
- 2.Elliott PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F, Charron P, Hagege AA, Lafont A, Limongelli G, Mahrholdt H, McKenna WJ, Mogensen J, Nihoyannopoulos P, Nistri S, Pieper PG, Pieske B, Rapezzi C, Rutten FH, Tillmanns C, Watkins H. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC) Eur Heart J. 2014;35:2733–2779. doi: 10.1093/eurheartj/ehu284. [DOI] [PubMed] [Google Scholar]
- 3.Maron BJ, Ho CY. Hypertrophic cardiomyopathy without hypertrophy: an emerging pre-clinical subgroup composed of genetically affected family members. JACC Cardiovasc Imaging. 2009;2:65–68. doi: 10.1016/j.jcmg.2008.09.008. [DOI] [PubMed] [Google Scholar]
- 4.Robinson P, Griffiths PJ, Watkins H, Redwood CS. Dilated and hypertrophic cardiomyopathy mutations in troponin and alpha-tropomyosin have opposing effects on the calcium affinity of cardiac thin filaments. Circ Res. 2007;101:1266–1273. doi: 10.1161/CIRCRESAHA.107.156380. [DOI] [PubMed] [Google Scholar]
- 5.Robinson P, Mirza M, Knott A, Abdulrazzak H, Willott R, Marston S, Watkins H, Redwood C. Alterations in thin filament regulation induced by a human cardiac troponin T mutant that causes dilated cardiomyopathy are distinct from those induced by troponin T mutants that cause hypertrophic cardiomyopathy. J Biol Chem. 2002;277:40710–40716. doi: 10.1074/jbc.M203446200. [DOI] [PubMed] [Google Scholar]
- 6.Baudenbacher F, Schober T, Pinto JR, Sidorov VY, Hilliard F, Solaro RJ, Potter JD, Knollmann BC. Myofilament Ca2+ sensitization causes susceptibility to cardiac arrhythmia in mice. J Clin Invest. 2008;118:3893–3903. doi: 10.1172/JCI36642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Silver PJ, Pinto PB, Dachiw J. Modulation of vascular and cardiac contractile protein regulatory mechanisms by calmodulin inhibitors and related compounds. Biochem Pharmacol. 1986;35:2545–2551. doi: 10.1016/0006-2952(86)90052-3. [DOI] [PubMed] [Google Scholar]
- 8.Hidaka H, Yamaki T, Naka M, Tanaka T, Hayashi H, Kobayashi R. Calcium-regulated modulator protein interacting agents inhibit smooth muscle calcium-stimulated protein kinase and ATPase. Mol Pharmacol. 1980;17:66–72. [PubMed] [Google Scholar]
- 9.Osawa M, Swindells MB, Tanikawa J, Tanaka T, Mase T, Furuya T, Ikura M. Solution structure of calmodulin-W-7 complex: the basis of diversity in molecular recognition. J Mol Biol. 1998;276:165–176. doi: 10.1006/jmbi.1997.1524. [DOI] [PubMed] [Google Scholar]
- 10.Adhikari BB, Wang K. Interplay of troponin- and myosin-based pathways of calcium activation in skeletal and cardiac muscle: the use of W7 as an inhibitor of thin filament activation. Biophys J. 2004;86:359–370. doi: 10.1016/S0006-3495(04)74112-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chyu KY, Babbidge SM, Zhao X, Dandillaya R, Rietveld AG, Yano J, Dimayuga P, Cercek B, Shah PK. Differential effects of green tea-derived catechin on developing versus established atherosclerosis in apolipoprotein E-null mice. Circulation. 2004;109:2448–2453. doi: 10.1161/01.CIR.0000128034.70732.C2. [DOI] [PubMed] [Google Scholar]
- 12.Tadano N, Du CK, Yumoto F, Morimoto S, Ohta M, Xie MF, Nagata K, Zhan DY, Lu QW, Miwa Y, Takahashi-Yanaga F, Tanokura M, Ohtsuki I, Sasaguri T. Biological actions of green tea catechins on cardiac troponin C. Br J Pharmacol. 2010;161:1034–1043. doi: 10.1111/j.1476-5381.2010.00942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Robertson IM, Li MX, Sykes BD. Solution structure of human cardiac troponin C in complex with the green tea polyphenol, (−)-epigallocatechin 3-gallate. J Biol Chem. 2009;284:23012–23023. doi: 10.1074/jbc.M109.021352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ludwig A, Lorenz M, Grimbo N, Steinle F, Meiners S, Bartsch C, Stangl K, Baumann G, Stangl V. The tea flavonoid epigallocatechin-3-gallate reduces cytokine-induced VCAM-1 expression and monocyte adhesion to endothelial cells. Biochem Biophys Res Commun. 2004;316:659–665. doi: 10.1016/j.bbrc.2004.02.099. [DOI] [PubMed] [Google Scholar]
- 15.Lorenz M, Wessler S, Follmann E, Michaelis W, Dusterhoft T, Baumann G, Stangl K, Stangl V. A constituent of green tea, epigallocatechin-3-gallate, activates endothelial nitric oxide synthase by a phosphatidylinositol-3-OH-kinase-, cAMP-dependent protein kinase-, and Akt-dependent pathway and leads to endothelial-dependent vasorelaxation. J Biol Chem. 2004;279:6190–6195. doi: 10.1074/jbc.M309114200. [DOI] [PubMed] [Google Scholar]
- 16.Townsend PA, Scarabelli TM, Pasini E, Gitti G, Menegazzi M, Suzuki H, Knight RA, Latchman DS, Stephanou A. Epigallocatechin-3-gallate inhibits STAT-1 activation and protects cardiac myocytes from ischemia/reperfusion-induced apoptosis. FASEB J. 2004;18:1621–1623. doi: 10.1096/fj.04-1716fje. [DOI] [PubMed] [Google Scholar]
- 17.Zeitz O, Rahman A, Hasenfuss G, Janssen PM. Impact of beta-adrenoceptor antagonists on myofilament calcium sensitivity of rabbit and human myocardium. J Cardiovasc Pharmacol. 2000;36:126–131. doi: 10.1097/00005344-200007000-00017. [DOI] [PubMed] [Google Scholar]
- 18.Dou Y, Arlock P, Arner A. Blebbistatin specifically inhibits actin-myosin interaction in mouse cardiac muscle. Am J Physiol Cell Physiol. 2007;293:C1148–C1153. doi: 10.1152/ajpcell.00551.2006. [DOI] [PubMed] [Google Scholar]
- 19.Davis J, Wen H, Edwards T, Metzger JM. Thin filament disinhibition by restrictive cardiomyopathy mutant R193H troponin I induces Ca2+-independent mechanical tone and acute myocyte remodeling. Circ Res. 2007;100:1494–1502. doi: 10.1161/01.RES.0000268412.34364.50. [DOI] [PubMed] [Google Scholar]
- 20.Kettlewell S, Walker NL, Cobbe SM, Burton FL, Smith GL. The electrophysiological and mechanical effects of 2,3-butane-dione monoxime and cytochalasin-D in the Langendorff perfused rabbit heart. Exp Physiol. 2004;89:163–172. doi: 10.1113/expphysiol.2003.026732. [DOI] [PubMed] [Google Scholar]
- 21.Tyska MJ, Hayes E, Giewat M, Seidman CE, Seidman JG, Warshaw DM. Single-molecule mechanics of R403Q cardiac myosin isolated from the mouse model of familial hypertrophic cardiomyopathy. Circ Res. 2000;86:737–744. doi: 10.1161/01.res.86.7.737. [DOI] [PubMed] [Google Scholar]
- 22.Miller G, Maycock J, White E, Peckham M, Calaghan S. Heterologous expression of wild-type and mutant beta-cardiac myosin changes the contractile kinetics of cultured mouse myotubes. J Physiol. 2003;548:167–174. doi: 10.1113/jphysiol.2002.031922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Seebohm B, Matinmehr F, Kohler J, Francino A, Navarro-Lopez F, Perrot A, Ozcelik C, McKenna WJ, Brenner B, Kraft T. Cardiomyopathy mutations reveal variable region of myosin converter as major element of cross-bridge compliance. Biophys J. 2009;97:806–824. doi: 10.1016/j.bpj.2009.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sommese RF, Sung J, Nag S, Sutton S, Deacon JC, Choe E, Leinwand LA, Ruppel K, Spudich JA. Molecular consequences of the R453C hypertrophic cardiomyopathy mutation on human beta-cardiac myosin motor function. Proc Natl Acad Sci USA. 2013;110:12607–12612. doi: 10.1073/pnas.1309493110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Belus A, Piroddi N, Scellini B, Tesi C, D'Amati G, Girolami F, Yacoub M, Cecchi F, Olivotto I, Poggesi C. The familial hypertrophic cardiomyopathy-associated myosin mutation R403Q accelerates tension generation and relaxation of human cardiac myofibrils. J Physiol. 2008;586:3639–3644. doi: 10.1113/jphysiol.2008.155952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Witjas-Paalberends ER, Piroddi N, Stam K, van Dijk SJ, Oliviera VS, Ferrara C, Scellini B, Hazebroek M, ten Cate FJ, van Slegtenhorst M, dos Remedios C, Niessen HW, Tesi C, Stienen GJ, Heymans S, Michels M, Poggesi C, van der Velden J. Mutations in MYH7 reduce the force generating capacity of sarcomeres in human familial hypertrophic cardiomyopathy. Cardiovasc Res. 2013;99:432–441. doi: 10.1093/cvr/cvt119. [DOI] [PubMed] [Google Scholar]
- 27.Kraft T, Witjas-Paalberends ER, Boontje NM, Tripathi S, Brandis A, Montag J, Hodgkinson JL, Francino A, Navarro-Lopez F, Brenner B, Stienen GJ, van der Velden J. Familial hypertrophic cardiomyopathy: functional effects of myosin mutation R723G in cardiomyocytes. J Mol Cell Cardiol. 2013;57:13–22. doi: 10.1016/j.yjmcc.2013.01.001. [DOI] [PubMed] [Google Scholar]
- 28.Malik FI, Hartman JJ, Elias KA, Morgan BP, Rodriguez H, Brejc K, Anderson RL, Sueoka SH, Lee KH, Finer JT, Sakowicz R, Baliga R, Cox DR, Garard M, Godinez G, Kawas R, Kraynack E, Lenzi D, Lu PP, Muci A, Niu C, Qian X, Pierce DW, Pokrovskii M, Suehiro I, Sylvester S, Tochimoto T, Valdez C, Wang W, Katori T, Kass DA, Shen YT, Vatner SF, Morgans DJ. Cardiac myosin activation: a potential therapeutic approach for systolic heart failure. Science. 2011;331:1439–1443. doi: 10.1126/science.1200113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jiang J, Wakimoto H, Seidman JG, Seidman CE. Allele-specific silencing of mutant Myh6 transcripts in mice suppresses hypertrophic cardiomyopathy. Science. 2013;342:111–114. doi: 10.1126/science.1236921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gedicke-Hornung C, Behrens-Gawlik V, Reischmann S, Geertz B, Stimpel D, Weinberger F, Schlossarek S, Precigout G, Braren I, Eschenhagen T, Mearini G, Lorain S, Voit T, Dreyfus PA, Garcia L, Carrier L. Rescue of cardiomyopathy through U7snRNA-mediated exon skipping in Mybpc3-targeted knock-in mice. EMBO Mol Med. 2013;5:1128–1145. doi: 10.1002/emmm.201202168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mearini G, Stimpel D, Geertz B, Weinberger F, Krämer E, Schlossarek S, Mourot-Filiatre J, Stöhr A, Dutshc A, Wijnker PJM, Braren I, Katus HA, Müller OJ, Voit T, Eschenhagen T, Carrier L. Mybpc3 gene therapy for neonatal cardiomyopathy enables longterm disease prevention in mice. Nat Commun. 2014;5:5515. doi: 10.1038/ncomms6515. [DOI] [PubMed] [Google Scholar]
- 32.Mamidi R, Li J, Gresham KS, Stelzer JE. Cardiac myosin binding protein-C: a novel sarcomeric target for gene therapy. Pflugers Arch. 2014;466:225–230. doi: 10.1007/s00424-013-1412-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Behrens-Gawlik V, Mearini G, Gedicke-Hornung C, Richard P, Carrier L. MYBPC3 in hypertrophic cardiomyopathy: from mutation identification to RNA-based correction. Pflugers Arch. 2014;466:215–223. doi: 10.1007/s00424-013-1409-7. [DOI] [PubMed] [Google Scholar]
- 34.Marziliano N, Merlini PA, Vignati G, Orsini F, Motta V, Bandiera L, Intrieri M, Veronese S. A case of compound mutations in the MYBPC3 gene associated with biventricular hypertrophy and neonatal death. Neonatology. 2012;102:254–258. doi: 10.1159/000339847. [DOI] [PubMed] [Google Scholar]
- 35.Dellefave LM, Pytel P, Mewborn S, Mora B, Guris DL, Fedson S, Waggoner D, Moskowitz I, McNally EM. Sarcomere mutations in cardiomyopathy with left ventricular hypertrabeculation. Circ Cardiovasc Genet. 2009;2:442–449. doi: 10.1161/CIRCGENETICS.109.861955. [DOI] [PubMed] [Google Scholar]
- 36.Lekanne Deprez RH, Muurling-Vlietman JJ, Hruda J, Baars MJ, Wijnaendts LC, Stolte-Dijkstra I, Alders M, van Hagen JM. Two cases of severe neonatal hypertrophic cardiomyopathy caused by compound heterozygous mutations in the MYBPC3 gene. J Med Genet. 2006;43:829–832. doi: 10.1136/jmg.2005.040329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xin B, Puffenberger E, Tumbush J, Bockoven JR, Wang H. Homozygosity for a novel splice site mutation in the cardiac myosin-binding protein C gene causes severe neonatal hypertrophic cardiomyopathy. Am J Med Genet A. 2007;143A:2662–2667. doi: 10.1002/ajmg.a.31981. [DOI] [PubMed] [Google Scholar]
- 38.Wessels MW, Herkert JC, Frohn-Mulder IM, Dalinghaus M, van den Wijngaard A, de Krijger RR, Michels M, de Coo IF, Hoedemaekers YM, Dooijes D. Compound heterozygous or homozygous truncating MYBPC3 mutations cause lethal cardiomyopathy with features of noncompaction and septal defects. Eur J Hum Genet. 2014 doi: 10.1038/ejhg.2014.211. doi:10.1038/ejhg.2014.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zahka K, Kalidas K, Simpson MA, Cross H, Keller BB, Galambos C, Gurtz K, Patton MA, Crosby AH. Homozygous mutation of MYBPC3 associated with severe infantile hypertrophic cardiomyopathy at high frequency among the Amish. Heart. 2008;94:1326–1330. doi: 10.1136/hrt.2007.127241. [DOI] [PubMed] [Google Scholar]
- 40.Schaefer E, Helms P, Marcellin L, Desprez P, Billaud P, Chanavat V, Rousson R, Millat G. Next-generation sequencing (NGS) as a fast molecular diagnosis tool for left ventricular noncompaction in an infant with compound mutations in the MYBPC3 gene. Eur J Med Genet. 2014;57:129–132. doi: 10.1016/j.ejmg.2014.02.015. [DOI] [PubMed] [Google Scholar]
- 41.Richard P, Charron P, Carrier L, Ledeuil C, Cheav T, Pichereau C, Benaiche A, Isnard R, Dubourg O, Burban M, Gueffet JP, Millaire A, Desnos M, Schwartz K, Hainque B, Komajda M. Hypertrophic cardiomyopathy: distribution of disease genes, spectrum of mutations and implications for molecular diagnosis strategy. Circulation. 2003;107:2227–2232. doi: 10.1161/01.CIR.0000066323.15244.54. [DOI] [PubMed] [Google Scholar]
- 42.Ho CY, Lever HM, DeSanctis R, Farver CF, Seidman JG, Seidman CE. Homozygous mutation in cardiac troponin T: implications for hypertrophic cardiomyopathy. Circulation. 2000;102:1950–1955. doi: 10.1161/01.cir.102.16.1950. [DOI] [PubMed] [Google Scholar]
- 43.Haberer K, Buffo-Sequeira I, Chudley AE, Spriggs E, Sergi C. A case of an infant with compound heterozygous mutations for hypertrophic cardiomyopathy producing a phenotype of left ventricular noncompaction. Can J Cardiol. 2014;30:1249 e1241–1243. doi: 10.1016/j.cjca.2014.05.021. [DOI] [PubMed] [Google Scholar]
- 44.Weterman MA, Barth PG, van Spaendonck-Zwarts KY, Aronica E, Poll-The BT, Brouwer OF, van Tintelen JP, Qahar Z, Bradley EJ, de Wissel M, Salviati L, Angelini C, van den Heuvel L, Thomasse YE, Backx AP, Nurnberg G, Nurnberg P, Baas F. Recessive MYL2 mutations cause infantile type I muscle fibre disease and cardiomyopathy. Brain. 2013;136:282–293. doi: 10.1093/brain/aws293. [DOI] [PubMed] [Google Scholar]
- 45.Ingles J, Doolan A, Chiu C, Seidman J, Seidman C, Semsarian C. Compound and double mutations in patients with hypertrophic cardiomyopathy: implications for genetic testing and counselling. J Med Genet. 2005;42:e59. doi: 10.1136/jmg.2005.033886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Maron BJ, Maron MS, Semsarian C. Double or compound sarcomere mutations in hypertrophic cardiomyopathy: a potential link to sudden death in the absence of conventional risk factors. Heart Rhythm. 2012;9:57–63. doi: 10.1016/j.hrthm.2011.08.009. [DOI] [PubMed] [Google Scholar]
- 47.Biagini E, Olivotto I, Iascone M, Parodi MI, Girolami F, Frisso G, Autore C, Limongelli G, Cecconi M, Maron BJ, Maron MS, Rosmini S, Formisano F, Musumeci B, Cecchi F, Iacovoni A, Haas TS, Bacchi Reggiani ML, Ferrazzi P, Salvatore F, Spirito P, Rapezzi C. Significance of sarcomere gene mutations analysis in the end-stage phase of hypertrophic cardiomyopathy. Am J Cardiol. 2014;114:769–776. doi: 10.1016/j.amjcard.2014.05.065. [DOI] [PubMed] [Google Scholar]
- 48.El-Saiedi SA, Seliem ZS, Esmail RI. Hypertrophic cardiomyopathy: prognostic factors and survival analysis in 128 Egyptian patients. Cardiol Young. 2014;24:702–708. doi: 10.1017/S1047951113001030. [DOI] [PubMed] [Google Scholar]
- 49.Keeler CE. Gene therapy. J Hered. 1947;38:294–298. [PubMed] [Google Scholar]
- 50.Kaye DM, Preovolos A, Marshall T, Byrne M, Hoshijima M, Hajjar R, Mariani JA, Pepe S, Chien KR, Power JM. Percutaneous cardiac recirculation-mediated gene transfer of an inhibitory phospholamban peptide reverses advanced heart failure in large animals. J Am Coll Cardiol. 2007;50:253–260. doi: 10.1016/j.jacc.2007.03.047. [DOI] [PubMed] [Google Scholar]
- 51.Pleger ST, Shan C, Ksienzyk J, Bekeredjian R, Boekstegers P, Hinkel R, Schinkel S, Leuchs B, Ludwig J, Qiu G, Weber C, Raake P, Koch WJ, Katus HA, Muller OJ, Most P. Cardiac AAV9-S100A1 gene therapy rescues post-ischemic heart failure in a preclinical large animal model. Sci Transl Med. 2011;3:92ra64. doi: 10.1126/scitranslmed.3002097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jessup M, Greenberg B, Mancini D, Cappola T, Pauly DF, Jaski B, Yaroshinsky A, Zsebo KM, Dittrich H, Hajjar RJ. Calcium upregulation by percutaneous administration of gene therapy in cardiac disease (CUPID): a phase 2 trial of intracoronary gene therapy of sarcoplasmic reticulum Ca2+-ATPase in patients with advanced heart failure. Circulation. 2011;124:304–313. doi: 10.1161/CIRCULATIONAHA.111.022889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Werfel S, Jungmann A, Lehmann L, Ksienzyk J, Bekeredjian R, Kaya Z, Leuchs B, Nordheim A, Backs J, Engelhardt S, Katus HA, Muller OJ. Rapid and highly efficient inducible cardiac gene knockout in adult mice using AAV-mediated expression of Cre recombinase. Cardiovasc Res. 2014;104:15–23. doi: 10.1093/cvr/cvu174. [DOI] [PubMed] [Google Scholar]
- 54.Mearini G, Stimpel D, Kramer E, Geertz B, Braren I, Gedicke-Hornung C, Precigout G, Muller OJ, Katus HA, Eschenhagen T, Voit T, Garcia L, Lorain S, Carrier L. Repair of Mybpc3 mRNA by 5′-trans-splicing in a mouse model of hypertrophic cardiomyopathy. Mol Ther Nucleic Acids. 2013;2:e102. doi: 10.1038/mtna.2013.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wally V, Murauer EM, Bauer JW. Spliceosome-mediated trans-splicing: the therapeutic cut and paste. J Invest Dermatol. 2012;132:1959–1966. doi: 10.1038/jid.2012.101. [DOI] [PubMed] [Google Scholar]
- 56.Woodley L, Valcarcel J. Regulation of alternative pre-mRNA splicing. Brief Funct Genomic Proteomic. 2002;1:266–277. doi: 10.1093/bfgp/1.3.266. [DOI] [PubMed] [Google Scholar]
- 57.Hammond SM, Wood MJ. Genetic therapies for RNA mis-splicing diseases. Trends Genet. 2011;27:196–205. doi: 10.1016/j.tig.2011.02.004. [DOI] [PubMed] [Google Scholar]
- 58.Duncker DJ, Bakkers J, Brundel BJ, Robbins J, Tardiff JC, Carrier L. Animal- and In Silico Models for the study of Sarcomeric Cardiomyopathies. Cardiovasc Res. 2015;105:439–448. doi: 10.1093/cvr/cvv006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Spoladore R, Maron MS, D'Amato R, Camici PG, Olivotto I. Pharmacological treatment options for hypertrophic cardiomyopathy: high time for evidence. Eur Heart J. 2012;33:1724–1733. doi: 10.1093/eurheartj/ehs150. [DOI] [PubMed] [Google Scholar]
- 60.Coppini R, Ferrantini C, Yao L, Fan P, Del Lungo M, Stillitano F, Sartiani L, Tosi B, Suffredini S, Tesi C, Yacoub M, Olivotto I, Belardinelli L, Poggesi C, Cerbai E, Mugelli A. Late sodium current inhibition reverses electromechanical dysfunction in human hypertrophic cardiomyopathy. Circulation. 2013;127:575–584. doi: 10.1161/CIRCULATIONAHA.112.134932. [DOI] [PubMed] [Google Scholar]
- 61.Johnson JN, Grifoni C, Bos JM, Saber-Ayad M, Ommen SR, Nistri S, Cecchi F, Olivotto I, Ackerman MJ. Prevalence and clinical correlates of QT prolongation in patients with hypertrophic cardiomyopathy. Eur Heart J. 2011;32:1114–1120. doi: 10.1093/eurheartj/ehr021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wagner S, Dybkova N, Rasenack EC, Jacobshagen C, Fabritz L, Kirchhof P, Maier SK, Zhang T, Hasenfuss G, Brown JH, Bers DM, Maier LS. Ca2+/calmodulin-dependent protein kinase II regulates cardiac Na+ channels. J Clin Invest. 2006;116:3127–3138. doi: 10.1172/JCI26620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ashpole NM, Herren AW, Ginsburg KS, Brogan JD, Johnson DE, Cummins TR, Bers DM, Hudmon A. Ca2+/calmodulin-dependent protein kinase II (CaMKII) regulates cardiac sodium channel Nav1.5 gating by multiple phosphorylation sites. J Biol Chem. 2012;287:19856–19869. doi: 10.1074/jbc.M111.322537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Shannon TR, Pogwizd SM, Bers DM. Elevated sarcoplasmic reticulum Ca2+ leak in intact ventricular myocytes from rabbits in heart failure. Circ Res. 2003;93:592–594. doi: 10.1161/01.RES.0000093399.11734.B3. [DOI] [PubMed] [Google Scholar]
- 65.Morotti S, Edwards AG, McCulloch AD, Bers DM, Grandi E. A novel computational model of mouse myocyte electrophysiology to assess the synergy between Na+ loading and CaMKII. J Physiol. 2014;592:1181–1197. doi: 10.1113/jphysiol.2013.266676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fischer TH, Herting J, Tirilomis T, Renner A, Neef S, Toischer K, Ellenberger D, Forster A, Schmitto JD, Gummert J, Schondube FA, Hasenfuss G, Maier LS, Sossalla S. Ca2+/calmodulin-dependent protein kinase II and protein kinase A differentially regulate sarcoplasmic reticulum Ca2+ leak in human cardiac pathology. Circulation. 2013;128:970–981. doi: 10.1161/CIRCULATIONAHA.113.001746. [DOI] [PubMed] [Google Scholar]
- 67.Toischer K, Rokita AG, Unsold B, Zhu W, Kararigas G, Sossalla S, Reuter SP, Becker A, Teucher N, Seidler T, Grebe C, Preuss L, Gupta SN, Schmidt K, Lehnart SE, Kruger M, Linke WA, Backs J, Regitz-Zagrosek V, Schafer K, Field LJ, Maier LS, Hasenfuss G. Differential cardiac remodeling in preload versus afterload. Circulation. 2010;122:993–1003. doi: 10.1161/CIRCULATIONAHA.110.943431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Harris KM, Spirito P, Maron MS, Zenovich AG, Formisano F, Lesser JR, Mackey-Bojack S, Manning WJ, Udelson JE, Maron BJ. Prevalence, clinical profile, and significance of left ventricular remodeling in the end-stage phase of hypertrophic cardiomyopathy. Circulation. 2006;114:216–225. doi: 10.1161/CIRCULATIONAHA.105.583500. [DOI] [PubMed] [Google Scholar]
- 69.Olivotto I, Cecchi F, Gistri R, Lorenzoni R, Chiriatti G, Girolami F, Torricelli F, Camici PG. Relevance of coronary microvascular flow impairment to long-term remodeling and systolic dysfunction in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2006;47:1043–1048. doi: 10.1016/j.jacc.2005.10.050. [DOI] [PubMed] [Google Scholar]
- 70.Pollick C. Muscular subaortic stenosis: hemodynamic and clinical improvement after disopyramide. N Engl J Med. 1982;307:997–999. doi: 10.1056/NEJM198210143071607. [DOI] [PubMed] [Google Scholar]
- 71.Sherrid MV, Barac I, McKenna WJ, Elliott PM, Dickie S, Chojnowska L, Casey S, Maron BJ. Multicenter study of the efficacy and safety of disopyramide in obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2005;45:1251–1258. doi: 10.1016/j.jacc.2005.01.012. [DOI] [PubMed] [Google Scholar]
- 72.Hartmann A, Kuhn J, Hopf R, Klepzig H, Standke R, Kober G, Maul FD, Hor G, Kaltenbach M. Effect of propranolol and disopyramide on left ventricular function at rest and during exercise in hypertrophic cardiomyopathy. Cardiology. 1992;80:81–88. doi: 10.1159/000174983. [DOI] [PubMed] [Google Scholar]
- 73.Kondo N, Mizukami M, Shibata S. Negative inotropic effects of disopyramide on guinea-pig papillary muscles. Br J Pharmacol. 1990;101:789–792. doi: 10.1111/j.1476-5381.1990.tb14158.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sherrid MV, Pearle G, Gunsburg DZ. Mechanism of benefit of negative inotropes in obstructive hypertrophic cardiomyopathy. Circulation. 1998;97:41–47. doi: 10.1161/01.cir.97.1.41. [DOI] [PubMed] [Google Scholar]
- 75.Rosing DR, Kent KM, Borer JS, Seides SF, Maron BJ, Epstein SE. Verapamil therapy: a new approach to the pharmacologic treatment of hypertrophic cardiomyopathy. I. Hemodynamic effects. Circulation. 1979;60:1201–1207. doi: 10.1161/01.cir.60.6.1201. [DOI] [PubMed] [Google Scholar]
- 76.Bonow RO, Rosing DR, Bacharach SL, Green MV, Kent KM, Lipson LC, Maron BJ, Leon MB, Epstein SE. Effects of verapamil on left ventricular systolic function and diastolic filling in patients with hypertrophic cardiomyopathy. Circulation. 1981;64:787–796. doi: 10.1161/01.cir.64.4.787. [DOI] [PubMed] [Google Scholar]
- 77.Gistri R, Cecchi F, Choudhury L, Montereggi A, Sorace O, Salvadori PA, Camici PG. Effect of verapamil on absolute myocardial blood flow in hypertrophic cardiomyopathy. Am J Cardiol. 1994;74:363–368. doi: 10.1016/0002-9149(94)90404-9. [DOI] [PubMed] [Google Scholar]
- 78.Force T, Bonow RO, Houser SR, Solaro RJ, Hershberger RE, Adhikari B, Anderson ME, Boineau R, Byrne BJ, Cappola TP, Kalluri R, LeWinter MM, Maron MS, Molkentin JD, Ommen SR, Regnier M, Tang WH, Tian R, Konstam MA, Maron BJ, Seidman CE. Research priorities in hypertrophic cardiomyopathy: report of a Working Group of the National Heart, Lung, and Blood Institute. Circulation. 2010;122:1130–1133. doi: 10.1161/CIRCULATIONAHA.110.950089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Vassallo P, Trohman RG. Prescribing amiodarone: an evidence-based review of clinical indications. JAMA. 2007;298:1312–1322. doi: 10.1001/jama.298.11.1312. [DOI] [PubMed] [Google Scholar]
- 80.Melacini P, Maron BJ, Bobbo F, Basso C, Tokajuk B, Zucchetto M, Thiene G, Iliceto S. Evidence that pharmacological strategies lack efficacy for the prevention of sudden death in hypertrophic cardiomyopathy. Heart. 2007;93:708–710. doi: 10.1136/hrt.2006.099416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Maier LS, Layug B, Karwatowska-Prokopczuk E, Belardinelli L, Lee S, Sander J, Lang C, Wachter R, Edelmann F, Hasenfuss G, Jacobshagen C. RAnoLazIne for the treatment of diastolic heart failure in patients with preserved ejection fraction: the RALI-DHF proof-of-concept study. JACC Heart Fail. 2013;1:115–122. doi: 10.1016/j.jchf.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 82.Pezhouman A, Madahian S, Stepanyan H, Ghukasyan H, Qu Z, Belardinelli L, Karagueuzian HS. Selective inhibition of late sodium current suppresses ventricular tachycardia and fibrillation in intact rat hearts. Heart Rhythm. 2014;11:492–501. doi: 10.1016/j.hrthm.2013.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Fernandes S, Hoyer K, Liu G, Wang WQ, Dhalla AK, Belardinelli L, Rajamani S. Selective inhibition of the late sodium current has no adverse effect on electrophysiological or contractile function of the normal heart. J Cardiovasc Pharmacol. 2014;63:512–519. doi: 10.1097/FJC.0000000000000075. [DOI] [PubMed] [Google Scholar]
- 84.Maier LS. Ca(2+)/calmodulin-dependent protein kinase II (CaMKII) in the heart. Adv Exp Med Biol. 2012;740:685–702. doi: 10.1007/978-94-007-2888-2_30. [DOI] [PubMed] [Google Scholar]
- 85.Dominguez-Rodriguez A, Ruiz-Hurtado G, Benitah JP, Gomez AM. The other side of cardiac Ca(2+) signaling: transcriptional control. Front Physiol. 2012;3:452. doi: 10.3389/fphys.2012.00452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kreusser MM, Backs J. Integrated mechanisms of CaMKII-dependent ventricular remodeling. Front Pharmacol. 2014;5:36. doi: 10.3389/fphar.2014.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ling H, Zhang T, Pereira L, Means CK, Cheng H, Gu Y, Dalton ND, Peterson KL, Chen J, Bers D, Brown JH. Requirement for Ca2+/calmodulin-dependent kinase II in the transition from pressure overload-induced cardiac hypertrophy to heart failure in mice. J Clin Invest. 2009;119:1230–1240. doi: 10.1172/JCI38022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pellicena P, Schulman H. CaMKII inhibitors: from research tools to therapeutic agents. Front Pharmacol. 2014;5:21. doi: 10.3389/fphar.2014.00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen X, Nakayama H, Zhang X, Ai X, Harris DM, Tang M, Zhang H, Szeto C, Stockbower K, Berretta RM, Eckhart AD, Koch WJ, Molkentin JD, Houser SR. Calcium influx through Cav1.2 is a proximal signal for pathological cardiomyocyte hypertrophy. J Mol Cell Cardiol. 2011;50:460–470. doi: 10.1016/j.yjmcc.2010.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kaltenbach M, Hopf R, Kober G, Bussmann WD, Keller M, Petersen Y. Treatment of hypertrophic obstructive cardiomyopathy with verapamil. Br Heart J. 1979;42:35–42. doi: 10.1136/hrt.42.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Westermann D, Knollmann BC, Steendijk P, Rutschow S, Riad A, Pauschinger M, Potter JD, Schultheiss HP, Tschope C. Diltiazem treatment prevents diastolic heart failure in mice with familial hypertrophic cardiomyopathy. Eur J Heart Fail. 2006;8:115–121. doi: 10.1016/j.ejheart.2005.07.012. [DOI] [PubMed] [Google Scholar]
- 92.Wagner S, Ruff HM, Weber SL, Bellmann S, Sowa T, Schulte T, Anderson ME, Grandi E, Bers DM, Backs J, Belardinelli L, Maier LS. Reactive oxygen species-activated Ca/calmodulin kinase IIdelta is required for late I(Na) augmentation leading to cellular Na and Ca overload. Circ Res. 2011;108:555–565. doi: 10.1161/CIRCRESAHA.110.221911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Herren AW, Bers DM, Grandi E. Post-translational modifications of the cardiac Na channel: contribution of CaMKII-dependent phosphorylation to acquired arrhythmias. Am J Physiol Heart Circ Physiol. 2013;305:H431–H445. doi: 10.1152/ajpheart.00306.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yao L, Fan P, Jiang Z, Viatchenko-Karpinski S, Wu Y, Kornyeyev D, Hirakawa R, Budas GR, Rajamani S, Shryock JC, Belardinelli L. Nav1.5-dependent persistent Na+ influx activates CaMKII in rat ventricular myocytes and N1325S mice. Am J Physiol Cell Physiol. 2011;301:C577–C586. doi: 10.1152/ajpcell.00125.2011. [DOI] [PubMed] [Google Scholar]
- 95.Pourrier M, Williams S, McAfee D, Belardinelli L, Fedida D. CrossTalk proposal: the late sodium current is an important player in the development of diastolic heart failure (heart failure with a preserved ejection fraction) J Physiol. 2014;592:411–414. doi: 10.1113/jphysiol.2013.262261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Olivotto I, Cecchi F, Poggesi C, Yacoub MH. Patterns of disease progression in hypertrophic cardiomyopathy: an individualized approach to clinical staging. Circ Heart Fail. 2012;5:535–546. doi: 10.1161/CIRCHEARTFAILURE.112.967026. [DOI] [PubMed] [Google Scholar]
- 97.Rastogi S, Sharov VG, Mishra S, Gupta RC, Blackburn B, Belardinelli L, Stanley WC, Sabbah HN. Ranolazine combined with enalapril or metoprolol prevents progressive LV dysfunction and remodeling in dogs with moderate heart failure. Am J Physiol Heart Circ Physiol. 2008;295:H2149–H2155. doi: 10.1152/ajpheart.00728.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Olivotto I, Girolami F, Nistri S, Rossi A, Rega L, Garbini F, Grifoni C, Cecchi F, Yacoub MH. The many faces of hypertrophic cardiomyopathy: from developmental biology to clinical practice. J Cardiovasc Transl Res. 2009;2:349–367. doi: 10.1007/s12265-009-9137-2. [DOI] [PubMed] [Google Scholar]
- 99.Ho CY, Sweitzer NK, McDonough B, Maron BJ, Casey SA, Seidman JG, Seidman CE, Solomon SD. Assessment of diastolic function with Doppler tissue imaging to predict genotype in preclinical hypertrophic cardiomyopathy. Circulation. 2002;105:2992–2997. doi: 10.1161/01.cir.0000019070.70491.6d. [DOI] [PubMed] [Google Scholar]
- 100.Haim TE, Dowell C, Diamanti T, Scheuer J, Tardiff JC. Independent FHC-related cardiac troponin T mutations exhibit specific alterations in myocellular contractility and calcium kinetics. J Mol Cell Cardiol. 2007;42:1098–1110. doi: 10.1016/j.yjmcc.2007.03.906. [DOI] [PubMed] [Google Scholar]
- 101.Knollmann BC, Kirchhof P, Sirenko SG, Degen H, Greene AE, Schober T, Mackow JC, Fabritz L, Potter JD, Morad M. Familial hypertrophic cardiomyopathy-linked mutant troponin T causes stress-induced ventricular tachycardia and Ca2+-dependent action potential remodeling. Circ Res. 2003;92:428–436. doi: 10.1161/01.RES.0000059562.91384.1A. [DOI] [PubMed] [Google Scholar]
- 102.Fraysse B, Weinberger F, Bardswell SC, Cuello F, Vignier N, Geertz B, Starbatty J, Kramer E, Coirault C, Eschenhagen T, Kentish JC, Avkiran M, Carrier L. Increased myofilament Ca2+ sensitivity and diastolic dysfunction as early consequences of Mybpc3 mutation in heterozygous knock-in mice. J Mol Cell Cardiol. 2012;52:1299–1307. doi: 10.1016/j.yjmcc.2012.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Semsarian C, Ahmad I, Giewat M, Georgakopoulos D, Schmitt JP, McConnell BK, Reiken S, Mende U, Marks AR, Kass DA, Seidman CE, Seidman JG. The L-type calcium channel inhibitor diltiazem prevents cardiomyopathy in a mouse model. J Clin Invest. 2002;109:1013–1020. doi: 10.1172/JCI14677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ho CY, Lakdawala NK, Cirino AL, Lipshultz SE, Sparks E, Abbasi SA, Kwong RY, Antman EM, Semsarian C, González A, López B, Diez J, Orav EJ, Colan SD, Seidman CE. Diltiazem treatment for pre-clinical hypertrophic cardiomyopathy sarcomere mutation carriers: a pilot randomized trial to modify disease expression. JACC Heart Fail. 2015;3:180–188. doi: 10.1016/j.jchf.2014.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ashrafian H, Redwood C, Blair E, Watkins H. Hypertrophic cardiomyopathy: a paradigm for myocardial energy depletion. Trends Genet. 2003;19:263–268. doi: 10.1016/S0168-9525(03)00081-7. [DOI] [PubMed] [Google Scholar]
- 106.Witjas-Paalberends ER, Ferrara C, Scellini B, Piroddi N, Montag J, Tesi C, Stienen GJ, Michels M, Ho CY, Kraft T, Poggesi C, van der Velden J. Faster cross-bridge detachment and increased tension cost in human hypertrophic cardiomyopathy with the R403Q MYH7 mutation. J Physiol. 2014;592:3257–3272. doi: 10.1113/jphysiol.2014.274571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Witjas-Paalberends ER, Guclu A, Germans T, Knaapen P, Harms HJ, Vermeer AM, Christiaans I, Wilde AA, Remedios CD, Lammertsma AA, van Rossum AC, Stienen GJ, van Slegtenhorst M, Schinkel AF, Michels M, Ho CY, Poggesi C, van der Velden J. Gene-specific increase in energetic cost of contraction in hypertrophic cardiomyopathy caused by thick filament mutations. Cardiovasc Res. 2014;103:248–257. doi: 10.1093/cvr/cvu127. [DOI] [PubMed] [Google Scholar]
- 108.Unno K, Isobe S, Izawa H, Cheng XW, Kobayashi M, Hirashiki A, Yamada T, Harada K, Ohshima S, Noda A, Nagata K, Kato K, Yokota M, Murohara T. Relation of functional and morphological changes in mitochondria to myocardial contractile and relaxation reserves in asymptomatic to mildly symptomatic patients with hypertrophic cardiomyopathy. Eur Heart J. 2009;30:1853–1862. doi: 10.1093/eurheartj/ehp184. [DOI] [PubMed] [Google Scholar]
- 109.Mahmod M, Francis JM, Pal N, Lewis A, Dass S, De Silva R, Petrou M, Sayeed R, Westaby S, Robson MD, Ashrafian H, Neubauer S, Karamitsos TD. Myocardial perfusion and oxygenation are impaired during stress in severe aortic stenosis and correlate with impaired energetics and subclinical left ventricular dysfunction. J Cardiovasc Magn Reson. 2014;16:29. doi: 10.1186/1532-429X-16-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Crilley JG, Boehm EA, Blair E, Rajagopalan B, Blamire AM, Styles P, McKenna WJ, Ostman-Smith I, Clarke K, Watkins H. Hypertrophic cardiomyopathy due to sarcomeric gene mutations is characterized by impaired energy metabolism irrespective of the degree of hypertrophy. J Am Coll Cardiol. 2003;41:1776–1782. doi: 10.1016/s0735-1097(02)03009-7. [DOI] [PubMed] [Google Scholar]
- 111.Timmer SA, Germans T, Brouwer WP, Lubberink M, van der Velden J, Wilde AA, Christiaans I, Lammertsma AA, Knaapen P, van Rossum AC. Carriers of the hypertrophic cardiomyopathy MYBPC3 mutation are characterized by reduced myocardial efficiency in the absence of hypertrophy and microvascular dysfunction. Eur J Heart Fail. 2011;13:1283–1289. doi: 10.1093/eurjhf/hfr135. [DOI] [PubMed] [Google Scholar]
- 112.Blair E, Redwood C, Ashrafian H, Oliveira M, Broxholme J, Kerr B, Salmon A, Ostman-Smith I, Watkins H. Mutations in the gamma(2) subunit of AMP-activated protein kinase cause familial hypertrophic cardiomyopathy: evidence for the central role of energy compromise in disease pathogenesis. Hum Mol Genet. 2001;10:1215–1220. doi: 10.1093/hmg/10.11.1215. [DOI] [PubMed] [Google Scholar]
- 113.Guclu A, Germans T, Witjas-Paalberends ER, Stienen GJ, Brouwer WP, Harms HJ, Marcus JT, Vonk AB, Stooker W, Yilmaz A, Klein P, Ten Berg JM, Kluin J, Asselbergs FW, Lammertsma AA, Knaapen P, van Rossum AC, van der Velden J. ENerGetIcs in hypertrophic cardiomyopathy: traNslation between MRI, PET and cardiac myofilament function (ENGINE study) Neth Heart J. 2013;21:567–571. doi: 10.1007/s12471-013-0478-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Krishnan J, Suter M, Windak R, Krebs T, Felley A, Montessuit C, Tokarska-Schlattner M, Aasum E, Bogdanova A, Perriard E, Perriard JC, Larsen T, Pedrazzini T, Krek W. Activation of a HIF1alpha-PPARgamma axis underlies the integration of glycolytic and lipid anabolic pathways in pathologic cardiac hypertrophy. Cell Metab. 2009;9:512–524. doi: 10.1016/j.cmet.2009.05.005. [DOI] [PubMed] [Google Scholar]
- 115.Holscher M, Schafer K, Krull S, Farhat K, Hesse A, Silter M, Lin Y, Pichler BJ, Thistlethwaite P, El-Armouche A, Maier LS, Katschinski DM, Zieseniss A. Unfavourable consequences of chronic cardiac HIF-1alpha stabilization. Cardiovasc Res. 2012;94:77–86. doi: 10.1093/cvr/cvs014. [DOI] [PubMed] [Google Scholar]
- 116.Sorajja P, Ommen SR, Nishimura RA, Gersh BJ, Berger PB, Tajik AJ. Adverse prognosis of patients with hypertrophic cardiomyopathy who have epicardial coronary artery disease. Circulation. 2003;108:2342–2348. doi: 10.1161/01.CIR.0000097110.55312.BF. [DOI] [PubMed] [Google Scholar]
- 117.Maron MS, Olivotto I, Maron BJ, Prasad SK, Cecchi F, Udelson JE, Camici PG. The case for myocardial ischemia in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2009;54:866–875. doi: 10.1016/j.jacc.2009.04.072. [DOI] [PubMed] [Google Scholar]
- 118.Morioka N, Shigematsu Y, Hamada M, Higaki J. Circulating levels of heart-type fatty acid-binding protein and its relation to thallium-201 perfusion defects in patients with hypertrophic cardiomyopathy. Am J Cardiol. 2005;95:1334–1337. doi: 10.1016/j.amjcard.2005.01.078. [DOI] [PubMed] [Google Scholar]
- 119.Cannon RO, III, Dilsizian V, O'Gara PT, Udelson JE, Schenke WH, Quyyumi A, Fananapazir L, Bonow RO. Myocardial metabolic, hemodynamic, and electrocardiographic significance of reversible thallium-201 abnormalities in hypertrophic cardiomyopathy. Circulation. 1991;83:1660–1667. doi: 10.1161/01.cir.83.5.1660. [DOI] [PubMed] [Google Scholar]
- 120.Maron BJ, Epstein SE, Roberts WC. Hypertrophic cardiomyopathy and transmural myocardial infarction without significant atherosclerosis of the extramural coronary arteries. Am J Cardiol. 1979;43:1086–1102. doi: 10.1016/0002-9149(79)90139-5. [DOI] [PubMed] [Google Scholar]
- 121.Greulich S, Schumm J, Grun S, Bruder O, Sechtem U, Mahrholdt H. Incremental value of late gadolinium enhancement for management of patients with hypertrophic cardiomyopathy. Am J Cardiol. 2012;110:1207–1212. doi: 10.1016/j.amjcard.2012.05.064. [DOI] [PubMed] [Google Scholar]
- 122.Lorenzoni R, Gistri R, Cecchi F, Olivotto I, Chiriatti G, Elliott P, McKenna WJ, Camici PG. Coronary vasodilator reserve is impaired in patients with hypertrophic cardiomyopathy and left ventricular dysfunction. Am Heart J. 1998;136:972–981. doi: 10.1016/s0002-8703(98)70152-8. [DOI] [PubMed] [Google Scholar]
- 123.Timmer SA, Germans T, Gotte MJ, Russel IK, Lubberink M, Ten Berg JM, Ten Cate FJ, Lammertsma AA, Knaapen P, van Rossum AC. Relation of coronary microvascular dysfunction in hypertrophic cardiomyopathy to contractile dysfunction independent from myocardial injury. Am J Cardiol. 2011;107:1522–1528. doi: 10.1016/j.amjcard.2011.01.029. [DOI] [PubMed] [Google Scholar]
- 124.Ahn HS, Kim HK, Park EA, Lee W, Kim YJ, Cho GY, Park JH, Sohn DW. Coronary flow reserve impairment in apical vs asymmetrical septal hypertrophic cardiomyopathy. Clin Cardiol. 2013;36:207–216. doi: 10.1002/clc.22095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.O'Gara PT, Bonow RO, Maron BJ, Damske BA, Van Lingen A, Bacharach SL, Larson SM, Epstein SE. Myocardial perfusion abnormalities in patients with hypertrophic cardiomyopathy: assessment with thallium-201 emission computed tomography. Circulation. 1987;76:1214–1223. doi: 10.1161/01.cir.76.6.1214. [DOI] [PubMed] [Google Scholar]
- 126.Huang L, Han R, Ai T, Sun Z, Bai Y, Cao Z, Morelli JN, Xia L. Assessment of coronary microvascular dysfunction in hypertrophic cardiomyopathy: first-pass myocardial perfusion cardiovascular magnetic resonance imaging at 1.5 T. Clin Radiol. 2013;68:676–682. doi: 10.1016/j.crad.2013.01.003. [DOI] [PubMed] [Google Scholar]
- 127.Karamitsos TD, Dass S, Suttie J, Sever E, Birks J, Holloway CJ, Robson MD, Jerosch-Herold M, Watkins H, Neubauer S. Blunted myocardial oxygenation response during vasodilator stress in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2013;61:1169–1176. doi: 10.1016/j.jacc.2012.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Yamanishi K, Fujita M, Araie E, Ohno A, Sasayama S, Okada E. Regional myocardial hypertrophy induced by repeated brief coronary occlusion in conscious dogs. Am J Cardiovasc Pathol. 1990;3:305–310. [PubMed] [Google Scholar]
- 129.Knoll R, Postel R, Wang J, Kratzner R, Hennecke G, Vacaru AM, Vakeel P, Schubert C, Murthy K, Rana BK, Kube D, Knoll G, Schafer K, Hayashi T, Holm T, Kimura A, Schork N, Toliat MR, Nurnberg P, Schultheiss HP, Schaper W, Schaper J, Bos E, Den Hertog J, van Eeden FJ, Peters PJ, Hasenfuss G, Chien KR, Bakkers J. Laminin-alpha4 and integrin-linked kinase mutations cause human cardiomyopathy via simultaneous defects in cardiomyocytes and endothelial cells. Circulation. 2007;116:515–525. doi: 10.1161/CIRCULATIONAHA.107.689984. [DOI] [PubMed] [Google Scholar]
- 130.Johansson B, Morner S, Waldenstrom A, Stal P. Myocardial capillary supply is limited in hypertrophic cardiomyopathy: a morphological analysis. Int J Cardiol. 2008;126:252–257. doi: 10.1016/j.ijcard.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 131.Krams R, Kofflard MJ, Duncker DJ, Von Birgelen C, Carlier S, Kliffen M, ten Cate FJ, Serruys PW. Decreased coronary flow reserve in hypertrophic cardiomyopathy is related to remodeling of the coronary microcirculation. Circulation. 1998;97:230–233. doi: 10.1161/01.cir.97.3.230. [DOI] [PubMed] [Google Scholar]
- 132.Maron BJ, Wolfson JK, Epstein SE, Roberts WC. Intramural (‘mall vessel’) coronary artery disease in hypertrophic cardiomyopathy. J Am Coll Cardiol. 1986;8:545–557. doi: 10.1016/s0735-1097(86)80181-4. [DOI] [PubMed] [Google Scholar]
- 133.Frid MG, Kale VA, Stenmark KR. Mature vascular endothelium can give rise to smooth muscle cells via endothelial-mesenchymal transdifferentiation: in vitro analysis. Circ Res. 2002;90:1189–1196. doi: 10.1161/01.res.0000021432.70309.28. [DOI] [PubMed] [Google Scholar]
- 134.Abe K, Toba M, Alzoubi A, Ito M, Fagan KA, Cool CD, Voelkel NF, McMurtry IF, Oka M. Formation of plexiform lesions in experimental severe pulmonary arterial hypertension. Circulation. 2010;121:2747–2754. doi: 10.1161/CIRCULATIONAHA.109.927681. [DOI] [PubMed] [Google Scholar]
- 135.Inoue K, Hamada M, Ohtsuka T, Hara Y, Shigematsu Y, Nakata S, Higaki J. Myocardial microvascular abnormalities observed by intravenous myocardial contrast echocardiography in patients with hypertrophic cardiomyopathy. Am J Cardiol. 2004;94:55–58. doi: 10.1016/j.amjcard.2004.03.030. [DOI] [PubMed] [Google Scholar]
- 136.Soliman OI, Geleijnse ML, Michels M, Dijkmans PA, Nemes A, van Dalen BM, Vletter WB, Serruys PW, ten Cate FJ. Effect of successful alcohol septal ablation on microvascular function in patients with obstructive hypertrophic cardiomyopathy. Am J Cardiol. 2008;101:1321–1327. doi: 10.1016/j.amjcard.2007.12.032. [DOI] [PubMed] [Google Scholar]
- 137.Davies JE, Whinnett ZI, Francis DP, Manisty CH, Aguado-Sierra J, Willson K, Foale RA, Malik IS, Hughes AD, Parker KH, Mayet J. Evidence of a dominant backward-propagating ‘uction’ wave responsible for diastolic coronary filling in humans, attenuated in left ventricular hypertrophy. Circulation. 2006;113:1768–1778. doi: 10.1161/CIRCULATIONAHA.105.603050. [DOI] [PubMed] [Google Scholar]
- 138.Olivotto I, Girolami F, Sciagra R, Ackerman MJ, Sotgia B, Bos JM, Nistri S, Sgalambro A, Grifoni C, Torricelli F, Camici PG, Cecchi F. Microvascular function is selectively impaired in patients with hypertrophic cardiomyopathy and sarcomere myofilament gene mutations. J Am Coll Cardiol. 2011;58:839–848. doi: 10.1016/j.jacc.2011.05.018. [DOI] [PubMed] [Google Scholar]
- 139.Basso C, Thiene G, Mackey-Bojack S, Frigo AC, Corrado D, Maron BJ. Myocardial bridging, a frequent component of the hypertrophic cardiomyopathy phenotype, lacks systematic association with sudden cardiac death. Eur Heart J. 2009;30:1627–1634. doi: 10.1093/eurheartj/ehp121. [DOI] [PubMed] [Google Scholar]
- 140.Deussen A, Ohanyan V, Jannasch A, Yin L, Chilian W. Mechanisms of metabolic coronary flow regulation. J Mol Cell Cardiol. 2012;52:794–801. doi: 10.1016/j.yjmcc.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 141.Konno T, Chen D, Wang L, Wakimoto H, Teekakirikul P, Nayor M, Kawana M, Eminaga S, Gorham JM, Pandya K, Smithies O, Naya FJ, Olson EN, Seidman JG, Seidman CE. Heterogeneous myocyte enhancer factor-2 (Mef2) activation in myocytes predicts focal scarring in hypertrophic cardiomyopathy. Proc Natl Acad Sci USA. 2010;107:18097–18102. doi: 10.1073/pnas.1012826107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Huke S, Venkataraman R, Faggioni M, Bennuri S, Hwang HS, Baudenbacher F, Knollmann BC. Focal energy deprivation underlies arrhythmia susceptibility in mice with calcium-sensitized myofilaments. Circ Res. 2013;112:1334–1344. doi: 10.1161/CIRCRESAHA.113.301055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Kubo T, Kitaoka H, Yamanaka S, Hirota T, Baba Y, Hayashi K, Iiyama T, Kumagai N, Tanioka K, Yamasaki N, Matsumura Y, Furuno T, Sugiura T, Doi YL. Significance of high-sensitivity cardiac troponin T in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2013;62:1252–1259. doi: 10.1016/j.jacc.2013.03.055. [DOI] [PubMed] [Google Scholar]
- 144.Alkon J, Friedberg MK, Manlhiot C, Manickaraj AK, Kinnear C, McCrindle BW, Benson LN, Addonizio LJ, Colan SD, Mital S. Genetic variations in hypoxia response genes influence hypertrophic cardiomyopathy phenotype. Pediatr Res. 2012;72:583–592. doi: 10.1038/pr.2012.126. [DOI] [PubMed] [Google Scholar]
- 145.Frenneaux MP, Counihan PJ, Caforio AL, Chikamori T, McKenna WJ. Abnormal blood pressure response during exercise in hypertrophic cardiomyopathy. Circulation. 1990;82:1995–2002. doi: 10.1161/01.cir.82.6.1995. [DOI] [PubMed] [Google Scholar]
- 146.Holloway CJ, Dass S, Suttie JJ, Rider OJ, Cox P, Cochlin LE, Jackson H, Fast AM, Johnson AW, Karamitsos TD, Neubauer S, Clarke K. Exercise training in dilated cardiomyopathy improves rest and stress cardiac function without changes in cardiac high energy phosphate metabolism. Heart. 2012;98:1083–1090. doi: 10.1136/heartjnl-2012-302145. [DOI] [PubMed] [Google Scholar]
- 147.Weiss RG, Gerstenblith G, Bottomley PA. ATP flux through creatine kinase in the normal, stressed, and failing human heart. Proc Natl Acad Sci USA. 2005;102:808–813. doi: 10.1073/pnas.0408962102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Udelson JE, Bonow RO, O'Gara PT, Maron BJ, Van Lingen A, Bacharach SL, Epstein SE. Verapamil prevents silent myocardial perfusion abnormalities during exercise in asymptomatic patients with hypertrophic cardiomyopathy. Circulation. 1989;79:1052–1060. doi: 10.1161/01.cir.79.5.1052. [DOI] [PubMed] [Google Scholar]
- 149.Cecchi F, Olivotto I, Gistri R, Lorenzoni R, Chiriatti G, Camici PG. Coronary microvascular dysfunction and prognosis in hypertrophic cardiomyopathy. N Engl J Med. 2003;349:1027–1035. doi: 10.1056/NEJMoa025050. [DOI] [PubMed] [Google Scholar]
- 150.Huke S, Knollmann BC. Increased myofilament Ca2+-sensitivity and arrhythmia susceptibility. J Mol Cell Cardiol. 2010;48:824–833. doi: 10.1016/j.yjmcc.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Abozguia K, Elliott P, McKenna W, Phan TT, Nallur-Shivu G, Ahmed I, Maher AR, Kaur K, Taylor J, Henning A, Ashrafian H, Watkins H, Frenneaux M. Metabolic modulator perhexiline corrects energy deficiency and improves exercise capacity in symptomatic hypertrophic cardiomyopathy. Circulation. 2010;122:1562–1569. doi: 10.1161/CIRCULATIONAHA.109.934059. [DOI] [PubMed] [Google Scholar]
- 152.Pound KM, Sorokina N, Ballal K, Berkich DA, Fasano M, Lanoue KF, Taegtmeyer H, O'Donnell JM, Lewandowski ED. Substrate-enzyme competition attenuates upregulated anaplerotic flux through malic enzyme in hypertrophied rat heart and restores triacylglyceride content: attenuating upregulated anaplerosis in hypertrophy. Circ Res. 2009;104:805–812. doi: 10.1161/CIRCRESAHA.108.189951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Ashrafian H, Czibik G, Bellahcene M, Aksentijevic D, Smith AC, Mitchell SJ, Dodd MS, Kirwan J, Byrne JJ, Ludwig C, Isackson H, Yavari A, Stottrup NB, Contractor H, Cahill TJ, Sahgal N, Ball DR, Birkler RI, Hargreaves I, Tennant DA, Land J, Lygate CA, Johannsen M, Kharbanda RK, Neubauer S, Redwood C, de Cabo R, Ahmet I, Talan M, Gunther UL, Robinson AJ, Viant MR, Pollard PJ, Tyler DJ, Watkins H. Fumarate is cardioprotective via activation of the Nrf2 antioxidant pathway. Cell Metab. 2012;15:361–371. doi: 10.1016/j.cmet.2012.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Ferrantini C, Belus A, Piroddi N, Scellini B, Tesi C, Poggesi C. Mechanical and energetic consequences of HCM-causing mutations. J Cardiovasc Transl Res. 2009;2:441–451. doi: 10.1007/s12265-009-9131-8. [DOI] [PubMed] [Google Scholar]