Abstract
To investigate the apoptosis-inducing effects of cetuximab combined with radiotherapy and hypothermia in human nasopharyngeal carcinoma CNE cells. CNE cells were treated with the radiation monotherapy, the radiation and hypothermia, the cetuximab and radiation, and the triple-combination treatment, respectively. MTT assay was performed to assess cell proliferation following treatments. Hoechst 33258 staining and flow cytometry analyses were used to detect apoptotic process. Western blot analysis was performed to determine the protein expression levels. Cetuximab monotherapy inhibited the proliferation of CNE cells. Hyperthermia alone inhibited EGFR expression, and prolonged hypothermia treatment resulted in declining EGFR expression levels in these cells. Moreover, Hoechst 33258 staining showed obvious apoptotic morphologies in the treatment groups. Flow cytometry analysis showed that the interventions dramatically increased the apoptosis rates in CNE cells, with the most potent effect for the triple-combination treatment. Western blot analysis showed that, in the treatment groups, the expression levels of Bax were increased, while the expression levels of Bcl-2 were decreased, leading to significantly elevated Bax/Bcl-2 ratios in these groups, with the highest ratio for the triple-combination treatment. Cetuximab combined with radiotherapy and hypothermia treatments could efficiently inhibit the proliferation of CNE cells, and enhance the cellular apoptotic processes via regulating the expression levels of Bax and Bcl-2. Our findings provide experimental evidence for the application of the combination therapy in clinical treatment of nasopharyngeal carcinoma.
Keywords: Cetuximab, hyperthermia, radiotherapy, combination treatment, nasopharyngeal carcinoma
Introduction
Nasopharyngeal carcinoma (NPC) is a common head and neck cancer in South China, with 10-30 cases per 100,000 in the general population [1]. Although radiation therapy has been used as the primary treatment option for NPC, the efficiency is not satisfactory [2]. Therefore, other therapeutic options, including biological gene therapy, targeted therapy, hyperthermia, and combination therapies [3,4], have been under investigation for NPC treatment for decades.
At present, targeted therapy has been one of the most active research areas for cancer treatment. Monoclonal antibody cetuximab exerts beneficial effects in the clinical treatment of NPC [5,6]. Cetuximab could specifically bind to and degrade the epidermal growth factor receptor (EGFR). In addition, cetuximab inhibits the binding of EGFR to its ligands and blocks the downstream signal transduction pathways, leading to proliferation inhibition and apoptosis induction in tumor cells [7].
On the other hand, hyperthermia treatment has attracted great attention in recent years, due to its high efficiency and well tolerance [8]. Hyperthermia could cause protein denaturation and induce apoptotic process in tumor cells [9,10]. Moreover, hyperthermia combined with radiotherapy has been associated with improved clinical outcomes. It has been reported that the efficiency of the combination treatment was superior to each of radiotherapy and hyperthermia treatment alone [11,12]. In addition, hyperthermia combined with chemotherapy would elevate the drug concentration within the tumor and enhance the anti-cancer effects. Based on these findings, it is of great significance to investigate the effects of cetuximab combined with radiotherapy and hypothermia treatments on NPC.
In this study, human nasopharyngeal carcinoma (CNE) cells were treated with cetuximab combined with radiotherapy and hypothermia, and the apoptosis-inducing effects of the combination treatments were investigated. Our findings provide experimental evidence for the application of the combination therapy in clinical treatment of NPC.
Materials and methods
Cell culture and grouping
Human nasopharyngeal carcinoma (CNE) cells were purchased from Shanghai Institute of Cell Bank, Chinese Academy of Science (Shanghai, China). CNE cells were grown in RPMI1640 complete medium (Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (FBS; Zhejiang Tianhang Biological Technology Co., Ltd., Hangzhou, Zhejiang, China), in a 37°C, 5% CO2 humidified incubator (Thermo Electron, Marietta, OH, USA). The culture medium was changed every 2 to 3 days.
These cells were divided into the following groups: (1) the control group that was free from intervention; (2) the radiotherapy groups that were subjected to radiation at 2, 4, 8, and 12 Gy, respectively; (3) the radiation and hypothermia groups that were first subjected to radiation at 2, 4, 8, 12, and 20 Gy, respectively, followed by hypothermia treatment at 41.8°C for 60 min; (4) the cetuximab and radiation groups that were first treated with 10 μg/ml cetuximab (ImClone, Branchburg, NJ, USA) for 48 h, and then subjected to radiation treatment at 2, 4, 8, and 12 Gy, respectively; (5) the triple-combination treatment groups, which were first treated with 10 μg/ml cetuximab for 48 h, and subsequently subjected to radiation (2, 4, 8, and 12 Gy, respectively) and hypothermia (43°C for 30 min) treatments.
MTT assay
The MTT assay was performed to determine the working concentration of cetuximab on CNE cells. Briefly, CNE cells in logarithmic growth phase were seeded onto 96-well culture plates at a density of 3 × 103 cells/well. These cells were subjected to the cetuximab treatment at different concentrations (0.05, 0.5, 1, 2, 10, 20, 100, and 200 μg/ml, respectively) for 48 h. 20 μl MTT (5 mg/ml; Sigma, St. Louis, MO, USA) was added into each well. After 4-h incubation, 100 μl DMSO (Amresco, Cleveland, Ohio, USA) was added, and the plate was shaken for 5 min. Absorbance (OD) at 490 nm was read on an automatic microplate reader (Biotek, Winooski, VT, USA), with a reference wavelength of 620 nm. Cell proliferation inhibition rate was calculated according to the following equation: cell proliferation inhibition rate (%) = (ODcontrol - ODtreatment) / ODcontrol × 100%. The treatment concentration between inhibiting concentration 20-30% (IC20 - IC30) was used in the following experiments.
Hoechst 33258 staining
Cells were seeded onto sterile coverslips placed in 30 mm2 culture dishes, and incubated overnight. After washed twice with double distilled water, cells were stained with 5 mg/L Hoechst 33258 (Sigma) for 5 min, and then observed under a fluorescence microscope (Leica, Leica, Germany).
Flow cytometry
CNE cells in logarithmic growth phase were seeded onto a 25 mm2 culture bottle at a density of 2 × 106 cells/bottle. Following indicated treatments as mentioned above, cells were further cultured for 24 h. For flow cytometry analysis, cells were collected and washed twice with pre-cold PBS. 400 μl binding buffer solution was used to re-suspend these cells to achieve a final concentration of 4 × 105 cells/ml. 5 μl annexin V-FITC (Bipec Biopharma Corp., Cambridge, MA, USA) was added to incubate the cells in dark at 4°C for 15 min, and then 10 μl PI solution (20 μg/ml) was added for incubation in dark at 4°C for 5 min. The apoptotic processes were detected by a flow cytometer.
Western blot analysis
Cells were harvested and lysed with 400 μl lysis solution (KeyGen, Nanjing, Jiangsu, China) containing PMSF on ice for 30 min. After centrifugation at 12,000 rpm at 4°C for 5 min, the supernatant was collected, and the concentration was adjusted with a BCA kit. Proteins were subjected to SDS-PAGE, and then electronically transferred onto a PVDF membrane. The blot was blocked with TBST containing 10% non-fat milk at room temperature for 1 h. Then the membrane was incubated with Bax (1:200 dilution; Santa Cruz, Santa Cruz, California), Bcl-2 (1:200 dilution; Santa Cruz), EGFR (1:200 dilution; Santa Cruz), and b-actin (1:200 dilution; Multisciences, Hangzhou, Zhejiang, China) primary antibodies, respectively, at 4°C overnight. After washed with TBST, goat anti-mouse IgG-AP (1:500 dilution; Sino-American Biotechnology Co., Ltd., Luoyang, Henan, China) was used to incubate the membrane at room temperature for 1 h, following by AP chromogenic reaction (Promega, Madison, WI, USA). b-actin was used as control. The optical density of protein bands were analyzed by a gel image analysis system (JS-380A; Shanghai Peiqing Science & Technology Co., Ltd., Shanghai, China).
To detect the effects of hypothermia on the expression of EGFR in CNE cells, these cells were subjected to hypothermia treatment for 0 min, 15 min, 30 min, 2 h, 6 h, 12 h, and 24 h, respectively, and then the protein expression levels of EGFR were assessed by the Western blot analysis.
Statistical analysis
Data were expressed as mean ± SD. SPSS15.0 software was used for statistical analysis. The t-test was used for the pair-wise comparison, and ANOVA was used for the multiple comparison. P < 0.05 was considered statistically significant.
Results
Effects of cetuximab monotherapy on CNE cell proliferation
To investigate the effects of cetuximab treatment on CNE cell proliferation, the MTT assay was performed. CNE cells were treated with cetuximab at 0.05, 0.5, 1, 2, 10, 20, 100, and 200 μg/ml, respectively, for 48 h, followed by cell proliferation measurement. Our results indicated that cetuximab treatments inhibited the proliferation of CNE cells, and the cell proliferation rate was increasing as the treatment concentration increased (Figure 1). According to the results, the treatment concentration between IC20 and IC30 values, i.e., 10 μg/ml, was used as the working concentration in the following experiments.
Figure 1.
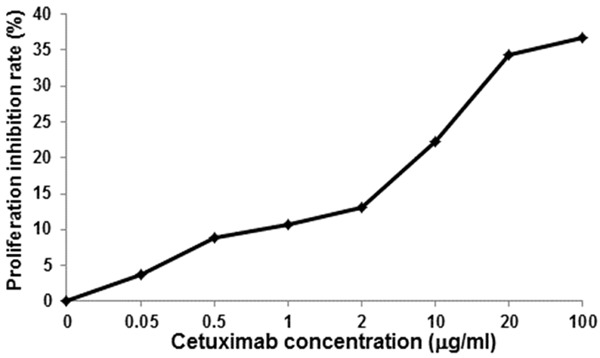
Cetuximab monotherapy inhibits the proliferation of CNE cells. MTT assay was performed to investigate the effects of cetuximab treatment alone on CNE cell proliferation, and the proliferation inhibition rates were calculated. CNE cells were treated with cetuximab at 0.05, 0.5, 1, 2, 10, 20, 100, and 200 μg/ml, respectively, for 48 h, followed by cell proliferation assessment.
Effects of hyperthermia monotherapy on EGFR expression in CNE cells
Next, we investigated the effects of hyperthermia on the expression of EGFR (the target of cetuximab) in CNE cells. These cells were subjected to hypothermia treatment for 0 min, 15 min, 30 min, 2 h, 6 h, 12 h, and 24 h, respectively, and the EGFR expression level was detected by Western blot analysis. Our result showed that hyperthermia could inhibit the protein expression of EGFR in CNE cells. Moreover, prolonged hypothermia treatment resulted in declining expression levels of EGFR in CNE cells (Figure 2). These results indicate that hyperthermia treatment could inhibit the EGFR expression in CNE cells. Since cetuximab has been known to target EGFR, synergistic effects would be expected for the combination treatments.
Figure 2.
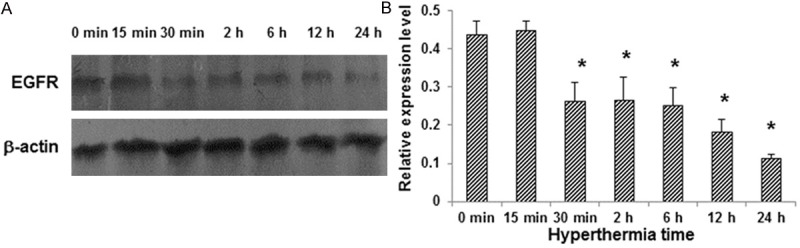
Hyperthermia inhibits the EGFR expression in CNE cells. A. CNE cells were subjected to hypothermia treatment for 0 min, 15 min, 30 min, 2 h, 6 h, 12 h, and 24 h, respectively, and the EGFR expression levels were detected by Western blot analysis. B. Statistical analysis of the EGFR expression levels in CNE cells. Compared with the control group, *P < 0.05.
Effects of cetuximab combination treatments on apoptotic process in CNE cells
In order to investigate the effects of cetuximab combination treatments on apoptosis in CNE cells, Hoechst 33258 staining and flow cytometry analyses were performed. Hoechst 33258 staining showed regular cellular morphology in control CNE cells, without apoptotic features (Figure 3A). In contrast, in the treatment groups (cetuximab, radiotherapy, hypothermia, and combination treatments, respectively), obvious apoptotic phenotypes were observed, including nuclear condensation. Moreover, the triple-combination treatment lead to the most significant morphological changes in CNE cells, where the apoptotic bodies were formed (Figure 3A). In addition, results from annexin V-FITC/PI staining and flow cytometry analysis showed that, compared with the control group, the treatments dramatically increased the apoptosis rates in CNE cells (Figure 3B, 3C). Moreover, the apoptosis rates in the triple-combination treatment groups were significantly higher than the groups treated with radiation or hypothermia treatment alone, as well as the group treated with cetuximab and radiation treatments (Figure 3C) (P < 0.05). Taken together, these results suggest that cetuximab combination therapy could enhance the apoptotic process in CNE cells, with the most obvious effect for the triple-combination treatment.
Figure 3.
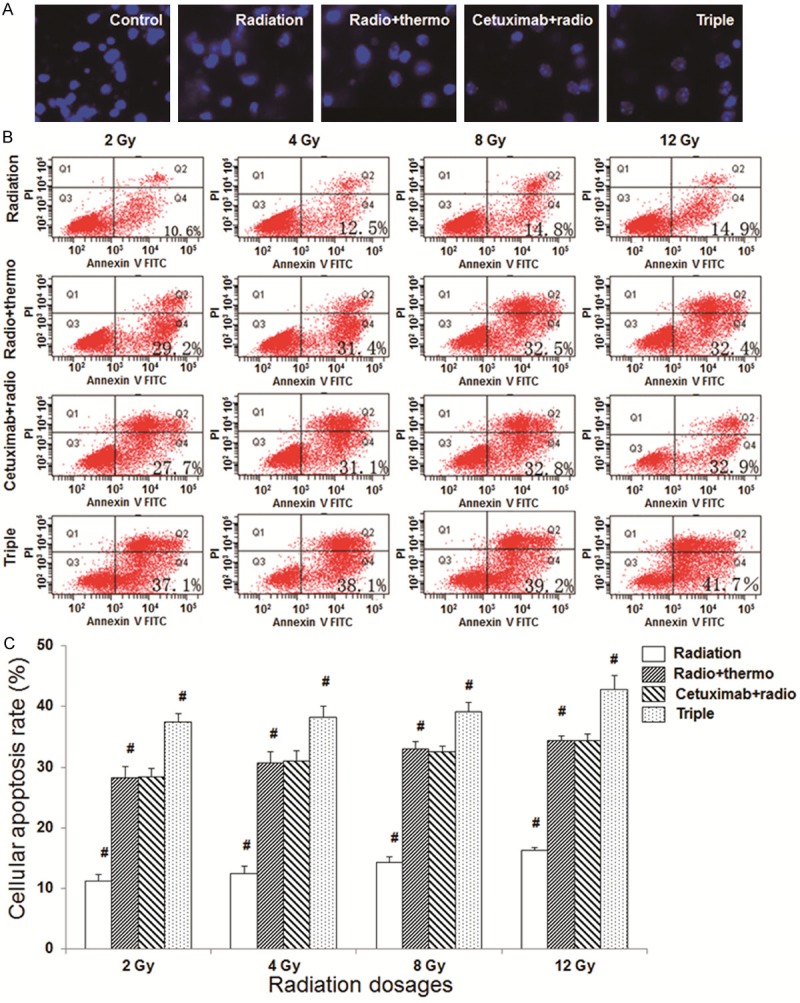
Cetuximab combination treatment promotes the apoptotic process in CNE cells. A. Hoechst 33258 staining was performed to detect the apoptosis in CNE cells (×200). B. Annexin V-FITC/PI staining and flow cytometry analyses were performed to detect apoptosis in CNE cells following treatments. The radiation dosages were set at 2, 4, 8, and 12 Gy, respectively. C. Statistical analysis of the apoptosis rates in CNE cells following treatments. Control, the control group; radiation, the radiation monotherapy group; radio + thermo, the radiation and hypothermia group; cetuximab + radio, the cetuximab and radiation group; triple, cetuximab combined with radiotherapy and hypothermia. Compared with the previous group, #P < 0.05.
Effects of cetuximab combination therapy on Bax and Bcl-2 expression levels in CNE cells
To further investigate the effects of cetuximab combination treatments on apoptosis in CNE cells, the expression levels of Bax and Bcl-2 were detected by Western blot analysis (Figure 4). Our results indicated that the expression levels of Bax were increased in the following order: the control group, the radiation monotherapy group, the radiation and hypothermia group, the cetuximab and radiation group, and the triple-combination treatment group, while the expression levels of Bcl-2 were decreased in the very same order, resulting in the increasing trend in the Bax/Bcl-2 ratio in these groups. The Bax/Bcl-2 ratios in all the intervention groups were significantly higher than the control group (P < 0.05), with the highest ratio for the triple-combination treatment group (P < 0.05).
Figure 4.
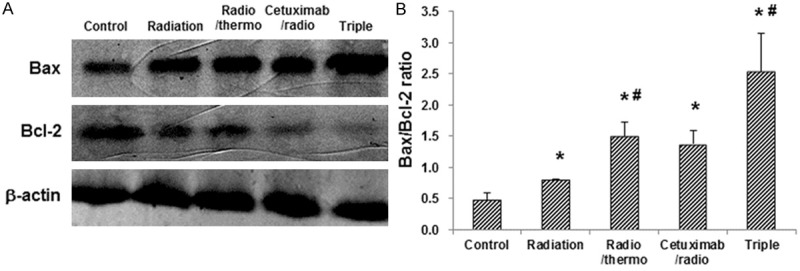
Cetuximab combination therapy regulates the Bax/Bcl-2 expression levels in CNE cells. A. CNE cells were subjected to the indicated treatments, and the expression levels of Bax and Bcl-2 in these cells were assessed by Western blot analysis, respectively. B. Statistical analysis of the Bax/Bcl-2 ratios in CNE cells following treatments. Compared with the control group, *P < 0.05; compared with the previous group, #P < 0.05.
Discussion
With the rapid development of molecular biology in recent years, the pathogenesis and development of tumor has been found to not only be associated with cell proliferation disturbance, but also with the dysfunction of apoptosis. Accordingly, apoptosis induction is of great significance in the clinical treatment of cancers [13]. Epidermal growth factor receptor (EGFR) is one member of ErbB cell surface receptor family, which is highly expressed and activated in various tumors, including nasopharyngeal carcinomas [14]. The extracellular domain of the receptor could bind to and phosphorylate the ligands, resulting in the formation of homo- or heterodimers, activating a series of downstream signaling molecules and thereby promoting cell proliferation and inhibiting apoptosis [15-17]. Therefore, EGFR has been recognized as one of the common targets in the treatment of tumors.
On the other hand, hyperthermia is attracting more and more attention in cancer treatment recently [8]. According to the treatment temperature, hyperthermia can be roughly divided into high-temperature (> 43°C) and low-temperature (≤ 43°C) treatments. High-temperature hyperthermia usually causes protein denaturation, leading to cell death, while low-temperature hyperthermia could induce cellular apoptosis [9,10]. It has been confirmed that, hyperthermia at 43°C for 30 min could induce widespread apoptotic process [18], and the combination of hyperthermia and chemotherapy would greatly elevate the apoptosis rate in tumor cells [19]. There are several advantages for hyperthermia in the cancer treatment: (1) Compared with normal cells, tumor cells are less tolerant to thermal treatments, which would generally be killed by 2 h treatment at 42°C. Since the heat sensitivity is higher for the tumor cells than the normal cells, the temperature in the tumor tissue following hyperthermia is usually higher than surrounding normal tumor tissues (37°C). Therefore, hyperthermia within a reasonable temperature range could directly kill the tumor cells, without causing damages to surrounding tissues. (2) Some tumor cells might be insensitive to radiation therapy, including cells in the S phase and the hypoxic cells. These cells are highly sensitive to the hyperthermia treatment. (3) Hyperthermia could partially repair the damages caused by the radiotherapy. (4) Since the tumor tissues are always linked with the abundant peripheral blood supply, the killing effects of hyperthermia on the peripheral cells are not as potent as that on the tumor cells. The treatment failure of hyperthermia is probably the tumor recurrence in surrounding tissues, while radiotherapy failure is mainly due to the local recurrence within the tumors. Therefore, the rational combination of hyperthermia and radiation therapies might overcome the disadvantages of the monotherapy [20,21]. (5) Hyperthermia could up-regulate the pro-apoptosis genes and down-regulate the apoptosis-suppressing genes [22]. (6) Hyperthermia enhances the cytotoxicity of certain chemotherapy drugs, such as cisplatin. It has been confirmed in vitro that the thermo treatment at 42°C for 2 h could enhance anti-cancer effects of several chemotherapy drugs by 10 to 100 times. Based on these backgrounds, it could be expected that the combination treatments would lead to synergistic effects in cancer treatment.
In this study, cetuximab combined with radiotherapy and hyperthermia was used to treat CNE cells. Our results showed that, the combination treatments could induce apoptotic process in these cells, with the most potent effects in the triple-combination treatment group. These results suggest that the combination treatments would provide synergistic effects in treating nasopharyngeal carcinoma cells, via promoting cellular apoptosis. Bcl-2 family members play important roles in the regulation of apoptosis. According to the protein function, Bcl-2 protein family could be divided into the following categories: Bcl-2, improving cell survival and inhibiting cellular apoptosis, and Bax, in contrast, promoting apoptotic process [23]. The expression levels of Bax and Bcl-2 in CNE cells following treatments were detected, and the results showed that, compared with the control group, all the treatments would up-regulate the expression levels of Bax, and down-regulate the Bcl-2 expression. Therefore, the Bax/Bcl-2 ratios were increased in the following order: the control group, the radiation monotherapy group, the radiation and hyperthermia treatment group, the cetuximab and radiation treatment group, and the triple-combination treatment group. These results suggest that cetuximab combined with radiation and hyperthermia treatments would induce apoptosis in CNE cells, probably through regulating the expression levels of Bax and Bcl-2.
Cetuximab could down-regulate the expression of EGFR, and block the binding of EGFR to its ligands, resulting in cellular apoptosis. In fact, EGFR is a transmembrane protein, whose membrane structure would be damaged by hyperthermia. Therefore, we speculate that hyperthermia might also regulate the expression of EGFR to promote apoptosis, achieving synergistic effects together with the cetuximab treatment. Our results showed that the expression levels of EFGR were decreased along with the hyperthermia treatment, which would be further declined when hyperthermia was combined with cetuximab. These results indicate that the synergistic effects between hyperthermia and cetuximab would be associated with the down-regulated expression levels of EGFR.
In conclusion, cetuximab combined with radiotherapy and hypothermia treatments could efficiently inhibit the proliferation of CNE cells, and enhance the cellular apoptotic processes via regulating the expression levels of Bax and Bcl-2. Our findings provide experimental evidence for the application of the combination treatments in clinical treatment of NPC.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81460460) and the Guangxi Health Research Project (200626).
Disclosure of conflict of interest
None.
References
- 1.Shang XL, Xie ZQ, Huang ZB. The risk factor of nasopharyngeal carcinoma. Modern Prevent Med. 2008;35:206–7. [Google Scholar]
- 2.Zhao C, Guan XX. The clinical research of combined therapy for nasopharyngeal carcinoma. Chinese Journal of Cancer Prevention and Treatment. 2006;13:0001–0004. [Google Scholar]
- 3.Ren H, Yang BF, Rainov NG. Receptor tyrosine kinases as therapeutic targets in malignant glioma. Rev Recent Clin Trials. 2007;2:87–101. doi: 10.2174/157488707780599384. [DOI] [PubMed] [Google Scholar]
- 4.Metro G, Finocchiaro G, Toschi L, Bartolini S, Magrini E, Cancellieri A, Trisolini R, Castaldini L, Tallini G, Crino L, Cappuzzo F. Epidermal growth factor receptor (EGFR) targeted therapies in non-small cell lung cancer (NSCLC) Rev Recent Clin Trials. 2006;1:1–13. doi: 10.2174/157488706775246157. [DOI] [PubMed] [Google Scholar]
- 5.Astsaturov I, Cohen RB, Harari P. EGFR-targeting monoclonal antibodies in head and neck cancer. Curr Cancer Drug Targets. 2007;7:650–65. doi: 10.2174/156800907782418365. [DOI] [PubMed] [Google Scholar]
- 6.Chan AT, Hsu MM, Goh BC, Hui EP, Liu TW, Millward MJ, Hong RL, Whang-Peng J, Ma BB, To KF, Mueser M, Amellal N, Lin X, Chang AY. Multicenter, phase II study of cetuximab in combination with carboplatin in patients with recurrent or metastatic nasopharyngeal carcinoma. J. Clin. Oncol. 2005;23:3568–76. doi: 10.1200/JCO.2005.02.147. [DOI] [PubMed] [Google Scholar]
- 7.Dutta PR, Maity A. Cellular responses to EGFR inhibitors and their relevance to cancer therapy. Cancer Lett. 2007;254:165–77. doi: 10.1016/j.canlet.2007.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moyer HR, Delman KA. The role of hyperthermia in optimizing tumor response to regional therapy. Int J Hyperthermia. 2008;24:251–61. doi: 10.1080/02656730701772480. [DOI] [PubMed] [Google Scholar]
- 9.Hildebrandt B, Wust P. The biologic rationale of hyperthermia. Cancer Treat Res. 2007;134:171–84. doi: 10.1007/978-0-387-48993-3_10. [DOI] [PubMed] [Google Scholar]
- 10.Peng YF, Zheng MH. Cellular and molecular mechanism and application of hyperthermia for tumor therapy. World Chinese Journal of Digestology. 2007;15:1319–23. [Google Scholar]
- 11.Li ZQ, Zhang MW, Li J, et al. Effects of radiochemotherapy and radiochemotherapy combined with hyperthermia in treating cervical lymph node metastasis of nasopharyngeal carcinoma. J Pract Med. 2004;20:532633. [Google Scholar]
- 12.Zheng Z, Zhang Y, Pan JJ, et al. Efficacy of heating treatment for cervical lymph node metastasis of nasopharyngeal carcinoma. Fujian Medical University. 2006;40:629631. [Google Scholar]
- 13.Melet A, Song K, Bucur O, Jagani Z, Grassian AR, Khosravi-Far R. Apoptotic pathways in tumor progression and therapy. Adv Exp Med Biol. 2008;615:47–79. doi: 10.1007/978-1-4020-6554-5_4. [DOI] [PubMed] [Google Scholar]
- 14.Mao RJ, Tan YM. Expressions of EGFR and p16 protein in nasopharyngeal cancer. Prac J Cancer. 1999;14:182–184. [Google Scholar]
- 15.Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG, Louis DN, Christiani DC, Settleman J, Haber DA. Activating mutations in the epidermal growth factor receptor underlying respon-siveness of non-small-cell lung cancer to Gefitinib. N Engl J Med. 2004;350:2129–39. doi: 10.1056/NEJMoa040938. [DOI] [PubMed] [Google Scholar]
- 16.Modjtahedi H, Affleck K, Stubberfield C, Dean C. EGFR blockade by tyrosine kinase inhibitor or monoclonal antibody inhibits growth, directs terminal differentiation and induces apoptosis in the human squamous cell carcinoma HN5. Int J Oncol. 1998;13:335–42. doi: 10.3892/ijo.13.2.335. [DOI] [PubMed] [Google Scholar]
- 17.Sordella R, Bell DW, Haber DA, Settleman J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science. 2004;305:1163–7. doi: 10.1126/science.1101637. [DOI] [PubMed] [Google Scholar]
- 18.Burd R, Dziedzic TS, Xu Y, Caligiuri MA, Subjeck JR, Repasky EA. Tumor cell apoptosis, lymphocyte recruitment and tumor vascular changes are induced by low temperature,long duration (fever-like) whole body hyperthermia. J Cell Physiol. 1998;177:137–47. doi: 10.1002/(SICI)1097-4652(199810)177:1<137::AID-JCP15>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 19.Tang R, Zhu ZG, Qu Y, Li JF, Ji YB, Cai Q, Liu BY, Yan M, Yin HR, Lin YZ. The impact of hyperthermic chemotherapy on human gastric cancer cell lines: preliminary results. Oncol Rep. 2006;16:631–41. [PubMed] [Google Scholar]
- 20.Constine LS, Milano MT, Friedman D, et al. Late Effects of Cancer treatment on Normal Tissues. In: Halperin EC, Perez CA, Brady LW, et al., editors. Perez and Brady’s Principles and Pra- ctice of Radiation Oncology. 5th edition. Philadelphia, Pa: Lippincott Williams & Wilkins; 2008. pp. 320–355. [Google Scholar]
- 21.Li G, Mitsumori M, Ogura M, Horii N, Kawamura S, Masunaga S, Nagata Y, Hiraoka M. Local hyperthermia combined with external irradiation for regional breast carcinoma. Int J Clin Oncol. 2004;9:179–83. doi: 10.1007/s10147-004-0395-3. [DOI] [PubMed] [Google Scholar]
- 22.Nikfarjam M, Muralidharan V, Malcontenti-Wilson C, Christophi C. The apoptotic response of liver and colorectal liver metastases to focal hyperthermic injury. Anticancer Res. 2005;25:1413–9. [PubMed] [Google Scholar]
- 23.Adams JM, Cory S. Bcl-2-regulated apoptosis: mechanism and therapeutic potential. Curr Opin Immunol. 2007;19:488–96. doi: 10.1016/j.coi.2007.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]


