Abstract
Management of the pregnant patient on peritoneal dialysis (PD) is potentially challenging because uterine enlargement may negatively affect catheter function and prescribed dwell volumes. Additional reports of the management of these patients are needed. Here, we describe a near-full-term delivery in a 27-year-old woman who had been on dialysis for 7 years. Peritoneal dialysis was continued during the entire pregnancy. In the third trimester, a higher delivered automated PD volume allowed for adequate clearance and control of volume status. A decision to hospitalize the patient to limit activity and facilitate the delivery of increased dialysate is believed to have contributed to the successful outcome for mother and infant. Our report discusses the management of this patient and reviews published dialysis prescriptions used during the third trimester of pregnancy in patients treated with PD.
Keywords: End-stage renal disease, automated peritoneal dialysis, pregnancy, hypertension, hemoperitoneum
Women with chronic kidney disease (CKD) who become pregnant are at increased risk of adverse maternal and fetal outcomes. A recent systematic review of pregnancy outcomes in women with CKD not requiring dialysis compared with women not having CKD showed a doubled risk for adverse maternal outcomes; in addition, premature births in the CKD cohort were twice as frequent (1). With the increased background risk seen in CKD, it is probable that patients with end-stage renal disease (ESRD) are at even higher risk. Women with ESRD rarely become pregnant, and of those who do, few carry the fetus to full term. Infertility because of amenorrhea and anovulation is a contributing factor, as is interference by intraperitoneal dialysate with migration of the ovum into the fallopian tubes (2). Pregnancy has been reported in 0.5% – 1.4% of female patients of childbearing age with ESRD, and despite that rarity, all such patients should receive pregnancy counseling (3). In most descriptions of pregnancy in ESRD, the patients were managed with hemodialysis (HD); the literature contains far fewer cases of a pregnant patient being managed with peritoneal dialysis (PD).
Despite the higher risk of these pregnancies, it is generally believed that maternal and fetal outcomes have improved in more modern cohorts. Infant survival was reported to be more than 50% in patients with ESRD in 1998, but recent reports of the use of intensified HD regimens and many PD reports suggest far better outcomes (4,5). For the PD patient who has become pregnant, PD offers specific benefits for management of the pregnancy, such as more continuous and gentle daily ultrafiltration, metabolic balance without the fluctuations noted in intermittent therapies, less anemia, avoidance of systemic anticoagulation, and a more liberal diet for maintaining maternal nutrition (6,7). But those potential benefits might be offset by uterine distention during the 3rd trimester, which complicates management of the pregnant patient on PD. Abdominal fullness necessitating a reduction in the dwell volume combined with increased clearance requirements, catheter displacement, drain pain, and dialysate flow disturbances can make management with PD more challenging. Nevertheless, a review of the literature shows successful pregnancies in ESRD patients managed with PD (Table 1).
TABLE 1.
Pregnancy Outcomes in End-Stage Renal Disease Patients Treated with Peritoneal Dialysis
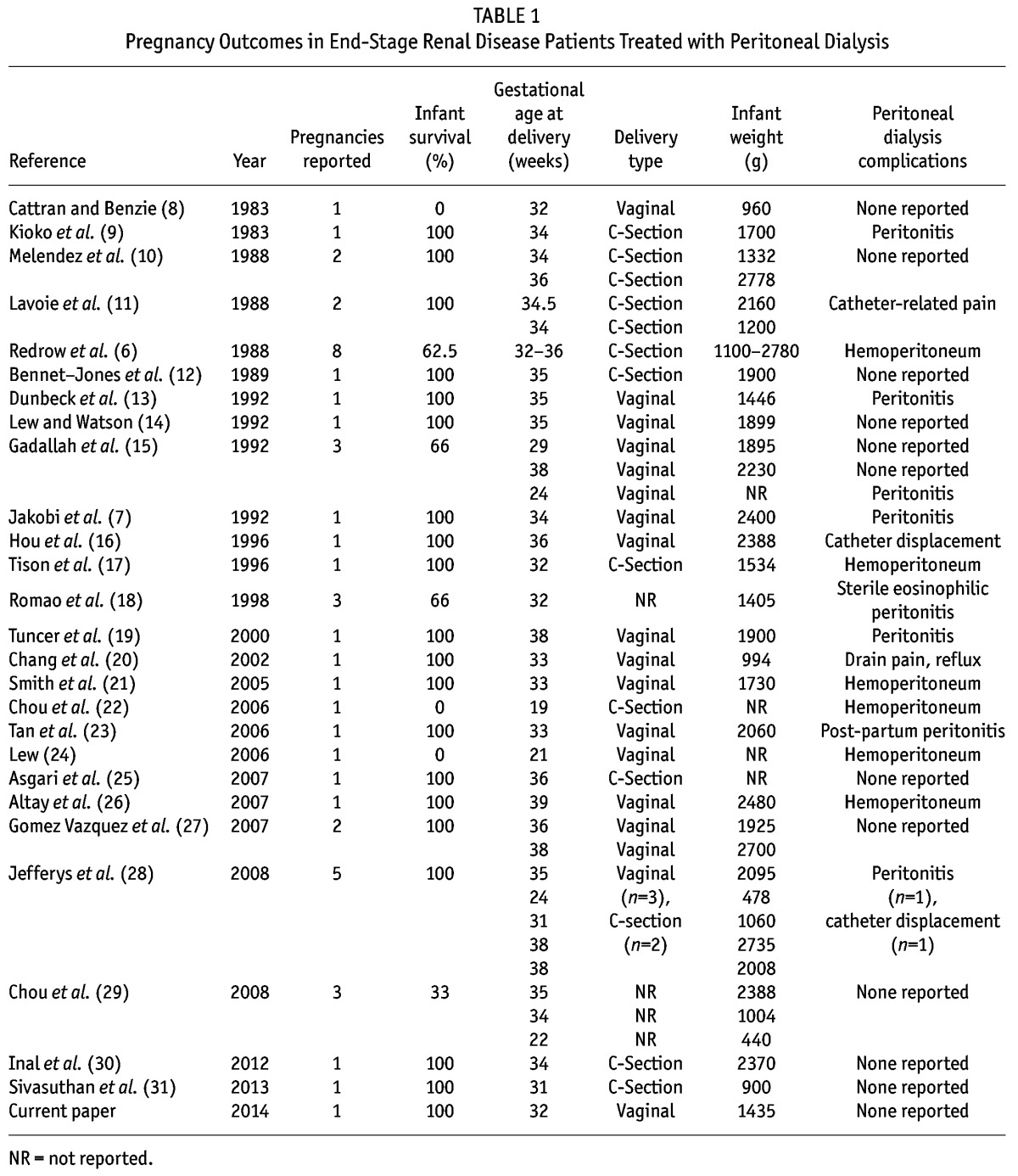
To add to that literature, we report the case of a 27-year-old woman who had been on dialysis for 7 years. She was maintained on HD for 4 years and then converted to PD. In the third year of her PD course, she became pregnant, and the decision was made to continue with PD throughout the pregnancy. Here, we describe the management of this patient, with particular detail about the PD prescription used during the 3rd trimester of pregnancy.
In addition to the specifics of the case report, this review focuses on dialysis prescription during the 3rd trimester. As mentioned earlier, uterine enlargement commonly requires a change in the PD prescription to allow for a reduction in the infused dialysate volume. To maintain adequate clearances, an increase in the total infused volume and prolonged APD schedules have been described. Uterine enlargement leading to catheter-related drain pain has been addressed with tidal PD prescriptions. We review the 3rd trimester PD prescriptions reported in the literature.
Case Report
Our patient, a 27-year-old Hispanic woman, had a past renal history of biopsy-proven anti-glomerular basement membrane disease that progressed to ESRD. She initiated HD in January 2004 at age 20. Her initial dialysis access was a tunneled vascular access catheter; she then transitioned to a forearm graft. She became pregnant shortly after initiating HD and delivered a baby in January 2005. There was no pre-eclampsia. The baby was delivered at 34 weeks and weighed 2.24 kg. The induced delivery was completed vaginally with no complications.
This patient elected to convert to PD in June 2010, initiating PD after a first failed access and a second operation for catheter placement. Shortly after PD start, she was found to be pregnant. Her prior menstrual cycles had been regular despite the ESRD, and on missing a period, she sought pregnancy testing, which was positive. The early course of her pregnancy was uneventful.
At 27 weeks, examination revealed a blood pressure of 124/86 mmHg, pulse 84 bpm, and respirations 12/min. The patient measured 160 cm and weighed 69 kg. The physical exam was notable for a regular pulse, no cardiac murmurs, and clear lung fields. The abdominal exam revealed a gravid uterus and PD catheter with the exit site in the left lower quadrant. The exit site appeared clean, but the subcutaneous cuff was beginning to protrude through the skin. The lower extremity exam showed very slight edema bilaterally.
At 27 weeks, the decision was made to admit the patient to the obstetrics and gynecology service in the hospital and to perform increased PD exchanges. In discussions between the medical caregivers, a decision was reached to target a delivered dialysis dose sufficient to maintain the patient’s blood urea nitrogen (BUN) at 50 mg/dL and her creatinine at 5.0 mg/dL.
Because of the size of the uterus, the initial prescription used a cycler that could deliver frequent but reduced dialysate volumes. Each evening, she was connected to the cycler for 12 hours, with eight 1-L dialysate exchanges being performed overnight. She was given extension tubing that allowed her, if needed, to ambulate within the room while attached to the cycler, thus giving her more freedom of movement during the prolonged time attached to the device. Her dialysate exchanges used combinations of 1.5% and 2.5% dextrose depending on her ultrafiltration needs, but the goal was to use the lowest dextrose concentration possible while maintaining volume status.
In addition to cycler therapy, the patient performed 4 daytime manual 1-L exchanges in her room. These low-volume manual exchanges were easy to perform, requiring approximately 10 minutes per exchange. However, the patient soon became unwilling to perform the 4 daytime manual exchanges, and so the decision was reached to extend her time on the cycler to 16 hours, delivering 12 cycles of 1-L dwell volumes. During the subsequent week, day-to-day decisions were made with the patient about the use of a longer time on the cycler or a shorter time combined with the performance of manual daytime exchanges. The daily variations ranged from 14 to 16 hours on the cycler, with 0 – 2 midday exchanges. She maintained urine output of up to 500 mL daily.
At 29 weeks, the overnight cycler prescription varied from 15 to 20 hours, with 14 – 18 exchanges of 750 mL dwell volumes. Beyond 30 weeks’ gestation, she was connected to the cycler for 18 hours daily, receiving up to 20 dialysate exchanges of 750 mL each.
During this PD regimen, the machine would occasionally sound an alarm either because of a kink in the dialysate tubing or because of the patient’s position. The alarms were rectified on each occasion by the patient checking the tubing pathway and changing position in bed. Otherwise the exchanges were uneventful. The patient was allowed to stop the cycler and disconnect if testing was required outside of the room. During the daily period when she was not connected to the cycler or doing manual exchanges, the patient was allowed to ambulate the room and hospital hallways. Her BUN measurements ranged from 39 mg/dL to 49 mg/dL for the 8 weeks preceding delivery.
At 32 weeks and 4 days, she developed abrupt onset of uterine contractions, and the amniotic sac broke. A Cesarian section had been planned, but before it could be arranged, the patient rapidly delivered the fetus’s feet and chest via the vagina, and the obstetrician continued to deliver the head with minor difficulty. The entire delivery was estimated to have occurred within 90 seconds. Initial Apgar scores were 3 and 4, and the infant’s weight was 1435 g. The infant was taken to the neonatal unit and received care for 8 weeks before discharge.
In the first week after delivery, the patient was maintained using a similar PD prescription of lower volumes and more frequent cycles (12 hours on the cycler with 10 exchanges of 1.2 – 1.5 L). She was discharged to resume her usual continuous ambulatory PD (CAPD) regimen.
Discussion
Successful pregnancy in PD has been reported in the literature, and yet the published reports surely represent some publication bias because of reporting only more favorable clinical outcomes (Table 1). Many of the pregnancies are notable for lower birthweight infants. Nevertheless, the reports document successful maternal and infant outcomes of pregnancy, and the outcomes are improved in more current cohorts compared with historical controls (32,33).
Delivering PD therapy in the advanced stages of pregnancy can be challenging. The enlarging uterus can create increased complaints of fullness, resulting in the need to lower dialysate dwell volumes. The enlarging uterus might contribute to slower drain times, drain pain, and gastroesophageal reflux. Those symptoms have been managed by reducing the infused dwell volume and using tidal regimens. Table 2 summarizes PD prescriptions used during the 3rd trimester in published cases and case series. Continuous ambulatory PD and automated PD (APD) regimens have both been described. Some reports noted the maintenance of full 2-L dwell volumes during pregnancy (18). However, most reports describe a reduction in the infused dwell volume, with or without additional exchanges. All reports suggested continuous therapy during the 3rd trimester, with no reports of a dry day during APD or a dry night during CAPD.
TABLE 2.
Peritoneal Dialysis Prescription During the Third Trimester

As noted in Table 2, many APD reports document PD prescriptions with reduced dwell volumes combined with an increased number of cycles. Increased total daily volumes were used in most cases. A common practice appears to be to increase the total daily dialysis volume in an attempt to increase clearance during pregnancy to account for urea generation by the fetus and to reduce maternal uremia (34). This empiric increase in total delivered volume in PD is supported somewhat in the HD literature. Asam Asamiya and colleagues reported on pregnancy outcomes in 28 patients receiving HD (35). They noted that the average BUN measurement was significantly lower in the successful pregnancy group than in the unsuccessful group. They also noted that the average hemoglobin level was significantly higher in the successful group than in the unsuccessful group (9.6 g/dL vs 8.3 g/dL). By performing a linear regression analysis of the relationship of maternal BUN with infant birthweight and gestational age, they determined that an acceptable gestational age of 32 weeks corresponded to a maternal BUN of 48 mg/dL. They recommended that an appropriate goal would be to keep maternal pre-dialysis BUN below 50 mg/dL and speculated that favorable fetal outcomes might be attributable to a less azotemic environment. The authors recommended that the foregoing targets be maintained as early in the pregnancy as can be instituted. Interestingly, a similar BUN recommendation can be found in the HD literature dating to 1991 (36).
Threshold BUN levels in pregnant PD patients that would improve outcomes are unknown, but based on the HD data, it might be reasonable to target a BUN lower than this HD target. The recommended BUN of less than 50 mg/dL in HD might need to be adjusted lower for PD in light of the understanding of the “peak urea hypothesis” (37). The lower urea target for PD is currently unknown, as is the targeted weekly Kt/V and creatinine clearance. Interpreting clearance targets for pregnancy in HD and PD is further hampered by the near ubiquitous lack of residual kidney function (RKF) being specified in the pregnant patients reported in the literature.
Our patient was managed using APD therapy with an extended period of connection to the cycler, allowing for additional cycling and an increase in the prescribed total volume (the target BUN being 50 mg/dL or less). The BUN values in the last 8 weeks of the pregnancy were below that 50 mg/dL target. Our patient was managed in a manner similar to that used by Smith and colleagues (21)—that is, she was hospitalized during the last weeks of gestation, thus allowing for more prolonged APD therapy with only periodic interruption to allow for ambulation or medical tests; otherwise, continuous cyclic therapy was maintained.
As mentioned, Table 2 lists other PD prescriptions. Notably, Chang and colleagues (20) described a tidal regimen designed to address drain pain and gastroesophageal reflux. The regimen left 15% of the initial infused dwell (tidal exchange volume of 85% of the initial fill), providing symptom resolution. Tidal regimens might also alleviate catheter drain dysfunction caused by the enlarging uterus. In addition, changing position during the drain phase can also reduce discomfort. Trials of draining in the recumbent or right or left side position can be used to identify the ideal position. Most descriptions of CAPD reported reduced infusion volumes and an increased number of manual exchanges. As mentioned, it is not possible to assess the delivered Kt/V of most regimens because of the infrequent reporting of maternal RKF.
Our literature review suggests that, compared with the dialysis patient who becomes pregnant while on long-standing dialysis, CKD patients who become pregnant and subsequently initiate PD (or HD) later in the pregnancy tend to experience better maternal and fetal outcomes (27,35). That observation might suggest that the degree of RKF favorably affects pregnancy outcomes (6), potentially implying that, in maternal patients with significant RKF, the increased dialysate infusion volumes (as in our case) might not be as fully indicated. Use of a target BUN as a therapeutic goal would perhaps allow for the contribution of PD therapy to be added to RKF and a delivered dialysate volume determined by the target BUN. Further research in this area is needed.
One of the common pregnancy complications in this population is polyhydramnios—presumably because of fetal urea osmotic diuresis, combined with maternal and fetal plasma volume expansion. In the HD literature, aggressive ultrafiltration was noted to resolve the polyhydramnios; however, excessive ultrafiltration may compromise placental circulation and perfusion of the fetal kidneys, leading some authors to suggest that mild-to-moderate polyhydramnios should be tolerated as an indicator of uteroplacental sufficiency (32,35). Increased PD regimens that result in lowering of fetal urea levels, combined with prudent maternal volume control, might minimize the development of severe polyhydramnios; again, however, some degree of minor or moderate polyhydramnios might perhaps be tolerated. Further investigation of this issue is also needed.
An additional complication frequently encountered is premature delivery (oftentimes precipitous), combined with low birthweight infants. Those possibilities suggest that the pregnancies be considered high-risk and that improvements in management are still needed.
Lastly, delivery of PD therapy is complicated by several technique-related concerns in the 3rd trimester. As Table 1 shows, reports describe complications of hemoperitoneum and peritonitis. Transient hemoperitoneum was felt to be secondary to catheter trauma and was managed with increased exchanges and cooled dialysate (26). Severe hemoperitoneum that does not clear might signal uterine trauma, uteroplacental detachment, or spontaneous abortion that might require surgical intervention (22,24). Peritonitis episodes were treated as they would be in the nonpregnant patient and resolved with the gestation continuing. No episodes of peritonitis during the later 3rd trimester that resulted in pregnancy termination were found.
Conclusions
Successful pregnancy outcomes have been reported in both CAPD and APD. A variety of PD prescriptions have been used in the management of the pregnant patient. In the 3rd trimester, most reports describe a reduction in dwell volume, combined with an increase in the number of exchanges. Automated PD has allowed for an even greater number of daily cycles, combined with lower dwell volumes, to deliver increased total daily dialysate exposure. In the pregnant PD patient, the effect of RKF and of achieved maternal plasma BUN in determining fetal outcome requires further clarification. Future reports should include information on RKF, prescribed weekly Kt/V, and achieved BUN to better understand the target BUN in the well-nourished pregnant patient on PD.
Disclosures
SG is an employee of Baxter Healthcare Corporation, Deerfield, IL, USA. The remaining authors have no financial conflicts of interest to declare.
REFERENCES
- 1. Nevis IF, Reitsma A, Dominic A, McDonald S, Thabane L, Akl EA, et al. Pregnancy outcomes in women with chronic kidney disease: a systematic review. Clin J Am Soc Nephrol 2011; 6:2587–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Holley JL, Reddy SS. Pregnancy in dialysis patients: a review of outcomes, complications, and management. Semin Dial 2003; 16:384–8. [DOI] [PubMed] [Google Scholar]
- 3. Hou S. Conception and pregnancy in peritoneal dialysis patients. Perit Dial Int 2001; 21(Suppl3): S290–4. [PubMed] [Google Scholar]
- 4. Okundaye I, Abrinko P, Hou S. Registry of pregnancy in dialysis patients. Am J Kidney Dis 1998; 31:766–73. [DOI] [PubMed] [Google Scholar]
- 5. Barua M, Hladunewich M, Keunen J, Pierratos A, McFarlane P, Sood M, et al. Successful pregnancies on nocturnal home hemodialysis. Clin J Am Soc Nephrol 2008; 3:392–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Redrow M, Cherem L, Elliott J, Mangalat J, Mishler RE, Bennett WM, et al. Dialysis in the management of pregnant patients with renal insufficiency. Medicine (Baltimore) 1988; 67:199–208. [DOI] [PubMed] [Google Scholar]
- 7. Jakobi P, Ohel G, Szylman P, Levit A, Lewin M, Paldi E. Continuous ambulatory peritoneal dialysis as the primary approach in the management of severe renal insufficiency in pregnancy. Obstet Gynecol 1992; 79:808–10. [PubMed] [Google Scholar]
- 8. Cattran DC, Benzie RJ. Pregnancy in a continuous ambulatory peritoneal dialysis patient. Perit Dial Int 1983; 3:13–14. [Google Scholar]
- 9. Kioko EM, Shaw KM, Clarke AD, Warren DJ. Successful pregnancy in a diabetic patients treated with continuous ambulatory peritoneal dialysis. Diabetes Care 1983; 6:298–300. [DOI] [PubMed] [Google Scholar]
- 10. Melendez R, Fraquero C, Gill P, Bakke T. Successful pregnancy with CAPD. ANNA J 1988; 15:280–1. [PubMed] [Google Scholar]
- 11. Lavoie SD, Johnson-Whittaker L, Huard PJ, Posen GA, Jindal SL, Garner P. Two successful pregnancies on CAPD. Adv Perit Dial 1988; 4:90–5. [Google Scholar]
- 12. Bennett-Jones DN, Aber GM, Baker K. Successful pregnancy in a patient treated with continuous ambulatory peritoneal dialysis. Nephrol Dial Transplant 1989; 4:583–5. [PubMed] [Google Scholar]
- 13. Dunbeck D, Klopstein K, Heroux J, Brencick K. Peritoneal dialysis patient completes successful pregnancy. ANNA J 1992; 19:269, 272. [PubMed] [Google Scholar]
- 14. Lew SQ, Watson IA. Urea and creatinine generation and removal in a pregnant patient receiving peritoneal dialysis. Adv Perit Dial 1992; 8:131–5. [PubMed] [Google Scholar]
- 15. Gadallah MF, Ahmad B, Karubian F, Campese VM. Pregnancy in patients on chronic ambulatory peritoneal dialysis. Am J Kidney Dis 1992; 20:407–10. [DOI] [PubMed] [Google Scholar]
- 16. Hou CH, Lee CN, Hung KY, Huang CH, Tsai TJ, Chen CY. An unexpected pregnancy causes poor drainage in automated peritoneal dialysis. Nephrol Dial Transplant 1996; 11:2335–7. [DOI] [PubMed] [Google Scholar]
- 17. Tison A, Lozowy C, Benjamin A, Usher R, Prichard S. Successful pregnancy complicated by peritonitis in a 35-year-old CAPD patient. Perit Dial Int 1996; 16(Suppl 1):S489–91. [PubMed] [Google Scholar]
- 18. Romão JE, Jr, Luders C, Kahhale S, Pascoal IJ, Abensur H, Sabbaga E, et al. Pregnancy in women on chronic dialysis. Nephron 1998; 78:416–22. [DOI] [PubMed] [Google Scholar]
- 19. Tuncer M, Trak B, Sapan M, Ozcan S, Süleymanlar G, Yakupoglu G, et al. Successful pregnancy complicated with peritonitis in a 25-year old Turkish CAPD patient. Perit Dial Int 2000; 20:349–50. [PubMed] [Google Scholar]
- 20. Chang H, Miller MA, Bruns FJ. Tidal peritoneal dialysis during pregnancy improves clearance and abdominal symptoms. Perit Dial Int 2002; 22:272–74. [PubMed] [Google Scholar]
- 21. Smith WT, Darbari S, Kwan M, O’Reilly-Green C, Devita MV. Pregnancy in peritoneal dialysis: a case report and review of adequacy and outcomes. Int Urol Nephrol 2005; 37:145–51. [DOI] [PubMed] [Google Scholar]
- 22. Chou CY, Ting IW, Hsieh FJ, Lee CN. Haemoperitoneum in a pregnant woman with peritoneal dialysis. Nephrol Dial Transplant 2006; 21:1454–5. [DOI] [PubMed] [Google Scholar]
- 23. Tan LK, Kanagalingam D, Tan HK, Choong HL. Obstetric outcomes in women with end-stage renal failure requiring renal dialysis. Int J Gynaecol Obstet 2006; 94:17–22. [DOI] [PubMed] [Google Scholar]
- 24. Lew SQ. Persistent hemoperitoneum in a pregnant patient receiving peritoneal dialysis. Perit Dial Int 2006; 26:108–11. [PubMed] [Google Scholar]
- 25. Asgari E, Bramham K, Shehata H, Makanjuola D. Successful pregnancy in a patient with end-stage renal failure secondary to HIV nephropathy on peritoneal dialysis. Nephrol Dial Transplant 2007; 22:3671. [DOI] [PubMed] [Google Scholar]
- 26. Altay M, Akay H, Parpucu H, Duranay M, Oguz Y. A rare case: full-term delivery in a lupus patient on CAPD. Perit Dial Int 2007; 27:711–12. [PubMed] [Google Scholar]
- 27. Gómez Vázquez JA, MartínezCalva IE, Mendíola Fernández R, Escalera León V, Cardona M, Noyola H. Pregnancy in end-stage renal disease patients and treatment with peritoneal dialysis: report of two cases. Perit Dial Int 2007; 27:353–8. [PubMed] [Google Scholar]
- 28. Jefferys A, Wyburn K, Chow J, Cleland B, Hennessy A. Peritoneal dialysis in pregnancy: a case series. Nephrology (Carlton) 2008; 13:380–3. [DOI] [PubMed] [Google Scholar]
- 29. Chou CY, Ting IW, Lin TH, Lee CN. Pregnancy in patients on chronic dialysis: a single center experience and combined analysis of reported results. Eur J Obstet Gynecol Reprod Biol 2008; 136:165–70. [DOI] [PubMed] [Google Scholar]
- 30. Inal S, Reis KA, Armağan B, Oneç K, Biri A. Successful pregnancy in an end-stage renal disease patient on peritoneal dialysis. Adv Perit Dial 2012; 28:140–1. [PubMed] [Google Scholar]
- 31. Sivasuthan G, Dahwa R, John GT, Ranganathan D. Dialysis and pregnancy in end stage kidney disease associated with lupus nephritis. Case Rep Med 2013; 2013:923581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Luders C, Castro MC, Titan SM, De Castro I, Elias RM, Abensur H, et al. Obstetric outcome in pregnant women on long-term dialysis: a case series. Am J Kidney Dis 2010; 56:77–85. [DOI] [PubMed] [Google Scholar]
- 33. Yang LY, Thia EWH, Tan LK. Obstetric outcomes in women with end-stage renal disease on chronic dialysis: a review. Obstet Med 2010; 3:48–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Okundaye I, Hou S. Management of pregnancy in women undergoing continuous ambulatory peritoneal dialysis. Adv Perit Dial 1996; 12:151–5. [PubMed] [Google Scholar]
- 35. Asam Asamiya Y, Otsubo S, Matsuda Y, Kimata N, Kikuchi K, Miwa N, et al. The importance of low blood urea nitrogen levels in pregnant patients undergoing hemodialysis to optimize birth weight and gestation age. Kidney Int 2009; 75:1217–22. [DOI] [PubMed] [Google Scholar]
- 36. Davison JM. Dialysis, transplantation, and pregnancy. Am J Kidney Dis 1991; 17:127–32. [DOI] [PubMed] [Google Scholar]
- 37. Keshaviah PR, Nolph KD, Van Stone JC. The peak concentration hypothesis: a urea kinetic approach to comparing adequacy of continuous ambulatory peritoneal dialysis (CAPD) and hemodialysis. Perit Dial Int 1989; 9:257–60. [PubMed] [Google Scholar]


