Abstract
The minimum information about a biofilm experiment (MIABiE) initiative has arisen from the need to find an adequate and scientifically sound way to control the quality of the documentation accompanying the public deposition of biofilm-related data, particularly those obtained using high-throughput devices and techniques. Thereby, the MIABiE consortium has initiated the identification and organization of a set of modules containing the minimum information that needs to be reported to guarantee the interpretability and independent verification of experimental results and their integration with knowledge coming from other fields. MIABiE does not intend to propose specific standards on how biofilms experiments should be performed, because it is acknowledged that specific research questions require specific conditions which may deviate from any standardization. Instead, MIABiE presents guidelines about the data to be recorded and published in order for the procedure and results to be easily and unequivocally interpreted and reproduced. Overall, MIABiE opens up the discussion about a number of particular areas of interest and attempts to achieve a broad consensus about which biofilm data and metadata should be reported in scientific journals in a systematic, rigorous and understandable manner.
Keywords: biofilm, microbial communities, data standardization, data interchange, machine-readable formats
Introduction
One of the major goals of microbial systems biology is to obtain comprehensive knowledge not only of individual cells, but also of entire microbial communities (Zengler & Palsson, 2012). In nature, these microbial communities can mostly be found in the form of biofilms, which are complex, three-dimensional aggregates of microbial cells enclosed in a self-produced polymeric matrix and living at interfaces (Hall-Stoodley et al., 2004).
While for planktonic individual cells the emergence of high-throughput methods and subsequent ‘omics’ disciplines has been fast, the evolution of technologies for scrutinizing sessile microbial communities has been slower. Nonetheless, similarly to what has happened in other research fields, the field of biofilms is starting to rely heavily on high-throughput techniques to boost the understanding of key phenomena related to microbial communities (Peeters et al., 2008; Azevedo, 2012). For instance, the development and standardization of high-throughput biofilm methods based upon microtitre plates assays (Ceri et al., 1999; Stepanović et al., 2000) has paved the way for the formation of similar biofilms in a large number of experiments. Some of these methods have even been validated by a standard setting organization (ASTM, 2012). Metagenomics, metaproteomics and other ‘omics’ technologies may then be applied to these structures in order to unveil the genome, transcriptome and proteome of the microbial community (Schmeisser et al., 2003; Ram et al., 2005; Pepperkok & Ellenberg, 2006; Gjersing et al., 2007; Yergeau et al., 2010). Other parameters that are crucial for a full understanding of a spatially structured ecosystem, such as the location of microorganisms and matrix components, may also be assessed in a high-throughput manner in the near future using specific microscopy methods (Pepperkok & Ellenberg, 2006; Azevedo, 2012).
The study of biofilms is hence becoming a data-intensive research field that must adapt to this new reality by deploying and enforcing novel methodologies in terms of data management and analysis. As in other areas, issues such as lack of data reproducibility, scarcity of standardized protocols, poor data quality and incomplete data sets significantly hamper the quality of published results (Huang & Gottardo, 2013). In fact, with the exception of data related to more established techniques developed for microbial communities as a whole (such as metagenomics), a large volume of biofilm data from published experiments lay in the private files of researchers. Public release of data in novel online platforms dedicated to biofilms, such as BiofOmics (Lourenço et al., 2012), is welcome but not sufficient. The lack of specific details about the experimental technique(s) employed, and the corresponding experimental conditions will still limit data interpretability by other researchers besides the authors (Sousa et al., 2012).
Data standardization and structuring in biofilm research is therefore crucial to allow researchers to understand, replicate and assess studies at an interlaboratory scale. This requires the definition of the minimum information that must be documented to ensure that an experiment on microbial biofilms is described unambiguously and comprehensively. Following this lead, this study presents a new standard initiative called the minimum information about a biofilm experiment (MIABiE), which is preparing a set of guidelines for the documentation of biofilm experiments and data, namely the minimum information checklists. Although the MIABiE consortium already consists of experts with a wide variety of research interests in biofilms, this initiative is expected to serve as a starting point for a broader, interdisciplinary community discussion. This concept provides a common platform which makes it easier to compare results obtained from different environments and settings.
Benefits of MIABiE
MIABiE allows researchers to summarize the purpose, methodology and principal conclusions of a biofilm study, with the following primary objectives:
assist authors in creating standard-compliant and structured machine-readable digital summaries of the data sets and other outputs related to particular biofilms studies;
assist authors in the process of writing comprehensive and unambiguous reports of their research, by bringing to their attention essential experimental details that should be specified in their papers;
facilitate the reproduction of experimental procedures, by formulating rules and guidelines to be met by methodology description;
enable incremental experimental designs, by using findings from previous experiments, either to complement or validate new results;
assist reviewers of journal articles in assessing the relevance of reported results;
underpin the development of specialized bioinformatics tools, both to produce the standard-compliant and structured machine-readable digital summaries, and to comply with more ambitious research goals and thus achieve a coordinated understanding of microbial communities;
to statistically evaluate those experimental parameters individually and interactively that are most important in influencing biofilm growth, development and response, and to identify possible ‘gaps’ which can be interrogated in silico or experimentally, allowing continual refinement of the model.
It is important to highlight that MIABiE does not intend to establish specific rules or provide standards on how biofilm experiments should be performed. Instead, MIABiE provides guidelines about the data to be recorded, considering the purpose of the study and the devices and techniques involved, in order for the procedure and the results to be easily reproducible and interpretable.
The MIABiE modules
The major challenge encountered when providing guidelines to document biofilm experiments and report their results is the complexity and variability of biofilm studies. For instance, studies may vary in the number and kind of conditions tested, the wide range of microorganisms that can be studied (both at the species and strain level), the inter- and intraspecies interactions these organisms may establish in multispecies biofilms, the initial physical and chemical conditions, the biofilm experimental model system (s) used, the type and number of analytical methods involved, specific data preprocessing, the number of technical and biological replicates performed, and the statistical method(s) used to analyse the data.
To better capture such complexity and variability in biofilm studies, the concept of module, already used for reporting minimum information guidelines in other fields (Taylor et al., 2007), was used. Each module addresses the standardized collection, integration, storage and dissemination of data on specific aspects of a biofilm study (e.g. antimicrobial susceptibility testing), biofilm-related device (e.g. the microtitre plate) or biofilm-related technique [e.g. the crystal violet (CV) assay]. Following an extensive discussion between members of the biofilm community, a set of 15 modules is proposed here. Each module represents a particular area of interest that critically influences the results of a biofilm experiment (Table 1); the set of modules can be easily expanded if necessary. The introduction and delineation of each module was based on the following criteria: (1) all biofilm-related experiments should comprehensively fit into one or more of the modules; (2) as most biofilm studies are greatly influenced by the type of device and operation mode used to develop them (Buckingham-Meyer et al., 2007), biofilm model systems were grouped according to their characteristics and mode of operation; (3) the many unique parameters associated with in vivo biofilm formation or biofilm formation on biotic or abiotic surfaces warrant the creation of distinct modules; and (4) techniques that are common to other research areas should be in modules of their own, delegating their description to the minimum information guidelines established by the corresponding initiative.
Table 1. Description of the modules proposed by MIABiE.
| Module | Short description |
|---|---|
| 1. Sample generation and study design | The description of the goal(s) and design of the experiment. This includes the identification of microorganisms (to the strain level, preferably with an identifier linking to an international Biological Resource Center) and environmental conditions tested, as well as the technologies used to form and analyse the biofilm |
| 2. Naturally occurring biofilms | Refers to biofilms formed previously in real settings such as in the environment (e.g. streams, rocks, soil, buildings), industry (e.g. process equipment and piping) and living beings (e.g. animals, humans, plants). It describes as extensively as possible the conditions under which the biofilm was formed, before sampling was carried out, or in situ examination |
| 3. Single- and multiwell reactors | Specifies the type(s) of well reactors used to develop biofilms in a standardized manner, together with process variables. Examples of such reactors are the microtitre plates (or 6/24/96-well tissue culture plates), the MatTek plates and the MBEC™device |
| 4. Continuously stirred flow reactors | Covers the use of stirred reactors with a continuous flow of media for biofilm formation and growth, and the documentation of process variables in in vitro biofilm development (temperature, pressure, residence time, flow rate, nutrient concentration and type, etc.). Examples of stirred flow reactors are the chemostat, annular reactors, CDC biofilm reactor, rotating disc reactor. |
| 5. Continuous plug flow reactors | Describes the various types of reactors that work in the continuous mode of operation (except for multiwells and stirred reactors) and associated process variables. Reactors included in this module are the modified Robbins device (MRD), flow cells, drip flow reactors, tubing reactors and microfluidic systems |
| 6. In vivo biofilm models | Provides protocols to study biofilm formation and development in in vivo animal models. Additionally, experimenters should inform about the device (catheter, beads, etc.) (if any) that is used to promote the growth of biofilms, providing information on the constituent material/biomaterial, eventual pre-and postprocessing of the material and the conditions used for biofilm growth, anatomical compartment of infection, infection procedure, recalcitrance to antimicrobial therapy, length of time of infection, and animal species and strain |
| 7. In vitro or ex vivo biofilm formation on biotic surfaces | Describes the protocols for biofilm formation in animal and vegetable tissue models. Experimenters should indicate the tissues (e.g. wound biofilm model, RHE model), the medium and the conditions used for developing biofilm on these surfaces |
| 8. Determination of antimicrobial susceptibility | Describes protocols to evaluate the activity of antimicrobial products (including antibiotics and disinfectants) against biofilms. This includes the parameters related with the antimicrobial agent(s) (name, company, dose, etc.), the preprocessing of antimicrobials (preparation of stock solutions, storage, etc.), the neutralization of the active component and protocols for susceptibility testing including pharmacokinetic [e.g. antibiotic concentrations (constant or fluctuating), exposure time] and the pharmacodynamic parameters (e.g. viability, resistance development, biomass, biofilm structure) and endpoints (e.g. minimum inhibitory, bactericidal or eradication concentrations). Information about removal and/or neutralization of the antimicrobial agents prior to downstream experiments should also be provided. Likewise, authors should describe the automation conditions of high-throughput screening campaigns, for example liquid-handling workstations and robotic protocols |
| 9. Culture-based biofilm assessment | Covers all the procedures related to CFU quantification and colony morphology classification in biofilms. These include details on sonication or scraping to remove the adhered cells, homogenization of the suspended cells (e.g. by vortexing), the type of plating medium used, incubation conditions, etc. |
| 10. Non-culture-based biofilm assessment | Characterizes other analytical techniques that are applied to biofilm samples prior to the final analysis, such as 4′,6-diamidino-2-phenylindole (DAPI), crystal violet (CV), Live/Dead, fluorescence in situ hybridization (FISH) and Dubois. Details on the technique include the final purpose of the technique, any reaction or interactions associated with the technique and any pretreatment of the sample |
| 11. Microscopy and flow cytometry methods | This module is specialized in flow cytometry and the different types of microscopy methods (such as CSLM) used in support of the analytical techniques, such as DAPI, Live/Dead, FISH, colony morphology. |
| 12. Spectrophotometric detection methods | Optical detection is used to support analytical techniques, such as CV, 2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide (XTT), Alamar Blue (resazurin), ATP detection and the Lowry protein assay, whose final values are dependent on absorbance, bioluminescence or fluorescence data. Data on the type of equipment used and wavelength(s) employed among other characteristics should be indicated |
| 13. ‘Omics’ and molecular methods | Encompasses the documentation of methods that evaluate and quantify the presence of molecules in biofilms (e.g. PCR) and the application of ‘omics’ methods such as proteomics, transcriptomics and metabolomics and their meta-version. MIABiE guidelines are compliant with guidelines for microarray-based transcriptomics (MIAME), quantitative real-time PCR (MIQE), proteomics (MIAPE) and metabolomics (CIMR), and will embrace any forthcoming guidelines emerging from other relevant ‘omics’ communities |
| 14. Statistical assessment | The statistical evaluation of the data should be fully disclosed and should include the type of test, whether or not data were transformed, significance levels used, etc. The ability to reproduce the experiment in other laboratory and obtain similar results is critical |
| 15. Bioinformatics resources and tools | Underpins data standardization and data interchange infrastructures, and the development of bioinformatics software specialized in biofilms data management and data analytics. This includes, among others: standardized terminologies specialized in biofilms, markup languages delivering computer-readable formats for representing biofilm models (e.g. similar to the systems biology markup language), databases to deposit biofilm data publicly, software for biofilm image analysis, and tools for drug virtual screening, and biofilm modelling and simulation |
As shown in Fig. 1, the modular approach is quite flexible, that is, as new devices, techniques or applications become increasingly popular, a new module can be created and integrated in this list without affecting the guidelines already in use in other modules. In fact, novel research questions often require modifying the published protocols and methods to achieve relevant answers. It is important that the modifications to the published methods be clearly documented to understand how the method itself evolves with the research data and knowledge gained.
Fig. 1.
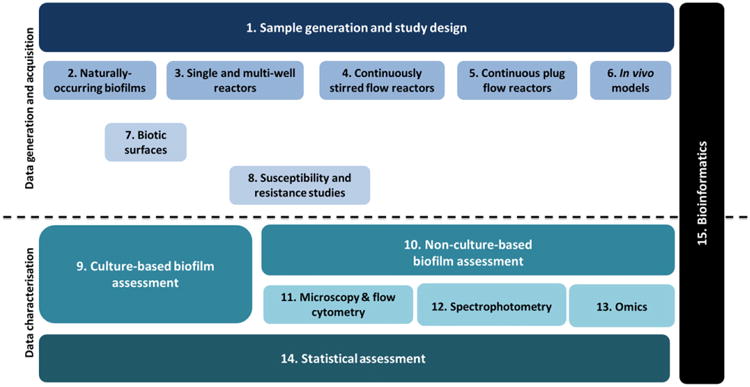
Schematic representation of the modules proposed by MIABiE.
A second major advantage is the delegation of responsibility, that is, the MIABiE consortium is focused on biofilm-specific data issues, relying on the guidelines of other consortia to link to other research areas.
Integration of MIABiE with other minimum information guidelines
As already mentioned, biofilm research is built upon biofilm-specific experiments and experiments from other scientific areas. To address this interrelation, MIABiE has engaged MIBBI (http://www.mibbi.org/), an initiative that provides a common portal with minimum information checklists, standards and guidelines from all areas of biological and biomedical sciences (Taylor et al., 2008).
At the core of the MIABiE strategy is the delegation of the documentation guidelines of non-biofilm-specific data. For instance, data coming from transcriptome, proteome and other ‘omic’ technologies applied to biofilm populations should be documented as suggested by MIAME (Brazma et al., 2001), MIAPE (Taylor et al., 2007) and similar guidelines (Fig. 2a). This would also be the case for specific techniques, such as fluorescence in situ hybridization or flow cytometry, for which minimum information guidelines have also been reported (Deutsch et al., 2008; Lee et al., 2008).
Fig. 2.
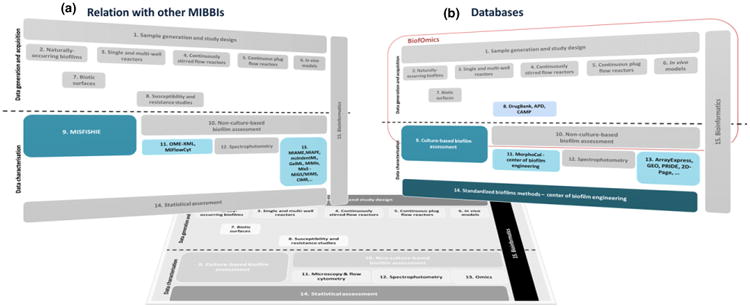
A schematic diagram, showing the relation between the different modules, MIBBIs and online resources; (a) the different sets of minimum information guidelines available; and (b) the databases where parameters and data can be accommodated. This scheme should not be interpreted as a fixed scheme, but rather as a starting point that will be adjusted as biofilm studies, databases and minimum information guidelines become integrated and evolve in time.
Availability of biofilm-centered databases and portals
For many years, the main source of biofilm information has been the scientific literature. This is hardly an optimal solution as manual curation of scientific literature is resource- and time-consuming (Lok, 2010; Lu & Hirschman, 2012). Far more important, biofilm-related publications do not usually have experimental or analytical raw data attached, which hampers attempts to reproduce certain experiments.
A strong indication that this situation is about to change is the emergence of databases and other public websites where data on biofilm experiments are made readily available (Inst. Pasteur, 2013; NASA, 2013; Proteome commons, 2013). Since 2012, the BiofOmics Web database offers a public site for experimental data and results from biofilm experiments (Lourenço et al., 2012). BiofOmics developers have committed to a trade-off between MIABiE guidelines and the donation of data, and data submitters are encouraged to comply with MIABiE guidelines, by creating a standardized Excel data workbook.
Although the BiofOmics platform can be extended to accommodate more data, the idea is not to cover all data, especially if they are not specific to biofilm experiments (Fig. 2b). Most of the results generated by Module 12, for instance, will be stored in ‘omics’ databases, such as the Gene Expression Omnibus (GEO) database (Edgar et al., 2002) and the PRoteomics IDEntifications (PRIDE) database (Wang et al., 2012), which keep data on the transcriptome and proteome of organisms, respectively. The use of these resources, rather than creating specialized ones, has two main advantages: it alleviates biofilm-centered databases of the burden of duplicating the storage of such large data volumes, while relying on the documentation guidelines established by ‘omics’ experts.
Use and creation of vocabularies and terminology standardization
MIABiE enforces the use of controlled vocabularies that facilitate the transmission of information, in terms of comprehensibility and interpretation. Ambiguity as the field develops its own terminology can occur not only within the field but also between different fields. For example, the term ‘extracellular matrix’ (ECM) has been used in plant and animal biology to refer to extracellular materials which may have a structural role (e.g. connective), but has been gaining use by biofilm researchers where it has been replacing what was previously known as ‘extracellular polysaccharides’ or more generically, as the complexities of the biofilm matrix have been revealed, ‘extracellular polymeric substances’ (EPS) (Flemming & Wingender, 2010). The use of ‘matrix’ to discuss the biofilm EPS has caused confusion, particularly when the biofilm might be investigated in clinical specimens where it is not clear whether ECM refers to the host or microbial derived fractions. Another term which can result in ambiguity is ‘substrate’. In some cases, this term is used, more in the engineering community, to refer to the surface that biofilms are grown on, while the more common usage is as a nutrient.
Regarding the data documenting a biofilm experiment, MIABiE has already looked into existing vocabularies. For instance, microorganisms and their sequences are catalogued in NCBI Taxonomy and GenBank (NCBI Resource Coordinators, 2013), strain numbers in international culture collections can be found in Straininfo.net (Dawyndt et al., 2005), and metabolism is described in pathway databases such as KEGG (Kanehisa et al., 2012) and BioCyc (Caspi et al., 2012). By cross-linking to these databases, researchers obviate the need to include this sort of data on their experiment files.
Portals such as the Open Biological and Biomedical Ontologies (OBO) foundry (http://www.obofoundry.org/) congregate a number of biological vocabularies potentially useful in biofilm descriptions (Ceusters & Smith, 2010). For instance, the PATO ontology (Beck et al., 2009), which describes phenotypic qualities generically, could be adapted or extended to include qualities specific of microbial communities. Likewise, systems biology approaches could benefit greatly from the efforts of Gene Ontology Consortium to annotate various biofilm aspects (Gene Ontology Consortium, 2013). Moreover, dictionaries on nonspecific information could be derived from databases and associated terminologies. For example, antimicrobial products, such as drugs and natural peptides, are being catalogued in Drug-Bank (Knox et al., 2011) and the Collection of Anti-Microbial Peptides (CAMP) (Thomas et al., 2010), respectively.
Vocabularies on biofilm specifics such as devices, techniques, materials and media are not available. Similarly, the qualitative description of biofilm features and behaviour, namely the morphology of the colonies when facing particular stresses, was subjective. Given their critical role in the description of the experiment, MIABiE initiated the elaboration of vocabularies covering these aspects, once again reaching out to the community for a consensus.
Final remarks
Similarly to what happened in other fields of Biology, the study of biofilms has entered the high-throughput era. Because data and knowledge accumulate rapidly, it is crucial to empower researchers with instant access to this information. New studies could be justified, integrated or contextualized by previous knowledge by comparing results from multiple, expected to be similar, experiments. Thereby, it seems only logical that to pursue high-quality research, biofilm experts engage into systematic data management and interchange.
Consequently, MIABiE proposes a standards initiative that intends to simplify the exchanging and comparison of biofilm data across different laboratories, by ensuring that authors document their experiments comprehensively and unequivocally in scientific publications. MIABiE also aims at complementing existing minimum information guidelines for other high-throughput fields, to assist in the completeness of data provided for a study. For instance, if an experiment on proteomics is carried out on biofilms, the data on proteomics might become less meaningful if the characterization of the microbiological sample is not complete. By providing adequate background to the microbiological sample, MIABiE ensures that results from such experiments can be better interpreted in the future.
Finally, MIABiE attempts to decrease the variability of results obtained from biofilm studies. Over time, this variability has become accepted as some sort of inevitability. While it is unlikely that this initiative will reduce variability to the level observed for individual cells, it will allow source discrimination into experimental variations, and intrinsic and extrinsic noise due to the microorganisms (Elowitz et al., 2002). This ability is important to assess the disparity of results obtained by different laboratories applying biofilm-related technologies under similar conditions, as well as to perform head-to-head comparisons between some of the most commonly used technologies, for example CV and XTT.
The success of MIABiE initiative depends on establishing a consensus within the biofilm community. At the moment, MIABiE modules and reporting requirements are being identified by the MIABiE consortium, but a broader community discussion should be pursued continuously. This discussion will be conducted either through e-mail discussion lists (details on the website, http://www.miabie.org) or on speciality conferences on biofilms. The first of these conference discussions was held at the Eurobiofilms 2013 meeting in Ghent, Belgium (9-12 September 2013).
This timely and well-written minireview describes a way forward towards establishing guidelines or modules for reporting on experiments with microbial biofilms. The authors review the rationale for and benefits of standardization and is a significant effort of great importance for the biofilm community.
Acknowledgments
The authors would like to thank Thomas Bjarnsholt and Adyary Fallarero for a critical revision of the manuscript. This work was supported by IBB-CEB; Fundação para a Ciência e Tecnologia (FCT); the European Community fund FEDER, through Program COMPETE, in the ambit of the FCT Project PTDC/SAU-SAP/113196/2009/FCOMP-01-0124-FEDER-0 16012; the European Union Seventh Framework Programme [FP7/REGPOT-2012-2013.1] under Grant Agreement No. 316265, BIOCAPS; the Agrupamento INBIOMED from DXPCTSUG-FEDER unha maneira de facer Europa (2012/273). T.C. would like to thank FWO Vlaanderen and the Interuniversity Attraction Poles Programme initiated by the Belgian Science Policy Office for funding.
References
- ASTM. ASTM E2799 – 12: Standard Test Method for Testing Disinfectant Efficacy against Pseudomonas aeruginosa Biofilm using the MBEC Assay. 2012 doi: 10.1520/E2799-12. [DOI] [Google Scholar]
- Azevedo NF. Biofilm formation on a microtiter-plate using artificial urine to mimic cathether-associated urinary tract infections. Biofilms 5 Int Conf B Abstr 2012 [Google Scholar]
- Beck T, Morgan H, Blake A, Wells S, Hancock JM, Mallon AM. Practical application of ontologies to annotate and analyse large scale raw mouse phenotype data. BMC Bioinformatics. 2009;10(suppl 5):S2. doi: 10.1186/1471-2105-10-S5-S2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brazma A, Hingamp P, Quackenbush J, et al. Minimum information about a microarray experiment (MIAME)-toward standards for microarray data. Nat Genet. 2001;29:365–371. doi: 10.1038/ng1201-365. [DOI] [PubMed] [Google Scholar]
- Buckingham-Meyer K, Goeres DM, Hamilton MA. Comparative evaluation of biofilm disinfectant efficacy tests. J Microbiol Methods. 2007;70:236–244. doi: 10.1016/j.mimet.2007.04.010. [DOI] [PubMed] [Google Scholar]
- Caspi R, Altman T, Dreher K, et al. The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of pathway/genome databases. Nucleic Acids Res. 2012;40:D742–D753. doi: 10.1093/nar/gkr1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceri H, Olson ME, Stremick C, Read RR, Morck D, Buret A. The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J Clin Microbiol. 1999;37:1771–1776. doi: 10.1128/jcm.37.6.1771-1776.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceusters W, Smith B. A unified framework for biomedical terminologies and ontologies. Stud Health Technol Inform. 2010;160:1050–1054. [PMC free article] [PubMed] [Google Scholar]
- Dawyndt P, Vancanneyt M, De Meyer H, Swings J. Knowledge accumulation and resolution of data inconsistencies during the integration of microbial information sources. IEEE Trans Knowl Data Eng. 2005;17:1111–1126. [Google Scholar]
- Deutsch EW, Ball CA, Berman JJ, et al. Minimum information specification for in situ hybridization and immunohistochemistry experiments (MISFISHIE) Nat Biotechnol. 2008;26:305–312. doi: 10.1038/nbt1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002;30:207–210. doi: 10.1093/nar/30.1.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elowitz MB, Levine AJ, Siggia ED, Swain PS. Stochastic gene expression in a single cell. Science. 2002;297:1183–1186. doi: 10.1126/science.1070919. [DOI] [PubMed] [Google Scholar]
- Flemming HC, Wingender J. The biofilm matrix. Nat Rev Microbiol. 2010;8:623–633. doi: 10.1038/nrmicro2415. [DOI] [PubMed] [Google Scholar]
- Gene Ontology Consortium. Gene Ontology annotations and resources. Nucleic Acids Res. 2013;41:D530–D535. doi: 10.1093/nar/gks1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gjersing EL, Herberg JL, Horn J, Schaldach CM, Maxwell RS. NMR metabolomics of planktonic and biofilm modes of growth in Pseudomonas aeruginosa. Anal Chem. 2007;79:8037–8045. doi: 10.1021/ac070800t. [DOI] [PubMed] [Google Scholar]
- Hall-Stoodley L, Costerton JW, Stoodley P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2004;2:95–108. doi: 10.1038/nrmicro821. [DOI] [PubMed] [Google Scholar]
- Huang Y, Gottardo R. Comparability and reproducibility of biomedical data. Brief Bioinform. 2013;14:391–401. doi: 10.1093/bib/bbs078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inst. Pasteur. Transcript profiling of Candida albicans biofilms. 2013 http://www.pasteur.fr/recherche/unites/Galar_Fungail/Biofilm/biofilm2.html.
- Kanehisa M, Goto S, Sato Y, Furumichi M, Tanabe M. KEGG for integration and interpretation of large-scale molecular data sets. Nucleic Acids Res. 2012;40:D109–D114. doi: 10.1093/nar/gkr988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knox C, Law V, Jewison T, et al. DrugBank 3.0: a comprehensive resource for “omics” research on drugs. Nucleic Acids Res. 2011;39:D1035–D1041. doi: 10.1093/nar/gkq1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JA, Spidlen J, Boyce K, et al. MIFlowCyt: the minimum information about a flow cytometry experiment. Cytometry A. 2008;73:926–930. doi: 10.1002/cyto.a.20623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lok C. Literature mining: speed reading. Nature. 2010;463:416–418. doi: 10.1038/463416a. [DOI] [PubMed] [Google Scholar]
- Lourenço A, Ferreira A, Veiga N, Machado I, Pereira MO, Azevedo NF. BiofOmics: a Web platform for the systematic and standardized collection of high-throughput biofilm data. PLoS One. 2012;7:e39960. doi: 10.1371/journal.pone.0039960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Z, Hirschman L. Biocuration workflows and text mining: overview of the BioCreative 2012 Workshop Track II. Database (Oxford) 2012;2012:bas043. doi: 10.1093/database/bas043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NASA (2013) http://www.nasa.gov/mission_pages/station/research/experiments/Micro2A.html
- NCBI Resource Coordinators. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2013;41:D8–D20. doi: 10.1093/nar/gks1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peeters E, Nelis HJ, Coenye T. Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. J Microbiol Methods. 2008;72:157–165. doi: 10.1016/j.mimet.2007.11.010. [DOI] [PubMed] [Google Scholar]
- Pepperkok R, Ellenberg J. High-throughput fluorescence microscopy for systems biology. Nat Rev Mol Cell Biol. 2006;7:690–696. doi: 10.1038/nrm1979. [DOI] [PubMed] [Google Scholar]
- Proteome commons. Proteome commons. 2013 https://proteome-commons.org/group-data.jsp?i=345.
- Ram RJ, Verberkmoes NC, Thelen MP, Tyson GW, Baker BJ, Blake RC, Shah M, Hettich RL, Banfield JF. Community proteomics of a natural microbial biofilm. Science. 2005;308:1915–1920. [PubMed] [Google Scholar]
- Schmeisser C, Stöckigt C, Raasch C, et al. Metagenome survey of biofilms in drinking-water networks. Appl Environ Microbiol. 2003;69:7298–7309. doi: 10.1128/AEM.69.12.7298-7309.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa AM, Ferreira A, Azevedo NF, Pereira MO, Lourenço A. Computational approaches to standard-compliant biofilm data for reliable analysis and integration. J Integr Bioinform. 2012;9:203. doi: 10.2390/biecoll-jib-2012-203. [DOI] [PubMed] [Google Scholar]
- Stepanović S, Vuković D, Dakić I, Savić B, Švabić-Vlahović M. A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J Microbiol Methods. 2000;40:175–179. doi: 10.1016/s0167-7012(00)00122-6. [DOI] [PubMed] [Google Scholar]
- Taylor CF, Paton NW, Lilley KS, et al. The minimum information about a proteomics experiment (MIAPE) Nat Biotechnol. 2007;25:887–893. doi: 10.1038/nbt1329. [DOI] [PubMed] [Google Scholar]
- Taylor CF, Field D, Sansone SA, et al. Promoting coherent minimum reporting guidelines for biological and biomedical investigations: the MIBBI project. Nat Biotechnol. 2008;26:889–896. doi: 10.1038/nbt.1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas S, Karnik S, Barai RS, Jayaraman VK, Idicula-Thomas S. CAMP: a useful resource for research on antimicrobial peptides. Nucleic Acids Res. 2010;38:D774–D780. doi: 10.1093/nar/gkp1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R, Fabregat A, Ríos D, et al. PRIDE Inspector: a tool to visualize and validate MS proteomics data. Nat Biotechnol. 2012;30:135–137. doi: 10.1038/nbt.2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yergeau E, Lawrence JR, Waiser MJ, Korber DR, Greer CW. Metatranscriptomic analysis of the response of river biofilms to pharmaceutical products, using anonymous DNA microarrays. Appl Environ Microbiol. 2010;76:5432–5439. doi: 10.1128/AEM.00873-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zengler K, Palsson BO. A road map for the development of community systems (CoSy) biology. Nat Rev Microbiol. 2012;10:366–372. doi: 10.1038/nrmicro2763. [DOI] [PubMed] [Google Scholar]


