Abstract
Background
Vitamin D deficiency and asthma are common at higher latitudes. Although vitamin D has important immunologic effects, its relation with asthma is unknown.
Objective
We hypothesized that a higher maternal intake of vitamin D during pregnancy is associated with a lower risk of recurrent wheeze in children at 3 y of age.
Design
The participants were 1194 mother-child pairs in Project Viva—a prospective prebirth cohort study in Massachusetts. We assessed the maternal intake of vitamin D during pregnancy from a validated food-frequency questionnaire. The primary outcome was recurrent wheeze, ie, a positive asthma predictive index (≥2 wheezing attacks among children with a personal diagnosis of eczema or a parental history of asthma).
Results
The mean (±SD) total vitamin D intake during pregnancy was 548 ± 167 IU/d. By age 3 y, 186 children (16%) had recurrent wheeze. Compared with mothers in the lowest quartile of daily intake (median: 356 IU), those in the highest quartile (724 IU) had a lower risk of having a child with recurrent wheeze [odds ratio (OR): 0.39; 95% CI: 0.25, 0.62; P for trend <0.001]. A 100-IU increase in vitamin D intake was associated with lower risk (OR: 0.81; 95% CI: 0.74, 0.89), regardless of whether vitamin D was from the diet (OR: 0.81; 95% CI: 0.69, 0.96) or supplements (OR: 0.82; 95% CI: 0.73, 0.92). Adjustment for 12 potential confounders, including maternal intake of other dietary factors, did not change the results.
Conclusion
In the northeastern United States, a higher maternal intake of vitamin D during pregnancy may decrease the risk of recurrent wheeze in early childhood.
Keywords: Vitamin D, pregnancy, dietary intake, childhood wheeze, asthma
INTRODUCTION
Vitamin D3 (cholecalciferol) is a vital nutrient available from food sources (eg, fortified milk) and nutritional supplements (1). The skin can also make vitamin D when exposed to sunlight. Recent studies suggest that vitamin D may have value in preventing or treating cancer and autoimmune diseases (2, 3). It has also become clear, especially in the northeastern United States (4, 5), that a large proportion of Americans have an inadequate vitamin D intake, as reflected by serum 25-hydroxyvitamin D concentrations (6).
Another public health problem in the northeastern United States is asthma. For example, the prevalence rates of asthma in New England are the highest in the nation (7). The national prevalence of asthma has increased from ≈3% in the 1970s to ≈8% in recent years (8). The onset of asthma occurs before 6 y of age in 80–90% of cases; 70% of cases occur before 3 y of age (9, 10). Asthma has multifactorial origins, but major risk factors have proven elusive. Some investigators have focused on dietary origins, but studies to date are mostly cross-sectional and have yielded inconsistent results (11). Although vitamin D is known to have important effects on immunologic function (12), the role of vitamin D on risk of asthma is unknown. The role of maternal diet on risk of asthma in offspring is a particularly intriguing topic given the growing evidence on the developmental origins of health and disease (13, 14) and the early age of asthma onset (15, 16).
We hypothesized that higher vitamin D intakes are protective against asthma in populations with inadequate vitamin D intakes. We formulated this hypothesis after noting the similarity between risk factors for vitamin D insufficiency and for asthma. In addition to the regional similarities noted above, vitamin D insufficiency is more common among African Americans (17) and among the obese (18). Temporal trends in the adoption of sun-screen use (19) and in reductions in milk intake (20) also support the hypothesized link between vitamin D and asthma. The purpose of the present study was to determine, among mothers living in the northeastern United States, whether a higher maternal intake of vitamin D intake during pregnancy is associated with a lower risk of asthma in children at 3 y of age.
SUBJECTS AND METHODS
Study design and subjects
Project Viva is a prospective cohort study examining prenatal factors in relation to outcomes of pregnancy and child health. Participants were recruited at 8 obstetric offices of Harvard Vanguard Medical Associates, a large multispecialty urban-suburban group practice in eastern Massachusetts. At the first study visit, directly after the woman’s initial clinical prenatal visit, we obtained informed consent, administered a brief interview, and provided a take-home self-administered questionnaire. At the second study visit, at 26 to 28 wk of gestation, we again administered a brief interview and provided a questionnaire. Project Viva participants gave birth at Brigham and Women’s Hospital or Beth Israel Deaconess Medical Center in Boston. Within 3 d after delivery, we interviewed the mothers. We reassessed mothers and children at 6 mo, 1 y, and annually thereafter. At the time of this analysis, the visits at age 3 y were almost complete.
Exclusion criteria included multiple gestation (eg. twins), the inability to answer questions in English, plans to move out of the area before delivery, and a gestational age >22 completed weeks at the initial prenatal clinical appointment. Additional details of recruitment and follow-up were presented elsewhere (21). Of the 2128 infants delivered in Project Viva, we excluded 228 because of missing diet assessment data from the first and second trimesters and an additional 37 with a gestational age of <34 completed weeks. Mothers of 368 of the remaining 1863 participants did not provide informed consent for child follow-up through age 3 y. Of the remaining 1495 infants, 76 were lost to follow-up and 225 have not yet exited the 3-y time window. The final sample for analysis consisted of 1194 participants. This sample appears representative of the overall study population. The human subjects committees of Harvard Pilgrim Health Care, Brigham and Women’s Hospital, and Beth Israel Deaconess Medical Center approved the study protocols.
Measurements
We obtained data directly from the participants and from medical records, as detailed previously (21). Briefly, maternal diet assessments at both the first and second visits were based on a validated 166-item semiquantitative food-frequency questionnaire (FFQ) (22). We slightly modified the FFQ for use in pregnancy from the FFQ used in the Nurses’ Health Study and other cohort studies (23). Modifications for use in pregnancy included changing the time referent, beverage section, and vitamin and supplement assessment. The FFQ used at the first visit reflected intakes in the first trimester; the time referent was “during this pregnancy.” In addition, we collected information on beverage intake during 2 specified time periods: before and after the participant learned that she was pregnant. To assess vitamin and supplement intakes during the first trimester, we administered a separate interview that assessed dose, duration, and brand or type of multivitamin, prescribed prenatal vitamin, and supplements. The FFQ used at the second visit (26–28 wk of gestation) reflected intakes during the second trimester; the time referent was “during the past 3 mo.” The second trimester instrument was the same as that used in the first trimester except that we assessed the use of vitamins and supplements as part of the self-completed FFQ and we collected beverage information during the one time period.
To calculate nutrient intakes, we used the Harvard nutrient composition database, which contains food-composition values from the US Department of Agriculture, and other sources (24). We calculated vitamin D intake for each FFQ, and the mean of these 2 values was the assigned exposure for each woman during pregnancy (25). We used a similar approach for retinol and calcium intakes. For the analyses based on foods rather than nutrients, we focused on milk (servings/d)—the major contributor to vitamin D intake in the diet. We also examined foods with possible effects on asthma risk (11, 26): fruit and vegetables (servings/d) and fish (servings/wk).
At the first visit in early pregnancy, in addition to diet assessment, we obtained information on maternal age, race-ethnicity, household income, education, marital status, last menstrual period (LMP), height, prepregnancy weight, gravidity, and history of atopic disorders. At the second visit in midpregnancy, we updated many of these variables. We calculated maternal prepregnancy body mass index as weight (in kg) divided by the square of height (in m). We calculated gestational age using the mother’s reported LMP, or from second trimester ultrasound if they differed by >10 d. From the hospital vital statistics record, we abstracted infant birth weight. We determined a birth weight–for–gestational age z value (fetal growth) by using US reference data (27). At the 6-mo and 1-y visits, we asked about infant feeding, vitamin supplements, and household exposures. At the 2-y visit, we also used a validated FFQ to assess the children’s vitamin D intakes (28).
Our primary outcome was based on the Asthma Predictive Index (29). We assessed wheeze status initially by asking 2 questions at each of the 3 annual visits: 1) Since your child was born (was 12 mo old, was 2 y old) has he or she ever had wheezing (or whistling in the chest)?, and 2) Since your child was born (was 12 mo old, was 2 y old), how many attacks or episodes of wheezing has he or she had? We defined recurrent wheeze as ≥2 wheezing attacks (summed from the 1-, 2-, and 3-y annual questionnaires) in children with a personal diagnosis of eczema or parental history of asthma.
Secondary wheeze outcomes included the larger group of children with “any wheeze” (ie, any report of wheezing during the first 3 y of life). Also, at the other end of the wheezing spectrum, we looked at 2 subsets of children with recurrent wheeze: 1) those who also had doctor-diagnosis of asthma (by maternal report), and 2) those with ≥4 wheezing attacks (summed over the annual questionnaires). These 2 subgroup analyses excluded subjects with an intermediate phenotype so that children who satisfied the more stringent case definitions were compared with children without any wheeze.
Finally, we examined 2 related outcomes: respiratory infection and eczema. We defined respiratory infection as a positive response to ≥1 of 3 questions on any of the annual questionnaires: Since your child was born (was 12 mo old, was 2 y old), have you been told by a health professional that he or she has bronchiolitis, pneumonia, bronchitis, croup, or any other respiratory infection? We defined eczema as a positive response to one question on any of the annual questionnaires: Have you ever been told by a health care professional that your child has eczema?
Data analysis
To assess the associations between vitamin D intake and clinical outcomes, we used multiple logistic regression models. We assessed the confounding effect of covariates by examining the association of vitamin D intake with outcomes before and after adding the covariates to the model. From the models, we present results using an odds ratio (OR) and 95% CI. We calculated the P for trend across quartiles using median values within each quartile. We examined effect modification of the association of maternal intake of vitamin D with risk of recurrent wheeze by 6 factors: prepregnancy BMI (dichotomized at median of 23.4 kg/m2), maternal calcium intake during pregnancy (1200 mg/d), maternal retinol intake during pregnancy (2500 IU/d), parental history of asthma (yes or no), white maternal race (yes or no), and season of LMP. We first put an interaction term in the model (continuous total vitamin D × factor) and then ran multivariate models stratified by the factor. We performed all analyses using SAS version 8.2 (SAS Institute, Cary, NC).
RESULTS
The mean (±SD) age of mothers at enrollment was 32.5 ± 4.9 y. Most of the mothers (72%) had at least a college education, and 65% lived in households with an annual income of >$70 000. The mean (±SD) prepregnancy BMI was 24.5 ±5.1, and 10% of the mothers smoked during the index pregnancy. Approximately half of the children were male (51%) and 74% were white. Their mean (±SD) birth weight was 3.51 ±0.52 kg, and the mean (±SD) gestational age was 39.6 ±1.5 wk. Children were breastfed for a mean (±SD) of 6.4 ±4.5 mo, and 53% were born into homes with other children aged <12 y. Of the mothers, 31% had either asthma (17%) or eczema (20%); 26% of the fathers had either asthma (14%) or eczema (15%).
The mean (±SD) total vitamin D intake during pregnancy was 548 ±167 IU/d, with an average of 225 IU from food and 319 IU from supplements. Maternal intake of vitamin D during pregnancy was <400 IU/d in 19% of women. Milk was the primary food contributor to vitamin D intake during pregnancy, accounting for 53% of intake; other substantial contributors were fish (18%) and cold cereal (9%).
Maternal vitamin D intake was associated with several factors that might affect risk of asthma (Table 1). Mothers with a higher intake of vitamin D were slightly older, less overweight, of higher socioeconomic status, less likely to smoke during pregnancy, and more likely to have a personal history of eczema. They consumed a little more fish but their intake of fruit and vegetables did not differ. Maternal vitamin D intake was not associated with conception during the winter months (ie, fall birthday), sex, birth weight, or gestational age. However, children of women with a higher maternal intake of vitamin D were more likely to be born to white mothers, to have been breastfed longer, to take a vitamin supplement in the first 6 mo of life, and to consume more vitamin D from foods at age 2 y. For the children, the mean (±SD) vitamin D intake from foods was 249 ± 101 IU/d; milk contributed 71% of vitamin D, fish 1%, and cold cereal 17%. The home environment of mothers with a high vitamin D intake was characterized by less passive smoke exposure and fewer siblings aged <12 y, but no difference in exposure to pets or other common allergens was found.
TABLE 1.
Characteristics of 1194 mother-infant pairs in Project Viva according to maternal intake of vitamin D during pregnancy1
| Quartile of vitamin D intake during pregnancy
|
P for trend2 | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| Vitamin D intake (IU) | |||||
| Median | 356 | 513 | 603 | 724 | |
| Range | 60–445 | 446–562 | 563–658 | 659–1145 | |
| No. of subjects | 298 | 299 | 299 | 298 | |
| Maternal and family history | |||||
| Maternal age at enrollment (y) | 31.3 ± 5.73 | 32.6 ± 4.6 | 32.7 ± 4.5 | 33.5 ± 4.4 | < 0.001 |
| Prepregnancy BMI (kg/m2) | 25.7 ± 6.0 | 24.2 ± 4.9 | 24.1 ± 4.5 | 24.0 ± 4.6 | < 0.001 |
| Maternal dietary intake | |||||
| Vitamin D from foods (IU) | 164 ± 76 | 186 ± 78 | 228 ± 75 | 320 ± 95 | < 0.001 |
| Vitamin D from supplements (IU) | 163 ± 107 | 322 ± 86 | 370 ± 67 | 421 ± 86 | < 0.001 |
| Calcium intake (mg) | 1109 ± 284 | 1327 ± 301 | 1447 ± 299 | 1620 ± 335 | < 0.001 |
| Vitamin A intake (μg RE) | 1663 ± 579 | 2087 ± 577 | 2328 ± 572 | 2582 ± 692 | < 0.001 |
| Fruit and vegetables (servings/d) | 5.8 ± 2.9 | 5.9 ± 2.4 | 6.1 ± 2.5 | 6.0 ± 3.1 | 0.17 |
| Fish (servings/wk) | 1.7 ± 1.4 | 1.8 ± 1.3 | 1.9 ± 1.4 | 2.1 ± 1.7 | < 0.001 |
| College graduate (%) | 53 | 74 | 80 | 82 | < 0.001 |
| Household income >$70 000/y (%) | 55 | 66 | 68 | 71 | < 0.001 |
| Mother smoked during index pregnancy (%) | 13 | 12 | 9 | 6 | 0.001 |
| Family history | |||||
| Maternal asthma (%) | 19 | 16 | 21 | 13 | 0.09 |
| Maternal eczema (%) | 15 | 21 | 18 | 24 | 0.02 |
| Maternal asthma or eczema (%) | 30 | 30 | 32 | 31 | 0.62 |
| Paternal asthma (%) | 18 | 17 | 9 | 14 | 0.05 |
| Paternal eczema (%) | 15 | 17 | 13 | 14 | 0.38 |
| Paternal asthma or eczema (%) | 30 | 28 | 21 | 25 | 0.05 |
| Child and home environment | |||||
| Maternal LMP during winter (%) | 18 | 21 | 24 | 23 | 0.08 |
| Boy (%) | 50 | 52 | 48 | 53 | 0.68 |
| White (%) | 63 | 78 | 78 | 79 | < 0.001 |
| Birth weight (kg) | 3.48 ± 0.52 | 3.53 ± 0.55 | 3.53 ± 0.48 | 3.51 ± 0.51 | 0.54 |
| Gestational age at birth (wk) | 39.5 ± 1.5 | 39.6 ± 1.5 | 39.8 ± 1.4 | 39.6 ± 1.4 | 0.31 |
| Fetal growth (birth-weight-for-GA z score) | 0.16 ± 1.0 | 0.25 ± 1.0 | 0.19 ± 0.9 | 0.20 ± 0.9 | 0.72 |
| Dietary intake during childhood | |||||
| Breastfeeding for ≥9 mo (%) | 32 | 37 | 41 | 49 | < 0.001 |
| Duration of breastfeeding (mo) | 5.7 ± 4.5 | 6.0 ± 4.6 | 6.7 ± 4.5 | 7.3 ± 4.4 | < 0.001 |
| Vitamin supplement use in first 6 mo (%) | 10 | 15 | 18 | 20 | < 0.001 |
| Vitamin D intake from foods at age 2 y (IU) | 225 ± 108 | 248 ± 103 | 251 ± 85 | 267 ± 106 | < 0.001 |
| Passive smoke exposure at home (%) | 14 | 6 | 5 | 6 | < 0.001 |
| Number of children aged <12 y at home | 1.8 ± 0.8 | 1.7 ± 0.8 | 1.6 ± 0.7 | 1.6 ± 0.7 | < 0.001 |
| Furry pet at home (%) | 40 | 42 | 42 | 41 | 0.80 |
| Home cockroach exposure (%) | 4 | 4 | 4 | 3 | 0.50 |
| Moisture or mildew in home (%) | 28 | 28 | 27 | 30 | 0.82 |
RE, retinol equivalents; LMP, last menstrual period; GA, gestational age.
Based on Mantel-Haenszel chi-square test for categorical variables and linear regression for continuous variables.
x̄ ± SD (all such values).
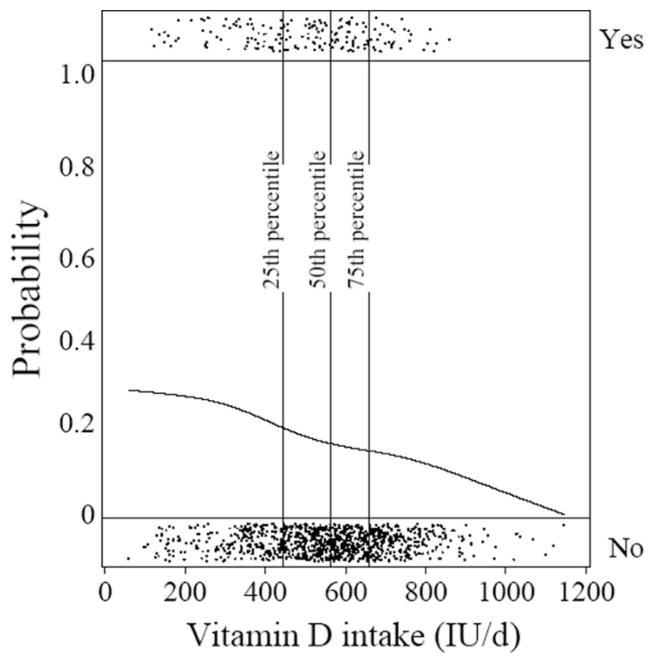
The unadjusted inverse linear association between maternal vitamin D intake and risk of recurrent wheeze in offspring is shown in Figure 1. Because individual observations would otherwise be difficult to distinguish, we added random noise to the display of each observation. The smoothed line shows the approximate probability of recurrent wheeze for each observed value of vitamin D intake. This strong inverse association is confirmed in Table 2 (P for trend <0.001). Compared with mothers in the lowest quartile of daily intake (median within quartile: 356 IU), those in the highest quartile (median: 724 IU) had a lower risk of having a child with recurrent wheeze at age 3 y (OR: 0.39; 95% CI: 0.25, 0.62). The strong inverse association between maternal vitamin D intake and recurrent wheeze was robust to controlling for a variety of potential confounders, including traditional asthma risk factors and putative dietary risk factors such as fish, fruit, and vegetables (Table 2). Further adjustment for a variety of micronutrients (eg, vitamin C, vitamin E, and zinc) did not affect the results (data not shown). In contrast with the decreased risk of recurrent wheeze, maternal intake of vitamin D was not associated with risk of respiratory infections or early childhood eczema (Table 2).
FIGURE 1.
Unadjusted inverse linear association between maternal vitamin D intake and risk of recurrent wheeze in offspring. The top band of dots (Yes) represents children who developed recurrent wheeze, and the bottom band of dots (No) represents children who did not develop recurrent wheeze. The vertical lines represent percentiles of maternal vitamin D intake.
TABLE 2.
Odds ratios (and 95% CIs) for maternal intake of vitamin D during pregnancy and risk of clinical outcomes in children at 3 y of age (n = 1194 mother-infant pairs)
| Quartile of vitamin D intake by mother during pregnancy
|
P for trend1 | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| Vitamin D intake (IU) | |||||
| Median | 356 | 513 | 603 | 724 | |
| Range | 60–445 | 446–562 | 563–658 | 659–1145 | |
| No. of subjects | 298 | 299 | 299 | 298 | |
| Recurrent wheeze (n = 186 cases) | |||||
| Unadjusted | 1.00 | 0.55 (0.36, 0.84) | 0.55 (0.36, 0.83) | 0.39 (0.25, 0.62) | < 0.001 |
| Multivariate model 12 | 1.00 | 0.49 (0.30, 0.80) | 0.56 (0.34, 0.92) | 0.41 (0.24, 0.70) | 0.001 |
| Multivariate model 23 | 1.00 | 0.47 (0.28, 0.77) | 0.54 (0.33, 0.89) | 0.38 (0.22, 0.65) | < 0.001 |
| Respiratory infection (n = 540 cases) | |||||
| Unadjusted | 1.00 | 0.68 (0.48, 0.96) | 0.92 (0.65, 1.30) | 0.68 (0.48, 0.96) | 0.09 |
| Multivariate model 12 | 1.00 | 0.73 (0.51, 1.06) | 1.01 (0.70, 1.45) | 0.74 (0.51, 1.08) | 0.28 |
| Multivariate model 23 | 1.00 | 0.74 (0.51, 1.06) | 1.01 (0.70, 1.46) | 0.75 (0.52, 1.09) | 0.32 |
| Eczema (n = 428 cases) | |||||
| Unadjusted | 1.00 | 1.00 (0.71, 1.39) | 1.03 (0.74, 1.44) | 1.10 (0.79, 1.53) | 0.58 |
| Multivariate model 12 | 1.00 | 0.88 (0.60, 1.27) | 0.94 (0.65, 1.37) | 0.95 (0.65, 1.39) | 0.88 |
| Multivariate model 23 | 1.00 | 0.86 (0.59, 1.25) | 0.93 (0.64, 1.36) | 0.92 (0.63, 1.35) | 0.75 |
From logistic regression models using the vitamin D variable; the median value within each quartile was used as the predictor variable.
Adjusted for sex, birth weight, income, maternal age, maternal prepregnancy BMI, passive smoking exposure, breastfeeding duration at 1 y, number of children <12 y of age in household, maternal history of asthma, and paternal history of asthma. For eczema analyses, the model adjusted for parental history of eczema instead of asthma.
Adjusted for the 10 factors in model 1 plus intakes of fish and fruit and vegetables.
To further explore these results, we examined risk according to vitamin D intake, both during pregnancy and over the first 2 y of life. As shown in Table 1, women with a higher vitamin D intake during pregnancy were more likely to have children who used vitamin supplements and ate more foods with vitamin D. Nevertheless, the association of maternal vitamin D intake with recurrent wheeze (Table 2; model 2) did not change with further control for the child’s use of a vitamin supplement at age 6 mo (OR: 0.39; 95% CI: 0.23, 0.67; P for trend < 0.001) or for the child’s vitamin D intake from foods at 2 y (OR: 0.34; 95% CI: 0.20, 0.60; P for trend < 0.001). This issue is addressed in another way in Figure 2: the mothers and children are stratified by the vitamin D intake of each group, with high-low cutoffs set at 400 IU for mothers and 200 IU for children. The data in the figure show that the decreased risk of recurrent wheeze was largely, if not entirely, due to a high maternal intake of vitamin D during pregnancy.
FIGURE 2.
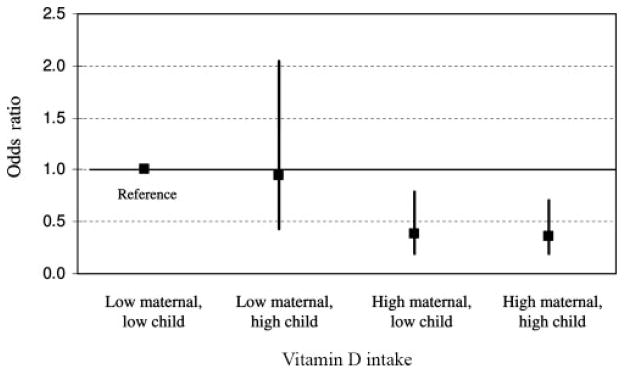
Risk of recurrent wheeze stratified by vitamin D intake. (Groups are split at 400 IU vitamin D for mothers during pregnancy and at 200 IU vitamin D for children at age 2 y.) Odds ratios are adjusted for the 12 factors in multivariate model 2 (see Table 2). In a formal test for interaction, the main effect of maternal vitamin D intake was significant (P <0.001), but the main effect of child vitamin D intake and the interaction term for maternal-child vitamin D intake were not (P > 0.80 for both).
The risk of recurrent wheeze with maternal vitamin D intake, expressed as a 100-IU/d increase rather than in categories, is shown in Table 3. The results closely resemble the strong inverse association shown in Table 2. Moreover, the results were similar for vitamin D from food only and for vitamin D from supplements only. Further adjustment for race-ethnicity and college education did not change the inverse association between maternal vitamin D intake and risk of recurrent wheeze (OR: 0.81; 95% CI: 0.72, 0.90). We also examined the potential effect of 2 dietary factors correlated with vitamin D intake: retinol (r = 0.53) and calcium (r = 0.57). Neither nutrient confounded the strong inverse association between maternal vitamin D intake and risk of recurrent wheeze (Table 3).
TABLE 3.
Odds ratios (and 95% CIs) for maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age (n = 1194 mother-infant pairs)
| Vitamin D intake (per 100 IU/d)
|
|||
|---|---|---|---|
| Total | Food only | Supplement only | |
| Unadjusted | 0.81 (0.74, 0.89) | 0.81 (0.69, 0.96) | 0.82 (0.73, 0.92) |
| Multivariate model A1 | 0.80 (0.72, 0.90) | 0.79 (0.65, 0.96) | 0.82 (0.71, 0.95) |
| Multivariate model B2 | — | 0.80 (0.66, 0.97) | 0.82 (0.71, 0.95) |
| Multivariate model C3 | 0.79 (0.69, 0.91) | — | — |
| Multivariate model D4 | 0.81 (0.71, 0.93) | — | — |
Adjusted for sex, birth weight, income, maternal age, maternal prepregnancy BMI, passive smoking exposure, breastfeeding duration at 1 y, number of children <12 y of age in household, maternal history of asthma, paternal history of asthma, and intakes of fish and fruit and vegetables.
Adjusted for the 12 factors in model A plus maternal intake of vitamin D from supplements (in food-only models) and vitamin D from foods (in supplement-only models).
Adjusted for the 12 factors in model A plus maternal intake of calcium.
Adjusted for the 12 factors in model A plus maternal intake of retinol.
To further explore the spectrum of wheezing illnesses of childhood, we looked at the relation of maternal vitamin D intake with risk of any wheeze (n = 416 cases) and with subsets of the primary outcome: recurrent wheeze with doctor-diagnosed asthma (n =132 cases) and recurrent wheeze with ≥4 wheezing episodes (n = 86 cases). For each 100-IU increase in maternal vitamin D intake, we observed a somewhat weaker but still significantly lower risk of any wheeze (OR: 0.89; 95% CI: 0.82, 0.97). The inverse association appeared stronger for the most stringent wheeze outcomes: children with recurrent wheeze and doctor-diagnosed asthma (OR: 0.82; 95% CI: 0.71, 0.93) or recurrent wheeze with ≥4 wheezing episodes (OR: 0.79; 95% CI: 0.67, 0.93). All of these values were adjusted for the 12 factors in model A (Table 3).
As noted earlier, a major dietary source of vitamin D is fortified milk. Accordingly, we examined the association between milk intake by mothers during pregnancy and risk of clinical outcomes in their children. Compared with mothers who did not drink milk (who had a median vitamin D intake of 479 IU from other foods and supplements), and after adjustment for 12 factors, risk of recurrent wheeze was lowest among those drinking 1–1.9 cups/d (OR: 0.35; 95% CI: 0.16, 0.80). The associations were less extreme for those drinking <1 cup/d (OR: 0.57; 95% CI: 0.27, 1.24) or ≥2 cups/d (OR: 0.45; 95% CI: 0.20, 1.02). The overall P value for milk in this multivariate model was 0.046, whereas the P for linear trend (across categories of increasing milk intake) was 0.13.
Finally, we explored effect modification of the association of maternal intake of vitamin D with risk of recurrent wheeze. Only season of LMP showed a likely interaction (P = 0.06). Maternal intake of vitamin D was associated with a lower risk of recurrent wheeze when the LMP was in the winter. The multivariate OR per 100-IU/d increase was 0.62 (95% CI: 0.47, 0.83) for LMP in winter compared with 0.85 (95% CI: 0.75, 0.97) in other seasons.
DISCUSSION
In this prospective cohort study of almost 1200 mother-child pairs from the northeastern United States, a higher maternal intake of vitamin D during pregnancy was associated with a lower risk of recurrent wheeze in children 3 y of age. The inverse association was independent of many potential confounders, including adjustment for fish intake, fruit and vegetable intake, early use of a vitamin supplement, and child’s intake of vitamin D at age 2 y. The association was present for vitamin D from either foods or nutritional supplements. In addition, among children conceived in the winter months and therefore born in the fall (a time of low ultraviolet light B exposure), the inverse association between maternal intake of vitamin D and risk of recurrent wheeze was stronger.
In recent years, vitamin D has been a focus of growing interest in public health nutrition (2, 3). Vitamin D insufficiency is common in the United States, especially in northern latitudes (4, 5). This insufficiency is multifactorial but probably stems from a combination of decreased dietary intake of vitamin D (eg, from fortified milk or fish) and decreased sun exposure (eg, because of lifestyle choices or increased use of sunscreen). Indeed, northern populations receive too few ultraviolet light B rays between November and March to generate sufficient vitamin D in the skin (4), and studies suggest that one cannot drink enough milk to overcome this shortfall (30). The reemergence of childhood rickets in recent years (31) has led the American Academy of Pediatrics to recommend vitamin D supplementation for high-risk groups (32). Most nutritionists rely on the 1997 Institute of Medicine recommendations (33), but a growing number are recommending that high-risk populations take a daily supplement of 800 to 1000 IU (34).
At present, very little is known about how vitamin D affects the lungs. Some studies report findings that are in general agreement with ours. For example, Black and Scragg (35) recently reported cross-sectional data from the third National Health and Nutrition Examination Survey and found that serum 25-hydroxyvitamin D was inversely associated with forced expiratory volume in 1 s and forced vital capacity in the US general population. The authors and the accompanying editorial (36) called for studies to determine whether vitamin D is of any benefit in patients with respiratory disease. Concurrent with this population-based study, Xystrakis et al (37) in London administered vitamin D to a small group of healthy persons and steroid-resistant asthmatic patients and found that the intervention enhanced subsequent responsiveness to dexamethasone for induction of interleukin (IL)-10. The authors suggested that vitamin D could potentially increase the therapeutic response to glucocorticoids in steroid-resistant asthma patients.
In contrast, a birth cohort in northern Finland reported that regular vitamin D supplementation in the first year of life increased the risks of developing atopy, allergic rhinitis, and asthma by age 31 y (38). The authors, however, collected no information on maternal intakes of the vitamin, and control for major confounders was limited. Furthermore, recall bias may have affected the ascertainment of early life asthma and allergies. Although these results are consistent with historical concerns about the potential adverse effects of milk intake on asthma and allergic diseases, most studies do not support this hypothesis (39). Indeed, the few epidemiologic studies that have examined the issue have reported an inverse association between milk intake and risk of asthma in young children (40, 41).
Laboratory research suggests several potential mechanisms for how vitamin D can affect risk of asthma. For example, several recent genetic association studies (42–45) suggest that polymorphisms of the VDR gene are related to obstructive pulmonary disease. Other proposed mechanisms include modulation of antigen-presenting cells, such as macrophages (46, 47), and the generation of regulatory T cells (48, 49) that express potentially inhibitory cytokines (IL-10 and transforming growth factor β), and the ability to potently inhibit antigen-specific T cell activation (50). A murine model of pulmonary eosinophilic inflammation showed that vitamin D supplementation of adult mice led to changes in cytokines, immunoglobulin E concentrations, and airway eosinophilia during allergen sensitization (51). Although many laboratory studies suggest that vitamin D induces a shift in the balance between T helper subset 1 (Th1)– and Th2-type cytokines toward Th2 dominance (12), Pichler et al (52) found that in CD4+ and CD8+ human cord blood cells, vitamin D inhibits not only IL-12–generated interferon γ production but also suppresses IL-4 and IL-4-induced expression of IL-13. If a shift away from the fetal Th2-type pattern in humans is linked with asthma protection, our maternal intake findings are consistent with the cytokine findings of Pichler et al. Thus, the differences between the studies on the Th1-Th2 dominance may lie in the timing of exposure of the cells to vitamin D (ie, prenatal versus postnatal); the response of naïve T cells to vitamin D exposure may differ from that of mature cells when exposed to vitamin D (53). Another possibility is that the association depends on the vitamin D status of the individual. In other words, lower vitamin D intakes (eg, to replete a deficiency state) may have different consequences than relatively high-dose supplementation, whereas an excess of vitamin D may indeed have opposite effects. These hypotheses merit further investigation.
Our study has several potential limitations. The early age of outcome measurement limits our ability to generalize these findings from recurrent wheeze to actual asthma. Many children who wheeze before 3 y of age have transient episodes and do not go on to have asthma (54). However, the chosen 3-y outcome carries an almost 10-fold increased risk of developing asthma by age 6 y (29). Continued follow-up of our birth cohort will determine whether the findings translate to a decreased risk of asthma later in childhood.
Another limitation is that all participants came from Massachusetts, were predominantly white, and were of higher socioeconomic status; the generalizability of the observed associations merits further study. However, on the basis of a growing body of literature on vitamin D, we anticipate stronger associations among African Americans and Hispanics living in the northern United States. Large interindividual differences in time spent outdoors and in sunscreen use will complicate the generalizability of these diet-asthma associations to regions with more sunlight exposure.
In summary, we found a strong inverse association between maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. The protective association was present for vitamin D from both diet and nutritional supplements and was observed in the context of apparently adequate intakes of vitamin D (33) for most women in this northeastern US population. Pregnant and lactating women are known to be at higher risk of vitamin D deficiency (55), and our findings provide additional support for efforts to improve the nutritional status of this population, including recommended intakes of >400 IU/d. The primary implications of our findings are in the research arena, eg, the need for additional prospective studies to confirm or refute our vitamin D–asthma hypothesis. If others replicate our findings, we would support the initiation of randomized trials of vitamin D repletion in populations at high-risk of asthma morbidity and mortality. The low cost and safety of vitamin D–containing foods and supplements could provide a very attractive intervention for the primary prevention of asthma.
Acknowledgments
We thank the participants and staff of Project Viva.
CAC, AAL, JWR-E, STW, DRG, and MWG designed the study, secured the funding, managed the data collection, and supervised the study. SLR-S and KK analyzed the data. CAC drafted the manuscript. All authors provided critical revisions for important intellectual content.
Footnotes
Supported by grants HL64925, HD34568, HL68041, and HL75504 from the National Institutes of Health (Bethesda, MD); the Harvard Medical School and Harvard Pilgrim Health Care Foundation (Boston, MA); and the Massachusetts General Hospital Center for D-receptor Activation Research (Boston, MA).
Reprints not available.
None of the authors had a conflict of interest to disclose.
References
- 1.Feldman D, Pike JW, Glorieux FH. Vitamin D. 2. Amsterdam, Netherlands: Elsevier Academic Press; 2005. [Google Scholar]
- 2.Holick MF. Vitamin D. Importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am J Clin Nutr. 2004;79:362–71. doi: 10.1093/ajcn/79.3.362. [DOI] [PubMed] [Google Scholar]
- 3.Garland CF, Garland FC, Gorham ED, et al. The role of vitamin D in cancer prevention. Am J Public Health. 2006;96:252–61. doi: 10.2105/AJPH.2004.045260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J Clin Endocrinol Metab. 1988;67:373–8. doi: 10.1210/jcem-67-2-373. [DOI] [PubMed] [Google Scholar]
- 5.Thomas MK, Lloyd-Jones DM, Thadhani RI, et al. Hypovitaminosis D in medical inpatients. N Engl J Med. 1998;338:777–83. doi: 10.1056/NEJM199803193381201. [DOI] [PubMed] [Google Scholar]
- 6.Hollis BW. Circulating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: implications for establishing a new effective dietary intake recommendation for vitamin D. J Nutr. 2005;135:317–22. doi: 10.1093/jn/135.2.317. [DOI] [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. Self-reported asthma prevalence and control among adults—United States, 2001. MMWR Morb Mortal Wkly Rep. 2003;52:381–4. [PubMed] [Google Scholar]
- 8.Mannino DM, Homa DM, Akinbami LJ, Moorman JE, Gwynn C, Redd SC. Surveillance for asthma–United States, 1980–1999. MMWR Morb Mortal Wkly Rep Surveill Summ. 2002;51:1–13. [PubMed] [Google Scholar]
- 9.Yunginger JW, Reed CE, O’Connell EJ, Melton JL, III, O’Fallon WM, Silverstein MD. A community-based study of the epidemiology of asthma: incidence rates, 1964–1983. Am Rev Respir Dis. 1992;146:888–94. doi: 10.1164/ajrccm/146.4.888. [DOI] [PubMed] [Google Scholar]
- 10.Wainwright C, Isles AF, Francis PW. Asthma in children. Med J Aust. 1997;167:218–23. [PubMed] [Google Scholar]
- 11.McKeever TM, Britton J. Diet and asthma. Am J Respir Crit Care Med. 2004;170:725–9. doi: 10.1164/rccm.200405-611PP. [DOI] [PubMed] [Google Scholar]
- 12.Cantorna MT, Zhu Y, Froicu M, Wittke A. Vitamin D status, 1,25-dihydroxyvitamin D3, and the immune system. Am J Clin Nutr. 2004;80(suppl):1717S–20S. doi: 10.1093/ajcn/80.6.1717S. [DOI] [PubMed] [Google Scholar]
- 13.Gillman MW. Developmental origins of health and disease. N Engl J Med. 2005;353:1848–50. doi: 10.1056/NEJMe058187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Litonjua AA, Rifas-Shiman SL, Ly NP, et al. Maternal antioxidant intake in pregnancy and wheezing illnesses in children at 2 y of age. Am J Clin Nutr. 2006;84:903–11. doi: 10.1093/ajcn/84.4.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gern JE, Lemanske RF, Busse WW. Early life origins of asthma. J Clin Invest. 1999;104:837–43. doi: 10.1172/JCI8272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Warner JA, Jones CA, Jones AC, Warner JO. Prenatal origins of allergic disease. J Allergy Clin Immunol. 2000;105(suppl):S493–8. doi: 10.1016/s0091-6749(00)90049-6. [DOI] [PubMed] [Google Scholar]
- 17.Nesby-O’Dell S, Scanlon KS, Cogswell ME, et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: third National Health and Nutrition Examination Survey, 1988–1994. Am J Clin Nutr. 2002;76:187–92. doi: 10.1093/ajcn/76.1.187. [DOI] [PubMed] [Google Scholar]
- 18.Arunabh S, Pollack S, Yeh J, Aloia JF. Body fat content and 25-hydroxyvitamin D levels in healthy women. J Clin Endocrinol Metab. 2003;88:157–61. doi: 10.1210/jc.2002-020978. [DOI] [PubMed] [Google Scholar]
- 19.Matsuoka LY, Wortsman J, Hanifan N, Holick MF. Chronic sunscreen use decreases circulating concentrations of 25-hydroxyvitamin D. A preliminary study. Arch Dermatol. 1988;124:1802–4. [PubMed] [Google Scholar]
- 20.Striegel-Moore RH, Thompson D, Affenito SG, et al. Correlates of beverage intake in adolescent girls: the National Heart, Lung, and Blood Institute Growth and Health Study. J Pediatr. 2006;148:183–7. doi: 10.1016/j.jpeds.2005.11.025. [DOI] [PubMed] [Google Scholar]
- 21.Gillman MW, Rich-Edwards JW, Rifas-Shiman S, Lieberman E, Kleinman K, Lipshultz S. Maternal age and other predictors of newborn blood pressure. J Pediatr. 2004;144:240–5. doi: 10.1016/j.jpeds.2003.10.064. [DOI] [PubMed] [Google Scholar]
- 22.Fawzi WW, Rifas-Shiman SL, Rich-Edwards JW, Willett WC, Gillman MW. Calibration of a semi-quantitative food frequency questionnaire in early pregnancy. Ann Epidemiol. 2004;14:754–62. doi: 10.1016/j.annepidem.2004.03.001. [DOI] [PubMed] [Google Scholar]
- 23.Willett WC. Nutritional epidemiology. 2. New York, NY: Oxford University Press; 1998. [Google Scholar]
- 24.US Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, release 14. Washington, DC: USDA; 2001. [Google Scholar]
- 25.Rifas-Shiman SL, Rich-Edwards JW, Willett WC, Kleinman KP, Oken E, Gillman MW. Changes in dietary intake from the first to the second trimester of pregnancy. Paediatr Perinat Epidemiol. 2006;20:35–42. doi: 10.1111/j.1365-3016.2006.00691.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Devereux G, Seaton A. Diet as a risk factor for atopy and asthma. J Allergy Clin Immunol. 2005;115:1109–17. doi: 10.1016/j.jaci.2004.12.1139. [DOI] [PubMed] [Google Scholar]
- 27.Oken E, Kleinman KP, Rich-Edwards JW, Gillman MW. A nearly continuous measure of birth weight for gestational age using a United States national reference. BMC Pediatr. 2003;3:6. doi: 10.1186/1471-2431-3-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Blum RE, Wei EK, Rockett HR, et al. Validation of a food frequency questionnaire in Native American and Caucasian children 1 to 5 years of age. Matern Child Health J. 1999;3:167–72. doi: 10.1023/a:1022350023163. [DOI] [PubMed] [Google Scholar]
- 29.Castro-Rodriguez JA, Holberg CJ, Wright AL, Martinez FD. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med. 2000;162:1403–6. doi: 10.1164/ajrccm.162.4.9912111. [DOI] [PubMed] [Google Scholar]
- 30.Tangpricha V, Pearce EN, Chen TC, Holick MF. Vitamin D insufficiency among free-living healthy young adults. Am J Med. 2002;112:659–62. doi: 10.1016/s0002-9343(02)01091-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rajakumar K, Thomas SB. Reemerging nutritional rickets: a historical perspective. Arch Pediatr Adolesc Med. 2005;159:335–41. doi: 10.1001/archpedi.159.4.335. [DOI] [PubMed] [Google Scholar]
- 32.Greer FR. Issues in establishing vitamin D recommendations for infants and children. Am J Clin Nutr. 2004;80(suppl):1759S–62S. doi: 10.1093/ajcn/80.6.1759S. [DOI] [PubMed] [Google Scholar]
- 33.Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Food and Nutrition Board, Institute of Medicine. DRI: Dietary Reference Intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, DC: National Academy Press; 1999. [Google Scholar]
- 34.Hanley DA, Davison KS. Vitamin D insufficiency in North America. J Nutr. 2005;135:332–7. doi: 10.1093/jn/135.2.332. [DOI] [PubMed] [Google Scholar]
- 35.Black PN, Scragg R. Relationship between serum 25-hydroxyvitamin D and pulmonary function in the Third National Health and Nutrition Examination Survey. Chest. 2005;128:3792–8. doi: 10.1378/chest.128.6.3792. [DOI] [PubMed] [Google Scholar]
- 36.Wright RJ. Make no bones about it: increasing epidemiologic evidence links vitamin D to pulmonary function and COPD. Chest. 2005;128:3781–3. doi: 10.1378/chest.128.6.3781. [DOI] [PubMed] [Google Scholar]
- 37.Xystrakis E, Kusumakar S, Boswell S, et al. Reversing the defective induction of IL-10-secreting regulatory T-cells in glucocorticoid-resistant asthma patients. J Clin Invest. 2006;116:146–55. doi: 10.1172/JCI21759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hypponen E, Sovio U, Wjst M, et al. Infant vitamin D supplementation and allergic conditions in adulthood: northern Finland birth cohort 1966. Ann N Y Acad Sci. 2004;1037:84–95. doi: 10.1196/annals.1337.013. [DOI] [PubMed] [Google Scholar]
- 39.Wuthrich B, Schmid A, Walther B, Sieber R. Milk consumption does not lead to mucus production or occurrence of asthma. J Am Coll Nutr. 2005;24(suppl):547S–55S. doi: 10.1080/07315724.2005.10719503. [DOI] [PubMed] [Google Scholar]
- 40.Wijga AH, Smit HA, Kerkhof M, et al. Association of consumption of products containing milk fat with reduced asthma risk in pre-school children: the PIAMA birth cohort study. Thorax. 2003;58:567–72. doi: 10.1136/thorax.58.7.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hijazi N, Abalkhail B, Seaton A. Diet and childhood asthma in a society in transition: a study in urban and rural Saudi Arabia. Thorax. 2000;55:775–9. doi: 10.1136/thorax.55.9.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Laufs J, Andrason H, Sigvaldason A, et al. Association of vitamin D binding protein variants with chronic mucus hypersecretion in Iceland. Am J Pharmacogenomics. 2004;4:63–8. doi: 10.2165/00129785-200404010-00007. [DOI] [PubMed] [Google Scholar]
- 43.Poon AH, Laprise C, Lemire M, et al. Association of vitamin D receptor genetic variants with susceptibility to asthma and atopy. Am J Respir Crit Care Med. 2004;170:967–73. doi: 10.1164/rccm.200403-412OC. [DOI] [PubMed] [Google Scholar]
- 44.Raby BA, Lazarus R, Silverman EK, et al. Association of vitamin D receptor gene polymorphisms with childhood and adult asthma. Am J Respir Crit Care Med. 2004;170:1057–65. doi: 10.1164/rccm.200404-447OC. [DOI] [PubMed] [Google Scholar]
- 45.Wjst M. Variants in the vitamin D receptor gene and asthma. BMC Genet. 2005;6:2. doi: 10.1186/1471-2156-6-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Griffin MD, Xing N, Kumar R. Vitamin D and its analogs as regulators of immune activation and antigen presentation. Annu Rev Nutr. 2003;23:117–45. doi: 10.1146/annurev.nutr.23.011702.073114. [DOI] [PubMed] [Google Scholar]
- 47.Lin R, White JH. The pleiotropic actions of vitamin D. Bioessays. 2004;26:21–8. doi: 10.1002/bies.10368. [DOI] [PubMed] [Google Scholar]
- 48.Gregori S, Giarratana N, Smiroldo S, Uskokovic M, Adorini L. A 1alpha,25-dihydroxyvitamin D(3) analog enhances regulatory T-cells and arrests autoimmune diabetes in NOD mice. Diabetes. 2002;51:1367–74. doi: 10.2337/diabetes.51.5.1367. [DOI] [PubMed] [Google Scholar]
- 49.Meehan MA, Kerman RH, Lemire JM. 1,25-Dihydroxyvitamin D3 enhances the generation of nonspecific suppressor cells while inhibiting the induction of cytotoxic cells in a human MLR. Cell Immunol. 1992;140:400–9. doi: 10.1016/0008-8749(92)90206-5. [DOI] [PubMed] [Google Scholar]
- 50.Schwartz RH. Natural regulatory T cells and self-tolerance. Nat Immunol. 2005;6:327–30. doi: 10.1038/ni1184. [DOI] [PubMed] [Google Scholar]
- 51.Matheu V, Back O, Mondoc E, Issazadeh-Navikas S. Dual effects of vitamin D-induced alteration of TH1/TH2 cytokine expression: enhancing IgE production and decreasing airway eosinophilia in murine allergic airway disease. J Allergy Clin Immunol. 2003;112:585–92. doi: 10.1016/s0091-6749(03)01855-4. [DOI] [PubMed] [Google Scholar]
- 52.Pichler J, Gerstmayr M, Szepfalusi Z, Urbanek R, Peterlik M, Willheim M. 1 alpha,25(OH)2D3 inhibits not only Th1 but also Th2 differentiation in human cord blood T cells. Pediatr Res. 2002;52:12–8. doi: 10.1203/00006450-200207000-00005. [DOI] [PubMed] [Google Scholar]
- 53.Annesi-Maesano I. Perinatal events, vitamin D, and the development of allergy. Pediatr Res. 2002;52:3–5. doi: 10.1203/00006450-200207000-00003. [DOI] [PubMed] [Google Scholar]
- 54.Martinez FD, Wright AL, Taussig LM, Holberg CJ, Halonen M, Morgan WJ. Asthma and wheezing in the first six years of life. N Engl J Med. 1995;332:133–8. doi: 10.1056/NEJM199501193320301. [DOI] [PubMed] [Google Scholar]
- 55.Hollis BW, Wagner CL. Assessment of dietary vitamin D requirements during pregnancy and lactation. Am J Clin Nutr. 2004;79:717–26. doi: 10.1093/ajcn/79.5.717. [DOI] [PubMed] [Google Scholar]