Abstract
Background and Aims Following the consensus view for unitary origin and conserved function of stomata across over 400 million years of land plant evolution, stomatal abundance has been widely used to reconstruct palaeo-atmospheric environments. However, the responsiveness of stomata in mosses and hornworts, the most basal stomate lineages of extant land plants, has received relatively little attention. This study aimed to redress this imbalance and provide the first direct evidence of bryophyte stomatal responsiveness to atmospheric CO2.
Methods A selection of hornwort (Anthoceros punctatus, Phaeoceros laevis) and moss (Polytrichum juniperinum, Mnium hornum, Funaria hygrometrica) sporophytes with contrasting stomatal morphologies were grown under different atmospheric CO2 concentrations ([CO2]) representing both modern (440 p.p.m. CO2) and ancient (1500 p.p.m. CO2) atmospheres. Upon sporophyte maturation, stomata from each bryophyte species were imaged, measured and quantified.
Key Results Densities and dimensions were unaffected by changes in [CO2], other than a slight increase in stomatal density in Funaria and abnormalities in Polytrichum stomata under elevated [CO2].
Conclusions The changes to stomata in Funaria and Polytrichum are attributed to differential growth of the sporophytes rather than stomata-specific responses. The absence of responses to changes in [CO2] in bryophytes is in line with findings previously reported in other early lineages of vascular plants. These findings strengthen the hypothesis of an incremental acquisition of stomatal regulatory processes through land plant evolution and urge considerable caution in using stomatal densities as proxies for paleo-atmospheric CO2 concentrations.
Keywords: Atmospheric CO2, bryophytes, carbon dioxide, evolution, hornworts, mosses, palaeo-atmospheric environment, stomatal density, Anthoceros punctatus, Phaeoceros laevis, Polytrichum juniperinum, Mnium hornum, Funaria hygrometrica
INTRODUCTION
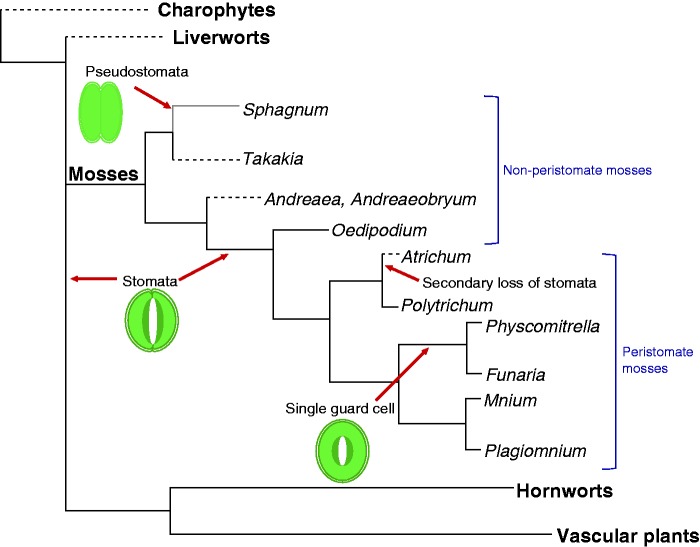
Stomata are considered one of the crucial adaptations in the evolution of the land flora and the development of the terrestrial landscape and atmosphere on Earth. These microscopic pores on the plant epidermis first appeared in the fossil record more than 400 million years ago, some 50–60 million years after the first land plants (Edwards et al., 1998). Today they are found on the sporophyte generations of all land plant groups with exceptions only in the liverworts, the earliest moss lineages and a few derived hornwort clades (Fig. 1). Exposure to high atmospheric CO2 concentration ([CO2]) has been shown to consistently result in a reduction of stomatal density (number of stomata per mm2) and index (ratio of stomata to epidermal cells) in the newly developed leaves of many vascular plant species (Woodward, 1987; Beerling et al., 1998). Today, the general consensus view is that stomatal morphology is conserved throughout land plants (Edwards et al., 1998) and that their primary function is related to the regulation of gas and water exchange, [CO2] being a key activator of stomatal frequency (Woodward, 1987; Woodward and Bazzaz, 1988). This is in line with Francis Darwin’s observations of more than a century ago (Darwin, 1898). Consequently, stomatal abundance in the fossilized remains of ancient plants has been widely used as a proxy to reconstruct palaeo-atmospheric [CO2] (e.g. Beerling et al., 1995; Beerling and Woodward, 1997; Berner, 1998; Beerling et al., 2001; Beerling and Royer, 2002). However, it should be noted that, though the size of stomata in angiosperms, commonly measured as guard cell length, has been shown to be positively correlated with genome size (Beaulieu et al., 2008), a similar relationship is absent in bryophytes (Table 1).
Fig. 1.
Single-phylogram scenario illustrating key land plant lineages (bold text) and moss genera and the appearance of stomata in modern plants. Dashed lines indicate absence of stomata and solid black lines their presence. This phylogram with liverworts basal (Qiu et al., 2006, 2007; Liu et al., 2014) indicates a single origin of stomata and multiple losses, whereas an alternative topology with hornworts basal implies multiple origins (Haig, 2013; Wickett et al., 2014).
Table 1.
Features of the stomata of a selection of bryophytes showing the lack of any clear cut relationships between guard cell numbers, sizes, aperture sizes and genome sizes. The size and numbers data are our own (with mean values in bold) except where indicated otherwise
| Taxon | Number/sporophyte | Size: length × width (μm) | Position | Spacing | Orientation | Pore type | Aperture surface area (μm2) | Guard cell number | Subsidiary cells | +/–calyptra | 1C value (pg) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mosses | ||||||||||||
| Sphagnales | ||||||||||||
| Sphagnum palustre | ∼400 | 42·7 ± 4·2 × 43·2 ± 6·0 | Superficial | Regular | Longitudinal | Absent | – | 2 | − | + | 0·92a 0·57b | |
| Sphagnum tenellum | ∼200 | 33·2 ± 5·1 × 34·7 ± 6·6 | Superficial | Regular | Longitudinal | Absent | – | 2 | − | + | 0·47a | |
| Oedipodiales | ||||||||||||
| Oedipodium griffithianum | ∼60 | 46·8 ± 7·4 × 36·5 ± 7·0 | Superficial | Regular | Longitudinal | Long | NA | 2 | +/− | − | NA | |
| Polytrichales | ||||||||||||
| Atrichum undulatum | 0 | – | – | – | – | – | – | − | + | 0·73–1·95c | ||
| Polytrichum formosum | 180–200 | 72·4 ± 8·9 × 48·7 ± 9·3 | Superficial | Irregular | Mainly longitudinal | Long | NA | 2 (3, 4) | + | + | 0·53c | |
| Polytrichum juniperinum | 42–49–55 | 83 ± 1·2 × 57 ± 1·1 | Superficial | Irregular | Mainly longitudinal | Long | 176·6 ± 12·6 | 2 (3, 4) | + | + | 0·42c | |
| Funariales | ||||||||||||
| Funaria hygrometrica | 160–178–220 | 41 ± 0·74 × 33 ± 0·59 | Superficial | Regular | Longitudinal | Long | 90·5 ± 1·9 (17–29)f | 1 | + | − | 0·40–0·94b,c | |
| Physcomitrella patens | 12–14 | 29·9 ± 1·1 × 25·1 ± 2·2 | Superficial | Slightly irregular | Random | Round | Green 39·4 ± 1·6; mature 42·2 ± 1·5 (13–17 µm2)f | 1 | − | − | 0·55d | |
| Bryales | ||||||||||||
| Mnium hornum | 21–30–38 | 45 ± 0·7 × 41 ± 0·8 | Sunken | Slightly irregular | Random | Round | 26·2 ± 1·5 | 2 | + | − | 0·88c | |
| 0·53b | ||||||||||||
| Plagiomnium cuspidatum | ∼60 | 28–32 × 25–28 | Sunken | Slightly irregular | Random | Round | NA | 2 | + | − | 1·3c | |
| Hornworts | ||||||||||||
| Anthoceros punctatus | Low density: 3–4·5/mm2 | Large: 55 ± 0·7 × 34 ± 0·3 | Superficial | Regular | Longitudinal | Long | 159·5 ± 8·9 | 2 | − | Inside involucre | 0·18e | |
| Phaeoceros laevis | Low density: 2·5–4/mm2 | Large: 67 ± 1·0 × 38 ± 0·6 | Superficial | Regular | Longitudinal | Long | 242·8 ± 9·2 | 2 | − | Inside involucre | 0·24e | |
aTemsch et al., 1998; bRenzaglia et al., 1995; cVoglmayr, 2000; dRensing et al., 2008; eBainard and Villarreal, 2013; fChater et al., 2011.
NA, not available.
The stomatal response to [CO2] is developmental, involving long-distance signalling from mature to new leaves (Lake et al., 2001, 2002). This serves to maximize water use efficiency under high [CO2] and to ensure optimal photosynthesis (Brodribb and McAdam, 2013). Some groups of land plants, however, including cycads, other gymnosperms and ferns, have recently been found to be unresponsive to such changes in [CO2] (Brodribb et al., 2009; Haworth et al., 2011). To date, similar studies examining stomatal responses to [CO2], including those relevant to plant evolutionary timescales (e.g. Berner, 2006), across a suite of non-vascular plants are missing. However, it is widely assumed that stomata have evolved once and their functioning and regulation were conserved from mosses through to angiosperms (Franks and Beerling, 2009). Underpinning these tenets of structural and functional congruence for the early evolution of the stomatal ‘toolkit’ some 400 million years ago are recent demonstrations in the moss Physcomitrella (Chater et al., 2011) and the lycopod Selaginella (Ruszala et al., 2011) of the same mechanisms actively regulating stomatal movements as those found in angiosperms, particularly pore closure responses to the plant hormone abscisic acid (ABA). This hormone is also associated with desiccation tolerance in several plant groups, including the mosses (Bopp and Werner, 1993; Mayaba et al., 2001; Stark et al., 2007), and has been shown to initiate stomatal closure under elevated [CO2] (Chater et al., 2014). The discovery that numerous stomatal genes, including those determining density, are common to both vascular plants and mosses (Chater et al., 2011), further supports the hypothesis that the first stomata to evolve more than 400 million years ago in non-vascular plants were analogous to their modern angiosperm counterparts both in function and in their active regulation via ABA-mediated opening and closing. Counter to this, based on their findings that stomata in a group of six ferns and a lycophyte do not respond to ABA by closure of stomatal pores, Brodribb and McAdam (2011) raised the hypothesis that ‘early-diverging clades of vascular land plants may preserve an ancestral stomatal behaviour that predates much of the complexity present in angiosperm stomatal responses’. Their findings support this, suggesting that ABA-mediated ‘active’ control of stomata is likely to have evolved after the divergence of the ferns and lycophytes.
However, a crucial missing piece in the jigsaw of stomatal evolution and function is the responsiveness of bryophyte stomata to [CO2]. In the only previous studies on the effects of [CO2] on stomata in non-vascular plants, Chater et al. (2011) recorded larger apertures in the mosses Physcomitrella and Funaria grown in the absence of CO2. Baars and Edwards (2008) reported a decrease in stomatal size and density in Leptobryum pyriforme, but absolute number of stomata per capsule remained the same at 10 times ambient [CO2]. These results were interpreted as general growth responses with stomatal numbers pre-programmed in the closed determinate development of moss sporophytes, and highlight the need for further studies on a range of taxa (Baars and Edwards, 2008). Such developmental constraints are absent in hornworts, where stomata are produced continuously from derivatives of the sporophyte meristem (Pressel et al., 2014). The responsiveness of hornwort stomata to [CO2] potentially holds particular significance, given their placement as sister group to all land plants in a study by Wickett et al. (2014) and contrary to their position as sister to vascular plants in other phylogenies (Qiu et al., 2006, 2007; Liu et al., 2014). It must therefore be underlined that considerable uncertainty remains as to the precise relationships between the bryophyte groups at the foot of the land plant tree.
Previous studies have suggested that hornwort stomata close in response to environmental stimuli and exogenous application of ABA (Hartung et al., 1987; Bopp and Werner, 1993; Hartung, 2010). Conversely, it has also been reported that application of ABA does not elicit stomatal closure in hornworts (Lucas and Renzaglia, 2002). Consequently, the function and CO2 responsiveness of stomata in hornworts, in addition to mosses, and their significance in the evolution of active control of stomata in land plants remain unclear. In the present study, we redress this imbalance and challenge the widely accepted dogma that the responsiveness of stomata to [CO2] in terms of density and opening is conserved across the land plant phylogeny through careful experimentation and cytological observation. Specifically, we address the following questions: (1) Are stomatal numbers on moss and hornwort sporophytes affected in the same way as those in angiosperms by elevated [CO2] representative of atmospheric concentrations in the Palaeozoic (Berner, 2006)? (2) Do guard cell lengths and apertures in bryophytes change when subjected to representative Palaeozoic [CO2] throughout development?
MATERIALS AND METHODS
Plant materials and growth
Wild plants from the same populations of three mosses (Polytrichum juniperinum, Mnium hornum and Funaria hygrometrica) with young sporophytes prior to capsule expansion and two hornworts (Anthoceros punctatus, Phaeoceros laevis) with sporophytes protruding just 1–2 mm above the tops of the involucres, were collected in southern England between early January and late March 2014. Extensive observations on the hornwort populations for studies on stomatal differentiation (Pressel et al., 2014) indicate that there is little or no variation between clones. The three mosses were selected to include: (1) species with sufficient numbers of stomata per sporophyte to allow a change of 10–15 % or more in number to be readily detected (Physcomitrella, with only 14 stomata, is unsuitable); (2) Polytrichum, the sister group to all other stomata-producing mosses; (3) stomata with apertures of different sizes and shapes, including free-floating apertures (i.e. the single guard cells in Funaria; Fig. 1); and (4) contrasting stomatal configurations: superficial versus sunken and whether or not they are covered by a calyptra (Table 1). Vouchers of all the specimens are housed in the Natural History Museum, London.
Wild-collected plants were transferred into seed trays filled with inert acid-washed silica sand within controlled growth environment chambers (BDR16, Conviron, Canada) prior to sporophyte development. Plants were maintained under the following conditions throughout the development of the sporophyte: 50 µmol m−2s−1 irradiance (representing half light-saturating conditions for non-vascular plants; Nobel, 1999; Fletcher et al., 2006); relative humidity 70 %; day:night temperatures 15 °C:12 °C; and day length 12 h. Atmospheric CO2 concentrations within the growth chambers were monitored using CARBOCAP GMP343 CO2 sensors (Vaisala, UK) and maintained by gaseous CO2 addition. Experimental plants were grown at either 440 p.p.m. [CO2] (F. hygrometrica, n = 30; M. hornum, n = 50; P. juniperinum, n = 50; P. laevis, n = 95; A. punctatus, n = 30) or 1500 p.p.m. [CO2] (F. hygrometrica, n = 30; M. hornum, n = 49; P. juniperinum, n = 50; P. laevis, n = 50; A. punctatus, n = 30) and were rotated within cabinets regularly. All plants were misted daily with an artificial rainwater solution. Both cabinets and contents were alternated every 2 weeks to avoid pseudo-replication. The three mosses were harvested after their capsules had become fully expanded with late stage sporogenous cells to mature spores, whilst the hornworts were harvested after their sporophytes had reached at least 2 cm in length.
Stomatal measurements
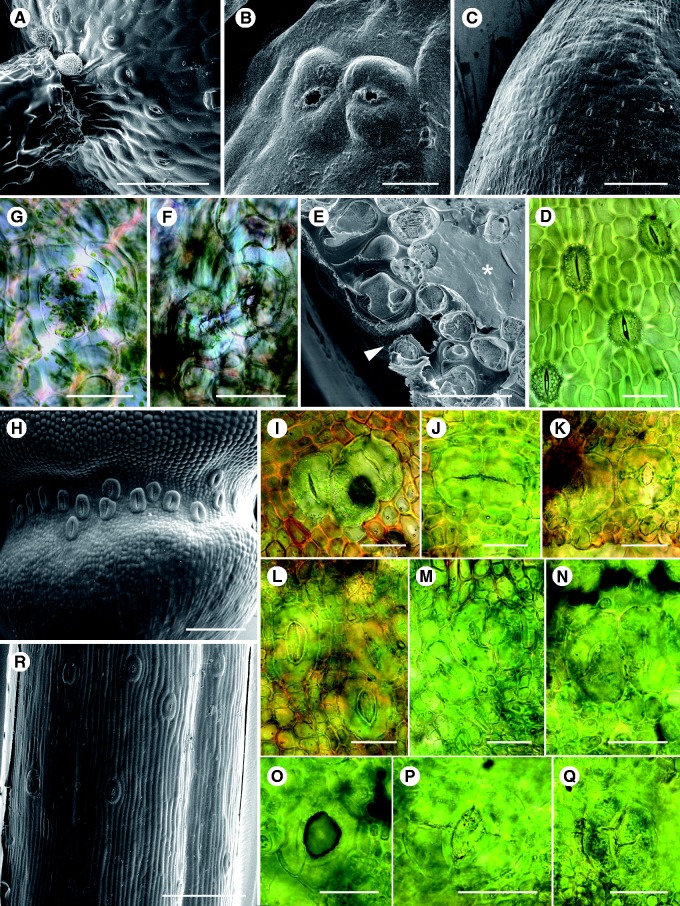
Sporophytes of all experimental plants, except those of Mnium, were cut longitudinally, mounted in water on slides with the external surfaces uppermost. In Mnium it is impossible to obtain accurate measurements from surface views as many of the deeply sunken stomata are obscured by the overarching epidermal cells; in this species the sporophytes were cut into quarters longitudinally, the spongy photosynthetic tissues scraped away and measurements taken from the inside view (Fig. 3F, G). Stomata were imaged with a Zeiss Axioscop 2 microscope equipped with an AxioCam MRc digital camera and numbers, aperture width and length and guard cell length were measured using the autocalibrated Axiovision Microscope Software. For mosses, every stoma/sporophyte was measured; for hornworts, every stoma from the first 1 cm of the sporophytes immediately above the top of the involucres was measured (Pressel et al., 2014), ensuring that all the stomata measured had developed well after the start of the elevated [CO2] treatment. Stomata of a selection of wild collected plants were also imaged by cryoscanning electron microscopy using the method of Duckett et al. (2009).
Fig. 3.
Light (D, F, G, I–Q) and cryoscanning electron (A–C, H, R) micrographs of moss and hornwort stomata. (A, B) Physcomitrella patens: 12–14 stomata slightly irregularly spaced (e.g. the paired stomata in B) and randomly orientated around the capsule base; pores are round and subsidiary cells absent. (C, D) In the closely related F. hygrometrica the numerous stomata are axially orientated and regularly spaced. Also note the radial arrangement of the epidermal cells around the long-pored stomata (D); compare with hornworts (R). (E–G) Mnium hornum stomata sunk in deep pits. Note the liquid-filled subtending intercellular spaces (*) in (E). Stomata are often irregularly spaced [see the paired stomata in (F)] and have small round pores (F, G). (H–Q) Polytrichum juniperinum (H–K, grown at 440 p.p.m. [CO2]; L–Q, grown at 1500 p.p.m. [CO2]). Note the predominately axially arranged long-pored stomata frequently occurring in multiple groups (H–K). Abnormalities occur on almost all sporophytes and these increase under elevated CO2, as does the size of some of the apertures (L–Q). (J) A pair of stomata with a shared pore. (M–P) Stomata with abnormal pores. (O) Stoma with massive aperture. (P) Stoma with four guard cells. (R) Sporophyte of the hornwort A. punctatus. Note the regularly spaced axial stomata lacking subsidiary cells. Scale bars: (C, H, R) = 200 µm; (A) = 100 µm; (D–G, I–Q) = 50 µm; (B) = 20 µm.
Statistics
Effects of plant species and [CO2]a on stomatal abundance and aperture were tested using two-way ANOVA with post hoc Tukey testing where indicated. Data were checked for normality and homogeneity of variance prior to ANOVA. Student’s t-tests were performed where indicated in the text. All statistics were carried out using Minitab v 12.21 (Minitab Inc., USA).
RESULTS
Stomatal abundance
We observed no differences in stomatal abundance on sporophytes of the mosses M. hornum and P. juniperinum or in the hornworts P. laevis and A. punctatus when grown under 440 or 1500 p.p.m. [CO2] (Fig. 2A). There was a small increase in stomatal abundance of F. hygrometrica sporophytes that underwent development at 1500 p.p.m. [CO2] compared with those at ambient [CO2] [two-sample t (27 d.f.) = −4·17, P = 0·0003] (Fig. 2A).
Fig. 2.
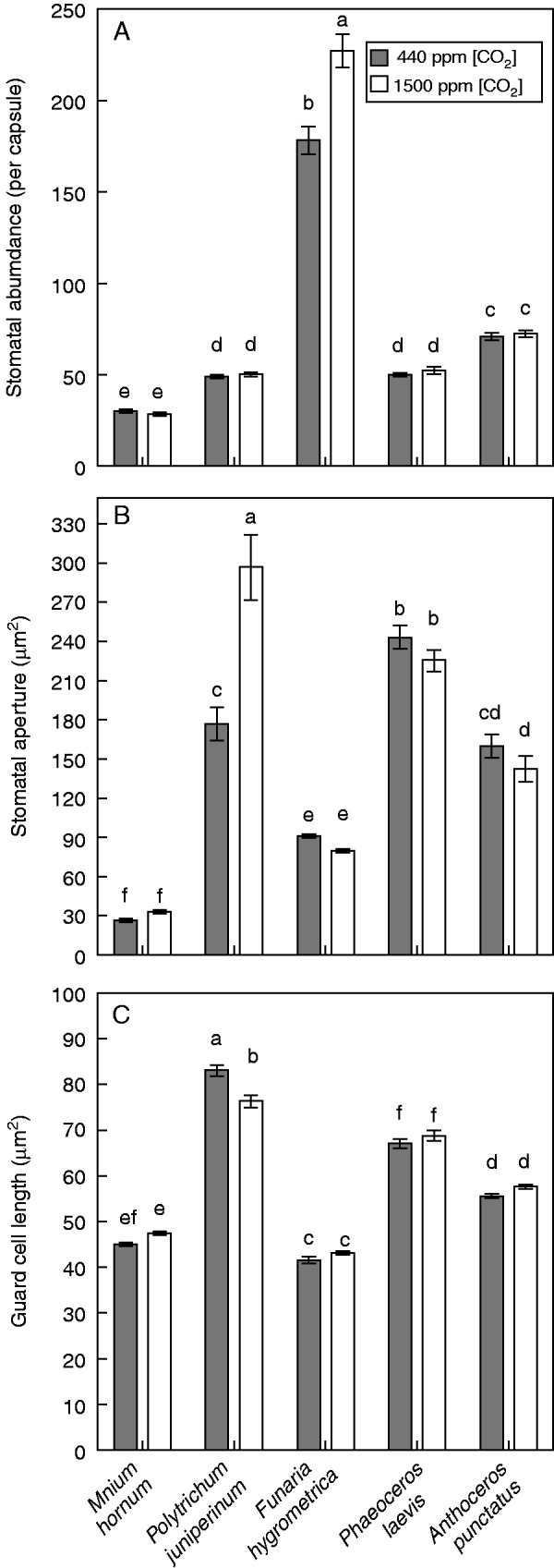
(A) Stomatal abundance on sporophyte generations of plants grown under 440 p.p.m. [CO2] (grey bars) and a replicated Palaeozoic [CO2] of 1500 p.p.m. (white bars). Error bars show ±1 s.e.; different letters denote statistical significance where P < 0·05 (ANOVA, post hoc Tukey test), n = 50 (M. hornum), 50 (P. juniperinum), 15 (F. hygrometrica), 50 (P. laevis), 30 (A. punctatus). (B) Mean aperture of stomatal pores on five individual sporophytes of each non-vascular species studied. Sporophytes had all undergone complete development at 440 p.p.m. [CO2] (grey bars) or 1500 p.p.m. [CO2] (white bars). Error bars show ±1 s.e., n = 5. Different letters indicate where P < 0·05 (ANOVA, post hoc Tukey test). (C) Mean length of guard cells measured on five individual sporophytes of each species of bryophyte.
Stomatal aperture
The stomatal aperture of P. juniperinum was significantly larger in sporophytes that underwent development at 1500 p.p.m. [CO2] compared with those that matured under 440 p.p.m. [CO2] [two-sample t (72 d.f.) = −4·31, P = 0·0001] (Fig. 2B). There were no differences in stomatal aperture of any of the other moss or hornwort species examined (Fig. 2B).
Guard cell length
There were no significant differences in guard cell length between [CO2] levels in the mosses M. hornum and F. hygrometrica or the hornworts P. laevis and A. punctatus (Fig. 2C). Guard cell length was reduced in in P. juniperinum sporophytes that underwent development at 1500 p.p.m. [CO2].
DISCUSSION
Our results unequivocally demonstrate that stomata on the sporophytes of several extant species of mosses and hornworts are non-responsive to changes in [CO2] in terms of stomatal numbers, guard cell length and stomatal aperture dimensions (Fig. 2, Table 2).
Table 2.
Summary of differences and interactions (F ratio from ANOVA) in stomatal abundance and aperture on sporophytes of moss and hornwort species grown under 440 and 1500 p.p.m. [CO2] (Fig. 2)
| Plant species | CO2 treatment | Species × CO2 | |
|---|---|---|---|
| Stomatal abundance on sporophyte | 198·06*** | 0·67 | 2·44 |
| Stomatal aperture (µm) | 70·19*** | 3·20 | 0·15 |
ANOVA has 1279 d.f.
For stomatal abundance at ambient and elevated [CO2], respectively: M. hornum, n = 50 and 49; P. juniperinum, n = 50 and 50; P. laevis, n = 95 and 50; A. punctatus, n = 30 and 30.
For stomatal aperture, n = 5.
***P < 0·001, post hoc Tukey test.
The small numerical increase in stomatal abundance of Funaria sporophytes that underwent development at 1500 p.p.m. [CO2] (Fig. 2A) is contrary to the reduction in stomatal abundance that would be expected if bryophyte stomata were analogous to angiosperms in their [CO2] response. The increase in stomatal abundance in Funaria is opposite to the small decrease reported previously in Leptobryum (Baars and Edwards, 2008), although in this moss the lower stomatal abundance was elicited by a 10-fold increase in ambient [CO2] and was due to an increase in capsule length, the overall number of stomata per capsule remaining unaltered (Baars and Edwards, 2008). The significantly larger apertures in P. juniperinum sporophytes grown under elevated [CO2] (Fig. 2B) are also contrary to expectation and, together with the misaligned and abnormal stomata recorded in this moss under 1500 p.p.m. [CO2] (Fig. 3L–Q), are almost certainly the result of slightly altered sporophyte development, as also seen in Leptobryum (Baars and Edwards, 2008). Whereas under 440 p.p.m. [CO2] individual sporophytes of P. juniperinum usually have from one to three stomata with three or four guard cells (Fig. 3H–K), under 1500 p.p.m. [CO2] we found many more occurring as groups and up to 25 % with abnormal guard cells. The teratologies included misplaced, extra and incomplete walls dividing the guard cells (Fig. 3L–Q). These malformations recall the asymmetrical stomata along the dehiscence grooves in hornworts, attributed to differential cell expansion (Pressel et al., 2014). Indeed, CO2-induced changes in epidermal cell growth patterns, perhaps as a consequence of increased carbon assimilation, are the simplest explanation for the stomatal abnormalities observed in Polytrichum under elevated [CO2].
Our results are in line with those of some previous studies on lycophytes and ferns showing that these basal groups of vascular plants lack aperture closure responses to [CO2] (Brodribb and McAdam, 2013). These findings and the recent demonstration that angiosperms are the only group of land plants that utilize calcium-based signalling pathways led Brodribb and McAdam (2011) to argue for an incremental acquisition of stomatal regulatory processes. This is contrary to the perhaps more widely held view, based on a large body of physiological and molecular evidence, that these are evolutionarily ancient and that physiologically active stomatal control evolved before the divergence of the bryophytes (Brodribb and McAdam, 2011; Chater et al., 2011; Ruszala et al., 2011). Another possible scenario for the evolution of stomatal functionality is neofunctionalization following whole-genome replication, given that ABA is also associated with desiccation tolerance in mosses (Bopp and Werner, 1993; Mayaba et al., 2001; Stark et al., 2007).
Even more problematic for the notion of stomatal functional continuity across land plants is the lack of stomata in the basal moss lineages (Fig. 1). These are absent in Takakiopsida and Andreaeopsida (here, as in liverworts, dehiscence is by the splitting of lidless capsules) and the paired cells adorning the capsules in Sphagnum are now regarded as pseudostomata since they are enclosed by the calyptra until maturation of the sporophytes and lack both open pores and subjacent intercellular spaces (Duckett et al., 2009). Their primary role appears to be facilitation of capsule desiccation leading to spore discharge rather than regulation of gaseous exchange.
Whereas in vascular plants stomatal densities and numbers make sense in terms of their regulatory role, the same is not true of mosses, where numbers (and absences) differ enormously even between closely related genera with very similar ecologies (Table 1, Fig. 3A–D, H). Equally perplexing is the absence of any relationship in bryophytes between stomatal dimensions and genome sizes. Thus, hornworts have some of the largest stomata and the smallest genome sizes. Those of M. hornum are larger than those of Plagiomnium cuspidatum despite a smaller genome size, and the same is true when comparison is made between Funaria and Physcomitrella (Voglmayr, 2000; Renzaglia et al., 1995; Rensing et al., 2008) and between the two Polytrichum species (Table 1). Similarly, there are wide disparities between closely related taxa in pore shapes, the presence or absence of subsidiary cells and stomatal orientation (Table 1, Fig. 3A–H).
A further question mark over an active regulatory role and a significant contribution of CO2 ingress through the pores to sporophyte nutrition in mosses is that they open only when the sporophytes have almost reached their full dimensions, i.e. the bulk of their carbon must have been acquired either from the parent gametophytes via the placenta (Ligrone et al., 1993) or directly through the epidermal cells. Added to this are further complications: (1) the stomata in many mosses are either covered by the calyptra until sporophyte maturation or are tightly enveloped by perichaetial leaves (e.g. Physcomitrella); (2) unlike the exponential water loss from drying out gametophytes, moss sporophytes lose water very slowly whether or not they possess stomata; (3) though Chater et al. (2011) state that the stomata in Physcomitrella and Funaria close in response to various stimuli, their data actually show only small changes in aperture dimensions, unlike the complete closure seen in vascular plants. That stomatal responsiveness to environmental cues in these two mosses is restricted to the developmental stage when green capsules are expanding (Chater et al., 2011) is in line with a recent study showing that, in Funaria, guard cell walls are thin and flexible soon after pore formation and that a decrease in pectin content coupled with changes in wall architecture during development renders mature stomata immobile (Merced and Renzaglia, 2014). However, the discovery that the intercellular spaces in moss and hornwort sporophytes, unlike those in vascular plants, are liquid-filled until well after the stomata open (Pressel et al., 2014) (see also Fig. 3E) casts serious doubt on any role of young stomata in active regulation of gaseous exchange. It should also be underlined that there has never been an unequivocal demonstration of reversible aperture changes in peristomate mosses, and the possible presence of potassium fluxes between the guard cells and their neighbours has yet to be investigated. Taking all these factors into account, sporophyte desiccation rather than gas regulation seems the more likely primary role for moss stomata. Indeed, their location in most mosses around the base of the capsule seems more fitted for removal of water ascending the setae than provision of CO2.
Many of these arguments are equally applicable to hornworts, following their wide acceptance as the sister group to vascular plants (Qiu et al., 2006, 2007; Liu et al., 2014). Any case for stomatal structural and functional continuity needs to explain the plastid-determined division of the guard mother cells, inelastic guard cell walls and initially liquid-filled sporophytic intercellular spaces in hornworts (Pressel et al., 2014). It should also be underlined that a new configuration of the land plant tree based on phylotranscriptomics (Wickett et al., 2014) places hornworts at the base of the land plant tree, although this placement is currently under debate. Implicit in this new phylogeny, very different from liverworts as sister to all other land plants (Chang and Graham, 2011; Qiu et al., 1998; Gao et al., 2010) and liverworts, mosses and hornworts as successive sister groups to vascular plants (Qiu et al., 1998, 2006, 2007; Liu et al., 2014), is the loss of stomata in liverworts and their reacquisition in mosses.
Given these major issues about developmental, functional and evolutionary continuity between bryophyte and vascular plant stomata, our failure to detect any responses to elevated [CO2] is not surprising. This pattern of stomatal non-reponsiveness to [CO2] is likely to extend into other early non-vascular and vascular land plant lineages. Our findings lend further support to the hypothesis that active stomatal regulation and [CO2] responsiveness occurred later in the evolution of land plants (Brodribb and McAdam, 2011). Consequently, our data prompt considerable caution in using stomatal densities as proxies for past paleo-atmospheric CO2 concentrations where extant counterparts are not available.
ACKNOWLEDGEMENTS
We thank Dr Kate Allinson for technical assistance. We also thank the editor and three anonymous reviewers for their helpful comments on this manuscript. This work was supported by a Royal Society University Research Fellowship to D.D.C. and a Leverhulme Emeritus Fellowship to J.G.D.
LITERATURE CITED
- Baars C, Edwards D. 2008. Effects of elevated atmospheric CO2 on spore capsules of the moss Leptobryum pyriforme. Journal of Bryology 30: 36–40. [Google Scholar]
- Bainard JD, Villarreal JC. 2013. Genome size increases in recently diverged hornwort clades. Genome 56: 431–435. [DOI] [PubMed] [Google Scholar]
- Beerling DJ, Royer DL. 2002. Reading a CO2 signal from fossil stomata. New Phytologist 153: 387–397. [DOI] [PubMed] [Google Scholar]
- Beerling DJ, Woodward FI. 1997. Changes in land plant function over the Phanerozoic: reconstructions based on the fossil record. Botanical Journal of the Linnean Society 124: 137–153. [Google Scholar]
- Beerling DJ, Birks HH, Woodward FI. 1995. Rapid late-glacial atmospheric CO2 changes reconstructed from the stomatal density record of fossil leaves. Journal of Quaternary Science 10: 379–384. [Google Scholar]
- Beerling DJ, McElwain JC, Osborne CP. 1998. Stomatal responses of the ‘living fossil’ Ginkgo biloba L. to changes in atmospheric CO2 concentrations. Journal of Experimental Botany 49: 1603–1607. [Google Scholar]
- Beerling DJ, Osborne CP, Chaloner WG. 2001. Evolution of leaf-form in land plants linked to atmospheric CO2 decline in the Late Palaeozoic era. Nature 410: 352–354. [DOI] [PubMed] [Google Scholar]
- Berner RA. 1998. The carbon cycle and CO2 over Phanerozoic time: the role of land plants. Philosophical Transactions of the Royal Society of London Series B: Biological Sciences 353: 75–81. [Google Scholar]
- Beaulieu JM, Leitch IJ, Patel S, Pendharkar A, Knight CA. 2008. Genome size is a strong predictor of cell size and stomatal density in angiosperms. New Phytologist 179: 975–986. [DOI] [PubMed] [Google Scholar]
- Berner RA. 2006. GEOCARBSULF: a combined model for Phanerozoic atmospheric O2 and CO2. Gemochimica et Cosmochimica Acta 70: 5653–5664. [Google Scholar]
- Bopp M, Werner O. 1993. Abscisic acid and desiccation tolerance in mosses. Botanica Acta 106: 103–106. [Google Scholar]
- Brodribb TJ, McAdam SA. 2011. Passive origins of stomatal control in vascular plants. Science 331: 582–585. [DOI] [PubMed] [Google Scholar]
- Brodribb TJ, McAdam SA. 2013. Unique responsiveness of angiosperm stomata to elevated CO2 explained by calcium signalling. PLoS ONE 8: e82057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodribb TJ, McAdam SA, Jordan GJ, Feild TS. 2009. Evolution of stomatal responsiveness to CO2 and optimization of water-use efficiency among land plants. New Phytologist 183: 839–847. [DOI] [PubMed] [Google Scholar]
- Chang Y, Graham S. 2011. Inferring the higher-order phylogeny of mosses (Bryophyta) and relatives using a large, multigene plastid data set. American Journal of Botany 98: 839–849. [DOI] [PubMed] [Google Scholar]
- Chater C, Kamisugi Y, Movahedi M, et al. 2011. Regulatory mechanism controlling stomatal behavior conserved across 400 million years of land plant evolution. Current Biology 21: 1025–1029. [DOI] [PubMed] [Google Scholar]
- Chater C, Oliver J, Casson SA, Gray JE. 2014. Putting the brakes on: abscisic acid as a central environmental regulator of stomatal development. New Phytologist 202: 376–391. [DOI] [PubMed] [Google Scholar]
- Darwin F. 1898. Observations on stomata. Proceedings of the Royal Society of London Series B 63: 413–417. [Google Scholar]
- Duckett JG, Pressel S, P’ng KM, Renzaglia KS. 2009. Exploding a myth: the capsule dehiscence mechanism and the function of pseudostomata in Sphagnum. New Phytologist 183: 1053–1063. [DOI] [PubMed] [Google Scholar]
- Edwards D, Kerp H, Hass H. 1998. Stomata in early land plants: an anatomical and ecophysiological approach. Journal of Experimental Botany 49: 255–278. [Google Scholar]
- Fletcher BJ, Brentnall SJ, Quick WP, Beerling DJ. 2006. BRYOCARB: a process-based model of thallose liverwort carbon isotope fractionation in response to CO2, O2, light and temperature. Geochimica et Cosmochimica Acta 70: 5676–5691. [Google Scholar]
- Franks PJ, Beerling DJ. 2009. Maximum leaf conductance driven by CO2 effects on stomatal size and density over geologic time. Proceedings of the National Academy of Sciences of the USA 106: 10343–10347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haig D. 2013. Filial mistletoes: the functional morphology of moss sporophytes. Annals of Botany 111: 337–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartung W. 2010. The evolution of abscisic acid (ABA) and ABA function in lower plants, fungi and lichen. Functional Plant Biology 37: 806–812. [Google Scholar]
- Hartung W, Weiler E, Volk O. 1987. Immunochemical evidence that abscisic acid is produced by several species of Anthocerotae and Marchantiales. Bryologist 90: 393–400. [Google Scholar]
- Haworth M, Fitzgerald A, McElwain JC. 2011. Cycads show no stomatal-density and index response to elevated carbon dioxide and subambient oxygen. Australian Journal of Botany 59: 630–639. [Google Scholar]
- Gao L, SU YJ, Wang T. 2010. Plastid genome sequencing, comparative genomics, and phylogenomics: current status and prospects. Journal of Systematics and Evolution 48: 77–93. [Google Scholar]
- Lake JA, Quick WP, Beerling DJ, Woodward FI. 2001. Plant development: signals from mature to new leaves. Nature 411: 154. [DOI] [PubMed] [Google Scholar]
- Lake JA, Woodward FI, Quick WP. 2002. Long-distance CO2 signalling in plants. Journal of Experimental Botany 53: 183–193. [DOI] [PubMed] [Google Scholar]
- Ligrone R, Duckett JG, Renzaglia KS. 1993. The gametophyte-sporophyte junction in land plants. Advances in Botanical Research 19: 231–318. [Google Scholar]
- Liu Y, Cox CJ, Wang W, Goffinet B. 2014. Mitochondrial phylogenomics of early land plants: mitigating the effects of saturation, compositional heterogeneity, and codon-usage bias. Systematic Biology 63: 862–878. [DOI] [PubMed] [Google Scholar]
- Lucas JR, Renzaglia KS. 2002. Structure and function of hornwort stomata. Microscopy and Microanalysis 8: 1090–1091. [Google Scholar]
- Mayaba N, Beckett RP, Csintalan Z, Tuba Z. 2001. ABA increases the desiccation tolerance of photosynthesis in the Afromontane understorey moss Atrichum androgynum . Annals of Botany 8: 1093–1100. [Google Scholar]
- Merced A, Renzaglia K. 2014. Developmental changes in guard cell wall structure and pectin composition in the moss Funaria: implications for function and evolution of stomata. Annals of Botany 114: 1001–1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobel PS. 1999. Physicochemical and environmental plant physiology. London: Academic Press. [Google Scholar]
- Pressel S, Goral T, Duckett JG. 2014. Stomatal differentiation and abnormal stomata in hornworts. Journal of Bryology 36: 87–103. [Google Scholar]
- Qiu YL, Cho Y, Cox JC., Palmer JD. 1998. The gain of three mitochondrial introns identifies liverworts as the earliest land plants. Nature 394: 671–674. [DOI] [PubMed] [Google Scholar]
- Qiu YL, Li L, Wang B, et al. 2006. The deepest divergences in land plants inferred from phylogenomic evidence. Proceedings of the National Academy of Sciences of the USA 103: 15511–15516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu YL, Li L, Wang B, et al. 2007. A nonflowering land plant phylogeny inferred from nucleotide sequences of seven chloroplast, mitochondrial, and nuclear genes. International Journal of Plant Sciences 168: 691–708. [Google Scholar]
- Rensing SA, Lang D, Zimmer AD, et al. 2008. The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319: 64–69. [DOI] [PubMed] [Google Scholar]
- Renzaglia KS, Rasch EM, Pike LM. 1995. Estimates of nuclear DNA content in bryophyte sperm cells: phylogenetic considerations. American Journal of Botany 82: 18–25. [Google Scholar]
- Ruszala EM, Beerling DJ, Franks PJ., et al. 2011. Land plants acquired active stomatal control early in their evolutionary history . Current Biology 21: 1030–1035. [DOI] [PubMed] [Google Scholar]
- Stark LR, Oliver MJ, Mishler BD, McLetchie DN. 2007. Generational differences in response to desiccation stress in the desert moss Tortula inermis. Annals of Botany 99: 53–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temsch EM, Greilhuber J, Krisai R. 1998. Genome size in Sphagnum (peat moss). Botanica Acta 111: 325–330. [Google Scholar]
- Voglmayr H. 2000. Nuclear DNA amounts in mosses (Musci). Annals of Botany 85: 531–546. [Google Scholar]
- Wickett NJ, Mirarab S, Nguyen N, et al. 2014. Phylotranscriptomic analysis of the origin and early diversification of land plants. Proceedings of the National Academy of Sciences of the USA 111: E4859–E4868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward F, Bazzaz F. 1988. The responses of stomatal density to CO2 partial pressure. Journal of Experimental Botany 39: 1771–1781. [Google Scholar]
- Woodward FI. 1987. Stomatal numbers are sensitive to increases in CO2 from pre-industrial levels. Nature 327: 617–618. [Google Scholar]