Abstract
As aboriginal sources of medications, medicinal plants are used from the ancient times. Andrographis paniculata is one of the highly used potential medicinal plants in the world. This plant is traditionally used for the treatment of common cold, diarrhoea, fever due to several infective cause, jaundice, as a health tonic for the liver and cardiovascular health, and as an antioxidant. It is also used to improve sexual dysfunctions and serve as a contraceptive. All parts of this plant are used to extract the active phytochemicals, but the compositions of phytoconstituents widely differ from one part to another and with place, season, and time of harvest. Our extensive data mining of the phytoconstituents revealed more than 55 ent-labdane diterpenoids, 30 flavonoids, 8 quinic acids, 4 xanthones, and 5 rare noriridoids. In this review, we selected only those compounds that pharmacology has already reported. Finally we focused on around 46 compounds for further discussion. We also discussed ethnobotany of this plant briefly. Recommendations addressing extraction process, tissue culture, and adventitious rooting techniques and propagation under abiotic stress conditions for improvement of phytoconstituents are discussed concisely in this paper. Further study areas on pharmacology are also proposed where needed.
1. Introduction
Medicinal plant is an integral part of human life to combat the sufferings from the dawn of civilization [1]. It is estimated that more than 80,000 of total plant species have been identified and used as medicinal plants around the world [2]. Among these plants, more than 1300 plant species have been used traditionally in Malaysia where the knowledge is being passed down from generation to generation [3]. The indigenous medicinal plants and plant-derived drugs are the potential source of alternative medicine and are extensively used to treat various health ailments [4]. Use of the medicinal plants is a core component at primary health care level due to availability, acceptability, compatibility, and affordability. Dependency on these medicinal plants varies from country to country. It is estimated that about 75–80% of people of developing countries and about 25% of people of developed countries depend either directly or indirectly on medicinal plants for the first line of treatment [3, 5]. Therefore, people are encouraging indigenous production and processing of these medicinal plants to use in different cultures and religion for the treatment of various diseases. Moreover, importance and uses of medicinal plants are also stated in different religious books (i.e., the Holy Qur'an, the Bible). About 19 medicinal plants and 176 medicinal plants are mentioned in the Holy Qur'an [6] and the Holy Bible [7], respectively.
Andrographis paniculata (Burm. f.) Wall. ex Nees (AP) is an important medicinal plant and widely used around the world. It belongs to the family Acanthaceae. AP is used as a traditional herbal medicine in Bangladesh, China, Hong Kong, India, Pakistan, Philippines, Malaysia, Indonesia, and Thailand [8, 9] and is ethnobotanically used for the treatment of snake bite, bug bite, diabetes, dysentery, fever, and malaria [3]. In the Unani and Ayurvedic medicines, AP is one of the mostly used medicinal plants [8]. In recent times, commercial preparations of this plant extracts are also used in certain countries. However, the preparations yet need to be standardized for their better efficacy. The aerial part of AP is most commonly used; its extracts contain diterpenoids, diterpene glycosides, lactones, flavonoids, and flavonoid glycosides. Whole plant leaves and roots are also used as a folklore remedy for different diseases in Asia and Europe [8, 10]. AP has been reported to have a broad range of pharmacological effects including anticancer [11–18], antidiarrheal [19, 20], antihepatitis [21, 22], anti-HIV [23], antihyperglycemic [24–27], anti-inflammatory [28–32], antimicrobial, antimalarial [33, 34], antioxidant [35–37], cardiovascular [38, 39], cytotoxic [23], hepatoprotective [40–52], immunostimulatory [53–57], and sexual dysfunctions [58].
Since the AP is used for the treatment of many diseases in traditional medicinal systems, its intended benefits need to be evaluated critically. Therefore, this paper reviews the ethnobotany, some agronomic techniques, isolation and characterization of phytoconstituents, and pharmacological properties of AP. Additionally, chemical properties, biological functions, and possible mode of actions of phytoconstituents are also entertained. The literature searches were conducted in worldwide accepted scientific database PubMed (http://www.ncbi.nlm.nih.gov/pubmed), ScienceDirect (http://www.sciencedirect.com/), Scopus (http://www.scopus.com/), Web of Science (http://webofknowledge.com/), Springer Link (http://link.springer.com/), Wiley Online Library (http://onlinelibrary.wiley.com/), and advance search in Google scholar (http://scholar.google.com.my/), as well as recognized books, abstract, and some nonimpact, and nonindexed journals. The search strings used were “Andrographis paniculata” or “King of Bitters” or “Kalmegh” or “Hempedu bumi” or “Andrographolide.” Further relevant papers were searched in the aforementioned databases from the list of references of all available papers. The authors went through more than 300 full papers and a total of 243 peer-reviewed papers focused on plant description, agronomic techniques, traditional uses, bioactive compound extraction, phytoconstituents, and pharmacology properties (such as common cold, anti-inflammatory, antihyperglycemic, hepatoprotective, antibacterial, antiviral, antiparasitic, anticancer, immunomodulatory, cardiovascular, antihyperlipidemic, sexual dysfunctions, contraceptive, safety, and toxicity effects) were selected for this review. We briefly discussed recent scientific findings and proposed some fields where further study is needed.
2. Botany
2.1. Origin and Distribution
A. paniculata is native to Taiwan, Mainland China, and India. It is also commonly found in the tropical and subtropical Asia, Southeast Asia, and some other countries including Cambodia, Caribbean islands, Indonesia, Laos, Malaysia, Myanmar, Sri Lanka, Thailand, and Vietnam [59–61]. This plant is also found in different phytogeographical and edaphic zones of China, America, West Indies, and Christmas Island [61].
2.2. Plant Description
A. paniculata is an important medicinal plant of Andrographis genus. A total number of species of this genus varied in different reports, which comprises either 19 [64, 65], 28 [59, 66], 40 [62, 67], or 44 [68] species. The exact numbers of species of Andrographis genus are not validated yet. Total number of chromosomes of AP is 25 and 50 in gametophytic [69] and sporophytic [70] count, respectively. In addition, genotypic differences are important considerations to find out high yielding germplasms.
A. paniculata is an annual, branched, erect, and herbaceous plant which grows in hedgerows throughout the plane lands, hill slopes, waste ground, farms, moist habitat, seashores, and roadsides. It also can be cultivated in garden. Moist shady places, forests, and wastelands are preferable for their well development [59, 71]. The morphological and physiological data of AP are presented in Table 1 and Figure 1. This plant grows abundantly in Southern and Southeastern Asia including India, Java, Sri Lanka, Pakistan, and Indonesia, while it is cultivated in India, China, Thailand, Brunei, Indonesia, the West Indies such as Jamaica, Barbados, and Bahamas, Hong Kong, and the tropical areas in America and also in southwestern Nigeria [10, 71].
Table 1.
| Traits | Values/characteristics |
|---|---|
| Plant height | 30–110 cm |
| Stem | Dark green |
| Length | 30–100 cm |
| Diameter | 2–6 mm |
| Shape | Quadrangular with longitudinal furrows and wings on the angles of the young parts, slightly enlarged at the nodes |
| Leaves | Glabrous |
| Length | 2–12 cm |
| Width | 1–3 cm |
| Arrangement | Lanceolate |
| Shape | Pinnate, acute apex, entire margin |
| Flowers | White with rose-purple spots on the petals |
| Size | Small, in lax spreading axillary and terminal racemes or panicles |
| Seed | Capsules linear-oblong, acute at both ends |
| Size | 1.9 cm × 0.3 cm |
| Color | Yellowish brown |
| Shape | Subquadrate, numerous |
| Flowering and fruiting | December to April |
Figure 1.
Morphology of Andrographis paniculata. (a) Mature A. paniculata in polybag stage, (b) flowering stage, (c) harvested seeds, (d) in vitro seedling, (e) young A. paniculata in polybag, (f) adventitious roots of A. paniculata, and (g) vegetative seedlings. Single direction of arrow indicates the developmental stages and both directions of arrow denote vegetative propagation of plant (Photographs are taken from M.S. Hossain's research work, except (b)).
2.3. Taxonomic Hierarchy
Taxonomic hierarchy is as follows:
-
Domain: Eukaryota,
-
Kingdom: Plantae,
-
Subkingdom: Tracheobionta,
-
Superdivision: Spermatophyta,
-
Division: Angiosperma,
-
Class: Dicotyledonae,
-
Subclass: Gamopetalae,
-
Series: Bicarpellatae,
-
Order: Personales,
-
Family: Acanthaceae,
-
Subfamily: Acanthoideae,
-
Tribe: Justiciae,
-
Subtribe: Andrographideae,
-
Genus: Andrographis,
2.4. Vernacular Names
Generally, the tree is known as “King of Bitters” for its extremely bitter taste. In Malaysia, AP is traditionally known as “hempedu bumi” (bile of the earth). This plant has different names in different languages. The vernacular names of AP are presented in Table 2. In addition, local people easily recognize plant species with vernacular names instead of binomial names.
Table 2.
| Language | Name |
|---|---|
| Arabic | Quasabhuva |
| Assamese | Chiorta, Kalmegh |
| Azerbaijani | Acılar Şahı, Acılar Xanı (khanı) |
| Bengali | Kalmegh |
| Burmese | Se-ga-gyi |
| Chinese | Chuan Xin Lian |
| English | The Creat, King of Bitters |
| French | Chirette verte, Roi des amers |
| Gujarati | Kariyatu |
| Hindi | Kirayat, Kalpanath, |
| Indonesian | Sambiroto, Sambiloto |
| Japanese | Senshinren |
| Kannada | Nelaberu |
| Konkani | Vhadlem Kiratyem |
| Lao | La-Sa-Bee |
| Malay | Hempedu Bumi, Sambiloto |
| Malayalam | Nelavepu, Kiriyattu |
| Manipuri | Vubati |
| Marathi | Oli-kiryata, Kalpa |
| Mizo | Hnakhapui |
| Oriya | Bhuinimba |
| Panjabi | Chooraita |
| Persian | Nain-e Havandi |
| Philippines | Aluy, Lekha and Sinta |
| Russian | Andrografis |
| Sanskrit | Kalmegha, Bhunimba and Yavatikta |
| Scandinavian | Green Chiratta |
| Sinhalese | Hīn Kohomba or Heen Kohomba |
| Spanish | Andrografis |
| Tamil | Nilavembu |
| Telugu | Nilavembu |
| Thai | Fa-Talai-Jorn, Fah-talai-jon (jone) |
| Turkish | Acılar Kralı, Acı Paşa, Acı Bey |
| Urdu | Kalmegh, Kariyat, Mahatita |
| Vietnamese | Xuyên Tâm Liên |
2.5. Agronomic Techniques
Plants conventionally grow via seed culture. Planting and harvesting time has influenced the yield of plant [153, 154]. Usually, May to July is the recommended time for sowing AP seeds with a spacing of 30 × 15 cm that resulted in a plant density of 222 thousands plants/ha and average biomass of 3 t/ha [155, 156]. However, seed dormancy is a major constrain in AP cultivation commercially [157, 158]. Although hormonal media and hot water treatment have been suggested to overcome this problem [159, 160], this technique is not enough to meet the commercial quantities required due to variability among the seed-derived progenies and scanty and delayed rooting of seedlings [161, 162]. Therefore, nonconventional propagation methods such as plant tissue culture techniques are alternative methods to produce plenty of plantlets within a short time and improve phytochemical contents in AP. Tissue culture techniques have been applied for large scale propagation of AP [162–164]. Dandin and Murthy [164] reported that in vitro regenerated AP contains higher amount of andrographolide compared to the mother plants. Optimum production of andrographolide can be achieved in a short span by employing the suspension cultures of AP [165]. Plant tissue culture has also been successfully used to form new flavones from callus culture [166–168].
3. Ethnobotanical Uses
Ethnobotanically, the leaves and roots of AP have been used since centuries in Asia and Europe to cure the wide spectrum of health ailments. However, the whole plant is also used for certain limited purposes. Due to its “cold property” activity, it is recommended to be used to get rid of the body heat in fevers and to dispel toxins from the body. The plants are also recommended for the use in cases of leprosy, gonorrhea, scabies, boils, skin eruptions, and chronic and seasonal fever for its high “blood purifying” properties [8, 169]. The overall traditional uses of AP in different traditional medicinal systems (TMS) or countries are pointed out in Table 3. In addition, it is also widely used for medicinal purposes by the traditional practitioners, tribes, or community as a folklore remedies in different countries [9].
Table 3.
The ethnobotanical uses of A. paniculata.
| Country/TMS* | Traditional uses | Reference |
|---|---|---|
| Ayurvedic | Fever, liver diseases, torpid liver, vitiligo | [62] |
|
| ||
| Japan | Fever, common cold | [10] |
|
| ||
| Malaysia | Diabetes, hypertension | [10, 77] |
|
| ||
| Scandinavian | Fever, common cold | [10] |
|
| ||
| Traditional Bangladeshi medicine | Acute diarrhea, anorexia, bloating with burning sensations in the chest, blood purifier, common cold, constipation, cough, debility, diabetes, dysentery, edema, emesis, fever, headache, helminthiasis, indigestion, intestinal worms, leucorrhea, liver disorders, loss of appetite, low sperm count, lower urinary tract infections, lung infections, malaria, mucus, pharyngotonsillitis, sexual and skin disorders, splenomegaly, uncomplicated sinusitis, vertigo | [9] |
|
| ||
| Traditional Chinese medicine | Inflammation, fever, burn, carbuncle, cervical erosion, chicken pox, common cold, cough with thick sputum, detoxicant, detumescent, diarrhea dispel toxins of the body, dysentery, eczema, epidemic encephalitis B, fever, hepatitis, herpes zoster, laryngitis, mumps, neonatal subcutaneous annular ulcer, neurodermatitis, pelvic inflammation, pharyngitis, pharyngolaryngitis, pneumonia, respiratory infections, snake bites, sores, suppurative otitis media, tonsillitis, vaginitis | [8, 10] |
|
| ||
| Traditional Indian medicine | Diabetes, dysentery, enteritis, helminthiasis, herpes, peptic ulcer, skin infections (topical use), snake-bites (topical use) | [10] |
|
| ||
| Traditional Thai medicine | Fever, common cold, noninfectious diarrhea | [10] |
|
| ||
| Unani system of medicine | Anthelmintic, anti-inflammatory, antipyretic, aperient, astringent, boils, carminative, chronic and seasonal fevers, convalescence after fevers, diuretic, dysentery, dyspepsia associated with gaseous distension, emmenagogue, emollient, gastric and liver tonic, general debility, gonorrhea, irregular bowel habits, leprosy, loss of appetite, relieve griping, scabies, skin eruptions | [8] |
*Traditional Medicinal Systems.
4. Phytoconstituents
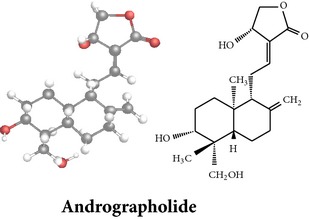
The aerial parts of AP have been described for its innumerous use in the extraction of phytoconstituents; however, leaves, stems, roots, and whole plants have also been reported for phytochemicals with pharmacological activities. The compositions of phytochemicals widely differ in terms of the part used, geography, season, and time of harvesting. M. Sharma and R. Sharma [76] reported that the highest amount of andrographolide, a major bioactive compound of AP, was found in the sample harvested after 110 days of cultivation followed by that just before flowering stage (130 days). The bioactive compounds were extracted with different types of solvents such as methanol (MeOH), ethanol (EtOH), hexane, acetone, acetone-water, chloroform (CHCl3), and dichloromethane from the whole plant, leaves, aerial parts, stems, and roots. Extraction procedure of bioactive compounds of AP from MeOH extracts, for example, is shown in Figure 2. In this extraction procedure, whole plant material of AP (11.5 kg) was shade dried, ground, extracted with methanol (10 L × 6) under reflux for 8 h, and filtered to give residue. After filtration, three residues of the extract were found: (i) CHCl3 residue (700 g), (ii) H2O soluble (1.5 kg), and (iii) H2O insoluble residue (150 g). The residues were then chromatographed to get specific fractions. These fractions were further chromatographed and followed several procedures to identify specific compound (Figure 2). A total of 32 bioactive compounds with seven ent-labdane diterpenoids, twelve flavonoids, and two quinic acid derivatives have been isolated and characterized by this procedure. Previous phytochemical studies of AP have reported more than 55 ent-labdane diterpenoids, 30 flavonoids, 8 quinic acids, 4 xanthones [132, 150], and 5 noriridoids, namely, andrographidoids A, B, C, D, and E [170, 171].
Figure 2.
Extraction of pure compounds of A. paniculata from MeOH extracts. Yield of pure compounds is in bracket in mg. Their structures have been elucidated mainly by 1D and 2D NMR and MS spectroscopic methods. SGCC: silica gel column chromatography; TLC: thin layer chromatography, MeOH: methanol; EtOAc: ethyl acetate, CHCl3: chloroform (adopted from [72]).
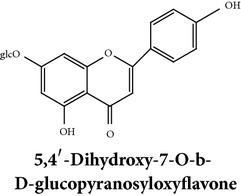
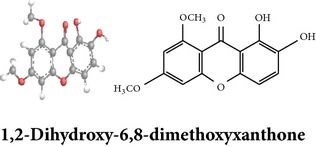
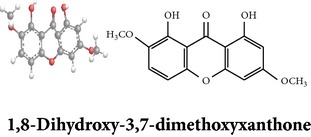
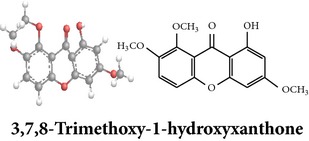
Zhang et al. [172] reported 3 new ent-labdane diterpenoids, namely, 19-norandrographolides A, B, and C from the ethanol extracts of the aerial parts of AP. Their structures have been established by HRESIMS and NMR spectral data in combination with X-ray crystallographic analysis; thus the 19-norandrographolide-A was identified as 3-dehydro-14-deoxy-19-norandrographolide. However, there is no report for pharmacological activity of these three compounds. The quinic acids were extracted only from the whole plants using the methanol solvents and they are functioning as an antiplatelet aggregator [72]. Recently, Arifullah et al. [141] demonstrated moderately potent antimicrobial and antioxidant activity of andrographolide and echiodinin extracted from acetone and methanol extracts of in vitro leaf callus of AP. Their structure elucidation was determined by electrospray ionization, MSD, NMR, and IR spectra. The yield of this compound was higher compared to the field plant. On the other hand, a novel flavonoid, 7, 8-dimethoxy-2′-hydroxy-5-O-β-d-glucopyranosyloxyflavone, together with 15 known flavonoids was isolated from the aerial parts of AP. The structure was elucidated on the basis of chemical and spectroscopic analysis. Significant antiproliferative activity of these flavonoids against human leukaemia HL-60 cell was also investigated [142]. Dua et al. [143] investigated four xanthones (1,2-dihydroxy-6,8-dimethoxyxanthone; 1,8-dihydroxy-3,7-dimethoxyxanthone; 3,7,8-trimethoxy-1-hydroxyxanthone; 4,8-dihydroxy-2,7-dimethoxyxanthone) from roots of AP using CHCl3 fraction and purity confirmed by HPLC. Xu et al. [171] isolated and established structure of 5 rare types of noriridoids with a known iridoid curvifloruside F from the ethanol extracts of roots. They also assayed antibacterial activity of these compounds, but none showed any inhibitory activity (MIC > 100 μg/mL). In this review, we provided available information on around 46 isolated phytoconstituents of AP with structure, chemical properties, IUPAC name, reported pharmacological activities, mode of extraction, and parts used (Table 4).
Table 4.
Preclinical pharmacology of isolated phytoconstituents of A. paniculata.
| "> | ||
|---|---|---|
| Compound 3D/2D, chemical properties/IUPAC name/part used/extracts/pharmacology | ||
|
| ||
| ent-labdane diterpenoids | ||
|
| ||
|
|
|
| Chemical properties: C20H30O5
Exact mass: 350.21; Mol. Wt.: 350.45; C, 68.54; H, 8.63; O, 22.83 IUPAC: (3E,4S)-3-[2-[(1R,4aS,5R,6R,8aS)-6-hydroxy-5-(hydroxymethyl)-5,8a-dimethyl-2-methylidene-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethylidene]-4-hydroxyoxolan-2-one Part used: L$,§,¢,, AeP∗,#,¤,œ,†,£,ð, WP£,ø, R¥ Extracts: M§,¤,¢,£,¥,ø,$,ð, E∗,#,$, Hœ, AW† Pharmacology: analgesic, antipyretic, antiulcerogenic [78], antiangiogenic [79], antiretroviral [33, 80], antithrombotic [81], anticancer [13, 16, 82–91], antidiabetic [25–27, 92–94], anti-inflammatory [95–100], anti-influenza [101, 102], antileishmaniasis [103], antiproliferative, proapoptotic [104, 105], antiurothelial [106], cardioprotective [107], cholestatic [108], hepatoprotective [40, 41], immunomodulatory [57, 90, 109–114], inhibition of Epstein Barr virus [115], protective activity against alcohol-induced hepatic, radiosensitiser [116], renal toxicity [117] |
Chemical properties: C26H40O8
Exact mass: 480.27; Mol. Wt.: 480.59; C, 64.98; H, 8.39; O, 26.63 IUPAC: 4-[2-[(4aS,5R,8aS)-5,8a-dimethyl-2-methylidene-5-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethyl]-2H-furan-5-one Part used: L$,§,¢,, WP£,ø, R¥, AeP∗,¤,¥,ð,† Exacts: M§,¤,¢,£,¥,ø,ð, E∗,$, AW† Pharmacology: antiherpes-simplex virus [33], anti-inflammatory [100, 118, 119], antioxidant [35], antiparasitic [34], chemosensitiser [120], hepatoprotective [9, 43] |
Chemical properties: C19H28O5
Exact mass: 336.19; Mol. Wt.: 336.42; C, 67.83; H, 8.39; O, 23.78 IUPAC: 3-(2-((1R,2R,4aR,5S,8aS)-decahydro-2-hydroxy-1-(hydroxymethyl)- 1,4a-dimethyl-6-oxonaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: L§,$,¢,‡, WP£,©,®,ø, AeP∗,¤,¥,#,ð,œ,† Extracts: M§,¤,¢,ø,©,ð, E∗,#,$, Hœ, AW† Pharmacology: antibacterial [121, 122], antifungal [123], hepatoprotective [124], immunomodulator [113], platelet activating factor antagonist [110], uterine smooth muscle relaxant [125], vasorelaxant, antihypertensive [126, 127] |
|
| ||
|
|
|
| Chemical properties: C20H28O4
Exact mass: 332.2; Mol. Wt.: 332.43; C, 72.26; H, 8.49; O, 19.25 IUPAC: 4-[(E)-2-[6-hydroxy-5-(hydroxymethyl)-5,8a-dimethyl-2-methylidene-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethenyl]-2H-furan-5-one Part used: L§,$,¢,‡, WP£,©,®,ø, AeP∗,¤,¥,#,ð,œ,† Extracts: M∗,#,§,¤,£,¥,®,ø,ð, E$, Hœ, DCM® Pharmacology: antidiabetic [128], antifungal [123], antioxidant, hepatoprotective [129], antiretroviral [80], antithrombotic [81], cholestatic [108], cytotoxic [130], vasorelaxant, antihypertensive [126, 127, 131], antiherpes [33], vasorelaxant, antihypertensive [126] |
Chemical properties: C19H28O5
Exact mass: 336.19; Mol. Wt.: 336.42; C, 67.83; H, 8.39; O, 23.78 IUPAC: 3-((2S,3aR,5aR,6R,7R,9aR,9bS)-dodecahydro-7-hydroxy-6-(hydroxymethyl)-3a,9a-dimethylnaphtho[2,1-b]furan-2-yl)furan-2(5H)-one Part used: L¢,, AeP#,¤,¥,†, WPø, R¥ Extracts: M¤,¢,¥,ø, E#, AW† Pharmacology: anti-inflammatory and anticancer [132], antiproliferative [133], cytotoxic [14], differentiation inducer [18], inhibits growth of Bacillus subtilis [134] |
Chemical properties: C27H42O10
Exact mass: 526.28; Mol. Wt.: 526.62; C, 61.58; H, 8.04; O, 30.38 IUPAC: NA Part used: L§,$,¢,‡, WP£,©,®,ø, AeP∗,¤,¥,#,ð,œ,† Extracts: M¤,£,‡,ð, E∗,#,$, AW†, PtE‡, CHL‡ Pharmacology: hepatoprotective [42] |
|
| ||
|
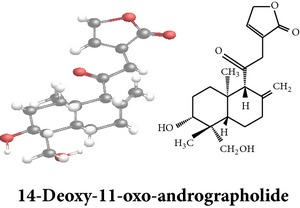
|
|
| Chemical properties: C20H30O3
Exact mass: 318.22; Mol. Wt.: 318.45; C, 75.43; H, 9.50; O, 15.07 IUPAC: 3-(2-((1R,4aS,5R)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylenenaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: L§,æ, AePβ,œ,† Extracts: Eβ,æ, Hœ Pharmacology: anti-inflammatory [135, 136] |
Chemical properties: C20H28O5
Exact mass: 348.19; Mol. Wt.: 348.43; C, 68.94; H, 8.10; O, 22.96 IUPAC: 3-(2-((1R,2R,4aR,5R,8aS)-decahydro-2-hydroxy-1-(hydroxymethyl)-1,4a-dimethyl-6-methylenenaphthalen-5-yl)-2-oxoethyl)furan-2(5H)-one Part used: L§,æ, AePβ,œ,† Extracts: M†, AW§ Pharmacology: antileishmaniasis [137] |
Chemical properties: C20H30O5
Exact mass: 350.21; Mol. Wt.: 350.45; C, 68.54; H, 8.63; O, 22.83 IUPAC: NA Part uses: AeP¤,†, WP£ Extracts: M¤,£, AW† Pharmacology: cell differentiation inducer [18] |
|
| ||
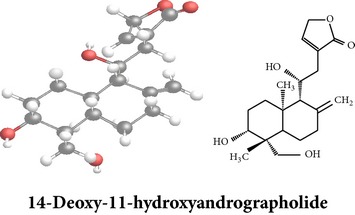
|
|
|
| Chemical properties: C21H30O5
Exact mass: 362.21; Mol. Wt.: 362.46; C, 69.59; H, 8.34; O, 22.07 IUPAC: 3-((E)-2-((1R,2R,4aR,5R,8aS)-decahydro- 2-hydroxy-1-(hydroxymethyl)-1,4a- dimethyl-6-methylenenaphthalen-5-yl)vinyl)-5-methoxyfuran-2(5H)-one Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C20H34O3
Exact mass: 322.25; Mol. Wt.: 322.48; C, 74.49; H, 10.63; O, 14.88 IUPAC: (Z)-2-(2-((1R,4aS,5R,8aS)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylenenaphthalen-5-yl)ethyl)but-2-ene-1,4-diol Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C26H40O9
Exact mass: 496.27; Mol. Wt.: 496.59; C, 62.88; H, 8.12; O, 29.00 IUPAC: 3-(2-((1R,2R,4aS,5R)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylene-2-(tetrahydro-3,4,5-trihydroxy-6-(hydroxymethyl)-2H-pyran-2-yloxy)naphthalene-5-yl)ethyl)furan-2(5H)-one Part used: AePþ, WP® Extracts: M®, Eþ Pharmacology: antibacterial [121, 122], antifungal [123] |
|
| ||
|
|
|
| Chemical properties: C20H30O5
Exact mass: 350.21, Mol. Wt.: 350.45, C, 68.54; H, 8.63; O, 22.83 IUPAC: NA Part used: AeP Extracts: M¤, AW† Pharmacology: antimicrobial [134] |
Chemical properties: C20H30O3
Exact mass: 318.22, Mol. Wt.: 318.45; C, 75.43; H, 9.50; O, 15.07 IUPAC: 3-(2-((1R,4aS,8aS)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylenenaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C28H44O8
Exact mass: 508.3, Mol. Wt.: 508.64; C, 66.12; H, 8.72; O, 25.16 IUPAC: 3-(2-(1-(((2R)-6-(ethoxymethyl)-tetrahydro-3,4,5-trihydroxy-2H-pyran-2-yloxy)methyl)-decahydro-1,4a-dimethyl- 6-methylenenaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: AeP Extracts: M¤ Pharmacology: cell differentiation inducer [18] |
|
| ||
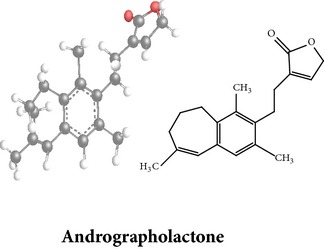
|
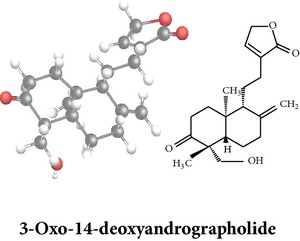
|
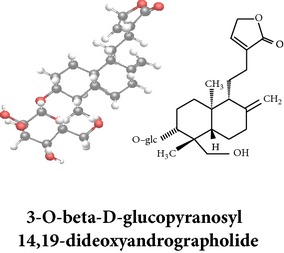
|
| Chemical properties: C20H24O2
Exact mass: 508.3; Mol. Wt.: 508.64; C, 66.12; H, 8.72; O, 25.16 IUPAC: 3-(2-(1-(((2R)-6-(ethoxymethyl)- tetrahydro-3,4,5-trihydroxy-2H-pyran-2-yloxy)methyl)-decahydro-1,4a-dimethyl-6-methylenenaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: AeP Extracts: E¬ Pharmacology: cytotoxic [139] |
Chemical properties: C20H28O4
Exact mass: 332.2; Mol. Wt.: 332.43; C, 72.26; H, 8.49; O, 19.25 IUPAC: 3-(2-((1R,4aS,5R,8aS)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylene-2-oxonaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C26H40O9
Exact mass: 496.27; Mol. Wt.: 496.59; C, 62.88; H, 8.12; O, 29.00 IUPAC: 3-(2-((1R,2R,4aS,5R,8aS)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylene-2-(tetrahydro-3,4,5-trihydroxy-6-(hydroxymethyl)-2H-pyran-2-yloxy)naphthalen-5-yl)ethyl)furan- 2(5H)-one Part used: AeP Extracts: AW† Pharmacology: antimicrobial [134] |
|
| ||
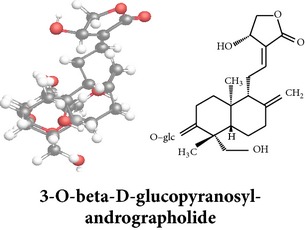
|
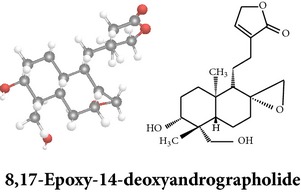
|
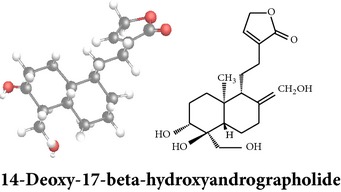
|
| Chemical properties: C26H40O10
Exact mass: 512.26; Mol. Wt.: 512.59; C, 60.92; H, 7.87; O, 31.21 IUPAC: (S,E)-dihydro-3-(2-((1R,2R,4aS,5R,8aS)-decahydro-1-(hydroxymethyl)-1,4a-dimethyl-6-methylene-2-(tetrahydro-3,4,5- trihydroxy-6-(hydroxymethyl)-2H-pyran-2-yloxy)naphthalen-5-yl)ethylidene)-4-hydroxyfuran-2(3H)-one Part used: AeP Extracts: AW† Pharmacology: antimicrobial [134] |
Chemical properties: C20H32O5
Exact mass: 352.22; Mol. Wt.: 352.47; C, 68.15; H, 9.15; O, 22.70 IUPAC: NA Part used: AeP Extracts: AW† Pharmacology: antimicrobial [134] |
Chemical properties: C20H32O5
Exact mass: 352.22; Mol. Wt.: 352.47; C, 68.15; H, 9.15; O, 22.70 IUPAC: 3-(2-((1R,2R,4aS,5R,8aS)-decahydro-2-hydroxy-1,6-bis(hydroxymethyl)-1,4a-dimethylnaphthalen-5-yl)ethyl)furan-2(5H)-one Part used: AeP Extracts: AW† Pharmacology: antimicrobial [134] |
|
| ||
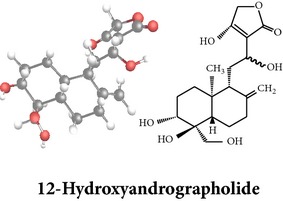
|
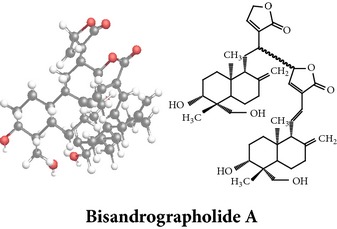
|
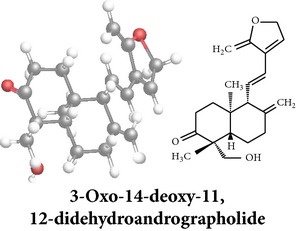
|
| Chemical properties: C19H28O7
Exact mass: 368.18; Mol. Wt.: 368.42; C, 61.94; H, 7.66; O, 30.40 IUPAC: NA Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C40H56O8
Exact mass: 664.4; Mol. Wt.: 664.87; C, 72.26; H, 8.49; O, 19.25 IUPAC: NA Part used: AeP Extracts: M¤ Pharmacology: cell differentiation inducer [18], transient receptor potential channel vanilloid-4 (TRPV-4), activator/analgesic, anti-inflammatory [140] |
Chemical properties: C21H28O3
Exact mass: 328.2; Mol. Wt.: 328.45; C, 76.79; H, 8.59; O, 14.61 IUPAC: (1R,4aR,5R,8aS)-octahydro-5-((1E)-2-(2,5-dihydro-2-methylenefuran-3-yl)vinyl)-1-(hydroxymethyl)-1,4a-dimethyl-6-methylenenaphthalen-2(1H)-one Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
|
| ||
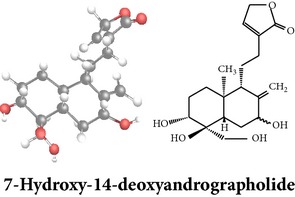
|
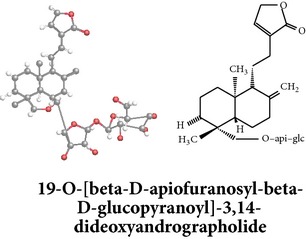
|
|
| Chemical properties: C19H28O6
Exact mass: 352.19; Mol. Wt.: 352.42; C, 64.75; H, 8.01; O, 27.24 IUPAC: 3-(2-((1S,2R,4aS,5S,8aS)-decahydro-1,2,7-trihydroxy-1-(hydroxymethyl)-4a-methyl-6-methylenenaphthalen-5-yl)ethyl)furan- 2(5H)-one Part used: AeP Extracts: E# Pharmacology: antiproliferative [138] |
Chemical properties: C31H48O12
Exact mass: 612.31; Mol. Wt.: 612.71; C, 60.77; H, 7.90; O, 31.34 IUPAC: NA Part used: AeP Extracts: AW† Pharmacology: antimicrobial [134] |
Echiodinin
Chemical properties: C16H12O5 Mol. Wt.: 285 IUPAC: NA Part used: in vitro Callus¶ Extracts: M¶, A¶ Pharmacology: antibacterial, antioxidant [141] |
|
| ||
| Flavonoids | ||
|
| ||
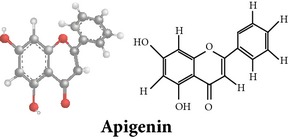
|
|
|
| Chemical properties: C15H10O4
Exact mass: 254.06; Mol. Wt.: 254.24; C, 70.86; H, 3.96; O, 25.17 IUPAC: 5,7-dihydroxy-2-phenyl-4H-chromen-4-one Part used: WPø Extracts: Mø Pharmacology: antiplatelet aggregator [72] |
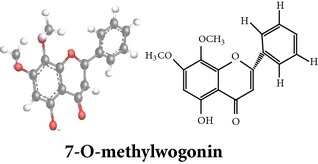
Chemical properties: C17H14O5
Exact mass: 298.08; Mol. Wt.: 298.29; C, 68.45; H, 4.73; O, 26.82 IUPAC: 5-hydroxy-7,8-dimethoxy-2-phenyl-4H- chromen-4-one Part used: WPø Extracts: Mø,£, H¥ Pharmacology: antiplatelet aggregator [72] |
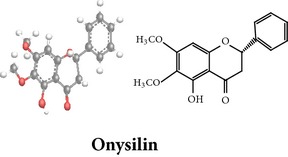
Chemical properties: C17H16O5
Exact mass: 300.1; Mol. Wt.: 300.31; C, 67.99; H, 5.37; O, 26.64 IUPAC: (S)-2,3-dihydro-5-hydroxy-6,7-dimethoxy-2-phenylchromen-4-one Part used: WPø Extracts: Mø Pharmacology: antiplatelet aggregator [72] |
|
| ||
|
|
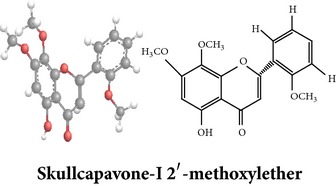
|
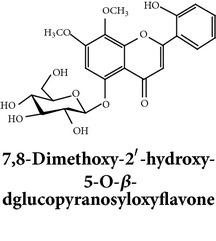
| Chemical properties: C23H25O11
Exact mass: 477.1391; Mol. Wt.: 477.1391 IUPAC: NA Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: C17H16O5
Exact mass: 300.1; Mol. Wt.: 300.31; C, 67.99; H, 5.37; O, 26.64 IUPAC: (S)-2,3-dihydro-5-hydroxy-7,8-dimethoxy-2-phenylchromen-4-one Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: C18H16O6
Exact mass: 328.09; Mol. Wt.: 328.32; C, 65.85; H, 4.91; O, 29.24 IUPAC: 5-hydroxy-7,8-dimethoxy-2-(2-methoxyphenyl)-4H- chromen-4-one Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
|
| ||
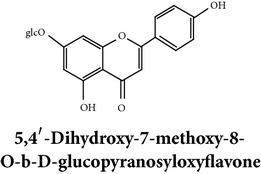
|
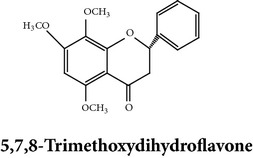
|
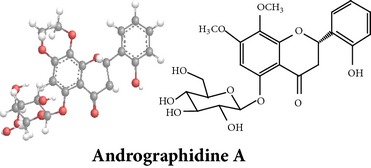
|
| Chemical properties: NA IUPAC: NA Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: NA IUPAC: NA Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: C23H26O10
Exact mass: 462.152597 Mol. Wt.: 462.44654 IUPAC: (2S)-7,8-dimethoxy-2-phenyl-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-2,3-dihydrochromen-4-one Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
|
| ||
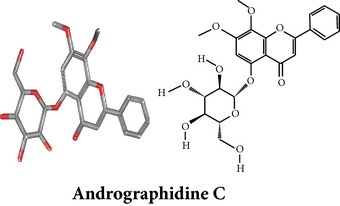
|
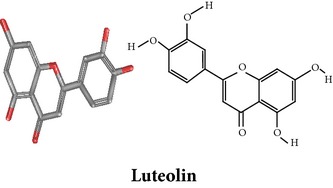
|
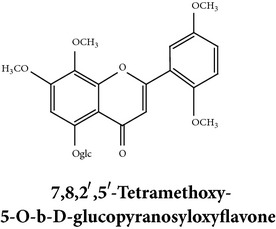
|
| Chemical properties: C23H24O10
Mol. Wt.: 460.43066, EM: 460.136947 IUPAC: 7,8-dimethoxy-2-phenyl-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: C15H10O6
Exact mass: 286.047738, Mol. Wt.: 286.2363, IUPAC: 2-(3,4-dihydroxyphenyl)-5,7-dihydroxychromen-4-one Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: — IUPAC: — Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
|
| ||
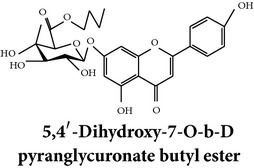
|
|
|
| Chemical properties: — IUPAC: — Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
Chemical properties: — IUPAC: — Part used: AeP~ Extracts: E~ Pharmacology: antiproliferative [142] |
|
|
| ||
| Xanthones | ||
|
| ||
|
|
|
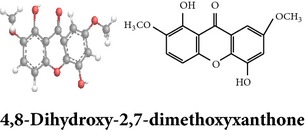
| Chemical properties: C15H12O6
Exact mass: 288.06; Mol. Wt.: 288.25; C, 62.50; H, 4.20; O, 33.30 IUPAC: 1,2-dihydroxy-6,8-dimethoxy-9H-xanthen-9-one Part used: R Extracts: sequential extraction with PtE, M, CHCl3 and water Pharmacology: antimalarial [143] |
Chemical properties: C15H12O6
Exact mass: 288.06; Mol. Wt.: 288.25; C, 62.50; H, 4.20; O, 33.30 IUPAC: 1,8-dihydroxy-2,6-dimethoxy-9H-xanthen-9-one Part used: R Extracts: sequential extraction with PtE, M, CHCl3 and water Pharmacology: antimalarial [143] |
Chemical properties: C16H14O6
Exact mass: 302.08; Mol. Wt.: 302.28; C, 63.57; H, 4.67; O, 31.76 IUPAC: 8-hydroxy-1,2,6-trimethoxy-9H-xanthen-9-one Part used: R Extracts: sequential extraction with PtE, M, CHCl3 and water Pharmacology: antimalarial [143] |
|
| ||
| Xanthones | Quinic acids | |
|
| ||
|
|
|
| Chemical properties: C15H12O6
Exact Mass: 288.06; Mol. Wt.: 288.25; C, 62.50; H, 4.20; O, 33.30 IUPAC: 1,5-dihydroxy-2,7-dimethoxy-9H-xanthen-9-one Part used: R Extracts: sequential extraction with PtE, M, CHCl3 and water Pharmacology: antimalarial [143] |
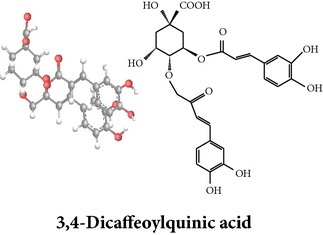
Chemical properties: C26H26O12
Exact mass: 530.14; Mol. Wt.: 530.48; C, 58.87; H, 4.94; O, 36.19 IUPAC: (1S,3R,4R,5R)-3-((E)-3-(3,4-dihydroxyphenyl)acryloyloxy)-4-((E)-4-(3,4-dihydroxyphenyl)-2-oxobut-3-enyloxy)-1,5-dihydroxycyclohexanecarboxylic acid Part used: WP Extracts: Mø Pharmacology: antiplatelet aggregator [72] |
|
L: leaves, AeP: aerial parts, WP: whole plants, R: roots, M: methanol, E: ethanol, H: hexane, A: acetone, AW: acetone water, PtE: petroleum ether, CHCl3: chloroform, DCM: dichloromethane, Mol. Wt.: molecular weight.
References: Arifullah¶ et al. 2013 [141]; Chen* et al. 2006 [144]; Chen# et al. 2008 [138]; Chen~ et al. 2014 [142]; Fujita§ et al. 1984 [145]; Jiβ et al. 2005 [135]; Liuæ et al. 2008 [136]; Matsuda¤ et al. 1994 [18]; Pramanick¢ et al. 2006 [146]; Rao£ et al. 2004 [147]; Reddy¥ et al. 2003 [148]; Reddyœ et al. 2005 [80]; Seth‡ et al. 2010 [149]; Shen† et al. 2006 [134]; Sule© et al. 2011 [121]; Sule® et al. 2012 [123]; Wang¬ et al. 2009 [139]; Wuø et al. 2008 [72]; Xu$ et al. 2010 [150]; Zhouþ et al. 2008 [151]; Zouð et al. 2010 [152].
5. Potential Pharmacology
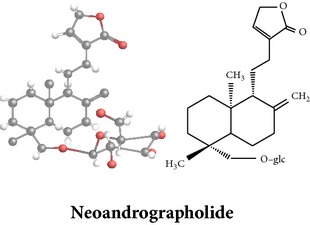
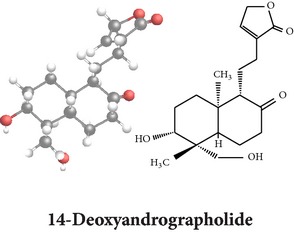
Extensive use of AP in traditional medicinal system has proven its efficacy over the past three decades. Several researches including in vitro, in vivo (animal), and clinical (human) studies have confirmed various pharmacological activities of AP extracts and products. A wide range of pharmacological activities such as antiplatelet aggregation activity [72], immunomodulatory activity [57, 109–113, 139], and other myriad health benefits [10, 75, 132] were observed among these researches. Andrographolide, a major ent-labdane diterpenoid of AP, is the largest contributor of many pharmacological activities. Other ent-labdane diterpenoids (such as neoandrographolide and 14-deoxyandrographolide), flavonoids, quinic acids, and xanthones are also reported for their significant contributions. A few of the reported works on pharmacology are summarized below.
5.1. Effect on Common Cold
A. paniculata is commonly used for the prevention and treatment of common cold in several communities. A double-blind, placebo-controlled study of 61 adult patients suffering from common cold used Kan Jang tablets (made from A. paniculata dried extract) for 5 days. Within the treatment period, significant clinical improvement was observed on day 4 for 1200 mg extract daily. Both groups showed significant reductions in clinical symptoms like shivering, sore throat, tiredness, muscular ache, rhinitis, sinus pains, and headache [173]. A placebo-controlled study was conducted in 1997 using Kan Jang tablets on 107 healthy students in a rural school as a dose of 2 tablets (200 mg) per day for 3 months to evaluate its efficacy to prevent common cold. The common cold was successfully prevented by Kan Jang tablets with 2.1-fold higher prevention rate compared to the placebo group [174]. Similar successful result was also demonstrated in another study including common cold with sinusitis in another place [175].
5.2. Anti-Inflammatory Effect
The anti-inflammatory activity of AP and its bioactive compounds (such as andrographolide and neoandrographolide) has been reported individually by many investigators [28–32, 95–100, 118, 119, 176]. AP extracts and andrographolide showed several anti-inflammatory activities, such as inhibition of intercellular adhesion molecule-1 expression in monocytes activated by tumor necrosis factor-α [177], suppression of inducible nitric oxide synthetase (iNOS) in RAW264.7 [29], cyclooxygenase-2 (COX-2) expression in neutrophils and microglial cells [95, 96], reduction of ERK1/2 phosphorylation in murine T cells [90] and IFN-γ, and IL-2 production [57, 90], and inhibition of TNF-α and GM-CSF induced by LPS [176]. In a study conducted by Hidalgo et al. [96], andrographolide was analyzed for the activation of NF-κB induced by platelet-activating factor (PAF) and N-formylmethionyl-leucyl-phenylalanine (fMLP) in HL-60 cells differentiated into neutrophils. NF-κB controls or contributes to the transcription of more than 200 genes that are involved in a variety of physiological and pathophysiological processes, particularly in immunity and inflammation. It is a critical regulator of cell differentiation, proliferation, and apoptosis and plays pivotal roles in the normal organ development and tumorigenesis [178]. However, the NF-κB can be activated via the signal transduction pathways which are multiple and complex. It was a surprising achievement that andrographolide significantly inhibited the binding of NF-κB to DNA, which is the final step of NF-κB activation and it also decreased the expression of cyclooxygenase-2 (COX-2) by inhibiting the activation of NF-κB in endothelial cells stimulated by PAF [179] and in microglial cells induced by lipopolysaccharide (LPS) [95]. In addition, the kidney inflammation can also be recovered using andrographolide due to its ability to inhibit the NF-κB activated by LPS in kidney [180].
Oral administration of andrographolide at a dose of 300 mg/kg daily had shown significant analgesic activity on acetic-induced writhing in mice and on the Randall-Selitto test in rats, but there was no effect on the hot plate test in mice [78]. Oral administration of andrographolide at 30, 100, and 300 mg/kg also showed anti-inflammatory activity in different models in rats [181]. Iruretagoyena et al. [57] suggested that treatment with andrographolide at a daily dose equal to 4 mg/kg significantly reduced an inflammatory demyelinating disease of the central nervous system, autoimmune encephalitis by inhibiting T cells in mice. The balancing of proinflammatory and anti-inflammatory cytokines is the result of anti-inflammatory performance of andrographolide and regulation of Th1, Th2, and Th17 transcription factors. Andrographolide also increased GATA3 mRNA expression but decreased T-bet and RORγt mRNA expressions [182].
Besides the AP and andrographolide, andrographolide derivatives (e.g., CHP1002 and andrographolide sulfonate) also exhibited anti-inflammatory properties [182, 183]. CHP1002, a novel synthetic derivative of andrographolide, significantly inhibited LPS-induced iNOS, COX-2 expressions, iNOS derived nitric oxide (NO), and COX-2 derived prostaglandin E2 (PGE2) production through the stimulation of heme oxygenase-1 (HO-1) expression in RAW264.7 macrophages. CHP1002 also significantly faded LPS-stimulated TNF-α, IL-1β, and IL-6 production [182]. Liu et al. [183] demonstrated that intraperitoneal administration of andrographolide sulfonate, a water soluble andrographolide (trade name: Xi-Yan-Ping injection), attenuated the severity of 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) induced colitis in mice. A dose of 1.25–5 mg/kg of andrographolide sulfonate significantly recovered the loss of body weights and diarrhea in colitis mice, prevented inflammatory damages of colons and proinflammatory cytokines such as IFN-γ, IL-17A, and TNF-α, and suppressed the functions of Th1 and Th17 which is a feasible strategy to control inflammatory bowel disease. The demonstrated results suggested that andrographolide sulfonate could be a strong therapeutic compound for the treatment of gastrointestinal inflammatory disorders.
5.3. Antihyperglycemic Effect
Inhibitions of α-glycosidase and α-amylase activity and stimulation of insulin sensitivity are considered as effective strategies to lower the level of postprandial blood glucose. These enzymes involved in digestion and absorption of carbohydrates resulting in postprandial increase of blood glucose [184]. Insulin resistance is mainly expressed by hyperinsulinemia and high blood glucose level and is associated with some metabolic hormonal abnormalities, such as dyslipidemia, abnormal uric acid metabolism, increased ovarian testosterone secretion, endothelial dysfunction, elevated procoagulant factors, and elevated inflammatory markers [185]. AP extracts and andrographolide effectively showed antihyperglycemic effect by (a) lowering blood glucose level through inhibition of α-glycosidase and α-amylase [26, 27, 111, 186]; (b) increasing insulin sensitivity and thus stimulating glucose uptake and oxidation by peripheral tissues [94]; (c) controlling abnormal lipid metabolism; (d) scavenging free radicals from circulation which disrupt the plasma membrane integrity resulting in decreased number of efficient plasma membrane receptors or transporter proteins necessary to uptake glucose from the blood stream [187]. Blood glucose lowering effect of AP was observed in both insulin-lacking diabetic rats and normal rats in several studies [25, 186, 188, 189].
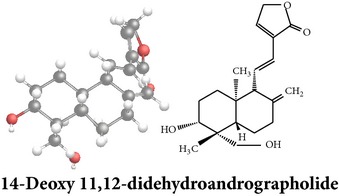
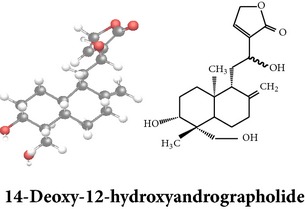
Andrographolide at a dose of 50 mg/kg effectively decreased blood glucose level, stimulated GLUT4 translocation [189], and improved diabetic rat's islet and beta cell functions [190]. Glucose induced hyperglycemia (orally administered) has been prevented by water extracts of AP in nondiabetic rats without affecting epinephrine-induced hyperglycemia [77]. Oral administration of ethanol extracts of AP significantly lowered the fasting blood glucose of human [26, 27, 93, 94]. Another bioactive compound, namely 14-deoxy-11,12-didehydroandrographolide, also showed the antihyperglycemic activity [128].
Besides controlling blood glucose level, andrographolide also effectively prevented the onset of insulitis in a dose dependent manner and thus delayed the onset and suppressed the development of diabetes in 30-week-old NOD mice. Andrographolide also regulates the Th1/Th2/Th17 homeostasis through which it may prevent β-cell death and inhibit T-cell infiltration into pancreatic islets and thereby prevent development of type 1 diabetes [191]. Recently, Augustine et al. [187] reported that AP decreases the blood glucose by increasing glucose utilization and oxidation, restoration of insulin signaling molecules in liver, and decreasing the serum lipid levels in high fat and sucrose induced type 2 diabetic rats without showing hypoglycemic effect. A combination of n-hexane insoluble fraction of AP (HIFA) with curcuminoids fraction of Curcuma xanthorrhiza rhizome (CFC) also significantly showed the antihyperglycemic effect on high-fructose-fat-fed rats [192]. The combination of HIFA-CFC could be a potential source to develop an antidiabetic agent. Therefore, the identification of more antihyperglycemic compounds of AP and combination of AP with other medicinal plants would be a focusing point of researchers for the better treatment option of the diabetic patients.
5.4. Hepatoprotective Effect
A. paniculata is widely used traditionally as a hepatoprotective agent and a stimulating agent for multiple enzymes of the liver. It is also used as an ingredient in the polyherbal preparations for the treatment of hepatic disorders in Ayurvedic and Unani medicine [8]. Along with different extracts of AP, andrographolide, neoandrographolide, 14-dexoyandrographolide, and 14-deoxy-11,12-didehydroandrographolide compounds are also reported to have hepatoprotective effect [40–42, 124, 129]. In a comparative study, the leaf extract and andrographolide was tested against the carbon tetrachloride- (CCl4-) induced hepatic microsomal lipid peroxidation. Only the leaf extract completely protected the high concentration CCl4-induced microsomal lipid peroxidation in vitro but not the andrographolide, which indicated that the hepatoprotective role is not solely due to the presence of andrographolide [48]. Similar effect of crude alcohol extracts of the AP leaves against CCl4-induced liver damage was also reported by Rana and Avadhoot [50]. Handa and Sharma [40] reported that andrographolide, methanol extract of whole plant, and andrographolide-free methanol extract improved liver histology in rats by 48.6%, 32%, and 15%, respectively, after CCl4-induced liver injury. Verma et al. [193] reported the effect of ethanol extract of AP on restoration of different enzyme after CCl4-induced liver injury. Further research using specific bioactive compounds is demanding for the better understanding of the hepatoprotective role played by the AP.
5.5. Antimicrobial and Antiparasitic Effect
5.5.1. Antibacterial Effect
Modern research has investigated the causes of extensive uses of AP in traditional healing systems as an antimicrobial agent to treat a variety of health morbidities of infectious origin. Leelarasamee et al. [194] reported that crude powder suspended in water to be devoid of in vitro antibacterial activity against Salmonella, Shigella, Escherichia coli, gram A Streptococci, and Staphylococcus aureus, even at a concentration of 25 mg/mL crude powder. However, over the last 3 decades, researchers reported that different types of extracts of A. paniculata possess potent antibacterial activity against various pathogenic and nonpathogenic bacteria. Nakanishi et al. [195] reported antibacterial activity of aqueous methanol (50% v/v) crude extracts of whole plant against Bacillus subtilis and Proteus vulgaris. Although Nakanishi et al. [195] reported the negative result against E. coli, ethanol extracts of aerial parts of AP were found to be effective in inhibiting E. coli growth along with other ten gram positive and gram negative bacteria species in an investigation conducted by Mishra et al. [196]. The aqueous extract showed significant antibacterial activity due to the combined effect of the isolated andrographolides and arabinogalactan proteins [197]. Similar effect was also mentioned by Fbricant and Farnsworth [198].
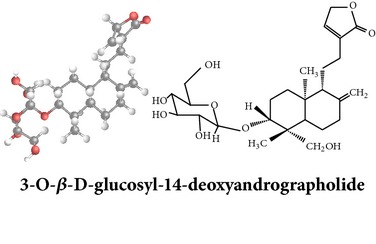
The antibacterial activity of three different extracts (dichloromethane, methanol, and aqueous) of AP whole plant was evaluated by Sule et al. [199] against 12 skin infection causing pathogenic bacterial strains. The extracts showed significant effects against all the tested bacterial strains in different concentrations likely 1000, 500, and 250 μg/disc. However, methanol extract showed the highest antibacterial activity against Enterococcus faecalis at 1000 μg/disc with an inhibition zone of 24 mm, and dichloromethane extract showed the least activity against Neisseria meningitis at 250 μg/disc with an inhibition zone of 6 mm. Similar results again were reported in another manuscript by Sule et al. [200] in the following year. In their study, they investigated antibacterial activity of methanol extract of whole plant against five human pathogenic bacteria S. aureus, Streptococcus pyogenes, Micrococcus luteus, Proteus mirabilis, and P. aeruginosa. Their results revealed that the highest inhibition (19.67 ± 0.76 mm) was exerted against S. aureus at 1000 μg/mL and the least (07.00 ± 1.50 mm) activity shown against P. aeruginosae at 250 μg/mL. However, it was a noteworthy result that the methanol extract exhibited more potent inhibitory activity against S. aureus (19.67 ± 0.76 mm) and Streptococcus pyogenes (16.00 ± 0.58 mm) at 1000 μg/mL compared to the antibiotic vancomycin (17.00 ± 1.05 mm and 14.50 ± 1.00 mm, resp.) [121]. Furthermore, they isolated and characterized two important antibacterial bioactive compounds, namely, 14-deoxyandrographolide (Figure 2) and 3-O-β-D-glucosyl-14-deoxyandrographolide, from the whole plant of AP and concluded that the mentioned efficacy of methanol extract had been shown due to the presence of these compounds (Table 4).
5.5.2. Antiviral Effect
Researchers investigated significant antiviral activity of AP besides other pharmacological activities in last two decades. Although they reported antiviral activity against limited viruses, such as dengue virus serotype 1 (DENV-1) [201], human papilloma virus type 16 (HPV16) [202], herpes simplex virus type 1 (HSV-1) [203], influenza A virus [102], and HIV [53, 55, 80, 204], their findings were very encouraging and noteworthy considering the life threatening role of these viruses in human community. The hot aqueous aerial parts extract of AP was reported for its significant antiviral activity to reduce the percentage of HIV antigen-positive H9 cells [55]. Recently, Tang et al. [201] reported that the methanol extract of AP possesses significant inhibition activity against DENV-1 in vitro assay. Another study has revealed that andrographolide suppressed HPV16 transcription activity, leading to the reduction of E6 oncoprotein and restored p53 [202].
Several bioactive compounds such as andrographolide, neoandrographolide, dehydroandrographolide, natural derivatives of andrographolide, namely, 14-deoxy-11,12-didehydroandrographolide and 14-deoxyandrographolide, and synthetic derivatives, namely, dehydroandrographolide succinic acid monoester (DAMS), 14-ά-lipoyl andrographolide (AL-1), 14-acetyl-3,9-isopropyl-ideneandrographolide, 14-acetylandrographolide, 3,14,19-triacetylandrographolide, and 3,9-isopropyl-ideneandrographolide have been shown to have significant antiviral activity against HIV, influenza A, and HSV-1 without any significant cytotoxic effect at virucidal concentrations. Andrographolide, isolated from ethanol extracts of whole plant of AP, showed a great promise in the treatment of HIV infections. It might be able to inhibit viral replication by interfering CDK (cyclin dependent kinase) activity, resulting in deregulation of HIV induced cell cycle [53, 80, 205]. The overall findings of andrographolide effects against different viruses indicate that andrographolide would be an effective agent for prevention and treatment of viral diseases.
5.5.3. Antiparasitic Effect
Antiparasitic activity of the AP extract is reported in certain articles. Dua et al. [143] investigated both in vitro and in vivo antimalarial activity of four xanthones (Table 4) isolated from roots against Plasmodium falciparum and Plasmodium berghei. One of the xanthones, 1,2-dihydroxy-6,8-dimethoxy-xanthone, showed substantial antiplasmodial activity during in vitro (4 μg/mL at IC50 value) and in vivo (62% parasitaemia reduction at 30 mg/Kg dose) study.
The water extract of dried leaves was found to be active against adult worms of Brugia malayi in vitro [206]. Recently, Padma et al. [207] evaluated the aqueous and methanol extracts for in vitro anthelmintic activity against adult earth worms Pheretima posthuma. The extracts showed significant results at the concentrations of 25 mg/mL, 50 mg/mL, and 75 mg/mL. However, the clinical relevancies of the antiparasitic studies are inconclusive due to obtaining the results at high concentration that may not be feasible clinically.
5.6. Anticancer Effect
Andrographolide exhibited both direct and indirect effects on cancer cells by inhibiting proliferation of cancer cells, cell-cycle arrests, or cell differentiation, enhancing body's own immune system against cancer cells; and inducting apoptosis and necrosis of cancer cells [208]. Dichloromethane fraction of methanol extract significantly inhibited the proliferation of HT-29 colon cancer cells. The major bioactive compound of AP, andrographolide, isolated from dichloromethane inhibited the growth of a diverse cancer cell representing different types of human cancers [11]. In contrast, recently Aditya et al. [209] reported that methanol extract of AP was found to be very less effective against both MCF-7 breast and HT-29 colon cancer cell lines. This low activity exhibited might be due to the low penetration power of the active principles.
Antiproliferative activities of andrographolide and isoandrographolide along with other 16 ent-labdane diterpenoids isolated from 85% ethanol extract of AP against human leukaemia HL-60 cells have also been investigated by Chen et al. [138]. These results showed that andrographolide and isoandrographolide were more effective than others. In a recent study, Chen et al. [142] identified a new flavonoid, 7, 8-dimethoxy-2′-hydroxy-5-O-β-d-glucopyranosyloxyflavone, isolated from the aerial parts of AP. This flavonoid exhibited potent antiproliferative activity against human leukaemia HL-60 cells with IC50 of 3.50 μM. Ethanol (70%) extracts and andrographolide were also found to be effective to increase the life spans of thymoma injected mice cells in an in vivo study [83]. In the following year, Geethangili et al. [210] showed the effective cytotoxic activity of ethanol extracts against human cancer cells including Jurkat (lymphocytic), PC-3 (prostate), HepG2 (hepatoma), and colon 205 (colonic) cancer cells. In another study, use of andrographolide at a dose 12 μg/mL for 36 h against HL-60 cells improved 27% in G0/G1 phase cells and significantly decreased cells number at S and G2/M phase [12]. Shi et al. [82] reported that andrographolide can inhibit human colorectal carcinoma (CRC) Lovo cell growth by G1–S phase arrest and induce the expression of cell-cycle inhibitory proteins p53, p21, and p16. These proteins repressed the activity of cyclin D1/Cdk4 and/or cyclin A/Cdk2, required for G1 to S phase transition. In a recent in vitro study, andrographolide has been shown to suppress the growth and invasion of CRC Lovo cells and trigger apoptosis. Besides the effect of andrographolide alone, andrographolide in combination with chemotherapeutics, cisplatin, is likely to represent a potential therapeutic strategy for CRC [211].
Andrographolide and its analogues exert direct inhibitory effect on cancer cells by inducing expression of cell cycle inhibitory proteins and depressing cyclin-dependent kinase (Cdk) resulting in blocking the cell cycle progression at G0/G1 [12, 13, 82, 212, 213]. Some other compounds also block the cell cycle progression at G2/M phase [210]. A novel semisynthetic analogue of andrographolide, DRF3188, exhibited anticancer activities against MCF 7 breast cancer cells at a lower dosage than andrographolide through similar mechanism [212]. Both the compounds block cell cycle at the G0-G1 phase through induction of the cell cycle inhibitor (p27) and concomitant decrease in the levels of Cdk4. Therefore, attention has been focused on the anticancer properties of pure components of AP and the molecular target of andrographolide that blocks G1 stage still needs to be determined.
5.7. Immunomodulatory Effect
Control of immune response by regulating nuclear factor of activated T cells (NFAT), a transcription factor essential for cytokine production during T-cell activation, is a widely known strategy. Preventing translocation of NFAT to nucleus is the target of several immunosuppressive agents (e.g., cyclosporin A, FK506) [112]. AP is known to exert several immunomodulatory properties. More than two decades ago, a laboratory test demonstrated that AP inhibited growth of human breast cancer cells similar to the drug tamoxifen [17]. Amroyan et al. [214] reported that andrographolide was effective to stop the clumping of blood platelets that lead to heart attacks and they also suggested that andrographolide has a major effect on activating the general defense functions of immune system by stimulating the production of antibodies as well as nonspecific immune responses such as increased macrophage phagocytosis. An in vitro study with the increased proliferation of lymphocytes and production of interleukin-2 (IL-2) confirmed the immunostimulatory activity of AP [12]. Three diterpene compounds of AP isolated from dichloromethane fraction of methanol extract showed augmented proliferation and IL-2 induction in human peripheral blood lymphocytes (HPBLs) at a low concentration [11]. In addition, the chronic consumption of the aqueous extract of AP also promoted the immune functions at 250 mg/kg and 500 mg/kg; however, 1000 mg/kg dose leading to development of autoimmune reactions, anaemia and multiple myeloma [215]. AP extract and andrographolide significantly promoted the lyses of natural killer (NK) cell-mediated target cells on day 5 after tumor induction. Antibody dependent cell mediated cytotoxicity (ADCC) and antibody dependent complement-mediated cytotoxicity (ACC) in metastatic tumor bearing animals were also enhanced significantly compared to the control by the treatment of AP extract and andrographolide. In addition, the levels of proinflammatory cytokines such as IL-1β, IL-6, GM-CSF, and TNF-α were also effectively reduced [216]. Carretta et al. [112] demonstrated that andrographolide reduces IL-2 production, extracellular signal regulated kinase- (ERK-) 1 and ERK-5 phosphorylation induced by anti-CD3 or phorbol myristate acetate and ionomycin (PMA/Ionomycin), and NF-κB activity in Jurkat cells and this effect can be related to a reduction in NFAT activity and an increase in c-jun-N-terminal kinase (JNK) phosphorylation. Moreover, andrographolide inhibited tumor growth in animals by stimulating the production of cytotoxic T lymphocytes [83]. Hence, the compounds modulate the host immune systems against these cells to confer the direct cytotoxicity to cancer cells. Based on the reported immunomodulatory properties, AP and andrographolide might be effective clinically to treat the autoimmune diseases.
5.8. Cardiovascular Effect
Cardiovascular diseases (CVDs) are the leading cause of death throughout the world. AP is used widely for improving the cardiac health in traditional medicinal systems. Several studies have investigated its activities in cardiovascular diseases [38, 81, 107, 131, 217–225]. Wang et al. [222] reported that AP is potential to increase the nitric oxide, cyclic guanosine monophosphate, and superoxide dismutase activity with declines of lipid peroxide and endothelin in an atherosclerotic rabbit model. In another study, Wang and Zhao [223] also investigated that the AP extracts can be able to prevent constriction of blood vessels and increase blood clotting time significantly in the pre- and postangioplasty procedures. The extracts inhibited the cell growth and DNA synthesis in a dose dependent manner, which is a similar mechanism like stents that prevent cell division.
Aqueous extracts and active constituents of AP showed significant antihypertensive activity in both spontaneously hypertensive rats and normotensive Wister-Kyoto rats [226], improved the blood pressure status in both pre- and post experimental myocardial infarction in animals [217, 218], and exhibited platelet antiaggregation in in vitro [81, 214] and ex vivo [227] assays. The existing reports suggested that AP can be used as alternative source of the treatment of CVDs. Further studies are necessary to know the insight of the mechanism of actions of specific constituents of AP and in clinical perspectives.
5.9. Antihyperlipidemic Effect
Hyperlipidemia is an important factor for atherosclerosis that leads to heart attack (obstruction occurs in the coronary arteries) and stroke (obstruction occurs in the arteries of the brain) [228–230]. Chen et al. [231] suggested the andrographolide as candidate therapeutic agent for atherosclerosis based on their research result. Recently, another study revealed antihyperlipidemic effects of andrographolide and neoandrographolide [225]. Yang et al. [225] reported the effects of andrographolide and neoandrographolide on hyperlipidemic mice induced by 75% yolk emulsion and hyperlipidemic rats induced by high fat emulsion. Andrographolide and neoandrographolide reduced triglyceride, total cholesterol, and low-density lipoprotein cholesterol in a dose dependent manner. Plasma aspartate transaminase and alanine transaminase levels were also significantly (P < 0.01) reduced compared to the positive control (Simvastatin). These compounds exhibited their lipid and lipoprotein reducing effects through downregulation of iNOS expression and upregulation of eNOS expression in aorta of hyperlipidemic rats [225]. The results of hypolipidemic study require further exploration of the molecular mechanism and the related signaling pathway.
5.10. Sexual Functions and Contraceptive Effect
AP and andrographolide showed earlier either contraceptive [58, 232–236], fertility [237–239], or no effects [240] in a variety of studies. AP showed contraceptive effects by terminating spermatogenesis in male albino rats [233, 236]. Zoha et al. [232] reported that there were no pregnant female mice that consumed AP mixed food daily after mating with the untreated male of potential fertility, which means that the AP has contraceptive effect on female mice. AP and andrographolide were effective to prevent cytokinesis of dividing spermatogenic cell lines resulting in stopped spermatogenesis [235, 236]. The antifertility effects of andrographolide are characterized by marked decreases of protein content along with significant increases of cholesterol, acid phosphatase, and alkaline phosphatase levels with appearance of fructose in the reproductive systems of rats [234]. Panossian et al. [241] reported negative results of AP's powdered extract on blood progesterone content in rats. However, the testosterone level with mounting frequency was significantly increased in mice at 4 weeks of andrographolide treatment. There was no toxicity of andrographolide (50 mg/kg) treatment for up to 8 weeks on number and motility of sperm. Further studies are needed to confirm the double stand activities of AP and andrographolide on female and male sexual behaviors. The investigation of specific mechanisms in regulation of fertility and contraceptive effects of AP and its active constituents would be valued to identify target point of controlling sexual behaviors.
5.11. Safety and Toxicity Effects
Generally, uses of AP as a medicine have been proved to be safe in various studies on mice, rats, and rabbits, as well as in in vitro assays and some clinical trials. Some conflicting results are also available. Few studies showed the toxic effect of AP on reproductive system by damaging the Sertoli cell in male gonads in albino rats. A dose of 25 and 50 mg/kg body weight for a period of 48 days demonstrated that antispermatogenic effect [58, 235, 236]. However, contradictory result was also demonstrated by numerous studies [237, 239, 240, 242–245]. The safety of AP extracts regarding the oral acute toxicity (>17 g/Kg, LD50) [237], testicular toxicity (>1 g/Kg, LD50) [240], and genotoxicity (5 g/Kg, LD50) [244] has been reported.
Due to AP's extreme bitterness, it may cause emesis. Some adverse effects including allergic reaction, gastric instability, fatigue, headache, loss of appetite, lymphadenopathy, diarrhea, metallic taste, and nausea are also observed in overdosing of AP extracts [63, 246]. It is suggested to avoid this plant during pregnancy due to ovulation preventive effects of the plant [232]. To date, all trials with few exceptions were for short duration; thus the prediction of safety for long term use would be farfetched.
6. Future Direction
Pharmacological activity of AP was investigated either using crude extracts or isolated bioactive compounds. Though the crude extract showed significant effects, isolation of bioactive compounds and investigation of pharmacology provide more specific knowledge especially about mechanism of actions of compounds. Conventional extraction processes (such as soxhlet extraction, maceration, and hydrodistillation) have been using by most of the researchers all over the world. However, selection of proper extraction methods is crucial for qualitative and quantitative studies of bioactive compounds derived from medicinal plant [247].
The reported pharmacology of bioactive compounds of AP is based on conventional extraction methods with few exceptions using different solvents, for example, methanol, ethanol, water, acetone, acetone-water, chloroform, and dichloromethane (Table 4). The conventional extraction techniques have some limitations including longer extraction time, costly and high purity solvent required, huge amount of solvent evaporation, low extraction selectivity, and thermal decomposition of thermolabile compounds. To overcome these problems, some new and promising extraction methods like ultrasound assisted extraction, enzyme-assisted extraction, microwave-assisted extraction, pulsed electric field assisted extraction, supercritical fluid extraction, and pressurized liquid extraction have been introduced which are termed as nonconventional extraction methods [248]. Some of these techniques are known as “green techniques” and are applicable for high yield and more purified compounds within a short time compared to classical methods. For example, extraction of phenolic antioxidants from five citrus peels (Yen Ben lemon, Meyer lemon, grapefruit, mandarin, and orange) by enzyme-assisted extraction was improved significantly with higher enzyme concentration [249]. Howard and Pandjaitan [250] extracted flavonoids from spinach by pressurized liquid extraction effectively. Therefore, it can be suggested that extraction of bioactive compounds from AP using nonconventional extraction methods of bioactive compounds would be valued to get high yield and purified compounds.
We mentioned earlier that tissue culture techniques have been applied successfully to form new flavones by differentiating callus culture. Jalal et al. [168] reported that differentiating tissue cultures of AP produce three new flavones, 5-hydroxy-7,8,2′-trimethoxy-, 5,2′-dihydroxy-7,8-dimethoxy-, and 5-hydroxy-7,8-dimethoxy-flavones. Arifullah et al. [141] isolated andrographolide and echiodinin from in vitro leaf callus of AP. In vitro root culture system can be exploited as a renewable source of minerals essential for designing effective drugs [167]. Other bioactive compounds can also be extracted via tissue culture. Adventitious roots from vegetative propagation of AP can also be an alternative source of plant materials for extraction of bioactive compounds from roots. We investigated a rapid and maximum adventitious roots in AP's shoot microcuttings applying soaking method at 3 mM IBA (Indole-3-butyric acid) (Figure 1(f)) (unpublished data). These adventitious roots can be further investigated to see the quality and content of bioactive compounds and pharmacological effects. Hence, adventitious rooting and plant tissue culture techniques can be alternative ways to meet the commercial demand of AP.
Stress alters growth and development of plants. In our investigation, we found that salinity stress showed deleterious effects on morphophysiological parameters including colour, plant height, leaf area, and root length of AP. It also causes less production of AP [251]. The stress conditions have a strong impact on the responsible metabolic pathways for the accumulation of the related natural products [252]. However, abiotic stress condition (as an agronomical approach) [253, 254] and biotechnological approaches (genetic transformation) [255] can be used to improve the content of some active compounds like flavonoids on a large scale in plants. Talei et al. [256] reported that high salinity level (16 dS/m) reached relatively higher andrographolide and neoandrographolide content compared to control. The increase in andrographolide and neoandrographolide contents at high salinity level (16 dS/m) was 177.57% and 131.18% compared to the control, respectively. Optimization of the yield of andrographolide and neoandrographolide content and investigation of other important bioactives of AP under abiotic stresses would be valued to meet the commercial demand of bioactive compounds of AP. To date, to our knowledge, any investigation of pharmacological activities of bioactive compounds obtained from salinity treated AP is not conducted yet. Therefore, it is very crucial to know the comparative efficacy of the compounds obtained from salinity treated and control AP. The obtained knowledge from the suggested study could also be used for further research design on AP for the betterment of human health.
7. Conclusion
The demand of AP is greatly increased in the past few years for its overwhelming therapeutic potentials. Available data on AP also clearly expresses a broad spectrum of pharmacological properties of this plant. Due to possessing extensive pharmacological activities, the AP can be safely regarded as one of the modern catholicons. However, the investigated pharmacological activities of AP need validation through the clinical study. Though several clinical studies were successfully completed without adverse effects or fatalities, most of them only investigated upper respiratory tract infections for a variety of conditions. Verification of the efficacy of other biological activities of AP including antidiabetic, anticancer, anti-inflammatory, and hepatoprotective activities, on human study subjects would bring a lot of benefits for the largest population of the globe. We assume that the AP could be useful as highly applied therapeutic agents for a variety of disorders in the near future to cure human diseases as well as some animal diseases. To fulfill this dream, the researchers might focus on multiplication of this plant to meet commercial demand besides the pharmacology study. Tissue culture techniques might be a good alternative to make AP available for researches (i.e., pharmacological study and phytochemical study to find new bioactive compounds) as well as conservation of this plant.
Acknowledgments
The authors would like to express their cordial thanks to Prof. Dr. Ahmed Jalal Khan Chowdhury from International Islamic University Malaysia, for his admirable inspiration. The authors' appreciation is also extended to Br. Abdur Razzak and Br. Md. Al Amin for their assistance in providing useful papers for the review. The authors are also grateful to Mr. Mohammad Ibrahim Auli Ullah from Universiti Sultan Zainal Abidin, Kuala Terengganu, Malaysia, for his critical proof reading of this paper.
Disclosure
This study was not involved in any handling of animals or live species.
Conflict of Interests
The authors have no conflict of interests to declare.
References
- 1.Chaudhary G., Goyal S., Poonia P. Lawsonia inermis Linnaeus: a phytopharmacological review. International Journal of Pharmaceutical Sciences and Drug Research. 2010;2(2):91–98. [Google Scholar]
- 2.Joy P., Thomas J., Mathew S., Skaria B. P. Medicinal plants. Tropical Horticulture. 1998;2:449–632. [Google Scholar]
- 3.Burkill I. H., Birtwistle W., Foxworthy F., Scrivenor J., Watson J. A Dictionary of the Economic Products of the Malay Peninsula. Kuala Lumpur, Malaysia: Ministry of Agriculture and Co-operatives; 1966. [Google Scholar]
- 4.Kavishankar G., Lakshmidevi N., Murthy S. M., Prakash H., Niranjana S. Diabetes and medicinal plants—a review. Journal of Pharmaceutical and Biomedical Sciences. 2011;2(3):65–80. [Google Scholar]
- 5.Hajiaghaee R., Akhondzadeh S. Herbal medicine in the treatment of Alzheimer's disease. Journal of Medicinal Plants. 2012;11(41):1–7. [Google Scholar]
- 6.Urbi Z., Hossain S., Hafizur Rahman K. M., Zayed T. M. Grape: a medicinal fruit species in the holy Qur'an and its ethnomedinical importance. World Applied Sciences Journal. 2014;30(3):253–265. doi: 10.5829/idosi.wasj.2014.30.03.81114. [DOI] [Google Scholar]
- 7.Duke J. A. Duke's Handbook of Medicinal Plants of the Bible. CRC Press, Taylor & Francis; 2007. [Google Scholar]
- 8.Akbar S. Andrographis paniculata: a review of pharmacological activities and clinical effects. Alternative Medicine Review. 2011;16(1):66–77. [PubMed] [Google Scholar]
- 9.Kabir M. H., Hasan N., Rahman M. M., et al. A survey of medicinal plants used by the Deb barma clan of the Tripura tribe of Moulvibazar district, Bangladesh. Journal of Ethnobiology and Ethnomedicine. 2014;10(1, article 19) doi: 10.1186/1746-4269-10-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jarukamjorn K., Nemoto N. Pharmacological aspects of Andrographis paniculata on health and its major diterpenoid constituent andrographolide. Journal of Health Science. 2008;54(4):370–381. doi: 10.1248/jhs.54.370. [DOI] [Google Scholar]
- 11.Ajaya Kumar R., Sridevi K., Vijaya Kumar N., Nanduri S., Rajagopal S. Anticancer and immunostimulatory compounds from Andrographis paniculata . Journal of Ethnopharmacology. 2004;92(2-3):291–295. doi: 10.1016/j.jep.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 12.Rajagopal S., Kumar R. A., Deevi D. S., Satyanarayana C., Rajagopalan R. Andrographolide, a potential cancer therapeutic agent isolated from Andrographis paniculata . Journal of Experimental Therapeutics and Oncology. 2003;3(3):147–158. doi: 10.1046/j.1359-4117.2003.01090.x. [DOI] [PubMed] [Google Scholar]
- 13.Cheung H.-Y., Cheung S.-H., Li J., et al. Andrographolide isolated from Andrographis paniculata induces cell cycle arrest and mitochondrial-mediated apoptosis in human leukemic HL-60 cells. Planta Medica. 2005;71(12):1106–1111. doi: 10.1055/s-2005-873128. [DOI] [PubMed] [Google Scholar]
- 14.Li W., Xu X., Zhang H., et al. Secondary metabolites from Andrographis paniculata . Chemical and Pharmaceutical Bulletin. 2007;55(3):455–458. doi: 10.1248/cpb.55.455. [DOI] [PubMed] [Google Scholar]
- 15.Harjotaruno S., Widyawaruyantil A., Zaini N. C. Apoptosis inducing effect of andrographolide on TD-47 human breast cancer cell line. African Journal of Traditional, Complementary and Alternative Medicines. 2008;4(3):345–351. doi: 10.4314/ajtcam.v4i3.31228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou J., Zhang S., Choon-Nam O., Shen H.-M. Critical role of pro-apoptotic Bcl-2 family members in andrographolide-induced apoptosis in human cancer cells. Biochemical Pharmacology. 2006;72(2):132–144. doi: 10.1016/j.bcp.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 17.Puri A., Saxena R., Saxena R. P., Saxena K. C., Srivastava V., Tandon J. S. Immunostimulant agents from Andrographis paniculata . Journal of Natural Products. 1993;56(7):995–999. doi: 10.1021/np50097a002. [DOI] [PubMed] [Google Scholar]
- 18.Matsuda T., Kuroyanagi M., Sugiyama S., Umehara K., Ueno A., Nishi K. Cell differentiation-inducing diterpenes from Andrographis paniculata Nees. Chemical and Pharmaceutical Bulletin. 1994;42(6):1216–1225. doi: 10.1248/cpb.42.1216. [DOI] [PubMed] [Google Scholar]
- 19.Gupta S., Choudhry M. A., Yadava J. N. S., Srivastava V., Tandon J. S. Antidiarrhoeal activity of diterpenes of Andrographis paniculata (Kal-Megh) against Escherichia coli enterotoxin in in vivo models. International Journal of Crude Drug Research. 1990;28(4):273–283. [Google Scholar]
- 20.Gupta S., Yadava J. N. S., Tandon J. S. Antisecretory (antidiarrhoeal) activity of Indian medicinal plants against Escherichia coli enterotoxin-induced secretion in rabbit and guinea pig ileal loop models. International Journal of Pharmacognosy. 1993;31(3):198–204. doi: 10.3109/13880209309082942. [DOI] [Google Scholar]
- 21.Sharma A., Singh R. T., Sehgal V., Handa S. S. Antihepatotoxic activity of some plants used in herbal formulations. Fitoterapia. 1991;62(2):131–138. [Google Scholar]
- 22.Tang W., Eisenbrand G. Andrographis paniculata (Burm. f.) Nees. In: Tang W., Eisenbrand G., editors. Chinese Drugs of Plant Origin Chemistry, Pharmacology, and Use in Raditional and Modern Medicine. Berlin, Germany: Springer; 1992. pp. 97–103. [Google Scholar]
- 23.Nanduri S., Nyavanandi V. K., Sanjeeva Rao Thunuguntla S., et al. Synthesis and structure-activity relationships of andrographolide analogues as novel cytotoxic agents. Bioorganic and Medicinal Chemistry Letters. 2004;14(18):4711–4717. doi: 10.1016/j.bmcl.2004.06.090. [DOI] [PubMed] [Google Scholar]
- 24.Yu B.-C., Hung C.-R., Chen W.-C., Cheng J.-T. Antihyperglycemic effect of andrographolide in streptozotocin-induced diabetic rats. Planta Medica. 2003;69(12):1075–1079. doi: 10.1055/s-2003-45185. [DOI] [PubMed] [Google Scholar]
- 25.Yu B. C., Chang C. K., Su C. F., Cheng J. T. Mediation of β-endorphin in andrographolide-induced plasma glucose-lowering action in type I diabetes-like animals. Naunyn-Schmiedeberg's Archives of Pharmacology. 2008;377(4–6):529–540. doi: 10.1007/s00210-007-0240-0. [DOI] [PubMed] [Google Scholar]
- 26.Subramanian R., Asmawi M. Z. Inhibition of α-glucosidase by Andrographis paniculata ethanol extract in rats. Pharmaceutical Biology. 2006;44(8):600–606. doi: 10.1080/13880200600896892. [DOI] [Google Scholar]
- 27.Subramanian R., Asmawi M. Z., Sadikun A. In vitro α-glucosidase and α-amylase enzyme inhibitory effects of Andrographis paniculata extract and andrographolide. Acta Biochimica Polonica. 2008;55(2):391–398. [PubMed] [Google Scholar]
- 28.Chiou W. F., Lin J. J., Chen C. F. Andrographolide suppresses the expression of inducible nitric oxide synthase in macrophage and restores the vasoconstriction in rat aorta treated with lipopolysaccharide. British Journal of Pharmacology. 1998;125(2):327–334. doi: 10.1038/sj.bjp.0702073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chiou W.-F., Chen C.-F., Lin J.-J. Mechanisms of suppression of inducible nitric oxide synthase (iNOS) expression in RAW 264.7 cells by andrographolide. British Journal of Pharmacology. 2000;129(8):1553–1560. doi: 10.1038/sj.bjp.0703191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sheeja K., Shihab P. K., Kuttan G. Antioxidant and anti-inflammatory activities of the plant Andrographis paniculata nees. Immunopharmacology and Immunotoxicology. 2006;28(1):129–140. doi: 10.1080/08923970600626007. [DOI] [PubMed] [Google Scholar]
- 31.Shen Y. C., Chen C. F., Chiou W. F. Andrographolide prevents oxygen radical production by human neutrophils: possible mechanism(s) involved in its anti-inflammatory effect. British Journal of Pharmacology. 2002;135(2):399–406. doi: 10.1038/sj.bjp.0704493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen Y.-C., Chen C.-F., Chiou W.-F. Suppression of rat neutrophil reactive oxygen species production and adhesion by the diterpenoid lactone andrographolide. Planta Medica. 2000;66(4):314–317. doi: 10.1055/s-2000-8537. [DOI] [PubMed] [Google Scholar]
- 33.Wiart C., Kumar K., Yusof M. Y., Hamimah H., Fauzi Z. M., Sulaiman M. Antiviral properties of ent-labdene diterpenes of Andrographis paniculata Nees, inhibitors of herpes simplex virus type 1. Phytotherapy Research. 2005;19(12):1069–1070. doi: 10.1002/ptr.1765. [DOI] [PubMed] [Google Scholar]
- 34.Misra P., Pal N. L., Guru P. Y., Katiyar J. C., Srivastava V., Tandon J. S. Antimalarial activity of Andrographis paniculata (Kalmegh) against Plasmodium berghei NK 65 in Mastomys natalensis. International Journal of Pharmacognosy. 1992;30(4):263–274. doi: 10.3109/13880209209054010. [DOI] [Google Scholar]
- 35.Kamdem R. E., Sang S., Ho C.-T. Mechanism of the superoxide scavenging activity of neoandrographolide—a natural product from Andrographis paniculata nees. Journal of Agricultural and Food Chemistry. 2002;50(16):4662–4665. doi: 10.1021/jf025556f. [DOI] [PubMed] [Google Scholar]
- 36.Singh R. P., Banerjee S., Rao A. R. Modulatory influence of Andrographis paniculata on mouse hepatic and extrahepatic carcinogen metabolizing enzymes and antioxidant status. Phytotherapy Research. 2001;15(5):382–390. doi: 10.1002/ptr.730. [DOI] [PubMed] [Google Scholar]
- 37.Akowuah G. A., Zhari I., Mariam A. Analysis of urinary andrographolides and antioxidant status after oral administration of Andrographis paniculata leaf extract in rats. Food and Chemical Toxicology. 2008;46(12):3616–3620. doi: 10.1016/j.fct.2008.09.008. [DOI] [PubMed] [Google Scholar]
- 38.Zhang C. Y., Tan B. K. H. Mechanisms of cardiovascular activity of Andrographis paniculata in the anaesthetized rat. Journal of Ethnopharmacology. 1997;56(2):97–101. doi: 10.1016/S0378-8741(97)01509-2. [DOI] [PubMed] [Google Scholar]
- 39.Tan B.-H., Zhang A. Andrographis paniculata and the cardiovascular system. Oxidative Stress and Disease. 2004;14:441–456. [Google Scholar]
- 40.Handa S. S., Sharma A. Hepatoprotective activity of andrographolide from Andrographis paniculata against carbontetrachloride. The Indian Journal of Medical Research. 1990;92:276–283. [PubMed] [Google Scholar]
- 41.Handa S. S., Sharma A. Hepatoprotective activity of andrographolide against galactosamine and paracetamol intoxication in rats. Indian Journal of Medical Research. 1990;92:284–292. [PubMed] [Google Scholar]
- 42.Kapil A., Koul I. B., Banerjee S. K., Gupta B. D. Antihepatoxic effects of major diterpenoid constituents of Andrographis paniculata . Biochemical Pharmacology. 1993;46(1):182–185. doi: 10.1016/0006-2952(93)90364-3. [DOI] [PubMed] [Google Scholar]
- 43.Chander R., Srivastava V., Tandon J. S., Kapoor N. K. Antihepatotoxic activity of diterpenes of Andrographis paniculata (Kal-Megh) against Plasmodium berghei-induced hepatic damage in Mastomys natalensis. International Journal of Pharmacognosy. 1995;33(2):135–138. doi: 10.3109/13880209509055213. [DOI] [Google Scholar]
- 44.Trivedi N., Rawal U. M. Hepatoprotective and toxicological evaluation of Andrographis paniculata on severe liver damage. Indian Journal of Pharmacology. 2000;32(5):288–293. [Google Scholar]
- 45.Trivedi N. P., Rawal U. M. Hepatoprotective and antioxidant property of Andrographis paniculata (Nees) in BHC induced liver damage in mice. Indian Journal of Experimental Biology. 2001;39(1):41–46. [PubMed] [Google Scholar]
- 46.Visen P. K. S., Saraswat B., Vuksan V., Dhawan B. N. Effect of andrographolide on monkey hepatocytes against galactosamine induced cell toxicity: an in-vitro study. Journal of Complementary and Integrative Medicine. 2007;4(1, article 10) [Google Scholar]
- 47.Saraswat B., Visen P. K. S., Patnaik G. K., Dhawan B. N. Effect of andrographolide against galactosamine-induced hepatotoxicity. Fitoterapia. 1995;66(5):415–420. [Google Scholar]
- 48.Choudhury B. R., Poddar M. K. Andrographolide and kalmegh (Andrographis paniculata) extract: in vivo and in vitro effect on hepatic lipid peroxidation. Methods and Findings in Experimental and Clinical Pharmacology. 1984;6(9):481–485. [PubMed] [Google Scholar]
- 49.Choudhury B. R., Haque S. J., Poddar M. K. In vivo and in vitro effects of kalmegh (Andrographis paniculata) extract and andrographolide on hepatic microsomal drug metabolizing enzymes. Planta Medica. 1987;53(2):135–140. doi: 10.1055/s-2006-962655. [DOI] [PubMed] [Google Scholar]
- 50.Rana A. C., Avadhoot Y. Hepatoprotective effects of Andrograhphis paniculata against carbon tetrachloride-induced liver damage. Archives of Pharmacal Research. 1991;14(1):93–95. doi: 10.1007/BF02857822. [DOI] [PubMed] [Google Scholar]
- 51.Visen P. K. S., Shukia B., Patnaik G. K., Dhawan B. N. Andrographolide protects rat hepatocytes against paracetamol-induced damage. Journal of Ethnopharmacology. 1993;40(2):131–136. doi: 10.1016/0378-8741(93)90058-D. [DOI] [PubMed] [Google Scholar]
- 52.Shukla B., Visen P. K. S., Patnaik G. K., Dhawan B. N. Choleretic effect of andrographolide in rats and guinea pigs. Planta Medica. 1992;58(2):146–149. doi: 10.1055/s-2006-961416. [DOI] [PubMed] [Google Scholar]
- 53.Calabrese C., Berman S. H., Babish J. G., et al. A phase I trial of andrographolide in HIV positive patients and normal volunteers. Phytotherapy Research. 2000;14(5):333–338. doi: 10.1002/1099-1573(200008)14:5<333::aid-ptr584>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 54.Otake T., Mori H., Morimoto M., et al. Screening of Indonesian plant extracts for anti-human immunodeficiency virus—type 1 (HIV-1) activity. Phytotherapy Research. 1995;9(1):6–10. doi: 10.1002/ptr.2650090103. [DOI] [Google Scholar]
- 55.Chang R. S., Ding L., Gai-Qing C., Qi-Choa P., Ze-Lin Z., Smith K. M. Dehydroandrographolide succinic acid monoester as an inhibitor against the human immunodeficiency virus (43225) Proceedings of the Society for Experimental Biology and Medicine. 1991;197(1):59–66. doi: 10.3181/00379727-197-43225. [DOI] [PubMed] [Google Scholar]
- 56.Basak A., Cooper S., Roberge A. G., Banik U. K., Chrétien M., Seidah N. G. Inhibition of proprotein convertases-1, -7 and furin by diterpines of Andrographis paniculata and their succinoyl esters. Biochemical Journal. 1999;338(1):107–113. doi: 10.1042/0264-6021:3380107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Iruretagoyena M. I., Tobar J. A., González P. A., et al. Andrographolide interferes with T cell activation and reduces experimental autoimmune encephalomyelitis in the mouse. Journal of Pharmacology and Experimental Therapeutics. 2005;312(1):366–372. doi: 10.1124/jpet.104.072512. [DOI] [PubMed] [Google Scholar]
- 58.Akbarsha M. A., Murugaian P. Aspects of the male reproductive toxicity/male antifertility property of andrographolide in albino rats: effect on the testis and the cauda epididymidal spermatozoa. Phytotherapy Research. 2000;14(6):432–435. doi: 10.1002/1099-1573(200009)14:6<432::AID-PTR622>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 59.Niranjan A., Tewari S. K., Lehri A. Biological activities of Kalmegh (Andrographis paniculata Nees) and its active principles-A review. Indian Journal of Natural Products and Resources. 2010;1(2):125–135. [Google Scholar]
- 60.Wu Z., Raven P. H., Hong D. Y., Garden M. B. Flora of China: Cucurbitaceae Through Valerianaceae with Annonaceae and Berberidaceae. Beijing, China: Science Press; 1996. [Google Scholar]
- 61.Benoy G. K., Animesh D. K., Aninda M., Priyanka D. K., Sandip H. An overview on Andrographis paniculata (burm. F.) Nees. International Journal of Research in Ayurveda and Pharmacy. 2012;3(6):752–760. doi: 10.7897/2277-4343.03610. [DOI] [Google Scholar]
- 62.Boopathi C. Andrographis spp.: a source of bitter compounds for medicinal use. Ancient Science of Life. 2000;19(3-4):164–168. [PMC free article] [PubMed] [Google Scholar]
- 63.Anju D., Jugnu G., Kavitha S., Arjun N., Sandeep D. A review on medicinal prospective of Andrographis paniculata Nees. Journal of Pharmaceutical and Scientific Innovation. 2012;1(1):1–4. [Google Scholar]
- 64.Patidar S., Gontia A. S., Upadhyay A., Nayak P. S. Biochemical constituents in Kalmegh (Andrographis paniculata Nees.) under various row spacing's and nitrogen levels. World Applied Sciences Journal. 2011;15(8):1095–1099. [Google Scholar]
- 65.Bhattacharya S., Puri S., Jamwal A., Sharma S. Studies on seed germination and seedling growth in Kalmegh (Andrographis paniculata Wall. Ex Nees) under abiotic stress conditions. International Journal of Science, Environment and Technology. 2012;1(3):197–204. [Google Scholar]
- 66.Gnanasekaran G., Murthy G. V. S. Lectotypifications in andrographis (Acanthaceae) Rheedea. 2012;22(2):77–79. [Google Scholar]
- 67.Rao Y. K., Damu A. G., Rao A. J., et al. Flavonoids from Andrographis viscosula. Chemical and Pharmaceutical Bulletin. 2003;51(12):1374–1376. doi: 10.1248/cpb.51.1374. [DOI] [PubMed] [Google Scholar]
- 68.Parixit B., Bharath C., Rajarajeshwari N., Ganapaty S. The genus Andrographis—a review. International Journal of Pharmaceutical Sciences. 2012;4(1):1835–1856. [Google Scholar]
- 69.Saggoo M., Bir S. Meiotic studies in certain members of family Acanthaceae from South India. The Journal of the Indian Botanical Society. 1986;65(3):310–315. [Google Scholar]
- 70.Roy S. K., Datta P. Chromosomal biotypes of Andrographis paniculata in India and Bangladesh. Cytologia. 1988;53(2):369–378. [Google Scholar]
- 71.Mishra S. K., Sangwan N. S., Sangwan R. S. Andrographis paniculata (Kalmegh): a review. Pharmacognosy Reviews. 2007;1(2):283–298. [Google Scholar]
- 72.Wu T.-S., Chern H.-J., Damu A. G., et al. Flavonoids and ent-labdane diterpenoids from Andrographis paniculata and their antiplatelet aggregatory and vasorelaxing effects. Journal of Asian Natural Products Research. 2008;10(1):17–24. doi: 10.1080/10286020701273627. [DOI] [PubMed] [Google Scholar]
- 73.USDA (United States Department of Agriculture) GRIN Taxonomy for Plants. April 2014, http://www.ars-grin.gov/cgi-bin/npgs/html/taxon.pl?414228.
- 74.Tropicos.org. Andrographis paniculata (Burm. f.) Wall. ex Nees. 2014, http://www.tropicos.org/Name/100007.
- 75.Valdiani A., Kadir M. A., Tan S. G., Talei D., Abdullah M. P., Nikzad S. Nain-e havandi andrographis paniculata present yesterday, absent today: a plenary review on underutilized herb of Iran's pharmaceutical plants. Molecular Biology Reports. 2012;39(5):5409–5424. doi: 10.1007/s11033-011-1341-x. [DOI] [PubMed] [Google Scholar]
- 76.Sharma M., Sharma R. Identification, purification and quantification of andrographolide from Andrographis paniculata (burm. F.) Nees by HPTLC at different stages of life cycle of crop. Journal of Current Chememical and Pharmaceutical Sciences. 2013;3(1):23–32. [Google Scholar]
- 77.Borhanuddin M., Shamsuzzoha M., Hussain A. H. Hypoglycaemic effects of Andrographis paniculata Nees on non-diabetic rabbits. Bangladesh Medical Research Council Bulletin. 1994;20(1):24–26. [PubMed] [Google Scholar]
- 78.Madav S., Tripathi H. C., Mishra S. K. Analgesic, antipyretic and antiulcerogenic effects of andrographolide. Indian Journal of Pharmaceutical Sciences. 1995;57(3):121–125. [Google Scholar]
- 79.Sheeja K., Guruvayoorappan C., Kuttan G. Antiangiogenic activity of Andrographis paniculata extract and andrographolide. International Immunopharmacology. 2007;7(2):211–221. doi: 10.1016/j.intimp.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 80.Reddy V. L. N., Reddy S. M., Ravikanth V., et al. A new bis-andrographolide ether from Andrographis paniculata nees and evaluation of anti-HIV activity. Natural Product Research. 2005;19(3):223–230. doi: 10.1080/14786410410001709197. [DOI] [PubMed] [Google Scholar]
- 81.Thisoda P., Rangkadilok N., Pholphana N., Worasuttayangkurn L., Ruchirawat S., Satayavivad J. Inhibitory effect of Andrographis paniculata extract and its active diterpenoids on platelet aggregation. European Journal of Pharmacology. 2006;553(1–3):39–45. doi: 10.1016/j.ejphar.2006.09.052. [DOI] [PubMed] [Google Scholar]
- 82.Shi M.-D., Lin H.-H., Lee Y.-C., Chao J.-K., Lin R.-A., Chen J.-H. Inhibition of cell-cycle progression in human colorectal carcinoma Lovo cells by andrographolide. Chemico-Biological Interactions. 2008;174(3):201–210. doi: 10.1016/j.cbi.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 83.Sheeja K., Kuttan G. Activation of cytotoxic T lymphocyte responses and attenuation of tumor growth in vivo by Andrographis paniculata extract and andrographolide. Immunopharmacology and Immunotoxicology. 2007;29(1):81–93. doi: 10.1080/08923970701282726. [DOI] [PubMed] [Google Scholar]
- 84.Ji L., Liu T., Liu J., Chen Y., Wang Z. Andrographolide inhibits human hepatoma-derived Hep3B cell growth through the activation of c-Jun N-terminal kinase. Planta Medica. 2007;73(13):1397–1401. doi: 10.1055/s-2007-990230. [DOI] [PubMed] [Google Scholar]
- 85.Manikam S. D., Stanslas J. Andrographolide inhibits growth of acute promyelocytic leukaemia cells by inducing retinoic acid receptor-independent cell differentiation and apoptosis. Journal of Pharmacy and Pharmacology. 2009;61(5):69–78. doi: 10.1211/jpp/61.01.0010. [DOI] [PubMed] [Google Scholar]
- 86.Liang F.-P., Lin C.-H., Kuo C.-D., Chao H.-P., Fu S.-L. Suppression of v-Src transformation by andrographolide via degradation of the v-Src protein and attenuation of the Erk signaling pathway. Journal of Biological Chemistry. 2008;283(8):5023–5033. doi: 10.1074/jbc.M705877200. [DOI] [PubMed] [Google Scholar]
- 87.Jiang C.-G., Li J.-B., Liu F.-R., Wu T., Yu M., Xu H.-M. Andrographolide inhibits the adhesion of gastric cancer cells to endothelial cells by blocking E-selectin expression. Anticancer Research. 2007;27(4 B):2439–2447. [PubMed] [Google Scholar]
- 88.Shi M.-D., Lin H.-H., Chiang T.-A., et al. Andrographolide could inhibit human colorectal carcinoma Lovo cells migration and invasion via down-regulation of MMP-7 expression. Chemico-Biological Interactions. 2009;180(3):344–352. doi: 10.1016/j.cbi.2009.04.011. [DOI] [PubMed] [Google Scholar]
- 89.Lee Y.-C., Lin H.-H., Hsu C.-H., Wang C.-J., Chiang T.-A., Chen J.-H. Inhibitory effects of andrographolide on migration and invasion in human non-small cell lung cancer A549 cells via down-regulation of PI3K/Akt signaling pathway. European Journal of Pharmacology. 2010;632(1–3):23–32. doi: 10.1016/j.ejphar.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 90.Burgos R. A., Seguel K., Perez M., et al. Andrographolide inhibits IFN-γ and IL-2 cytokine production and protects against cell apoptosis. Planta Medica. 2005;71(5):429–434. doi: 10.1055/s-2005-864138. [DOI] [PubMed] [Google Scholar]
- 91.Zhou J., Ong C.-N., Hur G.-M., Shen H.-M. Inhibition of the JAK-STAT3 pathway by andrographolide enhances chemosensitivity of cancer cells to doxorubicin. Biochemical Pharmacology. 2010;79(9):1242–1250. doi: 10.1016/j.bcp.2009.12.014. [DOI] [PubMed] [Google Scholar]
- 92.Zhang X. F., Tan B. K. H. Antihyperglycaemic and anti-oxidant properties of Andrographis paniculata in normal and diabetic rats. Clinical and Experimental Pharmacology and Physiology. 2000;27(5-6):358–363. doi: 10.1046/j.1440-1681.2000.03253.x. [DOI] [PubMed] [Google Scholar]
- 93.Subramanian R., Asmawi M. Z., Sadikun A. Effect of andrographolide and ethanol extract of Andrographis paniculata on liver glycolytic, gluconeogenic, and lipogenic enzymes in a type 2 diabetic rat model. Pharmaceutical Biology. 2008;46(10-11):772–780. doi: 10.1080/13880200802316079. [DOI] [Google Scholar]
- 94.Subramanian R., Asmawi M. Z., Sadikun A. Effect of ethanolic extract of Andrographis paniculata (Burm. F.) nees on a combination of fat-fed diet and low dose streptozotocin induced chronic insulin resistance in rats. Diabetologia Croatica. 2008;37(1):13–22. [Google Scholar]
- 95.Wang T., Liu B., Zhang W., Wilson B., Hong J.-S. Andrographolide reduces inflammation-mediated dopaminergic neurodegeneration in mesencephalic neuron-glia cultures by inhibiting microglial activation. Journal of Pharmacology and Experimental Therapeutics. 2004;308(3):975–983. doi: 10.1124/jpet.103.059683. [DOI] [PubMed] [Google Scholar]
- 96.Hidalgo M. A., Romero A., Figueroa J., et al. Andrographolide interferes with binding of nuclear factor-κB to DNA in HL-60-derived neutrophilic cells. British Journal of Pharmacology. 2005;144(5):680–686. doi: 10.1038/sj.bjp.0706105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Li J., Luo L., Wang X., Liao B., Li G. Inhibition of NF-κB expression and allergen-induced airway inflammation in a mouse allergic asthma model by andrographolide. Cellular & Molecular Immunology. 2009;6(5):381–385. doi: 10.1038/cmi.2009.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bao Z., Guan S., Cheng C., et al. A novel antiinflammatory role for andrographolide in asthma via inhibition of the nuclear factor-κb pathway. The American Journal of Respiratory and Critical Care Medicine. 2009;179(8):657–665. doi: 10.1164/rccm.200809-1516OC. [DOI] [PubMed] [Google Scholar]
- 99.Qin L.-H., Kong L., Shi G.-J., Wang Z.-T., Ge B.-X. Andrographolide inhibits the production of TNF-α and interleukin-12 in lipopolysaccharide-stimulated macrophages: role of mitogen-activated protein kinases. Biological and Pharmaceutical Bulletin. 2006;29(2):220–224. doi: 10.1248/bpb.29.220. [DOI] [PubMed] [Google Scholar]
- 100.Parichatikanond W., Suthisisang C., Dhepakson P., Herunsalee A. Study of anti-inflammatory activities of the pure compounds from Andrographis paniculata (burm.f.) Nees and their effects on gene expression. International Immunopharmacology. 2010;10(11):1361–1373. doi: 10.1016/j.intimp.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 101.Ko H.-C., Wei B.-L., Chiou W.-F. The effect of medicinal plants used in Chinese folk medicine on RANTES secretion by virus-infected human epithelial cells. Journal of Ethnopharmacology. 2006;107(2):205–210. doi: 10.1016/j.jep.2006.03.004. [DOI] [PubMed] [Google Scholar]
- 102.Chen J.-X., Xue H.-J., Ye W.-C., et al. Activity of andrographolide and its derivatives against influenza virus in vivo and in vitro . Biological and Pharmaceutical Bulletin. 2009;32(8):1385–1391. doi: 10.1248/bpb.32.1385. [DOI] [PubMed] [Google Scholar]
- 103.Sinha J., Mukhopadhyay S., Das N., Basu M. K. Targeting of liposomal andrographolide to L. donovani-infected macrophages in vivo. Drug Delivery. 2000;7(4):209–213. doi: 10.1080/107175400455137. [DOI] [PubMed] [Google Scholar]
- 104.Yang S., Evens A. M., Prachand S., et al. Mitochondrial-mediated apoptosis in lymphoma cells by the diterpenoid lactone andrographolide, the active component of Andrographis paniculata . Clinical Cancer Research. 2010;16(19):4755–4768. doi: 10.1158/1078-0432.CCR-10-0883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zhou J., Lu G.-D., Ong C.-S., Ong C.-N., Shen H.-M. Andrographolide sensitizes cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 up-regulation. Molecular Cancer Therapeutics. 2008;7(7):2170–2180. doi: 10.1158/1535-7163.MCT-08-0071. [DOI] [PubMed] [Google Scholar]
- 106.Sheeja K., Kuttan G. Protective effect of Andrographis paniculata and andrographolide on cyclophosphamide-induced urothelial toxicity. Integrative Cancer Therapies. 2006;5(3):244–251. doi: 10.1177/1534735406291984. [DOI] [PubMed] [Google Scholar]
- 107.Woo A. Y. H., Waye M. M. Y., Tsui S. K. W., Yeung S. T. W., Cheng C. H. K. Andrographolide up-regulates cellular-reduced glutathione level and protects cardiomyocytes against hypoxia/reoxygenation injury. Journal of Pharmacology and Experimental Therapeutics. 2008;325(1):226–235. doi: 10.1124/jpet.107.133918. [DOI] [PubMed] [Google Scholar]
- 108.Lee T.-Y., Lee K.-C., Chang H.-H. Modulation of the cannabinoid receptors by andrographolide attenuates hepatic apoptosis following bile duct ligation in rats with fibrosis. Apoptosis. 2010;15(8):904–914. doi: 10.1007/s10495-010-0502-z. [DOI] [PubMed] [Google Scholar]
- 109.Panossian A., Davtyan T., Gukassyan N., et al. Effect of andrographolide and Kan Jang—fixed combination of extract SHA-10 and extract SHE-3—on proliferation of human lymphocytes, production of cytokines and immune activation markers in the whole blood cells culture. Phytomedicine. 2002;9(7):598–605. doi: 10.1078/094471102321616409. [DOI] [PubMed] [Google Scholar]
- 110.Burgos R. A., Hidalgo M. A., Monsalve J., Labranche T. P., Eyre P., Hancke J. L. 14-Deoxyandrographolide as a platelet activating factor antagonist in bovine neutrophils. Planta Medica. 2005;71(7):604–608. doi: 10.1055/s-2005-871264. [DOI] [PubMed] [Google Scholar]
- 111.Xu Y., Chen A., Fry S., Barrow R. A., Marshall R. L., Mukkur T. K. S. Modulation of immune response in mice immunised with an inactivated Salmonella vaccine and gavaged with Andrographis paniculata extract or andrographolide. International Immunopharmacology. 2007;7(4):515–523. doi: 10.1016/j.intimp.2006.12.008. [DOI] [PubMed] [Google Scholar]
- 112.Carretta M. D., Alarcón P., Jara E., et al. Andrographolide reduces IL-2 production in T-cells by interfering with NFAT and MAPK activation. European Journal of Pharmacology. 2009;602(2-3):413–421. doi: 10.1016/j.ejphar.2008.11.011. [DOI] [PubMed] [Google Scholar]
- 113.Naik S. R., Hule A. Evaluation of immunomodulatory activity of an extract of andrographolides from Andographis paniculata . Planta Medica. 2009;75(8):785–791. doi: 10.1055/s-0029-1185398. [DOI] [PubMed] [Google Scholar]
- 114.Wang W., Wang J., Dong S.-F., et al. Immunomodulatory activity of andrographolide on macrophage activation and specific antibody response. Acta Pharmacologica Sinica. 2010;31(2):191–201. doi: 10.1038/aps.2009.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lin T.-P., Chen S.-Y., Duh P.-D., Chang L.-K., Liu Y.-N. Inhibition of the Epstein-Barr virus lytic cycle by andrographolide. Biological and Pharmaceutical Bulletin. 2008;31(11):2018–2023. doi: 10.1248/bpb.31.2018. [DOI] [PubMed] [Google Scholar]
- 116.Hung S.-K., Hung L.-C., Kuo C.-D., et al. Andrographolide sensitizes Ras-transformed cells to radiation in vitro and in vivo. International Journal of Radiation Oncology Biology Physics. 2010;77(4):1232–1239. doi: 10.1016/j.ijrobp.2010.01.007. [DOI] [PubMed] [Google Scholar]
- 117.Singha P. K., Roy S., Dey S. Protective activity of andrographolide and arabinogalactan proteins from Andrographis paniculata Nees. against ethanol-induced toxicity in mice. Journal of Ethnopharmacology. 2007;111(1):13–21. doi: 10.1016/j.jep.2006.10.026. [DOI] [PubMed] [Google Scholar]
- 118.Batkhuu J., Hattori K., Takano F., Fushiya S., Oshiman K.-I., Fujimiya Y. Suppression of NO production in activated macrophages in vitro and ex vivo by neoandrographolide isolated from Andrographis paniculata . Biological and Pharmaceutical Bulletin. 2002;25(9):1169–1174. doi: 10.1248/bpb.25.1169. [DOI] [PubMed] [Google Scholar]
- 119.Liu J., Wang Z.-T., Ji L.-L., Ge B.-X. Inhibitory effects of neoandrographolide on nitric oxide and prostaglandin E2 production in LPS-stimulated murine macrophage. Molecular and Cellular Biochemistry. 2007;298(1-2):49–57. doi: 10.1007/s11010-006-9349-6. [DOI] [PubMed] [Google Scholar]
- 120.Pfisterer P. H., Rollinger J. M., Schyschka L., Rudy A., Vollmar A. M., Stuppner H. Neoandrographolide from Andrographis paniculata as a potential natural chemosensitizer. Planta Medica. 2010;76(15):1698–1700. doi: 10.1055/s-0030-1249876. [DOI] [PubMed] [Google Scholar]
- 121.Sule A., Ahmed Q. U., Samah O. A., et al. Bioassay guided isolation of antibacterial compounds from Andrographis paniculata . The American Journal of Applied Sciences. 2011;8(6):525–534. doi: 10.3844/ajassp.2011.525.534. [DOI] [Google Scholar]
- 122.Ahmed Q. U., Samah O. A., Sule A. Andrographis paniculata (Burm. f) Wall. ex Ness: a potent antibacterial plant. In: Bobbarala V., editor. Antimicrobial Agents. InTech; 2012. pp. 345–360. [Google Scholar]
- 123.Sule A., Ahmed Q. U., Latip J., et al. Antifungal activity of Andrographis paniculata extracts and active principles against skin pathogenic fungal strains in vitro . Pharmaceutical Biology. 2012;50(7):850–856. doi: 10.3109/13880209.2011.641021. [DOI] [PubMed] [Google Scholar]
- 124.Roy D. N., Mandal S., Sen G., Mukhopadhyay S., Biswas T. 14-Deoxyandrographolide desensitizes hepatocytes to tumour necrosis factor-alpha-induced apoptosis through calcium-dependent tumour necrosis factor receptor superfamily member 1A release via the NO/cGMP pathway. British Journal of Pharmacology. 2010;160(7):1823–1843. doi: 10.1111/j.1476-5381.2010.00836.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Burgos R. A., Loyola M., Hidalgo M. A., Labranche T. P., Hancke J. L. Effect of 14-deoxyandrographolide on calcium-mediated rat uterine smooth muscle contractility. Phytotherapy Research. 2003;17(9):1011–1015. doi: 10.1002/ptr.1275. [DOI] [PubMed] [Google Scholar]
- 126.Zhang C. Y., Tan B. K. H. Effects of 14-deoxyandrographolide and 14-deoxy-11,12-didehydroandrographolide on nitric oxide production in cultured human endothelial cells. Phytotherapy Research. 1999;13(2):157–159. doi: 10.1002/(SICI)1099-1573(199903)13:2<157::AID-PTR388>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 127.Zhang C. Y., Tan B. K. H. Vasorelaxation of rat thoracic aorta caused by 14-deoxyandrographolide. Clinical and Experimental Pharmacology and Physiology. 1998;25(6):424–429. doi: 10.1111/j.1440-1681.1998.tb02226.x. [DOI] [PubMed] [Google Scholar]
- 128.Lee M.-J., Rao Y. K., Chen K., Lee Y.-C., Chung Y.-S., Tzeng Y.-M. Andrographolide and 14-deoxy-11,12-didehydroandrographolide from Andrographis paniculata attenuate high glucose-induced fibrosis and apoptosis in murine renal mesangeal cell lines. Journal of Ethnopharmacology. 2010;132(2):497–505. doi: 10.1016/j.jep.2010.07.057. [DOI] [PubMed] [Google Scholar]
- 129.Akowuah G. A., Zhari I., Mariam A., Yam M. F. Absorption of andrographolides from Andrographis paniculata and its effect on CCl4-induced oxidative stress in rats. Food and Chemical Toxicology. 2009;47(9):2321–2326. doi: 10.1016/j.fct.2009.06.022. [DOI] [PubMed] [Google Scholar]
- 130.Tan M., Kuroyanagi M., Sulaiman S., Najimudin N., Muhammad T. T. Cytotoxic activities of major diterpenoid constituents of Andrographis paniculata in a panel of human tumor cell lines. Pharmaceutical Biology. 2005;43(6):501–508. [Google Scholar]
- 131.Yoopan N., Thisoda P., Rangkadilok N., et al. Cardiovascular effects of 14-Deoxy-11,12-didehydroandrographolide and Andrographis paniculata extracts. Planta Medica. 2007;73(6):503–511. doi: 10.1055/s-2007-967181. [DOI] [PubMed] [Google Scholar]
- 132.Subramanian R., Asmawi M. Z., Sadikun A. A bitter plant with a sweet future? A comprehensive review of an oriental medicinal plant: Andrographis paniculata . Phytochemistry Reviews. 2012;11(1):39–75. doi: 10.1007/s11101-011-9219-z. [DOI] [Google Scholar]
- 133.He X., Zeng X., Hu H., Wu Y. Cytotoxic biotransformed products from andrographolide by Rhizopus stolonifer ATCC 12939. Journal of Molecular Catalysis B: Enzymatic. 2010;62(3-4):242–247. doi: 10.1016/j.molcatb.2009.11.002. [DOI] [Google Scholar]
- 134.Shen Y.-H., Li R.-T., Xiao W.-L., Lin Z.-W., Zhao Q.-S., Sun H.-D. ent-labdane diterpenoids from Andrographis paniculata . Journal of Natural Products. 2006;69(3):319–322. doi: 10.1021/np050160u. [DOI] [PubMed] [Google Scholar]
- 135.Ji L.-L., Wang Z., Dong F., Zhang W.-B., Wang Z.-T. Andrograpanin, a compound isolated from anti-inflammatory traditional Chinese medicine Andrographis paniculata, enhances chemokine SDF-1α-induced leukocytes chemotaxis. Journal of Cellular Biochemistry. 2005;95(5):970–978. doi: 10.1002/jcb.20464. [DOI] [PubMed] [Google Scholar]
- 136.Liu J., Wang Z.-T., Ge B.-X. Andrograpanin, isolated from Andrographis paniculata, exhibits anti-inflammatory property in lipopolysaccharide-induced macrophage cells through down-regulating the p38 MAPKs signaling pathways. International Immunopharmacology. 2008;8(7):951–958. doi: 10.1016/j.intimp.2007.12.014. [DOI] [PubMed] [Google Scholar]
- 137.Lala S., Nandy A. K., Mahato S. B., Basu M. K. Delivery in vivo of 14-deoxy-11-oxoandrographolide, an antileishmanial agent, by different drug carriers. Indian Journal of Biochemistry and Biophysics. 2003;40(3):169–174. [PubMed] [Google Scholar]
- 138.Chen L., Zhu H., Wang R., Zhou K., Jing Y., Qiu F. ent-labdane diterpenoid lactone stereoisomers from Andrographis paniculata . Journal of Natural Products. 2008;71(5):852–855. doi: 10.1021/np0704452. [DOI] [PubMed] [Google Scholar]
- 139.Wang G.-C., Wang Y., Williams I. D., et al. Andrographolactone, a unique diterpene from Andrographis paniculata . Tetrahedron Letters. 2009;50(34):4824–4826. doi: 10.1016/j.tetlet.2009.05.097. [DOI] [Google Scholar]
- 140.Smith P. L., Maloney K. N., Pothen R. G., Clardy J., Clapham D. E. Bisandrographolide from Andrographis paniculata activates TRPV4 channels. The Journal of Biological Chemistry. 2006;281(40):29897–29904. doi: 10.1074/jbc.M605394200. [DOI] [PubMed] [Google Scholar]
- 141.Arifullah M., Namsa N. D., Mandal M., Chiruvella K. K., Vikrama P., Gopal G. R. Evaluation of anti-bacterial and anti-oxidant potential of andrographolide and echiodinin isolated from callus culture of Andrographis paniculata Nees. Asian Pacific Journal of Tropical Biomedicine. 2013;3(8):604–610. doi: 10.1016/S2221-1691(13)60123-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Chen L.-X., He H., Xia G.-Y., Zhou K.-L., Qiu F. A new flavonoid from the aerial parts of Andrographis paniculata . Natural Product Research. 2014;28(3):138–143. doi: 10.1080/14786419.2013.856907. [DOI] [PubMed] [Google Scholar]
- 143.Dua V. K., Ojha V. P., Roy R., et al. Anti-malarial activity of some xanthones isolated from the roots of Andrographis paniculata . Journal of Ethnopharmacology. 2004;95(2-3):247–251. doi: 10.1016/j.jep.2004.07.008. [DOI] [PubMed] [Google Scholar]
- 144.Chen L.-X., Qiu F., Wei H., Qu G.-X., Yao X.-S. Nine new ent-labdane diterpenoids from the aerial parts of Andrographis paniculata . Helvetica Chimica Acta. 2006;89(11):2654–2664. doi: 10.1002/hlca.200690237. [DOI] [Google Scholar]
- 145.Fujita T., Fujitani R., Takeda Y., et al. On the diterpenoids of Andrographis paniculata: X-ray crystallographic analysis of andrographolide and structure determination of new minor diterpenoids. Chemical and Pharmaceutical Bulletin. 1984;32(6):2117–2125. [Google Scholar]
- 146.Pramanick S., Banerjee S., Achari B., et al. Andropanolide and isoandrographolide, minor diterpenoids from Andrographis paniculata: structure and X-ray crystallographic analysis. Journal of Natural Products. 2006;69(3):403–405. doi: 10.1021/np050211n. [DOI] [PubMed] [Google Scholar]
- 147.Rao Y. K., Vimalamma G., Rao C. V., Tzeng Y.-M. Flavonoids and andrographolides from Andrographis paniculata . Phytochemistry. 2004;65(16):2317–2321. doi: 10.1016/j.phytochem.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 148.Reddy M. K., Reddy M. V. B., Gunasekar D., Murthy M. M., Caux C., Bodo B. A flavone and an unusual 23-carbon terpenoid from Andrographis paniculata . Phytochemistry. 2003;62(8):1271–1275. doi: 10.1016/S0031-9422(03)00051-7. [DOI] [PubMed] [Google Scholar]
- 149.Seth S. K., Banerjee S., Kar T. Crystal structure and DFT calculations of andrographiside. Journal of Molecular Structure. 2010;965(1–3):45–49. doi: 10.1016/j.molstruc.2009.11.036. [DOI] [Google Scholar]
- 150.Xu C., Chou G. X., Wang Z. T. A new diterpene from the leaves of Andrographis paniculata Nees. Fitoterapia. 2010;81(6):610–613. doi: 10.1016/j.fitote.2010.03.003. [DOI] [PubMed] [Google Scholar]
- 151.Zhou K.-L., Chen L.-X., Zhuang Y.-L., Wang N.-L., Yao X.-S., Qiu F. Two new ent-labdane diterpenoid glycosides from the aerial parts of Andrographis paniculata . Journal of Asian Natural Products Research. 2008;10(10):939–943. doi: 10.1080/10286020802217432. [DOI] [PubMed] [Google Scholar]
- 152.Zou Q. Y., Li N., Dan C., Deng W. L., Peng S. L., Ding L. S. A new ent-labdane diterpenoid from Andrographis paniculata . Chinese Chemical Letters. 2010;21(9):1091–1093. doi: 10.1016/j.cclet.2010.05.002. [DOI] [Google Scholar]
- 153.Bhan M. K., Dhar A. K., Khan S., Lattoo S. K., Gupta K. K., Choudhary D. K. Screening and optimization of Andrographis paniculata (Burm.f.) Nees for total andrographolide content, yield and its components. Scientia Horticulturae. 2006;107(4):386–391. doi: 10.1016/j.scienta.2005.09.001. [DOI] [Google Scholar]
- 154.Nemade S., Mohod N., Wankhade S., Paturde J. Effect of planting and harvesting dates on yield and quality of kalmegh (Andrographis paniculata) Journal of Medicinal and Aromatic Plant Sciences. 2003;25(4):981–983. [Google Scholar]
- 155.Singh M., Singh A., Tripathi R., et al. Growth behavior, biomass and diterpenoid lactones production in Kalmegh (Andrographis paniculata Nees.) strains at different population densities. Agricultural Journal. 2011;6(3):115–118. [Google Scholar]
- 156.Parashar R., Upadhyay A., Singh J., Diwedi S. K., Khan N. A. Morpho-physiological evaluation of Andrographis paniculata at different growth stages. World Journal of Agricultural Sciences. 2011;7(2):124–127. [Google Scholar]
- 157.Talei D., Valdiani A., Abdullah M. P., Hassan S. A. A rapid and effective method for dormancy breakage and germination of king of bitters (Andrographis paniculata Nees.) seeds. Maydica. 2012;57(2):98–105. [Google Scholar]
- 158.Lattoo S. K., Dhar R. S., Khan S., et al. Comparative analysis of genetic diversity using molecular and morphometric markers in Andrographis paniculata (Burm. f.) Nees. Genetic Resources and Crop Evolution. 2008;55(1):33–43. doi: 10.1007/s10722-007-9212-y. [DOI] [Google Scholar]
- 159.Kumar B., Verma S. K., Singh H. P. Effect of temperature on seed germination parameters in Kalmegh (Andrographis paniculata Wall. ex Nees.) Industrial Crops and Products. 2011;34(1):1241–1244. doi: 10.1016/j.indcrop.2011.04.008. [DOI] [Google Scholar]
- 160.Kumar R. N., Chakraborty S., Nirmal K. Methods to break seed dormancy of Andrographis paniculata (Burm. f. Nees): an important medicinal herb of tropical Asia. Asian Journal of Experimental Biological Sciences. 2011;2(1):143–146. [Google Scholar]
- 161.Kataky A., Handique P. Micropropagation and screening of antioxidant potential of Andrographis paniculata (Burm. f) Nees. Journal of Hill Agriculture. 2010;1(1):13–18. [Google Scholar]
- 162.Martin K. P. Plant regeneration protocol of medicinally important Andrographis paniculata (Burm. f.) Wallich ex Nees via somatic embryogenesis. In Vitro Cellular and Developmental Biology-Plant. 2004;40(2):204–209. doi: 10.1079/IVP2003520. [DOI] [Google Scholar]
- 163.Purkayastha J., Sugla T., Paul A., Solleti S., Sahoo L. Rapid in vitro multiplication and plant regeneration from nodal explants of Andrographis paniculata: a valuable medicinal plant. In Vitro Cellular & Developmental Biology—Plant. 2008;44(5):442–447. doi: 10.1007/s11627-008-9156-8. [DOI] [Google Scholar]
- 164.Dandin V. S., Murthy H. N. Regeneration of Andrographis paniculata Nees: analysis of genetic fidelity and andrographolide content in micropropagated plants. African Journal of Biotechnology. 2012;11(61):12464–12471. [Google Scholar]
- 165.Gandi S., Rao K., Chodisetti B., Giri A. Elicitation of andrographolide in the suspension cultures of Andrographis paniculata . Applied Biochemistry and Biotechnology. 2012;168(7):1729–1738. doi: 10.1007/s12010-012-9892-4. [DOI] [PubMed] [Google Scholar]
- 166.Bowes B. The fine structure of wall modifications and associated structures in callus tissue of Andrographis paniculata Nees. New Phytologist. 1969;68(3):619–626. [Google Scholar]
- 167.Behera P. R., Nayak P., Barik D. P., Rautray T. R., Thirunavoukkarasu M., Chand P. K. ED-XRF spectrometric analysis of comparative elemental composition of in vivo and in vitro roots of Andrographis paniculata (Burm.f.) Wall. ex Nees-a multi-medicinal herb. Applied Radiation and Isotopes. 2010;68(12):2229–2236. doi: 10.1016/j.apradiso.2010.06.019. [DOI] [PubMed] [Google Scholar]
- 168.Jalal M. A. F., Overton K. H., Rycroft D. S. Formation of three new flavones by differentiating callus cultures of Andrographis paniculata . Phytochemistry. 1979;18(1):149–151. doi: 10.1016/S0031-9422(00)90934-8. [DOI] [Google Scholar]
- 169.Deng W. Outline of current clinical and pharmacological research on Andrographis paniculata in China. Newsletters of Chinese Herbal Medicine. 1978;10:27–31. [Google Scholar]
- 170.Sareer O., Ahad A., Umar S. Prophylactic and lenitive effects of Andrographis paniculata against common human ailments: an exhaustive and comprehensive reappraisal. Journal of Pharmaceutical Research and Opinion. 2012;2(10):138–162. [Google Scholar]
- 171.Xu C., Chou G.-X., Wang C.-H., Wang Z.-T. Rare noriridoids from the roots of Andrographis paniculata . Phytochemistry. 2012;77:275–279. doi: 10.1016/j.phytochem.2011.12.020. [DOI] [PubMed] [Google Scholar]
- 172.Zhang X.-Q., Wang G.-C., Ye W.-C., Li Q., Zhou G.-X., Yao X.-S. New diterpenoids from Andrographis paniculata (Burm. f.) Nees. Journal of Integrative Plant Biology. 2006;48(9):1122–1125. doi: 10.1111/j.1744-7909.2006.00308.x. [DOI] [Google Scholar]
- 173.Hancke J., Burgos R., Caceres D., Wikman G. A double-blind study with a new monodrug Kan Jang: decrease of symptoms and improvement in the recovery from common colds. Phytotherapy Research. 1995;9(8):559–562. doi: 10.1002/ptr.2650090804. [DOI] [Google Scholar]
- 174.Cáceres D. D., Hancke J. L., Burgos R. A., Wikman G. K. Prevention of common colds with Andrographis paniculata dried extract. A pilot double blind trial. Phytomedicine. 1997;4(2):101–104. doi: 10.1016/S0944-7113(97)80051-7. [DOI] [PubMed] [Google Scholar]
- 175.Melchior J., Palm S., Wikman G. Controlled clinical study of standardized Andrographis paniculata extract in common cold-a pilot trial. Phytomedicine. 1997;3(4):315–318. doi: 10.1016/S0944-7113(97)80002-5. [DOI] [PubMed] [Google Scholar]
- 176.Abu-Ghefreh A. A., Canatan H., Ezeamuzie C. I. In vitro and in vivo anti-inflammatory effects of andrographolide. International Immunopharmacology. 2009;9(3):313–318. doi: 10.1016/j.intimp.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 177.Habtemariam S. Andrographolide inhibits the tumour necrosis factor-α-induced upregulation of ICAM-1 expression and endothelial-monocyte adhesion. Phytotherapy Research. 1998;12(1):37–40. [Google Scholar]
- 178.Liu S. F., Malik A. B. NF-κB activation as a pathological mechanism of septic shock and inflammation. The American Journal of Physiology—Lung Cellular and Molecular Physiology. 2006;290(4):L622–L645. doi: 10.1152/ajplung.00477.2005. [DOI] [PubMed] [Google Scholar]
- 179.Marrache A. M., Gobeil F., Jr., Bernier S. G., et al. Proinflammatory gene induction by platelet-activating factor mediated via its cognate nuclear receptor. The Journal of Immunology. 2002;169(11):6474–6481. doi: 10.4049/jimmunol.169.11.6474. [DOI] [PubMed] [Google Scholar]
- 180.Pocock J., Gómez-Guerrero C., Harendza S., et al. Differential activation of NF-κB, AP-1, and C/EBP in endotoxin-tolerant rats: mechanisms for in vivo regulation of glomerular RANTES/CCL5 expression. The Journal of Immunology. 2003;170(12):6280–6291. doi: 10.4049/jimmunol.170.12.6280. [DOI] [PubMed] [Google Scholar]
- 181.Madav S., Tandan S. K., Lal J., Tripathi H. C. Anti-inflammatory activity of andrographolide. Fitoterapia. 1996;67(5):452–458. [Google Scholar]
- 182.Zhang B., Yan L., Zhou P., et al. CHP1002, a novel andrographolide derivative, inhibits pro-inflammatory inducible nitric oxide synthase and cyclooxygenase-2 expressions in RAW264.7 macrophages via up-regulation of heme oxygenase-1 expression. International Immunopharmacology. 2013;15(2):289–295. doi: 10.1016/j.intimp.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 183.Liu W., Guo W., Guo L., et al. Andrographolide sulfonate ameliorates experimental colitis in mice by inhibiting Th1/Th17 response. International Immunopharmacology. 2014;20(2):337–345. doi: 10.1016/j.intimp.2014.03.015. [DOI] [PubMed] [Google Scholar]
- 184.Kajaria D., Ranjana J., Tripathi Y. B., Tiwari S. In vitro a amylase and glucosidase inhibitory effect of ethanolic extract of antiasthmatic drug-Shirishadi. Journal of Advanced Pharmaceutical Technology and Research. 2013;4(4):206–209. doi: 10.4103/2231-4040.121415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Reaven G. The metabolic syndrome or the insulin resistance syndrome? Different names, different concepts, and different goals. Endocrinology and Metabolism Clinics of North America. 2004;33(2):283–303. doi: 10.1016/j.ecl.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 186.Chao W.-W., Lin B.-F. Isolation and identification of bioactive compounds in Andrographis paniculata (Chuanxinlian) Chinese Medicine. 2010;5, article 17:1–15. doi: 10.1186/1749-8546-5-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Augustine A. W., Narasimhan A., Vishwanathan M., Karundevi B. Evaluation of antidiabetic property of Andrographis paniculata powder in high fat and sucrose-induced type-2 diabetic adult male rat. Asian Pacific Journal of Tropical Disease. 2014;4(supplement 1):S140–S147. doi: 10.1016/S2222-1808(14)60429-1. [DOI] [Google Scholar]
- 188.Gupta R., Bajpai K. G., Johri S., Saxena A. M. An overview of Indian novel traditional medicinal plants with anti-diabetic potentials. African Journal of Traditional, Complementary and Alternative Medicines. 2008;5(1):1–17. [PMC free article] [PubMed] [Google Scholar]
- 189.Zhang Z., Jiang J., Yu P., Zeng X., Larrick J. W., Wang Y. Hypoglycemic and beta cell protective effects of andrographolide analogue for diabetes treatment. Journal of Translational Medicine. 2009;7, article 62 doi: 10.1186/1479-5876-7-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Nugroho A. E., Rais I. R., Setiawan I., et al. Pancreatic effect of andrographolide isolated from Andrographis paniculata (Burm. f.) Nees. Pakistan Journal of Biological Sciences. 2014;17(1):22–31. doi: 10.3923/pjbs.2014.22.31. [DOI] [PubMed] [Google Scholar]
- 191.Zhang C., Gui L., Xu Y., Wu T., Liu D. Preventive effects of andrographolide on the development of diabetes in autoimmune diabetic NOD mice by inducing immune tolerance. International Immunopharmacology. 2013;16(4):451–456. doi: 10.1016/j.intimp.2013.05.002. [DOI] [PubMed] [Google Scholar]
- 192.Nugroho A. E., Kusumaramdani G., Widyaninggar A., Anggoro D. P., Pramono S. Antidiabetic effect of combinations of n-hexane insoluble fraction of ethanolic extract of Andrographis paniculata with other traditional medicines. International Food Research Journal. 2014;21(2):785–789. [Google Scholar]
- 193.Verma V. K., Sarwa K. K., Kumar A., Zaman M. Comparison of hepatoprotective activity of Swertia chirayita and Andrographis paniculata plant of North-East India against CCl4 induced hepatotoxic rats. Journal of Pharmacy Research. 2013;7(7):647–653. doi: 10.1016/j.jopr.2013.07.008. [DOI] [Google Scholar]
- 194.Leelarasamee A., Trakulsomboon S., Sittisomwong N. Undetectable anti-bacterial activity of Andrographis paniculata (Burma) wall. ex ness. Journal of the Medical Association of Thailand. 1990;73(6):299–304. [PubMed] [Google Scholar]
- 195.Nakanishi K., Sasaki S., Kiang A. K., et al. Phytochemical survey of Malaysian plants preliminary chemical and pharmacological screening. Chemical and Pharmaceutical Bulletin. 1965;13(7):882–890. doi: 10.1248/cpb.13.882. [DOI] [PubMed] [Google Scholar]
- 196.Mishra U. S., Mishra A., Kumari R., Murthy P. N., Naik B. S. Antibacterial activity of ethanol extract of Andrographis paniculata . Indian Journal of Pharmaceutical Sciences. 2009;71(4):436–438. doi: 10.4103/0250-474X.57294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Singha P. K., Roy S., Dey S. Antimicrobial activity of Andrographis paniculata . Fitoterapia. 2003;74(7-8):692–694. doi: 10.1016/S0367-326X(03)00159-X. [DOI] [PubMed] [Google Scholar]
- 198.Fabricant D. S., Farnsworth N. R. The value of plants used in traditional medicine for drug discovery. Environmental Health Perspectives. 2001;109(1):69–75. doi: 10.1289/ehp.01109s169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Sule A., Ahmed Q. U., Samah O., Omar M. Screening for antibacterial activity of Andrographis paniculata used in Malaysian folkloric medicine: a possible alternative for the treatment of skin infections. Ethnobotanical Leaflets. 2010;2010(4):445–456. [Google Scholar]
- 200.Sule A., Ahmed Q. U., Samah O. A., Omar M. N. Bacteriostatic and bactericidal activities of Andrographis paniculata extracts on skin disease causing pathogenic bacteria. Journal of Medicinal Plants Research. 2011;5(1):7–14. [Google Scholar]
- 201.Tang L. I. C., Ling A. P. K., Koh R. Y., Chye S. M., Voon K. G. L. Screening of anti-dengue activity in methanolic extracts of medicinal plants. BMC Complementary and Alternative Medicine. 2012;12, article 3 doi: 10.1186/1472-6882-12-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Fangkham S., Ekalaksananan T., Aromdee C., et al. The effect of andrographolide on Human papillomavirus type 16 (HPV16) positive cervical cancer cells (SiHa) International Journal of Infectious Diseases. 2012;16(supplement 1):p. e80. [Google Scholar]
- 203.Aromdee C., Suebsasana S., Ekalaksananan T., Pientong C., Thongchai S. Stage of action of naturally occurring andrographolides and their semisynthetic analogues against herpes simplex virus type 1 in vitro. Planta Medica. 2011;77(9):915–921. doi: 10.1055/s-0030-1250659. [DOI] [PubMed] [Google Scholar]
- 204.Xu H. X., Wan M., Loh B. N., Kon O. L., Chow P. W., Sim K. Y. Screening of traditional medicines for their inhibitory activity against HIV-1 protease. Phytotherapy Research. 1996;10(3):207–210. doi: 10.1002/(SICI)1099-1573(199605)10:3<207::AID-PTR812>3.0.CO;2-U. [DOI] [Google Scholar]
- 205.Holt S., Comac L. Miracle Herbs: How Herbs Combine with Modern Medicine to Treat Cancer; Heart Disease, AIDS, and More. Caro, Mich, USA: Kensington; 1998. [Google Scholar]
- 206.Zaridah M. Z., Idid S. Z., Wan Omar A., Khozirah S. In vitro antifilarial effects of three plant species against adult worms of subperiodic Brugia malayi . Journal of Ethnopharmacology. 2001;78(1):79–84. doi: 10.1016/S0378-8741(01)00286-0. [DOI] [PubMed] [Google Scholar]
- 207.Padma Y., Narasimhudu C. L., Devi S., Natha N. M. B., Naga R. B., Philip G. H. In vitro anthelmintic activity of Andrographis paniculata (burm.f.) nees. International Journal of Pharmaceutical Research and Developement. 2011;3(3):202–205. [Google Scholar]
- 208.Vojdani A., Erde J. Regulatory T cells, a potent immunoregulatory target for CAM researchers: modulating tumor immunity, autoimmunity and alloreactive immunity (III) Evidence-Based Complementary and Alternative Medicine. 2006;3(3):309–316. doi: 10.1093/ecam/nel047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Aditya V. S. P. K., Kumar N. L., Mokkapati A. Evaluation of in vitro cytotoxicity of Andrographis paniculata, Duranta serratifolia and Albizzia lebbeck whole plant extracts by MTT assay against MCF-7 and HT-29 cell lines. Current Research in Microbiology and Biotechnology. 2014;2(2):351–353. [Google Scholar]
- 210.Geethangili M., Rao Y. K., Fang S.-H., Tzeng Y.-M. Cytotoxic constituents from Andrographis paniculata induce cell cycle arrest in Jurkat cells. Phytotherapy Research. 2008;22(10):1336–1341. doi: 10.1002/ptr.2493. [DOI] [PubMed] [Google Scholar]
- 211.Lin H.-H., Shi M.-D., Tseng H.-C., Chen J.-H. Andrographolide sensitizes the cytotoxicity of human colorectal carcinoma cells toward cisplatin via enhancing apoptosis pathways in vitro and in vivo. Toxicological Sciences. 2014;139(1):108–120. doi: 10.1093/toxsci/kfu032. [DOI] [PubMed] [Google Scholar]
- 212.Satyanarayana C., Deevi D. S., Rajagopalan R., Srinivas N., Rajagopal S. DRF 3188 a novel semi-synthetic analog of andrographolide: cellular response to MCF 7 breast cancer cells. BMC Cancer. 2004;4, article 26 doi: 10.1186/1471-2407-4-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Jada S. R., Subur G. S., Matthews C., et al. Semisynthesis and in vitro anticancer activities of andrographolide analogues. Phytochemistry. 2007;68(6):904–912. doi: 10.1016/j.phytochem.2006.11.031. [DOI] [PubMed] [Google Scholar]
- 214.Amroyan E., Gabrielian E., Panossian A., Wikman G., Wagner H. Inhibitory effect of andrographolide from Andrographis paniculata on PAF-induced platelet aggregation. Phytomedicine. 1999;6(1):27–31. doi: 10.1016/S0944-7113(99)80031-2. [DOI] [PubMed] [Google Scholar]
- 215.Bukoye O., Musbau A. Immune modulation potentials of aqueous extract of Andrographis paniculata leaves in male rat. Researcher. 2011;3(1):48–57. [Google Scholar]
- 216.Sheeja K., Kuttan G. Andrographis paniculata downregulates proinflammatory cytokine production and augments cell mediated immune response in metastatic tumor-bearing mice. Asian Pacific Journal of Cancer Prevention. 2010;11(3):723–729. [PubMed] [Google Scholar]
- 217.Zhao H., Fang W. Protective effects of Andrographis paniculata Nees on post-infarction myocardium in experimental dogs. Journal of Tongji Medical University. 1990;10(4):212–217. doi: 10.1007/BF02887933. [DOI] [PubMed] [Google Scholar]
- 218.Zhao H. Y., Fang W. Y. Antithrombotic effects of Andrographis paniculata nees in preventing myocardial infarction. Chinese Medical Journal. 1991;104(9):770–775. [PubMed] [Google Scholar]
- 219.Zhi-ling G., Hua-yue Z., Xin-hua Z. The effect of Andrographis Paniculata Nees (APN) in alleviating the myocardial ischemic reperfusion injury. Journal of Tongji Medical University. 1994;14(1):49–51. doi: 10.1007/BF02888059. [DOI] [PubMed] [Google Scholar]
- 220.Zhi-ling G., Hua-yue Z., Xin-hua Z. An experimental study of the mechanism of Andrographis paniculata nees (APN) in alleviating the Ca2+-overloading in the process of myocardial ischemie reperfusion. Journal of Tongji Medical University. 1995;15(4):205–208. doi: 10.1007/BF02887945. [DOI] [PubMed] [Google Scholar]
- 221.Guo Z.-L., Zhao H., Fu L. Protective effects of API0134 on myocardial ischemia and reperfusion injury. Journal of Tongji Medical University. 1996;16(4):193–197. doi: 10.1007/BF02888103. [DOI] [PubMed] [Google Scholar]
- 222.Wang H. W., Zhao H. Y., Xiang S. Q. Effects of Andrographis paniculata component on nitric oxide, endothelin and lipid peroxidation in experimental atherosclerotic rabbits. Chinese Journal of Integrated Traditional and Western Medicine. 1997;17(9):547–549. [PubMed] [Google Scholar]
- 223.Wang D. W., Zhao H. Y. Prevention of atherosclerotic arterial stenosis and restenosis after angioplasty with Andrographis paniculata nees and fish oil. Experimental studies of effects and mechanisms. Chinese Medical Journal. 1994;107(6):464–470. [PubMed] [Google Scholar]
- 224.Zhang C., Kuroyangi M., Tan B. K. H. Cardiovascular activity of 14-deoxy-11,12-didehydroandrographolide in the anaesthetised rat and isolated right atria. Pharmacological Research. 1998;38(6):413–418. doi: 10.1006/phrs.1998.0373. [DOI] [PubMed] [Google Scholar]
- 225.Yang T., Shi H.-X., Wang Z.-T., Wang C.-H. Hypolipidemic effects of andrographolide and neoandrographolide in mice and rats. Phytotherapy Research. 2013;27(4):618–623. doi: 10.1002/ptr.4771. [DOI] [PubMed] [Google Scholar]
- 226.Zhang C. Y., Tan B. Hypotensive activity of aqueous extract of Andrographis paniculata in rats. Clinical and Experimental Pharmacology and Physiology. 1996;23(8):675–678. doi: 10.1111/j.1440-1681.1996.tb01756.x. [DOI] [PubMed] [Google Scholar]
- 227.Zhang Y., Tang J., Zhang Y. Study of Andrographis paniculata extracts on antiplatelet aggregation and release reaction and its mechanism. Zhongguo Zhong Xi Yi Jie He Za Zhi. 1994;14(1):28–30. [PubMed] [Google Scholar]
- 228.Al-Attar A. M. Hypolipidemic effects of coenzyme Q10 in experimentally induced hypercholesterolemic model in female rats. The American Journal of Pharmacology and Toxicology. 2010;5(1):14–23. doi: 10.3844/ajptsp.2010.14.23. [DOI] [Google Scholar]
- 229.Briel M., Ferreira-Gonzalez I., You J. J., et al. Association between change in high density lipoprotein cholesterol and cardiovascular disease morbidity and mortality: systematic review and meta-regression analysis. British Medical Journal. 2009;338, article b92 doi: 10.1136/bmj.b92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Stamler J., Daviglus M. L., Garside D. B., Dyer A. R., Greenland P., Neaton J. D. Relationship of baseline serum cholesterol levels in 3 large cohorts of younger men to long-term coronary, cardiovascular, and all-cause mortality and to longevity. Journal of the American Medical Association. 2000;284(3):311–318. doi: 10.1001/jama.284.3.311. [DOI] [PubMed] [Google Scholar]
- 231.Chen J.-H., Hsiao G., Lee A.-R., Wu C.-C., Yen M.-H. Andrographolide suppresses endothelial cell apoptosis via activation of phosphatidyl inositol-3-kinase/Akt pathway. Biochemical Pharmacology. 2004;67(7):1337–1345. doi: 10.1016/j.bcp.2003.12.015. [DOI] [PubMed] [Google Scholar]
- 232.Zoha M. S., Hussain A. H., Choudhury S. A. Antifertility effect of andrographis paniculata in mice. Bangladesh Medical Research Council Bulletin. 1989;15(1):34–37. [PubMed] [Google Scholar]
- 233.Akbarsha M. A., Manivannan B., Hamid K. S., Vijayan B. Antifertility effect of Andrographis paniculata (Nees) in male albino rat. Indian Journal of Experimental Biology. 1990;28(5):421–426. [PubMed] [Google Scholar]
- 234.Janarthanan S. Antifertility effects of andrographolide in rats. Journal of Ecobiology. 1990;2(4):325–329. [Google Scholar]
- 235.Akbarsha M. A., Manivannan B. Biochemical changes in the testis and male accessory organs of albino rats on treatment with Andrographis paniculata Nees. Indian Journal of Comparative Animal Physiology. 1993;11(2):103–108. [Google Scholar]
- 236.Kamal R., Gupta R. S., Lohiya N. K. Plants for male fertility regulation. Phytotherapy Research. 2003;17(6):579–590. doi: 10.1002/ptr.1320. [DOI] [PubMed] [Google Scholar]
- 237.Burgos R. A., Caballero E. E., Sánchez N. S., Schroeder R. A., Wikman G. K., Hancke J. L. Testicular toxicity assesment of Andrographis paniculata dried extract in rats. Journal of Ethnopharmacology. 1997;58(3):219–224. doi: 10.1016/S0378-8741(97)00099-8. [DOI] [PubMed] [Google Scholar]
- 238.Mkrtchyan A., Panosyan V., Panossian A., Wikman G., Wagner H. A phase I clinical study of Andrographis paniculata fixed combination Kan Jang versus ginseng and valerian on the semen quality of healthy male subjects. Phytomedicine. 2005;12(6-7):403–409. doi: 10.1016/j.phymed.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 239.Sattayasai J., Srisuwan S., Arkaravichien T., Aromdee C. Effects of andrographolide on sexual functions, vascular reactivity and serum testosterone level in rodents. Food and Chemical Toxicology. 2010;48(7):1934–1938. doi: 10.1016/j.fct.2010.04.037. [DOI] [PubMed] [Google Scholar]
- 240.Allan J. J., Pore. M. P., Deepak M., Murali B., Mayachari. A. S., Agarwal A. Reproductive and fertility effects of an extract of andrographis paniculata in male wistar rats. International Journal of Toxicology. 2009;28(4):308–317. doi: 10.1177/1091581809339631. [DOI] [PubMed] [Google Scholar]
- 241.Panossian A., Kochikian A., Gabrielian E., et al. Effect of Andrographis paniculata extract on progesterone in blood plasma of pregnant rats. Phytomedicine. 1999;6(3):157–162. doi: 10.1016/S0944-7113(99)80003-8. [DOI] [PubMed] [Google Scholar]
- 242.Balu S., Boopathi C. A., Elango V. Antipyretic activities of some species of Andrographis wall. Ancient Science of Life. 1993;12(3-4):399–402. [PMC free article] [PubMed] [Google Scholar]
- 243.Jarukamjorn K., Don-in K., Makejaruskul C., et al. Impact of Andrographis paniculata crude extract on mouse hepatic cytochrome P450 enzymes. Journal of Ethnopharmacology. 2006;105(3):464–467. doi: 10.1016/j.jep.2005.11.024. [DOI] [PubMed] [Google Scholar]
- 244.Chandrasekaran C. V., Thiyagarajan P., Sundarajan K., et al. Evaluation of the genotoxic potential and acute oral toxicity of standardized extract of Andrographis paniculata (KalmCold) Food and Chemical Toxicology. 2009;47(8):1892–1902. doi: 10.1016/j.fct.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 245.Sharifuddin Y., Parry E. M., Parry J. M. The genotoxicity and cytotoxicity assessments of andrographolide in vitro. Food and Chemical Toxicology. 2012;50(5):1393–1398. doi: 10.1016/j.fct.2012.01.039. [DOI] [PubMed] [Google Scholar]
- 246.Kligler B., Ulbricht C., Basch E., et al. Andrographis paniculata for the treatment of upper respiratory infection: a systematic review by the natural standard research collaboration. Explore: The Journal of Science and Healing. 2006;2(1):25–29. doi: 10.1016/j.explore.2005.08.008. [DOI] [PubMed] [Google Scholar]
- 247.Smith R. M. Before the injection—modern methods of sample preparation for separation techniques. Journal of Chromatography A. 2003;1000(1-2):3–27. doi: 10.1016/S0021-9673(03)00511-9. [DOI] [PubMed] [Google Scholar]
- 248.Azmir J., Zaidul I. S. M., Rahman M. M., et al. Techniques for extraction of bioactive compounds from plant materials: a review. Journal of Food Engineering. 2013;117(4):426–436. doi: 10.1016/j.jfoodeng.2013.01.014. [DOI] [Google Scholar]
- 249.Li B. B., Smith B., Hossain M. M. Extraction of phenolics from citrus peels: II. Enzyme-assisted extraction method. Separation and Purification Technology. 2006;48(2):189–196. doi: 10.1016/j.seppur.2005.07.019. [DOI] [Google Scholar]
- 250.Howard L., Pandjaitan N. Pressurized liquid extraction of flavonoids from spinach. Journal of Food Science. 2008;73(3):C151–C157. doi: 10.1111/j.1750-3841.2007.00658.x. [DOI] [PubMed] [Google Scholar]
- 251.Hossain M. S., Ismail N. A., Phang I. C. Effect of salinity stress on morphology and physiology of Andtrographis paniculata . Proceedings of the International Association for Plant Biotechnology Congress; August 2014; Melbourne, Australia. Melbourne Convention and Exhibition Centre; p. p. 204. [Google Scholar]
- 252.Verpoorte R., van der Hejden R., ten Hoopen H. J. G., Memelink J. Metabolic engineering of plant secondary metabolite pathways for the production of fine chemicals. Biotechnology Letters. 1999;21(6):467–479. doi: 10.1023/A:1005502632053. [DOI] [Google Scholar]
- 253.Wahid A., Ghazanfar A. Possible involvement of some secondary metabolites in salt tolerance of sugarcane. Journal of Plant Physiology. 2006;163(7):723–730. doi: 10.1016/j.jplph.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 254.Olano C., Lombó F., Méndez C., Salas J. A. Improving production of bioactive secondary metabolites in actinomycetes by metabolic engineering. Metabolic Engineering. 2008;10(5):281–292. doi: 10.1016/j.ymben.2008.07.001. [DOI] [PubMed] [Google Scholar]
- 255.Yan Y.-P., Wang Z.-Z. Genetic transformation of the medicinal plant Salvia miltiorrhiza by Agrobacterium tumefaciens-mediated method. Plant Cell, Tissue and Organ Culture. 2007;88(2):175–184. doi: 10.1007/s11240-006-9187-y. [DOI] [Google Scholar]
- 256.Talei D., Valdiani A., Maziah M., Sagineedu S. R., Saad M. S. Analysis of the anticancer phytochemicals in Andrographis paniculata Nees. under salinity stress. BioMed Research International. 2013;2013:11. doi: 10.1155/2013/319047.319047 [DOI] [PMC free article] [PubMed] [Google Scholar]