Abstract
Objective
Nonspecific musculoskeletal pain can be difficult to manage in acute rehabilitation unit (ARU) patients. We investigated whether vitamin D status is a potential modifiable risk factor for nonspecific musculoskeletal pain in ARU patients.
Materials and Methods
This cross-sectional study focused on 414 adults from an inpatient ARU in Mission Viejo, California, between July 2011 and June 2012. On ARU admission, all patients had serum 25-hydroxyvitamin D (25(OH)D) levels measured and were assessed for nonspecific musculoskeletal pain. We performed multivariable logistic regression to test the association of serum 25(OH)D level with nonspecific musculoskeletal pain while adjusting for clinically relevant covariates.
Results
Among these 414 patients, mean (SD) 25(OH)D level was 29 (12) ng/mL, and 30% had nonspecific musculoskeletal pain. After adjustment for age, sex, race, body mass index, Functional Independence Measure score, Deyo-Charlson Comorbidity Index, fractures, steroid use, history of osteoporosis/osteomalacia, and patient type (orthopedic, cardiac, neurological, spinal cord injury, or traumatic brain injury), serum 25(OH)D level was inversely associated with nonspecific musculoskeletal pain (odds ratio [OR] per 10 ng/mL, 0.67; 95% confidence interval [CI], 0.48–0.82). When 25(OH)D level was dichotomized, patients with levels <20 ng/mL had higher odds of nonspecific musculoskeletal pain (OR, 2.33; 95% CI, 1.23–4.17) compared with patients with levels ≥20 ng/mL.
Conclusions
In adult patients, serum 25(OH)D level on admission to ARU was inversely associated with nonspecific musculoskeletal pain. These data support the need for randomized, controlled trials to test the role of vitamin D supplementation to improve nonspecific musculoskeletal pain in ARU patients.
Keywords: vitamin D, pain, musculoskeletal, acute rehabilitation
Clinical Relevancy Statement
Nonspecific musculoskeletal pain is highly prevalent in acute rehabilitation unit (ARU) patients. It can be difficult to manage using traditional pharmacological and nonpharmacological modalities. The study results suggest a potential role of vitamin D status in nonspecific musculoskeletal pain among ARU patients.
Introduction
Nonspecific musculoskeletal pain is common among inpatient acute rehabilitation unit (ARU) patients. Its prevalence varies greatly by the indication for admission, with values of 10%–70% having been reported.1–4 Nonspecific musculoskeletal pain is often difficult to manage since an identifiable pathology or anatomical basis for the pain cannot be found.5 Inadequately controlled pain not only is a problem in itself but also may exacerbate musculoskeletal weakness, depression, anxiety, and poor sleep quality—all major factors that influence the general well-being of ARU patients.6–9 Traditional pharmacological therapies include opioids and nonsteroidal anti-inflammatory drugs,10–12 which are of moderate benefit. Nontraditional pharmacological agents (such as calcium channel blockers, anticonvulsants, and muscle relaxants) have shown limited benefit.13–15 Furthermore, while nonpharmacological adjuncts such as acupuncture and massage therapy are thought to improve pain in ARU patients,16–18 evidence from adequately powered, randomized controlled trials (RCTs) is lacking.
Growing evidence suggests that vitamin D status may be a modifiable risk factor for various forms of musculoskeletal pain in ARU patients.19 Moreover, preliminary evidence from small clinical studies suggests that vitamin D supplementation improves pain in ambulatory patients with nonspecific musculoskeletal pain.20–22 Low serum 25-hydroxyvitamin D (25(OH)D) levels (the most widely recognized marker of total body vitamin D status23) are common in ARU patients.19 Indeed, 25(OH)D levels <20 ng/mL have been reported to affect as many as 49%–89% of ARU patients.24–26 Given the high prevalence of both low vitamin D status and inadequately controlled pain in ARU patients, our goal was to investigate whether serum 25(OH)D level is associated with nonspecific musculoskeletal pain in these potentially difficult-to-manage patients.
Materials and Methods
Study Cohort
We abstracted laboratory and administrative data from the electronic medical records of individuals admitted to a single, 21-bed ARU in Mission Viejo, California, over 1 year (July 2011 to June 2012). Mission Hospital is a 407-bed, short-term, acute care hospital. The ARU receives two-thirds of its referrals from Mission Hospital and the remaining one-third from other acute care hospitals and long-term care facilities from in and around Orange County. Given the retrospective nature of the study, a waiver of consent was granted by the local institutional review boards at Mission Hospital and Partners Healthcare.
We identified 427 adult admissions (age ≥18 years) over the 1-year study period. All patients were transferred directly from the intensive care unit (ICU) of a referring hospital or from a general ward after having received acute care services. We excluded all patients who did not have 25(OH)D levels measured on admission to the ARU (n = 1) and any readmissions during the study period (n = 12). The final study cohort was therefore composed of 414 patients.
Exposure of Interest and Comorbidities
The exposure of interest was serum 25(OH)D level obtained on admission to the ARU. For our analysis, we first considered 25(OH)D level as a continuous variable and then as a dichotomous variable (<20 ng/mL vs ≥20 ng/mL and <30 ng/mL vs ≥30 ng/mL). All cut points were adapted from existing national clinical guidelines.27,28 Serum 25(OH)D levels were determined by liquid chromatography–mass spectrometry29 in a Clinical Laboratory Improvement Amendments (CLIA)–certified laboratory (Quest Diagnostics, Inc, San Juan Capistrano, CA).
We used the International Classification of Diseases, Ninth Revision coding algorithms, which are well studied and validated,30 to derive the Deyo-Charlson Comorbidity Index to assess the burden of chronic illness in our study cohort. “Patient type” was determined by the primary admission diagnosis at the acute care facility before transfer to the ARU and was categorized as (1) orthopedic (eg, elective joint surgery, isolated traumatic long bone fractures), (2) cardiac (eg, cardiac surgery, status post–acute myocardial infarction), (3) neurological (eg, stroke, nontraumatic cerebral hemorrhage), (4) spinal cord injury, and (5) traumatic brain injury. Given the heterogeneity of vitamin D supplementation (eg, dose, frequency, and adherence) in the weeks before transfer to the ARU, we categorized ergocalciferol or cholecalciferol supplementation status as none, <1000 IU/d, or ≥1000 IU/d.
Outcome
The outcome assessed was the presence of nonspecific musculoskeletal pain following ARU admission, as defined by (1) complaints of persistent musculoskeletal pain, defined as >3 on the visual analog scale in the admitting ARU physician history and physical assessment, which persisted for >3 days and was not associated with a direct causative factor (eg, fracture, metastatic bone disease), or (2) a formal diagnosis of nonspecific musculoskeletal pain by a direct care provider trained in pain management. Individual medical records were reviewed to abstract information regarding pain assessments on admission to the ARU and for subsequent pain management interventions.
Statistical Analysis
Descriptive statistics were calculated for subjects with serum 25(OH)D levels <20 ng/mL vs those with levels ≥20 ng/mL. Continuous data were reported as means with standard deviations (SDs) or medians with interquartile ranges (IQRs). Comparison of characteristics was performed using t-test and Mann-Whitney analyses for normally distributed variables and for nonparametric variables, respectively. Categorical values were expressed as proportions and compared using χ2 tests. Locally weighted scatter plot smoothing (LOWESS) was used to graphically represent31,32 the relationship between 25(OH)D level and nonspecific musculoskeletal pain.
Since the presence of nonspecific musculoskeletal pain was defined as a dichotomous variable (present vs not present), logistic regression analyses were used to model the relationship between serum 25(OH)D levels and nonspecific musculoskeletal pain, while controlling for biologically plausible covariates. These covariates included age, sex, race, body mass index (BMI), Functional Independence Measure (FIM) score, Deyo-Charlson Comorbidity Index (DCCI), recent fractures, current or recent steroid use, documented clinical history of osteoporosis/osteomalacia (abstracted from individual intake comprehensive evaluation records), and patient type. Results are reported as odds ratios (ORs) with 95% confidence intervals (CIs).
On the basis of the existing literature related to the prevalence of nonspecific musculoskeletal pain in ARU patients1–4 and the growing recognition of suboptimal vitamin D status in these patients,6–9 we performed an a priori sample size calculation. We assumed that in patients with 25(OH)D levels <20 ng/mL, the prevalence of nonspecific musculoskeletal pain was 35% vs 15% in patients with 25(OH)D levels ≥20 ng/mL. To detect this difference with a power of 0.8 and α set at 0.05, we would require a minimum of 73 patients in each group. All analyses were performed in STATA 13.1 (StataCorp LP, College Station, TX). A 2-tailed P < .05 or 95% CI that did not span 1.00 was considered statistically significant.
Results
Baseline characteristics are shown in Table 1. The mean (SD) age was 67 (16) years. Most participants were male (53%) and white (86%). Mean (SD) BMI was 27 (6) kg/m2. The mean (SD) total FIM score and DCCI was 60 (13) and 3 (2), respectively. Mean (SD) serum 25(OH)D level at ARU admission was 29 (12) ng/mL. However, 2% of patients had 25(OH)D levels between 0 and 9.9 ng/mL, 16% between 10 and 19.9 ng/mL, 23% between 20 and 29.9 ng/mL, and 59% had levels of ≥30 ng/mL. Overall, 30% of patients met the study criteria for nonspecific musculoskeletal pain.
Table 1.
Demographic Factors and Baseline Clinical Information in Acute Rehabilitation Unit Patients According to Vitamin D Status (n = 414).
| Characteristic | 25(OH)D <20 ng/mL (n = 77) | 25(OH)D ≥20 ng/mL (n = 337) | P Value |
|---|---|---|---|
| Age, mean (SD), y | 65 (16) | 67 (16) | .32 |
| Sex, % | .12 | ||
| Female | 38 | 49 | |
| Male | 62 | 51 | |
| Race, % | .02 | ||
| Nonwhite | 25 | 12 | |
| White | 75 | 88 | |
| BMI, mean (SD), kg/m2 | 28 (6) | 26 (6) | .01 |
| DCCI, mean (SD) | 3 (2) | 2 (2) | <.001 |
| Total FIM, mean (SD) | 62 (12) | 60 (13) | <.001 |
| Fractures, % | .32 | ||
| No | 87 | 82 | |
| Yes | 13 | 18 | |
| Steroid use, % | .77 | ||
| No | 93 | 94 | |
| Yes | 7 | 6 | |
| Osteoporosis/osteomalacia, % | .65 | ||
| No | 90 | 88 | |
| Yes | 10 | 12 | |
| Vitamin D supplementation, % | <.001 | ||
| None | 99 | 83 | |
| <1000 IU | 1 | 15 | |
| ≥1000 IU | — | 2 | |
| Type of patient, % | .46 | ||
| Orthopedic | 30 | 29 | |
| Neurological | 48 | 50 | |
| Cardiac | 5 | 5 | |
| Spinal cord injury | — | 3 | |
| Traumatic brain injury | 17 | 13 | |
| Season of ARU admission, % | .48 | ||
| Summer | 54 | 59 | |
| Winter | 46 | 41 | |
| 25(OH)D, mean (SD), ng/mL | 15 (4) | 32 (11) | <.001 |
| NSMP, % | .02 | ||
| No | 61 | 83 | |
| Yes | 39 | 17 | |
| Medications, % | |||
| Acetaminophen | 90 | 96 | .10 |
| NSAIDs | — | — | — |
| Opioids | 92 | 77 | .003 |
| Other analgesics | — | — | — |
| Anticonvulsants | 6 | 10 | .30 |
| Antineoplastics | — | — | — |
| Antimicrobials | 5 | 3 | .47 |
| Antihypertensives | 4 | 1 | .17 |
| Antiretrovirals | — | — | — |
| HMG-CoA reductase inhibitors | 30 | 35 | .45 |
Significant P values (<.05) are shown in bold. ARU, acute rehabilitation unit; BMI, body mass index; DCCI, Deyo-Charlson Comorbidity Index; FIM, Functional Independence Measure; NSAIDs, nonsteroidal anti-inflammatory drugs; NSMP, nonspecific musculoskeletal pain; 25(OH)D, 25-hydroxyvitamin D; —, indicates <1%. Other analgesics = gabapentin, pregabalin, tricyclic antidepressants, tizanadine, cyclobenzaprine, baclofen. Anticonvulsants = phenytoin, carbamazepine, valproic acid. Antineoplastics = taxol, tamoxifen. Antimicrobials = clotrimazole, fluconazole, rifampin. Antihypertensives = nifedipine, spironolactone. Antiretrovirals = retonovir, sequinovir.
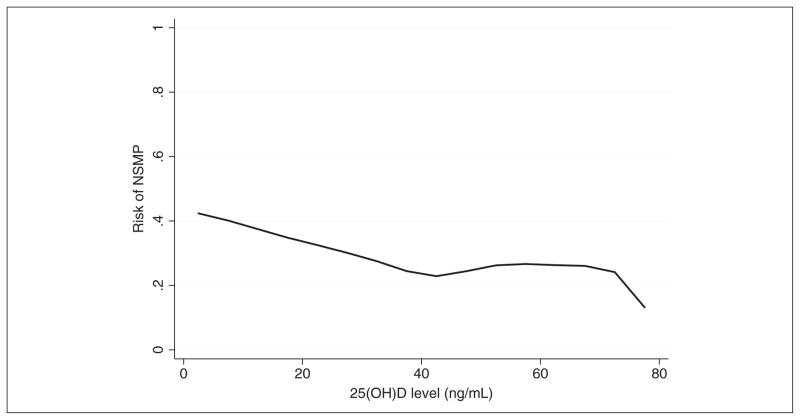
LOWESS plot demonstrated a near-inverse association between vitamin D status and nonspecific musculoskeletal pain (Figure 1) up to 25(OH)D levels of approximately 40 ng/mL. Between 25(OH)D levels of 40 ng/mL and 70 ng/mL, there was significant flattening of the curve. Logistic regression analysis, while controlling for biologically plausible covariates (Table 2), demonstrated an inverse association between admission serum 25(OH)D levels and nonspecific musculoskeletal pain (OR per 10 ng/mL, 0.67; 95% CI, 0.48–0.82). Further adjustment for vitamin D supplementation did not materially change these results (OR per 10 ng/mL, 0.68; 95% CI, 0.48–0.82), nor did additional adjustment for season of admission (OR per 10 ng/mL, 0.68; 95% CI, 0.49–0.80).
Figure 1.
Vitamin D status vs risk of nonspecific musculoskeletal pain. 25(OH)D, 25-hydroxyvitamin D; NSMP, nonspecific musculoskeletal pain. Locally weighted scatter plot smoothing used to represent the near-inverse linear association between 25(OH)D level and risk of nonspecific musculoskeletal pain.
Table 2.
Biologically Plausible Model to Test the Association of Admission 25-Hydroxyvitamin D Level With Nonspecific Musculoskeletal Pain in Acute Rehabilitation Unit Patients (n = 414).
| Covariate | Odds Ratio (95% Confidence Interval) |
|---|---|
| Age | 0.99 (1.20–1.65) |
| Sex | |
| Female | — |
| Male | 0.70 (0.42–1.15) |
| Race | |
| White | — |
| Nonwhite | 2.94 (1.32–6.67) |
| BMI | 0.97 (0.93–1.01) |
| DCCI | 1.41 (1.20–1.65) |
| Total FIM | 0.95 (0.93–0.98) |
| Fractures | |
| No | — |
| Yes | 2.22 (1.22–4.17) |
| Steroid use | |
| No | — |
| Yes | 2.42 (0.98–5.97) |
| Osteoporosis/osteomalacia | |
| No | — |
| Yes | 3.03 (1.43–6.67) |
| Type of patient | |
| Orthopedic | — |
| Neurological | 0.07 (0.04–0.11) |
| Cardiac | 0.03 (0.01–0.11) |
| Spinal cord injury | 0.54 (0.14–2.14) |
| Traumatic brain injury | 0.05 (0.02–0.14) |
| 25(OH)D | 0.96 (0.93–0.98) |
Statistically significant confidence intervals are noted in bold. —, indicates referent variable; BMI, body mass index; DCCI, Deyo-Charlson Comorbidity Index; FIM, Functional Independence Measure; 25(OH)D, 25-hydroxyvitamin D. Incident risk ratios are expressed per unit change in each covariate and are exponential for >1-unit change. For example, the incident risk ratio is 0.82 (0.965) for a 5-ng/mL increase in 25(OH)D level and 0.67 (0.9610) for a 10-ng/mL increase in 25(OH)D level.
When serum 25(OH)D levels were dichotomized using a threshold of 20 ng/mL, patients with serum 25(OH)D levels <20 ng/mL had higher odds of nonspecific musculoskeletal pain (adjusted OR, 2.33; 95% CI, 1.23–4.17) compared with patients with levels ≥20 ng/mL. Further adjustment for vitamin D supplementation or season of admission did not materially change these results (data not shown). Similarly, when serum 25(OH)D levels were dichotomized using a threshold of 30 ng/mL, patients with serum 25(OH)D levels <30 ng/mL had higher odds of nonspecific musculoskeletal pain (adjusted OR, 2.04; 95% CI, 1.15–3.57) compared with patients with levels ≥30 ng/mL. Further adjustment for vitamin D supplementation or season of admission did not materially change these results (data not shown).
Discussion
In this cross-sectional study, we investigated whether vitamin D status was associated with the presence of nonspecific musculoskeletal pain among individuals admitted for inpatient acute rehabilitation. We demonstrated that serum 25(OH)D level on admission to the ARU was inversely associated with persistent nonspecific musculoskeletal pain. Due to the observational nature of this study, a causal inference about the effect of vitamin D status on musculoskeletal pain is premature, but the observed association does support the need for RCTs to address this topic.
While vitamin D is widely recognized for its critical role in maintaining overall musculoskeletal health in humans,33 its potential role in regulating pain pathways is just starting to gain recognition. Indeed, studies have shown an association between 25(OH)D levels and pain in patients with osteoporosis,34 osteomalacia,35 failed back syndrome,21 neuropathic lesions,36 fibromyalgia,37 osteoarthritis,38 and rheumatoid arthritis.39 RCTs are limited, with the current evidence suggesting that vitamin D supplementation may benefit patients with fibromyalgia40; data regarding osteoporotic pain are less clear.41,42
The mechanism(s) by which vitamin D may help to regulate pain symptoms is not yet clear. Since vitamin D is critical for calcium homeostasis and preservation of bone health,43 it has been postulated that when 25(OH)D levels are low, impaired bone mineralization allows the osteoid matrix to absorb fluid and expand, causing outward pressure on the highly innervated periosteal tissues.19 Indeed, bone remodulation occurs in most ARU patients, even in the absence of fractures or orthopedic surgery.24 1,25-Dihydroxyvitamin D (1,25(OH)2D), the most biologically active vitamin D metabolite, also influences skeletal muscle calcium uptake, phosphate transport across the cell membrane, phospholipid metabolism, and muscle cell proliferation as well as differentiation.44 As such, immobility and muscular atrophy may be exacerbated in the setting of suboptimal vitamin D status23 and thus may increase the risk of muscular pain in ARU patients. Moreover, the prefrontal cortex, cingulate gyrus, hippocampus, basal forebrain, caudate/putamen, thalamus, substantia nigra, lateral geniculate nuclei, hypothalamus, and cerebellum all express the vitamin D receptor (VDR)—several regions have the capacity to locally convert circulating 25(OH)D to 1,25(OH)2D.45 In the central nervous system, 1,25(OH)2D has been shown to influence the production of neutrotrophic factors and calcium binding proteins in glial cells, which are important for repair, proliferation, and differentiation of neurons.46 Consequently, suboptimal vitamin D status may exacerbate neuropathic pain, which may have been caused by the underlying pathology in ARU patients (eg, stroke). Furthermore, recent studies have demonstrated that cells of the innate and adaptive immune system also express VDR.47 Low 25(OH)D levels are associated with depressed macrophage phagocytosis, attenuated chemotaxis, and proinflammatory cytokine production.48 Suboptimal vitamin D status may therefore contribute to chronic inflammation, which may result in persistent nonspecific pain symptoms. Since vitamin D appears to be essential for musculoskeletal, neuronal, and immune health, all factors that may influence pain symptoms in ARU patients, our findings suggest that vitamin D status may truly influence pain. Given the relatively limited options now available in the management of pain in many ARU patients,10–18 this possibility merits further investigation.
Although the study results are compelling, we must also consider the potential limitations of its observational nature, such as reverse causation and/or the lack of a randomly distributed exposure. Furthermore, despite adjusting for multiple potential covariates, there may still be residual confounding that contributed to the large observed differences in outcome. Specifically, low serum 25(OH)D levels may be a marker for the general condition of patients, for which we were unable to fully adjust. These issues complicate causal inferences and may decrease the generalizability of our results to all ARU patients. Moreover, the patients were selected from a single institution that is a referral center for ARU patients in Southern California, which may further affect the generalizability of our findings. Another potential limitation is that pain assessments are largely subjective, can be influenced by various physiologic and psychosomatic stressors, and may vary significantly over the temporal course of acute and chronic illness. Moreover, traditional assessment methods (eg, visual analog scale) may underestimate the presence of pain in patients with cognitive dysfunction (eg, after a stroke or traumatic brain injury). We also were unable to control for some potentially meaningful biomarkers such as parathyroid hormone, C-reactive protein, and calcium levels. These issues will need to be addressed by future studies.
Conclusion
Our results suggest that vitamin D status may be a modifiable risk factor for nonspecific musculoskeletal pain in ARU patients. We hypothesize that ideal 25(OH)D levels are associated with optimal musculoskeletal heath,43,44 appropriate expression of endogenous peptides within the central nervous system,45,46 and effective regulation of inflammatory responses47,48 following acute care hospitalization. This is particularly important, given that suboptimal 25(OH)D levels are highly prevalent in hospitalized patients who require subsequent acute rehabilitation.23–26 Further studies are needed to validate our findings, assess the potential benefit of optimizing 25(OH)D levels during acute rehabilitation, and identify the mechanism by which vitamin D may influence nonspecific musculoskeletal pain in ARU patients.
Acknowledgments
The authors thank James A. Cushing, MD, Lisa J. Maylish PT, CCM, Hongthao Pham, RN, BSN, and Veronica Searls, BA, for their valuable input and assistance with this project.
Footnotes
Financial disclosure: S.A.Q. received support from the National Institutes of Health (grant number 5T32GM007592).
References
- 1.Sullivan-Singh SJ, Sawyer K, Ehde DM, et al. Comorbidity of pain and depression among persons with traumatic brain injury. Arch Phys Med Rehabil. 2014;95:1100–1105. doi: 10.1016/j.apmr.2014.02.001. [DOI] [PubMed] [Google Scholar]
- 2.Pereira LS, Sherrington C, Ferreira ML, et al. Self-reported chronic pain is associated with physical performance in older people leaving aged care rehabilitation. Clin Interv Aging. 2014;9:259–265. doi: 10.2147/CIA.S51807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lin CH1, Chen KH, Chang CH, et al. Muscle pain intensity and pressure pain threshold changes in different periods of stroke patients. Am J Phys Med Rehabil. 2014;93:299–309. doi: 10.1097/PHM.0000000000000003. [DOI] [PubMed] [Google Scholar]
- 4.Zanca JM, Dijkers MP, Hammond FM, Horn SD. Pain and its impact on inpatient rehabilitation for acute traumatic spinal cord injury: analysis of observational data collected in the SCIRehab study. Arch Phys Med Rehabil. 2013;94:S137–S144. doi: 10.1016/j.apmr.2012.10.035. [DOI] [PubMed] [Google Scholar]
- 5.McBeth J, Jones K. Epidemiology of chronic musculoskeletal pain. Best Pract Res Clin Rheumatol. 2007;21:403–425. doi: 10.1016/j.berh.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 6.Feeney SL. The relationship between pain and negative affect in older adults: anxiety as a predictor of pain. J Anxiety Disord. 2004;18:733–744. doi: 10.1016/j.janxdis.2001.04.001. [DOI] [PubMed] [Google Scholar]
- 7.McKinley WO, Tewksbury MA, Godbout CJ. Comparison of medical complications following nontraumatic and traumatic spinal cord injury. J Spinal Cord Med. 2002;25:88–93. doi: 10.1080/10790268.2002.11753607. [DOI] [PubMed] [Google Scholar]
- 8.Dromerick A, Reding M. Medical and neurological complications during inpatient stroke rehabilitation. Stroke. 1994;25:358–361. doi: 10.1161/01.str.25.2.358. [DOI] [PubMed] [Google Scholar]
- 9.McKinley S1, Aitken LM, Alison JA, et al. Sleep and other factors associated with mental health and psychological distress after intensive care for critical illness. Intensive Care Med. 2012;38:627–633. doi: 10.1007/s00134-012-2477-4. [DOI] [PubMed] [Google Scholar]
- 10.Saulino M. Spinal cord injury pain. Phys Med Rehabil Clin North Am. 2014;25:397–410. doi: 10.1016/j.pmr.2014.01.002. [DOI] [PubMed] [Google Scholar]
- 11.O’Connor JP, Lysz T. Celecoxib, NSAIDs and the skeleton. Drugs Today (Barc) 2008;44:693–709. doi: 10.1358/dot.2008.44.9.1251573. [DOI] [PubMed] [Google Scholar]
- 12.Mitra R. The utilization of opiates in pain management: use or abuse. Eur J Phys Rehabil Med. 2013;49:93–96. [PubMed] [Google Scholar]
- 13.Rahman W, Dickenson AH. Voltage gated sodium and calcium channel blockers for the treatment of chronic inflammatory pain. Neurosci Lett. 2013;557(pt A):19–26. doi: 10.1016/j.neulet.2013.08.004. [DOI] [PubMed] [Google Scholar]
- 14.Chen L, Mao J. Update on neuropathic pain treatment: ion channel blockers and gabapentinoids. Curr Pain Headache Rep. 2013;17:359. doi: 10.1007/s11916-013-0359-2. [DOI] [PubMed] [Google Scholar]
- 15.Zorowitz RD, Smout RJ, Gassaway JA, Horn SD. Usage of pain medications during stroke rehabilitation: the Post-Stroke Rehabilitation Outcomes Project (PSROP) Top Stroke Rehabil. 2005;12:37–49. doi: 10.1310/C7MF-VLR0-CKDL-3C44. [DOI] [PubMed] [Google Scholar]
- 16.Chase T, Jha A, Brooks CA, Allshouse A. A pilot feasibility study of massage to reduce pain in people with spinal cord injury during acute rehabilitation. Spinal Cord. 2013;51:847–851. doi: 10.1038/sc.2013.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang R, Lao L, Ren K, Berman BM. Mechanisms of acupuncture-electroacupuncture on persistent pain. Anesthesiology. 2014;120:482–503. doi: 10.1097/ALN.0000000000000101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee JA, Park SW, Hwang PW, et al. Acupuncture for shoulder pain after stroke: a systematic review. J Altern Complement Med. 2012;18:818–823. doi: 10.1089/acm.2011.0457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shinchuk LM, Holick MF. Vitamin D and rehabilitation: improving functional outcomes. Nutr Clin Pract. 2007;22:297–304. doi: 10.1177/0115426507022003297. [DOI] [PubMed] [Google Scholar]
- 20.Huang W, Shah S, Long Q, Crankshaw AK, Tangpricha V. Improvement of pain, sleep, and quality of life in chronic pain patients with vitamin D supplementation. Clin J Pain. 2013;29:341–347. doi: 10.1097/AJP.0b013e318255655d. [DOI] [PubMed] [Google Scholar]
- 21.Waikakul S. Serum 25-hydroxy-calciferol level and failed back surgery syndrome. J Orthop Surg (Hong Kong) 2012;20:18–22. doi: 10.1177/230949901202000104. [DOI] [PubMed] [Google Scholar]
- 22.Abokrysha NT. Vitamin D deficiency in women with fibromyalgia in Saudi Arabia. Pain Med. 2012;13:452–458. doi: 10.1111/j.1526-4637.2011.01304.x. [DOI] [PubMed] [Google Scholar]
- 23.Heath KM, Elovic EP. Vitamin D deficiency: implications in the rehabilitation setting. Am J Phys Med Rehabil. 2006;85:916–923. doi: 10.1097/01.phm.0000242622.23195.61. [DOI] [PubMed] [Google Scholar]
- 24.Shinchuk LM, Morse L, Huancahuari N, Arum S, Chen TC, Holick MF. Vitamin D deficiency and osteoporosis in rehabilitation inpatients. Arch Phys Med Rehabil. 2006;87:904–908. doi: 10.1016/j.apmr.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 25.Schilling S. Epidemic vitamin D deficiency among patients in an elderly care rehabilitation facility. Dtsch Arztebl Int. 2012;109:33–38. doi: 10.3238/arztebl.2012.0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nemunaitis GA, Mejia M, Nagy JA, Johnson T, Chae J, Roach MJ. A descriptive study on vitamin D levels in individuals with spinal cord injury in an acute inpatient rehabilitation setting. PM R. 2010;2:202–208. doi: 10.1016/j.pmrj.2010.01.010. [DOI] [PubMed] [Google Scholar]
- 27.Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96:53–58. doi: 10.1210/jc.2010-2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–1930. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 29.Maunsell Z, Wright DJ, Rainbow SJ. Routine isotope-dilution liquid chromatography-tandem mass spectrometry assay for simultaneous measurement of the 25-hydroxy metabolites of vitamins D2 and D3. Clin Chem. 2005;51:1683–1690. doi: 10.1373/clinchem.2005.052936. [DOI] [PubMed] [Google Scholar]
- 30.Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43:1130–1139. doi: 10.1097/01.mlr.0000182534.19832.83. [DOI] [PubMed] [Google Scholar]
- 31.Cleveland W. Robust locally weighted regression and smoothing scatter-plots. J Am Stat Assoc. 1979;74:829–836. [Google Scholar]
- 32.Cleveland W, Devlin S. Locally-weighted regression: an approach to regression analysis by local fitting. J Am Stat Assoc. 1988;83:596–610. [Google Scholar]
- 33.Janssen HC, Samson MM, Verhaar HJ. Vitamin D deficiency, muscle function, and falls in elderly people. Am J Clin Nutr. 2002;75:611–615. doi: 10.1093/ajcn/75.4.611. [DOI] [PubMed] [Google Scholar]
- 34.Jansen JA, Haddad FS. High prevalence of vitamin D deficiency in elderly patients with advanced osteoarthritis scheduled for total knee replacement associated with poorer preoperative functional state. Ann R Coll Surg Engl. 2013;95:569–572. doi: 10.1308/003588413X13781990150374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bhan A, Rao AD, Rao DS. Osteomalacia as a result of vitamin D deficiency. Rheum Dis Clin North Am. 2012;38:81–91. doi: 10.1016/j.rdc.2012.03.008. [DOI] [PubMed] [Google Scholar]
- 36.Lee P, Chen R. Vitamin D as an analgesic for patients with type 2 diabetes and neuropathic pain. Arch Intern Med. 2008;168:771–772. doi: 10.1001/archinte.168.7.771. [DOI] [PubMed] [Google Scholar]
- 37.Jesus CA, Feder D, Peres MF. The role of vitamin D in pathophysiology and treatment of fibromyalgia. Curr Pain Headache Rep. 2013;17:355. doi: 10.1007/s11916-013-0355-6. [DOI] [PubMed] [Google Scholar]
- 38.Laslett LL, Quinn S, Burgess JR, et al. Moderate vitamin D deficiency is associated with changes in knee and hip pain in older adults: a 5-year longitudinal study. Ann Rheum Dis. 2014;73:697–703. doi: 10.1136/annrheumdis-2012-202831. [DOI] [PubMed] [Google Scholar]
- 39.Haque UJ, Bartlett SJ. Relationships among vitamin D, disease activity, pain and disability in rheumatoid arthritis. Clin Exp Rheumatol. 2010;28:745–747. [PubMed] [Google Scholar]
- 40.Wepner F, Scheuer R, Schuetz-Wieser B, et al. Effects of vitamin D on patients with fibromyalgia syndrome: a randomized placebo-controlled trial. Pain. 2014;155:261–268. doi: 10.1016/j.pain.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 41.Sanghi D, Mishra A, Sharma AC, et al. Does vitamin D improve osteoarthritis of the knee: a randomized controlled pilot trial. Clin Orthop Relat Res. 2013;471:3556–3562. doi: 10.1007/s11999-013-3201-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.McAlindon T, LaValley M, Schneider E, et al. Effect of vitamin D supplementation on progression of knee pain and cartilage volume loss in patients with symptomatic osteoarthritis: a randomized controlled trial. JAMA. 2013;309:155–162. doi: 10.1001/jama.2012.164487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Holick MF. Vitamin D: a millennium perspective. Cell Biochem. 2003;88:296–307. doi: 10.1002/jcb.10338. [DOI] [PubMed] [Google Scholar]
- 44.Vazquez G, de Boland AR, Boland R. Stimulation of Ca2+ release-activated Ca2+ channels as a potential mechanism involved in non-genomic 1,25(OH)2-vitamin D3-induced Ca2+ entry in skeletal muscle cells. Biochem Biophys Res Commun. 1997;239:562–565. doi: 10.1006/bbrc.1997.7501. [DOI] [PubMed] [Google Scholar]
- 45.Buell JS, Dawson-Hughes B. Vitamin D and neurocognitive dysfunction: preventing “D”ecline? Mol Aspects Med. 2008;29:415–422. doi: 10.1016/j.mam.2008.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Annweiler C, Allali G, Allain P, et al. Vitamin D and cognitive performance in adults: a systematic review. Eur J Neurol. 2009;16:1083–1089. doi: 10.1111/j.1468-1331.2009.02755.x. [DOI] [PubMed] [Google Scholar]
- 47.Adams JS, Hewison M. Unexpected actions of vitamin D: new perspectives on the regulation of innate and adaptive immunity. Nat Clin Pract Endocrinol Metab. 2008;4:80–90. doi: 10.1038/ncpendmet0716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kankova M, Luini W, Pedrazzoni M, et al. Impairment of cytokine production in mice fed a vitamin D3–deficient diet. Immunology. 1991;73:466–471. [PMC free article] [PubMed] [Google Scholar]