Abstract
Introduction
The effect of mean arterial pressure titration to a higher level on microcirculation in septic shock patients with previous hypertension remains unknown. Our goal is to assess the effect of mean arterial pressure titration to a higher level on microcirculation in hypertensive septic shock patients.
Methods
This is a single-center, open-label study. Hypertensive patients with septic shock for less than 24 hours after adequate fluid resuscitation and requiring norepinephrine to maintain a mean arterial pressure of 65 mmHg were enrolled. Mean arterial pressure was then titrated by norepinephrine from 65 mmHg to the normal level of the patient. In addition to hemodynamic variables, sublingual microcirculation was evaluated by sidestream dark field imaging.
Results
Nineteen patients were enrolled in the study. Increasing mean arterial pressure from 65 mmHg to normal levels was associated with increased central venous pressure (from 11 ± 4 to 13 ± 4 mmHg, P = 0.002), cardiac output (from 5.4 ± 1.4 to 6.4 ± 2.1 l/minute, P = 0.001), and central venous oxygen saturation (from 81 ± 7 to 83 ± 7%, P = 0.001). There were significant increases in small perfused vessel density (from 10.96 ± 2.98 to 11.99 ± 2.55 vessels/mm2, P = 0.009), proportion of small perfused vessels (from 85 ± 18 to 92 ± 14%, P = 0.002), and small microvascular flow index (from 2.45 ± 0.61 to 2.80 ± 0.68, P = 0.009) when compared with a mean arterial pressure of 65 mmHg.
Conclusions
Increasing mean arterial pressure from 65 mmHg to normal levels is associated with improved microcirculation in hypertensive septic shock patients.
Trial registration
Clinicaltrials.gov: NCT01443494; registered 28 September 2011.
Electronic supplementary material
The online version of this article (doi:10.1186/s13054-015-0866-0) contains supplementary material, which is available to authorized users.
Introduction
Septic shock is a major cause of death in critically ill patients and is characterized by hypotension and hypoperfusion [1]. Although mortality has been reported to be decreased due to the progress in therapy for septic shock [2,3], microcirculation abnormalities can still be present and contribute to hypoperfusion [4]. Microvascular dysfunction is associated with organ dysfunction and mortality [5,6]; moreover, early improvement in the microcirculation during resuscitation is associated with reduced organ failure [7]. Therefore, therapeutic strategies aimed at improving the microcirculation are warranted to mitigate hypoperfusion and organ failure.
Maintaining sufficient perfusion pressure might be an important way to improve the microcirculatory perfusion [8]. As one of the key determinants of perfusion pressure, adequate mean arterial pressure (MAP) might reduce hypoperfusion and organ failure. Correa and colleagues [9] have evaluated the effects of different MAP targets in septic pigs, and show that targeting a MAP between 50 and 60 mmHg in septic shock is associated with increased incidence of acute kidney injury when compared with a target MAP of between 75 and 85 mmHg. Moreover, Badin and colleagues [10] have reported that acute kidney injury is decreased with a MAP of at least 72 mmHg. However, which MAP levels should be targeted to improve microcirculation remains controversial [11-13].
The optimal MAP target in septic shock patients, especially in patients with previous hypertension, is still unknown [14]. The guidelines [15] for treating septic shock have recommended that MAP should be maintained at a level of at least 65 mmHg, and emphasized a higher blood pressure for patients with previous hypertension. A recent published randomized controlled study [16] has shown no difference in mortality in septic shock patients undergoing resuscitation with a high MAP target or a low MAP target. However, among chronic hypertensive patients, those in the high-target group require less renal replacement therapy, indicating that septic shock patients with chronic hypertension might need higher MAP to maintain microcirculation.
Our goal was to assess the effect on the microcirculation of MAP titration to normal levels in septic shock patients with previous hypertension. We hypothesized that the increase in MAP from 65 mmHg to the normal levels of patients is associated with improved sublingual microcirculation.
Materials and methods
Setting
This was a single-center, prospective, open-label study conducted in the intensive care unit of a tertiary care teaching hospital. The study protocol was approved by the Ethics Committee (Approval Number: 2011ZDllKY03.0) of Zhongda Hospital, School of Medicine, Southeast University, and informed consent was given by each patient or their next of kin.
Trial registration: Clinicaltrials.gov NCT01443494; registered 28 November 2011.
Study population
Patients admitted to the intensive care unit who met the following inclusion criteria were enrolled in the study: 1) patients with septic shock for less than 24 hours with a history of previous hypertension (septic shock was defined by the 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference [17]); 2) fluid resuscitation was performed according to the guidelines for treating septic shock to maintain the central venous pressure (CVP) at more than 8 mmHg and central venous oxygen saturation at more than 70%; 3) patients required norepinephrine (NE) to maintain a MAP of 65 mmHg. All patients were treated with mechanical ventilation, and received infusions of morphine and propofol. All patients had an arterial catheter and a central venous catheter in place.
Exclusion criteria were: pregnancy; age <18 years; inability to acquire the usual level of MAP; refusal of consent by the patient or relative; participation in other trials during the last 3 months; and hypertensive patients without antihypertensive therapy.
Interventions
As chronic hypertensive patients are supposed to have undergone more blood pressure measurements in daily life than normotensive patients, the average MAP acquired from a patient’s physical examination records over the last 2 years was taken to be the target MAP. If the medical records were incomplete, a detailed enquiry about the target MAP to their next of kin was performed. In addition, age, gender, duration of shock prior to inclusion, Acute Physiology And Chronic Health Evaluation II score, intensive care unit length of stay, in-hospital mortality, source of infection and comorbidities were also recorded.
Study design
After stabilization for 30 minutes, basal measurements including hemodynamic and microcirculatory measurements were taken 20 minutes apart, and the NE doses were increased to titrate MAP to the target level. Patients were allowed to stabilize for 30 minutes before new measurements were taken. Thus, there was 50 minutes between the baseline and the second measurement. During the process, no other changes in treatment were allowed. The decision to stop the research was made by the treating intensivist according to predetermined safety limits for each patient. The detailed safety limits are reported in Additional file 1.
Hemodynamic measurements
Serial measurements of NE dose, MAP, heart rate and CVP were performed. Noninvasive bioreactance cardiac output and stroke volume monitoring were obtained using the NICOM system (Cheetah Medical, Portland, OR, USA). Arterial and central venous gases were obtained to determine arterial pH, hemoglobin, lactate, arterial partial pressure of oxygen, arterial partial pressure of carbon dioxide, arterial hemoglobin saturation, arterial oxygen partial pressure to fractional inspired oxygen ratio and central venous hemoglobin saturation.
Microcirculatory measurements and analysis
Measurements of the sublingual microcirculation were obtained using sidestream dark field (SDF; Microscan, Microvision Medical, Amsterdam, The Netherlands). The SDF probe was placed under the sublingual area without pressure after removal of secretions. Video sequences of 20 seconds each were recorded from three different sublingual sites. These images were stored and later renumbered by an identifier blinded to the clinical course. Videos were converted to the AVI (audio video interleaved) file format with video processing software, and analysis for each image was performed by two different investigators. Finally, all the variables were averaged to yield a single value for statistical analysis. The microcirculatory flow index (MFI), based on determination of the predominant type of flow in four quadrants for small vessels and total vessels, was determined with a semi-quantitative methodology. Flow was characterized as no flow = 0, intermittent = 1, sluggish = 2, and continuous = 3, to reflect blood velocity, identical to that described in consensus conference recommendations [18]. MFI was calculated for all quadrants of the image and averaged for each sublingual site. Detailed methods are reported in Additional file 2.
Statistical analysis
Statistical analysis of the data was performed using SPSS 16.0 (IBM, Somers, NY, USA). Histograms and normal-quantile plots were examined and Kolmogorov-Smirnov test was performed to verify the normality of the distributions of the data. Data are presented as the mean ± SD if normally distributed or median (interquartile range) if not normally distributed. Categorical variables are presented as number and percentage. For continuous data, paired t test or Wilcoxon signed-rank test were used. Differences were considered significant at P < 0.05.
Results
Clinical characteristics of study subjects
The main characteristics of the 19 patients are presented in Table 1. Detailed enquiry about the target MAP to their next of kin was performed for all of the patients. The averaged value of MAP was 93 mmHg in our study, which was maintained in the range 80 to 110 mmHg under the control of medications. The mean duration of shock prior to study inclusion was 16 hours. In-hospital mortality was 53%. The most common source of sepsis was pneumonia.
Table 1.
Baseline patient characteristics
| Patient characteristic | (n = 19) |
|---|---|
| Age (years) | 74 ± 11 |
| Gender (%) | 13 males (68) |
| Duration of shock prior to inclusion (hours) | 16 ± 6 |
| APACHE II score | 24 ± 10 |
| Intensive care unit length of stay (days) | 12 ± 8 |
| In-hospital mortality (n (%)) | 10 (53) |
| Source of infection (n (%)) | |
| Respiratory infection | 11 (58) |
| Abdominal infection | 6 (32) |
| Bacteremia | 1 (5) |
| Urinary tract infection | 1 (5) |
| Comorbidities (n (%)) | |
| Chronic hypertension | 19 (100) |
| Diabetes | 2 (10) |
| Chronic renal failure | 2 (10) |
| Usual mean arterial pressure acquired from previous medical records (mmHg) | 93 ± 7 |
| Fluid infusion during the study (ml) | 185 ± 55 |
APACHE, Acute Physiology and Chronic Health Evaluation. Data presented as mean ± SD or n (%).
Effect on global hemodynamics
Increasing MAP from 65 mmHg to target level (from 71 ± 8 to 91 ± 7 mmHg, P < 0.001) by augmenting NE dose was associated with significantly increased CVP (from 11 ± 4 to 13 ± 4 mmHg, P = 0.002), cardiac output (from 5.4 ± 1.4 to 6.4 ± 2.1 l/minute, P = 0.001) and central venous oxygen saturation (from 81 ± 7 to 83 ± 7%, P = 0.001) (Table 2, Figure 1).
Table 2.
Hemodynamic variables
| MAP = 65 mmHg | Usual MAP | P | |
|---|---|---|---|
| Norepinephrine dose (μg/kg/minute) | 0.08 (0.04-0.34) | 0.25 (0.16-0.68)* | <0.001 |
| Systolic arterial pressure (mmHg) | 107 ± 19 | 140 ± 15* | <0.001 |
| Diastolic arterial pressure (mmHg) | 53 ± 7 | 66 ± 9* | <0.001 |
| Mean arterial pressure (mmHg) | 71 ± 8 | 91 ± 7* | <0.001 |
| Heart rate (beats/minute) | 100 ± 24 | 99 ± 21 | 0.793 |
| Central venous pressure (mmHg) | 11 ± 4 | 13 ± 4* | 0.002 |
| Cardiac output (l/minute) | 5.4 ± 1.4 | 6.4 ± 2.1* | 0.001 |
| Stroke volume (ml) | 57.5 ± 21.2 | 67.0 ± 26.7* | 0.003 |
| Urine output (ml/hour) | 56 ± 6 | 55 ± 6 | 0.921 |
| Hemoglobin (g/dl) | 9.4 ± 1.9 | 9.5 ± 2.0 | 0.605 |
| Arterial pH | 7.38 ± 0.06 | 7.39 ± 0.06 | 0.305 |
| Lactate (mmol/l) | 2.3 ± 2.3 | 2.2 ± 2.3 | 0.598 |
| Arterial partial pressure of oxygen (mmHg) | 132 ± 37 | 142 ± 51 | 0.250 |
| Arterial partial pressure of carbon dioxide (mmHg) | 30 ± 6 | 29 ± 6 | 0.303 |
| Arterial hemoglobin saturation (%) | 97.4 ± 1.8 | 98.0 ± 1.6 | 0.129 |
| PaO2:FiO2 ratio (mmHg) | 259 ± 99 | 291 ± 104* | 0.003 |
| Central venous hemoglobin saturation (%) | 81 ± 7 | 83 ± 7* | 0.001 |
*P < 0.05 versus baseline of mean arterial pressure 65 mmHg. MAP, mean arterial pressure; PaO2:FiO2, arterial oxygen partial pressure to fractional inspired oxygen. Data presented as means ± SD or median (interquartile range).
Figure 1.
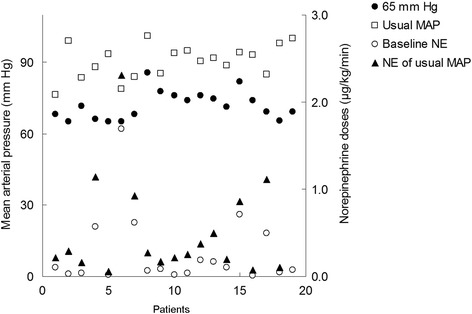
Baseline and target mean arterial pressure as well as baseline and final norepinephrine doses are displayed. MAP, mean arterial pressure. NE, norepinephrine.
Effect on sublingual microcirculation
Although the vascular density and heterogeneity index were not significantly altered, there were significant increases in small perfused vessel density (from 10.96 ± 2.98 to 11.99 ± 2.55 vessels/mm2, P = 0.009), proportion of small perfused vessels (from 85 ± 18 to 92 ± 14%, P = 0.002), and small MFI (from 2.45 ± 0.61 to 2.80 ± 0.68, P = 0.009) compared with a MAP of 65 mmHg (Table 3).
Table 3.
Sidestream dark field variables
| MAP = 65 mmHg | Usual MAP | P | |
|---|---|---|---|
| Small vessel density (vessels/mm2) | 11.29 ± 2.18 | 11.91 ± 2.14 | 0.129 |
| Total vessel density (vessels/mm2) | 12.35 ± 2.29 | 12.76 ± 2.09 | 0.227 |
| Small perfused vessel density (vessels/mm2) | 10.96 ± 2.98 | 11.99 ± 2.55* | 0.009 |
| Total perfused vessel density (vessels/mm2) | 11.34 ± 3.03 | 12.24 ± 2.55* | 0.021 |
| Proportion of small perfused vessels (%) | 85 ± 18 | 92 ± 14* | 0.002 |
| Proportion of total perfused vessels (%) | 92 ± 18 | 96 ± 12* | 0.029 |
| Small microvascular flow index | 2.45 ± 0.61 | 2.80 ± 0.68* | 0.009 |
| Total microvascular flow index | 2.70 ± 0.52 | 3.05 ± 0.51* | 0.002 |
| Heterogeneity index of small diameter vessels | 0.59 ± 0.32 | 0.47 ± 0.27 | 0.145 |
| Heterogeneity index of total diameter vessels | 0.36 ± 0.27 | 0.30 ± 0.19 | 0.330 |
*P < 0.05 versus baseline of mean arterial pressure 65 mmHg. MAP, mean arterial pressure. Data presented as means ± SD.
Meanwhile, little interindividual variability in small perfused vessel density (Figures 2 and 3), proportion of small perfused vessels (Figure 4) or MFI (Figure 5) appeared (see Additional file 3, a table listing the relationship between the changes of each microvascular variables and basal microvascular parameters).
Figure 2.
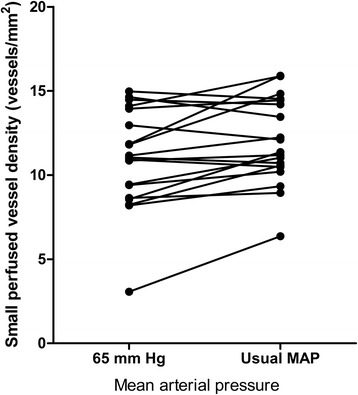
Results of small perfused vessel density are shown as the mean arterial pressure increased from 65 mmHg to usual level with norepinephrine. MAP, mean arterial pressure.
Figure 3.
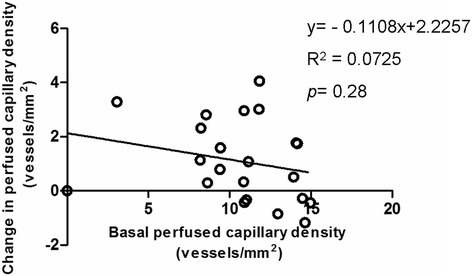
Relationship between the changes in small perfused vessel density, when mean arterial pressure increased from 65 mmHg to usual levels, with the basal small perfused vessel density at a mean arterial pressure of 65 mmHg.
Figure 4.
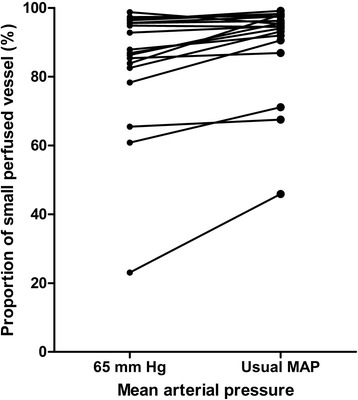
Results of proportion of small perfused vessels are shown as the mean arterial pressure increased from 65 mmHg to usual levels with norepinephrine. MAP, mean arterial pressure.
Figure 5.
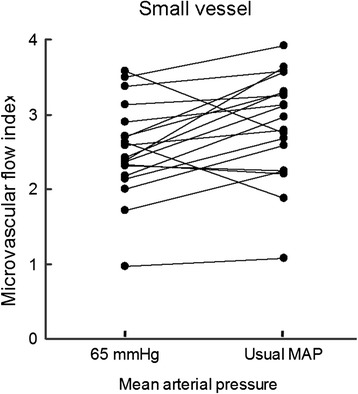
Results of microvascular flow index are shown as the mean arterial pressure increased from 65 mmHg to usual levels with norepinephrine. MAP, mean arterial pressure.
Discussion
The main finding of our study suggested that increasing MAP to the usual levels in early resuscitated hypertensive septic shock patients was associated with considerable microcirculatory improvements, characterized by increased perfused vessel density, a higher proportion of small perfused vessels and elevated MFI. Meanwhile, little individual variability developed.
Microcirculatory dysfunction plays an important role in the development of tissue hypoperfusion in patients with septic shock. Although early goal-directed therapy is reported to improve their outcome [2], the routine global hemodynamic parameters such as cardiac output, central or mixed venous oxygen saturation do not work as an effective perfusion index in most cases, and organ failure is still present after rapid correction of global hemodynamic alteration [6]. The microcirculation is responsible for regulating tissue perfusion to meet oxygen requirements and is associated with the development of organ dysfunction. Therefore, it is important to monitor and manage the microcirculation in order to augment perfusion in patients with septic shock.
An adequate MAP is typically essential to restore effective perfusion pressure and organ perfusion in septic shock patients. As the most common pre-existing condition in patients with septic shock [16], the Surviving Sepsis Campaign guidelines [15] have recommended a higher, but vague, blood pressure for patients with previous hypertension. Moreover, MAP targets in clinical trials varies substantially, ranging from 60 to 110 mmHg [19]. Few trials have focused on the effect of different MAP targets in septic shock patients with previous hypertension, let alone which MAP levels should be targeted to improve microcirculation.
Hypertension could cause a rightward shift in cerebral pressure-flow autoregulation [20], which suggests a higher MAP should be targeted in patients with these conditions to maintain organ perfusion. Moreover, a recently published study [16] has observed less renal replacement therapy in the high-target group in septic shock patients with previous hypertension, suggesting that higher MAP might reduce hypoperfusion and organ failure in these patients. However, few studies have looked at the microvascular responses to higher MAP titration in septic shock patients with previous hypertension.
In our study, increasing MAP from 65 mmHg to the usual levels of the patients resulted in improved microcirculatory function in these septic shock patients with previous hypertension, although the cause and effect are unclear. NE was used to titrate MAP to the usual level. As the key vasoactive agent recommended for restoring MAP in the treatment of septic shock, NE might not only perform as the vasopressor but also affect preload and tissue perfusion. Increasing the dose of NE significantly augments cardiac output by 11 to 17% [21], suggesting NE might recruit some blood from the large venous unstressed volume as another method of “endogenous fluid challenge”. Since fluid challenge is demonstrated to improve microvascular perfusion in the early phase of septic shock [22,23], NE-induced microcirculatory improvement might occur because of its role on arterial pressure preservation and blood recruitment.
An NE-induced cardiac output effect might be another reason for the improvement in the microcirculation: rapidly achieving a sufficient perfusion pressure could increase cardiac output in septic shock patients [24]. Georger and colleagues [25] found that NE administration is associated with an increase in MAP and cardiac index, which resulted in improvements of near-infrared spectroscopy variables in septic shock patients. In our study, the NE-induced increase in cardiac output was 1 l/minute on average, which might be another reason for the microcirculatory improvement.
In our study, fluid resuscitation was performed according to the guidelines to maintain a CVP of more than 8 mmHg and central venous oxygen saturation of more than 70% before patients were enrolled, and the basal measurements were taken after a MAP of 65 mmHg was achieved by fluids and NE. This differs from other studies where baseline measurements were taken in the unresuscitated septic shock patients. As a result, the sublingual microcirculation at baseline in our study seems to be less altered when compared with other studies. However, they were similar to microcirculatory parameters in resuscitated septic shock patients [11,12,22].
Considerable microcirculatory interpatient variations are reported in septic shock patients after MAP titration despite fluid resuscitation [12,26], which indicates that the level of MAP should be adapted in consideration of the interindividual effect. In this study, titration to a pressure closer to that of a given patient’s baseline aimed to reduce the interindividual variability, and reveals the microcirculatory alterations after MAP titration to a higher level in septic shock patients with previous hypertension.
Limitations
Our study has some limitations. Firstly, cardiac output was monitored by the NICOM system to minimize operative invasion. Although the most widely trusted technology for measuring cardiac output is still bolus thermodilution, NICOM is found to have similar monitoring capabilities compared with bolus thermodilution [27,28]. Secondly, the target MAP level was obtained from previous physical examination records or was given by relatives. Although patients with chronic hypertension often undergo more blood pressure measurements in daily life than normotensive patients, the target might be over- or underestimated. Moreover, higher doses of NE may be associated with arrhythmias and other adverse events, which were not observed in our study. Last, but not the least, no assessment of fluid responsiveness was undertaken in our study. NE might have different effects on the microcirculation in patients with or without fluid responsiveness.
It is also important to note that this study is not a controlled study, and lacks a control group and a return to baseline blood pressure. Each patient, therefore, served as his/her own control when increasing MAP from 65 mmHg to the usual level. In addition, the lack of normotensive controls makes the results difficult to extend to normotensive subjects. However, there are several studies [11,12] focusing on the role of different MAP targets in normotensive patients with septic shock. In a large multicenter, open-label trial [16], targeting a MAP of 80 to 85 mmHg, as compared with 65 to 70 mmHg, resulted in no significant differences in mortality for septic shock patients.
Conclusions
In this open-label intervention study, increasing MAP from 65 mmHg to the usual level in patients was associated with improved microcirculatory function in septic shock patients with previous hypertension.
Key messages
Increasing MAP from 65 mmHg to the usual level in patients by NE was associated with increased CVP, cardiac output and central venous oxygen saturation in hypertensive septic shock patients.
Increasing MAP from 65 mmHg to the usual level in patients by NE was associated with increased small perfused vessel density, proportion of small perfused vessels, and small microvascular flow index in hypertensive septic shock patients.
Acknowledgements
This work is partially supported by grants from the National Natural Science Foundations of China (81170057, 81201489, 81300043, 81300060, 81372093), grants from the Qinghai province basic research program (2012-Z-713) and grants from the Clinical Medicine Science and Technology program of Jiangsu Province (BL2013030).
Abbreviations
- CVP
central venous pressure
- MAP
mean arterial pressure
- MFI
microcirculatory flow index
- NE
norepinephrine
- SDF
sidestream dark field
Additional files
Study design: predetermined safety limits.
Microcirculatory measurements and analysis: detailed microcirculatory measurements and analysis.
Results: relationship between the changes of each microvascular variable and basal microvascular parameters.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
JYX participated in the design of the study, carried out the analysis and interpretation of data, and participated in drafting, editing and submitting the manuscript. SQM participated in drafting the manuscript, and made substantial contributions to microcirculatory measurements. CP made substantial contributions to data acquisition and statistical analysis. LL participated in data acquisition and analysis. SLH and HLH contributed to microcirculatory measurements. SXC and ARL contributed to microcirculatory data analysis. YZH contributed to the design and coordination of the study. FMG contributed to the design and coordination of the study. YY was responsible for conception and design, and revising the manuscript for important intellectual content. HBQ was responsible for conception and design, and revising the manuscript for important intellectual content. All authors read and approved the final manuscript.
Contributor Information
Jing-Yuan Xu, Email: xujingyuanmail@163.com.
Si-Qing Ma, Email: siqing177@sina.com.
Chun Pan, Email: panchun1982@gmail.com.
Hong-Li He, Email: hhl0408@163.com.
Shi-Xia Cai, Email: cccsssxxx1980@163.com.
Shu-Ling Hu, Email: shulinghu2013@163.com.
Ai-Ran Liu, Email: airanliu2013@gmail.com.
Ling Liu, Email: liulingdoctor@gmail.com.
Ying-Zi Huang, Email: yz_huang@126.com.
Feng-Mei Guo, Email: fmguo2003@139.com.
Yi Yang, Email: yiyiyang2004@163.com.
Hai-Bo Qiu, Email: haiboq2000@163.com.
References
- 1.Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–51. doi: 10.1056/NEJMra1208623. [DOI] [PubMed] [Google Scholar]
- 2.Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–77. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 3.Yealy DM, Kellum JA, Huang DT, Barnato AE, Weissfeld LA, Pike F, et al. A randomized trial of protocol-based care for early septic shock. N Engl J Med. 2014;370:1683–93. doi: 10.1056/NEJMoa1401602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.De Backer D, Creteur J, Preiser JC, Dubois MJ, Vincent JL. Microvascular blood flow is altered in patients with sepsis. Am J Respir Crit Care Med. 2002;166:98–104. doi: 10.1164/rccm.200109-016OC. [DOI] [PubMed] [Google Scholar]
- 5.Hernandez G, Boerma EC, Dubin A, Bruhn A, Koopmans M, Edul VK, et al. Severe abnormalities in microvascular perfused vessel density are associated to organ dysfunctions and mortality and can be predicted by hyperlactatemia and norepinephrine requirements in septic shock patients. J Crit Care. 2013;28:538.e9–14. doi: 10.1016/j.jcrc.2012.11.022. [DOI] [PubMed] [Google Scholar]
- 6.Sakr Y, Dubois MJ, De Backer D, Creteur J, Vincent JL. Persistent microcirculatory alterations are associated with organ failure and death in patients with septic shock. Crit Care Med. 2004;32:1825–31. doi: 10.1097/01.CCM.0000138558.16257.3F. [DOI] [PubMed] [Google Scholar]
- 7.Trzeciak S, McCoy JV, Phillip DR, Arnold RC, Rizzuto M, Abate NL, et al. Early increases in microcirculatory perfusion during protocol-directed resuscitation are associated with reduced multi-organ failure at 24 h in patients with sepsis. Intensive Care Med. 2008;34:2210–7. doi: 10.1007/s00134-008-1193-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Backer D, Donadello K, Taccone FS, Ospina-Tascon G, Salgado D, Vincent JL. Microcirculatory alterations: potential mechanisms and implications for therapy. Ann Intensive Care. 2011;1:27. doi: 10.1186/2110-5820-1-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Correa TD, Vuda M, Takala J, Djafarzadeh S, Silva E, Jakob SM. Increasing mean arterial blood pressure in sepsis: effects on fluid balance, vasopressor load and renal function. Crit Care. 2013;17:R21. doi: 10.1186/cc12495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Badin J, Boulain T, Ehrmann S, Skarzynski M, Bretagnol A, Buret J, et al. Relation between mean arterial pressure and renal function in the early phase of shock: a prospective, explorative cohort study. Crit Care. 2011;15:R135. doi: 10.1186/cc10253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jhanji S, Stirling S, Patel N, Hinds CJ, Pearse RM. The effect of increasing doses of norepinephrine on tissue oxygenation and microvascular flow in patients with septic shock. Crit Care Med. 2009;37:1961–6. doi: 10.1097/CCM.0b013e3181a00a1c. [DOI] [PubMed] [Google Scholar]
- 12.Dubin A, Pozo MO, Casabella CA, Palizas F, Jr, Murias G, Moseinco MC, et al. Increasing arterial blood pressure with norepinephrine does not improve microcirculatory blood flow: a prospective study. Crit Care. 2009;13:R92. doi: 10.1186/cc7922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thooft A, Favory R, Salgado DR, Taccone FS, Donadello K, De Backer D, et al. Effects of changes in arterial pressure on organ perfusion during septic shock. Crit Care. 2011;15:R222. doi: 10.1186/cc10462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beloncle F, Lerolle N, Radermacher P, Asfar P. Target blood pressure in sepsis: between a rock and a hard place. Crit Care. 2013;17:126. doi: 10.1186/cc12543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41:580–637. doi: 10.1097/CCM.0b013e31827e83af. [DOI] [PubMed] [Google Scholar]
- 16.Asfar P, Meziani F, Hamel JF, Grelon F, Megarbane B, Anguel N, et al. High versus low blood-pressure target in patients with septic shock. N Engl J Med. 2014;370:1583–93. doi: 10.1056/NEJMoa1312173. [DOI] [PubMed] [Google Scholar]
- 17.Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;2001:1250–6. doi: 10.1097/01.CCM.0000050454.01978.3B. [DOI] [PubMed] [Google Scholar]
- 18.De Backer D, Hollenberg S, Boerma C, Goedhart P, Buchele G, Ospina-Tascon G, et al. How to evaluate the microcirculation: report of a round table conference. Crit Care. 2007;11:R101. doi: 10.1186/cc6118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sevransky JE, Nour S, Susla GM, Needham DM, Hollenberg S, Pronovost P. Hemodynamic goals in randomized clinical trials in patients with sepsis: a systematic review of the literature. Crit Care. 2007;11:R67. doi: 10.1186/cc5948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Strandgaard S, Olesen J, Skinhoj E, Lassen NA. Autoregulation of brain circulation in severe arterial hypertension. Br Med J. 1973;1:507–10. doi: 10.1136/bmj.1.5852.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Monnet X, Jabot J, Maizel J, Richard C, Teboul JL. Norepinephrine increases cardiac preload and reduces preload dependency assessed by passive leg raising in septic shock patients. Crit Care Med. 2011;39:689–94. doi: 10.1097/CCM.0b013e318206d2a3. [DOI] [PubMed] [Google Scholar]
- 22.Ospina-Tascon G, Neves AP, Occhipinti G, Donadello K, Buchele G, Simion D, et al. Effects of fluids on microvascular perfusion in patients with severe sepsis. Intensive Care Med. 2010;36:949–55. doi: 10.1007/s00134-010-1843-3. [DOI] [PubMed] [Google Scholar]
- 23.Pottecher J, Deruddre S, Teboul JL, Georger JF, Laplace C, Benhamou D, et al. Both passive leg raising and intravascular volume expansion improve sublingual microcirculatory perfusion in severe sepsis and septic shock patients. Intensive Care Med. 2010;36:1867–74. doi: 10.1007/s00134-010-1966-6. [DOI] [PubMed] [Google Scholar]
- 24.Hamzaoui O, Georger JF, Monnet X, Ksouri H, Maizel J, Richard C, et al. Early administration of norepinephrine increases cardiac preload and cardiac output in septic patients with life-threatening hypotension. Crit Care. 2010;14:R142. doi: 10.1186/cc9207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Georger JF, Hamzaoui O, Chaari A, Maizel J, Richard C, Teboul JL. Restoring arterial pressure with norepinephrine improves muscle tissue oxygenation assessed by near-infrared spectroscopy in severely hypotensive septic patients. Intensive Care Med. 2010;36:1882–9. doi: 10.1007/s00134-010-2013-3. [DOI] [PubMed] [Google Scholar]
- 26.Boerma EC, Ince C. The role of vasoactive agents in the resuscitation of microvascular perfusion and tissue oxygenation in critically ill patients. Intensive Care Med. 2010;36:2004–18. doi: 10.1007/s00134-010-1970-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Marque S, Cariou A, Chiche JD, Squara P. Comparison between Flotrac-Vigileo and Bioreactance, a totally noninvasive method for cardiac output monitoring. Crit Care. 2009;13:R73. doi: 10.1186/cc7884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Squara P, Rotcajg D, Denjean D, Estagnasie P, Brusset A. Comparison of monitoring performance of bioreactance vs. pulse contour during lung recruitment maneuvers. Crit Care. 2009;13:R125. doi: 10.1186/cc7981. [DOI] [PMC free article] [PubMed] [Google Scholar]


