Abstract
Objective
To describe the relationship of delivery room cardiopulmonary resuscitation (DR-CPR) to short term outcomes of extremely preterm infants.
Study Design
This was a cohort study of 22-27+6/7 weeks gestational age infants during 2005-2011. DR-CPR was defined as chest compressions and/or epinephrine administration. Multivariable logistic regression was used to estimate odds ratios (OR) with 95% confidence intervals (CI) associated with DR-CPR; analysis was stratified by gestational age.
Results
Of 13 758 infants, 856 (6.2%) received DR-CPR. Infants 23+6/7 weeks 22-24-25+6/7 weeks . Infants receiving DR-CPR receiving DR-CPR had similar outcomes to had more severe intraventricular hemorrhage non-recipients (OR 1.36, 95% CI 1.07, 1.72). Infants 26-27+6/7 weeks receiving DR-CPR were more likely to die (OR 1.81, 95% CI 1.30, 2.51) and have intraventricular hemorrhage (OR 2.10, 95% CI 1.56, 2.82). Adjusted hospital DR-CPR rates varied widely (median 5.7%).
Conclusion
Premature infants receiving DR-CPR had worse outcomes. Mortality and morbidity varied by gestational age.
Introduction
Extreme prematurity is associated with mortality as well as short- and long- term health complications. During the 1980s and 1990s, there was a decrease in mortality rates of extremely premature infants.1, 2 However, recent studies suggest that the survival and neurodevelopmental outcomes of this population have reached a plateau.3, 4, 5, 6 Poor outcomes are even more prevalent in very low birth weight infants who have received delivery room cardiopulmonary resuscitation (DR-CPR), defined as chest compression and/or epinephrine.7, 8, 9, 10, 11, 12 A study of patients in the National Institute of Child Health and Development (NICHD) Neonatal Research Network (NRN) found that DRCPR was associated with increased mortality and neurodevelopmental impairment.10
Early studies of this population have been limited in the ability to identify potential associations between DR-CPR and outcomes at different gestational ages due to small numbers. The majority of more recent studies have focused on the experience of tertiary/regional (American Academy of Pediatrics (AAP) Level IV) neonatal intensive care units (NICUs), however a substantial portion of the care of preterm infants occurs at community level hospitals (AAP Level II and III).13 In fact, the majority of care of very preterm infants occurs at non-academic community level hospitals.14 Outcomes and practice patterns can vary according to the level of care and the general practice of providing DR-CPR in the community is not well described.
The question of the benefit or harm of DR-CPR in extremely preterm infants is a pressing one in the field of neonatal resuscitation. We performed a population-based study on the incidence of DR-CPR and its relationship to mortality and morbidities of extremely preterm infants (22+0/7-27+6/7 weeks), stratified by gestational age.
Methods
Study Population
The California Perinatal Quality Care Collaborative (CPQCC) prospectively collects data from the majority of NICUs in California. At the time of this study, the CPQCC included 130 NICUs, and we estimate that this dataset includes >95% of eligible premature infants in the state. The California Children's Services classifies NICUs into three levels which generally correspond to the American Academy of Pediatrics (AAP) Level designations as follows: Regional (Tertiary) NICUs (similar to AAP Level IV) provide mechanical ventilation and a full range of pediatric medical and surgical subspecialty services for patients, and outreach services to surrounding hospitals; Community NICUs (similar to AAP Level III) provide unrestricted care and ventilation to infants of all gestational ages and may have some availability of medical and surgical subspecialty services; Intermediate NICUs (similar to AAP Level II) provide care to a variably restricted population, ventilate only up to a specified number of hours, and refer all complicated cases to a higher level of care.13 However, there are some variations at the individual NICU level, as some Community NICUs may not offer surgical services, and not all Intermediate NICUs are equivalent to AAP Level II.15
The study cohort included infants born from January 2005 to December 2011, between 22+0/7 and 27+6/7 weeks gestation. Prior to applying additional exclusion criteria, there were 16 621 infants in the cohort. Infants included in the cohort were born in any one of the CPQCC associated NICUs, infants born at a non-CPQCC center were not included in the study. Exclusion criteria were similar to a previous multi-center study examining this question in the context of the NICHD NRN.10 In order to limit the possibility of errors in estimation of gestational age, infants who fell outside the 1st or 99th percentile for birth weight for their gestational age were excluded (n=76 infants). Infants with severe congenital abnormalities or congenital abnormalities of unknown severity were excluded (n = 957), as were infants with congenital viral infections (n = 73). Infants who received palliative care, defined as having received no resuscitation in the delivery room and died, or never received mechanical ventilation in the NICU and died, were also excluded (n = 1 757). The final analytic cohort included 13 758 infants.
Data analysis
DR-CPR was defined as chest compressions and/or administration of epinephrine in the delivery room. Per the CPQCC manual of definitions, delivery room chest compressions included any external cardiac massage performed immediately after birth.16 This was a binary variable, infants were not stratified based on duration of chest compressions. Delivery room intubation was not included in the identification of the DRCPR cohort. Student's t test or chi-squared test was used, as appropriate, to compare maternal and neonatal characteristics between those who did or did not receive DR-CPR. The Wilcoxon-Mann-Whitney test was used to compare Apgar scores. Variables examined included gestational age, birth weight, gender, multiple gestation, antenatal steroid exposure, maternal hypertension, chorioamnionitis, maternal bleeding, mode of delivery (vaginal or cesarean section), race, and Apgar scores. Data collection was performed by trained data abstractors based on definitions from the CPQCC and Vermont Oxford Network.16, 17
The primary outcome variable was death before hospital discharge, including if the death occurred after transfer to another hospital. Secondary outcomes included death in the first twelve hours of life, early onset sepsis (<72 hours after delivery), pneumothorax, patent ductus arteriosus, grade 3 or 4 intraventricular hemorrhage (IVH), cystic periventricular leukomalacia by 36 weeks corrected gestational age, a diagnosis of necrotizing enterocolitis (NEC), NEC requiring surgical intervention, postnatal steroids for chronic lung disease, postnatal steroids for any indication, and bronchopulmonary dysplasia (BPD, defined as an oxygen use at 36 weeks corrected GA). In cases in which infants were transferred out to another CPQCC NICU, the outcomes of in-hospital mortality, patent ductus arteriosus, IVH, periventricular leukomalacia, NEC, postnatal steroids, and BPD were followed across hospitals as part of the infant's initial hospital course.
A multivariable mixed regression model using the GLIMMIX procedure in SAS version 9.3 (SAS, Cary, NC) was used for risk adjustment. Based on previous literature, predictors in the model included gestation age, birth weight, gender, completion of antenatal steroids, and multiple gestation.18, 19 Hospital of birth was applied as a random effect. Analysis of the entire cohort, followed by subgroups stratified by gestational age was completed. Subgroups were as follows: 22 to 23+6/7 weeks GA, 24 to 25+6/7 weeks GA, 26 to 27+6/7 weeks GA. Odds ratios (OR) were estimated with 95% confidence intervals (CI). Sensitivity analyses for relevant secondary outcomes were performed for the entire cohort and after stratification based on gestational age. We excluded infants who died in the first twelve hours of life to test for possibility of bias due to early death.
In order to examine variation of care by hospital, we calculated rates of DR-CPR by birth hospital. We used a stepwise logistic regression model to account for significant factors that increased likelihood of receiving DR-CPR, and estimated a risk adjusted hospital rate for each hospital that cared for at least 20 eligible patients during the study period. Summary statistics were calculated for crude and adjusted rates. Institutional Board Review approval was obtained through Stanford University.
Results
During the study period, there were 13 758 infants who met inclusion criteria, 12 902 (93.8%) did not receive DR-CPR and 856 (6.2%) received DR-CPR. Infants who received DR-CPR were more likely to be younger, smaller, born in the setting of chorioamnionitis or a maternal bleed, and have lower 1 and 5 minute Apgar scores. Infants who received DR-CPR were less likely to be from multiple gestation pregnancies and have antenatal steroid exposure. There was no difference between the two groups in gender, mode of delivery, race, or presence of maternal hypertension (Table 1).
Table 1.
Characteristics of premature infants who received DR-CPR compared to infants who did not receive DR-CPR.
| DR-CPR n=856 | No DR-CPR n=12902 | P value | |
|---|---|---|---|
| GA in weeks (SD) | 24.9 (± 1.4) | 25-4 (± 1.4) | <0.0001 |
| Birth weight in grams (SD) | 767 (± 205) | 819 (± 204) | <0.0001 |
| Male sex | 53% | 54% | 0.70 |
| Multiple Birth | 18% | 24% | 0.0004 |
| Antenatal Steroids | 69% | 78% | <0.0001 |
| Maternal hypertension | 14% | 15% | 0.61 |
| Chorioamnionilis | 9% | 6% | <0.0001 |
| Maternal bleed | 26% | 21% | 0.0014 |
| Cesarean Delivery | 66% | 67% | 0.76 |
| Race | 0.19 | ||
| Non-Hispanic white | 17% | 14% | |
| Hispanic | 48% | 49% | |
| Blaek | 23% | 25% | |
| Asian PI | 9% | 9% | |
| Other | 5% | 3% | |
| Median 1 min APGAR seore (interquartile range) | 2 (1-3) | 5 (3-7) | <0.0001 |
| Median 5 min APGAR score (interquartile range) | 5 (3-7) | 7 (6-8) | <0.0001 |
DR-CPR – Delivery Room Cardiopulmonary Resuscitation; SD – Standard Deviation.
In multivariable regression analysis for the whole cohort of infants, those who received DR-CPR were more likely to have early onset sepsis, grade 3 or 4 IVH, receive post-natal steroids and die prior to hospital discharge. No difference was seen in the other secondary outcomes (Table 2). DR-CPR was not associated with improvements in the studied outcomes. In order to account for potential bias in early deaths leading to decreased incidence of secondary outcomes, we performed regression analyses for secondary outcomes excluding patients who died before 12 hours of age, and results were similar.
Table 2.
Outcomes of premature infants who received DR-CPR compared to infants who did not receive DR-CPR.
| Whole cohort | 22-23+6 | |||||||
|---|---|---|---|---|---|---|---|---|
| DR-CPR | No DR-CPR | DR-CPR | No DR-CPR | |||||
| n | 856 | 12902 | 142 | 1369 | ||||
| Outcome | n (%) | n (%) | Unadjusted p-value | AOR (95% CI) | n (%) | n (%) | Unadjusted p-value | AOR (95% CI) |
| In hospital mortality | 256 (30%) | 2509 (19%) | <0.0001 | 1.34 (1.12-1.59) | 84 (59%) | 802 (59%) | 0.90 | 1.13 (0.77-1.67) |
| Death<12 Ins | 67 (8%) | 619 (5%) | <0.0001 | 1.23 (0.91-1.65) | 36 (25%) | 334 (24%) | 0.80 | 1.30 (0.82-2.07) |
| Early onset sepsis (first 3d) | 38 (4%) | 315 (2%) | 0.003 | 1.77 (1.25-2.52) | 5 (4%) | 40 (3%) | 0.69 | 1.21 (0.47-3.14) |
| Pneumothorax | 30 (4%) | 289 (3%) | 0.42 | 1.06 (0.72-1.55) | 3 (2%) | 57 (4%) | 0.23 | 0.48 (0.15-1.57) |
| PDA (all intervention) | 520 (61%) | 7941 (62%) | 0.64 | 0.98 (0.84-1.14) | 66 (46%) | 682 (50%) | 0.45 | 0.85 (0.57-1.26) |
| Grade 3 or 4 IVH | 207 (24%) | 2054 (16%) | <0.0001 | 1.48 (1.25-1.76) | 35 (25%) | 355 (26%) | 0.74 | 0.92 (0.61-1.38) |
| PAX by 36 weeks | 36 (4%) | 497 (4%) | 0.60 | 1.01 (0.71-1.43) | 4 (3%) | 61 (4%) | 0.36 | 0.61 (0.22-1.70) |
| Late onset sepsis | 126 (15%) | 1623 (13%) | 0.07 | 1.14 (0.94-1.40) | 142 (13%) | 175 (13%) | 0.84 | 1.00 (0.59-1.68) |
| NEC diagnosis (stage not specified) | 36 (4%) | 451 (4%) | 0.27 | 1.16 (0.81-1.64) | 8 (6%) | 41 (3%) | 0.09 | 1.84 (0.83-4.09) |
| NEC requiring surgery | 21 (2%) | 289 (2%) | 0.68 | 1.04 (0.66-1.64) | 7(5%) | 31 (2%) | 0.05 | 2.29 (0.98-5.36) |
| Postnatal steroids for CLD | 68 (8%) | 870 (7%) | 0.18 | 1.14 (0.87-1.49) | 13 (9%) | 147 (11%) | 0.56 | 0.94 (0.50-1.77) |
| Postnatal steroids (all indications) | 353 (41%) | 4428 (34%) | <0.0001 | 1.24 (1.06-1.45) | 58 (41%) | 587 (43%) | 0.64 | 0.85 (0.58-1.24) |
| BPD (O2 at 36wk-int or cout) | 303 (35%) | 4686 (36%) | 0.59 | 0.96 (0.83-1.12) | 38 (27%) | 383 (28%) | 0.76 | 0.96 (0.62-1.48) |
| 24-25+6 | 26-27+6 | |||||||
|---|---|---|---|---|---|---|---|---|
| DR-CPR | No DR-CPR | DR-CPR | No DR-CPR | |||||
| n | 402 | 4891 | 312 | 6642 | ||||
| Outcome | n (%) | n (%) | Unadjusted p-value | AOR (95% CI) | n (%) | n (%) | Unadjusted p-value | AOR (95% CI) |
| In hospital mortality | 123 (31%) | 1146 (23%) | 0.001 | 1.26 (0.99-1.60) | 49 (16%) | 561 (8%) | <0.0001 | 1.81 (1.30-2.51) |
| Death<12 his | 22 (5%) | 220 (5%) | 0.37 | 1.02 (0.64-1.63) | 9 (3%) | 65 (1%) | 0.0013 | 2.35 (1.14-4.86) |
| Early onset sepsis (first 3d) | 16 (4%) | 141 (3%) | 0.21 | 1.42 (0.83-2.43) | 17 (5%) | 134 (2%) | <0.0001 | 2.84 (1.68-4.82) |
| Pneumothorax | 23 (6%) | 180 (4%) | 0.04 | 1.52 (0.97-2.40) | 4 (1%) | 152 (2%) | 0.24 | 0.57 (0.21-1.54) |
| PDA (all intervention) | 402 (68%) | 3371 (69%) | 0.83 | 1.09 (0.86-1.37) | 179 (57%) | 3888 (59%) | 0.68 | 0.91 (0.71-1.17) |
| Grade 3 or 4 IVH | 109 (27%) | 1019 (21%) | 0.003 | 1.36 (1.07-1.72) | 63 (20%) | 680 (10%) | <0.0001 | 2.10 (1.56-2.82) |
| PYL by 36 weeks | 20 (5%) | 210 (4%) | 0.52 | 1.12 (0.69-1.80) | 12 (4%) | 226 (3%) | 0.67 | 1.07 (0.59-1.94) |
| Late onset sepsis | 72 (18%) | 756 (15%) | 0.19 | 1.19 (0.91-1.57) | 35 (11%) | 692 (10%) | 0.65 | 1.05 (0.73-1.52) |
| NEC diagnosis (stage not specified) | 16 (4%) | 207 (4%) | 0.81 | 0.92 (0.55-1.56) | 12 (4%) | 203 (3%) | 0.43 | 1.22 (0.67-2.22) |
| NEC requiring surgery | 8 (2%) | 146 (3%) | 0.25 | 0.64 (0.31-1.32) | 6 (2%) | 112 (2%) | 0.75 | 1.14 (0.50-2.63) |
| Postnatal steroids for CLD | 39 (10%) | 452 (9%) | 0.76 | 1.14 (0.80-1.64) | 16 (5%) | 271 (4%) | 0.36 | 1.29 (0.75-2.23) |
| Postnatal steroids (all indications) | 207 (51%) | 2329 (44%) | 0.14 | 1.22 (0.98-1.52) | 88 (28%) | 1512 (23%) | 0.03 | 1.36 (1.03-1.79) |
| BPD (O2 at 36wk-int or cout) | 164 (41%) | 2130 (44%) | 0.28 | 0.94 (0.75-1.17) | 101 (32%) | 2173 (33%) | 0.9 | 0.91 (0.71-1.17) |
DR-CPR-Delivery Room Cardiopulmonary Resuscitation; AOR-Adjusted Odds Ratio; Cl-Confidence Interval; PDA-Patent Ductus Arteriosus; IVH-mtraventricularHemorrahge;PVL-PeriventricularLeukomalacia;NEC-Necrotizing Enterocolitis; CLD-Chronic Lung Disease; BPD-Bronchopulmonary Dysplasia
There were 1 511 infants in the 22 to 23+6/7 GA subgroup, of which 142 (9.4%) received DR-CPR. There were no differences in outcomes between those who did or did not receive DR-CPR for this GA group (Table 2). The 24 to 25+6/7 GA subgroup had 5 293 infants, of which 402 received DR-CPR (7.6%). Those who received DR-CPR were more likely to have grade 3 or 4 IVH (Table 2). In the 26 to 27+6/7 GA subgroup, there were 6 954 infants with 312 receiving DR-CPR (4.5%). Infants who received DR-CPR were more likely to die in the first twelve hours of life, have early onset sepsis, grade 3 or 4 IVH, require post-natal steroids, and die prior to hospital discharge. There was no difference between the two groups for other secondary outcomes (Table 2). DR-CPR was not associated with improvement of any of the studied outcomes for the individual subgroups. Notably, the relationship between DR-CPR and some outcomes differed according to gestational age. For example, grade 3 or 4 IVH was increased in both 24 to 25+6/7 week and 26 to 27+6/7 week infants, but not for 22 to 23+6/7 week infants. (Figure 1)
Figure 1.
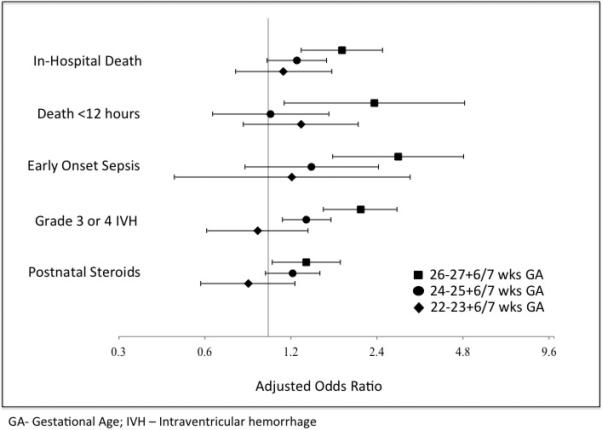
Forest Plot of key outcomes stratified by gestational age. Adjusted odds ratios of DR-CPR versus no DR-CPR with 95% confidence intervals.
DR-CPR rates differed by hospital level of care, with infants born at regional hospitals (324/4 417, 7.3%) more likely to receive DR-CPR than those born at community hospitals (423/7 784, 5.4%, p < 0.0001). Of the 115 hospitals that had at least 20 patients meeting study eligible criteria over the study period, there were 7 hospitals that had no patients receiving DR-CPR. The mean DR-CPR rate by hospital was 6.4%, median 5.5%, with interquartile range 2.7% to 9.5%. The highest hospital DR-CPR rate was 20.7%. With risk adjustment for multiple gestation, gestational age, and antenatal steroids, the mean rate was 6.3 %, median 5.7%, interquartile range 2.9% to 8.9%, with maximum hospital rate of 20.9% (Figure 2).
Figure 2.
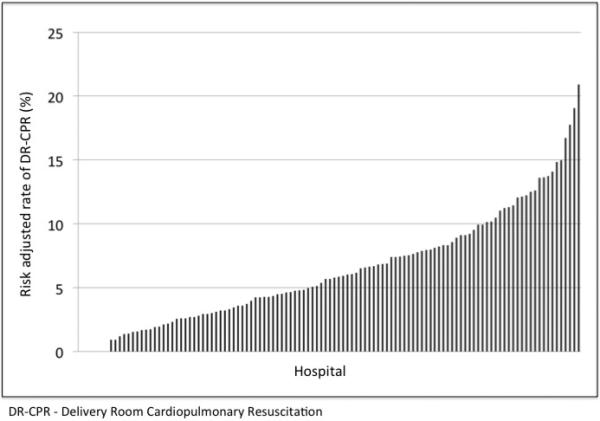
Adjusted DR-CPR rates by hospital of birth.
Discussion
In our population-based study of extremely preterm infants, those infants who received DR-CPR generally had worse outcomes than those who did not receive DRCPR. However, the degree of increased adverse outcomes differed by gestational age group. We also found notable variation between rates of DR-CPR across hospitals in California.
For the youngest GA group (22 to 23+6/7 weeks), DR-CPR was not associated with increased or decreased mortality or morbidity. However, the mortality rate of this subgroup, for both those who did and did not receive DR-CPR, was high (Table 2). More than half of these infants died, regardless of whether they received DR-CPR or not. Given the high mortality rate in the first 12 hours of life and prior to hospital discharge, DRCPR does not appear to be associated with differential short term outcomes in this population. The DR-CPR group was younger, smaller, had less antenatal steroid exposure and increased chorioamnionitis, all of which could contribute to increased mortality rate. It is possible that these mortality rates may have limited the observed rates of complications associated with prematurity. For example, the secondary outcome of NEC or BPD, which by definition is diagnosed at 36 weeks corrected GA, were probably less likely to be seen in this subgroup.
There were adverse outcomes associated with DR-CPR in the 24 to 25+6/7 week subgroup. Although the two groups did not have significantly different risk-adjusted mortality, there was a significantly increased risk for grade 3 and 4 IVH in infants who received DR-CPR. Previous literature has proposed that IVH in infants after DR-CPR is associated with blood pressure fluctuations during resuscitation.20 This could be related to variation in the quality and depth of chest compressions or the administration of epinephrine, given that it is vasoactive drug, which causes rapid changes in vascular tone and blood pressure. The association between Grade 3 and 4 IVH with DR-CPR in preterm infants has been previously reported by the NICHD NRN and Canadian Neonatal Network (CNN), however both reported additional adverse outcomes in populations that averaged 25 to 26 weeks.10, 12 This finding is different from our study and likely related to the stratification. Given that previous studies have looked at a wider range of infants, for example less than or greater than 1000g, it may be that the additional adverse outcomes reported actually primarily reflect those of the older infants. Although follow up data are not yet available for this cohort, it is known that extremely premature infants born between 24 to 28 weeks with grade 3 or 4 IVH have higher rates of neurodevelopmental impairment.21 Notably, one of the limitations regarding IVH outcomes is variable reporting between centers; CPQCC attempts to address this issue through data training and auditing, and by using a diagnosis of IVH that is based on radiology reports.
The findings of the cohort as a whole largely reflect those of the 26 to 27+6/7 week GA subgroup, which represents over 50% of the total cohort. The analysis of both the whole cohort and 26 to 27+6/7 GA infants who received DR-CPR showed increased mortality and morbidity associated with DR-CPR (Table 2). The adverse outcomes described mirror the findings of the NICHD NRN and CNN, though we did not find a difference in BPD rates.10, 12 While infants in the 26 to 27+6/7 week group were less likely to receive DR-CPR than 22 to 23+6/7 week infants, (4.6% versus 9.4%), they still had significant adverse outcomes. Although there may be presumptions that these relatively more mature infants could fare better, even with aggressive delivery room resuscitation, these results suggest that this may not be the case.
Our study demonstrates the importance of stratifying outcomes by GA when possible in observational studies of preterm infants (Figure 1). As physiologic differences may be present even with one to two weeks of in utero growth, therapies are likely to have differential effects based on gestational age. Even within a relatively narrowly defined group of extremely preterm infants, we found that the overall result was heavily influenced by the oldest and largest group of 26 to 27+6/7 week GA infants, and that results varied according to gestational age strata.
While some characteristics of those who did and did not receive DR-CPR were similar, such as gender and race, there were some differences that could impact neonatal outcomes. Higher birth weight, gestational age, and antenatal steroid exposure appear to be protective features, and we controlled for these variables in multivariable regression.18, 19 Another difference was the incidence of chorioamnionitis, which likely plays a role in the incidence of early onset sepsis and could explain the increase in early onset sepsis in the 26 to 27+6 week subgroup. There were also increased rates of maternal hemorrhage in the group receiving DR-CPR. It is unclear how maternal hemorrhage might affect the infant, however there may be certain circumstances like vasa previa that impact the infant's circulatory status and response to resuscitation. It may be that DR-CPR is a general marker of illness severity at the time of birth, as reflected in the lower Apgar scores of the DR-CPR cohort, and could modify interpretation of outcomes.
A previous study of primarily academic medical centers in the Neonatal Research Network found much higher rates of DR-CPR (15%) in a similar population, compared to 6% in our study.10 Additionally the CNN, which is also a population based data network reported a DR-CPR rate of 10.9% in infants <1000g.12 We found that DR-CPR rates were highest in Regional NICUs (7.3%), yet still relatively low compared to the aforementioned studies. The majority of infants in this study were born at hospitals with community NICUs and the DR-CPR rate was 5.4% at those hospitals. The differences between regional and community NICUs is notable and consistent with previous literature showing decreased mortality in extremely preterm infants born in hospitals with high volume and high level of care.22, 23 The wide variation in adjusted DR-CPR rates across hospitals, even with the acceptance of the Neonatal Resuscitation Program algorithm, suggests that there is not consistent implementation of the resuscitation algorithm in this population. (Figure 2).24 The marked variation in DR-CPR rates is likely influenced by a variety of factors including the philosophy and judgment of the bedside clinician, who may or may not be a neonatologist, and individual hospital resuscitation thresholds/policies. One might suggest that the wide range of experience of those resuscitating extremely preterm infants is a limitation of this study, however it is arguably a strength, given that it accurately reflects the population-based experience. Thus these findings truly represent the community experience in the United States and can be applied more broadly than previously available data.
This study is limited by the lack of availability of long term follow-up. Considering our findings of a differential effect of DR-CPR by gestational age, there is likely benefit to further outcomes research in the youngest gestational age infants who do survive. Although we did not find a difference in death or in-hospital morbidities, it is possible that there are impacts on development and long-term morbidities. Although not specific to 22 to 23+6 week infants, worse long term outcomes after DR-CPR have been reported.10
Although this study suggests that DR-CPR is a risk factor for adverse outcomes, given that outcomes are better for 26-27+6/7 week infants who received DR-CPR than 24-25+6/7 week infants who did not, these data continue to reinforce the importance of gestational age and perhaps suggest that older infants are potentially more appropriate DR-CPR candidates. These findings may add to discussions with families of extremely preterm infants. While there are potential confounding factors, for example if an infant is more acutely ill, physicians and families would like to assume that any intervention will only improve the chance of survival and well-being of their infants. Thus the discussion around the benefit versus harm of DR-CPR continues.
In our population-based cohort of extremely preterm infants, we found that DRCPR is associated with adverse outcomes, particularly for those infants at somewhat higher gestational age (26 to 27+6/7 weeks). For those infants born at 22 to 23+6/7 weeks, overall mortality and morbidity was high, but there was not a differential impact of DR-CPR. Although current guidelines for neonatal resuscitation do not specify differences across GA groups for chest compressions and epinephrine use, further studies are needed to determine whether there may be differences in response to DR-CPR according to GA for extremely preterm infants.
Acknowledgements
The project was supported by the University of California, San Francisco's Clinical and Translational Science Institute Resident Research Funding Program and grant number K23HD068400, Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD). The content is solely the responsibility of the authors and does not necessarily represent the official views of the Eunice Kennedy Shriver NICHD, or NIH.
Footnotes
Conflicts of Interest:
The authors of this paper have no conflicts of interest.
Contributor Information
Sara C. Handley, Department of Pediatrics, University of California, San Francisco.
Yao Sun, Department of Pediatrics, University of California, San Francisco.
Myra H. Wyckoff, Department of Pediatrics, University of Texas Southwestern Medical Center.
Henry C. Lee, Department of Pediatrics, Stanford University, California Perinatal Quality Care Collaborative.
References
- 1.Hack M, Friedman H, Fanaroff AA. Outcomes of extremely low birth weight infants. Pediatrics. 1996;98(5):931–937. [PubMed] [Google Scholar]
- 2.Lemons JA, Bauer CR, Oh W, Korones SB, Papile LA, Stoll BJ, et al. Very low birth weight outcomes of the National Institute of Child health and human development neonatal research network, January 1995 through December 1996. NICHD Neonatal Research Network. Pediatrics. 2001;107(1):E1. doi: 10.1542/peds.107.1.e1. [DOI] [PubMed] [Google Scholar]
- 3.Fanaroff AA, Stoll BJ, Wright LL, Carlo WA, Ehrenkranz RA, Stark AR, et al. Trends in neonatal morbidity and mortality for very low birthweight infants. Am J Obstet Gynecol. 2007;196(2):147, e141–148. doi: 10.1016/j.ajog.2006.09.014. [DOI] [PubMed] [Google Scholar]
- 4.Hintz SR, Kendrick DE, Vohr BR, Poole WK, Higgins RD. Changes in neurodevelopmental outcomes at 18 to 22 months' corrected age among infants of less than 25 weeks' gestational age born in 1993-1999. Pediatrics. 2005;115(6):1645–1651. doi: 10.1542/peds.2004-2215. [DOI] [PubMed] [Google Scholar]
- 5.Horbar JD, Badger GJ, Carpenter JH, Fanaroff AA, Kilpatrick S, LaCorte M, et al. Trends in mortality and morbidity for very low birth weight infants, 1991-1999. Pediatrics. 2002;110(1 Pt 1):143–151. doi: 10.1542/peds.110.1.143. [DOI] [PubMed] [Google Scholar]
- 6.Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010;126(3):443–456. doi: 10.1542/peds.2009-2959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lantos JD, Miles SH, Silverstein MD, Stocking CB. Survival after cardiopulmonary resuscitation in babies of very low birth weight. Is CPR futile therapy? N Engl J Med. 1988;318(2):91–95. doi: 10.1056/NEJM198801143180206. [DOI] [PubMed] [Google Scholar]
- 8.Shah PS. Extensive cardiopulmonary resuscitation for VLBW and ELBW infants: a systematic review and meta-analyses. J Perinatol. 2009;29(10):655–661. doi: 10.1038/jp.2009.71. [DOI] [PubMed] [Google Scholar]
- 9.Shah PS, Shah P, Tai KF. Chest compression and/or epinephrine at birth for preterm infants <32 weeks gestational age: matched cohort study of neonatal outcomes. J Perinatol. 2009;29(10):693–697. doi: 10.1038/jp.2009.70. [DOI] [PubMed] [Google Scholar]
- 10.Wyckoff MH, Salhab WA, Heyne RJ, Kendrick DE, Stoll BJ, Laptook AR. Outcome of extremely low birth weight infants who received delivery room cardiopulmonary resuscitation. J Pediatr. 2012;160(2):239–244. e232. doi: 10.1016/j.jpeds.2011.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Frontanes A, Garcia-Fragoso L, Garcia I, Rivera J, Valcarcel M. Outcome of very-low-birth-weight infants who received epinephrine in the delivery room. Resuscitation. 2011;82(4):427–430. doi: 10.1016/j.resuscitation.2010.11.020. [DOI] [PubMed] [Google Scholar]
- 12.Soraisham AS, Lodha AK, Singhal N, Aziz K, Yang J, Lee SK, et al. Neonatal outcomes following extensive cardiopulmonary resuscitation in the delivery room for infants born at less than 33 weeks gestational age. Resuscitation. 2014;85(2):238–243. doi: 10.1016/j.resuscitation.2013.10.016. [DOI] [PubMed] [Google Scholar]
- 13.Papile LA, Baley JE, Benitz W, Cummings J, Carlo WA, Kumar P, et al. Levels of neonatal care. Pediatrics. 2012;130(3):587–597. doi: 10.1542/peds.2012-1999. [DOI] [PubMed] [Google Scholar]
- 14.Lee HC, Bennett MV, Schulman J, Gould JB. Accounting for variation in length of NICU stay for extremely low birth weight infants. J Perinatol. 2013;33(11):872–876. doi: 10.1038/jp.2013.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. [2014 August 3];Perinatal Services Guidelines for Care: A Compilation of Current Standards. 2011 Available from: http://www.cdph.ca.gov/programs/rppc/Documents/MO-RPPC-PerinatalServicesGuidelines-CompilationofStandards-2011.pdf.
- 16. [2013 December 25];CPQCC Network Database 2008 Member Instructions for Electronic Data Submission (online) 2009 Version 03.09:[Available from: http://cpqcc.org/data/cpqcc_downloads.
- 17. [2013 December 25];Vermont Oxford Network Database Manual of Operations for Infants Born in 2010 (manual online) 2009 Release 14.0:[Available from: http://www.vtoxford.org/tools/2010ManualofOperationswithindex14.pdf.
- 18.Tyson JE, Parikh NA, Langer J, Green C, Higgins RD. Intensive care for extreme prematurity--moving beyond gestational age. N Engl J Med. 2008;358(16):1672–1681. doi: 10.1056/NEJMoa073059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee HC, Green C, Hintz SR, Tyson JE, Parikh NA, Langer J, et al. Prediction of death for extremely premature infants in a population-based cohort. Pediatrics. 2010;126(3):e644–650. doi: 10.1542/peds.2010-0097. [DOI] [PubMed] [Google Scholar]
- 20.Bada HS. Prevention of Intracranial Hemorrhage. NeoReviews. 2000;1(3):e48–e52. [Google Scholar]
- 21.Bolisetty S, Dhawan A, Abdel-Latif M, Bajuk B, Stack J, Lui K. Intraventricular Hemorrhage and Neurodevelopmental Outcomes in Extreme Preterm Infants. Pediatrics. 2014 doi: 10.1542/peds.2013-0372. [DOI] [PubMed] [Google Scholar]
- 22.Phibbs CS, Baker LC, Caughey AB, Danielsen B, Schmitt SK, Phibbs RH. Level and volume of neonatal intensive care and mortality in very-low-birth-weight infants. N Engl J Med. 2007;356(21):2165–2175. doi: 10.1056/NEJMsa065029. [DOI] [PubMed] [Google Scholar]
- 23.Watson SI, Arulampalam W, Petrou S, Marlow N, Morgan AS, Draper ES, et al. The effects of designation and volume of neonatal care on mortality and morbidity outcomes of very preterm infants in England: retrospective population-based cohort study. BMJ Open. 2014;4(7):e004856. doi: 10.1136/bmjopen-2014-004856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Textbook of Neonatal Resuscitation. 6th edn. American Academy of Pediatrics and American Heart Association; United States of America: 2011. [Google Scholar]


