Abstract
Purpose
Cartilage hair hypoplasia (CHH) is a rare metaphyseal chondrodysplasia characterized by short stature and short limbs, found primarily in Amish and Finnish populations. Cartilage hair hypoplasia is caused by mutations in the RMRP gene located on chromosome 9p13.3. The disorder has several characteristic orthopaedic manifestations, including joint laxity, limited elbow extension, ankle varus, and genu varum. Immunodeficiency is of concern in most cases. Although patients exhibit orthopaedic problems, the orthopaedic literature on CHH patients is scant at best. The objective of this study was to characterize the orthopaedic manifestations of CHH based on the authors’ unique access to the largest collection of CHH patients ever reported.
Methods
The authors examined charts and/or radiographs in 135 cases of CHH. We analyzed the orthopaedic manifestations to better characterize and further understand the orthopaedic surgeon’s role in this disorder. In addition to describing the clinical characteristics, we report on our surgical experience in caring for CHH patients.
Results
Genu varum, with or without knee pain, is the most common reason a patient with CHH will seek orthopaedic consultation. Of the cases reviewed, 32 patients had undergone surgery, most commonly to correct genu varum.
Conclusion
This paper characterizes the orthopaedic manifestations of CHH. Characterizing this condition in the orthopaedic literature will likely assist orthopaedic surgeons in establishing a correct diagnosis and appreciating the orthopaedic manifestations. It is important that the accompanying medical conditions are appreciated and evaluated.
Electronic supplementary material
The online version of this article (doi:10.1007/s11832-015-0646-z) contains supplementary material, which is available to authorized users.
Keywords: Pathoanatomy, McKusick-type metaphyseal chondrodysplasia, Cartilage hair hypoplasia
Introduction
McKusick et al. [1], in an extensive study of the Old Order Amish community in eastern Pennsylvania, clearly elucidated in 77 cases a distinct type of short-limb dwarfism termed cartilage hair hypoplasia, also known as McKusick metaphyseal chondrodysplasia. McKusick presented evidence that this disorder was inherited as an autosomal recessive with a frequency of 1–2 per thousand live births in the Old Order Amish [2].
Cartilage hair hypoplasia (CHH) has been reported in many countries and many peoples outside the Amish community [3–14]. The largest population of cases outside the Amish community is found in Finland, where Mäkitie in 1992 described the incidence as roughly 1:23,000 live births [15]. Mäkitie and colleagues, in several excellent publications, have described several associations of CHH beyond the presence of chondrodysplasia [5, 16–24]. CHH is associated with a broad spectrum of mild-to-moderate, cell-mediated immunodysfunction, including occasionally severe combined immune deficiency, neutropenia, lymphopenia, disordered erythrogenesis, and a predisposition to lymphoma [19–22, 25–37]. Recurring infections are commonly seen in 60 % of all patients, likely linked to the immunodeficiency. The severity is reflected in a report by Rider et al. [38] in which 32 % of their patients (25) had recurrent infections, with two requiring bone marrow transplantation [39–41]. Individuals affected with CHH can have marked impairment of T-lymphocyte function due to an intrinsic defect in cell proliferation. Beta cells and fibroblasts also show defective proliferation. Morbidity and mortality to varicella is clearly increased in CHH, but there is no apparent increase in susceptibility to severe or fatal infections with other viruses. Morbidity and mortality are directly related to immune dysfunction. The presence of infections, particularly in the first 2 years of life in children, is increased in frequency and hematologic malignancy is increased in adults. Mäkitie et al. [23] in 1999 reported an increased incidence of non-Hodgkin lymphoma and an excess risk of basal cell carcinoma in CHH. Others have reported similar findings, most commonly non-Hodgkin lymphoma [22, 23, 35, 36, 42–44]. Anemia is also commonly encountered and significantly correlated with the severity of immunodeficiency and growth failure.
It has been clearly documented that CHH is caused by mutations in the RMRP (RNA component of mitochondrial RNA processing endoribonuclease) gene located on chromosome 9p13–p12 and more recently narrowed to chromosome 9p13.3 [15, 16, 38, 45–60]. The RMRP 70A→G mutation is the most frequent ancestral mutation seen in Finnish and Old Order Amish people.
Patients with CHH are classically short at birth and the shortening may increase in the first 2 years of life. A very weak or absent pubertal growth spurt has been reported by Mäkitie.
Typically, the patients present with fine, sparse, light-colored, “silky” short hair that breaks easily. McKusick et al. [1, 2] and Coupe and Lowry [61] examined the hair grossly, microscopically, and in diameter. The hair has been demonstrated to be diminished in caliber and lacks a pigmented core, a finding also observed by van der Burgt et al. [3]. The hands are short and “pudgy” with marked excessive joint laxity, particularly of the metacarpal, phalangeal, and interphalangeal joints [1, 62]. The elbows typically lack full extension. Bowing of the lower extremities and disproportionate length of the fibula compared to the shortened tibia is consistent, although unproven as the cause of the ankle varus, resulting in ankle varus from relative fibular overgrowth (Fig. 1).
Fig. 1.
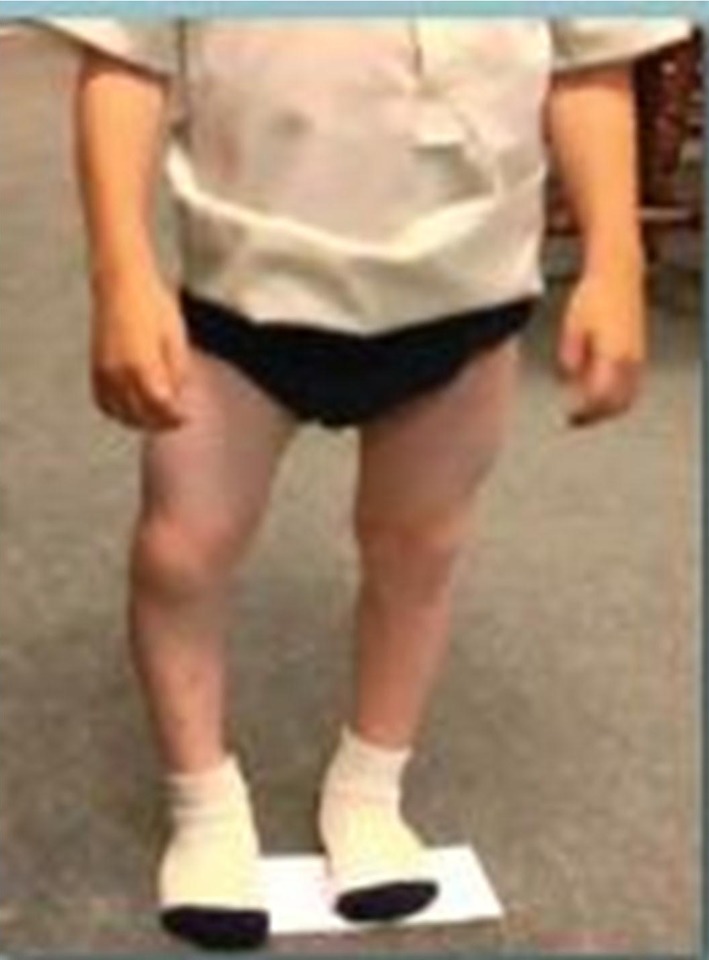
Frontal view demonstrating bowing of right femur and tibia in patient with cartilage hair hypoplasia
Mäkitie et al. reported that the median adult height was 131.1 cm (range 110.7–149.0 cm) for males and 122.5 cm (range 103.7–137.4 cm) for females [18]. The radius and ulna are commonly shorter than the humerus and the femur shorter than the tibia, with the tibia being more severely involved than the fibula.
Several nonskeletal issues are of considerable importance in association with CHH. In the McKusick series, a few patients had evidence of aganglionic colon, two patients died of varicella, and three others had had virulent infections with varicella. Intestinal malabsorption and Hirschsprung disease has been reported in association with CHH [1, 63]. Mäkitie et al. [24] estimated the incidence of these two disorders as occurring in 9 % of the patients. Prominence of the upper sternal region has been mentioned in the literature but is of no functional significance, but can be useful in diagnosis.
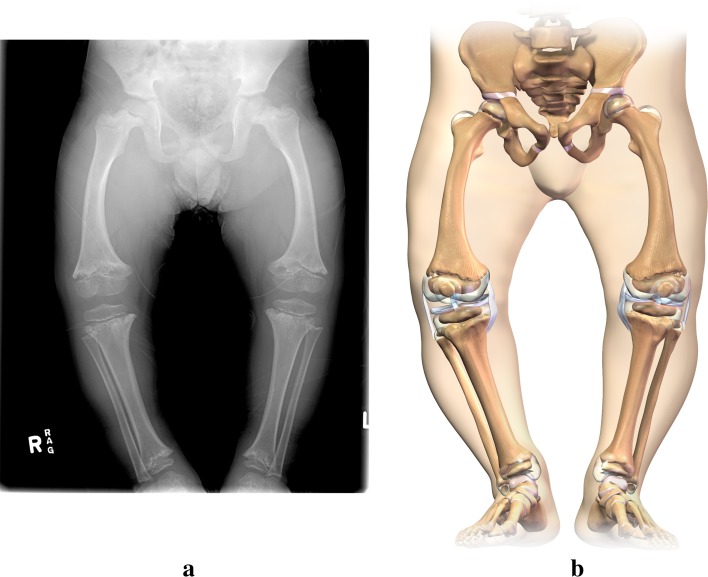
Typically, the ossification centers of the epiphyses are radiographically normal. Metaphyseal abnormalities reflecting the chondrodysplasia are manifested by flaring, cupping, marginal serration, fragmentation, and scalloping of the metaphyses of the tubular bones, most particularly seen at the knee [1, 3, 17, 64–66] (Fig. 2a, b).
Fig. 2.
a Anteroposterior radiograph of the lower extremity in cartilage hair hypoplasia demonstrating bowing. b Illustration depicting the osseous and cartilaginous deformities of lower extremities in cartilage hair hypoplasia
The hips are typically spared these findings. There are irregular cystic radiolucencies in the metaphyses with extension into the diaphyses. Abnormal epiphyseal shape occurs, seemingly as a consequence of the deformed metaphyseal region. There is marked shortening of the metacarpals, metatarsals, and phalanges, with metaphyseal cupping and “cone-shaped” epiphyses. Costochondral junctions have reflected mild flaring of the lower ribcage, slightly anterior angulated sternum, and lumbar lordosis. There is commonly delay in appearance of the proximal femoral epiphyses but absence of the proximal femoral metaphyseal changes that are typical at the knee. The entire metaphyseal region is widened and flared at the knee and ankle. The skull and spine are normal. Ray and Dorst [66] also reported mild-to-moderate C1 and C2 subluxation. Mäkitie et al. [17], however, reported in 1992 that none of 26 patients had cervical subluxation. Lachman [67] mentioned that several patients showed forward subluxation of C1 and C2 in flexion (lateral C-spine X-rays taken in neutral flexion and extension).
Mäkitie et al. demonstrated that the growth retardation in CHH correlates well with the severity of the metaphyseal changes [18, 68]. Ray and Dorst [66] reported that one-third of elbows showed lateral subluxation or dislocation of the radial head. Histologic examination was performed in one of McKusick’s cases and demonstrated paucity of cartilage cells and deficiency in columnar organization that was interpreted as cartilage hypoplasia. We could find no infrastructural histopathologic information in the literature to date. Prenatal diagnosis has been reported [69, 70].
A summary of the orthopaedic abnormalities that are typical in this disorder comprises significant joint laxity, particularly of the hands and feet, limited elbow extension, bowing of the lower leg and thigh, increased lumbar lordosis, varus deformity of the ankle due to fibular overgrowth, occasional atlantoaxial radiographic hypermobility, and scoliosis that rarely requires orthopaedic treatment.
Material: data
A unique opportunity was afforded the authors to access data on a large number of patients through clinical examinations, records, and radiographs of the largest accumulation of CHH cases so far encountered in North America. Approval from our institutional review board was obtained. Information was obtained from the files of the senior author (D.S.W.) on 135 patients with CHH comprising 63 males and 72 females: 65 Old Order Amish and 70 non-Amish patients. Documentation of diagnosis was obtained by clinical examination (26 patients), radiographic examination (82 patients), or genetic diagnosis (27 patients). Complete physical examination was available on 75 patients and the remainder was included based on radiographic assessment and/or genetic documentation.
For the purpose of this paper, we have arbitrarily divided the orthopaedic characteristics of CHH into typical (found in over 90 % of cases), frequent (50 % or more), occasional (<50 %), and rare. Radiographic abnormalities associated with CHH were similarly separated by frequency.
Results
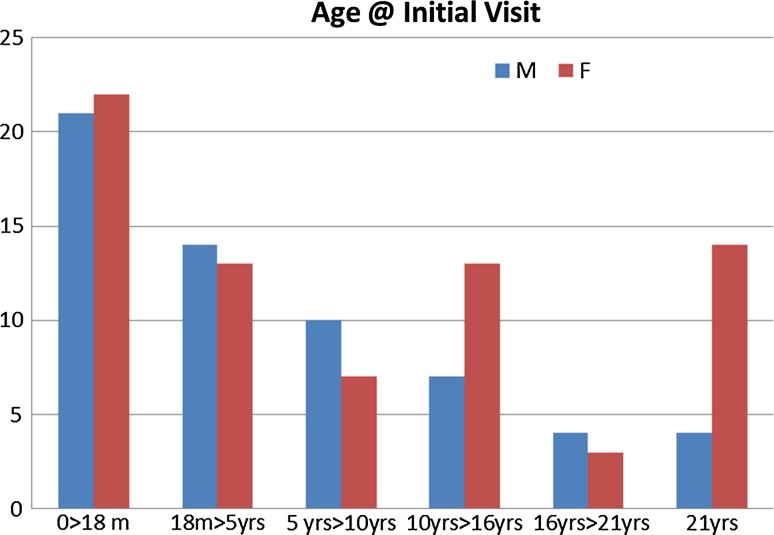
The age distribution of the patients at initial visit is shown in Fig. 3. The clinical findings seen in our population of 135 patients are recorded in Table 1 in order of their frequency. Likewise, in Table 2, radiographic features encountered are presented in accordance with their relative frequency. In our series, 27 % of the cases where radiographs were reviewed (19/71) showed coxa vara radiographically.
Fig. 3.
Patient age distribution at initial visit (m months, yrs years, M male, F female)
Table 1.
Frequency of clinical findings in CHH
| Clinical manifestation | Cases/patients reviewed | % Occurrence | Frequency |
|---|---|---|---|
| Short-limbed disproportionate dwarfism | 75/75 | 100 | Typical |
| Varus deformity of femur | 77/83 | 93 | Typical |
| Short pudgy hands with marked joint laxity | 75/75 | 100 | Typical |
| Varus deformity of tibia | 84/96 | 88 | Frequent |
| Diminished elbow extension | 56/69 | 81 | Frequent |
| Ankle varus deformity | 85/96 | 87 | Frequent |
Table 2.
Frequency of radiographic findings in CHH
| Radiographic manifestation | Cases/patients reviewed | % Occurrence | Frequency |
|---|---|---|---|
| Disproportionate short-limb dwarfism | 75/75 | 100 | Typical |
| Metaphyseal abnormalities of femur and tibia | 75/75 | 100 | Typical |
| Shortening of the metacarpals and phalanges | 75/75 | 100 | Typical |
| Genu varum | 85/96 | 87 | Frequent |
| Coxa vara | 19/71 | 27 | Occasional |
Several surgical procedures were performed on patients with angular and/or rotational deformity. In this series of patients, roughly 43 % had undergone surgical realignment of the lower extremities. The ages at surgery ranged from 2 to 27 years, with an average of 11.7 years and mean of 14.5 years. We did not encounter in our patients any cases that required cervical surgical stabilization (12 cases with lateral view of C-spine in neutral flexion and extension).
Discussion
It is interesting that over two-thirds of the patients in McKusick’s study derive ancestry from one John Miller who was married to Catherine Hochstetler. Over 70 % of the patients trace their ancestry to Jacob Hochstetler, a relative of Catherine. Eighty percent of parents of the 50 affected patients studied by McKusick were descended from either Jacob or Catherine Hochstetler, who immigrated to the United States in the mid-1700s. Amish migration has resulted in many cases seen in other states, particularly in Ohio at present [1, 2].
The orthopaedic manifestations that have clinical significance are related to the metaphyseal chondrodysplasia resulting in bowing, angulation, and rotational deformities. Femoral bowing and coxa vara are occasionally severe enough to warrant surgical intervention by osteotomy and realignment. In the lower leg, the shortening and bowing of the tibia and the ankle varus appears directly related to the relative overgrowth of the fibula.
It would appear that the loss of elbow extension, although typically seen, is most likely related to radial head subluxation or dislocation [27]. This is probably the best explanation, inasmuch as the other joints generally demonstrate increased laxity. Although typically there is lack of elbow extension, we have not encountered a single patient who had any issues of consequence with that loss of motion.
The most common mutation in the RMRP gene located on chromosome 9p13.3 is identical in the Old Order Amish population and the Finnish population. Although C1-2 subluxation has been described, we have not, in our series, encountered any cases that have required stabilization nor have we seen any cases of scoliosis that required orthopaedic management.
Coxa vara is likely due to a combination of relative discrepancy in growth of the upper femoral physis to trochanteric growth (slower proximal femoral growth plate in relation to trochanter and neck) combined with bowing and shortening of the femur, although this is admittedly speculative (Fig. 4). The severity of the bowing of the femur and tibia appears to correlate with the extent of metaphyseal radiographic changes.
Fig. 4.
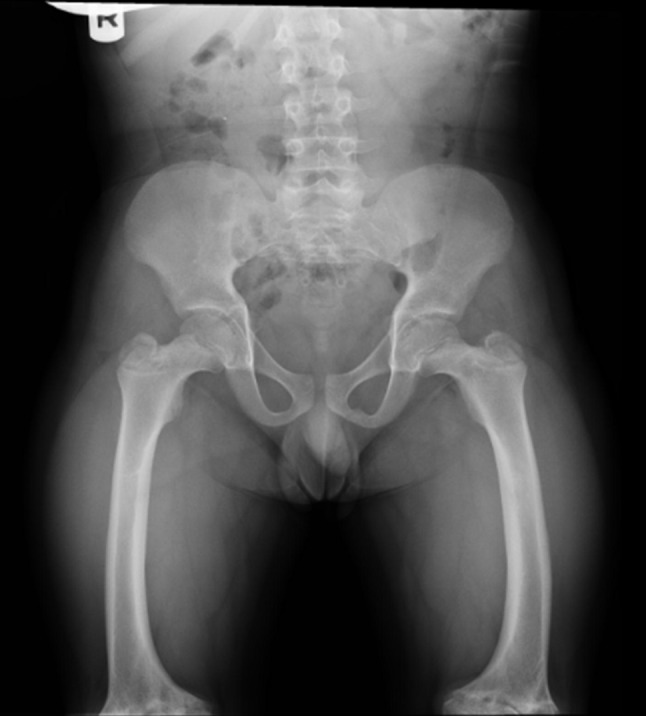
Anteroposterior radiograph demonstrating coxa vara and femoral bowing
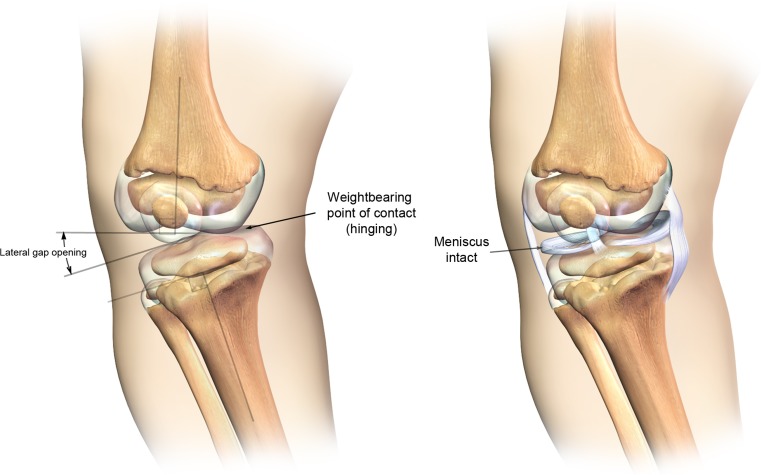
The metaphyseal alterations in the magnified illustration (Fig. 5) show a convoluted pattern of the bony metaphysis and junctural physis. Fibular overgrowth and joint laxity and genu varum coexisted but causation could not be established in this review. Computer reconstructions of the cartilaginous distal femoral condyles demonstrate a weight-bearing point of contact on the medial tibial plateau while the lateral portion of the knee shows an opening gap. Online Resource 1 depicts a rotating 3D computer model of Fig. 5.
Fig. 5.
Illustration of osseous and cartilaginous abnormalities in frontal view
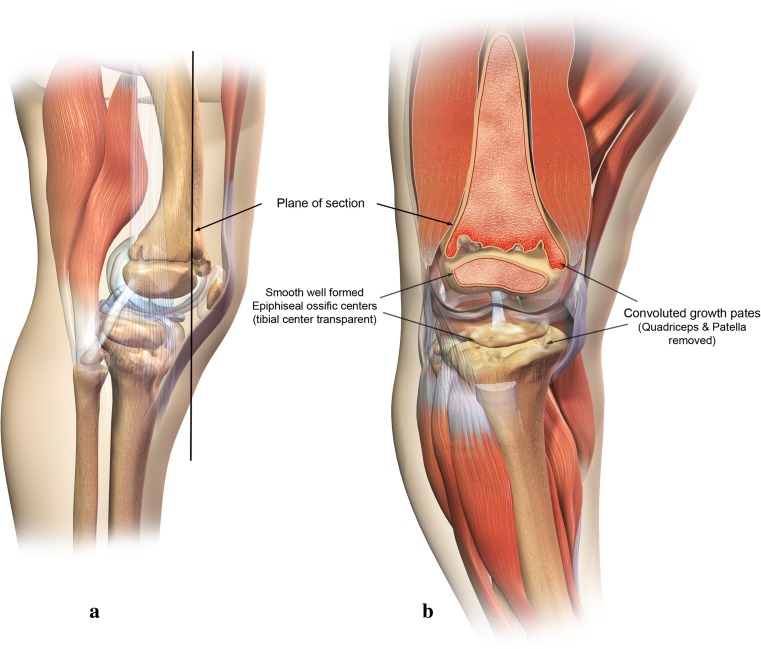
The convoluted pattern of the bony metaphysis likely reflects irregular cartilaginous invaginations into the ossification zone of the metaphysis from a disordered maturation process within the physis (Fig. 6a, b). Online Resource 2 depicts a rotating 3D computer model of Fig. 6.
Fig. 6.
a Lateral view illustration of typical knee anatomy in cartilage hair hypoplasia. b Anterior illustration of typical knee anatomy in cartilage hair hypoplasia (patella removed)
Likewise, even in the face of excessive laxity of the joints of the hand and concomitant shortness, we did not encounter any clinically significant complaints. Inasmuch as a large number of patients were Old Order Amish (65 patients), the usual types of employment probably correlate with the lack of complaints (blacksmith, farmer, lumber work, rough and finish carpenter).
Conclusion
The spectrum of orthopaedic manifestations typically and frequently seen in CHH has been elucidated in this paper based on this study of the largest group of patients so far reported in the North American literature. Thirty-four percent of 95 patients with the condition had undergone surgical realignment osteotomies of the long bones of the lower extremities.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
The authors wish to express their appreciation to Richard Pauli, M.D., Ph.D., for providing access to non-Amish patients’ information and to the Women’s Board of Akron Children’s Hospital for their research funding support. This study was supported in part by a grant from the Women’s Board of Akron Children’s Hospital.
Conflict of interest
The authors declare no conflict of interest.
References
- 1.McKusick VA, Eldridge R, Hostetler JA, Ruangwit U, Egeland JA. Dwarfism in the Amish. I. Cartilage-hair hypoplasia. Bull Johns Hopkins Hosp. 1965;116:285–326. [PubMed] [Google Scholar]
- 2.McKusick VA. Medical genetic studies of the amish: selected papers, assembled with commentary. Baltimore: Johns Hopkins University Press; 1978. [Google Scholar]
- 3.van der Burgt I, Haraldsson A, Oosterwijk JC, van Essen AJ, Weemaes C, Hamel B. Cartilage hair hypoplasia, metaphyseal chondrodysplasia type McKusick: description of seven patients and review of the literature. Am J Med Genet. 1991;41:371–380. doi: 10.1002/ajmg.1320410320. [DOI] [PubMed] [Google Scholar]
- 4.Lowry RB, Wood BJ, Birkbeck JA, Padwick PH. Cartilage-hair hypoplasia. A rare and recessive cause of dwarfism. Clin Pediatr (Phila) 1970;9:44–46. doi: 10.1177/000992287000900112. [DOI] [PubMed] [Google Scholar]
- 5.Mäkitie O, Sulisalo T, de la Chapelle A, Kaitila I. Cartilage-hair hypoplasia. J Med Genet. 1995;32:39–43. doi: 10.1136/jmg.32.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.RönningO Myllarniemi S, Perheentupa J. Craniofacial and dental characteristics of cartilage-hair hypoplasia. Cleft Palate J. 1978;15:49–55. [PubMed] [Google Scholar]
- 7.Sensenbrenner JA. Metaphyseal chondrodysplasia, McKusick type (cartilage hair hypoplasia) Birth Defects Orig Artic Ser. 1974;10:422–424. [PubMed] [Google Scholar]
- 8.Fryns JP, Pedersen JC, Pardon W, Van den Berghe H. Cartilage-hair hypoplasia. Acta Paediatr Belg. 1980;33:265–267. [PubMed] [Google Scholar]
- 9.Halle MA, Collipp PJ, Roginsky M. Cartilage-hair hypoplasia in childhood. N Y State J Med. 1970;70:2705–2708. [PubMed] [Google Scholar]
- 10.Siggers DC, Burke JB, Morris B, Normand IC, Tanner JM, Williamson DA. Cartilage hair hypoplasia. Postgrad Med. 1977;J53:473–479. doi: 10.1136/pgmj.53.622.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Irwin GA. Cartilage-hair hypoplasia (CHH) variant of familial metaphyseal dysostosis. Radiology. 1966;86:926–928. doi: 10.1148/86.5.926. [DOI] [PubMed] [Google Scholar]
- 12.Wiedemann HR, Spranger J, Kosenow W. Knorpel-Haar-Hypoplasia. Arch Kinderheilk. 1967;176:74–85. [PubMed] [Google Scholar]
- 13.Beals RK. Cartilage-hair hypoplasia. A case report. J Bone Joint Surg Am. 1968;50:1245–1249. [PubMed] [Google Scholar]
- 14.Boothby CB, Bower BD. Cartilage hair hypoplasia. Arch Dis Child. 1973;48:918–921. doi: 10.1136/adc.48.11.918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mäkitie O. Cartilage-hair hypoplasia in Finland: epidemiological and genetic aspects of 107 patients. J Med Genet. 1992;29:652–655. doi: 10.1136/jmg.29.9.652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mäkitie O, Sulisalo T, de la Chapelle A, Kaitila I. Cartilage-hair hypoplasia. J Med Genet. 1995;32:39–43. doi: 10.1136/jmg.32.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mäkitie O, Marttinen E, Kaitila I. Skeletal growth in cartilage-hair hypoplasia. A radiological study of 82 patients. Pediatr Radiol. 1992;22:434–439. doi: 10.1007/BF02013505. [DOI] [PubMed] [Google Scholar]
- 18.Mäkitie O, Perheentupa J, Kaitila I. Growth in cartilage-hair hypoplasia. Pediatr Res. 1992;31:176–180. doi: 10.1203/00006450-199202000-00018. [DOI] [PubMed] [Google Scholar]
- 19.Mäkitie O, Kaitila I, Savilahti E. Susceptibility to infections and in vitro immune functions in cartilage-hair hypoplasia. Eur J Pediatr. 1998;157:816–820. doi: 10.1007/s004310050943. [DOI] [PubMed] [Google Scholar]
- 20.Mäkitie O, Pukkala E, Kaitila I. Increased mortality in cartilage-hair hypoplasia. Arch Dis Child. 2001;84:65–67. doi: 10.1136/adc.84.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mäkitie O, Juvonen E, Dunkel L, Kaitila I, Siimes MA. Anemia in children with cartilage-hair hypoplasia is related to body growth and to the insulin-like growth factor system. J Clin Endocrinol Metab. 2000;85:563–568. doi: 10.1210/jcem.85.2.6339. [DOI] [PubMed] [Google Scholar]
- 22.Mäkitie O, Rajantie J, Kaitila I. Anaemia and macrocytosis—unrecognized features in cartilage-hair hypoplasia. Acta Paediatr. 1992;81:1026–1029. doi: 10.1111/j.1651-2227.1992.tb12168.x. [DOI] [PubMed] [Google Scholar]
- 23.Mäkitie O, Pukkala E, Teppo L, Kaitila I. Increased incidence of cancer in patients with cartilage-hair hypoplasia. J Pediatr. 1999;134:315–318. doi: 10.1016/S0022-3476(99)70456-7. [DOI] [PubMed] [Google Scholar]
- 24.Mäkitie O, Kaitila I, Rintala R. Hirschsprung disease associated with severe cartilage-hair hypoplasia. J Pediatr. 2001;138:929–931. doi: 10.1067/mpd.2001.113632. [DOI] [PubMed] [Google Scholar]
- 25.Kawasaki H, Kohdera U, Taniuchi S, Kobayashi Y. Cartilage-hair hypoplasia associated with IgG2 deficiency. Acta Paediatr Jpn. 1995;37:703–705. doi: 10.1111/j.1442-200X.1995.tb03409.x. [DOI] [PubMed] [Google Scholar]
- 26.Bocca G, Weemaes CM, van der Burgt I, Otten BJ. Growth hormone treatment in cartilage-hair hypoplasia: effects on growth and the immune system. J Pediatr Endocrinol Metab. 2004;17:47–54. doi: 10.1515/JPEM.2004.17.1.47. [DOI] [PubMed] [Google Scholar]
- 27.Wilson WG, Aylsworth AS, Folds JD, Whisnant JK. Cartilage-hair hypoplasia (metaphyseal chondrodysplasia, type McKusick) with combined immune deficiency: variable expression and development of immunologic functions in sibs. Birth Defects Orig Artic Ser. 1978;14:117–129. [PubMed] [Google Scholar]
- 28.Hong R. Associations of the skeletal and immune systems. Am J Med Genet. 1989;34:55–59. doi: 10.1002/ajmg.1320340111. [DOI] [PubMed] [Google Scholar]
- 29.Saulsbury FT, Winkelstein JA, Davis LE, Hsu SH, D’Souza BJ, Gutcher GR, Butler IJ. Combined immunodeficiency and vaccine-related poliomyelitis in a child with cartilage-hair hypoplasia. J Pediatr. 1975;86:868–872. doi: 10.1016/S0022-3476(75)80216-2. [DOI] [PubMed] [Google Scholar]
- 30.Castigli E, Irani AM, Geha RS, Chatila T. Defective expression of early activation genes in cartilage-hair hypoplasia (CHH) with severe combined immunodeficiency (SCID) Clin Exp Immunol. 1995;102:6–10. doi: 10.1111/j.1365-2249.1995.tb06628.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kooijman R, van der Burgt CJ, Weemaes CM, Haraldsson A, Scholtens EJ, Zegers BJ. T cell subsets and T cell function in cartilage-hair hypoplasia. Scand J Immunol. 1997;46:209–215. doi: 10.1046/j.1365-3083.1997.d01-112.x. [DOI] [PubMed] [Google Scholar]
- 32.Polmar SH, Pierce GF. Cartilage hair hypoplasia: immunological aspects and their clinical implications. Clin Immunol Immunopathol. 1986;40:87–93. doi: 10.1016/0090-1229(86)90071-1. [DOI] [PubMed] [Google Scholar]
- 33.Pierce GF, Polmar SH. Lymphocyte dysfunction in cartilage hair hypoplasia. II. Evidence for a cell cycle specific defect in T cell growth. Clin Exp Immunol. 1982;50:621–628. [PMC free article] [PubMed] [Google Scholar]
- 34.Trojak JE, Polmar SH, Winkelstein JA, Hsu S, Francomano C, Pierce GF, Scillian JJ, Gale AN, McKusick VA. Immunologic studies of cartilage-hair hypoplasia in the Amish. Johns Hopkins Med. 1981;J148:157–164. [PubMed] [Google Scholar]
- 35.Williams MS, Ettinger RS, Hermanns P, Lee B, Carlsson G, Taskinen M, Mäkitie O. The natural history of severe anemia in cartilage-hair hypoplasia. Am J Med Genet A. 2005;138:35–40. doi: 10.1002/ajmg.a.30902. [DOI] [PubMed] [Google Scholar]
- 36.Harris RE, Baehner RL, Gleiser S, Weaver DD, Hodes ME. Cartilage-hair hypoplasia, defective T-cell function, and Diamond-Blackfan anemia in an Amish child. Am J Med Genet. 1981;8:291–297. doi: 10.1002/ajmg.1320080306. [DOI] [PubMed] [Google Scholar]
- 37.Ashby GH, Evans DI. Cartilage hair hypoplasia with thrombocytopenic purpura, autoimmune haemolyticanaemia and cell-mediated immunodeficiency. J R Soc Med. 1986;79:113–114. doi: 10.1177/014107688607900216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rider NL, Morton DH, Puffenberger E, Hendrickson CL, Robinson DL, Strauss KA. Immunologic and clinical features of 25 Amish patients with RMRP 70 A→G cartilage hair hypoplasia. Clin Immunol. 2009;131:119–128. doi: 10.1016/j.clim.2008.11.001. [DOI] [PubMed] [Google Scholar]
- 39.Bordon V, Gennery AR, Slatter MA, Vandecruys E, Laureys G, Veys P, Qasim W, Friedrich W, Wulfraat NM, Scherer F, Cant AJ, Fischer A, Cavazzana-Calvo M, Bredius RG, Notarangelo LD, Mazzolari E, Neven B, Güngör T. Inborn error working party of the European bone marrow transplantation (EBMT) group. Clinical and immunologic outcome of patients with cartilage hair hypoplasia after hematopoietic stem cell transplantation. Blood. 2010;116:27–35. doi: 10.1182/blood-2010-01-259168. [DOI] [PubMed] [Google Scholar]
- 40.Guggenheim R, Somech R, Grunebaum E, Atkinson A, Roifman CM. Bone marrow transplantation for cartilage-hair-hypoplasia. Bone Marrow Transpl. 2006;38:751–756. doi: 10.1038/sj.bmt.1705520. [DOI] [PubMed] [Google Scholar]
- 41.Berthet F, Siegrist CA, Ozsahin H, Tuchschmid P, Eich G, Superti-Furga A, Seger RA. Bone marrow transplantation in cartilage-hair hypoplasia: correction of the immunodeficiency but not of the chondrodysplasia. Eur J Pediatr. 1996;155:286–290. doi: 10.1007/BF02002714. [DOI] [PubMed] [Google Scholar]
- 42.Taskinen M, Ranki A, Pukkala E, Jeskanen L, Kaitila I, Mäkitie O. Extended follow-up of the Finnish cartilage-hair hypoplasia cohort confirms high incidence of non-Hodgkin lymphoma and basal cell carcinoma. Am J Med Genet A. 2008;146A:2370–2375. doi: 10.1002/ajmg.a.32478. [DOI] [PubMed] [Google Scholar]
- 43.Eisner JM, Russell M. Cartilage hair hypoplasia and multiple basal cell carcinomas. J Am Acad Dermatol. 2006;54:S8–S10. doi: 10.1016/j.jaad.2005.03.055. [DOI] [PubMed] [Google Scholar]
- 44.Roberts MA, Arnold RM. Hodgkin’s lymphoma in a child with cartilage-hair hypoplasia: case report. Mil Med. 1984;149:280–281. [PubMed] [Google Scholar]
- 45.Hermanns P, Bertuch AA, Bertin TK, Dawson B, Schmitt ME, Shaw C, Zabel B, Lee B. Consequences of mutations in the non-coding RMRP RNA in cartilage-hair hypoplasia. Hum Mol Genet. 2005;14:3723–3740. doi: 10.1093/hmg/ddi403. [DOI] [PubMed] [Google Scholar]
- 46.Hermanns P, Tran A, Munivez E, Carter S, Zabel B, Lee B, Leroy JG. RMRP mutations in cartilage-hair hypoplasia. Am J Med Genet A. 2006;140:2121–2130. doi: 10.1002/ajmg.a.31331. [DOI] [PubMed] [Google Scholar]
- 47.Narra MB, Shearer WT. Cartilage-hair hypoplasia and severe allergy. J Allergy Clin Immunol. 2009;123:1418–1419. doi: 10.1016/j.jaci.2009.02.036. [DOI] [PubMed] [Google Scholar]
- 48.Baradaran-Heravi A, Thiel C, Rauch A, Zenker M, Boerkoel CF, Kaitila I. Clinical and genetic distinction of Schimke immuno-osseous dysplasia and cartilage-hair hypoplasia. Am J Med Genet A. 2008;146A:2013–2017. doi: 10.1002/ajmg.a.32406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nakashima E, Tran JR, Welting TJ, Pruijn GJ, Hirose Y, Nishimura G, Ohashi H, Schurman SH, Cheng J, Candotti F, Nagaraja R, Ikegawa S, Schlessinger D. Cartilage hair hypoplasia mutations that lead to RMRP promoter inefficiency or RNA transcript instability. Am J Med Genet A. 2007;143A:2675–2681. doi: 10.1002/ajmg.a.32053. [DOI] [PubMed] [Google Scholar]
- 50.Ridanpää M, van Eenennaam H, Pelin K, Chadwick R, Johnson C, Yuan B, vanVenrooij W, Pruijn G, Salmela R, Rockas S, Mäkitie O, Kaitila I, de la Chapelle A. Mutations in the RNA component of RNase MRP cause a pleiotropic human disease, cartilage-hair hypoplasia. Cell. 2001;104:195–203. doi: 10.1016/S0092-8674(01)00205-7. [DOI] [PubMed] [Google Scholar]
- 51.Ridanpää M, Sistonen P, Rockas S, Rimoin DL, Mäkitie O, Kaitila I. Worldwide mutation spectrum in cartilage-hair hypoplasia: ancient founder origin of the major 70A→G mutation of the untranslated RMRP. Eur J Hum Genet. 2002;10:439–447. doi: 10.1038/sj.ejhg.5200824. [DOI] [PubMed] [Google Scholar]
- 52.Ridanpää M, Jain P, McKusick VA, Francomano CA, Kaitila I. The major mutation in the RMRP gene causing CHH among the Amish is the same as that found in most Finnish cases. Am J Med Genet C Semin Med Genet. 2003;121C:81–83. doi: 10.1002/ajmg.c.20006. [DOI] [PubMed] [Google Scholar]
- 53.Notarangelo LD, Roifman CM, Giliani S. Cartilage-hair hypoplasia: molecular basis and heterogeneity of the immunological phenotype. Curr Opin Allergy Clin Immunol. 2008;8:534–539. doi: 10.1097/ACI.0b013e328310fe7d. [DOI] [PubMed] [Google Scholar]
- 54.Thiel CT, Mortier G, Kaitila I, Reis A, Rauch A. Type and level of RMRP functional impairment predicts phenotype in the cartilage hair hypoplasia-anauxetic dysplasia spectrum. Am J Hum Genet. 2007;81:519–529. doi: 10.1086/521034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Matesic D, Hagan JB. Cartilage-hair hypoplasia. Mayo Clin Proc. 2007;82:655. doi: 10.1016/S0025-6196(11)61181-6. [DOI] [PubMed] [Google Scholar]
- 56.Vatanavicharn N, Visitsunthorn N, Pho-iam T, Jirapongsananuruk O, Pacharn P, Chokephaibulkit K, Limwongse C, Wasant P. An infant with cartilage-hair hypoplasia due to a novel homozygous mutation in the promoter region of the RMRP gene associated with chondrodysplasia and severe immuno deficiency. J Appl Genet. 2010;51:523–528. doi: 10.1007/BF03208884. [DOI] [PubMed] [Google Scholar]
- 57.Sulisalo T, Sistonen P, Hästbacka J, Wadelius C, Mäkitie O, de la Chapelle A, Kaitila I. Cartilage-hair hypoplasia gene assigned to chromosome 9 by linkage analysis. Nat Genet. 1993;3:338–341. doi: 10.1038/ng0493-338. [DOI] [PubMed] [Google Scholar]
- 58.Sulisalo T, Francomano CA, Sistonen P, Maher JF, McKusick VA, de la Chapelle A, Kaitila I. High-resolution genetic mapping of the cartilage-hair hypoplasia (CHH) gene in Amish and Finnish families. Genomics. 1994;20:347–353. doi: 10.1006/geno.1994.1187. [DOI] [PubMed] [Google Scholar]
- 59.Sulisalo T, van der Burgt I, Rimoin DL, Bonaventure J, Sillence D, Campbell JB, Chitayat D, Scott CI, de la Chapelle A, Sistonen P, Kaitila I. Genetic homogeneity of cartilage-hair hypoplasia. Hum Genet. 1995;95:157–160. doi: 10.1007/BF00209394. [DOI] [PubMed] [Google Scholar]
- 60.Sulisalo T, Sillence D, Wilson M, Ryynänen M, Kaitila I. Early prenatal diagnosis of cartilage-hair hypoplasia (CHH) with polymorphic DNA markers. Prenat Diagn. 1995;15:135–140. doi: 10.1002/pd.1970150205. [DOI] [PubMed] [Google Scholar]
- 61.Coupe RL, Lowry RB. Abnormality of the hair in cartilage-hair hypoplasia. Dermatologica. 1970;141:329–334. doi: 10.1159/000252497. [DOI] [PubMed] [Google Scholar]
- 62.Brennan TE, Pearson RW. Abnormal elastic tissue in cartilage-hair hypoplasia. Arch Dermatol. 1988;124:1411–1414. doi: 10.1001/archderm.1988.01670090067015. [DOI] [PubMed] [Google Scholar]
- 63.leMerrer M, Briard ML, Chauvet ML, Maroteaux P. Autosomal recessive metaphyseal chondrodysplasia and Hirschsprung’s disease. Ann Pediatr (Paris) 1991;38:27–30. [PubMed] [Google Scholar]
- 64.Glass RB, Tifft CJ. Radiologic changes in infancy in McKusick cartilage hair hypoplasia. Am J Med Genet. 1999;86:312–315. doi: 10.1002/(SICI)1096-8628(19991008)86:4<312::AID-AJMG2>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 65.Giedion A. Phalangeal cone-shaped epiphyses of the hand: their natural history, diagnostic sensitivity, and specificity in cartilage hair hypoplasia and the trichorhinophalangeal syndromes I and II. Pediatr Radiol. 1998;28:751–758. doi: 10.1007/s002470050460. [DOI] [PubMed] [Google Scholar]
- 66.Ray HC, Dorst JP. Cartilage-hair hypoplasia. In: Kaufman H, editor. Intrinsic diseases of bone. Basel: S. Karger; 1973. [Google Scholar]
- 67.Lachman RS. The cervical spine in the skeletal dysplasias and associated disorders. Pediatr Radiol. 1997;27:402–408. doi: 10.1007/s002470050156. [DOI] [PubMed] [Google Scholar]
- 68.Conwell LS, Hermanns P, Zankl A. Short stature and metaphyseal dysplasia due to cartilage-hair hypoplasia. J Pediatr Endocrinol Metab. 2008;21:209–211. doi: 10.1515/JPEM.2008.21.3.209. [DOI] [PubMed] [Google Scholar]
- 69.Dungan JS, Emerson DS, Phillips OP, Shulman LP. Cartilage-hair hypoplasia syndrome: implications for prenatal diagnosis. Fetal Diagn Ther. 1996;11:398–401. doi: 10.1159/000264353. [DOI] [PubMed] [Google Scholar]
- 70.Alanay Y, Krakow D, Rimoin DL, Lachman RS. Angulated femurs and the skeletal dysplasias: experience of the international skeletal dysplasia registry (1988–2006) Am J Med Genet A. 2007;143A:1159–1168. doi: 10.1002/ajmg.a.31711. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.