Abstract
Background:
We aimed to test a new approach for repairing cerebrospinal fluid (CSF) leaks and to determine the demographic, diagnostic, and treatment factors associated with the successful management of intracranial complication. Owing to the high frequency of endoscopic surgeries and the low cost of medical care in Iran, we decided to report our experience of reconstruction after CSF leaks.
Methods:
We retrospectively reviewed our experience in the diagnosis and management of CSF rhinorrhoea in Iran between 2005 and 2012. The locations of all pre-repair leaks were identified using simple, readily available methods. The follow-up time ranged from 2 to 72 months.
Results:
Of the 37 participants, 59.5% were men and the mean age was 33 years. The success rate was 86.1%, and the most common aetiological factor was trauma (57%). The most common location was the ethmoidal fovea (45.9%), followed by the sphenoid sinus (24.3%), the cribriform plate (13.5%), and the posterior table of the frontal sinus (5.4%).
Conclusion:
Medical care in Iran has considerable budget restrictions. This study advocates a practical method of treatment for patients in similar circumstances, with a success rate of 86.1% when compared to the 90.6% achieved with other techniques. Keywords: cerebrospinal fluid leak, endoscopic, intracranial pressure ICP
Keywords: cerebrospinal fluid leak, endoscopic, intracranial pressure ICP, skull base defect
Introduction
Trauma and iatrogenic injury during functional endoscopic sinus surgery (FESS) are widely accepted causes of cerebrospinal fluid (CSF) rhinorrhoea. Traumatic CSF rhinorrhoea causes 90% of all CSF leaks, of which 80% are due to motor vehicle accidents (1). In Iran, car accident-related mortality and morbidities are among the highest in the world, and the management of CSF leaks has a significant impact on the Iranian health system. In addition, iatrogenic CSF rhinorrhoea can follow transcranial endoscopic surgery via the skull base, and the growing number of such procedures implies that surgeons must be able to manage CSF rhinorrhoea adequately (2).
Identifying the aetiology of a CSF leak is an important consideration prior to surgical decision making. Its location and size, the shape of the bony and meningeal defects and the presence of concomitant intracranial hypertension or meningoencephalocele should be accurately ascertained. Confirmation of the presence and location of CSF leaks can be provided by intrathecal fluorescein, which is advocated as an adjunct in the endoscopic endonasal identification of CSF leaks and has a reported success rate as high as 96%. However, despite pre-operative imaging and stereotactic navigation, the precise location often remains unknown.
At present, transcranial approaches are frequently used to repair anterior skull base defects. Complications of these approaches include highly reported failure rates, high craniotomy-associated morbidity and brain retraction, such as potential hematoma, seizures, and anosmia (3). Less invasive endoscopic endonasal approaches are limited to defects less than 1.5 cm, although some authors have found no correlation between the success of endoscopic repairs and the size of the resulting defect. Other investigators have used cartilage or bone to repair larger defects and have reported successful repairs for defects larger than 2 cm (4). Therefore, transnasal endoscopic repair of CSF leaks has become a pre-dominant therapy for paediatric CSF rhinorrhoea (5) because it reduces the incidence of each major complication, such as meningitis and intracranial abscess, to less than 1% (3).
There are multiple techniques for endoscopic leak repairs and no single approach has obvious superiority. Although CSF rhinorrhoea is a simple concept, making a diagnosis and locating the leak can be problematic and expensive. Therefore, we believe it is necessary to introduce a simple and cheap diagnostic strategy. In Iran, the frequency of endoscopic surgery is high, the likelihood of iatrogenic CSF leaks is high, and appropriate means of repairing such leaks is low and the cost of medical care is low. Therefore, this study aimed to design a remedial method to alleviate the problem caused by CSF leakage. We aimed to test a new approach for repairing CSF leaks and to determine the demographic, diagnostic and treatment factors associated with the successful management of intracranial complications.
Methods
Study design and patient selection
We retrospectively reviewed the demographics, aetiology, locations and follow-up results of all patients with CSF rhinorrhoea that were referred to our centre between 2005 and 2012. Data was collected by reviews of hospital and office records or by telephone surveys with targeted questions. Patients were assessed in terms of recurrence or persistence of symptoms, long-term complications such as meningitis, recurrent seizures, and chronic sinusitis. We then recorded the specific details of our diagnostic approach, including its success rate in identifying leaks pre-operatively, the surgical plans, and the graft choices.
Our pre-surgical diagnostic method was based on the typical facilities available to medical centres in Iran. Although a wide range of diagnostic methods was employed to confirm the existence of a CSF leak, we did not use expensive methods such as B2 transferrin analysis, metrizamide computed tomography (CT) cisternography, intraoperative navigation, intrathecal fluorescein for intraoperative CSF leak detection, fibrin glue or gasket seal with Medpore.
Diagnostic methods
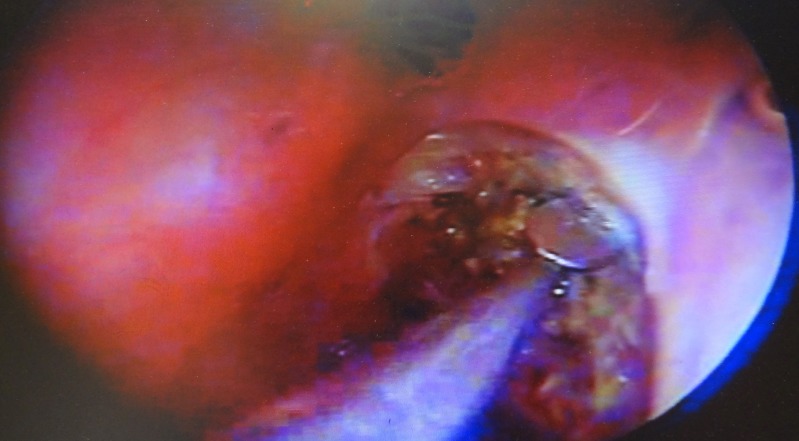
In the outpatient clinic or operating room, we performed endoscopic sinonasal examinations to detect and visualise the site and size of any leaks. The majority of CSF leaks were detectable by asking patients to perform Valsalva and squatting manoeuvres (Figure 1). Although these manoeuvres failed to show CSF leaks in some cases, we were often able to observe arachnoid herniation through the defects. We used intracranial pressure (ICP) augmentation techniques such as forced inspiration with the patient’s head positioned downward (6). We used a Queckenstedt test when these manoeuvres were impossible due to physical disability, noncompliance or lower limb trauma. This test raised ICP by bilateral jugular vein compression (7). However, a manual increment of the intrathoracic pressure, which determines the congestion and stasis of the internal jugular veins and a consequent increase of the intracranial pressure, was not very useful.
Figure 1:
Endoscopic examination in the examination room is performed after a squatting manoeuvre.
If the leaks could not be identified using the initial approach, patients were placed in a head-up position to facilitate a vertical position between the head and lens. If this failed to reveal the CSF leak, we would place the lens in the posterior choana and use transient augmentation of ICP as required. After a period, CSF typically penetrates the arachnoid layer, and the absence of clear visualisation of the CSF leak in the posterior choana may have reflected this phenomenon. We followed this manoeuvre by placing the lens in the osteomeatal complex and in the face of sphenoid to identify possible arachnoid herniation.
Operative approach and closure technique
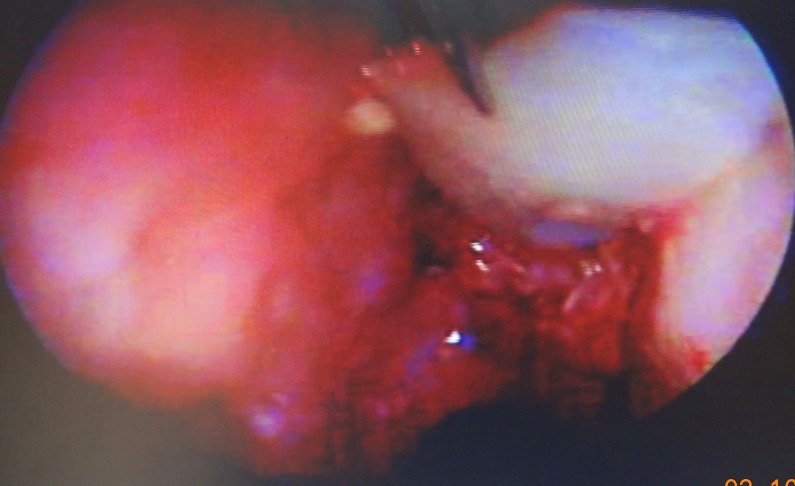
An underlay technique was selected for closure, with CSF pressure used to hold the graft in place. The defect site was prepared for grafting; dissection may not be easy in some areas, particularly in those adjacent to the cribriform plate. Mucosa was not placed in the epidural space because this may lead to an intracranial mucocele (7). Thus, we removed the mucosa around the defect with a round knife (Figure 2).
Figure 2:
Removing the mucosa around the defect with a round knife.
Choice of closure was based on the anatomical location and size of the defect. Our technique has been refined over time to use a variety of graft materials, although we recently have settled on a standard protocol. However, the specific graft material used was largely dependent on the availability during the initial approach and our preference to avoid a separate donor site. In general, we selected free cartilage grafts from the nasal septum with its perichondrium retained on one side. A good graft was trapezoidal in shape with suitable dimensions to close the defect; the smaller edge was 10 mm less than the larger edge, and the larger edge was 10 mm longer than the diameter of the defect (Figure 3). Sometimes, we employed a multilayer closure technique with two cartilages.
Figure 3:
The graft is in a trapezoid shape with the small side being 10 mm shorter than the larger edge, and the larger edge being 10 mm longer than the diameter of the defect.
Statistical analysis
The data were analysed using quantitative and qualitative tests. Categorical variables were compared using the chi-square and Fisher’s exact tests. For all calculations, P < 0.05 was considered statistically significant. This study was approved by the ethical committee of Shahid Beheshti University.
Results
We investigated 37 patients in total, including 22 males (59.5%) and 15 females (40.5%). The mean age at the time of surgery was 33 years. All patients had CSF rhinorrhoea, and the defect site was pre-operatively identified in all instances. Among the included patients, 21 (56.8%) had post-traumatic leaks, 11 (29.7%) had iatrogenic leaks and 5 (13.5%) had spontaneous leaks. The sites of the CSF leaks were the fovea in 17 (45.9%) cases, the sphenoid in 9 (24.3%), the cribriform plate in 5 (13.5%), the posterior table of the frontal sinus in 2 (5.4%) and the basal lamella in 1 (2.7%). Pre-repair complications associated with the CSF leaks included meningitis in 4 patients, encephalocele in 1 and pneumocephalus in 1. The patient data is summarised in Table 1.
Table 1 .
Patient Characteristics
| Trumatic | Hypophysectomy | Brain Stem Tumor | Spontenous | Tumor of Ethmoid | Polyposis | |
|---|---|---|---|---|---|---|
| No of patients | 21 | 8 | 1 | 5 | 1 | 1 |
| Age | 28.24 (SD 18.6) | 44.5 (SD 14.1) | 4 | 41 (SD 18.4) | 44 | 44 |
| Success | ||||||
| Yes | 18 (90) | 7 (87.5) | 1 (100) | 3 (60) | 1 (100) | 1 (100) |
| No | 2 (10) | 1 (12.5) | 0 (0) | 2 (40) | 0 (0) | 0 (0) |
| Gender | ||||||
| Male | 17 (81) | 3 (37.5) | 1 (100) | 0 (0) | 0 (0) | 1 (100) |
| Female | 4 (19) | 5 (62.5) | 0 (0) | 5 (100) | 1 (100) | 0 (0) |
| Site | ||||||
| Fovea | 13 (62) | 0 (0) | 0 (0) | 3 (60) | 1 (100) | 0 (0) |
| Fovea, sphenoid | 2 (9.5) | 8 (100) | 1 (100) | 0 (0) | 0 (0) | 0 (0) |
| Pos table of frontal | 2 (9.5) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Fovea and cribriform plate | 1 (4.8) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Cribriform plate | 3 (14.2) | 0 (0) | 0 (0) | 2 (40) | 0 (0) | 1 (100) |
CSF leaks were successfully repaired at the first attempt in 31 of 36 patients (86.1%), which is comparable with the reported average success rate in the literature (89.2%) (10–15). At the time of writing, the remaining 5 patients underwent a second attempt and achieved successful endoscopic repair of CSF rhinorrhoea. In these cases, the aetiologies for the CSF leak were posthypophysectomy and trauma in one and two patients, respectively. The aetiology of CSF leak was spontaneous in two patients. One patient developed chronic sinusitis and was treated with antibiotics.
There was no significant association between the success rate and gender (P = 0.138). Neither the success rates at different sites (P = 0.633) nor the aetiology of the leaks differed significantly (P = 0.557). The follow-up time ranged from 2 to 72 months.
Discussion
Risk factors for CSF leaks have been extensively discussed in the literature. Accidental trauma from closed head injury is the most common primary inciting event (44%), followed by surgical, iatrogenic trauma (29%). Both scenarios generally result in normal ICPs at the time of repair. Although accidental trauma is most often associated with small cracks that close spontaneously with conservative treatment (80%), the risk of infection is can be significant (30–40%). Furthermore, CSF rhinorrhoea is a complication that can be anticipated in sinus surgery, and the high numbers performed may increase its likelihood.
We use several key technical points to improve our closure rates. These are possible despite the technical and resource limitations of most of our operating room Although we cannot claim to have reduced the complication rate or to have achieved a better success rate, our technique is simple and can be readily applied to most clinical practices.
The diagnosis of CSF rhinorrhoea requires an appropriate chemical marker (1). At present, B2 transferrin has emerged as the preferred biochemical marker of CSF because it is highly specific for CSF, proving to be the most effective test currently available (8). However, B2 transferrin tests have several important limitations. First, reliable test results are dependent on laboratories receiving an adequate marker. This is because tests are not commonly available and the time involved in transportation may degrade the samples. Second, if the CSF leak is very slow or intermittent, the patient may be unable to provide sufficient nasal discharge for analysis. Third, it may not be possible to collect the nasal drainage from some elderly, disabled, or uncooperative patients because of difficulties in the actual collection process. Finally, B2 transferrin has been detected in aqueous humour and in the serum of patients with alcohol-induced chronic liver diseases.
Another strategy is based upon documenting the presence of communication between the intradural and extradural spaces (1). Metrizamide CT cisternography can confirm large communications, but cannot visualise small or intermittent leaks (16). A false negative result can be caused by the temporary cessation of leakage by swelling, inflammation or brain herniation through the defect (17). Furthermore, this assumes the availability of a neurosurgeon, which is not always possible.
No pre-dominant technique has been described in most reports, and the patching of bony dehiscence at the presumptive leak site has proved to be ineffective (16). We have found otology instruments more useful in our surgery because of the lack of specialist instruments in all operating rooms. Failure to repair the defect endoscopically may be related to the inability to localise the defect successfully, or to graft displacement, insufficient graft size or incomplete opposition of the graft to the skull base defect (18). At times, the optical distortion of the endoscope may also overestimate the size of the defect (9). In case of a sphenoid defect, it is safer to remove all of the mucosa to avoid mucocele formation completely (5). The defect needs to be repaired rather than simply sealing the CSF leak packing the sphenoid. Many defects are less than 1.5 cm and well-recognised disadvantages of the underlying technique are its limited accessibility, a lack of direct visualisation and the risk of neurovascular damage (1).
Several reasons led to our preference for free cartilage grafts from the nasal septum with perichondrium retained on one side. First, the grafts have a relatively low failure rate. Second, because a free bone graft can slide, a hole has to be made in the centre before placement, which makes grafting more difficult. Third, ambient ICP has a higher extrusion rate when soft tissues such as fascia or fat are used. In addition, if a defect is less than 1 mm or there are some concerns about the proximity of intracranial structures, such as the optic nerve or vessels, it may be preferable to avoid fat plugs requiring intradural dissection (8). Finally, a rozen can be inserted into a cartilage to ensure correct placement.
Reconstruction is to some extent dependent on defect size. Layered reconstruction that includes cartilage or a bone graft for solid support provides an optimal barrier for a medium-sized defect. The surgical technique chosen to be an underlay enables the graft to be maintained in position by CSF pressure, which is not possible with the overlay technique. Another recognised disadvantage of the underlay technique is limited accessibility with poor visualisation. Although a free bone graft may be used, its use is associated with a higher risk of neurovascular damage due to its sharp edges. In layered reconstruction, it should be mentioned that folding increases the risk of forming a functional dead space that can result in CSF accumulation.
For small defects, the best option is usually a filler graft, but care should be taken to ensure their correct placement during surgery. To the best of our knowledge, CSF leakage occurs in up to 80% of patients with nonsurgical trauma, and in 12–30% of all basilar skull fractures (3). Typically, a fracture in the posterior wall of the frontal sinus is not readily visualised by endoscopic methods (16) but is an indication for an intracranial approach (12,16,19). In this situation, we could place an endoscopic lens through the fracture line with a pair of forceps used in otologic surgeries. However, in the presence of several and multiple disruptions of the anterior skull base, the underlay technique is not possible, and the failure rate increases with an overlay technique. Therefore, the best way to deal with these injuries is to collaborate with neurosurgeons. We repaired such CSF leakages in this manner.
Perhaps the most notable outcome of this report is that we identified leaks and confirmed their locations, in all of the included patients. Importantly, we achieved this using the cheapest and the most readily available procedures coupled with simple and precise graft techniques. In addition, when compared to commonplace methods, our procedure can claim comparable success but with the same associated complications.
Managing rare cares presented their own difficulties. CSF fistulas in the lateral recesses may require an extended sphenoidotomy approach through the pterygopalatine fossa. Unfortunately, an underlay graft may not be possible because of the adjacent neurovascular structures (17); therefore, autologous fat is used as the tissue of choice for filling the tumour cavity (20) because it is accessible and durable. We have presented our experience with two patients who had CSF leaks of the lateral sphenoid wall that were repaired by endoscopic transnasal sinus obliteration with fat.
High-pressure CSF leaks may be either traumatic or non-traumatic. They are characterised by increased CSF pressure in conditions involving intracranial masses, or those caused by post-infective hydrocephalus or congenital hydrocephalus. As was stated by Mokri et al., undiagnosed intracranial hypertension can be dangerous when associated with both traumatic and nontraumatic leaks. Even when the duraplasty is correctly performed, the condition may cause surgical failure or malignant post-operative intracranial hypertension (19). Moreover, the transitory elevation of the intracranial pressure can lead to relapse (6,16). Although the use of lumbar drains remains controversial, they may be indicated to assess increased ICP (3).
Conclusion
Using our methods, reconstruction for CSF leaks was associated with a high success rate of approximately 86.1% at the first attempt and low morbidity. Moreover, the technique was applicable to the majority of CSF leaks, and open endoscopic repair is feasible for failed endoscopic repairs. Regarding patients with raised ICP, it appears best to advise them of the low treatment success rate and their probable need for additional procedures such as shunting.
Acknowledgments
We thank the research deputy of Shahid Beheshti University of Medical Sciences, Tehran, Iran, for his support. We also thank Dr Samira Chaybakhsh, PhD in biostatistics, for her collaboration during the data analysis. There were no financial conflicts for any of the authors.
Footnotes
Conflict of interest
None.
Funds
None.
Authors’ contributions
Conception and design, final approval of the article, provision of study materials or patient, collection and assembly of data, obtaining of funding and statistical expertise: AAS, AS
Analysis and interpretation of the data, drafting of the article and critical revision of the article for the important intellectual content: RJ
References
- 1.Paul WF, Bruce HH, Valeria JL. 5th ed. St Louis (MO): Mosby; 2010. Cummings Otolaryngology: Head and Neck Surgery; –785.pp. 796 [Google Scholar]
- 2.Nyquist GG, Anand VK, Schwartz TH. Endoscopic management of cerebrospinal fluid rhinorrhea. Operative Tech Otolaryngology-Head Neck Surg. 2011;22(3):229–231. doi: http://dx.doi.org/10.1016/j.otot. 2011.08.013 . [Google Scholar]
- 3.Prosser JD, Vender JR, Solares CA. Traumatic cerebrospinal fluid leaks. Otolaryngol Clin North Am. 2011;44(4):857–873. doi: 10.1016/j.otc.2011.06.007. doi: 10.1016/j.otc.2011.06.007 . [DOI] [PubMed] [Google Scholar]
- 4.Ismail AS, Costantino PD, Sen C. Transnasal transsphenoidal endoscopic repair of csf leakage using multilayer acellular dermis. Skull Base. 2007;17(2):125–132. doi: 10.1055/s-2007-970556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Peng A, Li Y, Xiao Z, Wu W. Exploration of endonasal endoscopic repair of paediatric cerebrospinal fluid rhinorrhea. Int J Pediatr Otorhinolaryngol. 2011;75(3):308–315. doi: 10.1016/j.ijporl.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 6.Presutti L, Mattioli F, Villari D, Marchioni D, Alicandri-Ciufelli M. Transnasal endoscopic treatment of cerebrospinal fluid leak: 17 years' experience. Acta Otorhinolaryngol Ital. 2009;29(4):191–196. [PMC free article] [PubMed] [Google Scholar]
- 7.Woodworth BA, Palmer JN. Spontaneous cerebrospinal fluid leaks. Curr Opin Otolaryngol Head Neck Surg. 2009;17(1):59–65. doi: 10.1097/MOO.0b013e3283200017. [DOI] [PubMed] [Google Scholar]
- 8.Briggs RJ, Wormald PJ. Endoscopic transnasal intradural repair of anterior skull base cerebrospinal fluid fistulae. J Clin Neurosci. 2004;11(6):597–599. doi: 10.1016/j.jocn.2003.09.011. [DOI] [PubMed] [Google Scholar]
- 9.Benjamin B, Satish G, James NP. State of the art cerebrospinal fluid leak and encephalocele repair. Operative Tech Otolaryngology-Head Neck Surg. 2006;17(1):49–57. doi: http://dx.doi.org/10.1016/j.otot.2005.12.002 . [Google Scholar]
- 10.Psaltis AJ, Schlosser RJ, Banks CA, Yawn J, Soler ZM. A systematic review of the endoscopic repair of cerebrospinal fluid leaks. Otolaryngol Head Neck Surg. 2012;147(2):196–203. doi: 10.1177/0194599812451090. doi: 10.1177/0194599812451090 . [DOI] [PubMed] [Google Scholar]
- 11.Daele JJ, Goffart Y, Machiels S. Traumatic, iatrogenic and spontaneous cerebrospinal fluid (CSF) leak: endoscopic repair. B -ENT. 2011;7(Suppl 17):47–60. [PubMed] [Google Scholar]
- 12.Gilat H, Rappaport Z, Yaniv E. Endoscopic transnasal cerebrospinal fluid leak repair: a 10 year experience. Isr Med Assoc J. 2011;13(10):597–600. [PubMed] [Google Scholar]
- 13.Lee DH, Lim SC, Joo YE. Treatment outcomes of endoscopic repairs of sinonasal cerebrospinal fluid leaks. J Craniofac Surg. 2011;22(4):1266–1270. doi: 10.1097/SCS.0b013e31821c6ad3. doi: 10.1097/SCS.0b013e31821c6ad3 . [DOI] [PubMed] [Google Scholar]
- 14.Banks CA, Palmer JN, Chiu AG, O'Malley BW Jr, Woodworth BA, Kennedy DW. Endoscopic closure of CSF rhinorrhea: 193 cases over 21 years. Otolaryngol Head Neck Surg. 2009;140(6):826–833. doi: 10.1016/j.otohns.2008.12.060. doi: 10.1016/j.otohns.2008.12.060 . [DOI] [PubMed] [Google Scholar]
- 15.Mirza S, Thaper A, McClelland L, Jones NS. Sinonasal cerebrospinal fluid leaks: management of 97 patients over 10 years. Laryngoscope. 2005;115(10):1774–1777. doi: 10.1097/01.mlg.0000175679.68452.75. [DOI] [PubMed] [Google Scholar]
- 16.Basu D, Haughey BH, Hartman JM. Determinants of success in endoscopic cerebrospinal fluid leak repair. Otolaryngol Head Neck Surg. 2006;135(5):769–773. doi: 10.1016/j.otohns.2006.05.025. [DOI] [PubMed] [Google Scholar]
- 17.Tosun F, Carrau RL, Snyderman CH, Kassam A, Celin S, Schaitkin B. Endonasal endoscopic repair of cerebrospinal fluid leaks of the sphenoid sinus. Arch Otolaryngol Head Neck Surg. 2003;129(5):576–580. doi: 10.1001/archotol.129.5.576. [DOI] [PubMed] [Google Scholar]
- 18.El-Banhawy OA, Halaka AN, El-Hafiz Shehab El-Dien, Ayad H. Subcranial transnasal repair of cerebrospinal fluid rhinorrhea with free autologous grafts by the combined overlay and underlay techniques. Minim Invasive Neurosurg. 2004;47(4):197–202. doi: 10.1055/s-2004-818513. [DOI] [PubMed] [Google Scholar]
- 19.Locatelli D, Rampa F, Acchiardi I, Bignami M, De Bernardi F, Castelnuovo P. Endoscopic endonasal approaches for repair of cerebrospinal fluid leaks: nine-year experience. Neurosurgery. 2006;58(4 Suppl 2):246–256. doi: 10.1227/01.NEU.0000193924.65297.3F. [DOI] [PubMed] [Google Scholar]
- 20.Tabaee A, Anand VK, Brown SM, Lin JW, Schwartz TH. Algorithm for reconstruction after endoscopic pituitary and skull base surgery. Laryngoscope. 2007;117(7):1133–1137. doi: 10.1097/MLG.0b013e31805c08c5. [DOI] [PubMed] [Google Scholar]