Abstract
Bone homeostasis seems to be controlled by delicate and subtle “cross talk” between the nervous system and “osteo-neuromediators” that control bone remodeling. The purpose of this study was to evaluate the effect of interactions between neuropeptides and human bone morphogenetic protein 2 (hBMP2) on human osteoblasts. We also investigated the effects of neuropeptides and hBMP2 on gap junction intercellular communication (GJIC). Osteoblasts were treated with neuropeptide Y (NPY), substance P (SP), or hBMP2 at three concentrations. At various intervals after treatment, cell viability was measured by the MTT assay. In addition, cellular alkaline phosphatase (ALP) activity and osteocalcin were determined by colorimetric assay and radioimmunoassay, respectively. The effects of NPY, SP and hBMP on GJIC were determined by laser scanning confocal microscopy. The viability of cells treated with neuropeptides and hBMP2 increased significantly in a time-dependent manner, but was inversely associated with the concentration of the treatments. ALP activity and osteocalcin were both reduced in osteoblasts exposed to the combination of neuropeptides and hBMP2. The GJIC of osteoblasts was significantly increased by the neuropeptides and hBMP2. These results suggest that osteoblast activity is increased by neuropeptides and hBMP2 through increased GJIC. Identification of the GJIC-mediated signal transduction capable of modulating the cellular activities of bone cells represents a novel approach to studying the biology of skeletal innervation.
Keywords: Neuropeptide, Bone morphogenetic protein, Osteoblast, Gap junction intercellular communication
Introduction
Bone alters its metabolic and anabolic activities in response to a variety of systemic and local factors, such as hormones and growth factors. In recent years, interest in the relationship between neuropeptides to bone processes has increased and represents one of the most interesting aspects in skeletal research. Classic observations describing an abundance of the nerve fibers in bone support a paradigm of nervous system influence on bone metabolism and anabolism. Also, many nerve-derived signaling molecules that might act as efferent agents on bone cells fall into the category of neuropeptides. Currently, five neuropeptides are known to be involved in the control of bone homeostasis, including calcitonin gene-related peptide (CGRP), neuropeptide Y (NPY), substance P (SP), vasoactive intestinal peptide (VIP), and tyrosine hydroxylase (TH) (1,2). Of these, CGRP is abundantly distributed in bone via sensory nerves, especially in epiphyseal trabecular bone, which strongly suggests that it influences bone metabolism (3). It has also been reported that adipose-derived stem cells (ADSCs) transduced with an adenoviral vector containing the GCRP gene (Ad-CGRP) had an increased potential to differentiate into osteoblasts in vitro (4). In this study, we focused on NPY and SP, in which interest has been increasing.
NPY may be a regulator of bone homeostasis, but its effect on osteoblast activity and the biological functions involving NPY receptors in bone homeostasis remain to be clarified. A functional analysis by Teixeira et al. (5) revealed the osteogenic potential of NPY, showing that osteoblast phenotype markers were significantly enhanced in osteoprogenitor cells stimulated by NPY, probably due to down-regulation of the Y1 receptor. Also, Lundberg et al. (6) suggested that an increase in the number of mesenchymal progenitors and altered Y1 receptor expression within bone cells in the absence of Y2 receptors are possible mechanisms for the increased bone mineralization in vivo and in vitro.
Some investigators have reported that SP can stimulate bone marrow stromal cell (BMSC) proliferation, protein production, and mineralization (7,8). Recently Goto et al. (9) demonstrated that SP treatment of calvarial osteoblasts increased mineralization and the expression of the bone-related proteins Runx2, type I collagen, and osteocalcin.
These data support the hypothesis that neuropeptide signaling stimulates bone formation and inhibits bone resorption. To further test this hypothesis, we selectively investigated the role of the two neuropeptides (NPY and SP). In addition, ALP activity and osteocalcin were assayed to profile some osteoblast features. The study examined the effects of neuropeptides on osteoblast cell viability, ALP activity, and osteocalcin in culture supernatants. Expression of their receptors by human osteoblasts was also evaluated.
Bone morphogenetic protein (BMP) is a multifunctional growth factor that belongs to the transforming growth factor-β (TGF-β) superfamily. Studies in transgenic and knockout mice and from animals and humans with naturally occurring BMP2 mutations and their signaling molecules have shown that BMP2 signaling plays a critical role in bone and cartilage development and postnatal bone formation (10). However, the combined effects of neuropeptides and BMP2 on bone metabolism are yet to be explored. In this study we observed their combined effects on human osteoblast viability and function.
In addition, gap junctions formed by connexins (Cx) play an important role in transmitting signals between bone cells such as osteoblasts and osteoclasts, the cells responsible for bone formation and bone remodeling, respectively. Gap junction intercellular communication (GJIC) has been shown to mediate the processes of osteoblast differentiation and bone formation. Furthermore, GJIC propagates Ca2+ signaling, conveys the anabolic effects of hormones and growth factors, and regulates gene transcription of osteoblast differentiation markers (11,12). To date, no information is available on the effects of neuropeptides on GJIC. Therefore, we also evaluated the effects of neuropeptides and hBMP2 on GJIC in osteoblasts.
Material and Methods
Cell culture
The research protocol was approved by the Ethics Committee of the Third Hospital of Hebei Medical University. The human osteoblast system used in this study was established and identified by our group and the normal human osteoblasts we used had been frozen in liquid nitrogen in our laboratory. After thawing and subculturing, the cells showed more than 85% viability and the same growth, morphologic, and genetic characteristics as before storage (13). The cells were cultured in Dulbecco's Modified Eagle Medium (DMEM; Gibco, USA) supplemented with 10% fetal bovine serum (Gibco), 100 U/mL penicillin and 100 μg/mL streptomycin. Incubation was conducted in 5% CO2 at 37°C. The medium was changed 3 times a week. Cells at the second or third passage were used for the experiments. The cells were prepared in the standard manner and cellular morphology was observed by scanning electron microscopy (SEM) with an S-800 microscope (Hitachi Inc., Japan). Cell suspensions were prepared for subsequent assays by washing with phosphate-buffered saline (PBS) and detached with 0.25% trypsin (Gibco). For the viability assay, the cells were placed in three 96-well plates and incubated in culture medium for 24 h. For the functional studies, cells from the second passage were seeded on four 24-well plates and incubated to confluence. After that, the medium was changed for modified medium supplemented with neuropeptides in different concentrations, depending on the experiments to be performed. After treatment, assays were performed for cell viability, ALP, and osteocalcin secretion. Cells growing in medium without any of the neuropeptides were used as controls. The arrangement of wells was randomized within the plates. The neuropeptides used in the study were human neuropeptide Y (NPY; Sigma, USA) and substance P (SP; Sigma). Human bone morphogenetic protein 2 (hBMP2) (Sigma) was obtained from the Fourth Military Medical University (China). All neuropeptides and hBMP2 were diluted with serum-free DMEM to appropriate concentrations.
Cell viability
Cell viability was measured by the MTT (3-4,5-dimethylthiazol 2,5-diphenyltetrazolium bromide) assay. Cells were seeded onto 96-well clear-bottomed plates at a density of 10,000 cells per well in 100 µL medium, incubated for 24 h, and treated with the neuropeptides and hBMP2 and their combination at final concentrations of 1, 0.1, and 0.01 μg/mL. Six replicates per concentration per treatment were assayed. After 24, 48, and 72 h, 20 µL of 5 mg/mL MTT solution was added to each well and incubated in a humidified 5% CO2 atmosphere at 37°C for 5 h. After incubation, the cells were washed with PBS. Subsequently, 100 µL of acidified isopropanol was added to each well, and the cells were incubated at room temperature for 10 min. Absorbance was measured by a microtiter plate reader (Organon Teknika Reader 230S, Austria) at 492 nm, with 620 nm as reference.
ALP activity
Cells (2×104 cell in 100 µL) from the second passage were seeded in four 24-well plates, incubated to confluence, and treated with NPY, SP, or hBMP2 at final concentrations of 0.1 μg/mL as well as combinations of 0.1 μg/mL hBMP2 with each neuropeptide. After 24 h, ALP activity was assayed in the cells in two plates. To simulate administration in clinical practice, the cells in the two remaining plates were cultured for an additional 3 days and then treated with the neuropeptides and hBMP2 again. After incubation for 24 h, the cells were assayed. The determinations were performed using 6 replicates of each treatment. To assay ALP activity, the remaining medium was removed, the cells were washed with PBS, and digested with 0.25% trypsin for 1 min. The resulting suspended cells were collected in Eppendorf tubes, distilled water was added, and the tubes were frozen in liquid nitrogen and thawed repeatedly 3-4 times to destroy the cell membranes. After centrifugation at 200 g for 1 min, the supernatants were collected and assayed with an Automatic Biochemistry Analyzer (Hitachi 7150, Japan) at 405 nm. All treatment results were compared with the control values (cell cultures in DMEM without neuropeptides and hBMP2).
Osteocalcin assay
Osteocalcin concentration was measured by an enzyme-linked immunosorbent assay (ELISA; Elabscience, China). Cells were cultured and prepared as described above for the ALP assay. NPY, SP, and hBMP2 were added at a final concentration of 0.1 μg/mL. For the osteocalcin assay, supernatants were collected in Eppendorf tubes to perform the determinations. A total of 6 replicates were tested for each treatment.
Neuropeptide and BMP2 receptors in human osteoblasts
To assay the expression of neuropeptide and hBMP2 receptors in human osteoblasts, cells grown to confluence on cover slips were fixed in 4% buffered formaldehyde, washed with distilled water, and incubated with 3% hydrogen peroxide for 5 min to eliminate the activity of the endogenous peroxidase. Immunocytochemical staining was performed with NPY, SP, and BMP2 kits (Sigma) following the protocol recommended by the manufacturer, with 3 replicates of each treatment. The cells were observed by phase contrast microscopy, and the Image-Pro Plus 5.0 (Media Cybernetics Inc., USA) image analysis software was used to measure the gray values of stained cells. The antigen levels of cells were inversely associated with the gray values. The mean gray value of 5 randomly selected fields in each cover slip was calculated. All the treatments were compared with the control cells, which were incubated only with secondary antibody.
GJIC in human osteoblasts
The effects of the neuropeptides and HBMP2 on GJIC were determined by fluorescence recovery after photobleaching (FRAP) using a laser scanning confocal microscope. Cells were seeded onto 96-well plates at a density of 5000 cells per well and incubated to confluence. The cells were rinsed twice with D-Hank's buffer and treated with 10 μmol/L CFDA (5,6-carboxy fluorescein diacetate; Sigma), at 37°C for 90 min, then rinsed with D-Hank's buffer again. Cells were then treated with NPY, SP, or hBMP2 at a final concentration of 0.1 μg/mL, and GJIC were measured with an MRC-1024 laser scanning confocal microscope imaging system (Bio-Rad, USA). The rate of fluorescence recovery (R) at 10 min after photobleaching was adopted as the functional index of GJIC.
Statistical analysis
Data are reported as means±SD. The Shapiro-Wilk test was performed to test the normality of data. Levene's test was used to assess the equality of variances. The t-test was carried out to compare data between two groups and analysis of variance (ANOVA) to compare data among three or more groups. The Student-Newman-Keuls multiple-range test was used to assess differences between groups. The SAS software package (version 9.1, SAS Institute Inc., USA) was used for statistical analysis. The results were considered statistically significant at P<0.05.
Results
Cell morphology
Thawed cells showed stable characteristics as previously described (13). The cellular morphology of human osteoblasts was observed using phase-contrast microscopy and SEM. On phase-contrast microscopic images (Figure 1A and B), we found that osteoblasts were fusiform or polygonal, with extensive cytoplasm and large, oval nuclei; SEM revealed that the cells were connected by ecphyma.
Figure 1. Cellular morphology of human osteoblasts. The osteoblast was thawed by directly putting the freezing tube into water at 40°C. Then, the frozen solution was removed by centrifugation (200 g for 1 min). The cells were cultured in Dulbecco's Modified Eagle Medium supplemented with 10% fetal bovine serum. The phase-contrast microscopic images of the cells showed that osteoblasts were fusiform or polygonal, with plenty of cytoplasm and large oval nuclei (A). SEM showed that the cells were connected by ecphyma (B).

Cell viability
Figure 2 shows the results of the cell viability assay following exposure to the neuropeptides and hBMP2 at concentrations of 1, 0.1, and 0.01 μg/mL for 24, 48, and 72 h. All treatments induced a significant increase in cell viability compared to the blank control group (P<0.05). Meanwhile, there were significant differences between the neuropeptides and hBMP2 at all concentrations at all times (P<0.05). The viability index of osteoblasts (absorbance at 492 nm) was inversely associated with the concentration of the neuropeptides, and positively associated with the duration of exposure, whereas the absorbance was positively associated with the concentration of hBMP2. As shown in Figure 3, the stimulating effect of neuropeptides on cell viability was reduced with the addition of hBMP2.
Figure 2. Cell viability assays. The graph shows the viability index of osteoblasts (absorbance) exposed to neuropeptide Y (NPY), substance P (SP) and human bone morphogenetic protein 2 (hBMP2). The absorbance was significantly different among the three treatments (P<0.05, ANOVA) and increased over time. Additionally, for the two neuropeptides, absorbance increased as the concentration decreased. However, for hBMP2, absorbance decreased as its concentration decreased.
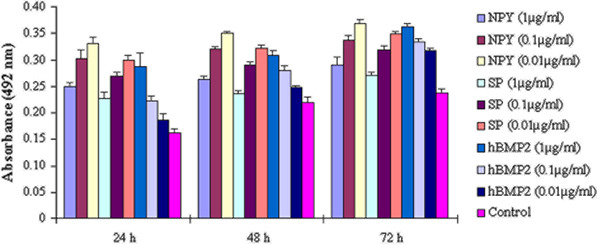
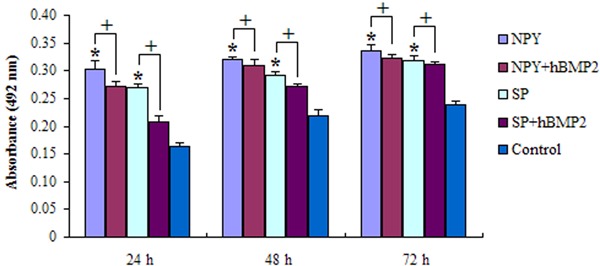
Figure 3. Cell viability of the effect of neuropeptides in combination with human bone morphogenetic protein 2 (hBMP2). It can be seen that the two neuropeptides [neuropeptide Y (NPY) and substance P (SP)] induced statistically significant increases (*P<0.05, compared to control; ANOVA) in cell viability, which rises over time, but the stimulating effect of neuropeptides decreased with the addition of hBMP2 (+P<0.05, ANOVA).
ALP activity
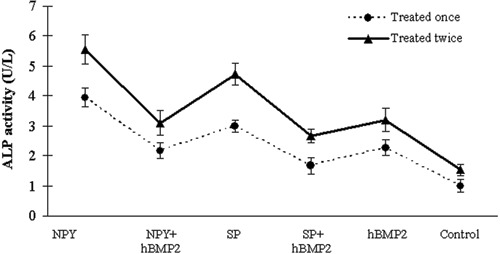
The ALP activity of the osteoblasts exposed to the neuropeptides and hBMP2 at a concentration of 0.1 μg/mL was evaluated (Figure 4). All treatments showed significantly higher ALP activities than the control group (P<0.05). Moreover, the ALP activity of the osteoblasts treated twice with the neuropeptides or hBMP2 was significantly higher than that of the osteoblasts treated only once (P<0.05). The osteoblasts exposed to 0.1 μg/mL NPY had the highest ALP activity. The ALP activity of the osteoblasts exposed to 0.1 μg/mL neuropeptides or hBMP2 alone was also significantly higher than that of the osteoblasts exposed to neuropeptides combined with hBMP2 (P<0.05).
Figure 4. Alkaline phosphatase (ALP) activity assay. The graph shows the ALP activity of the osteoblasts treated with 0.1 μg/mL neuropeptide Y (NPY), substance P (SP) and human bone morphogenetic protein 2 (hBMP2) alone, as well as their combined applications. Cells were treated once or twice. In this assay, all treatments induced statistically significant increases (P<0.05, ANOVA) in ALP activity after the osteoblasts were treated twice compared with those treated once. The levels of ALP activity in cells exposed to NPY were the highest. Moreover, the ALP activity of cells exposed to 0.1 μg/mL neuropeptides alone was also significantly higher than that of the osteoblasts exposed to neuropeptides combined with human bone morphogenetic protein 2.
Osteocalcin assay
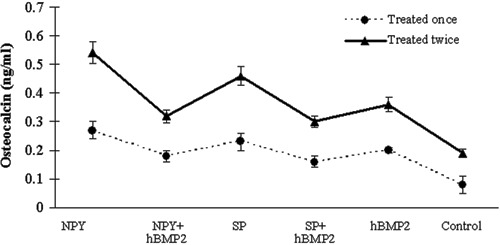
Neuropeptides and hBMP2 at a concentration of 0.1 μg/mL significantly increased the osteocalcin concentration compared with that of the control group (P<0.05) (Figure 5). Treating the osteoblasts twice with the neuropeptides or hBMP2 generated a significant increase in osteocalcin levels compared with treating the cells only once (P<0.05). Furthermore, the osteocalcin concentrations in osteoblasts exposed to 0.1 μg/mL neuropeptide or hBMP2 alone were higher than those in the osteoblasts exposed to their combinations (P<0.05).
Figure 5. Osteocalcin assay. The graph shows the osteocalcin of the osteoblasts treated once or twice with 0.1 μg/mL neuropeptide Y (NPY), substance P (SP) and human bone morphogenetic protein 2 (hBMP2) alone, as well as their combined applications. The osteoblasts exposed to 0.1 μg/mL NPY had the highest level of osteocalcin. The osteoblasts treated twice with the neuropeptides or hBMP2 displayed higher osteocalcin than those treated only once. Furthermore, the osteocalcin levels of cells exposed to 0.1 μg/mL neuropeptides alone were also significantly higher than those of the osteoblasts exposed to neuropeptides combined with hBMP2 (P<0.05, ANOVA).
Neuropeptides and BMP2 receptors in human osteoblast
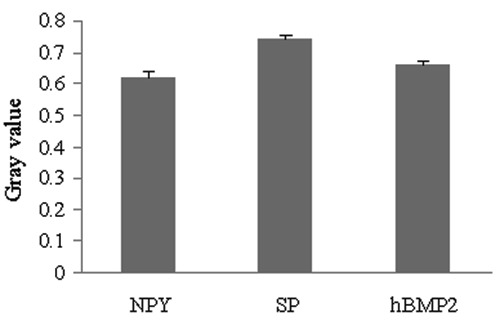
The expression of the neuropeptide (SP and NPY) and hBMP2 receptors in the human osteoblasts was measured by immunocytochemical assay and image semi-quantitative analysis. The expression level of receptors was inversely associated with the gray values of stained cells and positively associated with the antigen levels of the cells. The expression of neuropeptide and hBMP2 receptors was significantly higher than in the control group (P<0.05; Figure 6). The expression in human osteoblasts was the highest for NPY receptor and the lowest for SP receptor.
Figure 6. The expression level of neuropeptide Y (NPY), substance P (SP) and human bone morphogenetic protein 2 (hBMP2) receptors. The graph indicates the gray values of human osteoblasts, as measured with immunocytochemical assays. The expression level of the receptor was inversely associated with the gray value of the staining cell and positively associated with the antigen level of the cell. Therefore, the expression was the highest for NPY receptor and the lowest for SP receptor in human osteoblasts.
GJIC in human osteoblasts
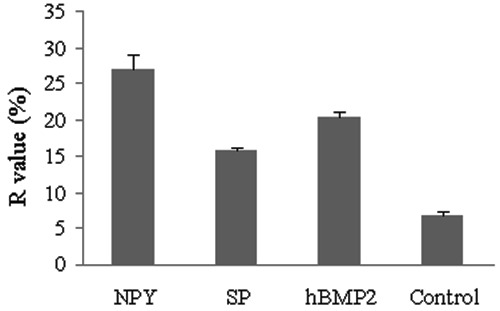
Using the FRAP method, the ratio of fluorescence recovery (R) of cells exposed to the neuropeptides and hBMP2 at the concentration of 0.1 μg/mL was between 15.8±0.4 and 27.0±1.9, whereas the cells in the control group had an R-value of 6.8±0.4. This demonstrated that the GJIC of osteoblasts was significantly increased by the neuropeptides and hBMP2 (P<0.05). The R-value of cells exposed to 0.1 μg/mL NPY (27.0±1.9) was the highest, followed by hBMP2 (20.2±0.7) and SP (15.8±0.4) (Figure 7).
Figure 7. Fluorescence recovery of cells. The graph displays the ratio of fluorescence recovery (R) of cells exposed to neuropeptide Y (NPY), substance P (SP) and human bone morphogenetic protein 2 (hBMP2) at the concentration of 0.1 μg/mL. The R-values of the osteoblasts after neuropeptides or hBMP2 treatments were all significantly higher than those of the blank control group. Meanwhile, cells exposed to NPY had the highest R-value, followed by hBMP2 and SP (P<0.05, ANOVA).
Discussion
Bone is abundantly innervated by small-diameter sensory nerves in the periosteum, bone marrow, and vascular canals. Our understanding of the homeostatic nature of bone remodeling has increased recently following the demonstration that neuropeptides and their receptors regulate osteoblast and osteoclast function in vivo (3,14). Because of the complexity of the mechanisms regulating bone remodeling, the experimental and mechanistic studies based on in vivo murine genetic and pharmacologic models are limited to some extent. However, in vitro studies have proved valuable for evaluating cell interactions and stimulating signals. Previous studies using osteoblastic cell lines derived from animal or human osteosarcomas have added to our understanding of the “osteo-neuromediators” involved in the control of osteoblast activity and hence bone formation. In this study, we used the normal human osteoblast cell lines established by our laboratory (13), which markedly improved the reliability of the research. The cells were fusiform and had numerous processes. Furthermore, they had bone-producing characteristics, with high levels of expression of ALP and osteocalcin. In the judgment of the investigators, the cells cultured in this study had the morphology and characteristics of osteoblasts (13), and could be used in subsequent experiments.
This study is unique in that it comprehensively investigated the concentration-dependent effects of NPY, SP, and hBMP2 on human osteoblasts, and focused specifically on cellular viability and function. The expression of NPY, SP, and hBMP2 receptors was also observed in osteoblasts. The presence of NPY, SP, and hBMP2 receptors in human osteoblasts indicated that they could directly modulate bone metabolism via receptor activation.
Several groups have studied the actions of neuropeptides on osteoblast cell viability, differentiation, or function, but the results of those studies have been contradictory, and few have examined the effects of various concentrations of neuropeptides. Sousa et al. (15) found that the absence of the Y1 receptor delayed fracture healing. Lee (16) also demonstrated that the NPY system, via the Y1 receptor, directly inhibited the differentiation of mesenchymal progenitor cells as well as the activity of mature osteoblasts. In addition, it was reported that BMP2 signaling regulates Y1 receptor gene expression, which raises the possibility that NPY acts in osteoblasts via an autocrine mechanism (17). Adamus (7) observed that alkaline phosphatase activity decreased at higher concentrations of SP (10−8 M) treatment. Another recent study reported that higher concentrations of SP (10−7 to 10−5 M) inhibited ALK activity, bone nodule formation, and osteocalcin gene expression in a dose-dependent fashion in fetal rat calvarial osteoblasts (18). Similar to the proliferative effects observed by Adamus et al. in rat bone marrow stromal cell (BMSC) cultures (7), in the current study we observed that osteoblast viability was inversely associated with the neuropeptide concentrations (10−7 to 10−9 M). Higher neuropeptide concentrations (1 μg/mL, corresponding to 10−7 M) had little effect on osteoblast viability, but hBMP2 treatment had a concentration-dependent effect on cellular viability (ranging from 0.01 μg/mL to 1 μg/mL, corresponding to 10−9 to 10−7 M). Paradoxically, Shih and Bernard found that similar concentrations of SP (ranging from 10−8 to 10−6 M) increased, rather than reduced, the number and size of bone colonies in a concentration-dependent manner (8). Similarly, Goto et al. (9) found that higher concentrations of SP (ranging from 10−8 to 10−6 M) increased the size of bone colonies in rat calvarial osteoblast cells. The different results in our study could be due to different protocols, different cell lines used in the studies, the duration of culturing, the diluent for neuropeptides, and the methods used to evaluate cell viability. We initially investigated only three orders of magnitude for the concentration of the neuropeptides in this study. The most effective concentration is still not clear. In addition, we assumed that higher neuropeptide concentrations might stimulate feedback inhibition of neuropeptides in some way.
In this study, ALP activity and osteocalcin were also upregulated by NPY and SP treatment, consistent with the findings of Goto et al. in rat calvarial osteoblasts (9). Moreover, ALP activity and osteocalcin levels were the highest after NPY treatment and the lowest after hBMP2. This suggests that NPY action was the most powerful, hBMP2 action was the weakest, and SP was intermediate between the other two. Collectively, these results suggest that neuropeptides stimulate osteogenesis in human osteoblasts. Previously, we observed that neuropeptide depletion in rats caused trabecular bone loss and inhibited bone formation (19). We also found that hereditary small-fiber sensory neuropathy in man was associated with neuropeptide loss, reduced bone mineral density (BMD), and increased bone fragility (20). These diverse data support our hypothesis that neuropeptide signaling contributes to the maintenance of bone mass by regulating osteogenic activity in osteoblasts.
BMPs, members of the TGF-β superfamily, play a pivotal role in the signaling network and are involved in nearly all processes associated with skeletal morphogenesis. The activity of BMPs was first identified in the 1960s (21), but the proteins responsible for bone induction remained unknown until the purification and sequencing of bovine BMP-3 (osteogenin) and the cloning of human BMP-2 and 4 in the late 1980s (22,23). The injection of BMP-2 locally over the surface of the calvariae of mice induced periosteal bone formation without a prior cartilage phase (24). Overexpression of a dominant-negative BMPR1B also inhibited osteoblast differentiation in osteoblast precursor cells (25). Luppen et al. (26) reported that BMP-2 restored mineralization in glucocorticoid (GC)-inhibited osteoblast cultures. Similarly, we found that hBMP2 stimulated human osteoblast viability and increased ALP activity and osteocalcin in cell culture. This would suggest a role for hBMP2s in the maintenance of a normal bone structure.
A number of studies have shown that the signals of BMPs are transferred through serine/threonine kinase receptors, which include both subtypes I and II (27,28). These receptors are expressed differentially in various tissues and are both indispensable for signal transduction. BMP signals are mediated by type I and II BMP receptors and their downstream molecules Smad1, 5, and 8. Phosphorylated Smad1, 5 and 8 proteins form a complex with Smad4 and are then translocated into the nucleus, where they interact with other transcription factors, such as Runx2 in osteoblasts (29,30).
BMP activities are regulated by many extracellular and intercellular proteins that interact with either BMPs or components of the BMP signaling pathways. Devlin (31) found that BMP antagonists bound to BMP and blocked BMP signaling, resulting in overexpression of noggin in mature osteoblasts and osteoporosis in mice. Yoshida (32) demonstrated that Tob negatively regulated osteoblast proliferation and differentiation by suppressing the activity of the BMP receptor-regulated Smad proteins. In Tob null mutant mice, BMP signaling was enhanced and bone formation was increased. Several investigators observed that Smurf1 was a Hect domain E3 ubiquitin ligase that interacted with Smad1 and 5 and mediated the degradation of those Smad proteins. Smurf1 was found to recognize bone-specific transcription factor Runx2 and mediate its degradation. Overexpression of Smurf1 in osteoblasts inhibited postnatal bone formation in mice (33).
We surveyed the combined action of neuropeptides and hBMP2 and demonstrated that the stimulatory effect of hBMP2 on osteoblasts was weakened when hBMP2 was combined with either NPY or SP. Immunocytochemical assay also detected NPY, SP, and hBMP2 receptors on the plasma membrane of human osteoblasts. The data support previous observations demonstrating that neuropeptides and hBMP2 regulated cellular viability and function by interacting with receptors on the plasma membrane of osteoblasts (2,3,16). The present study suggests that hBMP2 action was antagonized by neuropeptides in some manner at molecular levels analogous to that described above for hBMP2 and its binding proteins. Although the exact mechanism is still unclear, we postulate a requirement to temper the activity of locally produced skeletal regulatory factors. It is important to note that overexpression of skeletal regulatory factors may have serious detrimental consequences, such as osteopenia and fractures.
GJIC mediated by connexins, in particular Cx43, is important in regulating signal transmission among different bone cells, thereby regulating development, differentiation, modeling and remodeling of bone. GJIC regulates osteoblast formation, differentiation, survival, and apoptosis. Furthermore, osteocytes utilize GJIC to coordinate bone remodeling in response to anabolic factors and mechanical loading. These transmembrane channels allow continuity of cytoplasm and mediate the transfer of molecules between communicating cells (34-36). The biological importance of the communication mediated by Cx-forming channels in bone development is demonstrated by the low bone mass and osteoblast dysfunction in the Cx43-null mice and the skeletal malformations observed in occulodentodigital dysplasia (ODDD) caused by mutations in the Cx43 gene (37-39), but there are currently no available data on the effects of neuropeptides on GJIC between osteoblasts.
The novelty of this study is that we confirmed the stimulatory effect of neuropeptides and hBMP2 on GJIC in osteoblasts. The R-value of cells exposed to NPY was the highest, followed by hBMP2 and SP, which suggests that NPY had the strongest effect on regulation of osteoblast activity via promoting the GJIC between cells. This result was consistent with other observations that NPY had the strongest effect on osteoblast viability, ALP activity, and osteocalcin production. GJIC may serve as a means by which osteoblasts can act in synchrony and propagate locally generated signals throughout the skeletal tissue. Thus, we suppose that NPY, SP, and hBMP2 may enhance the GJIC between osteoblasts and then promote the transfer of molecules between communicating cells. In this way, they would improve the response of osteoblasts to various stimulating signals. The study data provided further support for the hypothesis that the stimulatory effects of neuropeptides and hBMP2 on osteoblasts and osteogenesis were due not only to the direct binding and activation of their receptors, but also to a direct action on the GJIC that coordinates bone remodeling. This study presents a novel role for neuropeptide control of bone homeostasis.
In conclusion, neuropeptides stimulated osteoblast viability and mineralization, whereas BMP2 promoted osteoblast activity with increasing concentrations. In addition, the stimulatory effects of neuropeptides or hBMP2 alone on osteoblasts were decreased when the neuropeptides and hBMP2 were combined. NPY, SP, hBMP2 receptors were found on osteoblasts. Moreover, neuropeptides and hBMP2 enhanced GJIC between osteoblasts. Given all these observations, it is tempting to hypothesize that neuropeptides and hBMP2 could be physiologic activators of bone formation in vivo, and that membrane-bound receptors are essential in maintaining skeletal homeostasis through regulating the proliferation and activation of osteoblasts by promoting signal transmission among bone cells. The regulation of GJIC in osteoblasts could be an additional mechanism for modulating bone metabolism. Furthering our understanding of the mechanisms underlying the involvement of the neuropeptide system in the control of bone homeostasis could lead to the development of therapies to improve bone mass in patients with diseases such as osteoporosis. However, we emphasize that the results reported here are preliminary and restricted by an inadequate knowledge of osteoblast cell biology and by the superficial understanding of the mechanism of bone remodeling. Future studies examining the role of neuropeptides signaling in animal models of bone acquisition and loss are required to further investigate these hypotheses.
Acknowledgments
We are grateful to Mrs. Huang Jinxiang for assistance with cell culture. We also express our gratitude to Liu Bo, Chen Xiao, Tian Ye, and Zhang Fei for their efforts in data preparation.
Footnotes
First published online.
References
- 1.Shi YC, Baldock PA. Central and peripheral mechanisms of the NPY system in the regulation of bone and adipose tissue. Bone. 2012;50:430–436. doi: 10.1016/j.bone.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 2.Togari A, Arai M, Mizutani S, Mizutani S, Koshihara Y, Nagatsu T. Expression of mRNAs for neuropeptide receptors and beta-adrenergic receptors in human osteoblasts and human osteogenic sarcoma cells. Neurosci Lett. 1997;233:125–128. doi: 10.1016/S0304-3940(97)00649-6. [DOI] [PubMed] [Google Scholar]
- 3.Imai S, Matsusue Y. Neuronal regulation of bone metabolism and anabolism: calcitonin gene-related peptide-, substance P-, and tyrosine hydroxylase-containing nerves and the bone. Microsc Res Tech. 2002;58:61–69. doi: 10.1002/jemt.10119. [DOI] [PubMed] [Google Scholar]
- 4.Fang Z, Yang Q, Xiong W, Li GH, Liao H, Xiao J, et al. Effect of CGRP-adenoviral vector transduction on the osteoblastic differentiation of rat adipose-derived stem cells. PLoS One. 2013;8: doi: 10.1371/journal.pone.0072738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Teixeira L, Sousa DM, Nunes AF, Sousa MM, Herzog H, Lamghari M. NPY revealed as a critical modulator of osteoblast function in vitro: new insights into the role of Y1 and Y2 receptors. J Cell Biochem. 2009;107:908–916. doi: 10.1002/jcb.22194. [DOI] [PubMed] [Google Scholar]
- 6.Lundberg P, Allison SJ, Lee NJ, Baldock PA, Brouard N, Rost S, et al. Greater bone formation of Y2 knockout mice is associated with increased osteoprogenitor numbers and altered Y1 receptor expression. J Biol Chem. 2007;282:19082–19091. doi: 10.1074/jbc.M609629200. [DOI] [PubMed] [Google Scholar]
- 7.Adamus MA, Dabrowski ZJ. Effect of the neuropeptide substance P on the rat bone marrow-derived osteogenic cells in vitro . J Cell Biochem. 2001;81:499–506. doi: 10.1002/1097-4644(20010601)81:3<499::AID-JCB1063>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 8.Shih C, Bernard GW. Neurogenic substance P stimulates osteogenesis in vitro . Peptides. 1997;18:323–326. doi: 10.1016/S0196-9781(96)00280-X. [DOI] [PubMed] [Google Scholar]
- 9.Goto T, Nakao K, Gunjigake KK, Kido MA, Kobayashi S, Tanaka T. Substance P stimulates late-stage rat osteoblastic bone formation through neurokinin-1 receptors. Neuropeptides. 2007;41:25–31. doi: 10.1016/j.npep.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 10.Wan M, Cao X. BMP signaling in skeletal development. Biochem Biophys Res Commun. 2005;328:651–657. doi: 10.1016/j.bbrc.2004.11.067. [DOI] [PubMed] [Google Scholar]
- 11.Stains JP, Civitelli R. Cell-to-cell interactions in bone. Biochem Biophys Res Commun. 2005;328:721–727. doi: 10.1016/j.bbrc.2004.11.078. [DOI] [PubMed] [Google Scholar]
- 12.Wrobel E, Leszczynska J, Przybylski J. [Role of Gap junctions in bone tissue] Postepy Biochem. 2011;57:411–417. [PubMed] [Google Scholar]
- 13.Ma WH, Zhang XM, Shi SS, et al. Establishment and identification of a normal human osteoblast system. Zhongguo Zuzhigongcheng Yanjiu yu Linchuang Kangfu. 2010;14:2661–2666. [Google Scholar]
- 14.Lee NJ, Herzog H. NPY regulation of bone remodelling. Neuropeptides. 2009;43:457–463. doi: 10.1016/j.npep.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 15.Sousa DM, McDonald MM, Mikulec K, Peacock L, Herzog H, Lamghari M, et al. Neuropeptide Y modulates fracture healing through Y1 receptor signaling. J Orthop Res. 2013;31:1570–1578. doi: 10.1002/jor.22400. [DOI] [PubMed] [Google Scholar]
- 16.Lee NJ, Doyle KL, Sainsbury A, Enriquez RF, Hort YJ, Riepler SJ, et al. Critical role for Y1 receptors in mesenchymal progenitor cell differentiation and osteoblast activity. J Bone Miner Res. 2010;25:1736–1747. doi: 10.1002/jbmr.61. [DOI] [PubMed] [Google Scholar]
- 17.Kurebayashi N, Sato M, Fujisawa T, Fukushima K, Tamura M. Regulation of neuropeptide Y Y1 receptor expression by bone morphogenetic protein 2 in C2C12 myoblasts. Biochem Biophys Res Commun. 2013;439:506–510. doi: 10.1016/j.bbrc.2013.09.014. [DOI] [PubMed] [Google Scholar]
- 18.Azuma H, Kido J, Ikedo D, Kataoka M, Nagata T. Substance P enhances the inhibition of osteoblastic cell differentiation induced by lipopolysaccharide from Porphyromonas gingivalis . J Periodontol. 2004;75:974–981. doi: 10.1902/jop.2004.75.7.974. [DOI] [PubMed] [Google Scholar]
- 19.Offley SC, Guo TZ, Wei T, Clark JD, Vogel H, Lindsey DP, et al. Capsaicin-sensitive sensory neurons contribute to the maintenance of trabecular bone integrity. J Bone Miner Res. 2005;20:257–267. doi: 10.1359/JBMR.041108. [DOI] [PubMed] [Google Scholar]
- 20.Maayan C, Becker Y, Gesundheit B, Girgis SI. Calcitonin gene related peptide in familial dysautonomia. Neuropeptides. 2001;35:189–195. doi: 10.1054/npep.2001.0863. [DOI] [PubMed] [Google Scholar]
- 21.Urist MR. Bone: formation by autoinduction. Science. 1965;150:893–899. doi: 10.1126/science.150.3698.893. [DOI] [PubMed] [Google Scholar]
- 22.Luyten FP, Cunningham NS, Ma S, Muthukumaran N, Hammonds RG, Nevins WB, et al. Purification and partial amino acid sequence of osteogenin, a protein initiating bone differentiation. J Biol Chem. 1989;264:13377–13380. [PubMed] [Google Scholar]
- 23.Wozney JM. The bone morphogenetic protein family and osteogenesis. Mol Reprod Dev. 1992;32:160–167. doi: 10.1002/mrd.1080320212. [DOI] [PubMed] [Google Scholar]
- 24.Chen D, Harris MA, Rossini G, Dunstan CR, Dallas SL, Feng JQ, et al. Bone morphogenetic protein 2 (BMP-2) enhances BMP-3, BMP-4, and bone cell differentiation marker gene expression during the induction of mineralized bone matrix formation in cultures of fetal rat calvarial osteoblasts. Calcif Tissue Int. 1997;60:283–290. doi: 10.1007/s002239900230. [DOI] [PubMed] [Google Scholar]
- 25.Chen D, Ji X, Harris MA, Feng JQ, Karsenty G, Celeste AJ, et al. Differential roles for bone morphogenetic protein (BMP) receptor type IB and IA in differentiation and specification of mesenchymal precursor cells to osteoblast and adipocyte lineages. J Cell Biol. 1998;142:295–305. doi: 10.1083/jcb.142.1.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Luppen CA, Smith E, Spevak L, Boskey AL, Frenkel B. Bone morphogenetic protein-2 restores mineralization in glucocorticoid-inhibited MC3T3-E1 osteoblast cultures. J Bone Miner Res. 2003;18:1186–1197. doi: 10.1359/jbmr.2003.18.7.1186. [DOI] [PubMed] [Google Scholar]
- 27.Koenig BB, Cook JS, Wolsing DH, Ting J, Tiesman JP, Correa PE, et al. Characterization and cloning of a receptor for BMP-2 and BMP-4 from NIH 3T3 cells. Mol Cell Biol. 1994;14:5961–5974. doi: 10.1128/MCB.14.9.5961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Macías-Silva M, Hoodless PA, Tang SJ, Buchwald M, Wrana JL. Specific activation of Smad1 signaling pathways by the BMP7 type I receptor, ALK2. J Biol Chem. 1998;273:25628–25636. doi: 10.1074/jbc.273.40.25628. [DOI] [PubMed] [Google Scholar]
- 29.Moustakas A, Heldin CH. From mono- to oligo-Smads: the heart of the matter in TGF-beta signal transduction. Genes Dev. 2002;16:1867–1871. doi: 10.1101/gad.1016802. [DOI] [PubMed] [Google Scholar]
- 30.Nishimura R, Kato Y, Chen D, Harris SE, Mundy GR, Yoneda T. Smad5 and DPC4 are key molecules in mediating BMP-2-induced osteoblastic differentiation of the pluripotent mesenchymal precursor cell line C2C12. J Biol Chem. 1998;273:1872–1879. doi: 10.1074/jbc.273.4.1872. [DOI] [PubMed] [Google Scholar]
- 31.Devlin RD, Du Z, Pereira RC, Kimble RB, Economides AN, Jorgetti V, et al. Skeletal overexpression of noggin results in osteopenia and reduced bone formation. Endocrinology. 2003;144:1972–1978. doi: 10.1210/en.2002-220918. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida Y, Tanaka S, Umemori H, Minowa O, Usui M, Ikematsu N, et al. Negative regulation of BMP/Smad signaling by Tob in osteoblasts. Cell. 2000;103:1085–1097. doi: 10.1016/S0092-8674(00)00211-7. [DOI] [PubMed] [Google Scholar]
- 33.Shi W, Chen H, Sun J, Chen C, Zhao J, Wang YL, et al. Overexpression of Smurf1 negatively regulates mouse embryonic lung branching morphogenesis by specifically reducing Smad1 and Smad5 proteins. Am J Physiol Lung Cell Mol Physiol. 2004;286:L293–L300. doi: 10.1152/ajplung.00228.2003. [DOI] [PubMed] [Google Scholar]
- 34.Civitelli R. Cell-cell communication in the osteoblast/osteocyte lineage. Arch Biochem Biophys. 2008;473:188–192. doi: 10.1016/j.abb.2008.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Civitelli R. Connexin43 modulation of osteoblast/osteocyte apoptosis: a potential therapeutic target? J Bone Miner Res. 2008;23:1709–1711. doi: 10.1359/jbmr.0811c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hashida Y, Nakahama K, Shimizu K, Akiyama M, Harada K, Morita I. Communication-dependent mineralization of osteoblasts via gap junctions. Bone. 2014;61:19–26. doi: 10.1016/j.bone.2013.12.031. [DOI] [PubMed] [Google Scholar]
- 37.McLachlan E, Plante I, Shao Q, Tong D, Kidder GM, Bernier SM, et al. ODDD-linked Cx43 mutants reduce endogenous Cx43 expression and function in osteoblasts and inhibit late stage differentiation. J Bone Miner Res. 2008;23:928–938. doi: 10.1359/jbmr.080217. [DOI] [PubMed] [Google Scholar]
- 38.Watkins M, Grimston SK, Norris JY, Guillotin B, Shaw A, Beniash E, et al. Osteoblast connexin43 modulates skeletal architecture by regulating both arms of bone remodeling. Mol Biol Cell. 2011;22:1240–1251. doi: 10.1091/mbc.E10-07-0571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chung DJ, Castro CH, Watkins M, Stains JP, Chung MY, Szejnfeld VL, et al. Low peak bone mass and attenuated anabolic response to parathyroid hormone in mice with an osteoblast-specific deletion of connexin43. J Cell Sci. 2006;119:4187–4198. doi: 10.1242/jcs.03162. [DOI] [PubMed] [Google Scholar]


