Abstract
It is tacitly understood that cell adhesion molecules (CAMs) are critically important for the development of cells, circuits, and synapses in the brain. What is less clear is what CAMs continue to contribute to brain structure and function after the early period of development. Here, we focus on the cadherin family of CAMs to first briefly recap their multidimensional roles in neural development and then to highlight emerging data showing that with maturity, cadherins become largely dispensible for maintaining neuronal and synaptic structure, instead displaying new and narrower roles at mature synapses where they critically regulate dynamic aspects of synaptic signaling, structural plasticity, and cognitive function. At mature synapses, cadherins are an integral component of multiprotein networks, modifying synaptic signaling, morphology, and plasticity through collaborative interactions with other CAM family members as well as a variety of neurotransmitter receptors, scaffolding proteins, and other effector molecules. Such recognition of the ever-evolving functions of synaptic cadherins may yield insight into the pathophysiology of brain disorders in which cadherins have been implicated and that manifest at different times of life.
1. INTRODUCTION
Our behaviors, thoughts, and actions reflect highly organized synaptic networks that are established principally during brain development, when molecular cues and neural activity collaborate to generate neural circuits that are progressively honed by sensory and motor experience during postnatal life (Benson, Colman, & Huntley, 2001). A large number of secreted and cell-surface molecular cues guide all phases of brain development, and key among these are structurally and functionally diverse families of cell adhesion molecules (CAMs). In particular, many different CAMs become concentrated at synapses, which are junctional sites of interneuronal communication, where they compose a transsynaptic adhesive apparatus that bridges rigidly apposed pre- and postsynaptic membranes across the intervening synaptic cleft (Benson & Huntley, 2012; Dalva, McClelland, & Kayser, 2007). There are two emergent features of synaptic CAMs that have modified our understanding of how CAMs contribute to the generation, maintenance, and pathological dysfunction of brain circuits. First, the function of CAMs is ever changing over developmental time, evolving in conformity with the complexities of the circuits they support. While initially providing spot welds of structural adhesion, they become dynamic and multifunctional signaling nodes that are integrated with a vast array of scaffolding, cytoskeletal, and effector proteins on both sides of the synapse, capable of exerting significant influence over neurotransmission. Second, CAMs of one family are unlikely to act in isolation of other CAM families, as recent data suggest surprisingly broad molecular cross talk between CAM families. Together, such emerging recognition of how and when CAMs interact has important implications for how they might contribute to brain disorders that manifest at different times in life. The goal of this chapter is to highlight some of these recent developments focusing on cadherins (calcium-dependent adherent proteins) and their varied roles in development and maintenance of synaptic circuits principally in the vertebrate nervous system.
2. THE CADHERIN SUPERFAMILY
Numerous reviews have comprehensively covered the organization and classification of the cadherin superfamily. Here, we outline the basics and refer the reader to other reviews for greater depth and detail (Hulpiau & van Roy, 2009; Yagi & Takeichi, 2000).
2.1. Classification and structure
Cadherins are an evolutionarily conserved family of adhesion proteins having first made their appearance in sponge epithelia where their contributions to cell–cell adhesion likely played a key role in the generation of multicellular animals (Abedin & King, 2008; Sakarya et al., 2007). Over the course of evolution, the cadherin repertoire has become extensive in vertebrates, containing 114 members in humans (Hulpiau, Gul, & van Roy, 2013). The superfamily is composed of Type I to Type IV cadherins, desmocollins, desmogleins, Flamingo/Celsr, clustered and nonclustered protocadherins (Pcdhs), Ret, Dachsous and Fat, calsyntenins, and primitive cadherins (Hulpiau & van Roy, 2009). All are membrane glycoproteins having two or more extracellular cadherin (EC) domains and conserved calcium-binding motifs. Type I and II cadherins, the desmosomal cadherins, Dachsous, Fat, Pcdhs, and calsyntenins engage in or regulate junctional adhesion in mammals. Some superfamily members appear to have evolved highly specialized functions. The desmosomal cadherins, which are specialized for the generation of desmosomes, and Dachsous and Fat bind heterophilically to generate a specialized adhesion site that helps to confer apical identity important for the generation of an organized cerebral cortex in mouse and planar cell polarity in Drosophila (Ishiuchi, Misaki, Yonemura, Takeichi, & Tanoue, 2009; Thomas & Strutt, 2012). Others, such as Flamingo in Drosophila and the Celsr family in vertebrates, as well as the calsyntenins, play important and varied roles in brain development that partially overlap with some classic cadherin functions. However, their mechanism(s) of action appears to be mostly distinct from other superfamily members (Berger-Muller & Suzuki, 2011; Boutin, Goffinet, & Tissir, 2012; Pettem et al., 2013; Ster et al., 2014; Um et al., 2014). Here, we will focus principally on those cadherins that participate in and regulate synapse adhesion in mammals: Type I and Type II classic cadherins and Pcdhs. Hereafter, we will refer to classic cadherins simply as “cadherins.”
Most cadherins engage in homophilic interactions mediated principally by N-terminal EC domains, but the binding interactions differ between groups. Homophilic adhesion between Type I cadherins is mediated by an N-terminal β-strand containing a conserved tryptophan (Trp2) in the first of five EC domains that engages a hydrophobic pocket in the apposing EC1 to generate an extended, curved trans dimer. Because the intermolecular interaction between the two apposing cadherins is the same as would be formed intramolecularly, the domains are “swapped” (Boggon et al., 2002; Shapiro et al., 1995; Shapiro & Weis, 2009). In the presence of physiological levels of calcium, the extracellular domain becomes more rigid (Nagar, Overduin, Ikura, & Rini, 1996; Pokutta, Herrenknecht, Kemler, & Engel, 1994) and trans interactions are favored over intramolec-ular interactions. Individual binding interactions are relatively weak, but when viewed by high-resolution electron microscopy, cadherin-based junctions can form highly ordered arrays, suggesting that strong adhesion is conferred by organized numbers (Al-Amoudi, Diez, Betts, & Frangakis, 2007). The degree to which cadherins interact in cis in vivo is not well understood. The binding interface is such that cis and trans interactions between EC1 domains are mutually exclusive, consistent with the increase in cis binding and loss of trans binding seen in the absence of calcium (Shapiro & Weis, 2009). At the same time, there may be some cis interactions that can coexist with trans binding. Crystal structure data suggest that specific cis interactions can form between EC1 and a site near the EC2–EC3 linker regions of some classic cadherins, and EM data suggest that there can be cis interactions at desmosomes (Shapiro & Weis, 2009; Wu, Vendome, Shapiro, Ben-Shaul, & Honig, 2011). In contrast, homophilic binding between T-cadherin (cadherin-13) occurs only via crossed contacts at a region between EC1 and EC2 (Ciatto et al., 2010), an alternate adhesive conformation utilized in other classic cadherins as well, either as a binding intermediate that facilitates transition into or out of the strand-swapped dimer (Harrison et al., 2010; Hong, Troyanovsky, & Troyanovsky, 2011; Nagar et al., 1996; Pertz et al., 1999), or as a preferred interaction under certain circumstances. Single molecule force measurements suggest that tensile force can promote the generation of crossed or X-dimers that become stronger when pulled and weaker when released (Manibog, Li, Rakshit, & Sivasankar, 2014; Rakshit, Zhang, Manibog, Shafraz, & Sivasankar, 2012), actions that would be useful for generating traction for cell or growth cone migration. Type II cadherins utilize an adhesion mechanism similar to that of Type I cadherins. They can form the crossed intermediate, but they have two conserved tryptophans in EC1 (Trp2 and Trp4) that bind a larger, apposing hydrophobic pocket (Patel et al., 2006).
Classic cadherins share a highly similar intracellular motif with a conserved juxtacellular domain and an extended β-catenin-binding motif (Huber & Weis, 2001). The juxtamembrane domain binds p120 and δ-catenins, which regulate lateral clustering, surface distribution, and interactions with secondary binding partners (Rubio, Curcio, Chauvet, & Bruses, 2005; Silverman et al., 2007; Yap, Niessen, & Gumbiner, 1998); β-catenin provides an interface with a wide variety of partners, including α-catenin, which provides a dynamic bridge to actin cytoskeleton (Yamada, Pokutta, Drees, Weis, & Nelson, 2005). Thus, different cadherins bind similar intracellular partners and similarly engage in intracellular anchoring and adhesion, and specificity is driven by trans engagement with particular cadherin partners as well as by binding preferentially to particular cis partners.
Protocadherins have six or seven EC repeats and a short cytoplasmic domain. They are divided into the clustered Pcdhs (α-, β-, and γ-Pcdhs), a term that refers to their genomic organization (Wu & Maniatis, 1999), and the nonclustered δ-Pcdhs (Hulpiau & van Roy, 2009). Multiple versions of α- and γ-Pcdhs are generated from mRNAs having different transcription start sites spliced to shared α or γ constant C-terminal exons (Sugino et al., 2000; Tasic et al., 2002; Wu et al., 2001). β-Pcdhs are generated similarly, but do not have a constant C-terminal domain (Hirano et al., 2012; Wang, Su, & Bradley, 2002). δ-Pcdhs are encoded by separate genes and have two or three conserved cytoplasmic motifs (Vanhalst, Kools, Staes, van Roy, & Redies, 2005).
A study of the molecular basis underlying adhesion between γ-Pcdhs indicates that binding is homophilic, only partially reliant on calcium, and requires the EC1 domain for binding but utilizes EC2 and EC3 (rather than EC1) for recognition (Schreiner & Weiner, 2010). γ-Pcdhs readily form cis multimers with themselves and other Pcdhs (Murata, Hamada, Morishita, Mutoh, & Yagi, 2004; Schreiner & Weiner, 2010) in a manner that suggests that combinatorial Pcdh tetramers will bind similar tetramers in trans (Schreiner & Weiner, 2010), conferring a rigorous recognition code. Based on the structure of α4-Pcdh (Morishita et al., 2006), Pcdhs have a much shallower hydrophobic pocket in their EC1 domain and lack the conserved Trp residues seen in classic cadherins, but the adhesive interactions shown by γ-Pcdhs strongly suggest that all Pcdhs are likely to be able to use their EC domains to generate adhesion. However, such adhesion must be highly regulated as homophilic adhesion between α-Pcdhs has not yet been demonstrated, and surface expression of α- and γ-Pcdhs is highly regulated. The C-terminal domain of γ-Pcdhs acts to retain most γ-Pcdhs in an intra-membranous pool (Fernandez-Monreal, Kang, & Phillips, 2009; O’Leary et al., 2011), and α-Pcdhs appear to use γ-Pcdhs to transit to the membrane surface (Murata et al., 2004). Additionally, some α-Pcdhs can engage in heterophilic interactions with integrins via an RGD motif in EC1 (Morishita et al., 2006; Mutoh, Hamada, Senzaki, Murata, & Yagi, 2004), and many Pcdhs have shown cis interactions with classic cadherins (Weiner & Jontes, 2013), suggesting that EC domains may confer cis as well as trans interaction specificity.
2.2. Cadherin localization in the central nervous system
Cadherins display distinct expression profiles in different regions, circuits, and cell types of the central nervous system (CNS). Their differential spatial and temporal distribution thus influences development and function of these highly structured areas. In general, Type I cadherins, and particularly N-cadherin, which has been the most extensively studied, are broadly expressed in the CNS, whereas Type II cadherins and protocadherins exhibit distinct, and partially overlapping, expression patterns.
The mammalian neocortex contains functionally and structurally distinct areas built upon a highly organized, laminated structure. While gradients of extracellular signaling molecules and transcription factors influence the formation of cortical regions and their laminar structure (reviewed in Sansom & Livesey, 2009), cadherins are also involved in these processes. N-Cadherin is required for development of proper laminar organization, as conditional knockout (KO) of N-cadherin, driven by Cre recombinase under the control of the D6 promoter which inactivates N-cadherin early in embryonic development, causes complete disruption of cortical laminar organization (Kadowaki et al., 2007). In contrast, extensive in situ hybridization studies have revealed that some Type I cadherins (e.g., R-cadherin), Type II cadherins, and Pcdhs mostly exhibit restricted and/or combinatorial expression that is specific to cells, layers and/or cortical regions (Bekirov, Needleman, Zhang, & Benson, 2002; Gil, Needleman, & Huntley, 2002; Hertel & Redies, 2011; Suzuki, Inoue, Kimura, Tanaka, & Takeichi, 1997; Lefebvre et al., 2012; Uemera et al., 2007). Adjacent cortical regions are delineated by unique expression patterns of one or more cadherin(s). For example, cadherin-8 is abundantly expressed in the anterior cingulate cortex (a medial cortical region), while cadherin-6 is concentrated in the adjacent primary motor cortex (Hertel & Redies, 2011). In contrast, cadherin-6, -8, and -10 are all expressed in somatosensory cortex, but exhibit only partially overlapping expression patterns (Gil et al., 2002). Additionally, regions that send projections to other cortical or subcortical areas display similar expression profiles as their targets, suggesting that specific cadherins, or combinations of cadherins, mediate the formation of brain circuits. For example, cadherin-8 is expressed by corticostriatal projection neurons in layer 5 of prefrontal cortex and by medium spiny neurons in striatum and becomes concentrated at striatal excitatory synapses early in postnatal development when this projection is establishing (Fig. 1), suggesting that cadherin-8 is involved in the formation and/or targeting of the corticostriatal projection (Friedman et al., 2014). Similarly, the expression pattern of cadherin-8 in structures of the trigeminal somatosensory system suggests a role in the precise synaptic targeting of specific projections from somatosensory thalamus to the somatosensory (barrel) cortex (Gil et al., 2002). Additionally, several different Type II cadherins are expressed differentially within rat hippocampus. Whereas in situ hybridization shows that cadherin-8 is expressed uniformly throughout CA1, CA3, and dentate gyrus, cadherin-10 is most abundantly expressed in CA1, and cadherin-9 in CA3 (Bekirov et al., 2002; Williams et al., 2011).
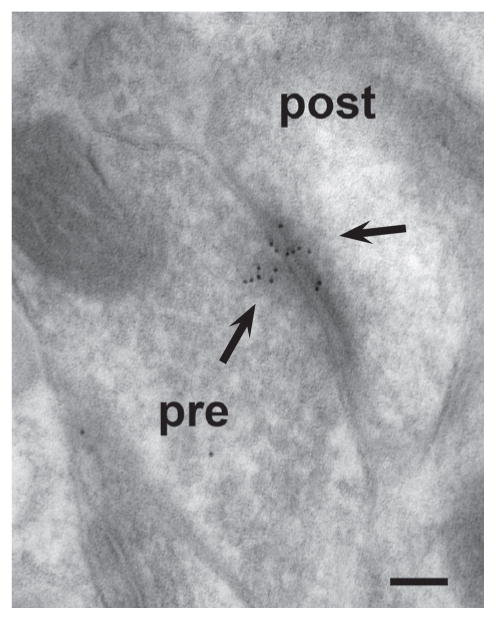
Figure 1.
Cadherin-8 is synaptically localized in striatum. This electron micrograph was taken from a section through the dorsal striatum of a P30 mouse that had been processed for immunogold localization of cadherin-8. The gold particles are clustered on both presynaptic (pre) and postsynaptic (post) sides of the asymmetric synapse (arrows), consistent with homophilic recognition, and this is a pattern of localization that is typical for classic cadherins in the brain. Bar, 100 nm.
Although Type I cadherins typically have a rather broad distribution, in the spinal cord, two different Type I cadherins delineate functionally distinct nociceptive synaptic pathways within lamina II of the dorsal horn. E-Cadherin is restricted to synapses formed by nonpeptidergic nociceptive afferents that occupy the inner portion of lamina II (Brock, Elste, & Huntley, 2004; Patil, Brock, Colman, & Huntley, 2009; Shimamura, Takahashi, & Takeichi, 1992), while N-cadherin is found at peptidergic nociceptive afferents that target the outer portion of lamina II (Brock et al., 2004).
Several types of neuroglia, the support cells of the CNS, also express classic cadherins. Astrocytes express N-cadherin and other Type I classic cadherins, but generally do not express Type II cadherins (Gil et al., 2002; Karpowicz et al., 2009; Tomaselli, Neugebauer, Bixby, Lilien, & Reichardt, 1988). During early embryonic development, radial glia, which can divide to form neural and glial progenitors, act as scaffolds to guide newly born neurons during migration to their target layer (Marin & Rubenstein, 2003; Miyata, Kawaguchi, Okano, & Ogawa, 2001). Cadherin-7 is expressed in radial glia and neurons in the basal plate of the spinal cord in chicken (Luo, Ju, & Redies, 2006), suggesting homophilic binding between two cell types aids in early radial migration.
Oligodendrocytes are myelin-producing glia that ensheath axons to facilitate saltatory conduction. Myelination is highly regulated by molecular cues that control specific axon–oligodendrocyte interactions (Bradl & Lassmann, 2010). Several studies suggest that N-cadherin plays an important role in oligodendrocyte targeting and adhesion with axons during the first steps of myelination. N-Cadherin is expressed in cultured oligodendrocytes and is concentrated on major processes in oligodendrocyte progenitors (OPs; Payne, Hemperly, & Lemmon, 1996). Blocking N-cadherin reduces OP migration on astrocyte monolayers (Schnadelbach et al., 2000) and impairs oligodendrocyte-axon adhesion in OPs cocultured on dorsal root ganglia explants or rat cerebellar slices (Schnadelbach, Ozen, Blaschuk, Meyer, & Fawcett, 2001). Recently, cadherin-19 was identified as a glial-specific cadherin that may be involved in myelination of the avian CNS. Although cadherin-19 expression is absent in rat brain (Takahashi & Osumi, 2005), oligodendrocytes in chicken spinal cord and brain selectively express cadherin-19 (Lin, Luo, & Redies, 2010).
3. FUNCTIONAL UNITS OF ADHESION AND SIGNALING: INTRA- AND INTER-CAM FAMILY CROSS TALK (TABLE 1)
Table 1.
Interactions between cadherins and transmembrane-binding partners
| Cadherin | cis/trans interactions | Purpose | Citation |
|---|---|---|---|
| N-Cadherin | δ-Pcdh-8/arcadlin | Activity-mediated induction of δ-Pcdh-8 binding in trans promotes N-cadherin internalization in cis; cis interactions also observed between PAPC and C-cadherin in Xenopus | Yasuda et al. (2007) and Chen and Gumbiner (2006) |
| PAPC (Xenopus homolog of δ-Pcdh-8/arcadlin) | Flrt3 (LRRP) PAPC | cis interactions appear to be via C-terminal domains. PAPC attenuates Flrt3 inhibition of C-cadherin adhesion | Chen and Gumbiner (2006) and Chen, Koh, Yoder, and Gumbiner (2009) |
| N-Cadherin | FGFR | N-Cadherin physically interacts with FGFR1 in breast cancer cells via Ig domains; blocking FGF2 inhibits N-cadherin- mediated growth | Suyama, Shapiro, Guttman, and Hazan (2002) and Williams et al. (2001) |
| N- and M-cadherin | CDO and BOC | Ig superfamily members CDO and BOC bind N-cadherin and M-cadherin in cis and promote muscle cell differentiation | Kang, Feinleib, Knox, Ketteringham, and Krauss (2003) |
| N-Cadherin | R-cadherin | cis or trans; does not appear to influence adhesion specificity | Shan et al. (2000) |
| N-Cadherin | GluA2 | GluA2 binds N-cadherin extracellular domain in cis or trans. Interaction needed to support GluA2 overexpression-mediated increase in dendritic spines and mEPSCs | Saglietti et al. (2007) |
| N-Cadherin | GluA1 | In vitro studies in HEK cells are suggestive of cis interactions that support surface GluA1 expression | Nuriya and Huganir (2006) |
| N-Cadherin | δ-Pcdh-19 | Cis interactions cooperate and promote adhesion mediated by δ-Pcdh-19; δ-Pcdh-17 acts similarly | Biswas, Emond, and Jontes (2010) and Emond, Biswas, Blevins, and Jontes (2011) |
| γ-Pcdh | Other γ-Pcdhs | Increases binding specificity. Heteromers form in cis and bind homomers in trans | Schreiner and Weiner (2010) |
| γ-Pcdhs | α-Pcdhs | cis interactions via C-termini as well as extracellular interactions. γ-Pcdhs promote surface distribution of α-Pcdhs | Murata et al. (2004) |
| Pcdh-α, -γ | Ret | Site of cis interaction is not clear. Stimulation by GDNF serves to stabilize both Ret and the Pcdhs | Schalm, Ballif, Buchanan, Phillips, and Maniatis (2010) |
| Pcdh- γC5 | γ2-GABAA receptor subunit | C-term cis interactions that may be unique to γC5. Promotes surface localization of GABAA receptors and appears to promote inhibitory synapse stability | Li et al. (2012) |
| Pcdh-α-4 (CNR1) | β1-integrin | In vitro studies suggest trans binding and adhesion between integrins expressed in HEK cells and the RGD site in Pcdh-α-4 | Mutoh et al. (2004) |
Since classic cadherins and many Pcdhs adhere homophilically in trans, some aspects of cadherin-based adhesion are symmetric with the same intra-cellular partners present on either side of a junction. However, since synapses are polarized, some pre- or postsynaptic-binding partners differ based on local availability or on regulated binding interactions. The complex C-terminal interactions have been reviewed in depth by others (Anderson & Benson, 2006; Arikkath & Reichardt, 2008; Brigidi & Bamji, 2011) and will not be detailed here, but should be noted as a key means by which the strength of cadherin-based adhesion can be adjusted. Fewer intracellular partners have been established for the Pcdhs and those that have been identified appear to be both molecularly and functionally unique to particular Pcdh families (Chen & Gumbiner, 2006; Chen et al., 2009; Nakao, Platek, Hirano, & Takeichi, 2008).
Pre- to postsynaptic adhesion between homophilic pairs gains a layer of specificity by the addition of cis- and trans-binding partners (Table 1). Classic cadherins commonly engage in cis interactions with Pcdhs (Weiner & Jontes, 2013). For example, N-cadherin can bind in cis with either Pcdh-19 or -17 and promote homophilic adhesion in trans between the Pcdhs (Biswas et al., 2010; Emond et al., 2011). Recent work shows that self-association between phrenic neurons during development requires expression of both cadherin-10 and Pcdh-10, suggesting that the two may be acting as an adhesive complex (Machado et al., 2014). α- and β-Pcdhs can be found in complexes with γ-Pcdhs (Han, Lin, Meng, & Wang, 2010), and γ-Pcdhs can form heteromeric tetramers in cis and partner with similar complexes in trans, increasing the specificity of interaction (Schreiner & Weiner, 2010). A more limited number of classic cadherins can form cis heteromers, but there is no evidence that they prefer heteromers over homomers in trans (Shan et al., 2000). Pcdh-8 (δ2 Pcdh, arcadlin) or PAPC serves to reduce adhesion by classic cadherins. In mouse neurons, Pcdh-8 promotes the internalization of N-cadherin (Yasuda et al., 2007), but interactions between PAPC and Flrt3 outline additional means to modulate adhesion (Chen et al., 2009).
Several additional heterophilic cis partners are particularly relevant for cadherin function at synapses. Pcdh-γC5 interacts directly with GABAA-γ receptor subunit via C-terminal domain regions. This interaction is not syn-aptogenic, but appears to augment the stability of receptors on the surface (Li et al., 2012). N-Cadherin can bind AMPA-type glutamate receptor subunits GluA1, GluA2, or GluA4, and at least for GluA2, such interactions can be in cis or in trans via an extracellular domain and appear to have a stabilizing effect on the surface levels of GluA subunits (Nuriya & Huganir, 2006; Saglietti et al., 2007). Indirect interactions between N-cadherin and neuro-ligins, which are probably mediated by the scaffolding protein S-SCAM (MAGI-2), positively modulate synapse formation in developing neurons (Aiga, Levinson, & Bamji, 2011; Jontes, Emond, & Smith, 2004; Stan et al., 2010). Notably, in cultured neurons less than 2 weeks old, N-cadherin is required for neuroligin to have its synaptogenic effect (Stan et al., 2010).
4. DEVELOPMENTAL PHASES OF CIRCUIT ASSEMBLY: EVOLVING ROLES OF THE CADHERIN-BASED ADHESIVE NETWORK
There are phases of circuit formation by which neurons in one brain area (the source) become synaptically coupled to neurons in another brain region (the target; Benson et al., 2001). These phases, which can be overlapping in time, include (1) the generation of source and target neurons (neurogenesis) in germinal zones and migration to their respective terminal locations; (2) the initiation, outgrowth, and targeting of axons from source neurons; (3) elaboration of postsynaptic dendrites and dendritic spines of target neurons; and (4) contact between pre- and postsynaptic membranes and subsequent synaptogenesis between source and target neurons. Cadherins have been implicated in all of these developmental phases, but their contributions at each step are mechanistically distinct (for a comprehensive review of developmental roles, see Hirano & Takeichi, 2012).
4.1. Neurogenesis and migration
A role for cadherin-based adhesive interactions in neurogenesis was speculated upon in early studies showing that neural tissue histogenesis was profoundly disrupted by cadherin function-neutralizing antibodies (Takeichi, Inuzuka, Shimamura, Matsunaga, & Nose, 1990). More recent studies have implicated N-cadherin in regulating proliferation of dopamine neuron progenitors in the ventral brainstem in mice (Sakane & Miyamoto, 2013). Similarly, N-cadherin regulates retinal neurogenesis in zebrafish (Nagashima, Barthel, & Raymond, 2013; Yamaguchi, Imai, Tonou-Fujimori, & Masai, 2010), where N-cadherin and Slit-Robo signaling collaborate to allow detachment of newborn retinal ganglion cells (RGCs) from the apical surface of the neuroepithelium following their terminal mitosis (Wong, Baudet, Norden, Leung, & Harris, 2012). Similar disruptions of N-cadherin-mediated apical attachments of neuroepithelial cells in mice lead to profoundly disorganized cortical structures (Kadowaki et al., 2007). Certain miRNAs (those belonging to the miR379–410 cluster) can bind directly to the 3′-UTR of N-cadherin transcripts and modulate N-cadherin levels in neocortical neural progenitor cells, which in turn controls the proliferation and subsequent migration of young neurons (Rago, Beattie, Taylor, & Winter, 2014). Dynamic trafficking of N-cadherin regulates migration of neocortical glutamatergic neurons along radial glial guides (Kawauchi et al., 2010). Additionally, cortical GABA interneurons require N-cadherin for both tangential migration (from their distant site of origin in the medial ganglionic eminence) to the neocortex and radial migration (which delivers them to their proper cortical layer; Luccardini et al., 2013).
4.2. Axon outgrowth and targeting
There are extensive and compelling data using both in vitro and in vivo approaches in different model systems showing that classic cadherins promote axon fasciculation and are a potent substrate for axonal outgrowth. Early studies showed that many different types of cultured neurons exhibit robust axonal outgrowth when grown on a purified N-cadherin substrate, on astrocytes, or on various cell lines expressing N-cadherin on their surface (Bixby & Zhang, 1990; Doherty, Skaper, Moore, Leon, & Walsh, 1992; Matsunaga, Hatta, Nagafuchi, & Takeichi, 1988; Tomaselli et al., 1988), effects that are blocked in the presence of N-cadherin-neutralizing antibodies (Doherty, Rowett, Moore, Mann, & Walsh, 1991; Drazba & Lemmon, 1990; Neugebauer, Tomaselli, Lilien, & Reichardt, 1988) or interfering peptides targeting the conserved HAV sequence in Type I cadherins (Blaschuk, Sullivan, David, & Pouliot, 1990). More recent genetic analyses in Drosophila and zebrafish also provide strong evidence for N-cadherin and R-cadherin in axon extension, growth cone guidance, and targeting (Babb et al., 2005; Bruses, 2011; Lee, Herman, Clandinin, Lee, & Zipursky, 2001; Nern, Zhu, & Zipursky, 2008; Schwabe, Neuert, & Clandinin, 2013). Cadherin dominant-negative reagents have also been used to investigate cadherin-based outgrowth and targeting. One such reagent, NcadΔE, lacks most of the ectodomain, but retains transmembrane and intracellular (catenin-binding) domains. As such, it inhibits the function of all classic cadherins by competing for β-catenin binding at the membrane (Bozdagi, Valcin, Poskanzer, Tanaka, & Benson, 2004; Kintner, 1992). Hip-pocampal neurons expressing NcadΔE exhibit significantly impaired axonal growth when grown on N-cadherin-expressing L-cells fibroblasts (Bozdagi et al., 2004), which do not express any classic cadherins endogenously (Nose, Nagafuchi, & Takeichi, 1988; Shan et al., 2000). When NcadΔE was expressed in vivo by Xenopus RGCs, about 70% of them failed to emit an axon (a failure of axonogenesis), and of those that did, their axon growth was significantly retarded, never reaching the optic tectum, their normal target (Riehl et al., 1996). Some Type II cadherins, such as cadherins 6B, 7, and 11, also promote axonal growth and branching (Barnes, Price, Wentzel, & Guthrie, 2010; Marthiens et al., 2005).
Mechanistically, the axon outgrowth-promoting effects of N-cadherin and other classic cadherins may involve association with coreceptors. Presenting cerebellar neurons with soluble N-cadherin-Fc causes coclustering of endogenous surface N-cadherin and the fibroblast growth factor (FGF) receptor and potently stimulates axon outgrowth in an FGF receptor-dependent manner (Utton, Eickholt, Howell, Wallis, & Doherty, 2001). The FGF receptor may be a common effector of classic cadherin-mediated outgrowth, as studies have shown that cadherin-11 promotes neurite outgrowth by recruiting the FGF receptor through ectodomain binding and triggering downstream effectors involving PLCγ/DAG lipase/CAM kinase and PI3 kinase pathways (Boscher & Mege, 2008; Williams, Mittal, Walsh, & Doherty, 1995; Williams, Walsh, & Doherty, 1994). Other studies support a model in which neurite outgrowth is coordinately regulated by molecular cross talk between N-cadherin and β1-integrin through differential coupling to the protein tyrosine kinase Fer (Arregui, Pathre, Lilien, & Balsamo, 2000). Intracellularly, cadherin-mediated outgrowth is also dependent on distinct signaling cascades initiated by protein tyrosine phosphatase-mu (PTPmu; Burden-Gulley & Brady-Kalnay, 1999; Oblander & Brady-Kalnay, 2010).
While many classic cadherins promote axon extension, others appear to constrain axon growth and targeting. For example, in hippocampal organotypic cultures, the axons of dentate gyrus granule neurons (mossy fibers) normally grow in a tightly fasciculated bundle to innervate CA3 pyramidal cell dendrites principally in stratum lucidum, as they do in vivo (Frotscher & Heimrich, 1995). However, treating hippocampal organotypic cultures with an exogenous cadherin-8 peptide inhibitor leads to exuberant mossy fiber growth, loss of fasciculation, and loss of laminar specificity (Bekirov, Nagy, Svoronos, Huntley, & Benson, 2008). These effects are not likely to represent off-target effects of N-cadherin blockade, because the cadherin-8 peptide inhibitor enhances axon outgrowth on a cadherin-8 substrate, but has no effects on outgrowth on an N-cadherin substrate. Similarly, the extension of motor neuron axons expressing T-cadherin is significantly impaired when grown on T-cadherin substrates (Fredette, Miller, & Ranscht, 1996). Together, these studies show that cadherins work coordinately to promote and constrain fasciculation and extension of growing axon tracts (Treubert-Zimmermann, Heyers, & Redies, 2002; Wöhrn et al., 1999).
In addition to affecting axon outgrowth, classic cadherins can also act as a “stop signal” to direct ingrowing axons to their correct terminal target layer or region. For example, N-cadherin becomes concentrated in ret-inorecipient layers of chick optic tectum contemporaneously with the arrival of retinal axons and subsequent synaptogenesis, but fails to do so in enucleated embryos (Yamagata, Herman, & Sanes, 1995). Treating tectal slices with N-cadherin function-blocking antibodies during this early period of ingrowth impairs laminar targeting of ingrowing retinal axons (Inoue & Sanes, 1997). Similarly, in the mammalian visual system, RGC axons in cadherin-6-deficient mice fail to innervate their appropriate visual targets, but instead project to inappropriate visual nuclei (Osterhout et al., 2011). In rodent barrel cortex—the part of primary somatosensory cortex containing an isomorphic map of the large facial whiskers of the contralateral snout in layer 4—thalamocortical inputs from two distinct nuclei, the ventral posterior medial (VPM) nucleus, and the medial division of the posterior nucleus (POm), both target layer 4 but these two thalamic streams terminate in mutually exclusive, interdigitating zones within layer 4. Studies have shown that N-cadherin becomes concentrated at thalamocortical synapses of one pathway (VPM-to-layer 4 synapses in barrel centers; Huntley & Benson, 1999), while cadherin-8 becomes concentrated at thalamocortical synapses of the other pathway (POm-to-layer 4 synapses in barrel septa; Gil et al., 2002) during the early postnatal period when these projections form. Such cadherin specificity and matching between source (thalamic nucleus) and target (layer 4 neurons) suggest that these cadherins play an important role in the targeting of these two converging, but separated thalamic input streams to layer 4. In partial confirmation of this idea, when N-cadherin function-neutralizing antibodies are applied to organotypic cocultures of thalamus and somatosensory cortex, thalamic axons grow normally through deep layers at the same rate as they grow through deep layers in untreated cocultures, but instead of terminating in their target layer 4, they grow inappropriately through layer 4 to reach the pial surface (Poskanzer, Needleman, Bozdagi, & Huntley, 2003). Direct evidence that Type II cadherins coordinate synaptic targeting and specificity comes from recent studies showing that cadherin-9 is required for selective targeting and synapse formation between hippocampal dentate gyrus (DG) granule neurons and CA3 pyramidal neurons (Williams et al., 2011). This study showed that in culture, DG granule neurons that had equal access to all hippocampal cell types, nonetheless preferentially formed synapses with their natural target, CA3 neurons, rather than indiscriminately with CA1 neurons. The molecular basis for such selective synaptic partnering involves cadherin-9, as loss of this cadherin from either DG neurons or CA3 neurons disrupted mossy fiber targeting and synapse formation.
Some members of the Pcdh family have also been implicated in axon targeting. For example, deletion of the constant region of the α-Pcdh gene cluster in mice leads to abnormal targeting of olfactory axons to their appropriate glomeruli in the olfactory bulb (Hasegawa et al., 2008) and to inhomogeneities in the density of serotonergic innervation in several structures throughout the brain (Katori et al., 2009). Other studies in mice have shown that genetic deletion of Pcdh-10 (a δ-Pcdh) impairs striatal axon outgrowth and impedes the thalamocortical projection through the ventral telenceph-alon (Uemura, Nakao, Suzuki, Takeichi, & Hirano, 2007). Inactivation of Celsr3 causes hypotrophy of the cortex and impedes tract formation of anterior commissure and corticospinal, corticofugal, and thalamocortical tracts in Celsr3 KO mice (Tissir, Bar, Jossin, De Backer, & Goffinet, 2005).
4.3. Elaboration of dendrites, arborization, and self-avoidance
Classic cadherins and γ-Pcdhs have been implicated in dendritic development (Garrett, Schreiner, Lobas, & Weiner, 2012; Hirano & Takeichi, 2012; Zhu & Luo, 2004). In dissociated hippocampal cultures, N-cadherin promotes dendritic outgrowth (Esch, Lemmon, & Banker, 2000), while in organotypic hippocampal slice cultures, interfering with N-cadherin leads to impaired dendritic arbors of CA3 pyramidal neurons attributed to both an arrest in initial dendrite growth and an increase in den-drite retraction (Bekirov et al., 2008). In Drosophila, DN-cadherin functions in dendrodendritic interactions between olfactory projection neurons that restrict dendritic arbors to a single glomerulus (Zhu & Luo, 2004). Similarly, activity-mediated enhancement of dendritic arborization in cultured hippo-campal neurons leads to increased surface expression of N-cadherin/ β-catenin and requires N-cadherin/β-catenin interactions between neurons (Tan, Peng, Song, Zheng, & Yu, 2010). The stabilization of dendritic arbors by classic cadherins may require interactions with other effectors. For example, in chick retina, RGC dendritic arbors are rapidly destabilized and retract following β1-integrin blockade; disrupting the juxtamembrane region of N-cadherin generates a β1-integrin inactivation signal leading to dendritic retraction (Marrs et al., 2006). In cultured cortical neurons, reducing levels of afadin, which is a Ras/Rap effector that associates with and regulates the cadherin/catenin system as well as nectins, Ephs/ephrins, and neurexins (Beaudoin et al., 2012; Buchert et al., 1999; Takai, Ikeda, Ogita, & Rikitake, 2008), leads to impaired dendritic growth (Srivastava et al., 2012). While blocking N-cadherin generally seems to impair dendritic growth, overexpression of N-cadherin, β-catenin, or αN-catenin in hippo-campal neurons promotes dendritic growth and complexity (Peng et al., 2009; Yu & Malenka, 2003).
An important binding partner of classic cadherins that also regulates development and maintenance of dendritic arbors is δ-catenin, a member of the p120-catenin family whose localization is restricted to dendrites and spines (Kosik, Donahue, Israely, Liu, & Ochiishi, 2005) and whose hemizygous loss in humans is associated with the mental retardation syndrome Cri-du-Chat (Medina, Marinescu, Overhauser, & Kosik, 2000). In cultured hippocampal neurons, dendritic branching is enhanced by overexpression of δ-catenin; conversely, dendritic morphogenesis is impaired by δ-catenin ablation (Arikkath et al., 2008; Kim et al., 2008). In contrast, in δ-catenin KO mice examined in vivo, cortical neuronal dendritic complexity was entirely normal through 5 postnatal weeks, but thereafter, significantly regressed, resulting in late postnatal abnormalities in cortical responsiveness and receptive field properties (Matter, Pribadi, Liu, & Trachtenberg, 2009). Because δ-catenin is a multifunctional protein with a number of interacting signaling and scaffolding partners in addition to cadherins (Brigidi & Bamji, 2011), it is not entirely clear whether or to what extent such regulation of dendritic development or maintenance by δ-catenin reflects interactions with cadherins.
Recent studies have shown that γ-Pcdhs regulate development of cortical pyramidal cell dendritic arbors (Garrett et al., 2012). Using a conditional allele that restricts disruption of the entire gene cluster to cortical pyramidal neurons and astrocytes starting in embryonic life, mutant pyramidal neurons failed to develop their typical complex dendritic arbors, particularly those that ramify in layer 1, but there were no obvious effects of deleting the γ-Pcdh gene cluster on migration, lamination, or synaptogenesis. In contrast to embryonic deletion, using an inducible mutant allele that disrupted the γ-Pcdh cluster after cortical dendritic arbors formed failed to affect dendritic complexity, demonstrating that γ-Pcdhs are required for the elaboration, but not maintenance, of cortical dendritic complexity. Functional analyses indicated that γ-Pcdhs promoted dendritic arborization by regulating a focal adhesion kinase/protein kinase C/MARCKS signal cascade (Garrett et al., 2012).
Dendrite self-avoidance, where dendritic branches of the same neuron repel each other so as to ensure even coverage of receptive territory (Grueber & Sagasti, 2010), is another function that has been attributed to γ-Pcdh. Conditionally ablating the γ-Pcdh gene cluster in retinal starburst amacrine cells or cerebellar Purkinje cells resulted in a cell-autonomous failure in dendritic self-avoidance (Lefebvre, Kostadinov, Chen, Maniatis, & Sanes, 2012). Interestingly, molecular replacement in KO neurons with a single γ-Pcdh isoform restored self-avoidance, but decreased interactions between dendrites of neighboring neurons. Thus, homophilic recognition between sister branches expressing the same γ-Pcdh isoforms mediated a repulsive signal to ensure self-avoidance. However, because neuronal expression of different γ-Pcdh isoforms is stochastic (Toyoda et al., 2014) and isoform interactions are both combinatorial (in cis) and strictly homo-philic (in trans; Schreiner & Weiner, 2010), dendritic branches from neighboring neurons are normally free to intermingle because of the low probability that neighboring neurons would express exactly the same combinations of isoforms.
A potential role for cadherin-8 in dendrite arborization and self-avoidance was recently described (Friedman et al., 2014). Cultured cortical neurons in which levels of cadherin-8 were significantly reduced by siRNA-mediated cadherin-8 knockdown exhibited increased numbers of dendritic branches and an increased number of dendrite self-crossings, suggesting a defect in dendrite self-avoidance.
4.4. Synaptogenesis, dendritic spine formation, and synaptic plasticity in developing neurons
Synaptogenesis is initiated by adhesion between heterophilic binding proteins, some of which are targeted to axons and the others to cell bodies and dendrites. Such synaptogenic proteins are specialized for the generation of excitatory or inhibitory synapses, and heterophilic binding ensures that resulting synapses are polarized, forming mostly between presynaptic axon terminals and postsynaptic cell bodies or dendrites. Neither classic cadherins nor Pcdhs alone can induce the formation of synapses, but strong evidence indicates that in young neurons, classic cadherins are essential partners in the combinatorial process of synapse adhesion and stabilization, and Pcdhs can act as priming factors promoting synaptogenesis (Garrett & Weiner, 2009; Li et al., 2012; Waites, Craig, & Garner, 2005).
Proteins that can induce the formation of synapses have been most commonly identified in an assay in which the protein in question is expressed in a nonneuronal cell line, like COS or HEK cells, that is cocultured with neurons and then assayed for the presence of heterologous “synapses” (Scheiffele, Fan, Choih, Fetter, & Serafini, 2000). While not sufficient for inducing synapses on their own, classic cadherins are required for the inductive process of synaptogenesis because synapses cannot be induced in neurons or cell lines that express synaptogenic proteins, but lack classic cadherins (Aiga et al., 2011; Jungling et al., 2006; Stan et al., 2010) or that overexpress inhibitors of classic cadherin adhesion (Bozdagi et al., 2004; Togashi et al., 2002). Consistent with this early and essential role, N-cadherin and β-catenin are concentrated and distributed evenly at both GABAergic and glutamatergic nascent junctions (Benson & Tanaka, 1998; Elste & Benson, 2006; Jontes et al., 2004; Yamagata et al., 1995), and synapses are lost when cadherins are removed or their adhesion is inhibited (Togashi et al., 2002; Zhang & Benson, 2001). Cadherin actions at synapses can be regulated selectively as β-catenin can be synthesized locally at sites of early contact (Huttelmaier et al., 2005; Taylor, Wu, Tai, & Schuman, 2013), and unprocessed N-cadherin retaining its N-terminal prodomain can compete with processed N-cadherin at synapses to reduce adhesion and decrease synapse number (Reines et al., 2012). Also, N-cadherin may be partially synaptogenic for cholinergic synapses as it can induce the formation of pre-synaptic terminals with heterologous cells when accompanied by p120-catenin, but such sites do not recruit postsynaptic cholinergic receptors and are not functional (Flannery & Bruses, 2012).
A principal role for the N-cadherin/β-catenin complex at nascent synapses is in the assembly of presynaptic vesicle clusters. Expression of the dominant-negative NcadΔE greatly reduces size, intensity, and function of presynaptic terminals (Bozdagi et al., 2004; Togashi et al., 2002), a finding that resembles cultured neurons expressing defective β-catenin (Bamji et al., 2003). While pan-cadherin manipulations show the strongest effects, stem-cell-derived neurons having a selective deletion of N-cadherin show impaired vesicle recruitment and short-term presynaptic plasticity ( Jungling et al., 2006). The mechanism is not fully worked out, but data from cultured neurons suggest that β-catenin tethered by N-cadherin binds to the scaffolding protein, Scribble, and this complex appears to recruit β-pix, a Rac/Cdc42 guanine-exchange factor that can enhance actin poly-merization (Sun, Aiga, Yoshida, Humbert, & Bamji, 2009; Sun & Bamji, 2011). Since the generation and maintenance of synaptic vesicle clusters in young neurons are also dependent on an intact F-actin cytoskeleton (Morales, Colicos, & Goda, 2000; Zhang & Benson, 2001), cadherins likely regulate vesicle clusters by promoting the generation of a physical restraint. Additionally, N-cadherin may promote synapse maturation by facilitating calcium influx in developing neurons through an N-cadherin/RhoA/ cytoskeletal cascade that affects voltage-gated calcium channels (Marrs, Theisen, & Bruses, 2009).
Classic cadherin adhesion also influences the normal generation and early stability of dendritic spines, highly specialized protrusions receiving the bulk of excitatory synaptic input. Neurons expressing a pan-cadherin dominant-negative construct produce thin and pointed dendritic protrusions that nevertheless retain synaptic contact (Togashi et al., 2002). The phenotype closely resembles that seen in neurons in which F-actin has been depolymerized (Zhang & Benson, 2001) and is reproduced in neurons lacking αN-catenin (Abe, Chisaka, Van Roy, & Takeichi, 2004; Togashi et al., 2002), which links classic cadherin tails to F-actin cytoskeleton. Collectively, the data support the view that classic cadherin adhesion recruits and probably stabilizes F-actin in developing dendritic spines.
Pcdhs have a pleiomorphic and less well understood impact on synapse formation. Based on their molecular diversity, timing of expression, and unique enrichment in vertebrate brain, most early work on Pcdhs conjectured that family members would drive synapse recognition and adhesion. There are few data that support this idea, but Pcdhs clearly regulate synaptogenesis (Weiner & Jontes, 2013). Mice lacking all γ-Pcdhs have a decreased density of excitatory and inhibitory synapses in spinal cord. While it was initially suspected that the decreased density could be due to the higher rates of cell death that are also seen in the absence of γ-Pcdhs (Wang, Weiner, et al., 2002), these two effects are separable as synapse density remains diminished even when the effects on cell death are bypassed by crossing the γ-Pcdhs KO mice to mice lacking the proapoptotic protein Bax (Weiner, Wang, Tapia, & Sanes, 2005). The synapse promoting effect appears to arise, at least in part, from γ-Pcdh expression in astrocytes and binding to neuronal γ-Pcdh. Astrocytes produce a variety of soluble priming factors that promote synaptogenesis (Ullian, Christopherson, & Barres, 2004; Waites et al., 2005), but this priming effect is substantially (albeit, transiently) inhibited when astrocytes lacking γ-Pcdhs are cocultured with wild-type spinal cord neurons, presumably by limiting neuron-astrocyte contact (Garrett & Weiner, 2009). Cultured hippocampal neurons lacking a subgroup of γ-Pcdhs (γ-Pcdh-C3, -C4, and -C5) have fewer GABAergic synapses. Here, however, the action is not on priming new synapse formation, but rather is on postsynaptic cis interactions between this group of γ-Pcdhs and GABAA receptor γ2 subunits, which appear to be important for stabilizing inhibitory synapses (Li et al., 2012). In an entirely different role, in dorsal striatum, δ-Pcdh-17 is dispensable for the normal targeting and generation of synapses, but appears to limit presynaptic vesicle number and distribution in adolescent mice (Hoshina et al., 2013). In all cases, the actions of particular Pcdhs appear to be restricted to particular neuron groups or areas.
The actions of classic and Pcdhs at the synapse appear to change over the course of synapse development. This has been best studied with classic cadherins. Once synapses have formed, classic cadherin localization assumes a more clustered distribution either within synapses, where they form discrete sites of adhesion, or sometimes within adjacent and extrasynaptic adhe-rens junctions (Elste & Benson, 2006; Uchida, Honjo, Johnson, Wheelock, & Takeichi, 1996). At this stage of synapse development, classic cadherins can be removed from synapses and the basic structure of synapses is still retained (Zhang & Benson, 2001). However, interfering with cadherin-based adhesion at this time alters the shape and/or stability of dendritic spines (Abe et al., 2004; Arikkath et al., 2009; Israely et al., 2004; Mendez, De Roo, Poglia, Klauser, & Muller, 2010) in the absence of synapse loss. Additionally, presynaptic N-cadherin is essential for maintaining vesicle availability during high-frequency stimulation ( Jungling et al., 2006).
In adolescent rodent hippocampus and in cultured hippocampal neurons older than about 3 weeks (“mature” cultures), N-cadherin regulates lasting forms of synapse plasticity. Lasting changes in synapses involve both changes in size (bigger synapses are stronger synapses) and changes in the efficacy of pre- to postsynaptic communication. In cultured neurons, strong stimulation of glutamate receptors induces the expansion of dendritic spine heads and a temporary relocalization of N-cadherin (Okamura et al., 2004; Tanaka et al., 2000; Yam et al., 2013). While the precise mechanism is not understood, maintained spine enlargement in cultured neurons requires N-cadherin and an intact actin cytoskeleton (Okamura et al., 2004). In what may be a related process, N-cadherin is also required for the stabilization of newly formed dendritic spines induced by long-term potentiation (LTP) in organotypic hippocampal slice cultures (Mendez et al., 2010). Data from cultured neurons suggest that such activity-driven changes in N-cadherin adhesion are mediated by regulated recruitment and stabilization of β-catenin to active synapses (Murase, Mosser, & Schuman, 2002), as well as by a more stable form of cadherin adhesion (Tanaka et al., 2000). In acute hippocampal slices, N-cadherin adhesion is required for the induction (Tang, Hung, & Schuman, 1998) and maintenance (Bozdagi, Shan, Tanaka, Benson, & Huntley, 2000; Bozdagi et al., 2010) of LTP at CA1 synapses. As mentioned, N-cadherin can interact in cis with AMPA receptor subunits GluA1 and GluA2, providing a direct mechanism to stabilize AMPA-type glutamate receptors at sites of synaptic adhesion. Additionally, the C-terminal domain of N-cadherin can bind directly to the scaffolding protein GRIP1, which can independently bind GluA2 (and probably GluA3) and motor protein KIF5. All of these proteins can be found in trafficking vesicles, suggesting a means by which increases in synapse size and activity could be coupled (Heisler et al., 2014).
The converse of synaptic LTP is synaptic long-term depression (LTD). Brief stimulation of cultured neurons with NMDA, which can induce the internalization of AMPA-type glutamate receptors and produce LTD, alters the regulated endocytosis of N-cadherin (Tai, Mysore, Chiu, & Schuman, 2007) and severs links between AKAP79 (A Kinase-Anchoring Protein) and an N-cadherin/β-catenin complex in a process likely to diminish phosphor-ylation and promote internalization of AMPA receptors (Dell’Acqua et al., 2006; Gorski, Gomez, Scott, & Dell’Acqua, 2005). Over a longer time scale (hours vs. minutes), strong stimulation in cultured hippocampal neurons promotes homophilic Pcdh-8 binding, activation of TAO2β kinase, a C-terminal Pcdh-8-binding partner, and MEK3 activator and increases p38 MAP kinase activity, all of which serves to promote N-cadherin recycling and decrease adhesion (Yasuda et al., 2007). Collectively, the data obtained from young to adolescent neurons suggest that strong, but dynamic N-cadherin-based synaptic adhesion stabilizes dendritic spines and can increase the strength of glutamatergic synapses. However, one caveat is that the effects of this ubiquitous glutamatergic synaptic cadherin are commonly used to make generalizations about the actions of the family, but they may be distinct. For example, mice lacking cadherin-11 show enhanced, rather than diminished, hippocampal LTP (Manabe et al., 2000).
5. BEYOND DEVELOPMENT: WHAT DOES THE CADHERIN–CATENIN TRANSSYNAPTIC NETWORK CONTRIBUTE TO BRAIN FUNCTION THROUGHOUT LIFE?
A great deal is now known about the multifaceted ways in which cadherins contribute to structural and functional development of brain circuits, particularly stages encompassing generation of synaptic connectivity, as attested to above. However, CAMs generally and cadherins particularly remain prominent components of synapses into adulthood, well beyond the developmental period of circuit and synapse formation. This raises some important questions: What do cadherins contribute to mature synapse and brain function? Are their developmental roles merely extended into maturity or do they take on different functions? If so, how are these functions achieved molecularly? It has generally not been possible to address these questions in the types of cell culture models that dominate studies of cadherin function during development because neurons grown in culture never achieve the level of maturity and functional complexity equivalent to neurons in an adult brain. Additionally, cell culture models cannot be used to assess behavioral consequences of altering the function of the cadherin–catenin transsynaptic apparatus. Accordingly, several recent studies have tackled these and related questions using similar Cre–LoxP-based strategies to conditionally manipulate cadherin and/or catenin levels, stability and function in the hippocampus of mice after the period of early brain development, and assessing connectivity, synaptic plasticity, and cognitive and behavioral performance in adulthood. The postnatal timing of the genetic manipulation in these studies ensures that the cadherin/catenin system is operational during early stages of brain development.
5.1. Cadherins become dispensible for maintaining hippocampal connectivity but regulate dynamic aspects of synaptic function and plasticity
Several recent studies used Cre–LoxP technology to postnatally manipulate cadherins/β-catenin with a Cre-driver line of mice in which Cre recombinase was driven by the α-calcium–calmodulin-dependent protein kinase type-II (αCaMKII) promoter (Tsien et al., 1996). This Cre-driver line ensures conditional gene manipulation starting around the third postna-tal week in hippocampal and other forebrain excitatory neurons, but does not affect GABA neurons or glial cells, which do not express αCaMKII. In one set of studies (Bozdagi et al., 2010; Nikitczuk et al., 2014), synaptic N-cadherin was ablated and β-catenin levels were lowered in excitatory neurons of hippocampus. In a second study (Mills et al., 2014), synaptic cadherins in hippocampal excitatory neurons were surface stabilized by a heterozygous mutation of β-catenin that rendered the molecule resistant to degradation. This mutant β-catenin showed enhanced association with synaptic cadherins, but otherwise retained all of its natural binding sites and functional abilities, including interactions with αN-catenin and components of the Wnt-signaling pathway. Because a pan-cadherin antibody was used for synaptic localization, it is not possible to know definitively which cadherin(s) were stabilized at the synapse by this particular β-catenin mutation. Remarkably, both of these studies showed that regardless of whether N-cadherin was eliminated (and β-catenin reduced) at synapses or cadherins (and β-catenin) were stabilized at synapses, adult hippocampal structure, dendritic organization, synapse and spine density, and baseline properties of synaptic neurotransmission were entirely indistinguishable from adult control (floxed) mice. Additionally, there were no (Bozdagi et al., 2010) or only very subtle (Mills et al., 2014) effects on presynaptic release. The lack of effects of two very different manipulations of synaptic cadherin/β-catenin levels and function on any of these parameters of hippocampal architecture, baseline neurotransmission, and presynaptic release was surprising, given the broad developmental roles the cadherin–catenin system has in establishing all of them. Instead, both sets of studies showed profound deficits in persistence of long-lasting forms of synaptic plasticity at mutant hippocampal synapses, as well as impairments in cognitive flexibility, and both point to the idea that deficits in trafficking or localization of particular glutamate receptor subunits underlie such synaptic plasticity and cognitive deficits. Previous studies had shown that through ectodomain interactions, N-cadherin could bind in cis with GluA1 to control its surface stability (Nuriya & Huganir, 2006), as well as in cis or in trans with GluA2 (Saglietti et al., 2007), but the significance of these interactions to adult synaptic and behavioral function in vivo had not been clear.
In the case of the conditional N-cadherin KO mice (Bozdagi et al., 2010; Nikitczuk et al., 2014), LTP and concurrent spine enlargement at adult CA1 synapses were both induced normally, but both became unstable, collapsing rapidly to baseline levels of synaptic strength or spine size, indicating a deficit in maintenance. In contrast, LTD of mutant CA1 synapses was unaffected. Normally, at wild-type CA1 synapses, both LTP and LTD require NMDA receptors (NMDARs) for induction (Collingridge & Bliss, 1987), but depend on modifications in the function and trafficking of AMPAR subunits for persistence (Malinow & Malenka, 2002). In particular, the insertion of GluA1 into the synaptic membrane, which is enabled by the scaffolding protein PSD95 as part of an activity-regulated delivery pathway (Ehlers, Heine, Groc, Lee, & Choquet, 2007; Opazo & Choquet, 2011; Passafaro, Piech, & Sheng, 2001; Schnell et al., 2002; Stein, House, Bredt, & Nicoll, 2003; Yang, Wang, Frerking, & Zhou, 2008; Yudowski et al., 2007), is required for, and links, the maintenance of both LTP and spine enlargement (Ehrlich & Malinow, 2004; Kopec, Li, Wei, Boehm, & Malinow, 2006; Kopec, Real, Kessels, & Malinow, 2007; Park, Penick, Edwards, Kauer, & Ehlers, 2004; Shi et al., 1999; Yang et al., 2008). Consistent with the plasticity deficits observed in the mutant mice, further analysis revealed that N-cadherin-deficient excitatory synapses contained normal levels and distribution of NMDAR subunits (hence, normal LTP/LTD induction), but significantly lower levels and altered distribution of both GluA1 and PSD95. This suggests that without N-cadherin, delivery, insertion, and/or trapping of GluA1 at the synaptic membrane is deficient, thus rendering both structural and functional forms of plasticity unstable. Additionally, one recent model proposed that N-cadherin is part of a transsynaptic molecular complex in which postsynaptic δ-catenin links N-cadherin to both the AMPARs and the NMDAR–CamKII complex, thus acting as a molecular “tag” at potentiated synapses to coordinately regulate LTP-associated changes in synaptic strength with pre- to postsynaptic synapse growth (Sanhueza & Lisman, 2013).
In contrast to changes in GluA1 and PSD95, levels and localization of GluA2 and its scaffolding protein S-SCAM/MAGI-2 were unaffected at N-cadherin-deficient synapses, which may have accounted for normal LTD and baseline properties of neurotransmission. LTD requires endocytocis of GluRs, particularly GluA2, from the synaptic membrane (Malinow & Malenka, 2002), while baseline neurotransmission at CA1 synapses is mediated mostly by non-GluA1-containing AMPARs (Danielson et al., 2012; Mainen, Jia, Roder, & Malinow, 1998; Zamanillo et al., 1999).
In the case of the cadherin/β-catenin-stabilized mutant mice (Mills et al., 2014), there were different plasticity deficits that were observed in comparison with the N-cadherin-deficient mice. Nevertheless, such deficits could also be accounted for by defective AMPAR trafficking that was largely complementary to that suggested by the N-cadherin-deficient mice. In the cadherin/β-catenin-stabilized mice, both the induction and the persistence of LTP at CA1 synapses were unaffected. In contrast, the persistence of NMDAR-dependent LTD was abolished. Unlike the N-cadherin-deficient mice, however, there were no effects of the β-catenin-stabilizing mutation on overall levels of GluA1, GluA2, NMDAR subunits, or PSD95. However, immunoelectron microscopy of CA1 synapses showed LTD-related effects on local synaptic trafficking of both cadherin and GluA1. Under basal conditions, mutant synapses had a greater accumulation of cadherin (as expected from stabilization) in comparison with floxed control slices, but a normal accumulation of GluA1. In contrast, inducing LTD in control slices (by bath-applied NMDA, a potent chemical LTD induction protocol) resulted in a reduction of both synaptic N-cadherin and GluA1, indicating that both are endocytosed from the synaptic membrane in response to NMDA. This is somewhat at odds with previous studies that have shown that NMDA treatment stabilizes surface N-cadherin via heightened β-catenin binding (Tai et al., 2007), while simultaneously accelerating GluR internalization to produce LTD (Beattie et al., 2000; Ehlers, 2000), a discrepancy likely attributable to vastly different experimental conditions between studies [dissociated neurons in culture, (Tai et al., 2007) vs acute slices from 1-year-old mice (Mills et al., 2014)]. Nevertheless, in mutant slices, NMDA treatment failed to change the synaptic accumulation of both cadherin and GluA1, suggesting that stabilized cadherins trapped GluRs in the synaptic membrane, thereby preventing GluR endocytocis and thus the LTD-associated decrease in synaptic signal strength. The fact that LTP was normal in these mutant mice presumably indicates that insertion/trapping of GluA1 in response to LTP is not affected by stabilized N-cadherin. N-cadherin-GluA2 interactions may also be important for regulating a form of LTD that requires metabotropic GluRs (Zhou, Hu, Passafaro, Xie, & Jia, 2011). Figure 2 highlights a model of how the cadherin/catenin system can differentially regulate trafficking and stability of GluRs at adult synapses to bidirectionally control persistent increases or decreases in synaptic strength all within the framework of existing connectivity.
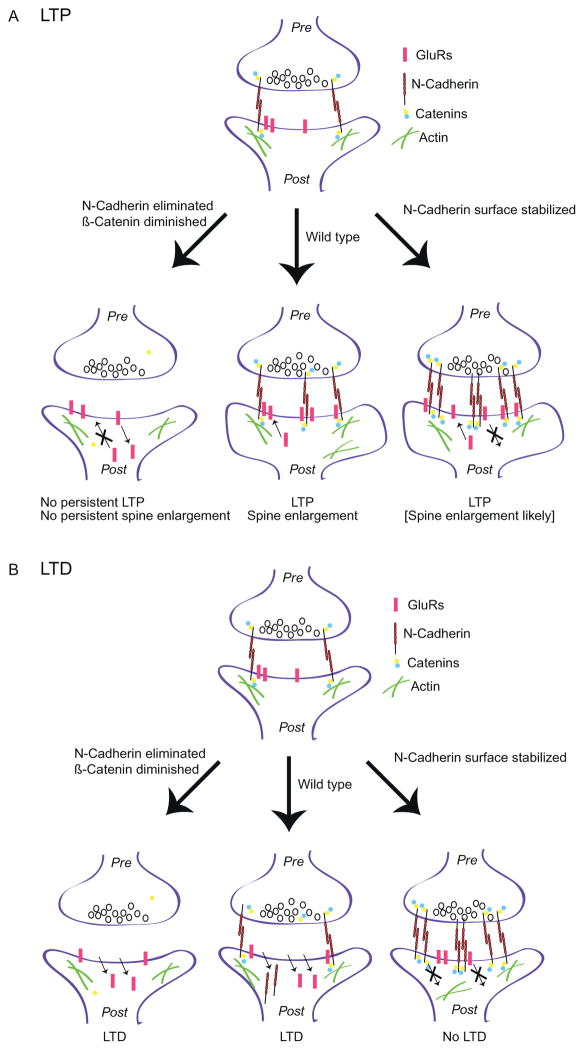
Figure 2.
Schematic diagram showing how the cadherin/catenin system can bidirec-tionally regulate synaptic plasticity by differentially controlling GluR subunit trafficking and/or stability at the surface. (A) During LTP, a synapse (top) becomes strengthened (wild-type synapse, bottom middle) by the insertion of GluRs into the membrane (small arrow in the spine head) and also undergoes a coordinated increase in the size of the spine head. N-Cadherin facilitates both processes by trapping and/or promoting GluR positioning through direct ectodomain interactions as well as through linkage to the actin cytoskeleton. In the case of the conditional N-cadherin deletion mutant (bottom left synapse), GluRs fail to insert and/or fail to become trapped at the synapse in the absence of N-cadherin (blocked arrow), thus persistent LTP and spine enlargement are both abolished. GluR endocytocis is presumably unaffected (small arrow), thus accounting for normal LTD in these mice. In the case of the cadherin/β-catenin stabilized mice (bottom right synapse), excessively stabilized surface N-cadherin has no effect on GluR insertion (small arrow), thus accounting for normal LTP in these mice. However, the surface-stabilized N-cadherin abnormally traps synaptic GluRs, thus preventing GluR endocytocis (blocked arrow) and blocking LTD. (B) During LTD, wild-type synapses (middle bottom) undergo endocytocis of both N-cadherin and GluRs from the synaptic surface (small arrows). In the N-cadherin conditional KO synapse (left synapse), GluR endocytocis is presumably not affected, thus yielding normal LTD. In contrast, when N-cadherin is surface stabilized (right synapse), GluRs are abnormally trapped, preventing endocytocis, thus these synapses fail to exhibit LTD. This cartoon is based on the studies of Bozdagi et al. (2010), Mills et al. (2014), and Nikitczuk et al. (2014).
In addition to apparent roles in trafficking or trapping of AMPA-type receptors during LTP or LTD, the N-cadherin/β-catenin system has also been shown to regulate trafficking and surface stabilization of kainate receptor subunits (Coussen et al., 2002) and nicotinic acetylcholine receptor subunits (Bruses, 2013). Thus, cadherins may function broadly to promote trafficking or stabilization of a variety of neurotransmitter receptor types.
Interestingly, not all classic cadherins respond similarly to plasticity-inducing synaptic activity in adulthood. For example, while both cadherin-8 and N-cadherin are localized to mature synapses formed by perforant path axons and DG granule neurons, an electrical stimulation paradigm that produces robust LTP and synaptic morphological remodeling at such synapses leads to the selective loss of cadherin-8 but the retention of N-cadherin (Huntley et al., 2012). Although the significance of such differential responses is unknown, the data suggest that activity-mediated loss and retention of different cadherins could promote both flexibility (enabling morphological plasticity) and stability (to maintain synaptic apposition).
5.2. Postnatal deletion of N-cadherin may alter the balance of excitation and inhibition
The studies discussed above show that altering N-cadherin/β-catenin function in mature hippocampal neurons has little or no effect on baseline properties of excitatory neurotransmission, but produces profound changes in plasticity of the excitatory synapses at which these molecules are normally located. However, further examination of the conditional N-cadherin KO mice showed that postnatal deletion of N-cadherin from excitatory synapses also had significant effects on GABAergic inhibitory synapses (Nikitczuk et al., 2014). This was surprising because N-cadherin is not normally found at hippocampal or other cortical GABA synapses in maturity (Benson & Tanaka, 1998; Huntley & Benson, 1999). When examined in adulthood, the density of puncta immunolabeled for pre- and postsynaptic molecular markers of GABA synapses was significantly elevated in the hippocampus of conditional N-cadherin KO mice, suggesting functionally enhanced inhibition. This possibility was indirectly confirmed by examining the severity of kainic acid-induced seizures in these adult mice as a proxy for overall levels of GABA inhibition, as GABA inhibition generally can regulate seizure threshold (Ben-Ari & Cossart, 2000). As expected from the anatomical results, adult conditional N-cadherin KO mice displayed a significantly diminished severity of kainic acid-induced seizures. The mechanism(s) behind such apparent changes in inhibitory synapses following the deletion of N-cadherin from excitatory synapses is not yet known, but there are several possibilities. One is that compensatory changes in levels or localization of other classic cadherins could have affected numbers of GABA synapses, as GABA synapse numbers are reduced in cultured hippocampal neurons when levels of E-cadherin, cadherin-11, or cadherin-13 are reduced (Fiederling, Ewert, Andreyeva, Jungling, & Gottmann, 2011; Paradis et al., 2007). Alternatively, PSD95 collaborates with different neuroligins to specify excitatory and inhibitory synapses (Graf, Zhang, Jin,Linhoff, & Craig, 2004; Levinson et al., 2005, 2010; Prange, Wong, Gerrow, Wang, & El-Husseini, 2004). The diminished levels of PSD95 in the N-cadherin KO mice could have altered neuroligin specificity resulting in the promotion of GABA synapses. Finally, since the conditional deletion of N-cadherin did not affect N-cadherin expression by GABAergic interneurons, it is possible that abnormally asymmetric (non-homophilic) partnering of N-cadherin presented on the surface of GABA neurons with other molecules, either in cis or in trans, could have promoted GABA synapse proliferation (Pielarski et al., 2013).
5.3. Deficits in cognitive flexibility in adult cadherin-mutant mice
Learning, memory, and other forms of information processing require various forms of persistent synaptic plasticity as well as a normal balance of excitation and inhibition (Silva, 2003; Yizhar et al., 2011) which are the very aspects of synaptic and network function that are altered in adult hippocampus when levels, localization, and function of the cadherin/ catenin system are perturbed. As expected from such cellular and molecular alterations, disrupting the cadherin/catenin system in adulthood produces selective deficits in the ability of mutant mice to adapt quickly to unexpectedly changing contingencies in several hippocampal-based behavioral tasks, an ability that wild-type mice readily display and which is called here “cognitive flexibility.” Such deficits in cognitive flexibility occur without overt affects on the ability of the mice to learn task rules initially.
In one set of studies using the adult conditional N-cadherin KO mice in which persistent LTP was impaired (Nikitczuk et al., 2014), animals were trained in a six-arm radial water maze to find the location of a submerged escape platform located at the end of one arm (the goal arm). Animals were placed randomly in one of the other five start arms at the beginning of each trial and were given one initial sample trial to find the escape platform by trial-and-error, then tested on subsequent match trials for memory of the position of the hidden platform in the goal arm, which remained constant during each day of testing, but changed across days. Performance measurements included entry into both correct and incorrect goal arms, swim-path lengths, and time to reach the platform (escape latency). Both mutant and control mice learned the general rules of the task at the same rates and performed similarly in the sample trials, where memory could not guide performance. However, in subsequent match trials, mutant mice were significantly impaired in their memory for spatial episodes. Interestingly, the pattern of errors made by the control mice suggested memory for the previous day’s goal arm (perseverative errors), while the mutant mice showed no evidence for perseverative errors. Control experiments ruled out any deficits in motivation, motor abilities, or sensory perceptual abilities. Interestingly, when the “cognitive load” of the task was increased by changing the position of the goal arm three times during each daily block of testing, both mutant and control mice performed similarly during the first 2 days of testing, but by the last 2 days of testing, while the control mice had adapted to the changing contingencies and performed significantly better across all measures than during the first 2 days, the mutant mice failed to adapt, revealing a deficit in the cognitive flexibility necessary to adapt to the changing task requirements.
A complimentary set of deficits in cognitive flexibility was observed in the cadherin/β-catenin-stabilized mutant mice in which LTD was deficient (Mills et al., 2014). Mutant and control mice were trained to find the location of a hidden escape platform using a standard Morris water maze. Like the N-cadherin conditional KO mice, the cadherin/β-catenin-stabilized mutant mice learned the initial rules of the task at similar rates to the control mice. However, after 5 days of testing where the platform remained in the same position, the escape platform was unexpectedly shifted to a new location. Control mice adapted quickly, finding the new location and eliminating entry into the previous goal location. In contrast, the mutant mice largely failed to learn the new position, instead displaying repeated entry into the previous goal position (perseverative errors). The ability to perform successfully in this task requires the extinction of the memory for the position of the initial goal platform, which is thought to require LTD (Dong et al., 2013). Thus, the deficient LTD displayed by these mutant mice likely contributed to their particular deficits in cognitive flexibility.
Other studies have shown that conditional deletion of β-catenin disrupted persistent storage of newly learned fear memories (Maguschak & Ressler, 2008), while application of exogenous HAV peptide blockers of Type I cadherins into dorsal hippocampus of adult mice impaired contextual fear conditioning (Schrick et al., 2007). Furthermore, mutations in δ-catenin have been associated with significant deficits in cognitive function (Israely et al., 2004), which may involve activity-dependent interactions between palmitoylated δ-catenin and N-cadherin that stabilizes N-cadherin at synapses and regulates GluR insertion (Brigidi et al., 2014).
6. CADHERIN-BASED TRANSSYNAPTIC NETWORKS IN PATHOPHYSIOLOGY OF BRAIN CIRCUITS
Various neurodevelopmental and psychiatric disorders are associated with genetic variations in proteins that mediate brain connectivity and synaptic function. Accumulating evidence implicates defects in cadherin-based adhesion and signal transduction in many of these disorders (Redies, Hertel, & Hubner, 2012). Aberrant function of Type I cadherins is also involved in some neurological disorders, as well as neuroprotective repair mechanisms following CNS injury. Finally, recent studies suggest that dysfunctional cadherin signaling may contribute to some types of neurodegen-erative disease.
6.1. Neurodevelopmental disorders
Autism spectrum disorders (ASDs) are a clinically heterogeneous group of neurodevelopmental conditions defined by the following core characteristics: impaired reciprocal social interactions, communication deficits, and repetitive restrictive behaviors (Pardo & Eberhart, 2007). Additionally, affected individuals commonly have comorbid anxiety disorders, attention deficit/hyperactivity disorder (ADHD), obsessive compulsive disorder (OCD), Tourette syndrome, or schizophrenia (Geurts et al., 2008; Rapoport, Chavez, Greenstein, Addington, & Gogtay, 2009; Simonoff et al., 2008). Extensive genetic studies have identified inherited and de novo copy number variants (CNVs) and single nucleotide polymorphisms (SNPs) in classic cadherins and Pcdhs that are associated with ASDs and comorbid disorders.
Data from the Autism Genome Project Consortium implicated a rare familial microdeletion on chromosome 16q21, containing only the gene encoding cadherin-8, in two families affected by autism and intellectual disability (ID; Pagnamenta et al., 2011). Genetic variations in nongenic regions that alter cadherin gene expression represent risk factors for ASD. Rare inherited CNVs in the intergenic region immediately upstream of cadherin-8 have been identified as new ASD susceptibility loci (Walker & Scherer, 2013). Genome-wide association studies have also uncovered autism-linked SNPs in the intergenic region between cadherin-9 and cadherin-10 genes on chromosome 5p14.1 (Wang et al., 2009). Homozygos-ity mapping of individuals with related parents revealed a large deletion within a protocadherin cluster proximal to the gene that encodes Pcdh-10 (Morrow et al., 2008), which promotes activity-dependent synapse elimination (Tsai et al., 2012). ASDs have a very high rate of de novo mutations in individuals with unaffected parents. A study of rare de novo CNVs found a microdeletion on 16q23, which disrupts cadherin-13 (Sanders et al., 2011).
Children with autism often exhibit ID (Matson & Shoemaker, 2009). Gene variants of cadherin-15 that disrupt its cell–cell adhesion function were found in patients with ID (Bhalla et al., 2008). Mutations in proteins that directly interact with cadherins have also been shown to be associated with ID. CTNND2 encodes δ-catenin which is deleted in Cri-du-Chat syndrome, a chromosomal disorder involving 5p chromosomal deletions that cause severe ID and speech impairment (Israely et al., 2004).
N-cadherin has numerous developmental and synaptic functions (discussed in the earlier sections), and its dysfunction is associated with multiple disorders; however until recently, no neurodevelopmental disease-linked N-cadherin mutation had been identified. Exon sequencing in a subset of OCD and Tourette’s patients identified rare single nucleotide missense variants in cadherin-2, which encodes N-cadherin (Moya et al., 2013). Although this study identified the domains that contain the genetic variants, it remains unknown how these SNPs affect N-cadherin function (Moya et al., 2013).
While several independent genome-wide association analyses have identified SNPs and CNVs in cadherin-13 that are associated with ADHD (Rivero et al., 2013), multiple studies have implicated cadherin-13 in a variety of neuropsychiatric disorders, including major depressive disorder (Sibille et al., 2009), substance abuse (Hart et al., 2012), schizophrenia (Borglum et al., 2014), and autism (Sanders et al., 2011). Although cadherin-13 contains the conserved extracellular domains of classic cadherins, it is linked to the cell membrane by a glycosylphosphatidylinositol anchor and lacks transmembrane and cytoplasmic domains (Ranscht & Dours-Zimmermann, 1991). Unlike other classic cadherins and Pcdhs, mutations in cadherin-13 may have pleiotropic effects due to its atypical structure and downstream signaling.
6.2. Seizure/epilepsy
A seizure-induced injury in the hippocampus promotes the loss of CA3 neurons and synaptic reorganization of mossy fibers that leads to de novo synapse formation and recurrent excitatory firing (Wenzel, Woolley, Robbins, & Schwartzkroin, 2000). N-Cadherin expression markedly increases 1–4 weeks following seizure induction in hippocampus, which is consistent with the time course of mossy fiber axon sprouting (Shan, Yoshida, Wu, Huntley, & Colman, 2002). Multiple familial and de novo mutations or whole-gene deletions of PCDH-19 (δ2 nonclustered Pcdh) have been linked to epilepsy and mental retardation limited to females (EFMR; Depienne et al., 2011; Dibbens et al., 2008; Jamal, Basran, Newton, Wang, & Milunsky, 2010). Affected EFMR individuals experience recurrent seizures that begin in infancy and mild to moderate ID and autism (Ryan et al., 1997). Due to the multiple mutations associated with Pcdh-19 loss of function, it is not surprising that EFMR patients have highly variable clinical features. However, unlike other cadherins associated with psychiatric disorders, PCDH-19 is located on the X-chromosome. Mono-zygotic twins carrying the same Pcdh-19 mutation can exhibit different clinical features (Higurashi et al., 2012), suggesting that random X inactivation of EFMR-linked PCDH-19 may lead to varying severity of symptoms (Depienne & LeGuern, 2012). Male carriers of the Pcdh-19 mutations do not show signs of epilepsy, but are susceptible to behaviors associated with ASDs (van Harssel et al., 2013).
6.3. CNS lesions, neuropathic pain, astrogliosis, and remyelination
Neuropathic pain is defined as abnormal pain sensations resulting from injuries to the peripheral or segmental spinal nerves, in part, stemming from the reorganization of microcircuits in the dorsal horn and dorsal root ganglia (Finnerup, Sindrup, & Jensen, 2007). In a mouse model of neuropathic pain, spinal nerve ligation and transection produce a persistent and abnormal responsiveness to nonnoxious mechanical stimuli that are otherwise normally tolerated and result in loss of E-cadherin from synapses formed by small-diameter primary afferents to spinal lamina II as well as a transient elevation of N-cadherin throughout lamina I and II (Brock et al., 2004; Patil et al., 2009). These changes together may mediate synaptic loss and reactive axon sprouting at the site of injury, or represent astrogliosis at the lesion site, leading to abnormal transmission of pain sensation.
N-cadherin expression is also upregulated following neocortical stab wound injury, which induces astrogliosis at the lesion site. In an inducible astrocyte-specific N-cadherin KO, there were fewer reactive astrocytes, but increased reactive microglia and neuronal cell death. These data indicate that N-cadherin is required for neuroprotection following brain injury through the maintenance of reactive astrogliosis (Kanemaru et al., 2013). Enhanced N-cadherin immunoreactivity has also been observed in early remyelinating lesions in mouse models of multiple sclerosis, with the highest expression in oligodendrocytes at lesion sites. N-Cadherin may confer this effect through homophilic adhesion between oligodendrocytes or between axons and oli-godendrocytes (Hochmeister et al., 2012).
6.4. Alzheimer’s disease
Although current evidence is limited, several studies have implicated dysfunctional N-cadherin/β-catenin signaling in Alzheimer’s disease (AD). AD is characterized pathologically by the formation of extracellular β-amyloid (Aβ)-containing plaques, intracellular neurofibrillary tangles consisting of hyperphosphorylated tau, and subsequent synaptic degeneration. Compared to age-matched controls, human AD brains exhibit reduced N-cadherin protein levels and increased p38 MAPK signaling (Ando et al., 2011). Extracellular Aβ oligomers promote the marked loss of synapses (Shankar et al., 2007) and lead to reduced N-cadherin expression and concomitant activation of p38 MAPK (Ando et al., 2011). Recent studies suggest that a γ-secretase-dependent, proteolytically derived C-terminal fragment of N-cadherin accelerates synaptic dysfunction by β-amyloid (Andreyeva et al., 2012).
In contrast, other studies report impaired N-cadherin metabolism that may be associated with AD. N-Cadherin undergoes sequential cleavage by the extracellular protease ADAM10 (A Disintegrin And Metalloprotease-10) following NMDA receptor stimulation (Reiss et al., 2005), and presenilin 1 (PS1), a γ-secretase complex component (Marambaud et al., 2003), to mediate cell–cell adhesion and downstream gene expression via β-catenin. In CHO cells, N-cadherin cleavage by ADAM10 is impaired by extracellular Aβ. Familial AD-linked mutations of PS1 (which also cleaves amyloid precursor protein to generate Aβ peptides) impede cleavage of N-cadherin (Uemura, Kuzuya, et al., 2007). To date, no other cadherins have been associated with AD. Although SNPs in Pcdh-11x were initially linked to AD susceptibility (Carrasquillo et al., 2009), recent studies have failed to replicate the results (Miar et al., 2011; Wu et al., 2010).
7. CONCLUSIONS
Cadherin-based adhesion and signaling regulate the development of neurons and their intricate, highly specific connections within the CNS through varied types of interactions and defined signaling cascades. With the development of genome-wide microarray technologies, the number of cadherins and associated proteins linked to neurodevelopmental disorders is growing, as multiple genetic variants in both coding and proximal nongenic regions are being identified, but we are only beginning to understand exactly how these variants affect synaptic function and contribute to disease pathophysiology. The emerging view that cadherins play an indispensable role in plasticity of mature synapses in hippocampus and elsewhere, and thus significantly influence cognitive abilities, suggests that cadherin/ catenin dysfunction later in life can have profound effects on memory and other aspects of higher-order brain function and support recent reports of dysfunctional N-cadherin signaling in AD. It is also critical to bear in mind that CAMs from different families are all part of a highly integrated network of synaptic proteins, probably rarely, if ever, operating in isolation of each other and other effectors. For example, the postnatal conditional deletion of synaptic β1-integrin leads to compensatory changes in both N-cadherin and neuroligins, but does not affect synaptic levels of SynCAM (Mortillo et al., 2012). The significance of this for human health is that the variable and complex phenotypes of certain disorders (e.g., autism) that are attributable to a particular genetic variant in one particular CAM (e.g., neu-roligin) may in fact reflect a number of compensatory readjustments in synaptic adhesion and signaling molecules that extend across multiple CAM families. Future studies should focus specifically on how different CAM family members influence the total repertoire of synaptic CAMs in order to uncover how these multifaceted proteins collaborate to mediate complex behaviors and pathological dysfunction in vertebrates.
Acknowledgments
We thank members of the Huntley and Benson laboratories for their contributions to the personal work discussed in this review. During the writing of this chapter, we were supported by grants from the National Institute of Mental Health (MH100600, MH095229, and MH099778); the Michael J. Fox Foundation; and an NIMH T32 Training Program in Mental Health grant (T32 MH096678).
References
- Abe K, Chisaka O, Van Roy F, Takeichi M. Stability of dendritic spines and synaptic contacts is controlled by alpha N-catenin. Nature Neuroscience. 2004;7:357–363. doi: 10.1038/nn1212. [DOI] [PubMed] [Google Scholar]
- Abedin M, King N. The premetazoan ancestry of cadherins. Science. 2008;319:946–948. doi: 10.1126/science.1151084. [DOI] [PubMed] [Google Scholar]
- Aiga M, Levinson JN, Bamji SX. N-cadherin and neuroligins cooperate to regulate synapse formation in hippocampal cultures. The Journal of Biological Chemistry. 2011;286:851–858. doi: 10.1074/jbc.M110.176305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Amoudi A, Diez DC, Betts MJ, Frangakis AS. The molecular architecture of cadherins in native epidermal desmosomes. Nature. 2007;450:832–837. doi: 10.1038/nature05994. [DOI] [PubMed] [Google Scholar]
- Anderson TR, Benson DL. Cadherin-mediated adhesion and signaling during vertebrate central synapse formation. In: Dityatev A, El-Husseini A, editors. Molecular mechanisms of synaptogenesis. New York, NY: Springer; 2006. pp. 83–93. [Google Scholar]
- Ando K, Uemura K, Kuzuya A, Maesako M, Asada-Utsugi M, Kubota M, et al. N-cadherin regulates p38 MAPK signaling via association with JNK-associated leucine zipper protein: Implications for neurodegeneration in Alzheimer disease. The Journal of Biological Chemistry. 2011;286:7619–7628. doi: 10.1074/jbc.M110.158477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreyeva A, Nieweg K, Horstmann K, Klapper S, Müller-Schiffmann A, Korth C, et al. C-terminal fragment of N-cadherin accelerates synapse destabilization by amyloid-β. Brain. 2012;135:2140–2154. doi: 10.1093/brain/aws120. [DOI] [PubMed] [Google Scholar]
- Arikkath J, Israely I, Tao Y, Mei L, Liu X, Reichardt LF. Erbin controls dendritic morphogenesis by regulating localization of delta-catenin. The Journal of Neuroscience. 2008;28:7047–7056. doi: 10.1523/JNEUROSCI.0451-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arikkath J, Peng IF, Ng YG, Israely I, Liu X, Ullian EM, et al. Delta-catenin regulates spine and synapse morphogenesis and function in hippocampal neurons during development. The Journal of Neuroscience. 2009;29:5435–5442. doi: 10.1523/JNEUROSCI.0835-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arikkath J, Reichardt LF. Cadherins and catenins at synapses: Roles in syn-aptogenesis and synaptic plasticity. Trends in Neurosciences. 2008;31:487–494. doi: 10.1016/j.tins.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arregui C, Pathre P, Lilien J, Balsamo J. The nonreceptor tyrosine kinase fer mediates cross-talk between N-cadherin and beta1-integrins. The Journal of Cell Biology. 2000;149:1263–1274. doi: 10.1083/jcb.149.6.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babb SG, Kotradi SM, Shah B, Chiappini-Williamson C, Bell LN, Schmeiser G, et al. Zebrafish R-cadherin (Cdh4) controls visual system development and differentiation. Developmental Dynamics. 2005;233:930–945. doi: 10.1002/dvdy.20431. [DOI] [PubMed] [Google Scholar]
- Bamji SX, Shimazu K, Kimes N, Huelsken J, Birchmeier W, Lu B, et al. Role of beta-catenin in synaptic vesicle localization and presynaptic assembly. Neuron. 2003;40:719–731. doi: 10.1016/s0896-6273(03)00718-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes SH, Price SR, Wentzel C, Guthrie SC. Cadherin-7 and cadherin-6B differentially regulate the growth, branching and guidance of cranial motor axons. Development. 2010;137:805–814. doi: 10.1242/dev.042457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beattie EC, Carroll RC, Yu X, Morishita W, Yasuda H, von Zastrow M, et al. Regulation of AMPA receptor endocytosis by a signaling mechanism shared with LTD. Nature Neuroscience. 2000;3:1291–1300. doi: 10.1038/81823. [DOI] [PubMed] [Google Scholar]
- Beaudoin GM, 3rd, Schofield CM, Nuwal T, Zang K, Ullian EM, Huang B, et al. Afadin, a Ras/Rap effector that controls cadherin function, promotes spine and excitatory synapse density in the hippocampus. The Journal of Neuroscience. 2012;32:99–110. doi: 10.1523/JNEUROSCI.4565-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bekirov IH, Nagy V, Svoronos A, Huntley GW, Benson DL. Cadherin-8 and N-cadherin differentially regulate pre- and postsynaptic development of the hippocampal mossy fiber pathway. Hippocampus. 2008;18:349–363. doi: 10.1002/hipo.20395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bekirov IH, Needleman LA, Zhang W, Benson DL. Identification and localization of multiple classic cadherins in developing rat limbic system. Neuroscience. 2002;115:213–227. doi: 10.1016/s0306-4522(02)00375-5. [DOI] [PubMed] [Google Scholar]
- Ben-Ari Y, Cossart R. Kainate, a double agent that generates seizures: Two decades of progress. Trends in Neurosciences. 2000;23:580–587. doi: 10.1016/s0166-2236(00)01659-3. [DOI] [PubMed] [Google Scholar]
- Benson DL, Colman DR, Huntley GW. Molecules, maps and synapse specificity. Nature Reviews. Neuroscience. 2001;2:899–909. doi: 10.1038/35104078. [DOI] [PubMed] [Google Scholar]
- Benson DL, Huntley GW. Building and remodeling synapses. Hippocampus. 2012;22:954–968. doi: 10.1002/hipo.20872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benson DL, Tanaka H. N-cadherin redistribution during synaptogenesis in hippocampal neurons. The Journal of Neuroscience. 1998;18:6892–6904. doi: 10.1523/JNEUROSCI.18-17-06892.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger-Muller S, Suzuki T. Seven-pass transmembrane cadherins: Roles and emerging mechanisms in axonal and dendritic patterning. Molecular Neurobiology. 2011;44:313–320. doi: 10.1007/s12035-011-8201-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhalla K, Luo Y, Buchan T, Beachem MA, Guzauskas GF, Ladd S, et al. Alterations in CDH15 and KIRREL3 in patients with mild to severe intellectual disability. American Journal of Human Genetics. 2008;83:703–713. doi: 10.1016/j.ajhg.2008.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biswas S, Emond MR, Jontes JD. Protocadherin-19 and N-cadherin interact to control cell movements during anterior neurulation. The Journal of Cell Biology. 2010;191:1029–1041. doi: 10.1083/jcb.201007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bixby JL, Zhang R. Purified N-cadherin is a potent substrate for the rapid induction of neurite outgrowth. The Journal of Cell Biology. 1990;110:1253–1260. doi: 10.1083/jcb.110.4.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaschuk OW, Sullivan R, David S, Pouliot Y. Identification of a cadherin cell adhesion recognition sequence. Developmental Biology. 1990;139:227–229. doi: 10.1016/0012-1606(90)90290-y. [DOI] [PubMed] [Google Scholar]
- Boggon TJ, Murray J, Chappuis-Flament S, Wong E, Gumbiner BM, Shapiro L. C-cadherin ectodomain structure and implications for cell adhesion mechanisms. Science. 2002;296:1308–1313. doi: 10.1126/science.1071559. [DOI] [PubMed] [Google Scholar]
- Borglum AD, Demontis D, Grove J, Pallesen J, Hollegaard MV, Pedersen CB, et al. Genome-wide study of association and interaction with maternal cytomeg-alovirus infection suggests new schizophrenia loci. Molecular Psychiatry. 2014;19:325–333. doi: 10.1038/mp.2013.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boscher C, Mege RM. Cadherin-11 interacts with the FGF receptor and induces neurite outgrowth through associated downstream signalling. Cellular Signalling. 2008;20:1061–1072. doi: 10.1016/j.cellsig.2008.01.008. [DOI] [PubMed] [Google Scholar]
- Boutin C, Goffinet AM, Tissir F. Celsr1-3 cadherins in PCP and brain development. Current Topics in Developmental Biology. 2012;101:161–183. doi: 10.1016/B978-0-12-394592-1.00010-7. [DOI] [PubMed] [Google Scholar]
- Bozdagi O, Shan W, Tanaka H, Benson DL, Huntley GW. Increasing numbers of synaptic puncta during late-phase LTP: N-cadherin is synthesized, recruited to synaptic sites, and required for potentiation. Neuron. 2000;28:245–259. doi: 10.1016/s0896-6273(00)00100-8. [DOI] [PubMed] [Google Scholar]
- Bozdagi O, Valcin M, Poskanzer K, Tanaka H, Benson DL. Temporally distinct demands for classic cadherins in synapse formation and maturation. Molecular and Cellular Neurosciences. 2004;27:509–521. doi: 10.1016/j.mcn.2004.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bozdagi O, Wang XB, Nikitczuk JS, Anderson TR, Bloss EB, Radice GL, et al. Persistence of coordinated long-term potentiation and dendritic spine enlargement at mature hippocampal CA1 synapses requires N-cadherin. The Journal of Neuroscience. 2010;30:9984–9989. doi: 10.1523/JNEUROSCI.1223-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradl M, Lassmann H. Oligodendrocytes: Biology and pathology. Acta Neu-ropathologica. 2010;119:37–53. doi: 10.1007/s00401-009-0601-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brigidi GS, Bamji SX. Cadherin-catenin adhesion complexes at the synapse. Current Opinion in Neurobiology. 2011;21:208–214. doi: 10.1016/j.conb.2010.12.004. [DOI] [PubMed] [Google Scholar]
- Brigidi GS, Sun Y, Beccano-Kelly D, Pitman K, Mobasser M, Borgland SL, et al. Palmitoylation of delta-catenin by DHHC5 mediates activity-induced synapse plasticity. Nature Neuroscience. 2014;17:522–532. doi: 10.1038/nn.3657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brock JH, Elste A, Huntley GW. Distribution and injury-induced plasticity of cadherins in relationship to identified synaptic circuitry in adult rat spinal cord. The Journal of Neuroscience. 2004;24:8806–8817. doi: 10.1523/JNEUROSCI.2726-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruses JL. N-cadherin regulates primary motor axon growth and branching during zebrafish embryonic development. The Journal of Comparative Neurology. 2011;519:1797–1815. doi: 10.1002/cne.22602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruses JL. Cell surface localization of alpha3beta4 nicotinic acetylcholine receptors is regulated by N-cadherin homotypic binding and actomyosin contractility. PLoS One. 2013;8:e62435. doi: 10.1371/journal.pone.0062435. http://dx.doi.org/10.1371/journal.pone.0062435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchert M, Schneider S, Meskenaite V, Adams MT, Canaani E, Baechi T, et al. The junction-associated protein AF-6 interacts and clusters with specific Eph receptor tyrosine kinases at specialized sites of cell-cell contact in the brain. The Journal of Cell Biology. 1999;144:361–371. doi: 10.1083/jcb.144.2.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burden-Gulley SM, Brady-Kalnay SM. PTPmu regulates N-cadherin-dependent neurite outgrowth. The Journal of Cell Biology. 1999;144:1323–1336. doi: 10.1083/jcb.144.6.1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrasquillo MM, Zou F, Pankratz VS, Wilcox SL, Ma L, Walker LP, et al. Genetic variation in PCDH11X is associated with susceptibility to late-onset Alzheimer’s disease. Nature Genetics. 2009;41:192–198. doi: 10.1038/ng.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Gumbiner BM. Paraxial protocadherin mediates cell sorting and tissue morphogenesis by regulating C-cadherin adhesion activity. The Journal of Cell Biology. 2006;174:301–313. doi: 10.1083/jcb.200602062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Koh E, Yoder M, Gumbiner BM. A protocadherin-cadherin-FLRT3 complex controls cell adhesion and morphogenesis. PLoS One. 2009;4:e8411. doi: 10.1371/journal.pone.0008411. http://dx.doi.org/10.1371/journal.pone.0008411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciatto C, Bahna F, Zampieri N, VanSteenhouse HC, Katsamba PS, Ahlsen G, et al. T-cadherin structures reveal a novel adhesive binding mechanism. Nature Structural & Molecular Biology. 2010;17:339–347. doi: 10.1038/nsmb.1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collingridge GL, Bliss TVP. NMDA receptors-their role in long-term potentiation. Trends in Neurosciences. 1987;10:288–293. [Google Scholar]
- Coussen F, Normand E, Marchal C, Costet P, Choquet D, Lambert M, et al. Recruitment of the kainate receptor subunit glutamate receptor 6 by cadherin/catenin complexes. The Journal of Neuroscience. 2002;22:6426–6436. doi: 10.1523/JNEUROSCI.22-15-06426.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalva MB, McClelland AC, Kayser MS. Cell adhesion molecules: Signalling functions at the synapse. Nature Reviews Neuroscience. 2007;8:206–220. doi: 10.1038/nrn2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danielson E, Zhang N, Metallo J, Kaleka K, Shin SM, Gerges N, et al. S-SCAM/MAGI-2 is an essential synaptic scaffolding molecule for the GluA2-containing maintenance pool of AMPA receptors. The Journal of Neuroscience. 2012;32:6967–6980. doi: 10.1523/JNEUROSCI.0025-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dell’Acqua ML, Smith KE, Gorski JA, Horne EA, Gibson ES, Gomez LL. Regulation of neuronal PKA signaling through AKAP targeting dynamics. European Journal of Cell Biology. 2006;85:627–633. doi: 10.1016/j.ejcb.2006.01.010. [DOI] [PubMed] [Google Scholar]
- Depienne C, LeGuern E. PCDH19-related infantile epileptic encephalopathy: An unusual X-linked inheritance disorder. Human Mutation. 2012;33:627–634. doi: 10.1002/humu.22029. [DOI] [PubMed] [Google Scholar]
- Depienne C, Trouillard O, Bouteiller D, Gourfinkel-An I, Poirier K, Rivier F, et al. Mutations and deletions in PCDH19 account for various familial or isolated epilepsies in females. Human Mutation. 2011;32:E1959–E1975. doi: 10.1002/humu.21373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dibbens LM, Tarpey PS, Hynes K, Bayly MA, Scheffer IE, Smith R, et al. X-linked protocadherin 19 mutations cause female-limited epilepsy and cognitive impairment. Nature Genetics. 2008;40:776–781. doi: 10.1038/ng.149. http://dx.doi.org/10.1002/humu.21373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doherty P, Rowett LH, Moore SE, Mann DA, Walsh FS. Neurite outgrowth in response to transfected N-CAM and N-cadherin reveals fundamental differences in neuronal responsiveness to CAMs. Neuron. 1991;6:247–258. doi: 10.1016/0896-6273(91)90360-c. [DOI] [PubMed] [Google Scholar]
- Doherty P, Skaper SD, Moore SE, Leon A, Walsh FS. A developmentally regulated switch in neuronal responsiveness to NCAM and N-cadherin in the rat hippocampus. Development. 1992;115:885–892. doi: 10.1242/dev.115.3.885. [DOI] [PubMed] [Google Scholar]
- Dong Z, Bai Y, Wu X, Li H, Gong B, Howland JG, et al. Hippocampal long-term depression mediates spatial reversal learning in the Morris water maze. Neuropharmacology. 2013;64:65–73. doi: 10.1016/j.neuropharm.2012.06.027. [DOI] [PubMed] [Google Scholar]
- Drazba J, Lemmon V. The role of cell adhesion molecules in neurite outgrowth on Muller cells. Developmental Biology. 1990;138:82–93. doi: 10.1016/0012-1606(90)90178-l. [DOI] [PubMed] [Google Scholar]
- Ehlers MD. Reinsertion or degradation of AMPA receptors determined by activity-dependent endocytic sorting. Neuron. 2000;28:511–525. doi: 10.1016/s0896-6273(00)00129-x. [DOI] [PubMed] [Google Scholar]
- Ehlers MD, Heine M, Groc L, Lee MC, Choquet D. Diffusional trapping of GluR1 AMPA receptors by input-specific synaptic activity. Neuron. 2007;54:447–460. doi: 10.1016/j.neuron.2007.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrlich I, Malinow R. Postsynaptic density 95 controls AMPA receptor incorporation during long-term potentiation and experience-driven synaptic plasticity. The Journal of Neuroscience. 2004;24:916–927. doi: 10.1523/JNEUROSCI.4733-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elste AM, Benson DL. Structural basis for developmentally regulated changes in cadherin function at synapses. The Journal of Comparative Neurology. 2006;495:324–335. doi: 10.1002/cne.20876. [DOI] [PubMed] [Google Scholar]
- Emond MR, Biswas S, Blevins CJ, Jontes JD. A complex of Protocadherin-19 and N-cadherin mediates a novel mechanism of cell adhesion. The Journal of Cell Biology. 2011;195:1115–1121. doi: 10.1083/jcb.201108115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esch T, Lemmon V, Banker G. Differential effects of NgCAM and N-cadherin on the development of axons and dendrites by cultured hippocampal neurons. Journal of Neurocytology. 2000;29:215–223. doi: 10.1023/a:1026515426303. [DOI] [PubMed] [Google Scholar]
- Fernandez-Monreal M, Kang S, Phillips GR. Gamma-protocadherin homo-philic interaction and intracellular trafficking is controlled by the cytoplasmic domain in neurons. Molecular and Cellular Neurosciences. 2009;40:344–353. doi: 10.1016/j.mcn.2008.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiederling A, Ewert R, Andreyeva A, Jungling K, Gottmann K. E-cadherin is required at GABAergic synapses in cultured cortical neurons. Neuroscience Letters. 2011;501:167–172. doi: 10.1016/j.neulet.2011.07.009. [DOI] [PubMed] [Google Scholar]
- Finnerup NB, Sindrup SH, Jensen TS. Chronic neuropathic pain: Mechanisms, drug targets and measurement. Fundamental & Clinical Pharmacology. 2007;21:129–136. doi: 10.1111/j.1472-8206.2007.00474.x. [DOI] [PubMed] [Google Scholar]
- Flannery RJ, Bruses JL. N-cadherin induces partial differentiation of cholinergic presynaptic terminals in heterologous cultures of brainstem neurons and CHO cells. Frontiers in Synaptic Neuroscience. 2012;4:1–11. doi: 10.3389/fnsyn.2012.00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fredette BJ, Miller J, Ranscht B. Inhibition of motor axon growth by T-cadherin substrata. Development. 1996;122:3163–3171. doi: 10.1242/dev.122.10.3163. [DOI] [PubMed] [Google Scholar]
- Friedman LG, Riemslagh FG, Sullivan JM, Mesias R, Williams FM, Huntley GW, et al. Cadherin-8 expression, synaptic localization and molecular control of neuronal form in prefrontal cortico-striatal circuits. The Journal of Comparative Neurology. 2014;523:75–92. doi: 10.1002/cne.23666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frotscher M, Heimrich B. Lamina-specific synaptic connections of hippocampal neurons in vitro. Journal of Neurobiology. 1995;26:350–359. doi: 10.1002/neu.480260307. [DOI] [PubMed] [Google Scholar]
- Garrett AM, Schreiner D, Lobas MA, Weiner JA. Gamma-protocadherins control cortical dendrite arborization by regulating the activity of a FAK/PKC/MAR-CKS signaling pathway. Neuron. 2012;74:269–276. doi: 10.1016/j.neuron.2012.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrett AM, Weiner JA. Control of CNS synapse development by {gamma}-protocadherin-mediated astrocyte-neuron contact. The Journal of Neuroscience. 2009;29:11723–11731. doi: 10.1523/JNEUROSCI.2818-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geurts HM, Grasman RP, Verte S, Oosterlaan J, Roeyers H, van Kammen SM, et al. Intra-individual variability in ADHD, autism spectrum disorders and Tour-ette’s syndrome. Neuropsychologia. 2008;46:3030–3041. doi: 10.1016/j.neuropsychologia.2008.06.013. [DOI] [PubMed] [Google Scholar]
- Gil OD, Needleman L, Huntley GW. Developmental patterns of cadherin expression and localization in relation to compartmentalized thalamocortical terminations in rat barrel cortex. The Journal of Comparative Neurology. 2002;453:372–388. doi: 10.1002/cne.10424. [DOI] [PubMed] [Google Scholar]
- Gorski JA, Gomez LL, Scott JD, Dell’Acqua ML. Association of an A-kinase-anchoring protein signaling scaffold with cadherin adhesion molecules in neurons and epithelial cells. Molecular Biology of the Cell. 2005;16:3574–3590. doi: 10.1091/mbc.E05-02-0134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf ER, Zhang X, Jin SX, Linhoff MW, Craig AM. Neurexins induce differentiation of GABA and glutamate postsynaptic specializations via neuroligins. Cell. 2004;119:1013–1026. doi: 10.1016/j.cell.2004.11.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grueber WB, Sagasti A. Self-avoidance and tiling: Mechanisms of dendrite and axon spacing. Cold Spring Harbor Perspectives in Biology. 2010;2:a001750. doi: 10.1101/cshperspect.a001750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han MH, Lin C, Meng S, Wang X. Proteomics analysis reveals overlapping functions of clustered protocadherins. Molecular & Cellular Proteomics. 2010;9:71–83. doi: 10.1074/mcp.M900343-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison OJ, Bahna F, Katsamba PS, Jin X, Brasch J, Vendome J, et al. Two-step adhesive binding by classical cadherins. Nature Structural & Molecular Biology. 2010;17:348–357. doi: 10.1038/nsmb.1784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hart AB, Engelhardt BE, Wardle MC, Sokoloff G, Stephens M, de Wit H, et al. Genome-wide association study of D-amphetamine response in healthy volunteers identifies putative associations, including cadherin 13 (CDH13) PLoS One. 2012;7:e42646. doi: 10.1371/journal.pone.0042646. http://dx.doi.org/10.1371/journal.pone.0042646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa S, Hamada S, Kumode Y, Esumi S, Katori S, Fukuda E, et al. The protocadherin-alpha family is involved in axonal coalescence of olfactory sensory neurons into glomeruli of the olfactory bulb in mouse. Molecular and Cellular Neurosciences. 2008;38:66–79. doi: 10.1016/j.mcn.2008.01.016. [DOI] [PubMed] [Google Scholar]
- Heisler FF, Lee HK, Gromova KV, Pechmann Y, Schurek B, Ruschkies L, et al. GRIP1 interlinks N-cadherin and AMPA receptors at vesicles to promote combined cargo transport into dendrites. Proceedings of the National Academy of Sciences of the United States of America. 2014;111:5030–5035. doi: 10.1073/pnas.1304301111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hertel N, Redies C. Absence of layer-specific cadherin expression profiles in the neocortex of the reeler mutant mouse. Cerebral Cortex. 2011;21:1105–1117. doi: 10.1093/cercor/bhq183. [DOI] [PubMed] [Google Scholar]
- Higurashi N, Shi X, Yasumoto S, Oguni H, Sakauchi M, Itomi K, et al. PCDH19 mutation in Japanese females with epilepsy. Epilepsy Research. 2012;99:28–37. doi: 10.1016/j.eplepsyres.2011.10.014. [DOI] [PubMed] [Google Scholar]
- Hirano K, Kaneko R, Izawa T, Kawaguchi M, Kitsukawa T, Yagi T. Single-neuron diversity generated by Protocadherin-beta cluster in mouse central and peripheral nervous systems. Frontiers in Molecular Neuroscience. 2012;5:1–13. doi: 10.3389/fnmol.2012.00090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirano S, Takeichi M. Cadherins in brain morphogenesis and wiring. Physiological Reviews. 2012;92:597–634. doi: 10.1152/physrev.00014.2011. [DOI] [PubMed] [Google Scholar]
- Hochmeister S, Romauch M, Bauer J, Seifert-Held T, Weissert R, Linington C, et al. Re-expression of N-cadherin in remyelinating lesions of experimental inflammatory demyelination. Experimental Neurology. 2012;237:70–77. doi: 10.1016/j.expneurol.2012.06.010. [DOI] [PubMed] [Google Scholar]
- Hong S, Troyanovsky RB, Troyanovsky SM. Cadherin exits the junction by switching its adhesive bond. The Journal of Cell Biology. 2011;192:1073–1083. doi: 10.1083/jcb.201006113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoshina N, Tanimura A, Yamasaki M, Inoue T, Fukabori R, Kuroda T, et al. Protocadherin 17 regulates presynaptic assembly in topographic corticobasal Ganglia circuits. Neuron. 2013;78:839–854. doi: 10.1016/j.neuron.2013.03.031. [DOI] [PubMed] [Google Scholar]
- Huber AH, Weis WI. The structure of the beta-catenin/E-cadherin complex and the molecular basis of diverse ligand recognition by beta-catenin. Cell. 2001;105:391–402. doi: 10.1016/s0092-8674(01)00330-0. [DOI] [PubMed] [Google Scholar]
- Hulpiau P, Gul IS, van Roy F. New insights into the evolution of metazoan cadherins and catenins. Progress in Molecular Biology and Translational Science. 2013;116:71–94. doi: 10.1016/B978-0-12-394311-8.00004-2. [DOI] [PubMed] [Google Scholar]
- Hulpiau P, van Roy F. Molecular evolution of the cadherin superfamily. The International Journal of Biochemistry & Cell Biology. 2009;41:349–369. doi: 10.1016/j.biocel.2008.09.027. [DOI] [PubMed] [Google Scholar]
- Huntley GW, Benson DL. Neural (N)-cadherin at developing thalamocortical synapses provides an adhesion mechanism for the formation of somatopically organized connections. The Journal of Comparative Neurology. 1999;407:453–471. [PubMed] [Google Scholar]
- Huntley GW, Elste AM, Patil SB, Bozdagi O, Benson DL, Steward O. Synaptic loss and retention of different classic cadherins with LTP-associated synaptic structural remodeling in vivo. Hippocampus. 2012;22:17–28. doi: 10.1002/hipo.20859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huttelmaier S, Zenklusen D, Lederer M, Dictenberg J, Lorenz M, Meng X, et al. Spatial regulation of beta-actin translation by Src-dependent phosphorylation of ZBP1. Nature. 2005;438:512–515. doi: 10.1038/nature04115. [DOI] [PubMed] [Google Scholar]
- Inoue A, Sanes JR. Lamina-specific connectivity in the brain: Regulation by N-cadherin, neurotrophins, and glycoconjugates. Science. 1997;276:1428–1431. doi: 10.1126/science.276.5317.1428. [DOI] [PubMed] [Google Scholar]
- Ishiuchi T, Misaki K, Yonemura S, Takeichi M, Tanoue T. Mammalian Fat and dachsous cadherins regulate apical membrane organization in the embryonic cerebral cortex. The Journal of Cell Biology. 2009;185:959–967. doi: 10.1083/jcb.200811030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Israely I, Costa RM, Xie CW, Silva AJ, Kosik KS, Liu X. Deletion of the neuron-specific protein delta-catenin leads to severe cognitive and synaptic dysfunction. Current Biology. 2004;14:1657–1663. doi: 10.1016/j.cub.2004.08.065. [DOI] [PubMed] [Google Scholar]
- Jamal SM, Basran RK, Newton S, Wang Z, Milunsky JM. Novel de novo PCDH19 mutations in three unrelated females with epilepsy female restricted mental retardation syndrome. American Journal of Medical Genetics. Part A. 2010;152A:2475–2481. doi: 10.1002/ajmg.a.33611. [DOI] [PubMed] [Google Scholar]
- Jontes JD, Emond MR, Smith SJ. In vivo trafficking and targeting of N-cadherin to nascent presynaptic terminals. The Journal of Neuroscience. 2004;24:9027–9034. doi: 10.1523/JNEUROSCI.5399-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jungling K, Eulenburg V, Moore R, Kemler R, Lessmann V, Gottmann K. N-cadherin transsynaptically regulates short-term plasticity at glutamatergic synapses in embryonic stem cell-derived neurons. The Journal of Neuroscience. 2006;26:6968–6978. doi: 10.1523/JNEUROSCI.1013-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadowaki M, Nakamura S, Machon O, Krauss S, Radice GL, Takeichi M. N-Cadherin mediates cortical organization in the mouse brain. Developmental Biology. 2007;304:22–33. doi: 10.1016/j.ydbio.2006.12.014. [DOI] [PubMed] [Google Scholar]
- Kanemaru K, Kubota J, Sekiya H, Hirose K, Okubo Y, Iino M. Calcium-dependent N-cadherin up-regulation mediates reactive astrogliosis and neuroprotection after brain injury. Proceedings of the National Academy of Sciences of the United States of Amer-ica. 2013;110:11612–11617. doi: 10.1073/pnas.1300378110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang JS, Feinleib JL, Knox S, Ketteringham MA, Krauss RS. Promyogenic members of the Ig and cadherin families associate to positively regulate differentiation. Proceedings of the National Academy of Sciences of the United States of America. 2003;100:3989–3994. doi: 10.1073/pnas.0736565100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karpowicz P, Willaime-Morawek S, Balenci L, DeVeale B, Inoue T, van der Kooy D. E-Cadherin regulates neural stem cell self-renewal. The Journal of Neuroscience. 2009;29:3885–3896. doi: 10.1523/JNEUROSCI.0037-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katori S, Hamada S, Noguchi Y, Fukuda E, Yamamoto T, Yamamoto H, et al. Protocadherin-alpha family is required for serotonergic projections to appropriately innervate target brain areas. The Journal of Neuroscience. 2009;29:9137–9147. doi: 10.1523/JNEUROSCI.5478-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawauchi T, Sekine K, Shikanai M, Chihama K, Tomita K, Kubo K, et al. Rab GTPases-dependent endocytic pathways regulate neuronal migration and maturation through N-cadherin trafficking. Neuron. 2010;67:588–602. doi: 10.1016/j.neuron.2010.07.007. [DOI] [PubMed] [Google Scholar]
- Kim H, Han JR, Park J, Oh M, James SE, Chang S, et al. Delta-catenin-induced dendritic morphogenesis. An essential role of p190RhoGEF interaction through Akt1-mediated phosphorylation. The Journal of Biological Chemistry. 2008;283:977–987. doi: 10.1074/jbc.M707158200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kintner C. Regulation of embryonic cell adhesion by the cadherin cytoplasmic domain. Cell. 1992;69:225–236. doi: 10.1016/0092-8674(92)90404-z. [DOI] [PubMed] [Google Scholar]
- Kopec CD, Li B, Wei W, Boehm J, Malinow R. Glutamate receptor exo-cytosis and spine enlargement during chemically induced long-term potentiation. The Journal of Neuroscience. 2006;26:2000–2009. doi: 10.1523/JNEUROSCI.3918-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopec CD, Real E, Kessels HW, Malinow R. GluR1 links structural and functional plasticity at excitatory synapses. The Journal of Neuroscience. 2007;27:13706–13718. doi: 10.1523/JNEUROSCI.3503-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosik KS, Donahue CP, Israely I, Liu X, Ochiishi T. Delta-catenin at the synaptic-adherens junction. Trends in Cell Biology. 2005;15:172–178. doi: 10.1016/j.tcb.2005.01.004. [DOI] [PubMed] [Google Scholar]
- Lee CH, Herman T, Clandinin TR, Lee R, Zipursky SL. N-Cadherin regulates target specificity in the Drosophila visual system. Neuron. 2001;30:437–450. doi: 10.1016/s0896-6273(01)00291-4. [DOI] [PubMed] [Google Scholar]
- Lefebvre JL, Kostadinov D, Chen WV, Maniatis T, Sanes JR. Protocadherins mediate dendritic self-avoidance in the mammalian nervous system. Nature. 2012;488:517–521. doi: 10.1038/nature11305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levinson JN, Chery N, Huang K, Wong TP, Gerrow K, Kang R, et al. Neuroligins mediate excitatory and inhibitory synapse formation: Involvement of PSD-95 and neurexin-1beta in neuroligin-induced synaptic specificity. The Journal of Biological Chemistry. 2005;280:17312–17319. doi: 10.1074/jbc.M413812200. [DOI] [PubMed] [Google Scholar]
- Levinson JN, Li R, Kang R, Moukhles H, El-Husseini A, Bamji SX. Postsynaptic scaffolding molecules modulate the localization of neuroligins. Neuroscience. 2010;165:782–793. doi: 10.1016/j.neuroscience.2009.11.016. [DOI] [PubMed] [Google Scholar]
- Li Y, Xiao H, Chiou TT, Jin H, Bonhomme B, Miralles CP, et al. Molecular and functional interaction between protocadherin-gammaC5 and GABAA receptors. The Journal of Neuroscience. 2012;32:11780–11797. doi: 10.1523/JNEUROSCI.0969-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J, Luo J, Redies C. Cadherin-19 expression is restricted to myelin-forming cells in the chicken embryo. Neuroscience. 2010;165:168–178. doi: 10.1016/j.neuroscience.2009.10.032. [DOI] [PubMed] [Google Scholar]
- Luccardini C, Hennekinne L, Viou L, Yanagida M, Murakami F, Kessaris N, et al. N-cadherin sustains motility and polarity of future cortical interneurons during tangential migration. The Journal of Neuroscience. 2013;33:18149–18160. doi: 10.1523/JNEUROSCI.0593-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo J, Ju MJ, Redies C. Regionalized cadherin-7 expression by radial glia is regulated by Shh and Pax7 during chicken spinal cord development. Neuroscience. 2006;142:1133–1143. doi: 10.1016/j.neuroscience.2006.07.038. [DOI] [PubMed] [Google Scholar]
- Machado CB, Kanning KC, Kreis P, Stevenson D, Crossley M, Nowak M, et al. Reconstruction of phrenic neuron identity in embryonic stem cell-derived motor neurons. Development. 2014;141:784–794. doi: 10.1242/dev.097188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maguschak KA, Ressler KJ. Beta-catenin is required for memory consolidation. Nature Neuroscience. 2008;11:1319–1326. doi: 10.1038/nn.2198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mainen ZF, Jia Z, Roder J, Malinow R. Use-dependent AMPA receptor block in mice lacking GluR2 suggests postsynaptic site for LTP expression. Nature Neuroscience. 1998;1:579–586. doi: 10.1038/2812. [DOI] [PubMed] [Google Scholar]
- Malinow R, Malenka RC. AMPA receptor trafficking and synaptic plasticity. Annual Review of Neuroscience. 2002;25:103–126. doi: 10.1146/annurev.neuro.25.112701.142758. [DOI] [PubMed] [Google Scholar]
- Manabe T, Togashi H, Uchida N, Suzuki SC, Hayakawa Y, Yamamoto M, et al. Loss of cadherin-11 adhesion receptor enhances plastic changes in hippocampal synapses and modifies behavioral responses. Molecular and Cellular Neurosciences. 2000;15:534–546. doi: 10.1006/mcne.2000.0849. [DOI] [PubMed] [Google Scholar]
- Manibog K, Li H, Rakshit S, Sivasankar S. Resolving the molecular mechanism of cadherin catch bond formation. Nature Communications. 2014;5:3941. doi: 10.1038/ncomms4941. [DOI] [PubMed] [Google Scholar]
- Marambaud P, Wen PH, Dutt A, Shioi J, Takashima A, Siman R, et al. A CBP binding transcriptional repressor produced by the PS1/epsilon-cleavage of N-cadherin is inhibited by PS1 FAD mutations. Cell. 2003;114:635–645. doi: 10.1016/j.cell.2003.08.008. [DOI] [PubMed] [Google Scholar]
- Marin O, Rubenstein JL. Cell migration in the forebrain. Annual Review of Neuroscience. 2003;26:441–483. doi: 10.1146/annurev.neuro.26.041002.131058. [DOI] [PubMed] [Google Scholar]
- Marrs GS, Honda T, Fuller L, Thangavel R, Balsamo J, Lilien J, et al. Den-dritic arbors of developing retinal ganglion cells are stabilized by beta 1-integrins. Molecular and Cellular Neurosciences. 2006;32:230–241. doi: 10.1016/j.mcn.2006.04.005. [DOI] [PubMed] [Google Scholar]
- Marrs GS, Theisen CS, Bruses JL. N-cadherin modulates voltage activated calcium influx via RhoA, p120-catenin, and myosin-actin interaction. Molecular and Cellular Neurosciences. 2009;40:390–400. doi: 10.1016/j.mcn.2008.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marthiens V, Gavard J, Padilla F, Monnet C, Castellani V, Lambert M, et al. A novel function for cadherin-11 in the regulation of motor axon elongation and fasciculation. Molecular and Cellular Neurosciences. 2005;28:715–726. doi: 10.1016/j.mcn.2004.12.001. [DOI] [PubMed] [Google Scholar]
- Matson JL, Shoemaker M. Intellectual disability and its relationship to autism spectrum disorders. Research in Developmental Disabilities. 2009;30:1107–1114. doi: 10.1016/j.ridd.2009.06.003. [DOI] [PubMed] [Google Scholar]
- Matsunaga M, Hatta K, Nagafuchi A, Takeichi M. Guidance of optic nerve fibres by N-cadherin adhesion molecules. Nature. 1988;334:62–64. doi: 10.1038/334062a0. [DOI] [PubMed] [Google Scholar]
- Matter C, Pribadi M, Liu X, Trachtenberg JT. Delta-catenin is required for the maintenance of neural structure and function in mature cortex in vivo. Neuron. 2009;64:320–327. doi: 10.1016/j.neuron.2009.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medina M, Marinescu RC, Overhauser J, Kosik KS. Hemizygosity of delta-catenin (CTNND2) is associated with severe mental retardation in cri-du-chat syndrome. Genomics. 2000;63:157–164. doi: 10.1006/geno.1999.6090. [DOI] [PubMed] [Google Scholar]
- Mendez P, De Roo M, Poglia L, Klauser P, Muller D. N-Cadherin mediates plasticity-induced long-term spine stabilization. The Journal of Cell Biology. 2010;189:589–600. doi: 10.1083/jcb.201003007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miar A, Alvarez V, Corao AI, Alonso B, Diaz M, Menendez M, et al. Lack of association between protocadherin 11-X/Y (PCDH11X and PCDH11Y) polymorphisms and late onset Alzheimer’s disease. Brain Research. 2011;1383:252–256. doi: 10.1016/j.brainres.2011.01.054. [DOI] [PubMed] [Google Scholar]
- Mills F, Bartlett TE, Dissing-Olesen L, Wisniewska MB, Kuznicki J, Macvicar BA, et al. Cognitive flexibility and long-term depression (LTD) are impaired following beta-catenin stabilization in vivo. Proceedings of the National Academy of Sciences of the United States of America. 2014;111:8631–8636. doi: 10.1073/pnas.1404670111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyata T, Kawaguchi A, Okano H, Ogawa M. Asymmetric inheritance of radial glial fibers by cortical neurons. Neuron. 2001;31:727–741. doi: 10.1016/s0896-6273(01)00420-2. [DOI] [PubMed] [Google Scholar]
- Morales M, Colicos MA, Goda Y. Actin-dependent regulation of neurotrans-mitter release at central synapses. Neuron. 2000;27:539–550. doi: 10.1016/s0896-6273(00)00064-7. [DOI] [PubMed] [Google Scholar]
- Morishita H, Umitsu M, Murata Y, Shibata N, Udaka K, Higuchi Y, et al. Structure of the cadherin-related neuronal receptor/protocadherin-alpha first extracellular cadherin domain reveals diversity across cadherin families. The Journal of Biological Chemistry. 2006;281:33650–33663. doi: 10.1074/jbc.M603298200. [DOI] [PubMed] [Google Scholar]
- Morrow EM, Yoo SY, Flavell SW, Kim TK, Lin Y, Hill RS, et al. Identifying autism loci and genes by tracing recent shared ancestry. Science. 2008;321:218–223. doi: 10.1126/science.1157657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mortillo S, Elste A, Ge Y, Patil SB, Hsiao K, Huntley GW, et al. Compensatory redistribution of neuroligins and N-cadherin following deletion of synaptic beta1-integrin. The Journal of Comparative Neurology. 2012;520:2041–2052. doi: 10.1002/cne.23027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moya PR, Dodman NH, Timpano KR, Rubenstein LM, Rana Z, Fried RL, et al. Rare missense neuronal cadherin gene (CDH2) variants in specific obsessive-compulsive disorder and Tourette disorder phenotypes. European Journal of Human Genetics. 2013;21:850–854. doi: 10.1038/ejhg.2012.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murase S, Mosser E, Schuman EM. Depolarization drives beta-catenin into neuronal spines promoting changes in synaptic structure and function. Neuron. 2002;35:91–105. doi: 10.1016/s0896-6273(02)00764-x. [DOI] [PubMed] [Google Scholar]
- Murata Y, Hamada S, Morishita H, Mutoh T, Yagi T. Interaction with protocadherin-gamma regulates the cell surface expression of protocadherin-alpha. The Journal of Biological Chemistry. 2004;279:49508–49516. doi: 10.1074/jbc.M408771200. [DOI] [PubMed] [Google Scholar]
- Mutoh T, Hamada S, Senzaki K, Murata Y, Yagi T. Cadherin-related neuronal receptor 1 (CNR1) has cell adhesion activity with beta1 integrin mediated through the RGD site of CNR1. Experimental Cell Research. 2004;294:494–508. doi: 10.1016/j.yexcr.2003.11.019. [DOI] [PubMed] [Google Scholar]
- Nagar B, Overduin M, Ikura M, Rini JM. Structural basis of calcium-induced E-cadherin rigidification and dimerization. Nature. 1996;380:360–364. doi: 10.1038/380360a0. [DOI] [PubMed] [Google Scholar]
- Nagashima M, Barthel LK, Raymond PA. A self-renewing division of zebrafish Muller glial cells generates neuronal progenitors that require N-cadherin to regenerate retinal neurons. Development. 2013;140:4510–4521. doi: 10.1242/dev.090738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakao S, Platek A, Hirano S, Takeichi M. Contact-dependent promotion of cell migration by the OL-protocadherin-Nap1 interaction. The Journal of Cell Biology. 2008;182:395–410. doi: 10.1083/jcb.200802069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nern A, Zhu Y, Zipursky SL. Local N-cadherin interactions mediate distinct steps in the targeting of lamina neurons. Neuron. 2008;58:34–41. doi: 10.1016/j.neuron.2008.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neugebauer KM, Tomaselli KJ, Lilien J, Reichardt LF. N-Cadherin, NCAM, and integrins promote retinal neurite outgrowth on astrocytes in vitro. The Journal of Cell Biology. 1988;107:1177–1187. doi: 10.1083/jcb.107.3.1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikitczuk JS, Patil SB, Matikainen-Ankney BA, Scarpa J, Shapiro ML, Benson DL, et al. N-Cadherin regulates molecular organization of excitatory and inhibitory synaptic circuits in adult hippocampus in vivo. Hippocampus. 2014;24:943–962. doi: 10.1002/hipo.22282. http://dx.doi.org/10.1002/hipo.22282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nose A, Nagafuchi A, Takeichi M. Expressed recombinant cadherins mediate cell sorting in model systems. Cell. 1988;54:993–1001. doi: 10.1016/0092-8674(88)90114-6. [DOI] [PubMed] [Google Scholar]
- Nuriya M, Huganir RL. Regulation of AMPA receptor trafficking by N-cadherin. Journal of Neurochemistry. 2006;97:652–661. doi: 10.1111/j.1471-4159.2006.03740.x. [DOI] [PubMed] [Google Scholar]
- Oblander SA, Brady-Kalnay SM. Distinct PTPmu-associated signaling molecules differentially regulate neurite outgrowth on E-, N-, and R-cadherin. Molecular and Cellular Neurosciences. 2010;44:78–93. doi: 10.1016/j.mcn.2010.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamura K, Tanaka H, Yagita Y, Saeki Y, Taguchi A, Hiraoka Y, et al. Cadherin activity is required for activity-induced spine remodeling. The Journal of Cell Biology. 2004;167:961–972. doi: 10.1083/jcb.200406030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Leary R, Reilly JE, Hanson HH, Kang S, Lou N, Phillips GR. A variable cytoplasmic domain segment is necessary for gamma-protocadherin trafficking and tubulation in the endosome/lysosome pathway. Molecular Biology of the Cell. 2011;22:4362–4372. doi: 10.1091/mbc.E11-04-0283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opazo P, Choquet D. A three-step model for the synaptic recruitment of AMPA receptors. Molecular and Cellular Neurosciences. 2011;46:1–8. doi: 10.1016/j.mcn.2010.08.014. [DOI] [PubMed] [Google Scholar]
- Osterhout JA, Josten N, Yamada J, Pan F, Wu SW, Nguyen PL, et al. Cadherin-6 mediates axon-target matching in a non-image-forming visual circuit. Neuron. 2011;71:632–639. doi: 10.1016/j.neuron.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagnamenta AT, Khan H, Walker S, Gerrelli D, Wing K, Bonaglia MC, et al. Rare familial 16q21 microdeletions under a linkage peak implicate cadherin 8 (CDH8) in susceptibility to autism and learning disability. Journal of Medical Genetics. 2011;48:48–54. doi: 10.1136/jmg.2010.079426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paradis S, Harrar DB, Lin Y, Koon AC, Hauser JL, Griffith EC, et al. An RNAi-based approach identifies molecules required for glutamatergic and GABAergic synapse development. Neuron. 2007;53:217–232. doi: 10.1016/j.neuron.2006.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardo CA, Eberhart CG. The neurobiology of autism. Brain Pathology. 2007;17:434–447. doi: 10.1111/j.1750-3639.2007.00102.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park M, Penick EC, Edwards JG, Kauer JA, Ehlers MD. Recycling endosomes supply AMPA receptors for LTP. Science. 2004;305:1972–1975. doi: 10.1126/science.1102026. [DOI] [PubMed] [Google Scholar]
- Passafaro M, Piech V, Sheng M. Subunit-specific temporal and spatial patterns of AMPA receptor exocytosis in hippocampal neurons. Nature Neuroscience. 2001;4:917–926. doi: 10.1038/nn0901-917. [DOI] [PubMed] [Google Scholar]
- Patel SD, Ciatto C, Chen CP, Bahna F, Rajebhosale M, Arkus N, et al. Type II cadherin ectodomain structures: Implications for classical cadherin specificity. Cell. 2006;124:1255–1268. doi: 10.1016/j.cell.2005.12.046. [DOI] [PubMed] [Google Scholar]
- Patil S, Brock JH, Colman DR, Huntley GW. GDNF-dependent modulation of cadherins in spinal dorsal horn in relationship to neuropathic pain. Pain. 2009;152:924–935. doi: 10.1016/j.pain.2011.01.017. [DOI] [PubMed] [Google Scholar]
- Payne HR, Hemperly JJ, Lemmon V. N-Cadherin expression and function in cultured oligodendrocytes. Brain Research. Developmental Brain Research. 1996;97:9–15. doi: 10.1016/s0165-3806(96)00124-1. [DOI] [PubMed] [Google Scholar]
- Peng YR, He S, Marie H, Zeng SY, Ma J, Tan ZJ, et al. Coordinated changes in dendritic arborization and synaptic strength during neural circuit development. Neuron. 2009;61:71–84. doi: 10.1016/j.neuron.2008.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pertz O, Bozic D, Koch AW, Fauser C, Brancaccio A, Engel J. A new crystal structure, Ca2+ dependence and mutational analysis reveal molecular details of E-cadherin homoassociation. The EMBO Journal. 1999;18:1738–1747. doi: 10.1093/emboj/18.7.1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettem KL, Yokomaku D, Luo L, Linhoff MW, Prasad T, Connor SA, et al. The specific alpha-neurexin interactor calsyntenin-3 promotes excitatory and inhibitory synapse development. Neuron. 2013;80:113–128. doi: 10.1016/j.neuron.2013.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pielarski KN, van Stegen B, Andreyeva A, Nieweg K, Jungling K, Redies C, et al. Asymmetric N-cadherin expression results in synapse dysfunction, synapse elimination, and axon retraction in cultured mouse neurons. PLoS One. 2013;8:e54105. doi: 10.1371/journal.pone.0054105. http://dx.doi.org/10.1371/journal.pone.0054105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pokutta S, Herrenknecht K, Kemler R, Engel J. Conformational changes of the recombinant extracellular domain of E-cadherin upon calcium binding. European Journal of Biochemistry. 1994;223:1019–1026. doi: 10.1111/j.1432-1033.1994.tb19080.x. [DOI] [PubMed] [Google Scholar]
- Poskanzer K, Needleman LA, Bozdagi O, Huntley GW. N-Cadherin regulates ingrowth and laminar targeting of thalamocortical axons. The Journal of Neurosci-ence. 2003;23:2294–2305. doi: 10.1523/JNEUROSCI.23-06-02294.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prange O, Wong TP, Gerrow K, Wang YT, El-Husseini A. A balance between excitatory and inhibitory synapses is controlled by PSD-95 and neuroligin. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:13915–13920. doi: 10.1073/pnas.0405939101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rago L, Beattie R, Taylor V, Winter J. miR379-410 cluster miRNAs regulate neurogenesis and neuronal migration by fine-tuning N-cadherin. The EMBO Journal. 2014;33:906–920. doi: 10.1002/embj.201386591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakshit S, Zhang Y, Manibog K, Shafraz O, Sivasankar S. Ideal, catch, and slip bonds in cadherin adhesion. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:18815–18820. doi: 10.1073/pnas.1208349109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranscht B, Dours-Zimmermann MT. T-Cadherin, a novel cadherin cell adhesion molecule in the nervous system lacks the conserved cytoplasmic region. Neuron. 1991;7:391–402. doi: 10.1016/0896-6273(91)90291-7. [DOI] [PubMed] [Google Scholar]
- Rapoport J, Chavez A, Greenstein D, Addington A, Gogtay N. Autism spec-trumdisordersandchildhood-onsetschizophrenia:Clinicalandbiologicalcontributionstoa relation revisited. Journal of the American Academy of Child and Adolescent Psychiatry. 2009;48:10–18. doi: 10.1097/CHI.0b013e31818b1c63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redies C, Hertel N, Hubner CA. Cadherins and neuropsychiatric disorders. Brain Research. 2012;1470:130–144. doi: 10.1016/j.brainres.2012.06.020. [DOI] [PubMed] [Google Scholar]
- Reines A, Bernier LP, McAdam R, Belkaid W, Shan W, Koch AW, et al. N-Cadherin prodomain processing regulates synaptogenesis. The Journal of Neuroscience. 2012;32:6323–6334. doi: 10.1523/JNEUROSCI.0916-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiss K, Maretzky T, Ludwig A, Tousseyn T, de Strooper B, Hartmann D, et al. ADAM10 cleavage of N-cadherin and regulation of cell-cell adhesion and beta-catenin nuclear signalling. The EMBO Journal. 2005;24:742–752. doi: 10.1038/sj.emboj.7600548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riehl R, Johnson K, Bradley R, Grunwald GB, Cornel E, Lilienbaum A, et al. Cadherin function is required for axon outgrowth in retinal ganglion cells in vivo. Neuron. 1996;17:837–848. doi: 10.1016/s0896-6273(00)80216-0. [DOI] [PubMed] [Google Scholar]
- Rivero O, Sich S, Popp S, Schmitt A, Franke B, Lesch KP. Impact of the ADHD-susceptibility gene CDH13 on development and function of brain networks. European Neuropsychopharmacology. 2013;23:492–507. doi: 10.1016/j.euroneuro.2012.06.009. [DOI] [PubMed] [Google Scholar]
- Rubio ME, Curcio C, Chauvet N, Bruses JL. Assembly of the N-cadherin complex during synapse formation involves uncoupling of p120-catenin and association with presenilin 1. Molecular and Cellular Neurosciences. 2005;30:611–623. [PubMed] [Google Scholar]
- Ryan SG, Chance PF, Zou CH, Spinner NB, Golden JA, Smietana S. Epilepsy and mental retardation limited to females: An X-linked dominant disorder with male sparing. Nature Genetics. 1997;17:92–95. doi: 10.1038/ng0997-92. [DOI] [PubMed] [Google Scholar]
- Saglietti L, Dequidt C, Kamieniarz K, Rousset MC, Valnegri P, Thoumine O, et al. Extracellular interactions between GluR2 and N-cadherin in spine regulation. Neuron. 2007;54:461–477. doi: 10.1016/j.neuron.2007.04.012. [DOI] [PubMed] [Google Scholar]
- Sakane F, Miyamoto Y. N-Cadherin regulates the proliferation and differentiation of ventral midbrain dopaminergic progenitors. Developmental Neurobiology. 2013;73:518–529. doi: 10.1002/dneu.22077. [DOI] [PubMed] [Google Scholar]
- Sakarya O, Armstrong KA, Adamska M, Adamski M, Wang IF, Tidor B, et al. A post-synaptic scaffold at the origin of the animal kingdom. PLoS One. 2007;2:e506. doi: 10.1371/journal.pone.0000506. http://dx.doi.org/10.1371/journal.pone.0000506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders SJ, Ercan-Sencicek AG, Hus V, Luo R, Murtha MT, Moreno-De-Luca D, et al. Multiple recurrent de novo CNVs, including duplications of the 7q11.23 Williams syndrome region, are strongly associated with autism. Neuron. 2011;70:863–885. doi: 10.1016/j.neuron.2011.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanhueza M, Lisman J. The CaMKII/NMDAR complex as a molecular memory. Molecular Brain. 2013;6:10. doi: 10.1186/1756-6606-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sansom SN, Livesey FJ. Gradients in the brain: The control of the development of form and function in the cerebral cortex. Cold Spring Harbor Perspectives in Biology. 2009;1:a002519. doi: 10.1101/cshperspect.a002519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schalm SS, Ballif BA, Buchanan SM, Phillips GR, Maniatis T. Phos-phorylation of protocadherin proteins by the receptor tyrosine kinase Ret. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:13894–13899. doi: 10.1073/pnas.1007182107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheiffele P, Fan J, Choih J, Fetter R, Serafini T. Neuroligin expressed in non-neuronal cells triggers presynaptic development in contacting axons. Cell. 2000;101:657–669. doi: 10.1016/s0092-8674(00)80877-6. [DOI] [PubMed] [Google Scholar]
- Schnadelbach O, Blaschuk OW, Symonds M, Gour BJ, Doherty P, Fawcett JW. N-Cadherin influences migration of oligodendrocytes on astro-cyte monolayers. Molecular and Cellular Neurosciences. 2000;15:288–302. doi: 10.1006/mcne.1999.0819. [DOI] [PubMed] [Google Scholar]
- Schnadelbach O, Ozen I, Blaschuk OW, Meyer RL, Fawcett JW. N-Cadherin is involved in axon-oligodendrocyte contact and myelination. Molecular and Cellular Neurosciences. 2001;17:1084–1093. doi: 10.1006/mcne.2001.0961. [DOI] [PubMed] [Google Scholar]
- Schnell E, Sizemore M, Karimzadegan S, Chen L, Bredt DS, Nicoll RA. Direct interactions between PSD-95 and stargazin control synaptic AMPA receptor number. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:13902–13907. doi: 10.1073/pnas.172511199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiner D, Weiner JA. Combinatorial homophilic interaction between gamma-protocadherin multimers greatly expands the molecular diversity of cell adhesion. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:14893–14898. doi: 10.1073/pnas.1004526107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrick C, Fischer A, Srivastava DP, Tronson NC, Penzes P, Radulovic J. N-cadherin regulates cytoskeletally associated IQGAP1/ERK signaling and memory formation. Neuron. 2007;55:786–798. doi: 10.1016/j.neuron.2007.07.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwabe T, Neuert H, Clandinin TR. A network of cadherin-mediated interactions polarizes growth cones to determine targeting specificity. Cell. 2013;154:351–364. doi: 10.1016/j.cell.2013.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan WS, Tanaka H, Phillips GR, Arndt K, Yoshida M, Colman DR, et al. Functional cis-heterodimers of N- and R-cadherins. The Journal of Cell Biology. 2000;148:579–590. doi: 10.1083/jcb.148.3.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan W, Yoshida M, Wu XR, Huntley GW, Colman DR. Neural (N-) cadherin, a synaptic adhesion molecule, is induced in hippocampal mossy fiber axonal sprouts by seizure. Journal of Neuroscience Research. 2002;69:292–304. doi: 10.1002/jnr.10305. [DOI] [PubMed] [Google Scholar]
- Shankar GM, Bloodgood BL, Townsend M, Walsh DM, Selkoe DJ, Sabatini BL. Natural oligomers of the Alzheimer amyloid-beta protein induce reversible synapse loss by modulating an NMDA-type glutamate receptor-dependent signaling pathway. The Journal of Neuroscience. 2007;27:2866–2875. doi: 10.1523/JNEUROSCI.4970-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro L, Fannon AM, Kwong PD, Thompson A, Lehmann MS, Grubel G, et al. Structural basis of cell-cell adhesion by cadherins. Nature. 1995;374:327–337. doi: 10.1038/374327a0. [DOI] [PubMed] [Google Scholar]
- Shapiro L, Weis WI. Structure and biochemistry of cadherins and catenins. Cold Spring Harbor Perspectives in Biology. 2009;1:a003053. doi: 10.1101/cshperspect.a003053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi SH, Hayashi Y, Petralia RS, Zaman SH, Wenthold RJ, Svoboda K, et al. Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science. 1999;284:1811–1816. doi: 10.1126/science.284.5421.1811. [DOI] [PubMed] [Google Scholar]
- Shimamura K, Takahashi T, Takeichi M. E-cadherin expression in a particular subset of sensory neurons. Developmental Biology. 1992;152:242–254. doi: 10.1016/0012-1606(92)90132-z. [DOI] [PubMed] [Google Scholar]
- Sibille E, Wang Y, Joeyen-Waldorf J, Gaiteri C, Surget A, Oh S, et al. A molecular signature of depression in the amygdala. The American Journal of Psychiatry. 2009;166:1011–1024. doi: 10.1176/appi.ajp.2009.08121760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva AJ. Molecular and cellular cognitive studies of the role of synaptic plasticity in memory. Journal of Neurobiology. 2003;54:224–237. doi: 10.1002/neu.10169. [DOI] [PubMed] [Google Scholar]
- Silverman JB, Restituito S, Lu W, Lee-Edwards L, Khatri L, Ziff EB. Synaptic anchorage of AMPA receptors by cadherins through neural plakophilin-related arm protein AMPA receptor-binding protein complexes. The Journal of Neuroscience. 2007;27:8505–8516. doi: 10.1523/JNEUROSCI.1395-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simonoff E, Pickles A, Charman T, Chandler S, Loucas T, Baird G. Psychiatric disorders in children with autism spectrum disorders: Prevalence, comorbidity, and associated factors in a population-derived sample. Journal of the American Academy of Child and Adolescent Psychiatry. 2008;47:921–929. doi: 10.1097/CHI.0b013e318179964f. [DOI] [PubMed] [Google Scholar]
- Srivastava DP, Copits BA, Xie Z, Huda R, Jones KA, Mukherji S, et al. Afadin is required for maintenance of dendritic structure and excitatory tone. The Journal of Biological Chemistry. 2012;287:35964–35974. doi: 10.1074/jbc.M112.363358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stan A, Pielarski KN, Brigadski T, Wittenmayer N, Fedorchenko O, Gohla A, et al. Essential cooperation of N-cadherin and neuroligin-1 in the transsynaptic control of vesicle accumulation. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:11116–11121. doi: 10.1073/pnas.0914233107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein V, House DR, Bredt DS, Nicoll RA. Postsynaptic density-95 mimics and occludes hippocampal long-term potentiation and enhances long-term depression. The Journal of Neuroscience. 2003;23:5503–5506. doi: 10.1523/JNEUROSCI.23-13-05503.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ster J, Steuble M, Orlando C, Diep TM, Akhmedov A, Raineteau O, et al. Calsyntenin-1 regulates targeting of dendritic NMDA receptors and dendritic spine maturation in CA1 hippocampal pyramidal cells during postnatal development. The Journal of Neuroscience. 2014;34:8716–8727. doi: 10.1523/JNEUROSCI.0144-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugino H, Hamada S, Yasuda R, Tuji A, Matsuda Y, Fujita M, et al. Geno-mic organization of the family of CNR cadherin genes in mice and humans. Genomics. 2000;63:75–87. doi: 10.1006/geno.1999.6066. [DOI] [PubMed] [Google Scholar]
- Sun Y, Aiga M, Yoshida E, Humbert PO, Bamji SX. Scribble interacts with beta-catenin to localize synaptic vesicles to synapses. Molecular Biology of the Cell. 2009;20:3390–3400. doi: 10.1091/mbc.E08-12-1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Bamji SX. beta-Pix modulates actin-mediated recruitment of synaptic vesicles to synapses. The Journal of Neuroscience. 2011;31:17123–17133. doi: 10.1523/JNEUROSCI.2359-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suyama K, Shapiro I, Guttman M, Hazan RB. A signaling pathway leading to metastasis is controlled by N-cadherin and the FGF receptor. Cancer Cell. 2002;2:301–314. doi: 10.1016/s1535-6108(02)00150-2. [DOI] [PubMed] [Google Scholar]
- Suzuki SC, Inoue T, Kimura Y, Tanaka T, Takeichi M. Neuronal circuits are subdivided by differential expression of Type-II classic cadherins in postnatal mouse brains. Molecular and Cellular Neurosciences. 1997;9:433–447. doi: 10.1006/mcne.1997.0626. [DOI] [PubMed] [Google Scholar]
- Tai CY, Mysore SP, Chiu C, Schuman EM. Activity-regulated N-cadherin endocytosis. Neuron. 2007;54:771–785. doi: 10.1016/j.neuron.2007.05.013. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Osumi N. Identification of a novel type II classical cadherin: Rat cadherin19 is expressed in the cranial ganglia and Schwann cell precursors during development. Developmental Dynamics. 2005;232:200–208. doi: 10.1002/dvdy.20209. [DOI] [PubMed] [Google Scholar]
- Takai Y, Ikeda W, Ogita H, Rikitake Y. The immunoglobulin-like cell adhesion molecule nectin and its associated protein afadin. Annual Review of Cell and Developmental Biology. 2008;24:309–342. doi: 10.1146/annurev.cellbio.24.110707.175339. [DOI] [PubMed] [Google Scholar]
- Takeichi M, Inuzuka H, Shimamura K, Matsunaga M, Nose A. Cadherin-mediated cell-cell adhesion and neurogenesis. Neuroscience Research. Supplement. 1990;13:S92–S96. doi: 10.1016/0921-8696(90)90036-3. [DOI] [PubMed] [Google Scholar]
- Tan ZJ, Peng Y, Song HL, Zheng JJ, Yu X. N-Cadherin-dependent neuron-neuron interaction is required for the maintenance of activity-induced dendrite growth. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:9873–9878. doi: 10.1073/pnas.1003480107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka H, Shan W, Phillips GR, Arndt K, Bozdagi O, Shapiro L, et al. Molecular modification of N-cadherin in response to synaptic activity. Neuron. 2000;25:93–107. doi: 10.1016/s0896-6273(00)80874-0. [DOI] [PubMed] [Google Scholar]
- Tang L, Hung CP, Schuman EM. A role for the cadherin family of cell adhesion molecules in hippocampal long-term potentiation. Neuron. 1998;20:1165–1175. doi: 10.1016/s0896-6273(00)80497-3. [DOI] [PubMed] [Google Scholar]
- Tasic B, Nabholz CE, Baldwin KK, Kim Y, Rueckert EH, Ribich SA, et al. Promoter choice determines splice site selection in protocadherin alpha and gamma pre-mRNA splicing. Molecular Cell. 2002;10:21–33. doi: 10.1016/s1097-2765(02)00578-6. [DOI] [PubMed] [Google Scholar]
- Taylor AM, Wu J, Tai HC, Schuman EM. Axonal translation of beta-catenin regulates synaptic vesicle dynamics. The Journal of Neuroscience. 2013;33:5584–5589. doi: 10.1523/JNEUROSCI.2944-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas C, Strutt D. The roles of the cadherins Fat and dachsous in planar polarity specification in drosophila. Developmental Dynamics. 2012;241:27–39. doi: 10.1002/dvdy.22736. [DOI] [PubMed] [Google Scholar]
- Tissir F, Bar I, Jossin Y, De Backer O, Goffinet AM. Protocadherin Celsr3 is crucial in axonal tract development. Nature Neuroscience. 2005;8:451–457. doi: 10.1038/nn1428. [DOI] [PubMed] [Google Scholar]
- Togashi H, Abe K, Mizoguchi A, Takaoka K, Chisaka O, Takeichi M. Cadherin regulates dendritic spine morphogenesis. Neuron. 2002;35:77–89. doi: 10.1016/s0896-6273(02)00748-1. [DOI] [PubMed] [Google Scholar]
- Tomaselli KJ, Neugebauer KM, Bixby JL, Lilien J, Reichardt LF. N-Cadherin and integrins: Two receptor systems that mediate neuronal process outgrowth on astrocyte surfaces. Neuron. 1988;1:33–43. doi: 10.1016/0896-6273(88)90207-3. [DOI] [PubMed] [Google Scholar]
- Toyoda S, Kawaguchi M, Kobayashi T, Tarusawa E, Toyama T, Okano M, et al. Developmental epigenetic modification regulates stochastic expression of clustered protocadherin genes, generating single neuron diversity. Neuron. 2014;82:94–108. doi: 10.1016/j.neuron.2014.02.005. [DOI] [PubMed] [Google Scholar]
- Treubert-Zimmermann U, Heyers D, Redies C. Targeting axons to specific fiber tracts in vivo by altering cadherin expression. The Journal of Neuroscience. 2002;22:7617–7626. doi: 10.1523/JNEUROSCI.22-17-07617.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai NP, Wilkerson JR, Guo W, Maksimova MA, DeMartino GN, Cowan CW, et al. Multiple autism-linked genes mediate synapse elimination via proteasomal degradation of a synaptic scaffold PSD-95. Cell. 2012;151:1581–1594. doi: 10.1016/j.cell.2012.11.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien JZ, Chen DF, Gerber D, Tom C, Mercer EH, Anderson DJ, et al. Subregion- and cell type-restricted gene knockout in mouse brain. Cell. 1996;87:1317–1326. doi: 10.1016/s0092-8674(00)81826-7. [DOI] [PubMed] [Google Scholar]
- Uchida N, Honjo Y, Johnson KR, Wheelock MJ, Takeichi M. The catenin/cadherin adhesion system is localized in synaptic junctions bordering transmitter release zones. Journal of Cell Biology. 1996;135:767–779. doi: 10.1083/jcb.135.3.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uemura K, Kuzuya A, Aoyagi N, Ando K, Shimozono Y, Ninomiya H, et al. Amyloid beta inhibits ectodomain shedding of N-cadherin via down-regulation of cell-surface NMDA receptor. Neuroscience. 2007;145:5–10. doi: 10.1016/j.neuroscience.2006.12.022. [DOI] [PubMed] [Google Scholar]
- Uemura M, Nakao S, Suzuki ST, Takeichi M, Hirano S. OL-protocadherin is essential for growth of striatal axons and thalamocortical projections. Nature Neuroscience. 2007;10:1151–1159. doi: 10.1038/nn1960. [DOI] [PubMed] [Google Scholar]
- Ullian EM, Christopherson KS, Barres BA. Role for glia in synaptogenesis. Glia. 2004;47:209–216. doi: 10.1002/glia.20082. [DOI] [PubMed] [Google Scholar]
- Um JW, Pramanik G, Ko JS, Song MY, Lee D, Kim H, et al. Calsyntenins function as synaptogenic adhesion molecules in concert with neurexins. Cell Reports. 2014;6:1096–1109. doi: 10.1016/j.celrep.2014.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Utton MA, Eickholt B, Howell FV, Wallis J, Doherty P. Soluble N-cadherin stimulates fibroblast growth factor receptor dependent neurite outgrowth and N-cadherin and the fibroblast growth factor receptor co-cluster in cells. Journal of Neurochemistry. 2001;76:1421–1430. doi: 10.1046/j.1471-4159.2001.00140.x. [DOI] [PubMed] [Google Scholar]
- Vanhalst K, Kools P, Staes K, van Roy F, Redies C. Delta-protocadherins: A gene family expressed differentially in the mouse brain. Cellular and Molecular Life Sciences. 2005;62:1247–1259. doi: 10.1007/s00018-005-5021-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Harssel JJ, Weckhuysen S, van Kempen MJ, Hardies K, Verbeek NE, de Kovel CG, et al. Clinical and genetic aspects of PCDH19-related epilepsy syndromes and the possible role of PCDH19 mutations in males with autism spectrum disorders. Neurogenetics. 2013;14:23–34. doi: 10.1007/s10048-013-0353-1. [DOI] [PubMed] [Google Scholar]
- Waites CL, Craig AM, Garner CC. Mechanisms of vertebrate syn-aptogenesis. Annual Review of Neuroscience. 2005;28:251–274. doi: 10.1146/annurev.neuro.27.070203.144336. [DOI] [PubMed] [Google Scholar]
- Walker S, Scherer SW. Identification of candidate intergenic risk loci in autism spectrum disorder. BMC Genomics. 2013;14:499. doi: 10.1186/1471-2164-14-499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Su H, Bradley A. Molecular mechanisms governing pcdh-gamma gene expression: Evidence for a multiple promoter and cis-alternative splicing model. Genes & Development. 2002;16:1890–1905. doi: 10.1101/gad.1004802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Weiner JA, Levi S, Craig AM, Bradley A, Sanes JR. Gamma protocadherins are required for survival of spinal interneurons. Neuron. 2002;36:843–854. doi: 10.1016/s0896-6273(02)01090-5. [DOI] [PubMed] [Google Scholar]
- Wang K, Zhang H, Ma D, Bucan M, Glessner JT, Abrahams BS, et al. Common genetic variants on 5p14.1 associate with autism spectrum disorders. Nature. 2009;459:528–533. doi: 10.1038/nature07999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiner JA, Jontes JD. Protocadherins, not prototypical: A complex tale of their interactions, expression, and functions. Frontiers in Molecular Neuroscience. 2013;6:4. doi: 10.3389/fnmol.2013.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiner JA, Wang X, Tapia JC, Sanes JR. Gamma protocadherins are required for synaptic development in the spinal cord. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:8–14. doi: 10.1073/pnas.0407931101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wenzel HJ, Woolley CS, Robbins CA, Schwartzkroin PA. Kainic acid-induced mossy fiber sprouting and synapse formation in the dentate gyrus of rats. Hippocampus. 2000;10:244–260. doi: 10.1002/1098-1063(2000)10:3<244::AID-HIPO5>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- Williams EJ, Mittal B, Walsh FS, Doherty P. A Ca2+/calmodulin kinase inhibitor, KN-62, inhibits neurite outgrowth stimulated by CAMs and FGF. Molecular and Cellular Neurosciences. 1995;6:69–79. doi: 10.1006/mcne.1995.1007. [DOI] [PubMed] [Google Scholar]
- Williams EJ, Walsh FS, Doherty P. The production of arachidonic acid can account for calcium channel activation in the second messenger pathway underlying neurite outgrowth stimulated by NCAM, N-cadherin, and L1. Journal of Neurochemistry. 1994;62:1231–1234. doi: 10.1046/j.1471-4159.1994.62031231.x. [DOI] [PubMed] [Google Scholar]
- Williams ME, Wilke SA, Daggett A, Davis E, Otto S, Ravi D, et al. Cadherin-9 regulates synapse-specific differentiation in the developing hippocampus. Neuron. 2011;71:640–655. doi: 10.1016/j.neuron.2011.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams EJ, Williams G, Howell FV, Skaper SD, Walsh FS, Doherty P. Identification of an N-cadherin motif that can interact with the fibroblast growth factor receptor and is required for axonal growth. The Journal of Biological Chemistry. 2001;276:43879–43886. doi: 10.1074/jbc.M105876200. [DOI] [PubMed] [Google Scholar]
- Wöhrn JC, Nakagawa S, Ast M, Takeichi M, Redies C. Combinatorial expression of cadherins in the tectum and the sorting of neurites in the tectofugal pathways of the chicken embryo. Neuroscience. 1999;90:985–1000. doi: 10.1016/s0306-4522(98)00526-0. [DOI] [PubMed] [Google Scholar]
- Wong GK, Baudet ML, Norden C, Leung L, Harris WA. Slit1b-Robo3 signaling and N-cadherin regulate apical process retraction in developing retinal ganglion cells. The Journal of Neuroscience. 2012;32:223–228. doi: 10.1523/JNEUROSCI.2596-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Q, Maniatis T. A striking organization of a large family of human neural cadherin-like cell adhesion genes. Cell. 1999;97:779–790. doi: 10.1016/s0092-8674(00)80789-8. [DOI] [PubMed] [Google Scholar]
- Wu Y, Vendome J, Shapiro L, Ben-Shaul A, Honig B. Transforming binding affinities from three dimensions to two with application to cadherin clustering. Nature. 2011;475:510–513. doi: 10.1038/nature10183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu ZC, Yu JT, Wang ND, Yu NN, Zhang Q, Chen W, et al. Lack of association between PCDH11X genetic variation and late-onset Alzheimer’s disease in a Han Chinese population. Brain Research. 2010;1357:152–156. doi: 10.1016/j.brainres.2010.08.008. [DOI] [PubMed] [Google Scholar]
- Wu Q, Zhang T, Cheng JF, Kim Y, Grimwood J, Schmutz J, et al. Comparative DNA sequence analysis of mouse and human protocadherin gene clusters. Genome Research. 2001;11:389–404. doi: 10.1101/gr.167301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagi T, Takeichi M. Cadherin superfamily genes: Functions, genomic organization, and neurologic diversity. Genes & Development. 2000;14:1169–1180. [PubMed] [Google Scholar]
- Yam PT, Pincus Z, Gupta GD, Bashkurov M, Charron F, Pelletier L, et al. N-Cadherin relocalizes from the periphery to the center of the synapse after transient synaptic stimulation in hippocampal neurons. PLoS One. 2013;8:e79679. doi: 10.1371/journal.pone.0079679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada S, Pokutta S, Drees F, Weis WI, Nelson WJ. Deconstructing the cadherin-catenin-actin complex. Cell. 2005;123:889–901. doi: 10.1016/j.cell.2005.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamagata M, Herman JP, Sanes JR. Lamina-specific expression of adhesion molecules in developing chick optic tectum. The Journal of Neuroscience. 1995;15:4556–4571. doi: 10.1523/JNEUROSCI.15-06-04556.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi M, Imai F, Tonou-Fujimori N, Masai I. Mutations in N-cadherin and a Stardust homolog, Nagie oko, affect cell-cycle exit in zebrafish retina. Mechanisms of Development. 2010;127:247–264. doi: 10.1016/j.mod.2010.03.004. [DOI] [PubMed] [Google Scholar]
- Yang Y, Wang XB, Frerking M, Zhou Q. Delivery of AMPA receptors to perisynaptic sites precedes the full expression of long-term potentiation. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:11388–11393. doi: 10.1073/pnas.0802978105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yap AS, Niessen CM, Gumbiner BM. The juxtamembrane region of the cadherin cytoplasmic tail supports lateral clustering, adhesive strengthening, and interaction with p120ctn. The Journal of Cell Biology. 1998;141:779–789. doi: 10.1083/jcb.141.3.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuda S, Tanaka H, Sugiura H, Okamura K, Sakaguchi T, Tran U, et al. Activity-induced protocadherin arcadlin regulates dendritic spine number by triggering N-cadherin endocytosis via TAO2beta and p38 MAP kinases. Neuron. 2007;56:456–471. doi: 10.1016/j.neuron.2007.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yizhar O, Fenno LE, Prigge M, Schneider F, Davidson TJ, O’Shea DJ, et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011;477:171–178. doi: 10.1038/nature10360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Malenka RC. Beta-catenin is critical for dendritic morphogenesis. Nature Neuroscience. 2003;6:1169–1177. doi: 10.1038/nn1132. [DOI] [PubMed] [Google Scholar]
- Yudowski GA, Puthenveedu MA, Leonoudakis D, Panicker S, Thorn KS, Beattie EC, et al. Real-time imaging of discrete exocytic events mediating surface delivery of AMPA receptors. The Journal of Neuroscience. 2007;27:11112–11121. doi: 10.1523/JNEUROSCI.2465-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamanillo D, Sprengel R, Hvalby O, Jensen V, Burnashev N, Rozov A, et al. Importance of AMPA receptors for hippocampal synaptic plasticity but not for spatial learning. Science. 1999;284:1805–1811. doi: 10.1126/science.284.5421.1805. [DOI] [PubMed] [Google Scholar]
- Zhang W, Benson DL. Stages of synapse development defined by dependence on F-actin. The Journal of Neuroscience. 2001;21:5169–5181. doi: 10.1523/JNEUROSCI.21-14-05169.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z, Hu J, Passafaro M, Xie W, Jia Z. GluA2 (GluR2) regulates meta-botropic glutamate receptor-dependent long-term depression through N-cadherin-dependent and cofilin-mediated actin reorganization. The Journal of Neuroscience. 2011;31:819–833. doi: 10.1523/JNEUROSCI.3869-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu H, Luo L. Diverse functions of N-cadherin in dendritic and axonal terminal arborization of olfactory projection neurons. Neuron. 2004;42:63–75. doi: 10.1016/s0896-6273(04)00142-4. [DOI] [PubMed] [Google Scholar]