Abstract
Aim
To evaluate the possibility of implementing a new scheme of rescue treatment after relapse or progression of high-grade glioma (HGG) treated at the first-line with bevacizumab and irinotecan (BVZ+CPT11), evaluating the response and toxicity of associating BVZ and fractionated stereotactic radiotherapy (BVZ+FSRT).
Materials and methods
We retrospectively analysed data from 59 patients with relapse of HGG. Nine patients with HGG relapse after treatment using the Stupp protocol that were treated with BVZ+CPT11 for progression between July 2007 and August 2012, after which the response was assessed according to the Revised Assessment in Neuro-Oncology (RANO) criteria. BVZ was administered at a dose of 10 mg/kg and FSRT up to a prescribed dose of 30 Gy, 500 cGy per fraction, three days a week. The median follow-up was 38 months.
Results
The treatment was well-tolerated by all patients. The response after nuclear magnetic resonance imaging (MRI) at 3–6 months was progression in two patients, stable disease in four, and three patients had a partial response. The median overall survival (OS) from diagnosis until death or the last control was 36.8 months. The median progression-free survival (PFS) was 10.8 months. The results from tumour sub-group analysis indicated that the PFS was not statistically significant although it seemed that it was higher in grade-III. The OS was higher in grade-III gliomas.
Conclusions
The combination of BVZ+FSRT as a second-line HGG relapse rescue treatment is well-tolerated and seems to offer promising results. We believe that multi-centre prospective studies are needed to determine the long-term efficacy and toxicity of this therapeutic approach.
Abbreviations: HGG, high-grade glioma; BVZ, bevacizumab; CPT11, irinotecan; FSRT, fractionated stereotactic radiotherapy; RANO, revised Assessment in Neuro-Oncology; MRI, magnetic resonance imaging; OS, overall survival; PFS, progression-free survival; TMZ, temozolomide; KPS, Karnofsky Performance Scale; VEGF, vascular endothelial growth factor; PTV, planning target volume; CAT, computed axial tomography; GTV, gross tumour volume; SEOM, Sociedad Española de Oncología Médica; ASCO, American Society of Clinical Oncology; CTCAE, common terminology criteria for adverse events; PET, positron emission tomography; CI, confidence interval; MGMT, O-6-methylguanine-DNA methyltransferase; HR, hazard ratio; CR, complete response; PR, partial response; SD, stable disease; SRS, stereotactic radiosurgery; PD, progressive disease; FLAIR, fluid-attenuated inversion recovery; NA, not applicable
Keywords: High-grade glioma, Rescue treatment, Bevacizumab, Fractionated stereotactic radiotherapy
1. Background
High-grade gliomas (HGGs) are heterogeneous entities that comprise primary glioblastomas, anaplastic oligodendrogliomas and gliomas, and secondary glioblastomas, together representing 25% of primary central nervous system tumours.1 Among patients affected with glioblastoma, less than 10% of those treated with the standard Stupp protocol2 (maximal surgical resection, external radiotherapy, and concomitant and adjuvant temozolomide [TMZ]) survive more than five years from diagnosis.3
HGG relapse treatment is changing. Although surgical resection is an option in relapse, the infiltrative nature of glioma makes complete tumour resection difficult during the rescue surgery and chemotherapy is being used with modest success because of chemoresistance and accumulated toxicity. Advances in the biology of HGGs allow targeted therapies to be developed, such as the use of bevacizumab (BVZ), a humanised monoclonal antibody against the vascular endothelial growth factor (VEGF) expressed in HGG cells, which used alone or in combination with external radiotherapy, is offering promising results.4
Fractionated stereotactic radiotherapy (FSRT) provides local control in HGG relapses with minimal morbidity. Case series and prospective and retrospective studies demonstrate efficacy and acceptable FSRT toxicity in the treatment of this disease.5
The purpose of this work was to evaluate the possibility of implementing a new relapse or progression treatment-scheme for HGG treated at the first-line with BVZ+CPT11, evaluating the response and toxicity of associating BVZ and fractionated stereotactic radiotherapy (BVZ+FSRT).
2. Materials and methods
2.1. Patient selection
This retrospective study was conducted with permission from the institutional review and ethics board of our centre; thus, we analysed data from 59 patients treated for HGG relapse between 2007 and 2014, of whom we found nine whose progression was treated with compassionate use with BVZ-CPT11 after a relapse following original Stupp protocol treatment, response was assessed using the RANO criteria (Table 1). After assessment by a multidisciplinary committee which discarded the possibility of any other type of treatment, BVZ was administered (dose of 10 mg/kg) on day two on the first day FSRT was started, FSRT was administered at a prescribed dose of 30 Gy, 500 cGy per fraction, three times a week, between July 2007 and August 2012.
Table 1.
RANO criteria.
| Criterion | CR | PR | SD | PD |
|---|---|---|---|---|
| T1 gadolinium-enhancing disease activity | None | ≥50%↓ | <50%↓ but <25%↑ | ≥25%↑a |
| T2/FLAIR disease activity | Stable or ↓ | Stable or ↓ | Stable or ↓ | ↑a |
| New lesion | None | None | None | Presenta |
| Corticosteroids | None | Stable or ↓ | Stable or ↓ | NAb |
| Clinical status | Stable or ↑ | Stable or ↑ | Stable or ↑ | ↓a |
| Response-requirement | All | All | All | Anya |
Abbreviations: RANO, Response Assessment in Neuro-Oncology; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; FLAIR, fluid-attenuated inversion recovery; NA, not applicable.
Progression occurs when this criterion is present.
Increased in corticosteroids alone will not be taken into account in determining progression in the absence of persistent clinical deterioration.
After the initial diagnosis, all the patients were treated by a total tumour excision followed by conventional external radiotherapy and concomitant and adjuvant TMZ (except for patient one where adjuvant chemotherapy was suspended because of haematological toxicity). All of them had relapsed after primary treatment, which was confirmed with nuclear magnetic resonance imaging (MRI) neuroimaging techniques.
The median follow-up was 38 months. All of them were in a good general condition, with no neurological deficits after primary treatment, with a Karnofsky Performance Scale (KPS) score above 70%, MGMT status was not tested; rescue surgery was discarded as the first option and was reserved as an option for use after the second-line treatment response in all of them.
2.2. Patient characteristics
The mean patient age was 54.8 years and the cohort consisted of five men and four women. The histology was: 2 patients with anaplasic oligodendroglioma, 2 grade II glioma and 3 glioblastoma. In 88.8% of the patients the primary tumour was located in a single cerebral lobe, presenting in the left frontal lobe in two patients, right temporal lobe in four of them, the right frontal lobe in one, and the right parietal lobe in the other. The primary location of the tumour was multiple in only one case. In eight patients the relapse was located at the base of or near the tumour, and of all the patients studied, 22.2% presented leptomeningeal spread (Table 2).
Table 2.
Characteristics of the study patients.
| n | % | |
|---|---|---|
| Sex | ||
| Men | 5 | 55.5 |
| Women | 4 | 44.4 |
| Age | ||
| <50 years | 2 | 22.2 |
| ≥50 years | 7 | 77.7 |
| Primary tumour location | ||
| Single lobe | 8 | 88.8 |
| Multiple lobes | 1 | 11.1 |
| Relapse location | ||
| Original tumour site or close | 8 | 88.8 |
| In another location | 1 | 11.1 |
| BVZ+CPT11 cycles | ||
| <10 | 6 | 66.6 |
| ≥10 | 3 | 33.3 |
| Toxicitya | ||
| Attributable to the treatment | 0 | 0 |
| Not attributable to the treatment | 9 | 100 |
| Time elapsedb | ||
| <12 months | 5 | 55.5 |
| ≥12 months | 4 | 44.4 |
| Radiological responsec | ||
| CR | 0 | 0 |
| PR | 3 | 33.3 |
| SD | 4 | 44.4 |
| PD | 2 | 22.2 |
| Average FSRT dosed | ||
| <29 Gy | 5 | 55.5 |
| ≥29 Gy | 4 | 44.4 |
Radio-necrosis confirmed by biopsy, a multiparametric nuclear magnetic resonance (MRI) study, or positron emission tomography (PET). BVZ, bevacizumab; CPT11, irinotecan.
Time between initial Stupp scheme treatment and administration of rescue treatment with BVZ+FSRT.
According to Revised Assessment in Neuro-Oncology (RANO) criteria.
Isodose of 95–103% to cover the lesion and 85–95% to cover the planning target volume (PTV).
66.6% of the patients received less than 10 neoadjuvant BVZ+CPT-11 cycles and the average time between administration of the Stupp scheme and the relapse treatment with BVZ+FSRT was 17.8 months. Systemic treatment scheme was based on recently published studies13 and it followed BVZ 10 mg/kg (1st and 15th day of 28 days) plus irinotecan 125 mg/m2. About patients evaluation, no quality of life specific tests were applied. A toxicity follow-up was performed in all cases, using the Barthel escale, Mini-Mental Score and ECOG performance status. The average FSRT dose administered to these patients was 29.2 Gy (covering the lesion with an isodose of between 95 and 103% with a planning target volume [PTV] of between 85 and 95%), this dose being less than 29 Gy in 55.5% of the patients.
2.3. Fractionated stereotactic radiotherapy technique
All patients underwent a computed axial tomography (CAT) simulation for radiotherapy treatment planning with cuts every 1.5 mm using a thermoplastic immobilisation mask (Brain-Lab system), as well as a T1-sequence gadolinium-contrast brain MRI with cuts every 1.5 mm, these images were merged with those from the CAT simulation. The gross tumour volume (GTV) was determined as the lesion captured by the T1 contrast sequence and the PTV was defined as the GTV plus a 4 mm margin. The treatment planning was carried out using a Brain SCAN planning system (Brainlab).
An overall dose of 30 Gy was administered (5 Gy per fraction in 6 fractions every other day; one patient received 6 Gy per fraction in 5 fractions) as determined by an isodose of 100%, using a single isocenter and an average of 13 non-coplanar beams (8–17 beams). The mean GTV was 11.14 cc (0.24–65.02 cc). Daily treatment positioning verification was performed using the ExacTrac® system. All the patients received corticosteroid therapy during the FSRT treatment period.
2.4. Systemic therapies and toxicity
We reviewed the clinical histories of all the patients to determine which systemic therapies were received by each of them before and after BVZ+FSRT treatment. Administration of systemic therapies was directed by the Medical Oncology service of the Castellon Provincial Hospital Consortium according to the therapeutic protocols recommended by the Spanish Society of Medical Oncology (Sociedad Española de Oncología Médica; SEOM) and the American Society of Clinical Oncology (ASCO) in coordination with the multidisciplinary neuro-oncology committee from the Provincial and General hospitals in Castellon.
Toxicity was determined based on the common terminology criteria for adverse events (CTCAE, version 4), which defines it as the appearance of new symptoms or the worsening of previous symptoms after administration of the treatment. Symptoms which were caused by progression of the disease were excluded.
2.5. Statistical analysis
Overall survival (OS) was considered from diagnosis and from the point of BVZ+FSRT treatment to death or last control and progression-free survival (PFS) were considered from diagnosis and from the point of BVZ+FSRT treatment to the date of diagnosis of tumour recurrence. Survival functions were calculated using the Kaplan–Meier method. For comparison of survival curves, a log-rank test was used.
The limit of statistical significance was established as a p value of alpha = 0.05. Statistical analysis was performed using SPSS (Statistical Package for Social Science) for Windows, version 17.0 (SPSS Inc., Chicago, IL, USA) and R software version 2.15.1.
3. Results
3.1. Response to rescue treatment with bevacizumab and fractionated stereotactic radiotherapy
At 3–6 months the disease progressed in two patients, was stable in four, and a partial response was obtained in three (according to the RANO criteria) as detected by MRI. When clinical or radiological progression was detected, a second round of BVZ-CPT11 treatment was administered in three cases and BVZ only in the other three after a new multidisciplinary committee evaluation.
Both treatments were administered every 15 days, with an average of 5.5 cycles (range: 2–8 cycles).
When the final clinical history of the patients was analysed, six had deceased, one had a partial response, and two remained with stable disease criteria.
3.2. Toxicity
The treatment was well-tolerated by all of the patients and the toxicity could not be clearly attributed to either of the two treatments. One patient presented with a diagnosis of radionecrosis indicated by an imaging test performed in another centre (non-multiparametric MRI). Clinically, this patient presented with occasional sphincter incontinence, bradypsychia, and clumsiness when walking which was attributed to the possible presence of radionecrosis and not to disease progression, but, as indicated, this could not be confirmed by biopsy or multiparametric MRI or positron emission tomography (PET) studies. No grade 4–5 toxicities occurred in relation to administration of BVZ or FSRT.
3.3. Survival
The median OS from diagnosis to death at the last control was 36.8 months (95% confidence interval [95% CI]: 25.3–48.3). The estimated mean OS was 40.7 months (95% CI: 27.8–53.6; Fig. 1).
Fig. 1.
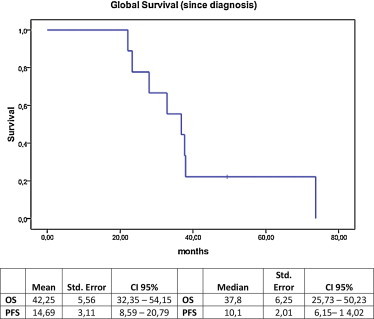
Overall survival from high-grade glioma diagnosis until death.
The median progression-free survival (PFS) from the point of BVZ+FSRT treatment was 6.4 months (95% CI: 2.0–10.8). The estimated average PFS was 12.1 months (95% CI: 4.4–19.7; Fig. 2).
Fig. 2.
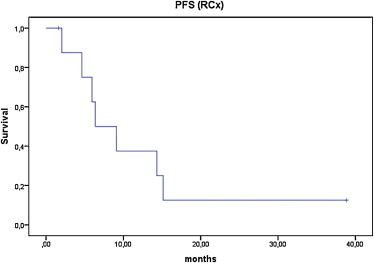
Progression-free survival after bevacizumab and fractionated stereotactic radiotherapy treatment.
The differences in PFS (since diagnosis) between the tumour subgroups were 5.3 months for grade-IV gliomas (95% CI: 4.0–6.6), 12.1 months for grade-III-OA gliomas (95% CI: 6.2–18.0), and the highest PFS was for grade-III gliomas at 15.4 months (95% CI: 1.4–29.4; Fig. 3).
Fig. 3.
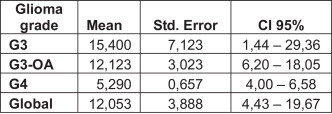
Progression-free survival by tumour subgroups.
The median progression-free survival (PFS) from the point of diagnosis to the first disease progression was 10.1 months (95% CI: 6.2–14.0). The estimated average PFS was 14.7 months (95% CI: 8.6–20.8). The differences in PFS between the tumour subgroups were higher for grade-III anaplastic oligodendrogliomas. The mean survival after BVZ+FSRT treatment was 15.3 months for grade-IV gliomas (95% CI: 3.8–26.8), 10 months for grade-III gliomas (95% CI: 2.86–17.40), and the highest PFS was for grade-III-OA gliomas at 23 months (95% CI: 8.8–37.13; Fig. 4).
Fig. 4.
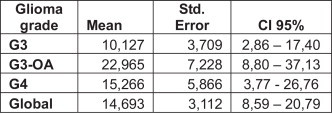
Overall survival by tumour subgroup after BVZ+FSRT treatment.
The OS was higher in grade-III gliomas (OS = 54.5 m) and anaplastic oligodendrogliomas (OS = 37.4 m) compared to grade-IV gliomas (OS = 24.5 m; Fig. 5).
Fig. 5.
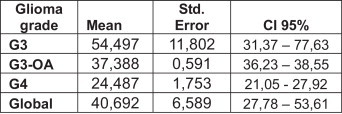
Overall survival by tumour subgroup since diagnosis.
It was clinically observed that the presence of more than one tumour lesion increases the risk of death and that this had a direct impact on the OS.
4. Discussion
The treatment choices for relapse patients with high-grade glioma are limited. The treatments used include surgical resection and systemic treatment with chemotherapy drugs or new molecular therapy agents until the administration of a second brain irradiation dose.
4.1. Rescue with surgery
The value of surgery has been considered controversial in the past and few studies have demonstrated promising results after surgery for relapsed HGG patients.
In general terms, the results are poor and surgical resection is sometimes associated with a higher risk of secondary effects given the naturally infiltrative nature of the disease.6
Park et al. recently generated an interesting tool to determine a prognostic scale applicable to the surgical intervention results from patients with relapsed glioma. This work took into account relevant factors that influence brain resection, such as the tumour volume, location, its association with vital brain areas, and factors related to the clinical condition of the patient, such as the KPS. They developed a point score index ranging from 0 to 3, good to poor, and demonstrated that patients with a final score of 3 benefitted very little form a surgical approach to their disease, and assert that the decision of which treatment to apply should be individualised for each patient taking the previously mentioned factors into account.7
4.2. Rescue with radiotherapy
With regard to administration of radiotherapy as a rescue treatment in relapsed HGG patients, conventional external radiotherapy is not generally possible because of the elevated risk of toxicity to the brain which can provoke an increase in neuro-cognitive deficits and necrosis induced by radiation.8
Multiple studies have demonstrated the security and efficacy of cerebral re-irradiation with distinct treatment schemes, including FSRT, radiosurgery, or non-conventional fractionation techniques; this second irradiation is always performed six months after the first irradiation with conventional external radiotherapy.6 However there is little experience of these studies and to date no phase-III trials have been published in the literature. Given that there are no randomised trials, data on the role of re-irradiation after progression of the disease or the appearance of radiation-induced cancers are scarce and poorly defined. The literature is limited to a series of small retrospective clinical case studies, which makes their interpretation difficult.9
Brachytherapy, which aims to deliver a localised dose of high-level radiation targeted at the tumour while protecting the normal cerebral tissue, points towards a modest improvement in the OS but has shown an elevated risk of cerebral necrosis (which lowers its percentage of effectiveness if tumour debulking has previously been performed). Chan et al. carried out a study on 24 relapsed HGG patients, administering low-dose rate brachytherapy with I125 (GliaSite®) in the previously operated area and evaluated the secondary effects (acute and delayed) and the OS. The majority of these effects were mild, infrequent, and were resolvable with symptomatic treatment, except in two cases that presented with symptoms of radio-necrosis. They observed that these two patients had received a dose of 50 Gy or more at a prescribed dose depth of more than 1 cm: the presence of an increased amount of tumour in these patients meant that they had to administer higher doses. They concluded that because they did not demonstrate an increase in OS at doses higher than 45 Gy, this might be a sufficient treatment dose with minimal risk of developing toxicity, provided that an initial optimal tumour resection is performed leaving minimal tumour residue.10
4.3. Re-irradiation and radio-necrosis
There is level-1 evidence for BVZ as a radio-necrosis treatment, the main risk of cerebral re-irradiation11,12 It has been demonstrated that molecular therapy with BVZ reverses the effects of radio-necrosis because (VEGF) is altered in radio-necrosis and BVZ acts on this factor. As described in their article, Chao et al. administered BVZ to 8 patients with cerebral radio-necrosis and showed a reduction in MRI contrast in all of them (T2 fluid-attenuated inversion recovery [FLAIR] and T1 post-gadolinium imaging). Moreover, it also helped reduce the corticosteroid dose. Levin et al.12 demonstrated that in a series of 14 patients all those treated with BVZ showed improvement in neurological signs and symptoms, but that there was no alteration in those treated with the placebo. Therefore, performing a second irradiation seems justified and safe if it is administered along with BVZ.
4.4. Molecular therapy
BVZ has shown an anti-tumour effect in HGG relapses,13 although the results are not clear in first-line treatment, as shown in the AVAglio and RTOG studies14,15 In the AVAGlio study on HGG patients previously treated with surgery, the addition of BVZ to the adjuvant treatment with TMZ+External beam Radiation Therapy (EBRT) did not significantly increase the OS, although it did increase the PFS by 4.4 months with an HR of 0.61, and improved the quality of life as shown by maintenance of a stable KPS score in this period, and reduced corticosteroid doses. However, this benefit was not seen as clearly in the RTOG 0825 study in which the patients’ symptoms and neuro-cognitive function deteriorated with BVZ. The justifications for these differences include, among others, statistical anomalies in the analysis of a data subset, given that studies on Quality of Life and Neuro-cognitive tests were only performed on a subgroup of patients in both studies, coupled with possible differences in the statistical methods used. The AVAglio study focussed only on the quality of life and not on neurocognitive data, and so, given that BVZ produces a larger gap at the blood-brain barrier and lowers gadolinium MRI-enhancement, there could have been radiologically occult disease progression in these patients which would justify the worsening of neuro-cognitive function observed in the RTOG 0825 study. Regarding the failure to increase OS, it is important to note that different factors can act to modify the response in different patients, such as the study design, treatment cross-effects, the presence of distinct patient molecular sub-types, or the presence of suboptimal BVZ concentrations at the site of action. Therefore, it is inevitable that VEGF may not be considered an optimal target in all HGG cases, or at least, not at all points of the disease, which would justify the appearance of the 10–20% prolongation of PFS and OS in patients treated with TMZ+EBRT+BVZ for recurrent glioblastoma.16,17 All of this makes it essential to know the molecular HGG profile of individual patients so as to be able to individualise the treatment and optimise resources.
4.5. Associated therapy
Turning to associated therapy (FSRT ± BVZ), there are multiple studies in the literature. The first one, a prospective study with 25 patients, was published in 2009 by Gutin et al. and they concluded that BVZ associated with FSRTin HGG relapse treatment was safe and well-tolerated.18 Niyazi et al. carried out a retrospective study with 30 patients, demonstrating that OS was higher in patients in whom BVZ was associated with the rescue treatment, and that the OS was also affected by the tumour grade and general condition of the patients.19 Several studies posit that the OS and the PFS are the highest where BVZ is associated with radiosurgery or FSRT (Table 3).
Table 3.
Studies associating bevacizumab with radio-surgery or fractionated stereotactic radiotherapy.
| Study | Patient No. | Treatment | Age (years) | KPS | Tumour volume n (cm3) | RTX dose (Gy) | Time between initial diagnosis and salvage RTX (months) | PFS (months) | OS from salvage RTX (months) | OS from initial diagnosis (months) | Radiation necrosis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hall et al. | 26 | SRS (LINAC) | 47 | 70 | 28 | 20 | 10 | NR | 6.5 | 18 | 14% |
| Sheirive et al. | 86 | SRS (LINAC) | 46 | 80 | 10.1 | 13 | 10.3 | NR | 10.2 | NR | 16% |
| Combs et al. | 32 | SRS (LINAC) | 56 | NR | 10 | 15 | 10 | 5 | 10 | 22 | 0% |
| Hsieh et al. | 26 | SRS (LINAC) | 58 | 70 | 21.6 | 12 | NR | NR | 10 | 16.7 | 31% |
| Mahajan et al. | 41 | SRS (LINAC) | 54 | NR | NR | NR | 11 | NR | 11 | 26 | NR |
| Vordemark et al. | 14 | HFSRT | 50 | 90 | 15 | 30 | 19 | 4.6 | 7.9 | NR | 0% |
| Kong et al. | 65 | SRS (GK) | 49 | 100 | 10.6 | 16 | NR | 4.6 | 13 | 23 | 24.4% |
| Patel et al. | 10 | HFSRT | 44 | 90 | 51.1 | NR | 14.9 | NR | 7.4 | 24.1 | NR |
| Patel et al. | 26 | SRS | 53 | 80 | 10.4 | 18 | 12.5 | NR | 8.4 | 24.4 | 7.6% |
| Gutin et al. | 20 | HFSRT+BVZ | 56 | 80 | 34 | 30 | 14.5 | 7.3 | 12.5 | NR | 0% |
| Fogh et al. | 105 | HFSRT | 53 | NR | 22 | 35 | 8 | NR | 11 | 23 | NR |
| Cuneo et al. | 42 | SRS (LINAC)+BVZ | 47 | 80 | 4.5 | 15 | 21 | 5.2 | 11.2 | 47 | 5% |
BVZ, bevacizumab; GK, gamma knife; HFSRT, hypofractionated stereotactic radiation therapy; KPS, Karnofsky performance status; LINAC, linear accelerator; OS, overall survival; PFS, progression free survival; RTX, radiation therapy; SRS, stereotactic radiosurgery; NR, not record.
One retrospective study, performed by Torcuator et al., compared 23 patients treated with either FSRT or stereotactic radiosurgery (SRS) after progression on an initial bevacizumab regimen to other 23 patients who, after progression to BVZ+chemotherapy, continued with more BVZ+chemotherapy. Patients treated with FSRT/SRS/BVZ had a longer median progression-free period (2.6 vs. 1.7 months), longer median post FSRT/SRS treatment survival (7.2 vs. 3.3 months) and higher radiographic response rate (22 vs. 0%). Thus, they concluded that FSRT or SRS followed by bevacizumab+chemotherapy may have a role for patients who progress on bevacizumab.20 Our modest study of 9 patients with HGG tumour relapse treated with FSRT+BVZ after a response with BVZ+CPT11, also showed higher OS and PFS figures, similar to those studies where FSRT+BVZ were performed as initial treatment in HGG relapse (Table 4).
Table 4.
Studies in which combined therapy is associated with high-grade glioma rescue treatment.
| Centre/author | Scheme | n | OS | OS after FSRT | PFS after FSRT |
|---|---|---|---|---|---|
| CHP Castellón-Eresa/Conde et al. | (BVZ+CPT11) BVZ+FSRT |
9 | 36.8 m | 15.7 m | 6.4 m |
| MSKCC/Gutin et al. | BVZ+FSRT | 24 | 32.1 m | 12.2 m | 7.5 m |
| LMU | BVZ+FSRT | 20 | – | 12 m | 8.1 m |
| Munich/Niyazi et al. | FSRT | 7 | 4.7 m | 5.8 m | |
| Michigan/Cabrera et al. | BVZ+FSRT | 15 | – | 14.4 m | 3.9 m |
| Torcuator et al. | (After progression to BVZ) BVZ+FSRT/SRS |
23 | – | 7.2 m | 2.7 m |
In 2013, the Memorial Sloan Kettering Cancer Centre published an update on its 2009 study on 22 patients in which they evaluated their clinical and radiological progression, showing that radiological progression is the only variable related with PFS.4 In the same year, Cabrera et al. observed, in a prospective study on 15 patients, that combined therapy did not produce grade 4–5 toxicity, neuro-cognitive deterioration, or a KPS decrease, and that there was a decrease in the MRI perfusion after radio-surgery or FSRT.3
To date, our retrospective study is the first to evaluate results from FSRT+BVZ after response to neoadjuvant therapy with BVZ+CPT11 in relapsed HGG in patients where FSRT was discarded as the first treatment to recurrence. Remaining tumour could not be identified on the control MRI in one patient after having received FSRT+BVZ treatment for six cycles. Another showed a 0.8 mm reduction in the size of the lesion after receiving 10 cycles and yet another showed a complete tumour response after 8 cycles. However, we do not yet have clear data which can guide us on what strategy should be followed after a response to BVZ+CPT11: if treatment should be continued or discontinued until progression, or if another type of treatment should be assessed.
Other factors which must be taken into account are response evaluation criteria. Several of referenced studies followed Macdonald criteria, but it is known that the RANO criteria are more suitable to evaluate response to anti-VEGF treatment. Therefore, these criteria were used in our study as in other studies, such as S. Comas et al., in which it is shown T2-Flair MRI sequence role in order to evaluate response, especially in neoadjuvant treatment.21
4.6. Prognostic scales
Various prognostic models have been developed to predict survival in patients newly diagnosed with HGG, above all, at the time of first-line treatment. Weller et al. evaluated the prognostic value of 11 molecular markers in patients treated with the Stupp scheme, reaching the conclusion that the only factor with prognostic significance was O-6-methylguanine-DNA methyltransferase (MGMT) gene methylation,22 which was associated with longer survival.23 In glioblastomas a DNA test can be carried out to determine if the MGMT gene promotor is methylated or not: a prognosis of significantly longer survival is associated with MGMT gene promoter methylation HGG patients compared to those with an unmethylated MGMT gene promoter, because the former can benefit from a better response to TMZ treatment.24
To improve prognostic predictions, these models have to be updated with data about HGG relapses. Thus Gorlia et al. conducted a combined study on the EORTC trials which analysed HGG relapses with the aim of validating the existing clinical prognostic factors, identifying markers which might be useful as future predictive factors for OS and PFS. Three hundred relapsed HGG patients were recruited to eight phase-I and -II studies (carried out by the EORTIC tumour group), in which the patients’ age, sex, clinical status (based on their performance status score), the presence of neurological deficits, the history of their disease, use of corticosteroid or anti-epileptic drugs, and the disease characteristics were measured. The analysis demonstrated that performance status, but not age, is a prognostic factor for OS and PFS in HGG relapses and that patients with large and multiple lesions are at a higher risk of death.25
5. Conclusions
OS and PFS in HGG relapse after treatment with surgery and FSRT+BVZ is very limited. We believe that after a first-line response to BVZ+CPT11, another option might be to explore the other types of treatments which were initially excluded. Rescue treatment with FSRT+BVZ was well-tolerated in this series of patients and seems to increase the OS and PFS. This was a retrospective study in which the administration of FSRT+BVZ was assessed only in patients who had had a second-line response and those in whom a surgical option was again discarded; hence, the low number of patients included, and this bias may explain the differences between different patient series in different studies, meaning that a prospective multicentre study is necessary to address this issue. Tissue and serological markers associated with the response should be evaluated, as well as changes in the CAT-MRI perfusion pattern in order to search for the optimum timing for administration of relapse rescue treatment in HGG relapse patients.
Conflict of interest
None declared.
Financial disclosure
None declared.
Acknowledgements
The authors wish to thank to the Provincial Hospital of Castellón Foundation, for their assistance in the development of this work and especially to Francisco García-Piñón, data manager and biostatistician, for his invaluable assistance in this work.
References
- 1.Mirimanoff R.O. High-grade gliomas: reality and hopes. Chin J Cancer. 2014;33(January (1)):1–3. doi: 10.5732/cjc.013.10215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stupp R., Hegi M.E., Mason W.P. Effects of radiotherapy with concomitant and adjuvant temozolamide versus radiotherapy alone on survival in glioblastoma in a randomized phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. [DOI] [PubMed] [Google Scholar]
- 3.Cabrera A.R., Cuneo K.C., Desjardins A. Concurrent stereotactic radiosurgery and bevacizumab in recurrent malignant gliomas: a prospective trial. Int J Radiat Oncol Biol Phys. 2013;86(5):873–879. doi: 10.1016/j.ijrobp.2013.04.029. [DOI] [PubMed] [Google Scholar]
- 4.Shapiro L.Q., Beal K., Goenka A. Patterns of failure after concurrent bevacizumab and hypofractionated stereotactic radiation therapy for recurrent high-grade glioma. Int J Radiat Oncol Biol Phys. 2013;85(3):636–642. doi: 10.1016/j.ijrobp.2012.05.031. [DOI] [PubMed] [Google Scholar]
- 5.Cuneo K.C., Vredenburgh J.J., Sampson J.H. Safety and efficacy of steretotactic radiosurgery and adjuvant bevacizumab in patients with recurrent malignant gliomas. Int J Radiat Oncol Biol Phys. 2012;82(5):2018–2024. doi: 10.1016/j.ijrobp.2010.12.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Combs S.E., Edler L., Rausch R., Welzel T., Wick W., Debus J. Generation and validation of a prognostic score to predict outcome after re-irradiation of recurrent glioma. Acta Oncol. 2013;52:147–152. doi: 10.3109/0284186X.2012.692882. [DOI] [PubMed] [Google Scholar]
- 7.Park J.K., Hodges T., Arko L. Scale to predict survival after surgery for recurrent glioblastoma multiforme. J Clin Oncol. 2010;28:3838–3843. doi: 10.1200/JCO.2010.30.0582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mayer R., Sminia P. Reirradiation tolerance of the human brain. Int J Radiat Oncol Biol Phys. 2008;70(5):1350–1360. doi: 10.1016/j.ijrobp.2007.08.015. [DOI] [PubMed] [Google Scholar]
- 9.Paulino A.C., Mai W.Y., Chintagumpala M., Taher A., Teh B.S. Radiation-induced malignant gliomas: is there a role for reirradiation? Int J Radiat Oncol Biol Phys. 2008;71(5):1381–1387. doi: 10.1016/j.ijrobp.2007.12.018. [DOI] [PubMed] [Google Scholar]
- 10.Chan T.A., Weingart J.D., Parisi M. Treatment of recurrent glioblastoma multiforme with GliaSite® brachytherapy. Int J Radiat Oncol Biol Phys. 2005;62(4):1133–1139. doi: 10.1016/j.ijrobp.2004.12.032. [DOI] [PubMed] [Google Scholar]
- 11.Chao S.T., Ahluwalia M.S., Barnett G.H. Challenges with the diagnosis and treatment of cerebral radiation necrosis. Int J Radiat Oncol Biol Phys. 2013;87(3):449–457. doi: 10.1016/j.ijrobp.2013.05.015. [DOI] [PubMed] [Google Scholar]
- 12.Levin V.A1, Bidaut L., Hou P. Randomized double-blind placebo-controlled trial of bevacizumab therapy necrosis of the central nervous system. Int J Radiat Oncol Biol Phys. 2011;79(5):1487–1495. doi: 10.1016/j.ijrobp.2009.12.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Friedman H.S., Prados M.D., Wen P.Y. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009;27(28):4733–4740. doi: 10.1200/JCO.2008.19.8721. [DOI] [PubMed] [Google Scholar]
- 14.Chinot O.L., de La Motte Rouge T., Moore N. AVAglio: Phase 3 trial of bevacizumab plus temozolomide and radiotherapy in newly diagnosed glioblastoma multiforme. Adv Ther. 2011;28(4):334–340. doi: 10.1007/s12325-011-0007-3. [DOI] [PubMed] [Google Scholar]
- 15.Soffietti R., Trevisan E., Rudà R. What have we learned from trials on antiangiogenic agents in glioblastoma? Expert Rev Neurother. 2014;14(January (1)):1–3. doi: 10.1586/14737175.2014.873277. [DOI] [PubMed] [Google Scholar]
- 16.Kreisl T.N., Zhang W., Odia Y. A phase II trial of single-agent bevacizumab in patients with recurrent anaplastic glioma. Neuro Oncol. 2011;13(October (10)):1143–1150. doi: 10.1093/neuonc/nor091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Khasraw M., Ameratunga M., Gommes C. Bevacizumab for the treatment of high-grade glioma: an update after phase III trials. Expert Opin Biol Ther. 2014;14(5):729–740. doi: 10.1517/14712598.2014.898060. [DOI] [PubMed] [Google Scholar]
- 18.Gutin P.H., Iwamoto F.M., Beal K. Safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys. 2009;75(1):156–163. doi: 10.1016/j.ijrobp.2008.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Niyazi M., Ganswindt U., Schwarz S.B. Irradiation and bevacizumab in high-grade glioma retreatment settings. Int J Radiat Oncol Biol Phys. 2012;82(1):67–76. doi: 10.1016/j.ijrobp.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 20.Torcuator R.G., Thind R., Patel M. The role of salvage reirradiation for malignant gliomas that progress on bevacizumab. J Neurooncol. 2010;97(May (3)):401–407. doi: 10.1007/s11060-009-0034-y. [DOI] [PubMed] [Google Scholar]
- 21.Comas S., Villà S., Etxaniz O. Patterns of relapse in glioblastoma after neoadjuvancy and radiation therapy. Rep Pract Oncol Radiother. 2013;18:S195. [Google Scholar]
- 22.Hegi M.E., Diserens A.C., Gorlia T. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005;352(10):997–1003. doi: 10.1056/NEJMoa043331. [DOI] [PubMed] [Google Scholar]
- 23.Krex D., Klink B., Hartmann C. Long-term survival with glioblastoma multiforme. Brain. 2007;130(10):2596–2606. doi: 10.1093/brain/awm204. [DOI] [PubMed] [Google Scholar]
- 24.Würth R., Barbieri F., Florio T. New molecules and old drugs as emerging approaches to selectively target human glioblastoma cancer stem cells. Biomed Res Int. 2014;2014:126586. doi: 10.1155/2014/126586. [Epub Jan 2, 2014] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gorlia T., Stupp R., Brandes A.A. New prognostic factors and calculators for outcome prediction in patients with recurrent glioblastoma: a pooled analysis of EORTC Brain Tumour Group phase I and II clinical trials. Eur J Cancer. 2012;48(8):1176–1184. doi: 10.1016/j.ejca.2012.02.004. [DOI] [PubMed] [Google Scholar]


