Abstract
Ascorbic acid (AsA) is the most abundant water-soluble antioxidant in plants, and it plays a crucial role in plant growth, development and abiotic stress tolerance. In the present study, six key Arabidopsis or rapeseed genes involved in AsA biosynthesis were constitutively overexpressed in an elite Japonica rice cultivar. These genes encoded the GDP-mannose pyrophosphorylase (GMP), GDP-mannose-3',5'-epimerase (GME), GDP-L-galactose phosphorylase (GGP), L-galactose-1-phosphate phosphatase (GPP), L-galactose dehydrogenase (GDH), and L-galactono-1,4-lactone dehydrogenase (GalLDH). The effects of transgene expression on rice leaf AsA accumulation were carefully evaluated. In homozygous transgenic seedlings, AtGGP transgenic lines had the highest AsA contents (2.55-fold greater than the empty vector transgenic control), followed by the AtGME and AtGDH transgenic lines. Moreover, with the exception of the AtGPP lines, the increased AsA content also provoked an increase in the redox state (AsA/DHA ratio). To evaluate salt tolerance, AtGGP and AtGME transgenic seedlings were exposed to salt stress for one week. The relative plant height, root length and fresh weight growth rates were significantly higher for the transgenic lines compared with the control plants. Altogether, our results suggest that GGP may be a key rate-limiting step in rice AsA biosynthesis, and the plants with elevated AsA contents demonstrated enhanced tolerance for salt stress.
Introduction
L-ascorbic acid (AsA), also known by its popular name vitamin C, is the most abundant water-soluble antioxidant in plants. It protects plants from oxidative damage by scavenging the free radicals and reactive oxygen species generated by photosynthesis, oxidative metabolism, and environmental stresses [1]. AsA has been shown to play a critical role in plant growth and development, as well as in tolerance of abiotic stresses such as drought [2], salinity [3,4], ozone [5], and high light intensity [6]. Additionally, AsA is a cofactor for many enzymes that are involved in multiple processes, including flavonoid and phytohormone biosynthesis as well as the xanthophyll cycle [7, 8].
To date, four alternative AsA biosynthesis pathways have been proposed in plants: the L-galactose [9], galacturonate [10,11], glucose [12] and myo-inositol pathways [13]. Among these pathways, the L-galactose pathway might have an important role in cereal AsA biosynthesis (Fig 1) [14]. For example, when rice seedlings were supplied with L-galactose and L-galactono-1,4-lactone, shoot AsA contents increased up to 5- and 4-fold, respectively; in contrast, for D-galacturonic acid, myo-inositol, D-glucuronolactone and L-guluno-1,4-lactone, AsA levels were similar to those of the untreated control plants [15]. At least ten enzymes in the L-galactose pathway are involved in converting D-glucose to AsA through the formation of GDP-D-mannose, GDP-L-galactose, L-galactose-1-P, L-galactose, and L-galactono-1,4-lactone intermediates (Fig 1).
Fig 1. The L-galactose pathway in higher plants [14].
Most of the genes involved in this pathway, e.g., GDP-mannose pyrophosphorylase (GMP) [16,17], GDP-mannose-3',5'-epimerase (GME) [3], GDP-L-galactose phosphorylase (GGP) [18,19], L-galactose-1-phosphate phosphatase (GPP) [20], L-galactose dehydrogenase (GDH) [21], and L-galactono-1,4-lactone dehydrogenase (GalLDH) [22,23], have been cloned, and several researchers have attempted to regulate AsA content by overexpressing them in transgenic plants. However, these studies each only focused on one gene, and few works have examined the effects of transgenic regulation of all of the above genes in one experiment, especially in rice, one of the most important crops consumed worldwide.
Feeding with AsA precursors during rice growth led to an increase in leaf AsA content, shedding light onto the development of cultivars with enhanced tolerance for key environmental stresses [24–26]. In contrast, the ND6172 rice mutant had approximately 20–30% lower AsA levels and exhibited negative effects on agronomic traits at ozone levels exceeding the current ambient concentrations [27]. Moreover, reducing GalLDH expression in rice was reported to increase lipid peroxidation, but also decrease foliar AsA levels [22]. These results revealed that the AsA content of rice leaves is a sensitive characteristic that might be consistent with stress tolerance. Furthermore, the AsA redox state, i.e., the ratio between ascorbate and dehydroascorbate (AsA/DHA), also plays important role in plant senescence, defense, and stress response [28–30]. The AsA/DHA ratio may depend on the plant species; in general, it is approximately 1.0 in 4-week-old rice seedlings [31, 32].
In the present study, the expression levels of genes involved in the rice L-galactose pathway were compared by quantitative RT-PCR. Six L-galactose pathway genes from Arabidopsis or rapeseed, including GMP, GME, GGP, GPP, GDH and GalLDH (Fig 1), were then overexpressed in rice and their effects on rice seedling AsA accumulation were carefully compared. This results of this analysis showed that GGP overexpression had the best AsA accumulation, followed by GME and GDH, and the abiotic stress tolerances of GGP and GME transgenic rice plants were subsequently evaluated.
Materials and Methods
Plant materials
The Japonica rice variety Nipponbare was used for quantitative expression analysis of the endogenous L-galactose pathway genes. Another elite Japonica cultivar, Wuyujing 3 (WY3), was used for generation of transgenic lines. All rice plants were grown in the field at the experimental farm of Yangzhou University (32°39’N, 119°42’E) under normal culture conditions. Developing endosperm, stems, sheaths and old leaves were sampled and collected at 20 days, and young leaves and roots were harvested from 4-week-old seedlings. The collected samples were immediately frozen in liquid nitrogen and stored at −80°C.
Transgenic vector construction
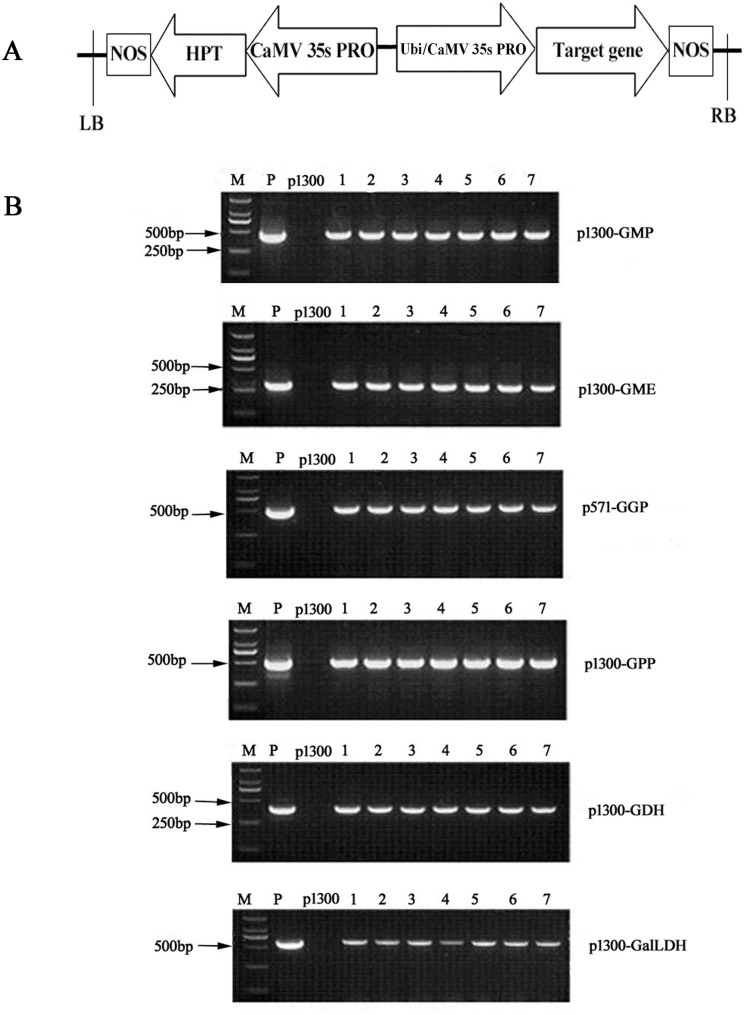
Arabidopsis AtGGP, AtGMP, AtGME, AtGPP, AtGDH and rapeseed BoGalLDH cDNAs (GenBank accession numbers AY093138, AF076484, NM_122767, NM_001035549, AJ417563, and Z97060, respectively) were previously cloned by RT-PCR [20]. To construct the transgenic vectors, the cDNAs were amplified to add suitable restriction enzymatic sites using the primers listed in S1 Table. The amplified cDNAs were then inserted into the pMD18-T vector (TAKARA, Dalian, China) and confirmed by sequence analysis. The full-length cDNAs, regulated by the maize Ubi or CaMV 35s promoters and NOS terminator, were cloned into the binary pCAMBIA1300 vector (www.cambia.com.au). With the exception of AtGGP, which was driven by the CaMV 35s promoter, the target genes were regulated by the maize Ubi promoter. The resulting binary vectors (Fig 2) containing the target genes and the corresponding empty pCAMBIA1300 control vector were transformed into Agrobacterium tumefacien strain EHA105 and subsequently used for rice transformation.
Fig 2. Rice transformation constructs (A) and PCR amplification of transformed genes (B).
A. Schematic representation of the transformation vectors used in this study. CaMV 35S pro, cauliflower mosaic virus 35S promoter; Ubi pro, maize ubiquitin promoter; NOS, nopaline synthase gene terminator; HPT, hygromycin resistance gene; LB, left border; RB, right border. B. PCR analysis of transgenic and control lines using specific primers for amplification of each transgene. Lanes 1–7, transgenic plants (T0 generation); P, positive control; M, DL2000 DNA marker (TAKARA, Dalian, China).
Rice transformation and confirmation
Calli derived from either immature or mature WY3 rice cultivar embryos were used as explants for Agrobacterium-mediated transformation. Tissue culture, transformation, selection and regeneration of transgenic plants were performed as described by Liu et al. [33]. Genomic DNA was extracted from leaves using the CTAB method [34], and PCR analysis was performed using transgene-specific primer sets (S1 Table) to detect the transgenes.
Quantitative RT-PCR
Total RNA was isolated as previously described [35]. A 1 μg sample of each RNA preparation was used for reverse transcription using the SuperScript First-Strand Synthesis System (Invitrogen, Carlsbad, CA) and oligo(dT)18 according to the manufacturer’s instructions. Aliquots of cDNA were used in 25 μl real-time PCR assay reactions using the SYBR Premix ExTaq kit (TaKaRa, Tokyo, Japan). The results were normalized against rice actin mRNA. The sequences of the real-time PCR assay primers are listed in S1 Table.
AsA and DHA measurements
AsA was extracted according to the method described by Zhou et al. [20]. Briefly, 50 mg of leaves from 4-week-old seedlings were harvested at 6 pm, ground in liquid nitrogen, mixed with 1 ml 0.1% oxalic acid in dim light, and placed on ice for 1 h. The samples were centrifuged at 10 000 g for 10 min and the supernatants were then pooled and filtered for measurement. For AsA assays measuring the reduced + oxidized forms, 25 μL (25 mmol L-1) DTT and 25 μL (320 mmol L-1) K2HPO4 were added to 500 μL supernatant, incubated for 15 minutes at room temperature, centrifuged, and then filtered for measurements. The amounts of DHA were determined as the difference between the two assays. Standard L-AsA solutions (1 mg ml-1) (Sigma, St. Louis, MO, USA) were freshly prepared prior to use. L-AsA HPLC analysis was performed using a C-18 reversed phase column (5 μm, 150×4.6 mm). Samples (10 μL) were injected using a Waters 2695 autosampler. The solvent system was 0.1% oxalic acid at a 1.0 mL min-1 flow rate, and the samples were analyzed with a UV detector at 245 nm.
Salt stress treatment
Germinated seeds from transgenic and control lines were placed in the 96-well PCR plate (without bottom) and pre-grown in modified Hoagland’s solution (S2 Table). The culture was under a light intensity of about 700 μmol photons m−2 s−1, 75% humidity and a 16/8-h light/dark photoperiod at 25°C for three weeks. Seedlings were then cultured in solutions containing 100 mM NaCl for one week. The solutions were renewed everyday, and three replications were used in salt stress evaluation experiments. The plant heights, root lengths and fresh weights were measured before and after salt stress, and the relative plant height, root length and fresh weight growth rates were calculated.
Relative membrane ion leakage analysis
The oxidative stress tolerance of the plants was assessed by ion leakage analysis as described by Wang et al. [36]. Ten leaf discs of 6 mm diameter were placed in a cuvette containing 20 mL distilled water, vacuumized for 30 min, and then surged for 10 h at room temperature to measure the initial electronic conductance (S1) using an osmometer (DDS-11A, SuoShen, Shanghai, China). The cuvette was heated in boiling water for 30 min and cooled to room temperature to determine the final electronic conductance (S2). The relative membrane ion leakage (%) was defined as 100× (S1/S2).
Statistical analysis
The data were analyzed using a statistical software package (SPSS v16.0). One-way analysis of variance (ANOVA) was used to compare the differences between the transgenic and control lines, followed by Tukey’s post hoc test. A p-value below 0.05 was considered to be statistically significant.
Results
Enriched expression of the L-galactose pathway in young rice leaves
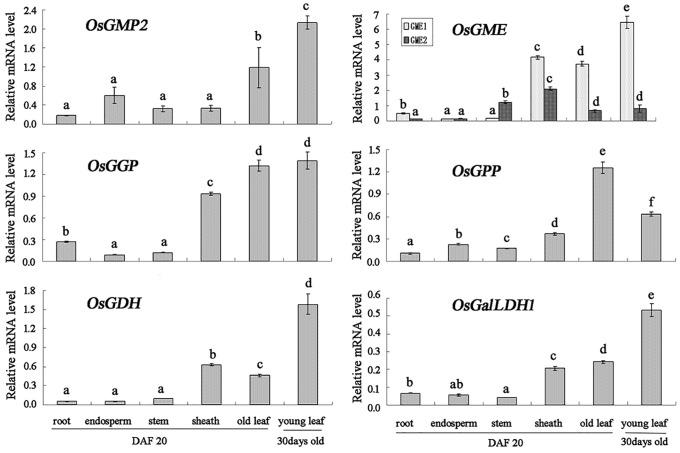
The rice genome contains only one copy each of the OsPMM, OsGGP and OsGPP genes, while there are two copies of the OsGME genes (OsGME1 and OsGME2) and OsGalLDH (OsGalLDH1 and OsGalLDH2) and three copies of OsGMP genes (OsGMP1, OsGMP2 and OsGMP3) [37]. To increase our understanding of rice AsA biosynthesis, we first performed expression profiling of the genes involved in the final six L-galactose pathway steps in various tissues. As there is high identity (more than 90%) among the coding sequences of three OsGMP genes, and also very high identity (98%) between OsGalLDH1 and OsGalLDH2, the expression analysis was only carried out by using the primer according to the OsGMP2 or OsGalLDH1, which might represent the whole of three OsGMP genes or two OsGalLDH genes, respectively. As shown in Fig 3, this analysis demonstrated expression of all tested genes in all tissues; however, their transcription levels were quite low in root, stem and developing endosperm samples compared with their levels in leaves. Moreover, their expression in young leaves from 4-week-old seedlings were much greater than in old leaves from plants 20 days after flowering, implying that the expression of L-galactose pathway was enriched in young rice leaves.
Fig 3. Expression of ascorbate biosynthetic genes in rice roots, endosperm, stems, sheaths, old and young leaves.
All tissues except young leaves were taken from DAF20 (20 days after flowering) plants. The young leaves were taken from 30 day-old plants. Data obtained by real-time RT-PCR were normalized using actin mRNA expression. Data shown are the average of three measurements with standard errors. Different letters indicate statistically significant differences at P<0.05 (ANOVA, Tukey post-host). The GeneBank accession numbers for OsGMP2, OsGME1, OsGME2, OsGGP, OsGPP, OsGDH, and OsGalLDH1 are AK122126, AK069385, AK102348, AK071188, AK071149, AK067039, and AK241565, respectively.
Generation and confirmation of homozygous transgenic rice
Expression cassettes (Fig 2A) containing the target gene or empty vector were transferred into WY3 Japonica rice by Agrobacterium-mediated transformation. More than 20 independent transformants were regenerated for each construct. All T0 transgenic rice plants were confirmed by PCR using primers specific to each target gene (Fig 2B). The results from field studies showed that there was no significant difference of the phenotype such as plant height, tiller number, flower time and seed production between the transgenic plants and their wild type under normal field condition. For each construct, at least three independent lines with homozygous and high-levels of transgene expression were chosen for further analysis. The transgenic plants derived from the empty vector p1300 (empty control) were also selected for further testing.
Transgene expression in rice leaves
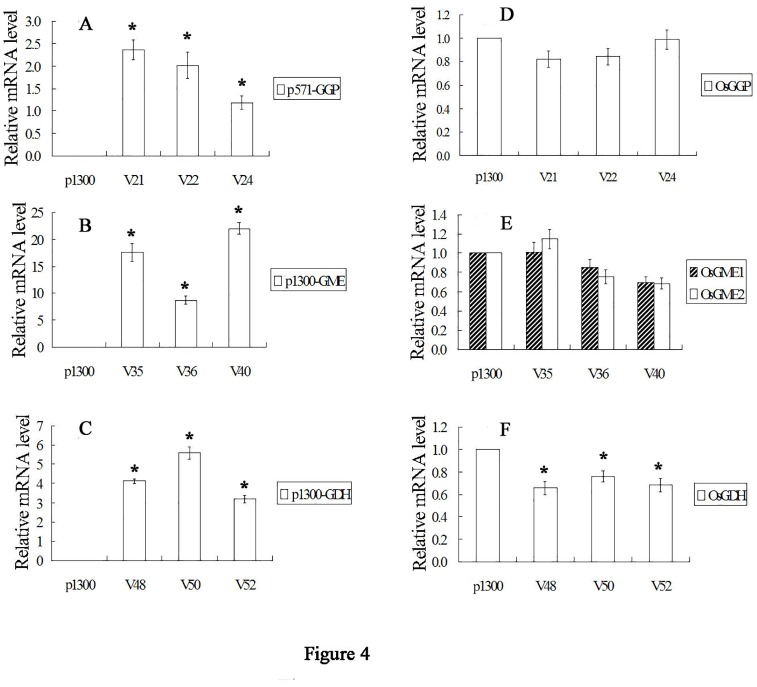
Transgene expression was detected by real-time RT-PCR, and the results of this analysis showed that the foreign transgenes could be expressed at high levels in rice leaves (Fig 4A–4C). The expression of endogenous genes in transgenic rice plants was also analyzed and compared with the empty control (Fig 4D–4F). In AtGDH transgenic rice leaves, endogenous OsGDH expression was slightly lower than empty control (Fig 4F); however, the expression levels of endogenous genes related to AsA biosynthesis in the other transgenic plants were quite similar to those of the empty control (Fig 4D and 4E). The results of this analysis suggested that the transgenes had limited or no effects on L-galactose pathway gene expression.
Fig 4. Relative gene expression in AtGGP, AtGME and AtGDH transgenic lines.
A, AtGGP; B, AtGME; C, AtGDH; D, OsGGP; E, OsGME1 and OsGME2; F, OsGDH. A-C, data obtained by real-time RT-PCR were normalized using actin mRNA expression. E-F, data obtained by real-time RT-RCR were normalized using actin expression and are expressed as a percentage of the control. Asterisk indicates values that are significantly different from those of empty control plants (Tukey’s test, P<0.05).
Effects of exogenous gene overexpression on AsA accumulation
To analyze and compare AsA levels among different transgenic rice plants, T6 generation homozygous transgenic lines and the empty p1300 control were grown simultaneously in the same field under normal culture conditions. The young, fully expanded fresh leaves of 30 day-old plants were collected and used for HPLC measurement of AsA and DHA levels. We used 0.1% oxalic acid as the extraction buffer and found that it provided high AsA stabilization and minimal interaction with the chromatographic system (Fig 5). Compared with the p1300 empty control, AsA levels were significantly increased (up to 2.55-fold) for all six target transgenic lines (Fig 6). Among the transgenic lines, the p571-GGP lines showed the best AsA accumulation, followed by the p1300-GME and p1300-GDH transgenic lines.
Fig 5. HPLC chromatograms of AsA standards (A), and extracts from the leaves of empty control (B) and p571-GGP-derived transgenic rice (C).
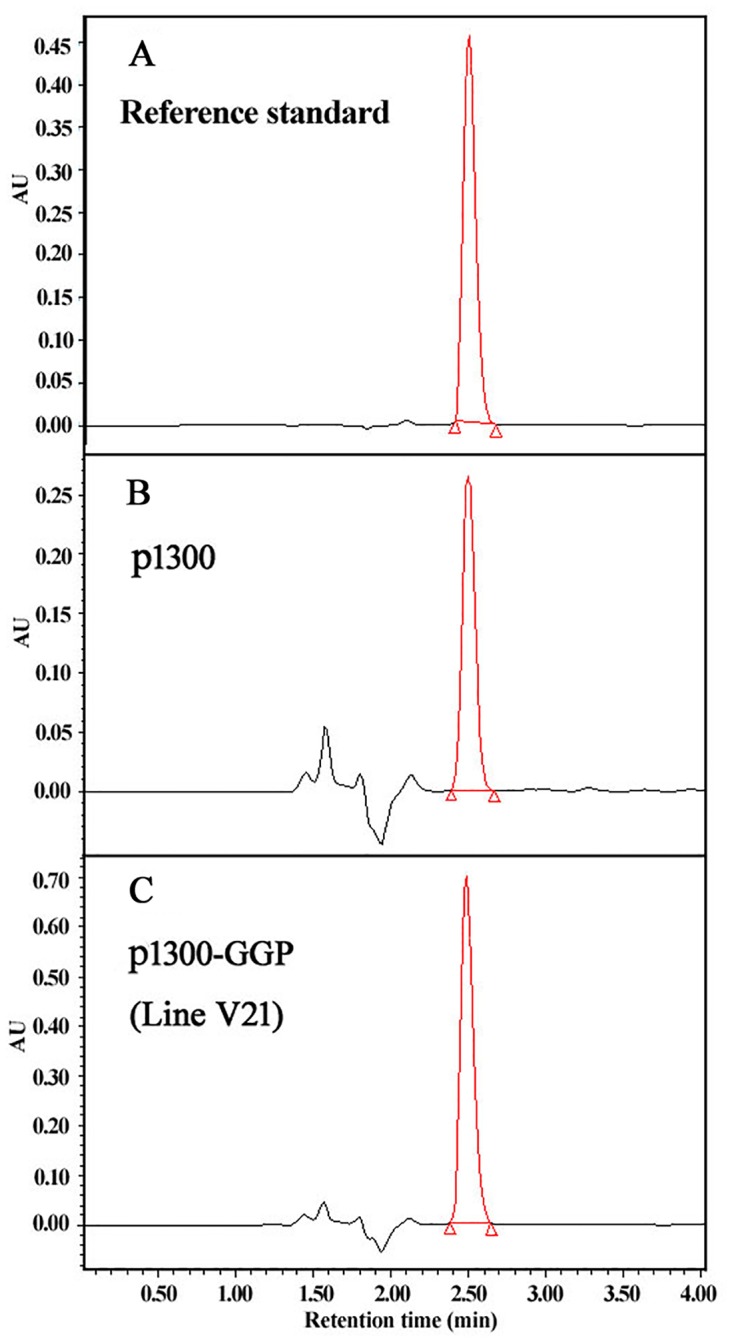
Fig 6. AsA and DHA contents and the ratio of AsA to DHA in each transgenic line.
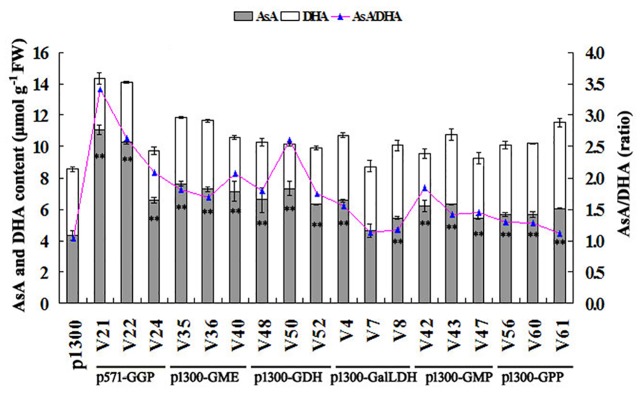
Data are the averages of thirty plants ± standard deviation. Double asterisk indicates values of AsA content are significantly different from those of empty control plants (Tukey’s test, P<0.01).
Effects of exogenous gene overexpression on the ascorbate redox state
As shown in Fig 6, the DHA levels were not significantly altered in the leaves of transgenic rice overexpressing the target genes. As the AsA levels increased significantly in these transgenic leaves, the redox state (AsA/DHA ratio) also increased in the transgenic lines, with the exception of the p1300-GPP lines. The ratio of AsA to DHA was approximately 1.04 in the empty control (p1300) plants, while it was 3.42, 2.64 and 2.08 in the leaves of p1300-GGP transgenic lines V21, V22 and V24, respectively (Fig 6).
Increased AsA content leads to enhanced salt stress tolerance
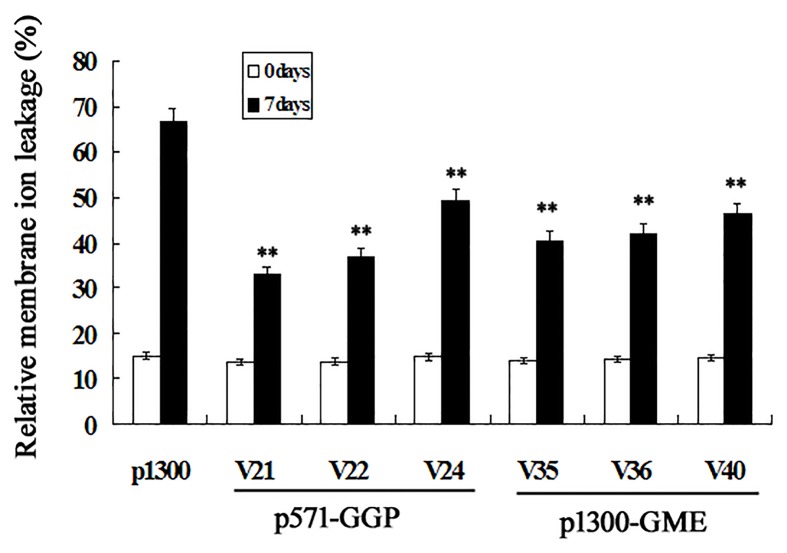
To investigate the effects of increased AsA content on transgenic rice salt tolerance, p1300-GGP and p1300-GME transgenic rice plants which were the two best in AsA accumulation were selected for stress tolerance testing. The 3-week old rice seedlings were transferred to Hoagland’s solution supplemented with 100 mM NaCl for one week. The plant heights, root lengths and fresh weights were measured before and after salt stress, and used to calculate the relative growth rates (Table 1). This analysis showed that the relative plant height and root length growth rates in most of the p1300-GME or p571-GGP transgenic lines tested were significantly greater (p<0.01) than those of the p1300 control plants. The fresh weights of the p1300 control plants were reduced after salt treatment due to wilting under salt stress; in contrast, the extent of wilting was reduced for the p571-GGP and p1300-GME transgenic lines. Consequently, the relative fresh weight growth rates of the p571-GGP and p1300-GME transgenic lines were significantly higher (p<0.01) than those of the p1300 control plants (Table 1). The ion leakage was measured to reflect the level of cellular damage after salt treatment. As shown in Fig 7, relative membrane ion leakage was not significantly different between wild-type and transgenic plants under normal conditions. However, after salt treatment, the relative membrane ion leakage of wild-type plants increased up to 67%, significantly higher (p<0.01) than that of transgenic plants. The results indicated that overexpression of AtGGP or AtGME could significantly improve rice seedling salt tolerance.
Table 1. The relative growth rate of plant height, root length and fresh weight growth rates of transgenic lines transferred to 100 mM NaCl for one week.
| Transgene | Line | Plant height (cm) before stress | Plant height (cm) after stress | The relative growth rate of plant height | Root length (cm) before stress | Root length (cm) after stress | The relative growth rate of root length | Fresh weight (mg/plant) before stress | Fresh weight (mg/plant) after stress | The relative growth rate of fresh weight |
|---|---|---|---|---|---|---|---|---|---|---|
| Empty | p1300 | 15.87±0.21 | 16.17±0.32 | +0.02 | 9.57±0.31 | 10.73±0.25 | +0.12 | 206.33±7.37 | 185.33±5.13 | -0.10 |
| AtGGP | V21 | 16.06±0.25 | 16.40±0.43 | +0.02 | 10.28±0.21 | 11.98±0.15** | +0.17** | 218.38±9.76 | 251.01±4.58** | +0.15** |
| V22 | 16.02±0.14 | 16.78±0.65* | +0.05** | 10.17±0.15 | 11.77±0.32** | +0.15* | 214.34±6.31 | 286.49±4.16** | +0.34** | |
| V24 | 15.47±0.15 | 16.80±0.26** | +0.09** | 10.13±0.15 | 11.37±0.21 | +0.12 | 208.21±8.79 | 241.67±7.81** | +0.16** | |
| AtGME | V35 | 15.41±0.21 | 16.94±0.21** | +0.10** | 9.67±0.40 | 11.62±0.13** | +0.20** | 209.46±7.10 | 235.08±6.03** | +0.12** |
| V36 | 15.20±0.36 | 17.18±0.23** | +0.13** | 9.54±0.17 | 11.53±0.26* | +0.21** | 202.33±8.22 | 247.29±7.21** | +0.22** | |
| V40 | 15.16±0.37 | 17.43±0.26** | +0.15** | 9.23±0.46 | 11.05±0.32 | +0.20** | 193.37±5.03 | 207.67±5.51* | +0.07** |
The data is presented as mean ± SD of at least twelve plants.
* and ** represent significant differences (Tukey’s test) at p≤0.05 and p≤0.01, respectively.
Fig 7. Relative membrane ion leakage in the leaves before and after salt stress treatment.
The data presented are the mean values of twelve plants ± standard deviation. Asterisk indicates values that are significantly different from those of empty control plants (Tukey’s test, P<0.05).
Discussion
In the last decade, great progress has been made in metabolic engineering of the L-galactose pathway; however, it is difficult to systematically compare the contributions of L-galactose pathway genes to AsA accumulation due to the differences in transgenes and plant species across studies.
In this study, six genes involved in the L-galactose pathway were overexpressed in rice. Our analysis showed that the p571-GGP transgenic lines had the highest AsA levels, with a 2.55-fold increase compared with the empty vector control; similar results were reported by Zhou et al. [20]. Transgenic tobacco plants overexpressing kiwifruit GGP showed an approximately 3-fold increase in AsA levels [38]. In 2014, Wang et al. [18] reported that overexpression of tomato GGP in tobacco led to a significant increase in the AsA content and AsA/DHA ratio. Therefore, we can conclude that GGP may be a key rate-limiting step in plant AsA biosynthesis, especially in Arabidopsis, tobacco and rice.
The reactions catalyzed by GMP and GME are located in the upper steps of the end of AsA biosynthesis, and they are also important for cell wall formation and membrane protein modification. Overexpression of tomato GMP in tobacco resulted in a 2-4-fold increase in AsA and improved temperature stress tolerance [17]. Expression of GMP increased AsA levels in tomatoes up to 70% in leaves, 50% in green fruit and 35% in red fruit [39]. In this report, p1300-GMP transgenic lines exhibited a half-fold increase in AsA levels compared with the controls; therefore, GMP does not appear to exert a major metabolic function during rice AsA biosynthesis. Overexpression of both SlGME1 and SlGME2 resulted in a 1.4-fold increase in the AsA contents of tomato leaves and fruits, and improved abiotic stress tolerance [3]. It was reported that the OsGME kinetic parameters seemed to be relatively low for rapid AsA biosynthesis [40]. Overexpression of AtGME in rice showed the next best enhancement of AsA levels. GME catalyzes the double epimerization of the D-mannose moiety at the 3' and 5' carbons to form L-galactose, and the reaction equilibrium is highly biased against AsA biosynthesis [10, 40]. Consequently, GDP-L-galactose formed by GME must be quickly converted to L-galactose-1-phosphate, otherwise abundant amount of GDP-L-galactose needs to accumulate in cells. Consequently, simultaneous overexpression of both GME and GGP may be suitable for strongly pulling the metabolic flux downwards and promoting rice AsA accumulation.
GDH and GalLDH are downstream genes in the L-galactose pathway. Overexpression of GDH in tobacco and Arabidopsis showed little effect on the increase in leaf AsA content [20, 21]. Our results revealed that AtGDH transgenic lines displayed the third best enhancement in AsA levels. Similar to our result, prior OsGalLDH overexpression in rice showed up to a 1.48-fold increase in AsA content [22]. However, Fukunaga et al. [15] reported that the activities of OsGDH and OsGalLDH were sufficient to convert an increased substrate to AsA. Thus, GDH and GalLDH might not be ideal candidates for co-transformation to increase AsA levels. Zhou et al. [20] reported that GPP overexpression in Arabidopsis resulted in an up to 1.5-fold increase in AsA levels compared with wild-type, and our analysis yielded similar results. Furthermore, for transgenic Arabidopsis, simultaneous overexpression of GGP and GPP increased leaf AsA contents up to 4.1-fold [20]. Consequently, it is suggested that the transgenic expression of both enzymes might shed light on the potential for increasing rice AsA content.
In general, plant responses to salinity by occurring in two phases, osmotic phase and ionic phase [41]. might be the best target for single according to the target genes used. nts, the test. which genes was quite similar with that In this study, overexpression of AtGGP or AtGME in rice led to improved osmotic stress tolerance. The growth rate of root and leaf of these transgenic lines were significantly higher than that in the control. The result of ion leakage analysis revealed the lesser cellular damage in transgenic lines than that in empty control. Similar results were obtained for transgenic plants overexpressing genes involved in AsA biosynthesis [2, 3, 18, 23, 42–45]. Therefore, AsA plays a key role in plants as an antioxidant in abiotic stress defense, and plants with higher AsA contents exhibited enhanced tolerance to these stresses.
Taking previous evidence and our present study together, it can be concluded that it is easy to elevate AsA levels in plants via metabolic engineering of AsA biosynthesis; however, the effects differed greatly according to the target genes used. GGP, GDH and GME may be the best three targets, and combining two or three target genes is a better approach for generating transgenic lines with increased AsA accumulation. Consequently, further experimental efforts are necessary to verify whether simultaneous overexpression of GGP and GME, or GGP and GPP would lead to a greater increase in rice AsA levels.
Supporting Information
(DOC)
(DOC)
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This study was supported by the National Key Basic Research Projects (2012CB944803), the National Major Special Project for the Development of Transgenic Organisms (2014ZX08001006-005), the National Science Foundation of China (31471459) to QQL, and the Independent Innovation Fund of Jiangsu Province (CX (13) 5084), China to GYZ. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Noctor G, Foyer CH. Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol. 1998; 49: 249–279. [DOI] [PubMed] [Google Scholar]
- 2. Hemavathi, Upadhyaya CP, Akula N, Kim HS, Jeon JH, Ho OM, et al. Biochemical analysis of enhanced tolerance in transgenic potato plants overexpressing D-galacturonic acid reductase gene in response to various abiotic stresses. Mol Breed. 2011; 28: 105–115. [Google Scholar]
- 3. Zhang C, Liu J, Zhang Y, Cai X, Gong P, Zhang J, et al. Overexpression of SlGMEs leads to ascorbate accumulation with enhanced oxidative stress, cold, and salt tolerance in tomato. Plant Cell Rep. 2011; 30: 389–398. 10.1007/s00299-010-0939-0 [DOI] [PubMed] [Google Scholar]
- 4. Venkatesh J, Upadhyaya CP, Yu JW, Hemavathi A, Kim DH, Strasser RJ, et al. Chlorophyll a fluorescence transient analysis of transgenic potato overexpressing D-galacturonic acid reductase gene for salinity stress tolerance. Hort Environ Biotechnol. 2012; 53: 320–328. [Google Scholar]
- 5. Feng Z, Pang J, Nouchi I, Kobayashi K, Yamakawa T, Zhu J. Apoplastic ascorbate contributes to the differential ozone sensitivity in two varieties of winter wheat under fully open-air field conditions. Environ Pollut. 2010; 158: 3539–3545. 10.1016/j.envpol.2010.08.019 [DOI] [PubMed] [Google Scholar]
- 6. Talla S, Riazunnisa K, Padmavathi L, Sunil B, Rajsheel P, Raghavendra AS. Ascorbic acid is a key participant during the interactions between chloroplasts and mitochondria to optimize photosynthesis and protect against photoinhibition. J Biosci. 2011; 36: 163–173. [DOI] [PubMed] [Google Scholar]
- 7. De Tullio MC, Arrigoni O. Hopes, disillusions and more hopes from vitamin C. Cell Mol Life Sci, 2004; 61: 209–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Debolt S, Melino V, Ford CM. Ascorbate as a biosynthetic precursor in plants. Ann Bot. 2007; 99: 3–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wheeler GL, Jones MA, Smirnoff N. The biosynthetic pathway of vitamin C in higher plants. Nature. 1998; 393: 365–369. [DOI] [PubMed] [Google Scholar]
- 10. Wolucka BA, Van Montagu M. GDP-mannose 3′, 5′-epimerase forms GDP-L-gulose, a putative intermediate for the de novo biosynthesis of vitamin C in plants. J Biol Chem. 2003; 278: 47483–47490. [DOI] [PubMed] [Google Scholar]
- 11. Agius F, González-Lamothe R, Caballero JL, Muñoz-Blanco J, Botella MA, Valpuesta V. Engineering increased vitamin C levels in plants by overexpression of a D-galacturonic acid reductase. Nat Biotechno. 2003; 21: 177–181. [DOI] [PubMed] [Google Scholar]
- 12. Jain AK, Nessler CL. Metabolic engineering of an alternative pathway for ascorbic acid biosynthesis in plants. Mol Breed. 2000; 6: 73–78. [Google Scholar]
- 13. Lorence A, Chevone BI, Mendes P, Nessler CL. myo-Inositol oxygenase offers a possible entry point into plant ascorbate biosynthesis. Plant Physiol. 2004; 134: 1200–1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Imai T, Ban Y, Yamamoto T, Moriguchi T. Ectopic overexpression of peach GDP-D-mannose pyrophosphorylase and GDP-D-mannose-3’,5’-epimerase in transgenic tobacco. Plant Cell Tiss Organ Cult. 2012; 111:1–13 [Google Scholar]
- 15. Fukunaga K, Fujikawa Y, Esaka M. Light regulation of ascorbic acid biosynthesis in rice via light responsive cis-elements in genes encoding ascorbic acid biosynthetic enzymes. Biosci Biotechnol Biochem. 2010; 74: 888–891. [DOI] [PubMed] [Google Scholar]
- 16. Badejo AA, Tanaka N, Esaka M. Analysis of GDP-D-mannose pyrophosphorylase gene promoter from acerola (Malpighia glabra) and increase in ascorbate content of transgenic tobacco expressing the acerola gene. Plant Cell Physiol. 2008; 49: 126–132. [DOI] [PubMed] [Google Scholar]
- 17. Wang HS, Yu C, Zhu ZJ, Yu XC. Overexpression in tobacco of a tomato GMPase gene improves tolerance to both low and high temperature stress by enhancing antioxidation capacity. Plant Cell Rep. 2011; 30: 1029–1040. 10.1007/s00299-011-1009-y [DOI] [PubMed] [Google Scholar]
- 18. Wang L, Meng X, Yang D, Ma N, Wang G, Meng Q. Overexpression of tomato GDP-l-galactose phosphorylase gene in tobacco improves tolerance to chilling stress. Plant Cell Rep. 2014; 33: 1441–1451. 10.1007/s00299-014-1627-2 [DOI] [PubMed] [Google Scholar]
- 19. Bulley S, Wright M, Rommens C, Yan H, Rassam M, Lin-Wang K, et al. Enhancing ascorbate in fruits and tubers through over-expression of the L-galactose pathway gene GDP-L-galactose phosphorylase. Plant Biotechnol. 2012; 10: 390–397. 10.1111/j.1467-7652.2011.00668.x [DOI] [PubMed] [Google Scholar]
- 20. Zhou Y, Tao QC, Wang ZN, Fan R, Li Y, Sun XF, et al. Engineering ascorbic acid biosynthetic pathway in Arabidopsis leaves by single and double gene transformation. Biol Plant. 2012; 56: 451–457. [Google Scholar]
- 21. Gatzek S, Wheeler GL, Smirnoff N. Antisense suppression of l-galactose dehydrogenase in Arabidopsis thaliana provides evidence for its role in ascorbate synthesis and reveals light modulated l-galactose synthesis. Plant J. 2002; 30: 541–553. [DOI] [PubMed] [Google Scholar]
- 22. Liu Y, Yu L, Wang R. Level of ascorbic acid in transgenic rice for L-galactono-1, 4-lactone dehydrogenase overexpressing or suppressed is associated with plant growth and seed set. Acta Physiol Plant. 2011; 33: 1353–1363. [Google Scholar]
- 23. Liu W, An HM, Yang M. Overexpression of Rosa roxburghii l-galactono-1, 4-lactone dehydrogenase in tobacco plant enhances ascorbate accumulation and abiotic stress tolerance. Acta Physiol Plant. 2013; 35: 1617–1624. [Google Scholar]
- 24. Chao YY, Hong CY, Kao CH. The decline in ascorbic acid content is associated with cadmium toxicity of rice seedlings. Plant Physiol Biochem. 2010; 48: 374–381. 10.1016/j.plaphy.2010.01.009 [DOI] [PubMed] [Google Scholar]
- 25. Yamane K, Rahman MS, Kawasaki M, Taniguchi M, Miyake H. Pretreatment with antioxidants decreases the effects of salt stress on chloroplast ultrastructure in rice leaf segments (Oryza sativa L.). Plant Prod Sci. 2004; 7: 292–300. [Google Scholar]
- 26. Guo Z, Tan H, Zhu Z, Lu S, Zhou B. Effect of intermediates on ascorbic acid and oxalate biosynthesis of rice and in relation to its stress resistance. Plant Physiol Biochem. 2005; 43: 955–962. [DOI] [PubMed] [Google Scholar]
- 27. Frei M, Wissuwa M, Pariasca-Tanaka J, Chen CP, Südekum KH, Kohno Y. Leaf ascorbic acid level—Is it really important for ozone tolerance in rice? Plant Physiol Biochem. 2012; 59: 63–70. 10.1016/j.plaphy.2012.02.015 [DOI] [PubMed] [Google Scholar]
- 28. Barth C, Moeder W, Klessig DF, Conklin PL. The timing of senescence and response to pathogens is altered in the ascorbate-deficient Arabidopsis mutant vitamin c-1. Plant Physiol. 2004; 134: 1784–1792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. López-Carbonell M, Munné-Bosch S, Alegre L. The ascorbate-deficient vtc-1 Arabidopsis mutant shows altered ABA accumulation in leaves and chloroplasts. J Plant Growth Regul. 2006; 25: 137–144. [Google Scholar]
- 30. Noctor G. Metabolic signalling in defence and stress: the central roles of soluble redox couples. Plant Cell Environ. 2006; 29: 409–425. [DOI] [PubMed] [Google Scholar]
- 31. Kwon SY, Choi SM, Ahn YO, Lee HS, Lee HB, Park YM, et al. Enhanced stress-tolerance of transgenic tobacco plants expressing a human dehydroascorbate reductase gene. J Plant Physiol. 2003; 160: 347–353. [DOI] [PubMed] [Google Scholar]
- 32. Wang Z, Xiao Y, Chen W, Tang K, Zhang L. Increased vitamin C content accompanied by an enhanced recycling pathway confers oxidative stress tolerance in Arabidopsis. J Integr Plant Biol. 2010; 52: 400–409 10.1111/j.1744-7909.2010.00921.x [DOI] [PubMed] [Google Scholar]
- 33. Liu QQ, Zhang JL, Wang ZY, Hong MM, Gu MH. A highly efficient transformation system mediated by Agrobacterium tumefaciens in rice (Oryza sativa L.). Acta Phytophysiol Sin. 1998; 24: 259–271. [Google Scholar]
- 34. Murray MG, Thompson WF. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980; 8: 4321–4325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Zhang GY, Liu RR, Xu G, Zhang P, Li Y, Tang KX, et al. Increased α-tocotrienol content in seeds of transgenic rice overexpressing Arabidopsis γ-tocopherol methyltransferase. Transgenic Res. 2013; 22: 89–99. 10.1007/s11248-012-9630-2 [DOI] [PubMed] [Google Scholar]
- 36. Wang LY, Li D, Deng YS, Lv W, Meng QW. Antisense-mediated depletion of tomato GDP-l-galactose phosphorylase increases susceptibility to chilling stress. J Plant Physiol. 2013; 170: 303–314 10.1016/j.jplph.2012.10.015 [DOI] [PubMed] [Google Scholar]
- 37. Höller S, Hajirezaei MR, von Wirén N, Frei M. Ascorbate metabolism in rice genotypes differing in zinc efficiency. Planta. 2014; 239: 367–379 10.1007/s00425-013-1978-x [DOI] [PubMed] [Google Scholar]
- 38. Laing WA, Wright MA, Cooney J, Bulley SM. The missing step of the L-galactose pathway of ascorbate biosynthesis in plants, an L-galactose guanyltransferase, increases leaf ascorbate content. Proc Natl Acad Sci USA. 2007; 104: 9534–9539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Cronje C, George GM, Fernie AR, Bekker J, Kossmann J, Bauer R. Manipulation of L-ascorbic acid biosynthesis pathways in Solanum lycopersicum: elevated GDP-mannose pyrophosphorylase activity enhances L-ascorbate levels in red fruit. Planta. 2012; 235: 553–564. 10.1007/s00425-011-1525-6 [DOI] [PubMed] [Google Scholar]
- 40. Watanabe K, Suzuki K, Kitamura S. Characterization of a GDP-d-mannose 3‘, 5’-epimerase from rice. Phytochemistry. 2006; 67: 338–346. [DOI] [PubMed] [Google Scholar]
- 41. Munns R, Tester M. Mechanisms of salinity tolerance. Annu Rev Plant Biol. 2008; 59: 651–681 10.1146/annurev.arplant.59.032607.092911 [DOI] [PubMed] [Google Scholar]
- 42. Eltayeb AE, Yamamoto S, Habora MEE, Yin L, Tsujimoto H, Tanaka K. Transgenic potato overexpressing Arabidopsis cytosolic AtDHAR1 showed higher tolerance to herbicide, drought and salt stresses. Breeding Sci. 2011; 61: 3–10. [Google Scholar]
- 43. Lisko KA, Torres R, Harris RS, Belisle M, Vaughan MM, Jullian B, et al. (2013) Elevating vitamin C content via overexpression of myo-inositol oxygenase and L-gulono-1, 4-lactone oxidase in Arabidopsis leads to enhanced biomass and tolerance to abiotic stresses. In Vitro Cell Dev Biol-Plant. 2013; 49: 643–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Lim MY, Pulla RK, Park JM, Harn CH, Jeong BR. Over-expression of L-gulono-γ-lactone oxidase (GLOase) gene leads to ascorbate accumulation with enhanced abiotic stress tolerance in tomato. In Vitro Cell Dev Biol-Plant. 2012; 48: 453–461. [Google Scholar]
- 45. Hemavathi, Upadhyaya CP, Young KE, Akula N, Kim HS, Oh OM, et al. Over-expression of strawberry D-galacturonic acid reductase in potato leads to accumulation of vitamin C with enhanced abiotic stress tolerance. Plant Sci. 2009; 177: 659–667. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOC)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.