Abstract
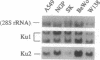
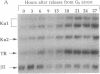
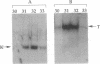
We have previously described purification and characterization of a nuclear protein, TREF, which interacts specifically with the transcriptional control element, TRA, of the human transferrin receptor (TR) gene. In this report we show that TREF can be separated into two functionally distinct DNA-binding activities. The first DNA-binding activity (TRAC) is highly specific for the 8-bp element TRA and the related Escherichia coli cAMP receptor binding site. This motif is homologous to the phorbol 12-tetradecanoate 13-acetate- and cAMP-responsive elements of eukaryotic genes and the regulatory proximal sequence elements of the U1 small nuclear RNA gene and is also present in the promoter of the Drosophila melanogaster yolk protein factor 1 gene. In striking contrast, the second activity exhibits high affinity for the ends of double-stranded DNA in a sequence-unspecific manner and is attributable to the heterodimeric Ku autoantigen. Notably, transcription of Ku is induced during mid-late G0/G1 with kinetics similar to the TR gene. Ku is a highly abundant nuclear protein possessing nonspecific affinity for the ends of DNA, whose biological role remains to be elucidated. A transcriptional role for this protein has been proposed, however, on the basis of studies attributing DNA sequence-specific binding activity, notably for TRA-like sequences described above, directly to the Ku heterodimer. The observation that Ku-mediated nonspecific DNA-binding activity copurifies with the TRA-specific activity, TRAC, clearly has implications for these and related studies. The unusual properties of TRAC activity and its relationship, if any, with the enigmatic Ku protein, are discussed.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Casey J. L., Di Jeso B., Rao K. K., Rouault T. A., Klausner R. D., Harford J. B. Deletional analysis of the promoter region of the human transferrin receptor gene. Nucleic Acids Res. 1988 Jan 25;16(2):629–646. doi: 10.1093/nar/16.2.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan J. Y., Lerman M. I., Prabhakar B. S., Isozaki O., Santisteban P., Kuppers R. C., Oates E. L., Notkins A. L., Kohn L. D. Cloning and characterization of a cDNA that encodes a 70-kDa novel human thyroid autoantigen. J Biol Chem. 1989 Mar 5;264(7):3651–3654. [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- De Lorenzo V., Herrero M., Giovannini F., Neilands J. B. Fur (ferric uptake regulation) protein and CAP (catabolite-activator protein) modulate transcription of fur gene in Escherichia coli. Eur J Biochem. 1988 May 2;173(3):537–546. doi: 10.1111/j.1432-1033.1988.tb14032.x. [DOI] [PubMed] [Google Scholar]
- Depper J. M., Leonard W. J., Drogula C., Krönke M., Waldmann T. A., Greene W. C. Interleukin 2 (IL-2) augments transcription of the IL-2 receptor gene. Proc Natl Acad Sci U S A. 1985 Jun;82(12):4230–4234. doi: 10.1073/pnas.82.12.4230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gottlieb T. M., Jackson S. P. The DNA-dependent protein kinase: requirement for DNA ends and association with Ku antigen. Cell. 1993 Jan 15;72(1):131–142. doi: 10.1016/0092-8674(93)90057-w. [DOI] [PubMed] [Google Scholar]
- Gunderson S. I., Knuth M. W., Burgess R. R. The human U1 snRNA promoter correctly initiates transcription in vitro and is activated by PSE1. Genes Dev. 1990 Dec;4(12A):2048–2060. doi: 10.1101/gad.4.12a.2048. [DOI] [PubMed] [Google Scholar]
- Hai T., Curran T. Cross-family dimerization of transcription factors Fos/Jun and ATF/CREB alters DNA binding specificity. Proc Natl Acad Sci U S A. 1991 May 1;88(9):3720–3724. doi: 10.1073/pnas.88.9.3720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton T. A. Regulation of transferrin receptor expression in concanavalin A stimulated and Gross virus transformed rat lymphoblasts. J Cell Physiol. 1982 Oct;113(1):40–46. doi: 10.1002/jcp.1041130109. [DOI] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III in DNA sequence analysis. Methods Enzymol. 1987;155:156–165. doi: 10.1016/0076-6879(87)55014-5. [DOI] [PubMed] [Google Scholar]
- Knuth M. W., Gunderson S. I., Thompson N. E., Strasheim L. A., Burgess R. R. Purification and characterization of proximal sequence element-binding protein 1, a transcription activating protein related to Ku and TREF that binds the proximal sequence element of the human U1 promoter. J Biol Chem. 1990 Oct 15;265(29):17911–17920. [PubMed] [Google Scholar]
- Larrick J. W., Cresswell P. Modulation of cell surface iron transferrin receptors by cellular density and state of activation. J Supramol Struct. 1979;11(4):579–586. doi: 10.1002/jss.400110415. [DOI] [PubMed] [Google Scholar]
- Lathe R. Synthetic oligonucleotide probes deduced from amino acid sequence data. Theoretical and practical considerations. J Mol Biol. 1985 May 5;183(1):1–12. doi: 10.1016/0022-2836(85)90276-1. [DOI] [PubMed] [Google Scholar]
- Lesley J. F., Schulte R. J. Inhibition of cell growth by monoclonal anti-transferrin receptor antibodies. Mol Cell Biol. 1985 Aug;5(8):1814–1821. doi: 10.1128/mcb.5.8.1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May G., Sutton C., Gould H. Purification and characterization of Ku-2, an octamer-binding protein related to the autoantigen Ku. J Biol Chem. 1991 Feb 15;266(5):3052–3059. [PubMed] [Google Scholar]
- Messier H., Fuller T., Mangal S., Brickner H., Igarashi S., Gaikwad J., Fotedar R., Fotedar A. p70 lupus autoantigen binds the enhancer of the T-cell receptor beta-chain gene. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2685–2689. doi: 10.1073/pnas.90.7.2685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mimori T., Ohosone Y., Hama N., Suwa A., Akizuki M., Homma M., Griffith A. J., Hardin J. A. Isolation and characterization of cDNA encoding the 80-kDa subunit protein of the human autoantigen Ku (p70/p80) recognized by autoantibodies from patients with scleroderma-polymyositis overlap syndrome. Proc Natl Acad Sci U S A. 1990 Mar;87(5):1777–1781. doi: 10.1073/pnas.87.5.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miskimins W. K. Interaction of multiple factors with a GC-rich element within the mitogen responsive region of the human transferrin receptor gene. J Cell Biochem. 1992 Aug;49(4):349–356. doi: 10.1002/jcb.240490405. [DOI] [PubMed] [Google Scholar]
- Miskimins W. K., McClelland A., Roberts M. P., Ruddle F. H. Cell proliferation and expression of the transferrin receptor gene: promoter sequence homologies and protein interactions. J Cell Biol. 1986 Nov;103(5):1781–1788. doi: 10.1083/jcb.103.5.1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitsis P. G., Wensink P. C. Identification of yolk protein factor 1, a sequence-specific DNA-binding protein from Drosophila melanogaster. J Biol Chem. 1989 Mar 25;264(9):5188–5194. [PubMed] [Google Scholar]
- Murphy J. T., Skuzeski J. T., Lund E., Steinberg T. H., Burgess R. R., Dahlberg J. E. Functional elements of the human U1 RNA promoter. Identification of five separate regions required for efficient transcription and template competition. J Biol Chem. 1987 Feb 5;262(4):1795–1803. [PubMed] [Google Scholar]
- Okayama H., Berg P. A cDNA cloning vector that permits expression of cDNA inserts in mammalian cells. Mol Cell Biol. 1983 Feb;3(2):280–289. doi: 10.1128/mcb.3.2.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouyang Q., Bommakanti M., Miskimins W. K. A mitogen-responsive promoter region that is synergistically activated through multiple signalling pathways. Mol Cell Biol. 1993 Mar;13(3):1796–1804. doi: 10.1128/mcb.13.3.1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owen D., Kühn L. C. Noncoding 3' sequences of the transferrin receptor gene are required for mRNA regulation by iron. EMBO J. 1987 May;6(5):1287–1293. doi: 10.1002/j.1460-2075.1987.tb02366.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauza C. D., Bleil J. D., Lennox E. S. The control of transferrin receptor synthesis in mitogen-stimulated human lymphocytes. Exp Cell Res. 1984 Oct;154(2):510–520. doi: 10.1016/0014-4827(84)90175-7. [DOI] [PubMed] [Google Scholar]
- Raveche E. S., Steinberg A. D., DeFranco A. L., Tjio J. H. Cell cycle analysis of lymphocyte activation in normal and autoimmune strains of mice. J Immunol. 1982 Sep;129(3):1219–1226. [PubMed] [Google Scholar]
- Reed J. C., Alpers J. D., Nowell P. C., Hoover R. G. Sequential expression of protooncogenes during lectin-stimulated mitogenesis of normal human lymphocytes. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3982–3986. doi: 10.1073/pnas.83.11.3982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves W. H., Sthoeger Z. M. Molecular cloning of cDNA encoding the p70 (Ku) lupus autoantigen. J Biol Chem. 1989 Mar 25;264(9):5047–5052. [PubMed] [Google Scholar]
- Roberts M. R., Miskimins W. K., Ruddle F. H. Nuclear proteins TREF1 and TREF2 bind to the transcriptional control element of the transferrin receptor gene and appear to be associated as a heterodimer. Cell Regul. 1989 Nov;1(1):151–164. doi: 10.1091/mbc.1.1.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stuiver M. H., Coenjaerts F. E., van der Vliet P. C. The autoantigen Ku is indistinguishable from NF IV, a protein forming multimeric protein-DNA complexes. J Exp Med. 1990 Oct 1;172(4):1049–1054. doi: 10.1084/jem.172.4.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trowbridge I. S., Lopez F. Monoclonal antibody to transferrin receptor blocks transferrin binding and inhibits human tumor cell growth in vitro. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1175–1179. doi: 10.1073/pnas.79.4.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trowbridge I. S., Omary M. B. Human cell surface glycoprotein related to cell proliferation is the receptor for transferrin. Proc Natl Acad Sci U S A. 1981 May;78(5):3039–3043. doi: 10.1073/pnas.78.5.3039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Violette S. M., Shashikant C. S., Salbaum J. M., Belting H. G., Wang J. C., Ruddle F. H. Repression of the beta-amyloid gene in a Hox-3.1-producing cell line. Proc Natl Acad Sci U S A. 1992 May 1;89(9):3805–3809. doi: 10.1073/pnas.89.9.3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaneva M., Wen J., Ayala A., Cook R. cDNA-derived amino acid sequence of the 86-kDa subunit of the Ku antigen. J Biol Chem. 1989 Aug 15;264(23):13407–13411. [PubMed] [Google Scholar]
- de Vries E., van Driel W., Bergsma W. G., Arnberg A. C., van der Vliet P. C. HeLa nuclear protein recognizing DNA termini and translocating on DNA forming a regular DNA-multimeric protein complex. J Mol Biol. 1989 Jul 5;208(1):65–78. doi: 10.1016/0022-2836(89)90088-0. [DOI] [PubMed] [Google Scholar]