Abstract
Hormones are involved in a plethora of processes including development and growth, metabolism, mood, and immune responses. These essential functions are dependent on the ability of the hormone to access its target tissue. In the case of endocrine hormones that are transported through the blood, this often means that the endothelium must be crossed. Many studies have shown that the concentrations of hormones and nutrients in blood can be very different from those surrounding the cells on the tissue side of the blood vessel endothelium, suggesting that transport across this barrier can be rate limiting for hormone action. This transport can be regulated by altering the surface area of the blood vessel available for diffusion through to the underlying tissue or by the permeability of the endothelium. Many hormones are known to directly or indirectly affect the endothelial barrier, thus affecting their own distribution to their target tissues. Dysfunction of the endothelial barrier is found in many diseases, particularly those associated with the metabolic syndrome. The interrelatedness of hormones may help to explain why the cluster of diseases in the metabolic syndrome occur together so frequently and suggests that treating the endothelium may ameliorate defects in more than one disease. Here, we review the structure and function of the endothelium, its contribution to the function of hormones, and its involvement in disease.
Hormones are essential for efficient dissemination of nutrients to the correct tissues, general metabolism, maintaining adequate blood pressure, and many other processes. There are numerous hormones in the body responsible for many essential functions. These can include sex development and characteristics (testosterone, estrogen), metabolism and nutrient storage (insulin), blood pressure (renin-angiotensin axis), the fight or flight response (epinephrine, norepinephrine), control of mood and sleep (serotonin), as well as many others. Hormones can be secreted from a variety of tissues, including the brain, and can act locally in a paracrine fashion or throughout the body. Insulin, although released from the pancreas, affects skeletal muscle, adipose tissue, and liver, among others. Similarly, angiotensin II can have vascular, neural, adrenal, and renal effects. Therefore, access of each hormone to its various target tissues must be efficiently regulated. Often to identify health problems, blood samples are taken to measure the concentration of hormones and nutrients in the blood, yet plasma is not representative of the environment at the cell surface. The physical and functional barrier separating blood from many organs and cells is the endothelium, which lines blood vessels throughout the body. This barrier regulates the access of hormones and nutrients to underlying target tissues.
The endothelium can be modified in many ways to increase or decrease access to the underlying tissue. Many hormones are themselves able to induce changes in the endothelial barrier to alter their own access. To investigate the ability of a hormone to improve its own access, we must first understand that changes can be made to modify the endothelial transport and access to the interstitium. We will provide evidence that a barrier exists, review the structure of the endothelium, and then investigate transport of hormones across the endothelium and functions of the endothelium that modify access to essential organs.
Evidence of a Barrier
Many studies investigate cellular responsiveness to hormones to determine the presence of receptors and signaling cascades, but cell reaction to the hormone in vitro may not be indicative of what happens physiologically in vivo. For example, glucose and amino acid concentrations in muscle and adipose tissue were found to be substantially different from circulating levels using microdialysis (54). Thus there is a barrier preventing the equilibration of the plasma with the interstitial space. Similar results have been found for other hormones; angiotensin, involved in regulation of blood pressure and fluid volume, is 1,000-fold higher in the renal interstitium than in plasma (81), suggesting a role as a paracrine rather than an endocrine hormone.
Studies have shown that the concentration of insulin at the cell surface is very different from that in plasma (1, 15, 82). This difference could contribute to impaired insulin response independent of cellular effects. This insulin gradient has been confirmed using microdialysis (40, 82) as well as by direct interstitial access (7) and lymph measurements (1, 15). Based on insulin extraction across a limb, transendothelial transport is more likely to limit interstitial concentrations than hormone uptake or disposal (93). This disequilibrium between the plasma and interstitium is suggestive of a barrier preventing equilibration between the two compartments.
Although this barrier can affect the ultimate concentration reached at the cell surface in relation to the plasma levels, it can also have a temporal effect, delaying the increase in hormone concentration, a representation of which is shown in FIGURE 1. It was noted that insulin-mediated glucose uptake lagged behind the increase in plasma insulin but matched well with insulin receptor kinase activation in muscle (35). In cultured skeletal muscle cells, insulin-mediated glucose uptake occurs almost immediately (47, 74), and the uptake rate is constant during exposure. Thus the time delay seen in vivo (35) suggests an intrinsic delay in insulin access to the myocyte. Glucose disposal in dogs exposed to venous insulin infusion corresponded with the time for lymph insulin increase, yet the increase in arterial insulin was much quicker (57). This temporal relationship confirms the concept that the time required for insulin to reach the interstitial space is the limiting factor for insulin-mediated glucose uptake: only once insulin is present at the muscle cell surface can it bind to receptors to cause glucose uptake. The correlation between insulin concentration and glucose uptake is strongest with interstitial (lymph) insulin concentrations (r = 0.92; P < 0.001) rather than vein (r = 0.77; P < 0.001) or arterial (r = 0.19; P > 0.2) concentrations (15). Therefore, the concentration of insulin at the surface of the target cell, rather than in the blood, is the best predictor for the rate of insulin-mediated glucose uptake. We have shown that diffusion throughout the interstitium is rapid in healthy tissue (15); as such, it seems that the endothelium provides the main barrier to equilibration with the plasma (4). Sampling the fluid at the cell surface is thus essential to understanding the ability of the cell to respond to its environment, and the ability of hormones to access the target tissue is essential for their action.
FIGURE 1. Increase of the hormone in plasma.
Once insulin is infused at time 0, the increase of the hormone in plasma is very rapid (time shown in minutes); however, more time is required for the increased concentration on the other side of the endothelium, the interstitial space. The interstitial concentration temporally matches the hormones’ measured effect, glucose uptake.
The Endothelium as a Structural Barrier
The endothelium lines the blood vessels throughout the body, from the larger arteries and veins down to the small capillaries that branch through organs. Transport of nutrients and hormones across this barrier is essential for the appropriate functioning of the body. Originally, the capillary endothelium was thought to be an inert semipermeable barrier; however, the concept of a purely physical endothelial barrier has been replaced by our understanding of a more complex and functional one.
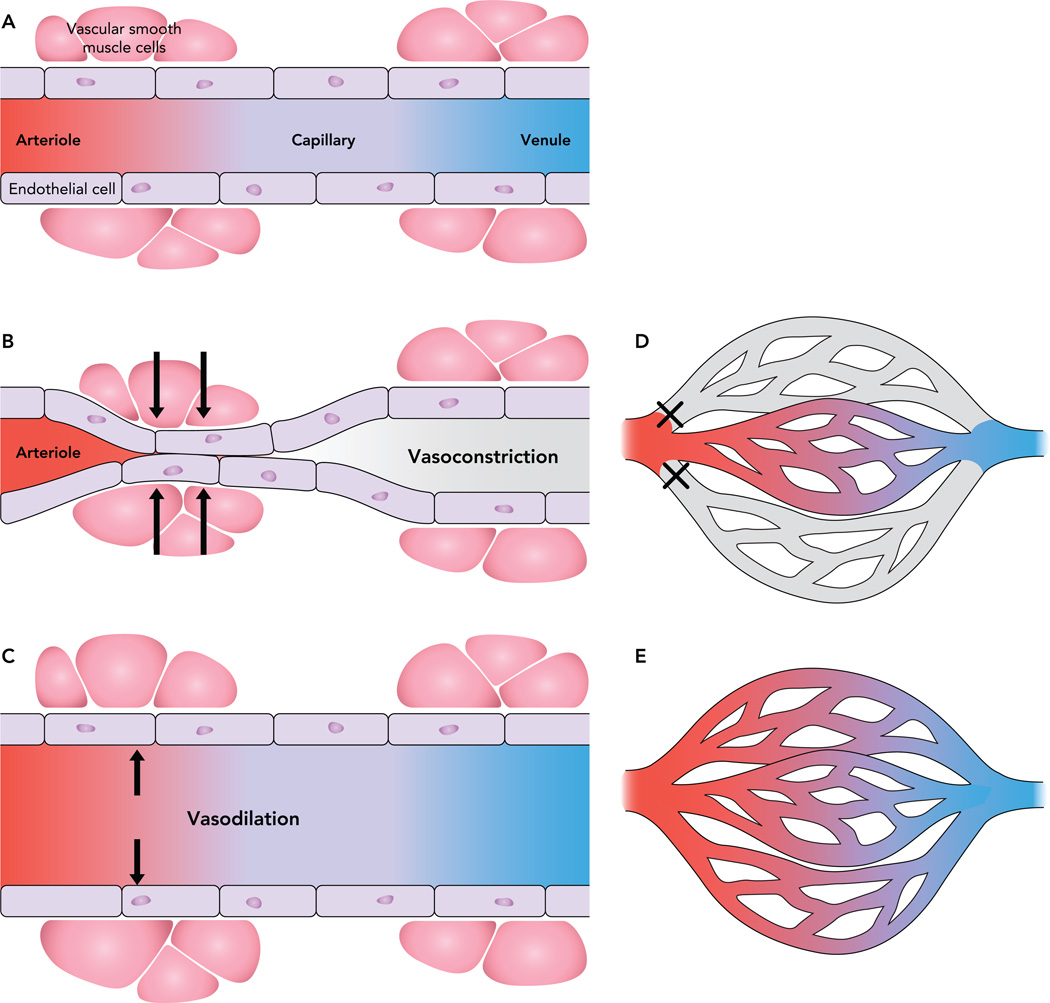
The endothelium consists of endothelial cells on the luminal side of the blood vessel, with an underlying layer of smooth muscle cells (FIGURE 2). Isolated arterioles show that endothelial cells can prevent small water-soluble molecules diffusing from the luminal surface to the smooth muscle cell layer (52), whereas lipid-soluble compounds diffuse rapidly from either side of the arteriole, suggesting a selectivity of the endothelial cell (52). Early work used the molecular sieving ability of capillaries to estimate pore size (70). Studies that showed a change in the resistance of the capillary wall due to plasma volume expansion concluded that the pores can stretch (78). Although the larger arteries and arterioles contain vascular smooth muscle cells, much of the exchange of molecules occurs across the capillary wall where there are usually no vascular smooth muscle cells. Some exchange can also occur at the postcapillary venules, which often do not possess smooth muscle cells.
FIGURE 2. Structure of typical blood vessel.
The endothelial cell lines the blood vessel with an underlying basement membrane, and vascular smooth muscle cells are present on arteries and larger venules. Capillaries do not contain vascular smooth muscle cells and are the major site of exchange for nutrients, hormones, gases, and macromolecules. Any molecule for exchange must 1) move through the blood, 2) pass through the glycocalyx, 3) cross the endothelial barrier, and 4) diffuse through the interstitial space to the target tissue where it can 5) interact with the target tissue.
The glycocalyx is a permeable endothelial surface layer consisting of glycoproteins that extend into the blood vessel. The glycocalyx is involved in the regulation of vascular permeability, mediation of shear stress, and decreasing adhesion [reviewed by Becker et al. (5)], and can regulate the ability of various molecules to access the endothelium based on size and charge (95), thus limiting access to the endothelial cells.
It has long been known that the structure of the endothelium can vary among tissues (34). Depending on the tissue, the junctions between the endothelial cells can be tight or open to large pores. Because of this, the ability of hormones or other nutrients to access the target tissue will depend on the structure of the endothelium within that particular tissue (reviewed in Refs. 2, 56). The skeletal muscle, heart, lung, and brain each have continuous endothelium, where there are tight junctions between cells, and most transport must occur through the cell. Even within this class, there are differences. The blood-brain barrier is a highly specialized endothelial barrier with very tight control, as reviewed by Deli et al. (24). The pancreas and intestine are examples of tissue that contain fenestrated endothelium, with pores allowing more access to the underlying tissue than would be permitted by continuous endothelium. However, the liver, spleen, and bone marrow have discontinuous endothelium with large holes, which allow rapid equilibration of plasma with the underlying tissue. This exposure of the liver to plasma changes may explain its earlier susceptibility to insulin resistance, for example (44). The role of the endothelium in hormone transport varies between organs and can depend on the physical ability of the endothelium to block transport.
The Endothelium as a Functional Barrier
The location of the endothelium is such that it is often the first structure to be exposed to any changes in blood and is, therefore, the first to be able to respond to such changes. Although the endothelium can function as a physical barrier, endocytosis and transcytosis (66, 79) have been observed in endothelial cells, suggesting a more active capacity of the endothelium to modulate access to the underlying tissue. In that sense, the endothelium can act as a “gateway” by which hormones or other signals can reach target cells. Interestingly, many hormones can have direct effects on the endothelium and as such may be able to modify the endothelium to alter their effects.
Hormones Can Increase Surface Area for Diffusion
Increasing the surface area for diffusion can increase access to underlying cells; an increased area for exchange to underlying tissue allows more rapid equilibration.
Some hormones can regulate the formation of new blood vessels, which will increase the surface area available for diffusion. These include angiotensin II, endothelin, growth hormone, insulin, and vasopressin, among others (18); hormones can induce angiogenesis either directly through actions on endothelial cells or indirectly by regulating proangiogenic factors like vascular endothelial growth factor [reviewed by Clapp et al. (18)]. There are also anti-angiogenic hormones, including somatostatin and ghrelin.
Aside from this chronic effect on surface area, acute manipulations of the vascular system can alter surface area much more dynamically.
Vasodilation/Vasoconstriction
As shown in FIGURE 2, the general structure of the endothelium includes vascular smooth muscle cells. These are able to induce vasoconstriction or vasodilation, thus modulating the diameter of the vessel (FIGURE 3). Known vasoactive hormones include serotonin, norepinephrine, insulin, and endothelin, among others. Nitric oxide (NO) is the major effector of vasodilation; its release from endothelial cells by nitric oxide synthase (NOS) leads to relaxation. The sex steroid estradiol rapidly activates eNOS (16), and although eNOS activation by other sex steroids, such as testosterone, progesterone, or di-hydrosterone is controversial (16), administration of estradiol (38), progesterone (65), testosterone (101), and other sex hormones causes vasodilation, whereas serotonin causes vasoconstriction (67). Among the varied effects on nutrient disposal and storage, insulin also has hemodynamic effects: at supraphysiological concentrations, insulin increases blood flow (50), although this finding has led to some controversy. Since bulk blood flow changes are only seen at supraphysiological doses of insulin, the relevance at the physiological level was questioned. However, at physiological concentrations, insulin vasodilates blood vessels (86); this increase in the diameter of the vessel increases the surface area available for diffusion. Since the degree of microvascular surface area is related to insulin sensitivity (28, 77), vasodilation can have significant metabolic effects without changes in bulk blood flow. Insulin causes the release of NO and also endothelin, a potent vasoconstrictor that opposes NO (88). In this way, the vascular effects of insulin are a balance between vasodilation and vasoconstriction.
FIGURE 3. Vascular smooth muscle cells lining the arteriole can modulate blood flow through capillaries.
The vascular smooth muscle cells lining the arteriole can modulate blood flow through capillaries (A). Vasoconstriction in response to certain stimuli can prevent blood flow through the vessel (B). Other factors can dilate the vessel, allowing more blood flow through the vessel (C). Capillary recruitment refers to the perfusion of previously unperfused vessels. In a tissue bed, the arteries supply a network of capillaries, yet at rest not all vessels are perfused (D). Vasoactive hormones can increase the number of perfused vessels (E), thus increasing the ability of the hormone to access more of the tissue.
In a simple system, vasodilation would increase, and vasoconstriction decrease, surface area for exchange and, therefore, metabolism. However, divergent effects on metabolism in the face of similar pressure response suggest a complexity in the effect of vasoactive hormones on the vasculature. For example, the vasoconstrictors serotonin and norephinephrine engage different areas of the microvasculature (61). Norepinephrine stimulates muscle metabolism as measured by oxygen uptake, yet serotonin decreases the oxygen uptake and metabolism of the muscle (20). Endothelin, a vasoconstrictor, has been shown to have divergent effects on metabolism, wherein low doses stimulate metabolism but higher doses lead to a generalized constriction and inhibition of metabolism (46). Blocking the vascular effects of endothelin also prevented the metabolism effects, suggesting that the vascular effects themselves were causing the changes in oxygen uptake. Interestingly, insulin was also able to block these effects (46). Therefore, a more complicated mechanism is required to explain the effects of vasoconstrictors that increase metabolism (e.g., norepinephrine and endothelin) where vasoconstriction changes the nature of blood flow through muscle.
Capillary Recruitment
Although vasodilation can increase the surface area of each vessel for nutrient exchange, vasoconstriction and vasodilation can also alter the surface area throughout the tissue in a process known as capillary recruitment. At rest, only a small portion of the capillaries through tissue are exposed to blood flow, but this can be altered by dilating or constricting the feed arterioles (FIGURE 3). For example, exercise increases the number of perfused capillaries in skeletal muscle to improve delivery of oxygen for muscle activity (69). Norepinephrine causes vasoconstriction, leading to the perfusion of more small vessels than under control conditions, and has a stimulatory effect on muscle metabolism as measured by oxygen uptake (61). If more of the vessels are perfused, the distance that molecules must diffuse to access the underlying tissue decreases: some hormones have this ability, thus increasing access to the underlying tissue. Although the ability of the hormone itself to access the tissue is important, it can also have a much more general effect on nutrient supply and even metabolism.
Techniques to measure capillary recruitment can investigate the breakdown of molecules by endothelium-bound enzymes; the rate of breakdown gives an indication of the endothelial surface area exposed to flow (67) and has shown microvascular effects of insulin (69, 102) and serotonin (67); microdialysis (60, 62) and Laser Doppler (19) have also been used to show insulin-mediated capillary recruitment. Also, contrast-enhanced ultrasound has demonstrated that the microvascular effects of insulin are an early effect regulating insulin-mediated glucose uptake (92, 94). NO blockade decreases insulin-mediated capillary recruitment and also impairs glucose uptake in humans (91). Since NO antagonism should only prevent the vascular effects of insulin, it reinforces the concept that endothelial function has an impact on the metabolism of the target tissue.
Transport of Hormones Across the Endothelium
The main function of the endothelium is as a semi-permeable barrier between plasma and the tissue; therefore, there are limited ways for hormones or other solutes to leave the vasculature. As discussed, in some tissues, hormone transport can occur through large pores, in other cases even tight junctions may permit paracellular “leak” (FIGURE 4). This involves passage through the junctions between the cells and is usually restricted to small molecules, such as urea, glucose, and ions; some have proposed that insulin may also use this method (73), although it is thought to be too big for this process. Paracellular transport would be considered a more passive role of the endothelium, yet it is increased specifically in venules during inflammation by opening the junctions between endothelial cells and can be modified by hormones such as ghrelin (49). Hormones can directly affect the permeability of a vessel, for example, angiotensin II increases vascular permeability factor gene expression (99), and angiotensin II-induced endothelial cell hyperpermeability can be inhibited by the hormone adiponectin (100).
FIGURE 4. Methods of transport across the endothelial barrier.
In the tissues that contain pores or fenestrations, solutes can pass freely from the blood to the abluminal surface. However, in continuous, un-fenestrated endothelium, small molecules can move paracellularly through the tight junctions between the endothelial cells. Larger molecules can cross using vesicular transport. Invaginations of the plasma membrane called caveolae may contain receptors for more specific transcytosis.
Since endothelial permeability is dependent of the size of the solute, the pathway for transport of larger molecules such as hormones is different from that for smaller molecules (56). Aside from paracellular transport or movement through pores, transport through the endothelial cell can also occur.
Transendothelial Transport
Transcytosis, or the pathway that couples endocytosis to exocytosis on the opposite plasma membrane, is a function of endothelial cells (66, 79). Tracers are transported through the endothelial cell via vesicles (FIGURE 4), but vascular permeability varies depending on the vessel (23). Studies following the transport of the hormone human chorionic gonadotropin (hCG) through the vessel demonstrated internalization by coated pits and vesicles on the luminal side of the endothelium and showed in a time-dependent manner the transfer through tubulovesicular endosomes and large smooth vesicles to the abluminal space (37). Vesicular transport has also been shown for albumin (56) through albumin binding proteins on the endothelial cell surface. Leptin transport across the capillary endothelium was shown to be transcellular rather than paracellular and associated with plasmalemmal vesicles (12).
Caveolae are invaginations of the plasma membrane that occur on endothelial cells, and are involved in endocytosis and signal transduction. Hormones can be dependent on caveolae for their eNOS effects, such as estrogen (43), or for altering signaling cascades, such as leptin, an adipokine involved in energy homeostasis (80). Caveolae defects alter microvascular permeability (76) through a NOS-dependent pathway and may be compensated for by increased paracellular transport through larger interendothelial junctions (66). Although the contribution of caveolae includes signaling and trafficking within the cell (71), their role in vesicular trafficking and transcytosis has been much studied (reviewed in Ref. 66). Studies have suggested that caveolae are required for insulin transport through endothelial cells (97); however, much of the transported fluid is not receptor bound (87). Therefore caveolae can be involved in hormone signaling cascades, changes in endothelial permeability to alter access to the underlying tissue, or hormone transcytosis, which may or may not be bound to receptors.
Receptor-Mediated Transport
Although many hormone receptors can be expressed on endothelial cells, the receptor may contribute more to signaling and vascular effects as described above than to transport across the endothelium. Transport of hormones across the endothelial barrier is specific to the hormone. Insulin is possibly the most studied of these and has been shown to be concentrated in the endothelial cells relative to both plasma and muscle (96). Some studies have concluded that transport is saturable (26, 40) and, as such, must be receptor mediated; however, others have shown no saturation even at concentrations in the pharmacological range (18 µU·min−1·kg−1) (85), claiming that there is no evidence for receptor-mediated transport (10, 85) or that any receptor-mediated transport is not quantitatively significant for insulin transport (73). Interestingly, inactivation of the insulin receptor on endothelial cell has no effect under basal conditions but alters expression of vasoactive mediators and may play a role in maintaining vascular tone and regulation of insulin sensitivity (89). Perhaps the most likely explanation is that the insulin receptors are involved, but at higher doses the IGF-1 receptors also contribute to insulin transport (96). The insulin receptors present on endothelial cells have been shown to be involved in the uptake of insulin (98), and more recent studies have shown that a knockout mouse model of endothelial IRS-2 is insulin resistant, and impaired insulin signaling in endothelial cells reduces insulin-mediated glucose uptake as well as impaired eNOS phosphorylation and capillary recruitment (48). Studies of human chorionic gonadotropin have shown transcytosis in the testicular microvasculature occurs via vesicles from the luminal to large vesicles open on the abluminal surface; this occurs through a receptor-mediated process (37). Receptor-mediated transcytosis of leptin (12) and growth factors (55) in other cell types have been shown, although information specific to the endothelium is scarce.
Endothelial Dysfunction: The Endothelium in Disease
Endothelial dysfunction is broadly used to describe any defect in the endothelium; however, it often refers to an impaired NO system, either by reduced NO production or increased breakdown due to oxidative stress. Endothelial dysfunction has been observed in hypertension, dyslipidemia, diabetes, obesity, and inflammation (32), and can be assessed in a variety of ways (reviewed in Ref. 32). Endothelial dysfunction can develop from otherwise healthy adaptive responses that become excessive or inappropriate (3).
Cardiovascular Disease
As mentioned, NO is responsible for the vasodilation effects of many hormones. However, NO also prevents platelet adhesion and aggregation, leukocyte adhesion and migration, and smooth muscle cell proliferation, when these abilities are lost, atherosclerosis can develop. Hyperlipemia leads to increased transcytosis of LDL, leading to accumulation of lipoprotein in the intima, and a cascade of events that lead to plaque formation and atherosclerosis (79). Decreased permeability in an experimental model of hypertension (13) and hypertensive humans with the metabolic syndrome (25) has been detected and may promote atherosclerosis development as well as impair the transendothelial transport of hormones.
Diabetes
Although impairments in cellular insulin response have been noted (6, 17, 29, 36), some studies show that signaling is not altered in insulin-resistant individuals (47), suggesting the contribution of a factor other than cellular response to whole body insulin sensitivity. Some studies have shown increased permeability in diabetic microcirculation mediated by hyperglycemia (75) or advanced glycation end products, the result of prolonged hyperglycemia (8), and acute hyperglycemia also increases glycocalyx permeability (103). However, decreased ability of a substance to reach the interstitial fluid was found in Type 2 diabetic subjects (39); since permeability of the endothelium is increased, it becomes more likely that delivery of the substance to the tissue is impaired. Consistent with this, capillary recruitment is inhibited in diabetic states (reviewed in Ref. 68). In a model of experimental insulin resistance, transendothelial transport was impaired, and it was proposed that, although cellular defects could also contribute, transport was responsible for one-third of the insulin resistance observed (58). So far, the correlation between interstitial insulin concentration and local glucose uptake (15) has held in insulin-resistant studies, where there was no significant increase in interstitial insulin when insulin was injected into the muscle and there was no change in local glucose uptake (14, 45). Therefore, there still appears to be a linear correlation between interstitial insulin concentration and local glucose uptake, suggesting the importance of transendothelial transport in insulin action.
Obesity
Decreased permeability (83) and microvascular function (84) have been detected in experimental obesity and also in obese humans (42). A high-fat diet reduces the distribution volume of macromolecules without altering the vascular volume, suggesting reduced delivery to tissues (27). Obese humans had impaired endothelium-dependent vasodilation in visceral adipose tissue, whereas nonendothelium-dependent vasodilation was unaffected (31), and the concurrent increase in inflammation suggested a potential link to atherosclerosis.
Metabolic Syndrome
Many hormones are tied to the actions of others. For example, endothelin is intrinsically involved in the vascular actions of insulin (30), stimulates catecholamine release (epinephrine and norepinephrine) in DOCA-salt hypertension (51), and is also involved in the vascular effects of steroids (progestins and androgens) (65). Norepinephrine and epinephrine are altered by insulin or insulin’s effects (53), yet epinephrine also modulates the renin-angiotensin system (81), which is involved in hypertension. Obesity impairs microvascular responses to insulin before a significant effect on metabolic processes (22, 84), and this has been proposed as a potential cause of the related disorders of insulin resistance and hypertension (90). In this way, the cluster of diseases that are associated with the metabolic syndrome begin to show their interrelatedness in terms of hormone effects. For example, angiotensin-converting enzyme inhibition can restore insulin-mediated capillary recruitment in late-stage diabetes (21). Insulin-resistant hypertensive patients with metabolic syndrome show systemically altered capillary permeability and blunted endothelial-mediated vasodilation (25), and this endothelial dysfunction could affect the delivery of any or all hormones to their target tissue.
Endothelium as a Target for Treatment
Since it has been shown that endothelium function is impaired in many diseases, the endothelium can be targeted to improve access to underlying tissue or to restore the functionality, thereby ameliorating some of the effects of the disease. The endothelium has been postulated as a target in chronic kidney disease, using, for example, erythropoietin, which protects endothelial cell function and integrity (33), and the cardiovascular benefits of resveratrol were investigated in retinal vascular disease (59), which is often associated with diabetes. The endothelium has even been suggested as a target for treatment of arthritis (72). Acute hyperglycemia increases glycocalyx permeability and decreases functional capillary density (103) as well as causing endothelial dysfunction and coagulation activation in vivo (63), and as such the glycocalyx is a potential target for treatment (5).
Perhaps the more interesting aspect of endothelial treatment is the ability to ameliorate several aspects of the metabolic syndrome at once. For example, activators of AMPK, such as AICAR (aminoimidazole carboxamide ribonucleotide), activate eNOS and recruit microcirculation (9) but also improve glucose tolerance and lower blood pressure in insulin resistance (11). Angiotensin II receptor blockade appears to improve numerous aspects of the metabolic syndrome, including blood pressure and insulin sensitivity (64), and improves microvascular function in hypertension (41). Although it is likely that there are other factors that contribute to disease, the contribution of the endothelium to disease and its potential role as a drug target should be considered.
Future Directions
Many studies on hormones use either plasma concentrations or in vitro preparations to determine hormone action and efficiency, yet the access of the hormone to the target tissue in vivo is a major determinant for action. There is much that is still unknown about how specific hormones cross the endothelial barrier and how this transport rate may be affected in disease. Insulin is possibly the best-studied of the hormones, yet the specific mechanism of transport is still being clarified (97). For many other hormones, the involvement of receptors in transendothelial transport is unknown. Studies of many diseases would benefit from a more complete understanding of the ability of hormones to access the target tissue, a factor that may be manipulated to improve disease.
In conclusion, the endothelium behaves in many tissues as both a structural and a functional barrier, and many hormones can have either a direct or indirect effect on the endothelium. By increasing the capillary surface area available for nutrient and hormone exchange, the access of the hormone and other nutrients to the target tissue, as well as whole body metabolism, can be altered by effects on the endothelium. These effects can be chronic, such as angiogenesis. However, the acute vascular effects such as vasoconstriction and vasodilation, which can potentially lead to changes in the distribution of flow, are more likely to have a rapid effect on access. The method of transport of a particular hormone across the endothelial barrier can also affect the activity of the hormone. This transendothelial transport can occur in a passive form such as leakage or diffusion but can also use a more regulated receptor-mediated transport system. Since many diseases, including hypertension, obesity, diabetes, cardiovascular disease, and atherosclerosis, exhibit some form of endothelial dysfunction, understanding the role of the endothelium in each of these diseases is essential. The interrelatedness of the actions of a range of hormones and their associated diseases suggests that the endothelium could be a good primary target, and in many cases restoring functionality of the endothelium may help in part ameliorate the disease.
Acknowledgments
This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases Grants DK27619 and DK29867.
Footnotes
No conflicts of interest, financial or otherwise, are declared by the author(s).
Author contributions: C.M.K. prepared figures; C.M.K. drafted manuscript; C.M.K. and R.N.B. edited and revised manuscript; C.M.K. and R.N.B. approved final version of manuscript.
References
- 1.Ader M, Bergman RN. Importance of transcapillary insulin transport to dynamics of insulin action after intravenous glucose. Am J Physiol Endocrinol Metab. 1994;266:E17–E25. doi: 10.1152/ajpendo.1994.266.1.E17. [DOI] [PubMed] [Google Scholar]
- 2.Aird WC. Phenotypic heterogeneity of the endothelium: II. Representative vascular beds. Circ Res. 2007;100:174–190. doi: 10.1161/01.RES.0000255690.03436.ae. [DOI] [PubMed] [Google Scholar]
- 3.Aird WC. Endothelium in health and disease. Pharmacol Rep. 2008;60:139–143. [PubMed] [Google Scholar]
- 4.Barrett EJ, Eggleston EM, Inyard AC, Wang H, Li G, Chai W, Liu Z. The vascular actions of insulin control its delivery to muscle and regulate the rate-limiting step in skeletal muscle insulin action. Diabetologia. 2009;52:752–764. doi: 10.1007/s00125-009-1313-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Becker BF, Chappell D, Bruegger D, Annecke T, Jacob M. Therapeutic strategies targeting the endothelial glycocalyx: acute deficits, but great potential. Cardiovasc Res. 2010;87:300–310. doi: 10.1093/cvr/cvq137. [DOI] [PubMed] [Google Scholar]
- 6.Bjornholm M, Zierath JR. Insulin signal transduction in human skeletal muscle: identifying the defects in Type II diabetes. Biochem Soc Trans. 2005;33:354–357. doi: 10.1042/BST0330354. [DOI] [PubMed] [Google Scholar]
- 7.Bodenlenz M, Schaupp LA, Druml T, Sommer R, Wutte A, Schaller HC, Sinner F, Wach P, Pieber TR. Measurement of interstitial insulin in human adipose and muscle tissue under moderate hyperinsulinemia by means of direct interstitial access. Am J Physiol Endocrinol Metab. 2005;289:E296–E300. doi: 10.1152/ajpendo.00431.2004. [DOI] [PubMed] [Google Scholar]
- 8.Bonnardel-Phu E, Wautier JL, Schmidt AM, Avila C, Vicaut E. Acute modulation of albumin microvascular leakage by advanced glycation end products in microcirculation of diabetic rats in vivo. Diabetes. 1999;48:2052–2058. doi: 10.2337/diabetes.48.10.2052. [DOI] [PubMed] [Google Scholar]
- 9.Bradley EA, Eringa EC, Stehouwer CD, Korstjens I, Nieuw Amerongen GP, Musters R, Sipkema P, Clark MG, Rattigan S. Activation of AMP-activated protein kinase by 5-aminoimidazole-4-carboxamide-1-beta-d-ribofuranoside in the muscle microcirculation increases nitric oxide synthesis and microvascular perfusion. Arterioscler Thromb Vasc Biol. 2010;30:1137–1142. doi: 10.1161/ATVBAHA.110.204404. [DOI] [PubMed] [Google Scholar]
- 10.Brunner F, Wascher TC. Contribution of the endothelium to transcapillary insulin transport in rat isolated perfused hearts. Diabetes. 1998;47:1127–1134. doi: 10.2337/diabetes.47.7.1127. [DOI] [PubMed] [Google Scholar]
- 11.Buhl ES, Jessen N, Pold R, Ledet T, Flyvbjerg A, Pedersen SB, Pedersen O, Schmitz O, Lund S. Long-term AICAR administration reduces metabolic disturbances and lowers blood pressure in rats displaying features of the insulin resistance syndrome. Diabetes. 2002;51:2199–2206. doi: 10.2337/diabetes.51.7.2199. [DOI] [PubMed] [Google Scholar]
- 12.Cammisotto PG, Gingras D, Bendayan M. Transcytosis of gastric leptin through the rat duodenal mucosa. Am J Physiol Gastrointest Liver Physiol. 2007;293:G773–G779. doi: 10.1152/ajpgi.00260.2007. [DOI] [PubMed] [Google Scholar]
- 13.Chakir M, Plante GE, Maheux P. Reduction of capillary permeability in the fructose-induced hypertensive rat. Am J Hypertens. 1998;11:563–569. doi: 10.1016/s0895-7061(97)00411-1. [DOI] [PubMed] [Google Scholar]
- 14.Chiu JD, Kolka CM, Richey JM, Harrison LN, Zuniga E, Kirkman EL, Bergman RN. Experimental hyperlipidemia dramatically reduces access of insulin to canine skeletal muscle. Obesity (Silver Spring) 2009;17:1486–1492. doi: 10.1038/oby.2009.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chiu JD, Richey JM, Harrison LN, Zuniga E, Kolka CM, Kirkman EL, Ellmerer M, Bergman RN. Direct administration of insulin into skeletal muscle reveals that the transport of insulin across the capillary endothelium limits the time course of insulin to activate glucose disposal. Diabetes. 2008;57:828–835. doi: 10.2337/db07-1444. [DOI] [PubMed] [Google Scholar]
- 16.Chow RW, Handelsman DJ, Ng MK. Minireview: rapid actions of sex steroids in the endothelium. Endocrinology. 2010;151:2411–2422. doi: 10.1210/en.2009-1456. [DOI] [PubMed] [Google Scholar]
- 17.Ciaraldi TP, Abrams L, Nikoulina S, Mudaliar S, Henry RR. Glucose transport in cultured human skeletal muscle cells. Regulation by insulin and glucose in nondiabetic and non-insulin-dependent diabetes mellitus subjects. J Clin Invest. 1995;96:2820–2827. doi: 10.1172/JCI118352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Clapp C, Thebault S, Jeziorski MC, Martinez DLE. Peptide hormone regulation of angiogenesis. Physiol Rev. 2009;89:1177–1215. doi: 10.1152/physrev.00024.2009. [DOI] [PubMed] [Google Scholar]
- 19.Clark AD, Barrett EJ, Rattigan S, Wallis MG, Clark MG. Insulin stimulates laser Doppler signal by rat muscle in vivo, consistent with nutritive flow recruitment. Clin Sci (London) 2001;100:283–290. [PubMed] [Google Scholar]
- 20.Clark AD, Youd JM, Rattigan S, Barrett EJ, Clark MG. Heterogeneity of laser Doppler flowmetry in perfused muscle indicative of nutritive and non-nutritive flow. Am J Physiol Heart Circ Physiol. 2001;280:H1324–H1333. doi: 10.1152/ajpheart.2001.280.3.H1324. [DOI] [PubMed] [Google Scholar]
- 21.Clerk LH, Vincent MA, Barrett EJ, Lankford MF, Lindner JR. Skeletal muscle capillary responses to insulin are abnormal in late-stage diabetes and are restored by angiogensin-converting enzyme inhibition. Am J Physiol Endocrinol Metab. 2007;293:E1804–E1809. doi: 10.1152/ajpendo.00498.2007. [DOI] [PubMed] [Google Scholar]
- 22.Clerk LH, Vincent MA, Jahn LA, Liu Z, Lindner JR, Barrett EJ. Obesity blunts insulin-mediated microvascular recruitment in human forearm muscle. Diabetes. 2006;55:1436–1442. doi: 10.2337/db05-1373. [DOI] [PubMed] [Google Scholar]
- 23.Debbage PL, Solder E, Seidl S, Hutzler P, Hugl B, Ofner D, Kreczy A. Intravital lectin perfusion analysis of vascular permeability in human micro- and macro-blood vessels. Histochem Cell Biol. 2001;116:349–359. doi: 10.1007/s004180100328. [DOI] [PubMed] [Google Scholar]
- 24.Deli MA, Abraham CS, Kataoka Y, Niwa M. Permeability studies on in vitro blood-brain barrier models: physiology, pathology, and pharmacology. Cell Mol Neurobiol. 2005;25:59–127. doi: 10.1007/s10571-004-1377-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dell’Omo G, Penno G, Pucci L, Mariani M, Del Prato S, Pedrinelli R. Abnormal capillary permeability and endothelial dysfunction in hypertension with comorbid metabolic syndrome. Atherosclerosis. 2004;172:383–389. doi: 10.1016/j.atherosclerosis.2003.11.013. [DOI] [PubMed] [Google Scholar]
- 26.Eggleston EM, Jahn LA, Barrett EJ. Hyperinsulinemia rapidly increases human muscle microvascular perfusion but fails to increase muscle insulin clearance: evidence that a saturable process mediates muscle insulin uptake. Diabetes. 2007;56:2958–2963. doi: 10.2337/db07-0670. [DOI] [PubMed] [Google Scholar]
- 27.Ellmerer M, Hamilton-Wessler M, Kim SP, Huecking K, Kirkman E, Chiu J, Richey J, Bergman RN. Reduced access to insulin-sensitive tissues in dogs with obesity secondary to increased fat intake. Diabetes. 2006;55:1769–1775. doi: 10.2337/db05-1509. [DOI] [PubMed] [Google Scholar]
- 28.Ellmerer M, Kim SP, Hamilton-Wessler M, Hucking K, Kirkman E, Bergman RN. Physiological hyperinsulinemia in dogs augments access of macromolecules to insulin-sensitive tissues. Diabetes. 2004;53:2741–2747. doi: 10.2337/diabetes.53.11.2741. [DOI] [PubMed] [Google Scholar]
- 29.Eriksson J, Koranyi L, Bourey R, Schalin-Jantti C, Widen E, Mueckler M, Permutt AM, Groop LC. Insulin resistance in Type 2 (non-insulin-dependent) diabetic patients and their relatives is not associated with a defect in the expression of the insulin-responsive glucose transporter (GLUT-4) gene in human skeletal muscle. Diabetologia. 1992;35:143–147. doi: 10.1007/BF00402546. [DOI] [PubMed] [Google Scholar]
- 30.Eringa EC, Stehouwer CD, Merlijn T, Westerhof N, Sipkema P. Physiological concentrations of insulin induce endothelin-mediated vasoconstriction during inhibition of NOS or PI3-kinase in skeletal muscle arterioles. Cardiovasc Res. 2002;56:464–471. doi: 10.1016/s0008-6363(02)00593-x. [DOI] [PubMed] [Google Scholar]
- 31.Farb MG, Ganley-Leal L, Mott M, Liang Y, Ercan B, Widlansky ME, Bigornia SJ, Fiscale AJ, Apovian CM, Carmine B, Hess DT, Vita JA, Gokce N. Arteriolar function in visceral adipose tissue is impaired in human obesity. Arterioscler Thromb Vasc Biol. 2012;32:467–473. doi: 10.1161/ATVBAHA.111.235846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Flammer AJ, Luscher TF. Three decades of endothelium research: from the detection of nitric oxide to the everyday implementation of endothelial function measurements in cardiovascular diseases. Swiss Med Wkly. 2010;140:w13122. doi: 10.4414/smw.2010.13122. [DOI] [PubMed] [Google Scholar]
- 33.Fliser D. Perspectives in renal disease progression: the endothelium as a treatment target in chronic kidney disease. J Nephrol. 2010;23:369–376. [PubMed] [Google Scholar]
- 34.Florey The endothelial cell. Br Med J. 1966;2:487–490. doi: 10.1136/bmj.2.5512.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Freidenberg GR, Suter S, Henry RR, Nolan J, Reichart D, Olefsky JM. Delayed onset of insulin activation of the insulin receptor kinase in vivo in human skeletal muscle. Diabetes. 1994;43:118–126. doi: 10.2337/diab.43.1.118. [DOI] [PubMed] [Google Scholar]
- 36.Garvey WT, Maianu L, Zhu JH, Brechtel-Hook G, Wallace P, Baron AD. Evidence for defects in the trafficking and translocation of GLUT4 glucose transporters in skeletal muscle as a cause of human insulin resistance. J Clin Invest. 1998;101:2377–2386. doi: 10.1172/JCI1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ghinea N, Mai TV, Groyer-Picard MT, Milgrom E. How protein hormones reach their target cells. Receptor-mediated transcytosis of hCG through endothelial cells. J Cell Biol. 1994;125:87–97. doi: 10.1083/jcb.125.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gilligan DM, Badar DM, Panza JA, Quyyumi AA, Cannon RO., III Acute vascular effects of estrogen in postmenopausal women. Circulation. 1994;90:786–791. doi: 10.1161/01.cir.90.2.786. [DOI] [PubMed] [Google Scholar]
- 39.Gudbjornsdottir S, Sjostrand M, Strindberg L, Lonnroth P. Decreased muscle capillary permeability surface area in Type 2 diabetic subjects. J Clin Endocrinol Metab. 2005;90:1078–1082. doi: 10.1210/jc.2004-0947. [DOI] [PubMed] [Google Scholar]
- 40.Herkner H, Klein N, Joukhadar C, Lackner E, Langenberger H, Frossard M, Bieglmayer C, Wagner O, Roden M, Muller M. Transcapillary insulin transfer in human skeletal muscle. Eur J Clin Invest. 2003;33:141–146. doi: 10.1046/j.1365-2362.2003.01106.x. [DOI] [PubMed] [Google Scholar]
- 41.Jonk AM, Houben AJ, Schaper NC, de Leeuw PW, Serne EH, Smulders YM, Stehouwer CD. Acute angiotensin II receptor blockade improves insulin-induced microvascular function in hypertensive individuals. Microvasc Res. 2011;82:77–83. doi: 10.1016/j.mvr.2011.04.002. [DOI] [PubMed] [Google Scholar]
- 42.Keske MA, Clerk LH, Price WJ, Jahn LA, Barrett EJ. Obesity blunts microvascular recruitment in human forearm muscle after a mixed meal. Diabetes Care. 2009;32:1672–1677. doi: 10.2337/dc09-0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim HP, Lee JY, Jeong JK, Bae SW, Lee HK, Jo I. Nongenomic stimulation of nitric oxide release by estrogen is mediated by estrogen receptor alpha localized in caveolae. Biochem Biophys Res Commun. 1999;263:257–262. doi: 10.1006/bbrc.1999.1348. [DOI] [PubMed] [Google Scholar]
- 44.Kim SP, Ellmerer M, Van Citters GW, Bergman RN. Primacy of hepatic insulin resistance in the development of the metabolic syndrome induced by an isocaloric moderate-fat diet in the dog. Diabetes. 2003;52:2453–2460. doi: 10.2337/diabetes.52.10.2453. [DOI] [PubMed] [Google Scholar]
- 45.Kolka CM, Harrison LN, Lottati M, Chiu JD, Kirkman EL, Bergman RN. Diet-induced obesity prevents interstitial dispersion of insulin in skeletal muscle. Diabetes. 2009;59:619–626. doi: 10.2337/db09-0839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kolka CM, Rattigan S, Richards S, Clark MG. Metabolic and vascular actions of endothelin-1 are inhibited by insulin-mediated vasodilation in perfused rat hindlimb muscle. Br J Pharmacol. 2005;145:992–1000. doi: 10.1038/sj.bjp.0706263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Krutzfeldt J, Kausch C, Volk A, Klein HH, Rett K, Haring HU, Stumvoll M. Insulin signaling and action in cultured skeletal muscle cells from lean healthy humans with high and low insulin sensitivity. Diabetes. 2000;49:992–998. doi: 10.2337/diabetes.49.6.992. [DOI] [PubMed] [Google Scholar]
- 48.Kubota T, Kubota N, Kumagai H, Yamaguchi S, Kozono H, Takahashi T, Inoue M, Itoh S, Takamoto I, Sasako T, Kumagai K, Kawai T, Hashimoto S, Kobayashi T, Sato M, Tokuyama K, Nishimura S, Tsunoda M, Ide T, Murakami K, Yamazaki T, Ezaki O, Kawamura K, Masuda H, Moroi M, Sugi K, Oike Y, Shimokawa H, Yanagihara N, Tsutsui M, Terauchi Y, Tobe K, Nagai R, Kamata K, Inoue K, Kodama T, Ueki K, Kadowaki T. Impaired insulin signaling in endothelial cells reduces insulin-induced glucose uptake by skeletal muscle. Cell Metab. 2011;13:294–307. doi: 10.1016/j.cmet.2011.01.018. [DOI] [PubMed] [Google Scholar]
- 49.Kwan RO, Cureton E, Dozier K, Curran B, Sadjadi J, Victorino GP. Ghrelin decreases microvascular leak during inflammation. J Trauma. 2010;68:1186–1191. doi: 10.1097/TA.0b013e3181bb9878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Laakso M, Edelman SV, Brechtel G, Baron AD. Decreased effect of insulin to stimulate skeletal muscle blood flow in obese man. A novel mechanism for insulin resistance. J Clin Invest. 1990;85:1844–1852. doi: 10.1172/JCI114644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lange DL, Haywood JR, Hinojosa-Laborde C. Endothelin enhances and inhibits adrenal catecholamine release in deoxycorticosterone acetate-salt hypertensive rats. Hypertension. 2000;35:385–390. doi: 10.1161/01.hyp.35.1.385. [DOI] [PubMed] [Google Scholar]
- 52.Lew MJ, Rivers RJ, Duling BR. Arteriolar smooth muscle responses are modulated by an intramural diffusion barrier. Am J Physiol Heart Circ Physiol. 1989;257:H10–H16. doi: 10.1152/ajpheart.1989.257.1.H10. [DOI] [PubMed] [Google Scholar]
- 53.Liang C, Doherty JU, Faillace R, Maekawa K, Arnold S, Gavras H, Hood WB., Jr Insulin infusion in conscious dogs. Effects on systemic and coronary hemodynamics, regional blood flows, and plasma catecholamines. J Clin Invest. 1982;69:1321–1336. doi: 10.1172/JCI110572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Maggs DG, Jacob R, Rife F, Lange R, Leone P, During MJ, Tamborlane WV, Sherwin RS. Interstitial fluid concentrations of glycerol, glucose, and amino acids in human quadricep muscle and adipose tissue. Evidence for significant lipolysis in skeletal muscle. J Clin Invest. 1995;96:370–377. doi: 10.1172/JCI118043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Maratos-Flier E, Kao CY, Verdin EM, King GL. Receptor-mediated vectorial transcytosis of epidermal growth factor by Madin-Darby canine kidney cells. J Cell Biol. 1987;105:1595–1601. doi: 10.1083/jcb.105.4.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mehta D, Malik AB. Signaling mechanisms regulating endothelial permeability. Physiol Rev. 2006;86:279–367. doi: 10.1152/physrev.00012.2005. [DOI] [PubMed] [Google Scholar]
- 57.Miles PD, Levisetti M, Reichart D, Khoursheed M, Moossa AR, Olefsky JM. Kinetics of insulin action in vivo. Identification of rate-limiting steps. Diabetes. 1995;44:947–953. doi: 10.2337/diab.44.8.947. [DOI] [PubMed] [Google Scholar]
- 58.Miles PD, Li S, Hart M, Romeo O, Cheng J, Cohen A, Raafat K, Moossa AR, Olefsky JM. Mechanisms of insulin resistance in experimental hyper-insulinemic dogs. J Clin Invest. 1998;101:202–211. doi: 10.1172/JCI1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nagaoka T, Hein TW, Yoshida A, Kuo L. Resveratrol, a component of red wine, elicits dilation of isolated porcine retinal arterioles: role of nitric oxide and potassium channels. Invest Ophthalmol Vis Sci. 2007;48:4232–4239. doi: 10.1167/iovs.07-0094. [DOI] [PubMed] [Google Scholar]
- 60.Newman JM, Di Maria CA, Rattigan S, Clark MG. Nutritive blood flow affects microdialysis O/I ratio for [14C]ethanol and 3H2O in perfused rat hindlimb. Am J Physiol Heart Circ Physiol. 2001;281:H2731–H2737. doi: 10.1152/ajpheart.2001.281.6.H2731. [DOI] [PubMed] [Google Scholar]
- 61.Newman JM, Dora KA, Rattigan S, Edwards SJ, Colquhoun EQ, Clark MG. Norepinephrine and serotonin vasoconstriction in rat hindlimb control different vascular flow routes. Am J Physiol Endocrinol Metab. 1996;270:E689–E699. doi: 10.1152/ajpendo.1996.270.4.E689. [DOI] [PubMed] [Google Scholar]
- 62.Newman JM, Ross RM, Richards SM, Clark MG, Rattigan S. Insulin and contraction increase nutritive blood flow in rat muscle in vivo determined by microdialysis of L-[14C]glucose. J Physiol. 2007;585:217–229. doi: 10.1113/jphysiol.2007.138818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nieuwdorp M, van Haeften TW, Gouverneur MC, Mooij HL, van Lieshout MH, Levi M, Meijers JC, Holleman F, Hoekstra JB, Vink H, Kastelein JJ, Stroes ES. Loss of endothelial glycocalyx during acute hyperglycemia coincides with endothelial dysfunction and coagulation activation in vivo. Diabetes. 2006;55:480–486. doi: 10.2337/diabetes.55.02.06.db05-1103. [DOI] [PubMed] [Google Scholar]
- 64.Pershadsingh HA. Treating the metabolic syndrome using angiotensin receptor antagonists that selectively modulate peroxisome proliferator-activated receptor-gamma. Int J Biochem Cell Biol. 2006;38:766–781. doi: 10.1016/j.biocel.2005.08.006. [DOI] [PubMed] [Google Scholar]
- 65.Perusquia M, Hernandez R, Morales MA, Campos MG, Villalon CM. Role of endothelium in the vasodilating effect of progestins and androgens on the rat thoracic aorta. Gen Pharmacol. 1996;27:181–185. doi: 10.1016/0306-3623(95)00091-7. [DOI] [PubMed] [Google Scholar]
- 66.Predescu SA, Predescu DN, Malik AB. Molecular determinants of endothelial transcytosis and their role in endothelial permeability. Am J Physiol Lung Cell Mol Physiol. 2007;293:L823–L842. doi: 10.1152/ajplung.00436.2006. [DOI] [PubMed] [Google Scholar]
- 67.Rattigan S, Appleby GJ, Miller KA, Steen JT, Dora KA, Colquhoun EQ, Clark MG. Serotonin inhibition of 1-methylxanthine metabolism parallels its vasoconstrictor activity and inhibition of oxygen uptake in perfused rat hindlimb. Acta Physiol Scand. 1997;161:161–169. doi: 10.1046/j.1365-201X.1997.00215.x. [DOI] [PubMed] [Google Scholar]
- 68.Rattigan S, Bussey CT, Ross RM, Richards SM. Obesity, insulin resistance, and capillary recruitment. Microcirculation. 2007;14:299–309. doi: 10.1080/10739680701282796. [DOI] [PubMed] [Google Scholar]
- 69.Rattigan S, Wheatley C, Richards SM, Barrett EJ, Clark MG. Exercise and insulin-mediated capillary recruitment in muscle. Exerc Sport Sci Rev. 2005;33:43–48. [PubMed] [Google Scholar]
- 70.Renkin EM. Filtration, diffusion, and molecular sieving through porous cellulose membranes. J Gen Physiol. 1954;20:225–243. [PMC free article] [PubMed] [Google Scholar]
- 71.Rippe B, Rosengren BI, Carlsson O, Venturoli D. Transendothelial transport: the vesicle controversy. J Vasc Res. 2002;39:375–390. doi: 10.1159/000064521. [DOI] [PubMed] [Google Scholar]
- 72.Sakuta T, Morita Y, Satoh M, Fox DA, Kashihara N. Involvement of the renin-angiotensin system in the development of vascular damage in a rat model of arthritis: effect of angiotensin receptor blockers. Arthritis Rheum. 2010;62:1319–1328. doi: 10.1002/art.27384. [DOI] [PubMed] [Google Scholar]
- 73.Salvetti F, Cecchetti P, Janigro D, Lucacchini A, Benzi L, Martini C. Insulin permeability across an in vitro dynamic model of endothelium. Pharm Res. 2002;19:445–450. doi: 10.1023/a:1015187410909. [DOI] [PubMed] [Google Scholar]
- 74.Sarabia V, Lam L, Burdett E, Leiter LA, Klip A. Glucose transport in human skeletal muscle cells in culture. Stimulation by insulin and metformin. J Clin Invest. 1992;90:1386–1395. doi: 10.1172/JCI116005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Scalia R, Gong Y, Berzins B, Zhao LJ, Sharma K. Hyperglycemia is a major determinant of albumin permeability in diabetic microcirculation: the role of mu-calpain. Diabetes. 2007;56:1842–1849. doi: 10.2337/db06-1198. [DOI] [PubMed] [Google Scholar]
- 76.Schubert W, Frank PG, Woodman SE, Hyogo H, Cohen DE, Chow CW, Lisanti MP. Microvascular hyperpermeability in caveolin-1 (−/−) knock-out mice. Treatment with a specific nitric-oxide synthase inhibitor, l-NAME, restores normal microvascular permeability in Cav-1 null mice. J Biol Chem. 2002;277:40091–40098. doi: 10.1074/jbc.M205948200. [DOI] [PubMed] [Google Scholar]
- 77.Serne EH, Ijzerman RG, Gans RO, Nijveldt R, de Vries G, Evertz R, Donker AJ, Stehouwer CD. Direct evidence for insulin-induced capillary recruitment in skin of healthy subjects during physiological hyperinsulinemia. Diabetes. 2002;51:1515–1522. doi: 10.2337/diabetes.51.5.1515. [DOI] [PubMed] [Google Scholar]
- 78.Shirley HH, Jr, Wolfram CG, Wasserman K, Mayerson HS. Capillary permeability to macromolecules: stretched pore phenomenon. Am J Physiol. 1957;190:189–193. doi: 10.1152/ajplegacy.1957.190.2.189. [DOI] [PubMed] [Google Scholar]
- 79.Simionescu M, Gafencu A, Antohe F. Transcytosis of plasma macromolecules in endothelial cells: a cell biological survey. Microsc Res Tech. 2002;57:269–288. doi: 10.1002/jemt.10086. [DOI] [PubMed] [Google Scholar]
- 80.Singh P, Peterson TE, Sert-Kuniyoshi FH, Jensen MD, Somers VK. Leptin upregulates caveolin-1 expression: implications for development of atherosclerosis. Atherosclerosis. 2011;217:499–502. doi: 10.1016/j.atherosclerosis.2010.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Siragy HM, Howell NL, Ragsdale NV, Carey RM. Renal interstitial fluid angiotensin. Modulation by anesthesia, epinephrine, sodium depletion, and renin inhibition. Hypertension. 1995;25:1021–1024. doi: 10.1161/01.hyp.25.5.1021. [DOI] [PubMed] [Google Scholar]
- 82.Sjostrand M, Holmang A, Lonnroth P. Measurement of interstitial insulin in human muscle. Am J Physiol Endocrinol Metab. 1999;276:E151–E154. doi: 10.1152/ajpendo.1999.276.1.E151. [DOI] [PubMed] [Google Scholar]
- 83.St Pierre P, Bouffard L, Papirakis ME, Maheux P. Increased extravasation of macromolecules in skeletal muscles of the Zucker rat model. Obesity (Silver Spring) 2006;14:787–793. doi: 10.1038/oby.2006.91. [DOI] [PubMed] [Google Scholar]
- 84.St Pierre P, Genders AJ, Keske MA, Richards SM, Rattigan S. Loss of insulin-mediated microvascular perfusion in skeletal muscle is associated with the development of insulin resistance. Diabetes Obes Metab. 2010;12:798–805. doi: 10.1111/j.1463-1326.2010.01235.x. [DOI] [PubMed] [Google Scholar]
- 85.Steil GM, Ader M, Moore DM, Rebrin K, Bergman RN. Transendothelial insulin transport is not saturable in vivo. No evidence for a receptor-mediated process. J Clin Invest. 1996;97:1497–1503. doi: 10.1172/JCI118572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tack CJJ, Schefman AEP, Willems JL, Thien T, Lutterman JA, Smits P. Direct vasodilator effects of physiological hyperinsulin-aemia in human skeletal muscle. Eur J Clin Invest. 1996;26:772–778. doi: 10.1046/j.1365-2362.1996.2020551.x. [DOI] [PubMed] [Google Scholar]
- 87.Tuma PL, Hubbard AL. Transcytosis: crossing cellular barriers. Physiol Rev. 2003;83:871–932. doi: 10.1152/physrev.00001.2003. [DOI] [PubMed] [Google Scholar]
- 88.Verma S, Yao L, Stewart DJ, Dumont AS, Anderson TJ, McNeill JH. Endothelin antagonism uncovers insulin-mediated vasorelaxation in vitro and in vivo. Hypertension. 2001;37:328–333. doi: 10.1161/01.hyp.37.2.328. [DOI] [PubMed] [Google Scholar]
- 89.Vicent D, Ilany J, Kondo T, Naruse K, Fisher SJ, Kisanuki YY, Bursell S, Yanagisawa M, King GL, Kahn CR. The role of endothelial insulin signaling in the regulation of vascular tone and insulin resistance. J Clin Invest. 2003;111:1373–1380. doi: 10.1172/JCI15211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Villela NR, Kramer-Aguiar LG, Bottino DA, Wiernsperger N, Bouskela E. Metabolic disturbances linked to obesity: the role of impaired tissue perfusion. Arq Bras Endocrinol Metabol. 2009;53:238–245. doi: 10.1590/s0004-27302009000200015. [DOI] [PubMed] [Google Scholar]
- 91.Vincent MA, Barrett EJ, Lindner JR, Clark MG, Rattigan S. Inhibiting NOS blocks microvascular recruitment and blunts muscle glucose uptake in response to insulin. Am J Physiol Endocrinol Metab. 2003;285:E123–E129. doi: 10.1152/ajpendo.00021.2003. [DOI] [PubMed] [Google Scholar]
- 92.Vincent MA, Clerk LH, Lindner JR, Klibanov AL, Clark MG, Rattigan S, Barrett EJ. Microvascular recruitment is an early insulin effect that regulates skeletal muscle glucose uptake in vivo. Diabetes. 2004;53:1418–1423. doi: 10.2337/diabetes.53.6.1418. [DOI] [PubMed] [Google Scholar]
- 93.Vincent MA, Clerk LH, Rattigan S, Clark MG, Barrett EJ. Active role for the vasculature in the delivery of insulin to skeletal muscle. Clin Exp Pharmacol Physiol. 2005;32:302–307. doi: 10.1111/j.1440-1681.2005.04188.x. [DOI] [PubMed] [Google Scholar]
- 94.Vincent MA, Dawson D, Clark AD, Lindner JR, Rattigan S, Clark MG, Barrett EJ. Skeletal muscle microvascular recruitment by physiological hyperinsulinemia precedes increases in total blood flow. Diabetes. 2002;51:42–48. doi: 10.2337/diabetes.51.1.42. [DOI] [PubMed] [Google Scholar]
- 95.Vink H, Duling BR. Capillary endothelial surface layer selectively reduces plasma solute distribution volume. Am J Physiol Heart Circ Physiol. 2000;278:H285–H289. doi: 10.1152/ajpheart.2000.278.1.H285. [DOI] [PubMed] [Google Scholar]
- 96.Wang H, Liu Z, Li G, Barrett EJ. The vascular endothelial cell mediates insulin transport into skeletal muscle. Am J Physiol Endocrinol Metab. 2006;291:E323–E332. doi: 10.1152/ajpendo.00047.2006. [DOI] [PubMed] [Google Scholar]
- 97.Wang H, Wang AX, Barrett EJ. Caveolin-1 is required for vascular endothelial insulin uptake. Am J Physiol Endocrinol Metab. 2011;300:E134–E144. doi: 10.1152/ajpendo.00498.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wang H, Wang AX, Liu Z, Barrett EJ. Insulin signaling stimulates insulin transport by bovine aortic endothelial cells. Diabetes. 2008;57:540–547. doi: 10.2337/db07-0967. [DOI] [PubMed] [Google Scholar]
- 99.Williams B, Baker AQ, Gallacher B, Lodwick D. Angiotensin II increases vascular permeability factor gene expression by human vascular smooth muscle cells. Hypertension. 1995;25:913–917. doi: 10.1161/01.hyp.25.5.913. [DOI] [PubMed] [Google Scholar]
- 100.Xu SQ, Mahadev K, Wu X, Fuchsel L, Donnelly S, Scalia RG, Goldstein BJ. Adiponectin protects against angiotensin II or tumor necrosis factor alpha-induced endothelial cell monolayer hyperpermeability: role of cAMP/PKA signaling. Arterioscler Thromb Vasc Biol. 2008;28:899–905. doi: 10.1161/ATVBAHA.108.163634. [DOI] [PubMed] [Google Scholar]
- 101.Yue P, Chatterjee K, Beale C, Poole-Wilson PA, Collins P. Testosterone relaxes rabbit coronary arteries and aorta. Circulation. 1995;91:1154–1160. doi: 10.1161/01.cir.91.4.1154. [DOI] [PubMed] [Google Scholar]
- 102.Zhang L, Vincent MA, Richards SM, Clerk LH, Rattigan S, Clark MG, Barrett EJ. Insulin sensitivity of muscle capillary recruitment in vivo. Diabetes. 2004;53:447–453. doi: 10.2337/diabetes.53.2.447. [DOI] [PubMed] [Google Scholar]
- 103.Zuurbier CJ, Demirci C, Koeman A, Vink H, Ince C. Short-term hyperglycemia increases endothelial glycocalyx permeability and acutely decreases lineal density of capillaries with flowing red blood cells. J Appl Physiol. 2005;99:1471–1476. doi: 10.1152/japplphysiol.00436.2005. [DOI] [PubMed] [Google Scholar]