Abstract
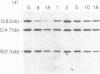
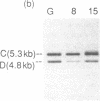
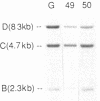
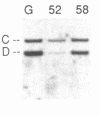
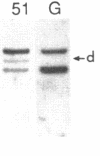
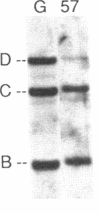
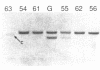
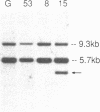
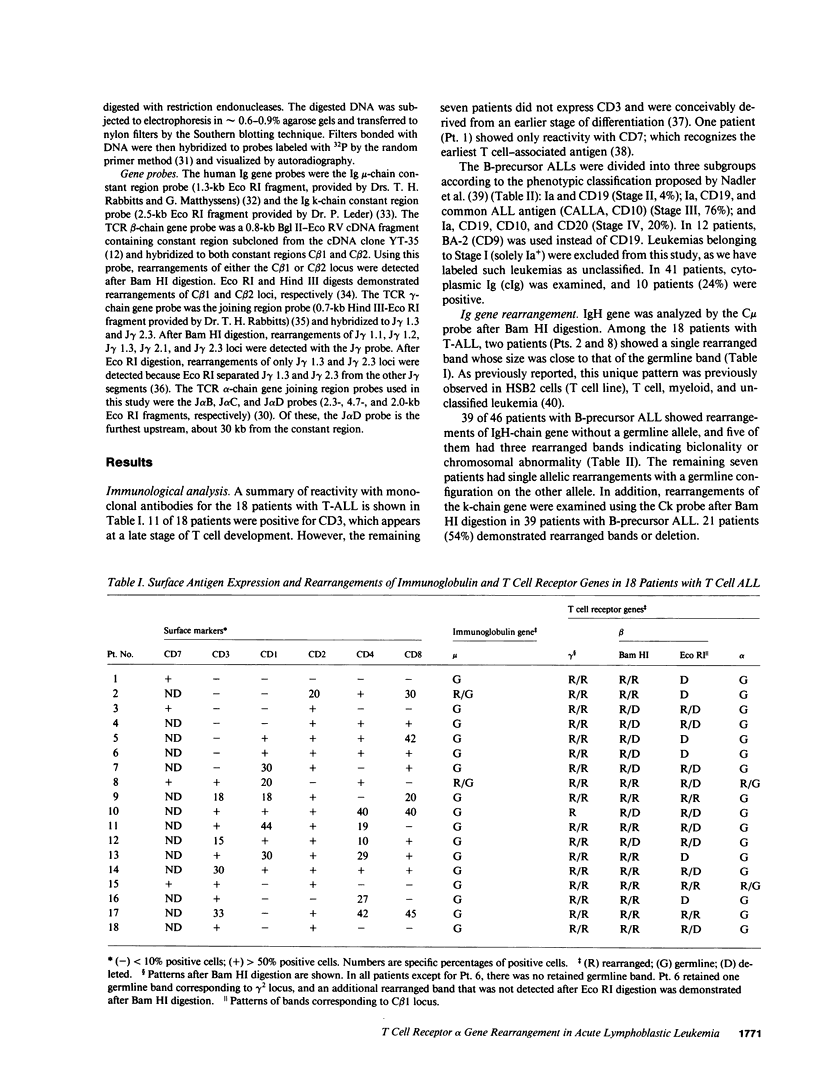
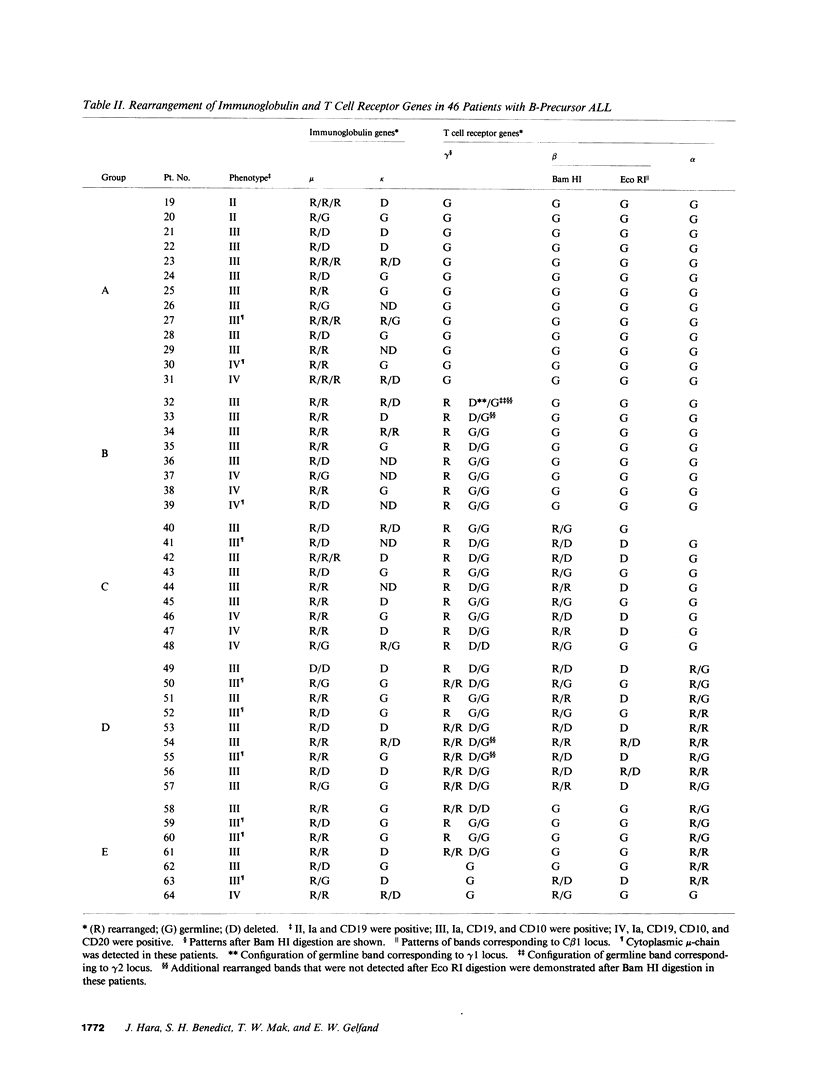
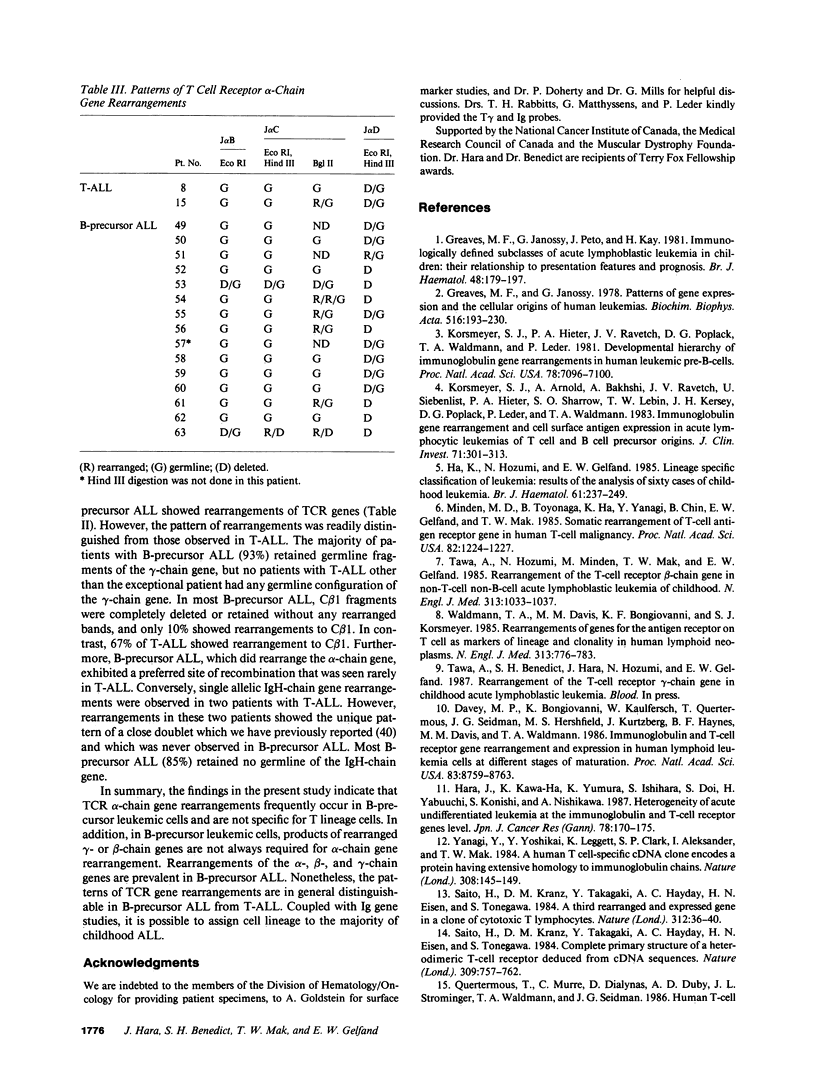
We have analyzed T cell receptor alpha-chain gene configuration using three genomic joining (J) region probes in 64 children with acute lymphoblastic leukemia (ALL). 11 out of 18 T-ALLs were T3 positive; alpha-chain gene rearrangements were demonstrated in only two of 18, indicating that the majority of T-ALLs would have rearrangements involving J alpha segments located upstream of these probes. In contrast, 15 out of 46 B-precursor ALLs showed rearrangements of the alpha-chain gene and J alpha segments located approximately 20-30 kb upstream of the constant region were involved in 13 of these patients. Nine of 15 B-precursor ALLs with rearranged alpha-chain genes had rearrangements of both gamma- and beta-chain genes, whereas the remaining six had no rearrangements of gamma- and beta-chain genes. These findings indicated that alpha-chain gene rearrangement is not specific for T lineage cells and gamma- and/or beta-chain gene rearrangement does not appear essential for alpha-chain gene rearrangement, at least in B-precursor leukemic cells.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acuto O., Fabbi M., Bensussan A., Milanese C., Campen T. J., Royer H. D., Reinherz E. L. The human T-cell receptor. J Clin Immunol. 1985 May;5(3):141–157. doi: 10.1007/BF00915505. [DOI] [PubMed] [Google Scholar]
- Alt F. W. Exclusive immunoglobulin genes. Nature. 1984 Dec 6;312(5994):502–503. doi: 10.1038/312502a0. [DOI] [PubMed] [Google Scholar]
- Bank I., DePinho R. A., Brenner M. B., Cassimeris J., Alt F. W., Chess L. A functional T3 molecule associated with a novel heterodimer on the surface of immature human thymocytes. Nature. 1986 Jul 10;322(6075):179–181. doi: 10.1038/322179a0. [DOI] [PubMed] [Google Scholar]
- Brenner M. B., McLean J., Dialynas D. P., Strominger J. L., Smith J. A., Owen F. L., Seidman J. G., Ip S., Rosen F., Krangel M. S. Identification of a putative second T-cell receptor. Nature. 1986 Jul 10;322(6075):145–149. doi: 10.1038/322145a0. [DOI] [PubMed] [Google Scholar]
- Brenner M. B., Trowbridge I. S., Strominger J. L. Cross-linking of human T cell receptor proteins: association between the T cell idiotype beta subunit and the T3 glycoprotein heavy subunit. Cell. 1985 Jan;40(1):183–190. doi: 10.1016/0092-8674(85)90321-6. [DOI] [PubMed] [Google Scholar]
- Davey M. P., Bongiovanni K. F., Kaulfersch W., Quertermous T., Seidman J. G., Hershfield M. S., Kurtzberg J., Haynes B. F., Davis M. M., Waldmann T. A. Immunoglobulin and T-cell receptor gene rearrangement and expression in human lymphoid leukemia cells at different stages of maturation. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8759–8763. doi: 10.1073/pnas.83.22.8759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Foon K. A., Todd R. F., 3rd Immunologic classification of leukemia and lymphoma. Blood. 1986 Jul;68(1):1–31. [PubMed] [Google Scholar]
- Furley A. J., Mizutani S., Weilbaecher K., Dhaliwal H. S., Ford A. M., Chan L. C., Molgaard H. V., Toyonaga B., Mak T., van den Elsen P. Developmentally regulated rearrangement and expression of genes encoding the T cell receptor-T3 complex. Cell. 1986 Jul 4;46(1):75–87. doi: 10.1016/0092-8674(86)90861-5. [DOI] [PubMed] [Google Scholar]
- Greaves M. F., Janossy G., Peto J., Kay H. Immunologically defined subclasses of acute lymphoblastic leukaemia in children: their relationship to presentation features and prognosis. Br J Haematol. 1981 Jun;48(2):179–197. [PubMed] [Google Scholar]
- Greaves M., Janossy G. Patterns of gene expression and the cellular origins of human leukaemias. Biochim Biophys Acta. 1978 Oct 27;516(2):193–230. doi: 10.1016/0304-419x(78)90008-2. [DOI] [PubMed] [Google Scholar]
- Ha K., Hozumi N., Hrincu A., Gelfand E. W. Lineage specific classification of leukaemia: results of the analysis of sixty cases of childhood leukaemia. Br J Haematol. 1985 Oct;61(2):237–249. doi: 10.1111/j.1365-2141.1985.tb02822.x. [DOI] [PubMed] [Google Scholar]
- Ha K., Minden M., Hozumi N., Gelfand E. W. Immunoglobulin gene rearrangement in acute myelogenous leukemia. Cancer Res. 1984 Oct;44(10):4658–4660. [PubMed] [Google Scholar]
- Ha K., Minden M., Hozumi N., Gelfand E. W. Immunoglobulin mu-chain gene rearrangement in a patient with T cell acute lymphoblastic leukemia. J Clin Invest. 1984 Apr;73(4):1232–1236. doi: 10.1172/JCI111310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ha K., Minden M., Hozumi N., Gelfand E. W. Single allelic C mu gene rearrangements in patients with T cell and undifferentiated leukemia. Leuk Res. 1986;10(1):1–8. doi: 10.1016/0145-2126(86)90098-6. [DOI] [PubMed] [Google Scholar]
- Haars R., Kronenberg M., Gallatin W. M., Weissman I. L., Owen F. L., Hood L. Rearrangement and expression of T cell antigen receptor and gamma genes during thymic development. J Exp Med. 1986 Jul 1;164(1):1–24. doi: 10.1084/jem.164.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara J., Kawa-Ha K., Yumura K., Ishihara S., Doi S., Yabuuchi H., Konishi S., Nishikawa A. Heterogeneity of acute undifferentiated leukemia at the immunoglobulin and T-cell receptor genes level. Jpn J Cancer Res. 1987 Feb;78(2):170–175. [PubMed] [Google Scholar]
- Hieter P. A., Max E. E., Seidman J. G., Maizel J. V., Jr, Leder P. Cloned human and mouse kappa immunoglobulin constant and J region genes conserve homology in functional segments. Cell. 1980 Nov;22(1 Pt 1):197–207. doi: 10.1016/0092-8674(80)90168-3. [DOI] [PubMed] [Google Scholar]
- Ikuta K., Hattori M., Wake K., Kano S., Honjo T., Yodoi J., Minato N. Expression and rearrangement of the alpha, beta, and gamma chain genes of the T cell receptor in cloned murine large granular lymphocyte lines. No correlation with the cytotoxic spectrum. J Exp Med. 1986 Aug 1;164(2):428–442. doi: 10.1084/jem.164.2.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korsmeyer S. J., Arnold A., Bakhshi A., Ravetch J. V., Siebenlist U., Hieter P. A., Sharrow S. O., LeBien T. W., Kersey J. H., Poplack D. G. Immunoglobulin gene rearrangement and cell surface antigen expression in acute lymphocytic leukemias of T cell and B cell precursor origins. J Clin Invest. 1983 Feb;71(2):301–313. doi: 10.1172/JCI110770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korsmeyer S. J., Hieter P. A., Ravetch J. V., Poplack D. G., Waldmann T. A., Leder P. Developmental hierarchy of immunoglobulin gene rearrangements in human leukemic pre-B-cells. Proc Natl Acad Sci U S A. 1981 Nov;78(11):7096–7100. doi: 10.1073/pnas.78.11.7096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanier L. L., Weiss A. Presence of Ti (WT31) negative T lymphocytes in normal blood and thymus. Nature. 1986 Nov 20;324(6094):268–270. doi: 10.1038/324268a0. [DOI] [PubMed] [Google Scholar]
- Lefranc M. P., Forster A., Rabbitts T. H. Rearrangement of two distinct T-cell gamma-chain variable-region genes in human DNA. 1986 Jan 30-Feb 5Nature. 319(6052):420–422. doi: 10.1038/319420a0. [DOI] [PubMed] [Google Scholar]
- Lefranc M. P., Rabbitts T. H. Two tandemly organized human genes encoding the T-cell gamma constant-region sequences show multiple rearrangement in different T-cell types. Nature. 1985 Aug 1;316(6027):464–466. doi: 10.1038/316464a0. [DOI] [PubMed] [Google Scholar]
- Minden M. D., Mak T. W. The structure of the T cell antigen receptor genes in normal and malignant T cells. Blood. 1986 Aug;68(2):327–336. [PubMed] [Google Scholar]
- Minden M. D., Toyonaga B., Ha K., Yanagi Y., Chin B., Gelfand E., Mak T. Somatic rearrangement of T-cell antigen receptor gene in human T-cell malignancies. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1224–1227. doi: 10.1073/pnas.82.4.1224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadler L. M., Korsmeyer S. J., Anderson K. C., Boyd A. W., Slaughenhoupt B., Park E., Jensen J., Coral F., Mayer R. J., Sallan S. E. B cell origin of non-T cell acute lymphoblastic leukemia. A model for discrete stages of neoplastic and normal pre-B cell differentiation. J Clin Invest. 1984 Aug;74(2):332–340. doi: 10.1172/JCI111428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelicci P. G., Knowles D. M., 2nd, Dalla Favera R. Lymphoid tumors displaying rearrangements of both immunoglobulin and T cell receptor genes. J Exp Med. 1985 Sep 1;162(3):1015–1024. doi: 10.1084/jem.162.3.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pittaluga S., Raffeld M., Lipford E. H., Cossman J. 3A1 (CD7) expression precedes T beta gene rearrangements in precursor T (lymphoblastic) neoplasms. Blood. 1986 Jul;68(1):134–139. [PubMed] [Google Scholar]
- Quertermous T., Strauss W. M., Van Dongen J. J., Seidman J. G. Human T cell gamma chain joining regions and T cell development. J Immunol. 1987 Apr 15;138(8):2687–2690. [PubMed] [Google Scholar]
- Rabbitts T. H., Forster A., Milstein C. P. Human immunoglobulin heavy chain genes: evolutionary comparisons of C mu, C delta and C gamma genes and associated switch sequences. Nucleic Acids Res. 1981 Sep 25;9(18):4509–4524. doi: 10.1093/nar/9.18.4509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raulet D. H., Garman R. D., Saito H., Tonegawa S. Developmental regulation of T-cell receptor gene expression. Nature. 1985 Mar 7;314(6006):103–107. doi: 10.1038/314103a0. [DOI] [PubMed] [Google Scholar]
- Reinherz E. L., Kung P. C., Goldstein G., Levey R. H., Schlossman S. F. Discrete stages of human intrathymic differentiation: analysis of normal thymocytes and leukemic lymphoblasts of T-cell lineage. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1588–1592. doi: 10.1073/pnas.77.3.1588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Royer H. D., Acuto O., Fabbi M., Tizard R., Ramachandran K., Smart J. E., Reinherz E. L. Genes encoding the Ti beta subunit of the antigen/MHC receptor undergo rearrangement during intrathymic ontogeny prior to surface T3-Ti expression. Cell. 1984 Dec;39(2 Pt 1):261–266. doi: 10.1016/0092-8674(84)90003-5. [DOI] [PubMed] [Google Scholar]
- Saito H., Kranz D. M., Takagaki Y., Hayday A. C., Eisen H. N., Tonegawa S. A third rearranged and expressed gene in a clone of cytotoxic T lymphocytes. Nature. 1984 Nov 1;312(5989):36–40. doi: 10.1038/312036a0. [DOI] [PubMed] [Google Scholar]
- Saito H., Kranz D. M., Takagaki Y., Hayday A. C., Eisen H. N., Tonegawa S. Complete primary structure of a heterodimeric T-cell receptor deduced from cDNA sequences. 1984 Jun 28-Jul 4Nature. 309(5971):757–762. doi: 10.1038/309757a0. [DOI] [PubMed] [Google Scholar]
- Sangster R. N., Minowada J., Suciu-Foca N., Minden M., Mak T. W. Rearrangement and expression of the alpha, beta, and gamma chain T cell receptor genes in human thymic leukemia cells and functional T cells. J Exp Med. 1986 Jun 1;163(6):1491–1508. doi: 10.1084/jem.163.6.1491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snodgrass H. R., Dembić Z., Steinmetz M., von Boehmer H. Expression of T-cell antigen receptor genes during fetal development in the thymus. Nature. 1985 May 16;315(6016):232–233. doi: 10.1038/315232a0. [DOI] [PubMed] [Google Scholar]
- Snodgrass H. R., Kisielow P., Kiefer M., Steinmetz M., von Boehmer H. Ontogeny of the T-cell antigen receptor within the thymus. Nature. 1985 Feb 14;313(6003):592–595. doi: 10.1038/313592a0. [DOI] [PubMed] [Google Scholar]
- Tawa A., Hozumi N., Minden M., Mak T. W., Gelfand E. W. Rearrangement of the T-cell receptor beta-chain gene in non-T-cell, non-B-cell acute lymphoblastic leukemia of childhood. N Engl J Med. 1985 Oct 24;313(17):1033–1037. doi: 10.1056/NEJM198510243131701. [DOI] [PubMed] [Google Scholar]
- Waldmann T. A., Davis M. M., Bongiovanni K. F., Korsmeyer S. J. Rearrangements of genes for the antigen receptor on T cells as markers of lineage and clonality in human lymphoid neoplasms. N Engl J Med. 1985 Sep 26;313(13):776–783. doi: 10.1056/NEJM198509263131303. [DOI] [PubMed] [Google Scholar]
- Yanagi Y., Yoshikai Y., Leggett K., Clark S. P., Aleksander I., Mak T. W. A human T cell-specific cDNA clone encodes a protein having extensive homology to immunoglobulin chains. Nature. 1984 Mar 8;308(5955):145–149. doi: 10.1038/308145a0. [DOI] [PubMed] [Google Scholar]
- Yancopoulos G. D., Blackwell T. K., Suh H., Hood L., Alt F. W. Introduced T cell receptor variable region gene segments recombine in pre-B cells: evidence that B and T cells use a common recombinase. Cell. 1986 Jan 31;44(2):251–259. doi: 10.1016/0092-8674(86)90759-2. [DOI] [PubMed] [Google Scholar]