Abstract
Treatment of peripheral nerve injuries remains a challenge to modern medicine due to the complexity of the neurobiological nerve regenerating process. There is a greater challenge when the transected nerve ends are not amenable to primary end-to-end tensionless neurorraphy. When facing a segmental nerve defect, great effort has been made to develop an alternative to the autologous nerve graft in order to circumvent morbidity at donor site, such as neuroma formation, scarring and permanent loss of function. Tubolization techniques have been developed to bridge nerve gaps and have been extensively studied in numerous experimental and clinical trials. The use of a conduit intends to act as a vehicle for moderation and modulation of the cellular and molecular ambience for nerve regeneration. Among several conduits, vein tubes were validated for clinical application with improving outcomes over the years. This article aims to address the investigation and treatment of segmental nerve injury and draw the current panorama on the use of vein tubes as an autogenous nerve conduit.
Keywords: peripheral nerve injury, nerve graft, nerve conduit, Wallerian degeneration, neurotrophic factors, veins, autografts, nerve regeneration
Peripheral nerve injuries are a common entity in the treatment of hand disorders. They are most commonly related to direct mechanical trauma, and can be associated with other soft and bone tissue lesions (Taylor et al., 2008). Nerve regeneration is a complex process that has been extensively studied. The peripheral nervous system has an intrinsic ability for regeneration and repair. However, despite this regenerative potential, function is rarely restored spontaneously after an injury to the human peripheral nerve (Mackinnon et al., 1988; Brushart, 2011).
Transection of axons implicates in morphologic and metabolic changes throughout the whole length of the neuron. From the moment of injury, the cell body is signalized and the regeneration process starts (Hanz et al., 2006). While the distal nerve stump undergoes Wallerian degeneration, the proximal stump retracts and the Schwann cells undergo apoptosis (Maggi et al., 2003). The axon sprouts and a growth cone is formed at the tip of each sprout, interacting with activated and proliferating Schwann cells (Geuna et al., 2009). The nerve injury itself results in decreasing of motoneurons in the spinal cord. In an experimental study from Grecco et al. (2003) with Wistar rats, the number of motoneurons labeled in the spinal cord decreased an average of 20% due to an isolated tibial nerve injury, with no technical interference by the surgeon.
Treatment of peripheral nerve injury is a challenge to modern medicine. Capacity of regeneration is related to the age of the patient, mechanism of injury, level of the injury, and local biologic factors, such as the extent and severity at the injury site (Lee et al., 2000), although predicting in which cases a good or insufficient nerve regeneration can be expected is not possible with present studies (Paprottka et al., 2013). The presence of devitalized tissue, hematoma, or bacterial contamination impairs the process of regeneration, as well as the decision of the optimal timing of nerve repair. Even with the improvement of microsurgical techniques, functional recovery is not complete, even with early primary repair, due to insufficient nerve regeneration, loss of motor and sensitive neurons, mismatch between fibers and topographic changes on target organs (Wiberg et al., 2002). Newer publications on digital nerve injuries following end-to-end coaptation demonstrated significantly better sensory recovery outcomes than older ones (Paprottka et al., 2013).
There is a greater challenge when the transected nerve ends are not amenable to primary end-to-end tensionless neurorraphy, either by retraction of the fibers or by trauma with loss of substance. An experimental study demonstrated that epineural neurorraphy within a vein tube performed without a gap between nerve ends showed better results for nerve regeneration than the same procedure with a gap (Santos et al., 2012). This may demonstrate the difficulty in providing adequate regeneration when a segmental defect is present, maybe due to axon and fiber mismatch and lesser neurotrophic factors stimulation and release when there is no contact between nerve ends.
When surgeons face this scenario, most surgeons worldwide still rely upon an autologous nerve graft for restoring continuity of the nerve. However, autologous nerve grafting is associated with morbidity at donor site, including neuroma formation, scarring and permanent loss of function. Furthermore, donor nerves have limited availability and are often of small caliber, resulting in mismatch between nerve and graft dimensions (Battiston et al., 2005; Pabari et al., 2009; Rinker et al., 2011; Griffin et al., 2013; Paprottka et al., 2013). When a nerve graft was used in a rat model, a loss of approximately 35% of motoneurons was found, when compared to the control group. Thus, the nerve graft may act blocking partially the migration of regenerating axons, added to the possible death of motoneurons caused by the injury itself (Grecco et al., 2003). Therewith, functional results following nerve grafts remain far from ideal. In a recent meta-analysis on digital nerve repair, Paprottka et al. (2013) collected data from 87 publications, with a total of 2,997 nerve repairs. With nerve graft the proportion of good sensory recovery outcome was 42% (95% confidence interval: 33–52%) and excellent in 25% (95% confidence interval: 15–37%). The study aimed to compare the outcomes of digital nerve repair with different methods, including end-to-end and end-to-side coaptations, nerve grafts, artificial conduit, vein, muscle, and muscle-in-vein reconstructions, and replantations. No specific technique showed significant superiority in providing a good/excellent outcome, despite the heterogeneity of the publications included. In conclusion, the authors claim that surgical decision making should be based on clinical experience and personal preference of the surgeon, based on the gap length and donor site morbidity caused by graft material harvesting.
To circumvent these problems, tremendous interest in developing an alternative to nerve autograft has emerged over the last three decades. In order to bridge nerve gaps, tubolization techniques have been developed and favorably applied for repair of gap lesions of digital or mixed nerves (Weber et al., 2005; Pabari et al., 2010; Ray et al. 2010; Boyd et al., 2011). The use of a conduit as a vehicle for moderation and modulation of the cellular and molecular ambience for nerve regeneration has been widely investigated (Meek et al., 2002; Konofaos et al., 2013; Lin et al., 2013; Pabari et al., 2014; Rinker et al., 2014).
Tubular materials intent to connect nerve stumps, contain neural tissue, create a microenvironment in which neurotrophic and neurotropic factors can act, guiding the regenerating axons, while protecting the nerve from the surrounding tissue. Combination of biological, physical and chemical factors has made the study of nerve tubes a complex process, rising tremendous interest in the fields of medicine, biomedicine and bioengineering. The ideal tubular material has not yet been established. Several materials, either of biologic origin or synthetically fabricated, have been applied for these purposes. The possibility of connecting nerve stumps with a synthetic conduit has been subject of intense analysis over the past few decades (Fields et al., 1989; Wang et al., 1993; Battiston et al., 2005; Pabari et al., 2009, 2014; Ray et al., 2010; Boyd et al., 2011; Lin et al., 2013). The ideal conduit would be made of a low-cost, biologically inert material that is, biocompatible, thin, flexible, transparent, inhibitor of inflammatory processes such fibrosis, gliomas, neuromas, swelling, ischemia, and adhesions, and facilitator of the processes that contribute to regeneration, accumulating of factors that promote nerve growth (Brunelli et al., 1994). Currently, there are seven U.S. Food and Drug Administration/Conformit Europe-approved synthetic nerve conduits, produced primarily from biodegradable materials, such as woven polyglycolic acid (PGA), type I collagen, poly-DL-lactide-caprolactone (PCL), among others (Pabari et al., 2014). However, although synthetic conduits are convenient and time saving, they add expense to the surgical procedure and carry the risk of foreign body reaction and extrusion.
Since the first attempts of using vein grafts for bridging nerve defects in humans by Wrede (1909), which successfully repaired a median nerve of a male patient with a 45-mm-long vein tube, satisfactory functional results have been obtained in cases of repair of distal sensory nerve defects of 3 cm or less. Although arteries, muscles and tendons were studied for bridging nerve defects, vein tubes has received the most attention among researchers (Chiu et al., 1982; Ristiano et al., 1989; Walton et al., 1989). There are several advantages to using vein conduits. The tissue composition of veins is similar to that of nerve tissue. The basal lamina, which constitutes the endothelium of the vein, is rich in laminin, as well as the media while the adventitia is composed primarily of collagen. The vein walls are resilient enough to act as a barrier against scar ingrowth and have the permeability to allow diffusion of the proper nutrients (Wang et al., 1993; Tseng et al., 2003). Proponents cite the ample supply for harvesting vein grafts from many areas of the body, the low cost, and the relatively minor degree of additional injury compared with nerve grafts. Moreover, veins are nonimmunogenic, cause less inflammatory reaction, and are available in a wide variety of sizes. They also can provide a mechanical support for the regenerating axonal cone, offering a protected biochemical milieu, away from surrounding tissue (Allet et al., 2003).
Vein tubes were validated for clinical application as autogenous nerve conduits with improving outcomes over the years (Chiu et al., 1990, 1999; Tang et al., 1993). When comparing direct repair, conventional nerve grafting and autogenous vein grafts as nerve grafts in segmental nerve injuries of 3 cm or less, Chiu et al. (1990) observed uniformly significant symptom relief and satisfactory sensory function, with a superior two-point discrimination in the direct repair group and probably in the conventional nerve grafting patients. The same author published a review (Chiu, 1999) reaffirming the effectiveness of autogenous venous nerve conduits for reconstruction of peripheral nerve injuries with a gap of 3 cm or less, and foresees the use of vein tubes as a vehicle for moderation and modulation of the cellular and molecular ambience for nerve regeneration. When comparing biological conduits, Rinker et al. (2011) found that sensory recovery after nerve reconstruction using autogenous vein tubes was equivalent to that with PGA conduit in a prospective randomized clinical trial with a mean nerve gap of 10 mm (gap lengths: 4–25 mm), with similar cost profile and fewer postoperative complications. Furthermore, a meta-analysis demonstrated that, for gap lengths up to 4 cm, vein reconstructions did not reveal any significant difference in sensory recovery outcome when compared to artificial conduits (Paprottka et al., 2013).
According to Tseng et al. (2003), histologic analysis revealed that the process of nerve regeneration through an autologous vein conduit occurs in four phases: the hematoma phase, cellular migration phase, axonal advancement phase, and myelination and maturation phase. An experimental study by Raimondo et al. (2005) enlightened the understanding of the behavior of proliferating glial cells along a combined muscle-vein autograft. The skeletal muscle fibers not only avoid the vein collapse but also assist axon regrowth and Schwann cell migration by scaffolding these migrating cells in the basal lamina, even without preliminary degeneration. Axonal growth into the conduit lags behind Schwann-cell migration, which is a prompt phenomenon. Perineurial cells are present in a large amount inside graft conduits and begin to envelop axons and Schwann cells, generating an appropriate endoneurial environment. Furthermore, a small number of macrophages inside the tube may be related to the absence of degenerating axons.
Although vein conduits have shown potential for nerve repair, opponents are concerned with the possibility of lumen collapse possibly impending nerve regeneration. However, it has been shown in experimental studies that the lumen of the vein conduit remains patent throughout the process of nerve regeneration (Tseng et al., 2003). Furthermore, the conduit length and nerve diameter are being recognized as variables for clinical outcome in nerve reconstructions with tubes (Moore et al., 2009). Another disadvantage of vein graft is the presence of valves inside the lumen. They can provide obstruction of regenerating axons and result in neuroma formation. Therefore, valveless sections of donor veins should be elected or the vein conduit can be pulled inside-out to invert the normal orientation and place the adventitial layer within the lumen as described by Wang et al. (1993, 1995). The authors investigated the regenerating process of sciatic nerve injuries with a 10 mm gap in rats, demonstrating that regeneration on the inside-out vein graft side had superior functional results (faster conduction velocities) and improved histological results (greater axon counts) compared with the nerve grafted side. This technique provides contact between the regenerating axons and the adventitial surface, providing an environment rich with collagen, laminin, and Schwann cells, increasing vascularization and providing improved histological and functional results.
Filling the vein tube intents to limit the risk of collapse (Figure 1). The use of nerve or muscle as intraluminal additives has proven benefits of supplying neurotrophic factors and extracellular matrix, facilitating nerve regeneration by promoting Schwann cell migration, cell proliferation, and guidance of the axonal growth cone (Brunelli et al., 1993; Raimondo et al., 2005). Efforts to limit the risk of vein collapse and provide biological additives for nerve regeneration have led to the filling of vein grafts with a diverse range of substances (Figure 2).
Figure 1.
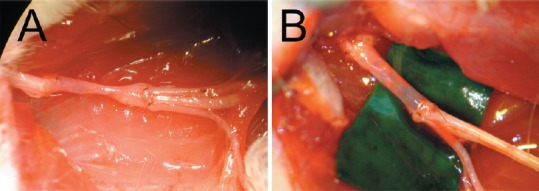
The empty vein graft tends to collapse (A). Filling the conduit with a diverse range of substances (B) intents to provide scaffold and enhance the nerve regenerating process (Sabongi et al., 2014).
Figure 2.
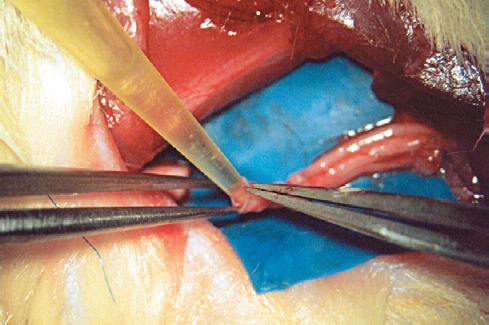
Filling material implanted in an autologous vein graft is used as a nerve conduit (Sabongi et al., 2014).
Muscle-vein-combined graft conduits have been broadly devised and effectively employed for segmental nerve injuries. In addition to preventing vein collapse, a combined muscle-in-vein autograft supplies a good pathway for regenerating axons and Schwann-cell migration, while vein walls prevent dispersion of muscle fibers, scar tissue invasion and provide a good blood supply (Raimondo et al., 2005). Experimental study in rat's tibial nerve injury with a 8 mm gap, Fernandes et al. (2007) demonstrated encouraging outcomes with vein grafts filled with degenerated muscle, with similar results as nerve graft repair when comparing the number of spinal cord motoneurons positive for retrograde tracer. Another experimental study in rats by Geuna et al. (2003) investigated the histological activity of Schwann cells in sciatic nerve injuries with a gap bridged with a vein graft filled with muscle. The authors reported a progressive invasion of Schwann cells immunopositive for glial fibrillar acid protein, a protein that is specifically expressed in glial cell; and proliferating cell nuclear antigen, a protein that is expressed by cells during DNA synthesis. These findings suggest that cell proliferation continues to occur inside the combined vein graft, and that a favorable environment that can potentiate the early regeneration phases is established. Manoli et al. (2014) conducted a retrospective clinical trial in order to compare regeneration results after digital nerve reconstruction with muscle-in-vein conduits, nerve autografts, or direct suture. In a total of 46 patients with 53 digital nerve injuries with a segmental nerve injury ranging between 1 and 6 cm, no statistically significant differences between all three groups could be found. The authors also emphasized that after harvesting a nerve graft, reduction of sensibility at the donor site occurred in 10 of 14 cases but only in one case after harvesting a muscle-in-vein conduit. Other attempts in maintaining the conduit patency were investigated by using minced nerve tissue inside the lumen. In an experimental sudy by Sahin et al. (2014), a 1 cm tibial nerve defect was repaired by vein graft filled with minced nerve tissue. The authors found no difference in functional results (with tibial functional index) and number of myelinated fibers between nerve graft and minced nerve groups. They concluded that this enriched vein graft technique could be applied as an alternative to nerve grafts in different clinical scenarios, such as using the nerve tissue at the injury site to fill the vein; using a smaller nerve graft to provide nerve fragments for longer gaps; or repair two nerve defects with a single nerve graft.
Neurotrophic factors or other pharmacological aids, extra-cellular matrix proteins, transplantable cells, and hydrogels or 3D scaffolds have been postulated to enrich the nerve guide, in an effort to surpass the histological and functional results of the nerve graft repair (Faroni et al., 2014). Several small molecules, peptides, hormones and growth factors have been suggested as potential candidates to improve the regenerating process and limit neuronal death following the injury. Fibroblast growth factor (FGF) was able to improve the neuronal count in the spinal cord of rats following segmental nerve repair (Hirakawa et al., 2007). Topically administered betamethasone inside the vein conduit lumen also resulted in improvement of functional recovery and quantitative morphometric indices of sciatic nerve segmental injuries in rats (Mohammadi et al., 2013). Similarly, platelet-rich plasma (PRP) was used as a gelled material to scaffold the vein tube and enhance nerve regeneration, resulting in improved functional outcome in a rat model (Kim et al., 2014; Sabongi et al., 2014).
With modern advances in regenerative medicine, great hope is placed in the potential of cell-based therapies. The theoretical benefit of increasing the number of Schwann cells inside the lumen is based in the pathophysiology of nerve regeneration and the microenvironment through which regenerating axons reach their peripheral targets. Measures to increase these cells migration and mitosis, such as Schwann-cell-derived growth factors failed to provide the same improvement than directly injecting autogenous Schwann cells inside nerve conduits. The incorporation of Schwann cells has been beneficial and detailed investigation endorses the advantages. This strategy may overcome the gap length barrier of 3 cm currently recommended for clinical use of vein tubes (Chiu et al., 1990; Boyd et al., 2011; Griffin et al., 2013). Strauch et al. (2001) demonstrated excellent nerve regeneration through a 6cm autogenous venous nerve conduit with the addition of autologous Schwann cells in segmental injuries of peroneal nerves in rabbits. Likewise, Zhang et al. (2002) evaluated the regeneration of a 4 cm gap in a rabbit tibial nerve model, comparing a vein graft alone, vein graft with intraluminal injection of Schwann cell suspension and autogenous vein graft. The authors found no nerve regrotwh in the empty vein subjects, while the formation of new nerve fascicles with myelination was confirmed within veins filled with Schwann cells. Electrophysiologic evaluation showed a faster average motor nerve conduction velocity in the enriched vein graft group compared to the nerve grafting group. Recent progressions in bioengineering Schwann-cell enriched conduits and cultivation of Schwann cells may push even further the boundaries of gap length, improving morphological and functional restoration of the nerve by facilitating the nerve regenerating process.
The addition of stem cells to the nerve conduit, expecting their differentiation into neurons and glia has also been researched. Adipose or bone marrow-derived mesenchymal stem cells can be induced to differentiate into Schwann-like cells. Fernandes et al. (2008) compared the outcomes of nerve autografts and venous grafts containing mononuclear bone marrow cells in sciatic nerve-lesioned rats. Results demonstrated a significantly higher vessel density in the bone marrow enriched vein grafts. This is attributable, in part, to the ability of these cells to secrete molecules that participate in angiogenesis, such as fibroblast growth factor, vascular endothelial growth factor, as well as a range of cytokines. Promoting angiogenesis is expected to be favorable for axon growth, since peripheral nerves are well-vascularized structures.
Although the superiority of filled vein conduits to autograft has not been established, positive outcomes have been demonstrated suggesting that there is some evidence that the composite conduit is more effective than vein alone in the clinical setting (Manoli et al., 2014). The treatment of critical nerve gaps continues to be an active area of clinical and scientific inquiry. Currently, no clear advantages of a specific surgical technique for digital nerve repair could be proven (Griffin et al., 2013; Paprottka et al., 2013). Therewith, direct tension-free nerve repair is still the method of choice, and for extended nerve defects, different techniques seem feasible for bridging the gap. Decision-making has to be based upon the peculiarities of each method, surgical experience, and clinical setting.
The main goal of restoring normal function after a nerve injury, especially when a segmental defect is present, is prospering as we progress in understanding the complex biologic process of nerve injury and regeneration. This will yield inductive strategies to harness and refine the nerve's innate regenerative ability. The quest for alternatives in the treatment of nerve injuries with a gap have led to remarkable progress in many areas of knowledge, already providing a large benefit for the development of surgical techniques, tissue engineering, cell therapy, and neurobiology. Since the first description of suturing the nerve stumps of a transected nerve by Gabriele Ferrara (Artico et al., 1996), peripheral nerve regeneration treatment has evolved to a greater understanding of the neurobiological process of nerve injury and refined microsurgical techniques. Several clinical and experimental essays indicate that there may be viable alternatives to successfully replace the traditional nerve grafts with conduits, such as vein autografts. Future perspectives aim in a combined approach to the regenerating process, focusing not only in a scaffold that can support growing axons but also improve Schwann cell migration and deliver growth-promoting factors inside the lumen. As novel interventions and techniques develop in other fields, this may lead to a breakthrough on the treatment of lesions in the peripheral nervous system, and perhaps revolutionize all nerve injury care.
References
- Allet MA, Leite VM, Albertoni WA, Fernandes FAS, Fernandes M, Faloppa F. Graft versus graft covered with vein conduits in nerve repairs. Rev Bras Ortop. 2003;38:193–200. [Google Scholar]
- Artico M, Cervoni L, Nucci F, Giuffré R. Birthday of peripheral nervous system surgery: the contribution of Gabriele Ferrara (1543-1627) Neurosurgery. 1996;39:380–383. doi: 10.1097/00006123-199608000-00030. [DOI] [PubMed] [Google Scholar]
- Battiston B, Geuna S, Ferrero M, Tos P. Nerve repair by means of tubulization: literature review and personal clinical experience comparing biological and synthetic conduits for sensory nerve repair. Microsurgery. 2005;25:258–267. doi: 10.1002/micr.20127. [DOI] [PubMed] [Google Scholar]
- Boyd KU, Nimigan AS, Mackinnon SE. Nerve reconstruction in the hand and upper extremity. Clin Plast Surg. 2011;38:643–660. doi: 10.1016/j.cps.2011.07.008. [DOI] [PubMed] [Google Scholar]
- Brunelli GA, Battiston B, Vigasio A, Brunelli G, Marocolo D. Bridging nerve defects with combined skeletal muscle and vein conduits. Microsurgery. 1993;14:247–251. doi: 10.1002/micr.1920140407. [DOI] [PubMed] [Google Scholar]
- Brunelli GA, Vigasio A, Brunelli GR. Different conduits in peripheral nerve surgery. Microsurgery. 1994;15:176–178. doi: 10.1002/micr.1920150307. [DOI] [PubMed] [Google Scholar]
- Brushart TM. New York; Oxford: Oxford University Press; 2011. Nerve Repair. [Google Scholar]
- Chiu DT, Janecka I, Krizek TJ, Wolff M, Lovelace RE. Autogenous vein graft as a conduit for nerve regeneration. Surgery. 1982;91:226–233. [PubMed] [Google Scholar]
- Chiu DT. Autogenous venous nerve conduits. A review. Hand Clin. 1999;15:667–671. [PubMed] [Google Scholar]
- Chiu DTW, Strauch B. A prospective clinical evaluation of autogenous vein grafts used as a nerve conduit for distal sensory nerve defects of 3 cm or less. Plast Reconstr Surg. 1990;86:928–934. doi: 10.1097/00006534-199011000-00015. [DOI] [PubMed] [Google Scholar]
- Faroni A, Mobasseri SA, Kingham PJ, Reid AJ. Peripheral nerve regeneration: experimental strategies and future perspectives. Adv Drug Deliv Rev. 2014 doi: 10.1016/j.addr.2014.11.010. doi: 10.1016/j.addr.2014.11.010. [DOI] [PubMed] [Google Scholar]
- Fernandes M, Valente SG, Amado D, Fernandes MJDS, Naffah-Mazzacoratti MDG, Santos JBGD, Faloppa F, Leite VM. Comparative study between autogenous graft and muscular graft covered with autogenous vein tube in wistar rats’ tibial nerves using the fluoro-gold® as a neuronal marker. Acta Ortop Bras. 2007;15:97–100. [Google Scholar]
- Fernandes M, Valente SG, Fernandes MJ, Félix EP, Mazzacoratti Mda G, Scerni DA, dos Santos JB, Leite VM, Faloppa F. Bone marrow cells are able to increase vessels number during repair of sciatic nerve lesion. J Neurosci Methods. 2008;170:16–24. doi: 10.1016/j.jneumeth.2007.12.009. [DOI] [PubMed] [Google Scholar]
- Fields RD, Le Beau JM, Longo FM, Ellisman MH. Nerve regeneration through artificial tubular implants. Prog Neurobiol. 1989;33:87–134. doi: 10.1016/0301-0082(89)90036-1. [DOI] [PubMed] [Google Scholar]
- Geuna S, Raimondo S, Nicolino S, Boux E, Fornaro M, Tos P, Battiston B, Perroteau I. Schwann-cell proliferation in muscle-vein combined conduits for bridging rat sciatic nerve defects. J Reconstr Microsurg. 2003;19:119–123. doi: 10.1055/s-2003-37818. [DOI] [PubMed] [Google Scholar]
- Geuna S, Raimondo S, Ronchi G, Di Scipio F, Tos P, Czaja K, Fornaro M. Chapter 3: histology of the peripheral nerve and changes occurring during nerve regeneration. Int Rev Neurobiol. 2009;87:27–46. doi: 10.1016/S0074-7742(09)87003-7. [DOI] [PubMed] [Google Scholar]
- Grecco MAS, Leite VM, Albertoni WM, Santos JBGD, Hirakawa CK, Faloppa F, Valente SG. A study on nerve regeneration in tibial nerves of wistars rats, using Fluoro-Gold® as a neuronal tracer. Acta Ortop Bras. 2003;11:225–229. [Google Scholar]
- Griffin JW, Hogan MV, Chhabra AB, Deal DN. Peripheral nerve repair and reconstruction. J Bone Joint Surg Am. 2013;95:2144–2151. doi: 10.2106/JBJS.L.00704. [DOI] [PubMed] [Google Scholar]
- Hanz S, Fainzilber M. Retrograde signaling in injured nerves: the axon reaction revisited. J Neurochem. 2006;99:13–19. doi: 10.1111/j.1471-4159.2006.04089.x. [DOI] [PubMed] [Google Scholar]
- Hirakawa CK, Grecco MAS, Leite VM, Faloppa F. Fibroblasts growth factor and nerve fragments effect on tibial nerve regeneration in rats: a comparative study. Acta Ortop Bras. 2007;15:114–117. [Google Scholar]
- Kim JY, Jeon WJ, Kim DH, Rhyu IJ, Kim YH, Youn I, Park JW. An inside-out vein graft filled with platelet-rich plasma for repair of a short sciatic nerve defect in rats. Neural Regen Res. 2014;9:1351–1357. doi: 10.4103/1673-5374.137587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konofaos P, Ver Halen JP. Nerve repair by means of tubulization: past, present, future. J Reconstr Microsurg. 2013;29:149–164. doi: 10.1055/s-0032-1333316. [DOI] [PubMed] [Google Scholar]
- Lee SK, Wolfe SW. Peripheral nerve injury and repair. J Am Acad Orthop Surg. 2000;8:243–252. doi: 10.5435/00124635-200007000-00005. [DOI] [PubMed] [Google Scholar]
- Lin MY, Manzano G, Gupta R. Nerve allografts and conduits in peripheral nerve repair. Hand Clin. 2013;29:331–348. doi: 10.1016/j.hcl.2013.04.003. [DOI] [PubMed] [Google Scholar]
- Mackinnon S, Dellon AL. New York: Thieme; 1988. Surgery of the peripheral nerve. [Google Scholar]
- Maggi SP, Lowe JB, Mackinnon SE. Pathophysiology of nerve injury. Clin Plast Surg. 2003;30:109–126. doi: 10.1016/s0094-1298(02)00101-3. [DOI] [PubMed] [Google Scholar]
- Manoli T, Schulz L, Stahl S, Jaminet P, Schaller HE. Evaluation of sensory recovery after reconstruction of digital nerves of the hand using muscle-in-vein conduits in comparison to nerve suture or nerve autografting. Microsurgery. 2014;34:608–615. doi: 10.1002/micr.22302. [DOI] [PubMed] [Google Scholar]
- Meek MF, Coert JH. Clinical use of nerve conduits in peripheral-nerve repair: review of the literature. J Reconstr Microsurg. 2002;18:97–109. doi: 10.1055/s-2002-19889. [DOI] [PubMed] [Google Scholar]
- Mohammadi R, Amini K, Eskafian H. Betamethasone-enhanced vein graft conduit accelerates functional recovery in the rat sciatic nerve gap. J Oral Maxillofac Surg. 2013;71:786–792. doi: 10.1016/j.joms.2012.08.009. [DOI] [PubMed] [Google Scholar]
- Moore AM, Kasukurthi R, Magill CK, Farhadi HF, Borschel GH, Mackinnon SE. Limitations of conduits in peripheral nerve repairs. Hand. 2009;4:180–186. doi: 10.1007/s11552-008-9158-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pabari A, Yang SY, Seifalian AM, Mosahebi A. Modern surgical management of peripheral nerve gap. J Plast Reconstr Aesthet Surg. 2010;63:1941–1948. doi: 10.1016/j.bjps.2009.12.010. [DOI] [PubMed] [Google Scholar]
- Pabari A, Lloyd-Hughes H, Seifalian AM, Mosahebi A. Nerve conduits for peripheral nerve surgery. Plast Reconstr Surg. 2014;133:1420–1430. doi: 10.1097/PRS.0000000000000226. [DOI] [PubMed] [Google Scholar]
- Paprottka FJ, Wolf P, Harder Y, Kern Y, Paprottka PM, Machens HG, Lohmeyer JA. Sensory recovery outcome after digital nerve repair in relation to different reconstructive techniques: meta-analysis and systematic review. Plast Surg Int. 2013 doi: 10.1155/2013/704589. doi: 10.1155/2013/704589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raimondo S, Nicolino S, Tos P, Battiston B, Giacobini‐Robecchi MG, Perroteau I, Geuna S. Schwann cell behavior after nerve repair by means of tissue‐engineered muscle-vein combined guides. J Comp Neurol. 2005;489:249–259. doi: 10.1002/cne.20625. [DOI] [PubMed] [Google Scholar]
- Ray WZ, Mackinnon SE. Management of nerve gaps: autografts, allografts, nerve transfers, and end-to-side neurorrhaphy. Exp Neurol. 2010;223:77–85. doi: 10.1016/j.expneurol.2009.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rinker B, Liau JY. A prospective randomized study comparing woven polyglycolic acid and autogenous vein conduits for reconstruction of digital nerve gaps. J Hand Surg Am. 2011;36:775–781. doi: 10.1016/j.jhsa.2011.01.030. [DOI] [PubMed] [Google Scholar]
- Rinker B, Vyas KS. Clinical applications of autografts, conduits, and allografts in repair of nerve defects in the hand: current guidelines. Clin Plast Surg. 2014;41:533–550. doi: 10.1016/j.cps.2014.03.006. [DOI] [PubMed] [Google Scholar]
- Risitano G, Cavallaro G, Lentini M. Autogenous vein and nerve grafts: a comparative study of nerve regeneration in the rat. J Hand Surg Br. 1989;14:102–104. doi: 10.1016/0266-7681(89)90027-2. [DOI] [PubMed] [Google Scholar]
- Sabongi RG, De Rizzo LA, Fernandes M, Valente SG, Gomes dos Santos JB, Faloppa F, Leite VM. Nerve regeneration: is there an alternative to nervous graft? J Reconstr Microsurg. 2014;30:607–616. doi: 10.1055/s-0034-1372477. [DOI] [PubMed] [Google Scholar]
- Sahin C, Karagoz H, Kulahci Y, Sever C, Akakin D, Kolbasi B, Ulkur E, Peker F. Minced nerve tissue in vein grafts used as conduits in rat tibial nerves. Ann Plast Surg. 2014;73:540–546. doi: 10.1097/SAP.0000000000000060. [DOI] [PubMed] [Google Scholar]
- Santos EBD, Fernandes M, Santos JBGD, Leite VM, Valente SG, Faloppa F. Study of tibial nerve regenration in Wistar rats in primary neurorrhaphy with and without gap, wrapped in vein segments. Acta Ortop Bras. 2012;20:165–169. doi: 10.1590/S1413-78522012000300006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauch B, Rodriguez DM, Diaz J, Yu HL, Kaplan G, Weinstein DE. Autologous Schwann cells drive regeneration through a 6-cm autogenous venous nerve conduit. J Reconstr Microsurg. 2001;17:589–595. doi: 10.1055/s-2001-18812. [DOI] [PubMed] [Google Scholar]
- Tang JB, Gu YQ, Song YS. Repair of digital nerve defect with autogenous vein graft during flexor tendon surgery in zone 2. J Hand Surg Br. 1993;18:449–453. doi: 10.1016/0266-7681(93)90144-5. [DOI] [PubMed] [Google Scholar]
- Taylor CA, Braza D, Rice JB, Dillingham T. The incidence of peripheral nerve injury in extremity trauma. Am J Phys Med Rehabil. 2008;87:381–385. doi: 10.1097/PHM.0b013e31815e6370. [DOI] [PubMed] [Google Scholar]
- Tseng CY, Hu G, Ambron RT, Chiu DT. Histologic analysis of Schwann cell migration and peripheral nerve regeneration in the autogenous venous nerve conduit (AVNC) J Reconstr Microsurg. 2003;19:331–340. doi: 10.1055/s-2003-42502. [DOI] [PubMed] [Google Scholar]
- Walton RL, Brown RE, Matory WE, Jr, Borah GL, Dolph JL. Autogenous vein graft repair of digital nerve defects in the finger: a retrospective clinical study. Plast Reconstr Surg. 1989;84:944–949. [PubMed] [Google Scholar]
- Wang KK, Costas PD, Bryan DJ, Jones DS, Seckel BR. Inside-out vein graft promotes improved nerve regeneration in rats. Microsurgery. 1993;14:608–618. doi: 10.1002/micr.1920140914. [DOI] [PubMed] [Google Scholar]
- Wang KK, Costas PD, Bryan DJ, Eby PL, Seckel BR. Inside-out vein graft repair compared with nerve grafting for nerve regeneration in rats. Microsurgery. 1995;16:65–70. doi: 10.1002/micr.1920160205. [DOI] [PubMed] [Google Scholar]
- Weber RV, Mackinnon SE. Bridging the neural gap. Clin Plast Surg. 2005;32:605–616. doi: 10.1016/j.cps.2005.05.003. [DOI] [PubMed] [Google Scholar]
- Wiberg M, Terenghi G. Will it be possible to produce peripheral nerves? Surg Technol Int. 2002;11:303–310. [PubMed] [Google Scholar]
- Wrede L. Uberbruckung eines nervendefektes mittels seidennhat und lebenden venenstuckes. Dtsch Med Wochenschr. 1909;35:1125. [Google Scholar]
- Zhang F, Blain B, Beck J, Zhang J, Chen Z, Chen ZW, Lineaweaver WC. Autogenous venous graft with one-stage prepared Schwann cells as a conduit for repair of long segmental nerve defects. J Reconstr Microsurg. 2002;18:295–300. doi: 10.1055/s-2002-30186. [DOI] [PubMed] [Google Scholar]


