Abstract
Oncogenesis frequently is accompanied by rampant genome instability, which fuels genetic heterogeneity and resistance to targeted cancer therapy. We have developed an approach that allows precise, quantitative measurement of genome instability in high-throughput format in the Saccharomyces cerevisiae model system. Our approach takes advantage of the strongly DNA damage-inducible gene RNR3, in conjunction with the reporter synthetic genetic array methodology, to infer mutants exhibiting genome instability by assaying for increased Rnr3 abundance. We screen for genome instability across a set of ~1000 essential and ~4200 nonessential mutant yeast alleles in untreated conditions and in the presence of the DNA-damaging agent methylmethane sulfonate. Our results provide broad insights into the cellular processes and pathways required for genome maintenance. Through comparison with existing genome instability screens, we isolated 130 genes that had not previously been linked to genome maintenance, 51% of which have human homologs. Several of these homologs are associated with a genome instability phenotype in human cells or are causally mutated in cancer. A comprehensive understanding of the processes required to prevent genome instability will facilitate a better understanding of its sources in oncogenesis.
Keywords: ribonucleotide reductase, DNA repair, DNA damage, DNA replication, genome instability
Genome maintenance is critical for cell viability and frequently is compromised in cancer. Loss of genome stability can be caused by exposure to sources of DNA damage, or defects in DNA repair or DNA replication genes. Genome instability can result in a diverse set of effects on DNA, including point mutations, small insertions and deletions, chromosome translocation or truncation, loss of heterozygosity, and even changes in ploidy (reviewed in Aguilera and Gomez-Gonzalez 2008; Storchova and Pellman 2004). Cancer cells frequently suffer from increased mutation rates and abnormal karyotypes, indicative of genome instability (reviewed in Vogelstein et al. 2013). In fact, genome instability underlies the genetic heterogeneity that stymies targeted cancer therapy (Glickman and Sawyers 2012; Vogelstein et al. 2013). Understanding the set of genes that function to maintain genome stability is critical to address the sources of genome instability in cancer. In somatic cancers especially, these sources remain unclear (Negrini et al. 2010).
Several genome-wide screens assaying genome instability have been conducted with the budding yeast S. cerevisiae (Alvaro et al. 2007; Alver et al. 2013; Andersen et al. 2008; Askree et al. 2004; Cheng et al. 2012; Huang et al. 2003; Stirling et al. 2011; Smith et al. 2004; Yu et al. 2006). Because genome instability has diverse consequences, these screens have used a wide array of assays. Many focus on the consequences of genome instability on DNA, by assaying loss of genetic markers, increased mutation rate, or changes in telomere length (Andersen et al. 2008; Askree et al. 2004; Huang et al. 2003; Stirling et al. 2011). Others focus on assaying the relocalization of DNA repair or DNA damage checkpoint proteins to nuclear foci, which indicates their active involvement in genome maintenance (Alvaro et al. 2007; Cheng et al. 2012). Another approach is to assay for activation of the DNA damage response (DDR) pathway, a signaling pathway that coordinates DNA repair processes with the cell-cycle (reviewed in Harrison and Haber 2006; Putnam et al. 2009). The DDR causes strong transcriptional induction of the gene RNR3, and assaying Rnr3 abundance has been used to identify genome maintenance genes (Zhou and Elledge 1992). However, the Rnr3 abundance screen has yet to be combined with modern reagents and methodology for yeast genetics, such as the nonessential deletion mutant collection (Giaever et al. 2002), and synthetic genetic array (SGA) technology (Tong and Boone 2006).
In S. cerevisiae, activation of the DDR begins when the single-stranded binding protein, RPA, coats exposed tracts of single-stranded DNA (ssDNA). Next, ssDNA-RPA is bound by Ddc2, in complex with the large phosphatidylinositol 3′ kinase-like kinase Mec1 (Rouse and Jackson 2002; Zou and Elledge 2003). Mec1 is activated subsequently through interactions with multiple nearby protein assemblies, the most prominent being the 9-1-1 (Mec3-Rad17-Ddc1) clamp (Majka et al. 2006; Navadgi-Patil and Burgers 2009). Another large phosphatidylinositol 3′ kinase-like kinase, Tel1, is uniquely recruited to and activated at double-stand breaks (Falck et al. 2005; Nakada et al. 2003). Activated Mec1 and Tel1 propagate the damage signal by hyperphosphorylating the signaling adaptor Rad9, which mediates phosphorylation of the transducing kinase Rad53 (Naiki et al. 2004; Schwartz et al. 2002; Sweeney et al. 2005). Rad53 amplifies the DNA damage signaling through extensive autophosphorylation (Sun et al. 1998). In addition, Rad53 phosphorylates downstream targets, communicating the presence of DNA damage to diverse cellular processes. Two well-understood effects of Rad53 activation are the inhibition of late origin firing during S-phase (Zegerman and Diffley 2010; Lopez-Mosqueda et al. 2010) and prevention of sister chromatid separation in anaphase (Sanchez et al. 1999; Agarwal et al. 2003), both of which allow time for DNA repair to occur.
Another well-studied effect of Rad53 activation is the transcriptional induction of the ribonucleotide reductase (RNR) complex, which catalyzes the rate-limiting step in dNTP synthesis. Many repair processes involve a DNA synthesis step and increased dNTP levels promote survival after DNA damage (Chabes et al. 2003). In S. cerevisiae, the RNR complex is encoded by two small (RNR2 and RNR4) and two large subunits (RNR1 and RNR3) (reviewed in Sanvisens et al. 2013). In a normal cell cycle, two Rnr1 subunits associate with Rnr2 and Rnr4 in an α2ββ’ architecture, with Rnr3 abundance being negligible. DDR activation induces the transcription of all four RNR subunits, albeit by different mechanisms and to different extents. RNR2, RNR3 and RNR4 contain 13bp damage response elements (DREs) in their promoters, which are bound by the transcriptional repressor Crt1/Rfx1 (Huang et al. 1998). Active Rad53 induces transcription of these RNR genes by phosphorylating the kinase Dun1, which in turn hyperphosphorylates Crt1, causing its dissociation from DNA (Huang et al. 1998). RNR1 lacks DREs in its promoter and is induced in a Mec1-Rad53−dependent but Dun1-independent manner that involves a different transcription factor, Ixr1 (Tsaponina et al. 2011).
Rnr3 is an ideal read-out of DDR pathway activation, and by extension genome instability. First, it is a well-characterized transcriptional target of the DNA damage response (Huang et al. 1998). RNR3 up-regulation in response to exogenous DNA-damaging agents like methylmethane sulfonate (MMS) has been demonstrated at both the mRNA and protein level and depends on known DDR kinases (Huang et al. 1998; Li and Reese 2001; Tsaponina et al. 2011). Second, mutation of several well-characterized DNA repair and replication genes leads to constitutive expression of RNR3, demonstrating that RNR3 can be induced by both exogenous (i.e., environmental) and endogenous (i.e., genetic perturbation) sources of genome instability (Tang et al. 2009; Davidson et al. 2012). Finally, expression of RNR3 is negligible in the absence of perturbation, but it is precipitously induced in response to DNA damage, by far the greatest induction of all the RNR genes (Elledge et al. 1993). The original Constitutive RNR Three (CRT) screen used an RNR3-URA3 transcriptional fusion and spontaneous mutagenesis as means to identify mutants causing genome instability (Zhou and Elledge 1992). We sought to complement this approach using modern yeast genetic tools, incorporating the Rnr3 assay into a comprehensive genome-wide screen.
We generated a fluorescent reporter for RNR3 expression compatible with reporter synthetic genetic array (R-SGA) technology, allowing us to systematically measure Rnr3 abundance across ~5200 yeast mutants (Kainth et al. 2009). We identified 150 mutants with increased Rnr3 abundance in the absence of exogenous perturbation and, in a second screen, 200 mutants with increased Rnr3 abundance in the presence of the DNA damaging agent MMS. We identified known repressors of RNR3 transcription, such as crt1∆, validating our reporter system and saw high enrichment for processes known to impinge upon genome maintenance, such as DNA replication and repair, validating the use of Rnr3 abundance as an effective indicator of genome instability. Furthermore, by systematic comparison with existing genome instability datasets, we determined that 133 mutants, representing 130 different genes, 51% of which have human orthologs, are unique to our Rnr3 read-out of genome instability,
Materials and Methods
Rnr3 R-SGA
Two independent crosses of GBY691 (MATα RNR3-GFP::HIS3MX leu2∆0 his3∆1 met15∆0 lyp1∆ can1pr::RPL39pr-tdTomato::CaURA3::can1∆::STE2pr-LEU2) with the yeast nonessential deletion collection and a set of conditional temperature-sensitive alleles of essential genes (G. Tan and C. Boone, unpublished data) were performed following standard SGA procedures (Tong and Boone 2006). Final arrays were pinned in duplicate on either SD/MSG –his –leu –ura +200 mg/mL G418 (untreated) or YPD supplemented with 0.03% MMS, and grown for 20 hr before fluorescence scanning. Media was poured 48 hr before use, allowing for drying sufficient to prevent condensation on the fluorescence scanner. The Typhoon Trio Variable Mode Imager (GE Healthcare) was used to acquire GFP (488-nm laser, 520/40 BP emission filter) and tdTomato (532-nm laser, 610/30 BP emission filter) fluorescence values. For the essential temperature-sensitive mutants, all growth was conducted at 23°. After fluorescence imaging, colony size data were acquired by individually photographing plates with a Canon PowerShot G2 4.0 megapixel digital camera using Remote Capture software. Data analysis followed essentially what is described in Kainth et al. (2009), with small variations. To summarize, background-subtracted green fluorescent protein (GFP) and tdTomato intensities were computed for each colony from .GEL images using GenePix Pro version 3.0 software. Colony size information was calculated from individual photographs using Qt ColonyImager software, version 1.0.1. Border colonies, small colonies (colony area < 500 pixels), and colonies with aberrantly low tdTomato values (bottom 0.05%) were removed before further analysis. log2(Rnr3-GFP/tdTomato) values were calculated and LOESS normalized for each replicate experiment. Using the log2(Rnr3-GFP/tdTomato) ratio as a metric for Rnr3 abundance has the advantage that dividing by tdTomato corrects for any colony size-dependent intensity effects. Finally, normalized log2(Rnr3-GFP/tdTomato) values were averaged across all replicate experiments and a Z-score calculated (Supporting Information, Table S1 and Table S2; Raw R-SGA data, Supporting Information, File S1). All analysis was performed in R.
Gene Ontology (GO) enrichment analysis
The GO Annotations Database was downloaded on September 23, 2014 (http://geneontology.org/page/download-annotations). A hypergeometric test was used to compute P-values for all GO terms within the biological process, molecular function and cellular component ontologies. GO terms with greater than 2000 associated genes were omitted as they are often too general to be informative. Similarly, for the biological process ontology, GO terms with less than 10 associated genes were omitted to remove very specific and largely redundant terms. P-values were adjusted for multiple hypothesis testing using the Benjamini-Hochberg method with a cut-off of FDR < 0.05.
Homology analysis
The inParanoid8 database for Saccharomyces cerevisiae to Homo sapiens homology was downloaded (http://inparanoid.sbc.su.se/cgi-bin/e.cgi) to determine yeast-human homology relationships. The set of human cancer genes casually mutated in cancer were downloaded from the Cancer Gene Consensus (http://cancer.sanger.ac.uk/cancergenome/projects/census/).
Results
A genome-wide screen for mutants with increased genome instability using Rnr3 abundance
RNR3 exhibits a strong and specific induction in response to genome instability in S. cerevisiae. We sought to adapt RNR3 expression into a reporter for genome instability that would be amenable to a genome-wide mutant screen. To this end, we adopted the R-SGA methodology (Kainth et al. 2009), which allows for high-throughput acquisition of the abundance of a protein of interest in the context of a yeast mutant collection. We used R-SGA to query Rnr3 abundance across the set of yeast nonessential deletion mutants, containing ~4200 open-reading frame deletions (Giaever et al. 2002), as well as a set of ~1000 kanMX-marked, temperature-sensitive alleles that cover ~600 essential genes (G. Tan and C. Boone, unpublished data).
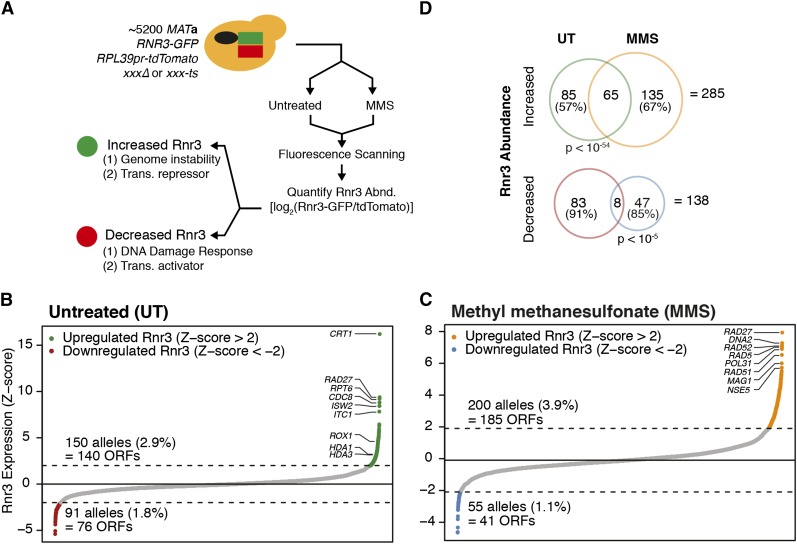
To conduct R-SGA, a strain containing endogenous RNR3 C-terminally tagged with GFP and expressing tdTomato constitutively from the RPL39 promoter was mated with both the non-essential gene deletion and conditional temperature-sensitive (TS) essential gene mutant collections using the SGA methodology (Tong and Boone 2006). The result was a collection of strains harboring RNR3-GFP, RPL39pr-tdTomato, and either a unique nonessential gene deletion (xxxΔ) or a TS essential gene allele (xxx-TS). Using a fluorescence scanner, we assayed the GFP and tdTomato intensities across the mutant strains arrayed as whole colonies on agar plates, and then we calculated a normalized log2(GFP/tdTomato) ratio, which is indicative of Rnr3 abundance. The screen was conducted in quadruplicate and on both untreated (UT) media and media containing the DNA-damaging agent MMS (Figure 1A). The average correlation across replicate screens was R = 0.73 and R = 0.95 for the nonessential deletion collection and temperature-sensitive collection, respectively. The correlations compare favorably with previous R-SGA screens, where an average of R = 0.77 was observed across 27 screens (Kainth et al. 2009).
Figure 1.
Assaying Rnr3 abundance across ~5200 yeast mutants, untreated (UT) and in methylmethane sulfonate (MMS). (A) Schematic of the Rnr3 R-SGA screening methodology. ~5200 yeast mutant strains containing RNR3-GFP and RPL39pr-tdTomato were either untreated or treated with MMS before fluorescence scanning and Rnr3 abundance quantification. Potential cellular functions of mutants with increased or decreased Rnr3 are described. (B) Distribution of Rnr3 abundances across all mutants screened in UT. (C) Distribution of Rnr3 abundance in MMS. (D) Overlap between UT and MMS screens for mutants with increased and decreased Rnr3 abundance. Both overlaps are significant. Total number of mutants unique to each condition is shown as a percentage, as well as the total number of mutants with increased and decreased Rnr3 across both conditions.
We hypothesized that mutants causing increased RNR3 expression in the absence of exogenous perturbation (untreated conditions) would unveil two classes of genes: (1) those directly regulating RNR3 transcription and (2) those involved in the maintenance of genome stability (Figure 1A). By further performing the Rnr3 R-SGA screen in the presence of MMS (an exogenous source of DNA damage), we aimed to isolate genome maintenance factors that function specifically in tolerance of MMS damage.
We used a Z-score based threshold to identify mutants with the greatest change in Rnr3 abundance, defining those mutants with Z > 2 (corresponding to an Rnr3 abundance two standard deviations above the mean) as having increased Rnr3 and those with Z < −2 as having decreased Rnr3. Using these thresholds, we identified 150 mutants (2.9% of those screened) with increased Rnr3 abundance in untreated conditions and 91 with decreased Rnr3 abundance (Figure 1B, and Table 1, Table S3, and Table S4). In MMS, we identified 200 mutants (3.9%) with increased Rnr3 and 55 mutants with decreased Rnr3 (Figure 1C, Table 1, Table S5, and Table S6). A larger fraction of mutants with increased Rnr3 abundance were nonessential (~66% for UT and MMS); however, a greater relative proportion of all essential mutants screened had increased Rnr3 abundance (e.g., ~34% of the 150 mutants with increased Rnr3 in UT are essential, although essential genes comprise only ~19% of the mutants screened). A tendency for mutation of essential genes to more readily cause genome instability is consistent with a previous study that similarly used both essential and nonessential mutant collections (Stirling et al. 2011).
Table 1. Evolutionary conservation of putative novel genome instability genes.
| Untreated | MMS | |||||
|---|---|---|---|---|---|---|
| Screened | Increased Rnr3 | Increased Rnr3 and Novel | Screened | Increased Rnr3 | Increased Rnr3 and Novel | |
| Genes | 5143 | 150 | 66 | 5121 | 200 | 85 |
| Orthologs | 1989 (39%) | 78 (52%) | 33 (50%) | 1973 (39%) | 106 (53%) | 44 (52%) |
| γH2AX | 356 (7%) | 11 (7%) | 3 (5%) | 352 (7%) | 17 (8.5%) | 6 (7%) |
| CC | 67 (1.3%) | 3 (2%) | 1 (1.5%) | 67 (1.3%) | 5 (2.5%) | 3 (3.5%) |
Gene mutants with an Rnr3 abundance Z-score >2 are indicated. Putative novel genome instability genes are defined in Figure 4. Orthologs were identified by the inParanoid algorithm. Genes with increased gH2AX foci following depletion were defined by Paulsen et al., 2009. Genes included in the Cancer Census are indicated by CC. MMS, methylmethane sulfonate.
Altogether, we identified a total of 285 mutants that cause increased Rnr3 abundance and 138 mutants with decreased Rnr3 abundance. Both the UT and MMS screen shared a significant number of mutants with increased and decreased Rnr3 levels. Nevertheless, we identified a large number of mutants unique to each condition (Figure 1D). Indeed, MMS treatment resulted in the identification of an additional 135 mutants with increased and 47 mutants with decreased Rnr3, indicating a change in the complement of genes that determine Rnr3 abundance when DNA damage is present.
Mutants with increased Rnr3 abundance
We first focused our attention on those mutants that exhibited an increase in Rnr3 abundance, which we expected to fall into two distinct functional classes: (1) those directly regulating RNR3 transcription and (2) those involved in the maintenance of genome instability.
In a normal cell cycle, RNR3 expression is inhibited by the transcriptional repressor Crt1/Rfx1, which binds to three DREs located upstream RNR3′s promoter (Huang et al. 1998). Consistent with this, a CRT1 deletion mutant had by far the greatest abundance of Rnr3 in our R-SGA assay conducted in untreated conditions (Figure 1B). In addition, several other genes with established connections to RNR transcriptional repression, including ISW2, HDA1, and ROX1 (Klinkenberg et al. 2006; Sharma et al. 2007; Zhang and Reese 2004), possessed an increased Rnr3 abundance upon deletion (Table S3). Our data also implicate ITC1, a binding partner of ISW2 (Goldmark et al. 2000), and HDA3, which forms a complex with HDA1 (Carmen et al. 1996), in the transcriptional repression of Rnr3, although this has not been explicitly demonstrated previously. For both complexes, Rnr3 induction upon deletion of either of the two constituent members was highly similar, suggesting that R-SGA can yield precise quantitative data on protein abundance. Together, these results confirm that the R-SGA methodology identifies known regulators of Rnr3 transcription.
Next, we sought to establish whether Rnr3 R-SGA could recover mutants belonging to our second expected class of hits: genes functioning to maintain genome stability. To take a broad and unbiased approach, we used the GO database, a curated set of terms describing known properties of the protein product of a gene. First, we examined the localization of genes whose mutation causes increased Rnr3 abundance. GO analysis using the cellular component ontology indicated that mutants with increased Rnr3 abundance are significantly enriched for genes that reside within the nucleus (Table S7). In fact, 79 of 140 genes with increased Rnr3 abundance have a nuclear localization, suggesting a direct role in genome maintenance. Analysis of the molecular function ontology supported the idea that many of the proteins with elevated Rnr3 abundance could directly influence genome stability. Namely, 31 genes possess direct DNA binding activity and 12 interact with chromatin (Table S7). Moreover, several genes contain domains that function to modify DNA topology, such as those possessing “DNA-dependent ATPase activity” (GO:0004003) and “helicase activity” (GO:0004386).
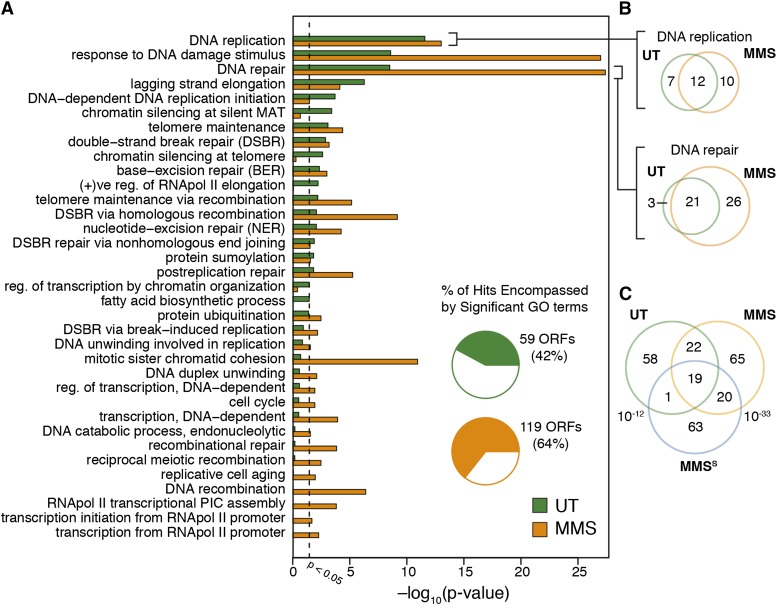
Finally, we examined the biological processes enriched among mutants with increased Rnr3 abundance in untreated conditions (Figure 2A). These terms overwhelmingly reflected processes that impinge upon genome stability. “DNA replication” (GO:0006260) was the most significantly enriched term, consistent with this biological process posing the greatest risk to genome stability in an unperturbed cell. Functionally similar GO terms “DNA repair” (GO:0006281) and “response to DNA damage stimulus” (GO:00060974) also were highly enriched. In aggregate, the 20 significantly enriched GO biological process terms included 59 unique mutants, covering 42% of all genes with increased Rnr3 abundance.
Figure 2.
Significant biological processes present among mutants with increased Rnr3 abundance. (A) Breakdown of all biological process Gene Ontology (GO) terms significantly enriched among genes with increased Rnr3 in either the untreated (UT; green) or methylmethane sulfonate (MMS; yellow) screen. Terms are ordered from top by significance in the UT screen. Pie charts present the fraction of genes with increased Rnr3 annotated with at least one significant GO term. (B) Overlap of genes within the biological processes “DNA replication” (GO:0006260) and “DNA repair” (GO:0006281) between UT and MMS. (C) Overlap of nonessential genes with increased Rnr3 abundance with a set of nonessential mutants identified as MMS sensitive (MMSS). Both screens overlap significantly, but MMS has almost twice as many shared genes.
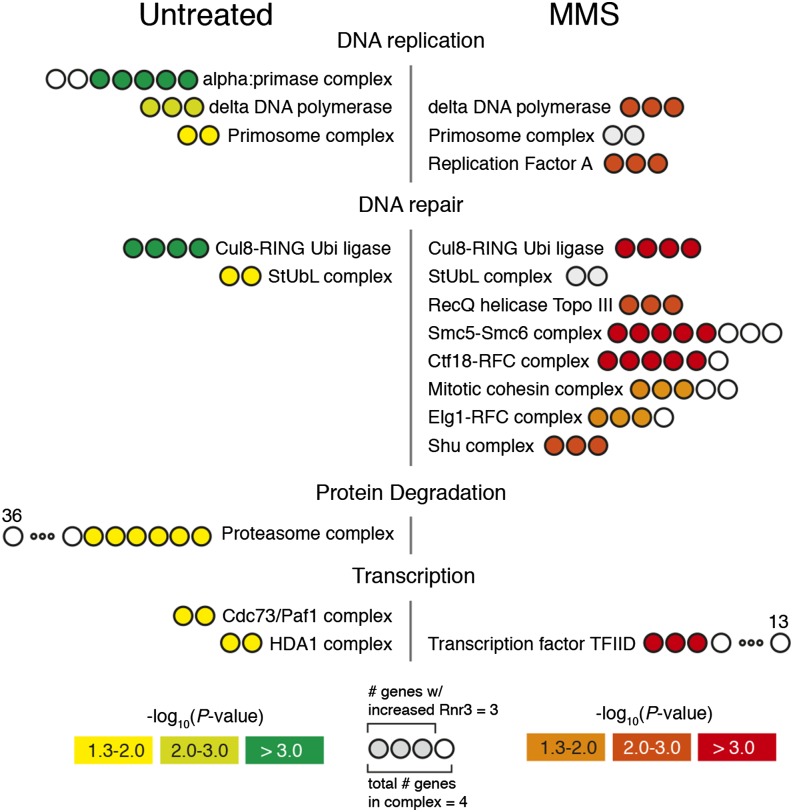
In addition to uncovering many biological processes associated with genome instability, we also identified several protein complexes associated with the maintenance of genome stability (Figure 3). Interestingly, two of these complexes, the Cul8-RING Ubiquitin ligase complex and the StUbL (i.e., SUMO-targeted Ubiquitin ligase) complex are related to ubiquitin transactions. All three members of lagging strand polymerase, DNA polymerase δ (POL3, POL31, POL32), were recovered, as well as both core members of DNA primase (PRI1, PRI2).
Figure 3.
Significant protein complexes present among mutants with increased Rnr3 abundance. Protein complexes with significant enrichment in either the untreated or MMS screen are shown. Number of circles corresponds to the number of genes within the complex, filled circles indicates the number of those genes with increased Rnr3 abundance. Color corresponds to the significance of complex enrichment. Two complexes from the MMS screen with insignificant enrichment P-values are indicated in gray, as both their constituent genes were identified [StUbL (i.e., SUMO-targeted Ubiquitin ligase) complex and Primosome complex]. Complexes were derived from the GO cellular component term set (Table S7 and Table S8). MMS, methylmethane sulfonate; GO, Gene Ontology.
Mutants with increased Rnr3 abundance in MMS
We sought to expand our effort to understand genome stability to conditions of induced DNA damage. To this end, we repeated the Rnr3 R-SGA screen in the context of the carcinogen and DNA damaging agent MMS. MMS is a monofunctional DNA alkylating agent that primarily methylates DNA on N7-deoxyguanine and N3-deoxyadenine (Beranek 1990; Lawley 1989). The N3-methyladenine lesion in particular is thought to impede replication fork progression, which can lead to replication fork collapse (Beranek 1990). We reasoned that mutants with a greater increase in Rnr3 abundance in the presence of MMS would have functions specific to repair of MMS damage.
Results from the MMS Rnr3 R-SGA screen presented some commonalities with the UT Rnr3 R-SGA screen. For example, mutants with increased Rnr3 abundance in MMS were more likely to reside in the nucleus and have biochemical activities relevant to DNA maintenance (Table S8). Once again, “DNA replication” (GO:0006260), “DNA repair” (GO:0006821), and “response to DNA damage stimulus” (GO:0006974) were the three most significantly enriched GO biological process terms (Figure 2A). However, nearly double the number of genes associated with and the DDR were identified in the presence of MMS. In fact, the MMS screen identified nearly all the DNA repair annotated genes with increased Rnr3 abundance in UT conditions, plus an additional 26 DNA repair annotated genes (Figure 2B).
We wanted to assess the degree to which treatment with MMS resulted in the identification of mutants associated specifically with MMS repair. The N3-methyladenine lesions produced by MMS treatment are addressed through three mechanisms of repair: (1) base excision repair, (2) recombinational repair and (3) postreplication repair (Xiao et al. 1996). “Base-excision repair” (GO:0006284) and “postreplication repair” (GO:0006301) were significantly enriched terms in both UT and MMS screens; however, more BER and PRR genes were identified in the presence of MMS (6/15 BER genes in MMS vs. 5/15 in UT; 7/11 PRR genes in MMS vs. 4/11 in UT; Figure 2A). Notably, the base-excision repair gene MAG1 (i.e., 3-MethylAdenine DNA Glycosylase), the deletion of which increases the expression of RNR3 in MMS (Jia and Xiao 2003), was identified specifically in the MMS Rnr3 R-SGA screen. The greater enrichment of postreplication repair was driven in part by identification of the entire E3 ubiquitin ligase complex containing RAD5, UBC13, and MMS2, which poly-ubquitylates proliferating cell nuclear antigen in an early step of error-free postreplication repair (Hoege et al. 2002). The final mechanism used in repair of MMS damage, “recombinational repair” (GO:0000725), was only enriched in the MMS screen (Figure 2A). Here, we identified all members of the Shu complex (CSM2, PSY3, SHU1), which mediates recombinational repair of stalled replication forks (Figure 3) (Ball et al. 2009). Other mutants found specifically in MMS were MPH1, which is thought to drive regression of stalled replication forks, and MMS4 and MUS81, which play an important role in processing of stalled replication forks for homologous recombination (Choi et al. 2010; Osman and Whitby 2007). Finally, we noted a prominent difference between the two screens with regards to the process “mitotic sister chromatid cohesion” (GO:0007064), which was highly enriched only in the presence of MMS (Figure 2A).
As further evidence of the ability of our Rnr3 R-SGA screen to capture mutants involved in MMS repair, we compared mutants from both UT and MMS screens with a set of non-essential deletion mutants previously identified as being MMS sensitive (Figure 2C) (Chang et al. 2002). Both the UT and MMS Rnr3 R-SGA screen significantly overlapped with the MMS sensitive dataset. However, the MMS Rnr3 R-SGA screen had twice as many mutants in common with the MMS sensitivity screen as did the UT Rnr3 R-SGA screen.
Comparison with previous genome instability screens
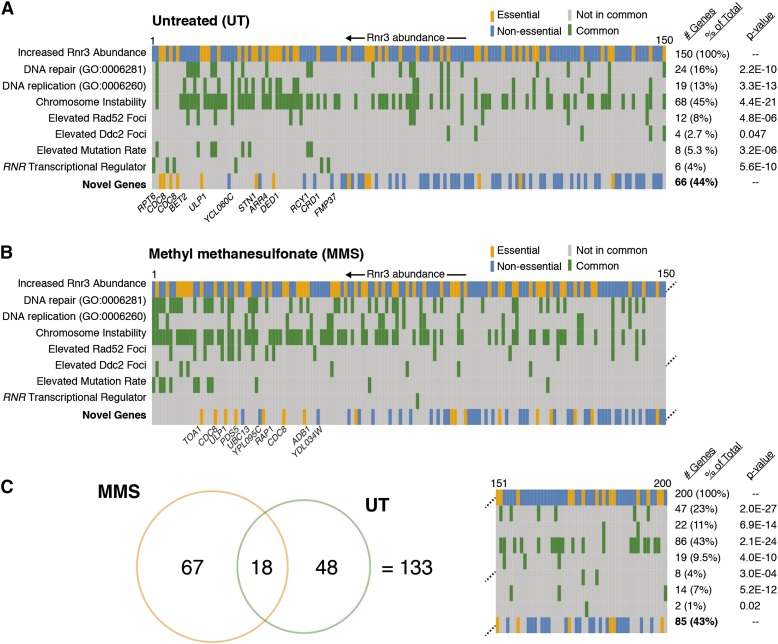
Several genome instability studies have been conducted in S. cerevisiae (Alvaro et al. 2007; Alver et al. 2013; Andersen et al. 2008; Askree et al. 2004; Cheng et al. 2012; Huang et al. 2003; Scholes et al. 2001; Smith et al. 2004; Stirling et al. 2011). Many of these studies have made use of the same collection of nonessential deletion mutants and more recently the use of hypomorphic or temperature-sensitive mutant collections similar to the one we used has been reported (Cheng et al. 2012; Stirling et al. 2011). We used this existing pool of data to conduct a thorough assessment of whether any of the 285 mutants with increased Rnr3 abundance could potentially represent novel genome maintenance genes. We focused on conducting a comparison to studies that directly assayed genome instability, as drug sensitivity and global protein localization studies can capture effects additional to genome maintenance (Tkach et al. 2012). To this end, we chose four major and orthogonal genome instability screens done in yeast to cross reference with our dataset: (1) 33 nonessential mutants possessing an elevated mutation rate (Huang et al. 2003), (2) 87 essential mutants with increased Ddc2 foci (Cheng et al. 2012), an indication of exposed ssDNA tracts, (3) 89 nonessential mutants with elevated Rad52 foci (Alvaro et al. 2007), an indication of double strand DNA breaks, and (4) 692 essential and nonessential mutants with chromosome instability (Stirling et al. 2011), roughly defined as increased loss of genetic material, as assessed by several different assays. In addition, we collected genes with the GO annotations “DNA repair” (GO:0006281) and “DNA replication” (GO:0006260), to capture mutants with a known association to these processes that haven’t been captured in high-throughput genome instability studies. We systematically annotated each mutant with increased Rnr3 abundance for its presence in any of the four genome instability datasets or two GO terms, and also annotated known transcriptional regulators of RNR (Figure 4).
Figure 4.
Systematic comparison with existing genome instability screens and genome maintenance-related GO terms. (A) Putative novel genome maintenance genes are identified by comparative gene subtraction. The 150 mutants with increased Rnr3 abundance in UT conditions are represented in each row, in order of decreasing Rnr3 abundance. In the top row, yellow rectangles represent essential and blue rectangles represent nonessential mutants. In the following seven rows, mutants are colored green if they are present in the data set listed at left, and gray otherwise. In the bottom row, all previously identified mutants are removed, leaving the putative novel mutants. Note that the majority of novel mutants cluster toward the right, indicating they have a lower Rnr3 abundance. The table on the right gives the number of genes with increased Rnr3 present in each data set, as well as the significance of the overlap between the datasets. (B) Same as in (A), but for the MMS screen. (C) Overlap between the putative novel genome maintenance genes identified in UT and MMS screens. A total of 133 novel mutants are identified. GO, Gene Ontology MMS, methylmethane sulfonate; UT, untreated.
From the pool of mutants with increased Rnr3 in untreated conditions, 84 mutants could be assigned to at least one of the above annotations leaving 66 mutants (44%) as potentially novel genome maintenance genes (Table S9). Similarly, 115 mutants with increased Rnr3 abundance in MMS had at least one genome instability annotation, leaving 85 (43%) mutants unannotated (Table S10). For both the UT and MMS screen, putative novel mutants had a tendency to be ranked lower than those associated with existing genome instability datasets. Overlap of the two sets of putative novel mutants revealed 18 shared mutants, resulting in an aggregate of 133 novel mutants identified (Figure 4C). Among these genes, GO enrichment produced no significantly enriched terms, suggesting that the putative novel mutants are not functionally cohesive and derive from a wide array of biological processes. Some of the putative novel mutants have inferable connections to genome maintenance, despite not being identified in previous genome instability screens. For example, we identified two alleles of CDC8 (cdc8-1 and cdc8-2), which is involved in dTTP synthesis (Jong et al. 1984; Sclafani and Fangman 1984), and therefore likely causes genome instability by affecting DNA replication (Hartwell 1973). Others are less easily linked to genome maintenance, such as DED1 (again supported by two alleles, ded1-95 and ded1-199), an ATP-dependent RNA helicase important for translation initiation (Chuang et al. 1997; Iost et al. 1999).
Evolutionary conservation of mutants with increased Rnr3 abundance
We used the ortholog cluster database generated by the inParanoid algorithm to evaluate the extent to which genes with increased Rnr3 abundance are conserved from yeast to human (Sonnhammer and Ostlund 2014). The core of the inParanoid algorithm is a reciprocal best-match BLAST search between proteomes of two species of interest, which is used to identify ortholog pairs. In total, the inParanoid algorithm assigns 2386 yeast genes a human ortholog, corresponding to 36% of the yeast genome. We identified 78 (52%) yeast genes with human orthologs among the set of genes with increased Rnr3 abundance in untreated conditions (Table S11). Similarly, 106 (53%) of the mutants with increased Rnr3 in MMS had a human ortholog (Table S12). Interestingly, considering only our putative novel set of mutants has little effect on the fraction of genes conserved: 33 (50%) novel UT and 44 (52%) novel MMS mutants have human orthologs (Table 1).
We compared the human orthologs of our genes causing increased Rnr3 abundance to two additional datasets. The first was a set of human genes identified as having increased H2AX phosphorylation following small interfering RNA knock-down (Paulsen et al. 2009). H2AX is phosphorylated at residue Ser139 in response to double-strand breaks by DNA damage checkpoint kinases, and serves as an indicator of genome instability in human cells (Paulsen et al. 2009). We found 11 and 17 of the mutants with increased Rnr3 abundance in UT and MMS had a human homolog with increased H2AX phosphorylation (Table 1, Table S11, and Table S12), suggesting their role in genome maintenance is conserved. Of these, eight belong to our set of putative novel mutants. In addition, we asked whether any of the human homologs of yeast mutants with increased Rnr3 were present in the Cancer Gene Census, which is a list of genes with mutations that have been causally linked to oncogenesis (Forbes et al. 2014; Futreal et al. 2004). In doing so, we identified a total of seven mutants present in the cancer census that have increased Rnr3 abundance in yeast, four of which were associated with our putative novel set.
Discussion
We have complemented a previous version of the Rnr3 assay (Zhou and Elledge 1992) by using yeast functional genomics reagents and methodologies. Instead of screening mutants generated via spontaneous mutagenesis, we us the nonessential gene deletion collection and conditional temperature-sensitive gene mutant collection. We introduce an Rnr3 reporter into both of these mutant collections using R-SGA. The resultant assay is an extremely high-throughput method to screen for genome instability: from start to finish it entails only an SGA procedure followed by ~1 hr of fluorescence scanning of whole colonies arrayed on agar plates (for the ~4800 mutants in the yeast nonessential deletion collection). Moreover, the method is compatible with drug treatment, insofar as the drug of interest is soluble in agar plates.
The Zhou and Elledge Rnr3 assay identified nine complementation groups (CRT1 to CRT9) exhibiting increased Rnr3 (Zhou and Elledge 1992). Of these, the associated gene for all but CRT2 is known. These genes are the transcriptional regulators CRT1/RFX1, SSN6, and TUP1; the RNR subunits RNR1, RNR2,and RNR4; and CDC21. Our R-SGA Rnr3 screen robustly identified CRT1/RFX1. Alleles of SSN6, TUP1, RNR1, RNR2, and RNR4 were not present in the mutant collections screened. An allele of CDC21 was screened in our R-SGA assay and did not have increased Rnr3 abundance, although another gene in the de novo pyrimidine synthesis pathway, CDC8, was identified.
Although here we focused on mutants causing increased Rnr3, we also identified 138 mutants with decreased Rnr3 (Table S4 and Table S6). Interestingly, in the MMS Rnr3 R-SGA screen, we noted that mutants with decreased Rnr3 abundance were significantly enriched for genes associated with the DDR pathway (data not shown). This result makes sense, as Rnr3 induction in the presence of damage depends on an intact DDR and suggests a further class of genes may be identified through screening Rnr3 abundance in the presence of damage.
A number of genome instability screens have been previously conducted in S. cerevisiae. This existing set of screens provides a valuable resource for comparative analysis. For example, after systematic comparison of our Rnr3 R-SGA dataset with four major, orthogonal genome instability studies, as well as GO terms associated with genome maintenance, we conclude that 45% of the 285 mutants we identified have no previous connection to maintenance of the genome. We attribute this substantial number of novel mutants to Rnr3 providing a different measure of genome instability relative to earlier screens. Existing genome instability screens can be roughly divided into two classes, based on the nature of their read-out: (1) those that directly assay alterations in the genetic material (such as mutation of the canavinine reporter, chromosome loss, loss of the MAT locus, telomere length, etc.) and (2) those that infer the presence of DNA damage through monitoring the formation of “damage foci” by fluorescence microscopy. Distinct from these approaches, the Rnr3 R-SGA identifies mutants that cause induction of the DDR.
We sought to assess the extent to which insights in yeast can be transferred to human and, in particular, to our understanding of genome instability in cancer. Approximately 52% of the mutants exhibiting increased Rnr3 abundance have human homologs, compared with 36% of all yeast genes. The enrichment for homologs is partly reflective of the fact that core genome maintenance processes, such as DNA replication, are highly conserved (75% of genes associated with the GO term “DNA replication” have human homologs). However, even in considering only the putative novel set of mutants, which lack known connections to replication or repair, we still note that 51% have human homologs. Furthermore, in about 14% of cases, we are able to provide evidence of conservation of the genome instability phenotype from yeast to human, simply by comparing to an existing genome instability screen. Although neither the UT nor the MMS Rnr3 R-SGA screen was significantly enriched for human genome instability genes, we note that, just as in comparisons between genome instability screens in yeast, differences in read-out between human and yeast screens can produce nonoverlapping results. Other factors likely frustrate such a direct approach to assessing phenotypic conservation, such as extent of inactivation of the gene (knock-down vs. deletion and hypomorph) and cell-type specific effects in human. Lastly, we recovered a handful of genes with human homologs that are part of the Cancer Gene Consensus, demonstrating that yeast genome instability studies can be used to provide disease relevant functional annotations to human cancer genes.
More than half of all genes causally implicated in cancer are oncogenes (Vogelstein et al. 2013). Indeed, gene duplication or hyperactivity is a frequent occurrence in cancer, yet the extent to which this causes genome instability is unknown. Gene overexpression collections now exist in yeast (Douglas et al. 2012; Hu et al. 2007; Sopko et al. 2006), and can be readily combined with the Rnr3 R-SGA method, providing insights into the relationship between oncogenes and genome instability.
Acknowledgments
We thank Bryan-Joseph San Luis for help with colony size analysis, Brenda Andrews for strains, and David Gallo, Attila Balint, and Raphael Loll-Krippleber for comments on the manuscript. This work was supported by grants from the Canadian Institutes of Health Research (MOP-79368 to G.W.B., and MOP-102629 to C.B.) and the National Institutes of Health (1R01 HG005853 to C.B.).
Footnotes
Supporting information is available online at http://www.g3journal.org/lookup/suppl/doi:10.1534/g3.115.016576/-/DC1
Communicating editor: A. Gasch
Literature Cited
- Agarwal R., Tang Z., Yu H., Cohen-Fix O., 2003. Two distinct pathways for inhibiting pds1 ubiquitination in response to DNA damage. J. Biol. Chem. 278: 45027–45033. [DOI] [PubMed] [Google Scholar]
- Aguilera A., Gomez-Gonzalez B., 2008. Genome instability: a mechanistic view of its causes and consequences. Nat. Rev. Genet. 9: 204–217. [DOI] [PubMed] [Google Scholar]
- Alvaro D., Lisby M., Rothstein R., 2007. Genome-wide analysis of Rad52 foci reveals diverse mechanisms impacting recombination. PLoS Genet. 3: e228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alver B., Jauert P. A., Brosnan L., O’Hehir M., Vandersluis B., et al. , 2013. A whole genome screen for minisatellite stability genes in stationary phase yeast cells. G3 (Bethesda) 3: 741–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen M. P., Nelson Z. W., Hetrick E. D., Gottschling D. E., 2008. A genetic screen for increased loss of heterozygosity in Saccharomyces cerevisiae. Genetics 179: 1179–1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Askree S. H., Yehuda T., Smolikov S., Gurevich R., Hawk J., et al. , 2004. A genome-wide screen for Saccharomyces cerevisiae deletion mutants that affect telomere length. Proc. Natl. Acad. Sci. USA 101: 8658–8663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ball L. G., Zhang K., Cobb J. A., Boone C., Xiao W., 2009. The yeast Shu complex couples error-free post-replication repair to homologous recombination. Mol. Microbiol. 73: 89–102. [DOI] [PubMed] [Google Scholar]
- Beranek D. T., 1990. Distribution of methyl and ethyl adducts following alkylation with monofunctional alkylating agents. Mutat. Res. 231: 11–30. [DOI] [PubMed] [Google Scholar]
- Carmen A. A., Rundlett S. E., Grunstein M., 1996. HDA1 and HDA3 are components of a yeast histone deacetylase (HDA) complex. J. Biol. Chem. 271: 15837–15844. [DOI] [PubMed] [Google Scholar]
- Chabes A., Georgieva B., Domkin V., Zhao X., Rothstein R., et al. , 2003. Survival of DNA damage in yeast directly depends on increased dNTP levels allowed by relaxed feedback inhibition of ribonucleotide reductase. Cell 112: 391–401. [DOI] [PubMed] [Google Scholar]
- Chang M., Bellaoui M., Boone C., Brown G. W., 2002. A genome-wide screen for methyl methanesulfonate-sensitive mutants reveals genes required for S phase progression in the presence of DNA damage. Proc. Natl. Acad. Sci. USA 99: 16934–16939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng E., Vaisica J. A., Ou J., Baryshnikova A., Lu Y., et al. , 2012. Genome rearrangements caused by depletion of essential DNA replication proteins in Saccharomyces cerevisiae. Genetics 192: 147–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi K., Szakal B., Chen Y. H., Branzei D., Zhao X., 2010. The Smc5/6 complex and Esc2 influence multiple replication-associated recombination processes in Saccharomyces cerevisiae. Mol. Biol. Cell 21: 2306–2314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang R.-Y., Weaver P. L., Liu Z., Chang T.-H., 1997. Requirement of the DEAD-Box protein ded1p for messenger RNA translation. Science 275: 1468–1471. [DOI] [PubMed] [Google Scholar]
- Davidson M. B., Katou Y., Keszthelyi A., Sing T. L., Xia T., et al. , 2012. Endogenous DNA replication stress results in expansion of dNTP pools and a mutator phenotype. EMBO J. 31: 895–907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douglas A.C., Smith A.M., Sharifpoor S., Yan Z., Durbic T., et al. , 2012. Functional analysis with a barcoder yeast gene overexpression system. G3 (Bethesda) 2: 1279–1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elledge S. J., Zhou Z., Allen J. B., Navas T. A., 1993. DNA damage and cell cycle regulation of ribonucleotide reductase. BioEssays 15: 333–339. [DOI] [PubMed] [Google Scholar]
- Falck J., Coates J., Jackson S. P., 2005. Conserved modes of recruitment of ATM, ATR and DNA-PKcs to sites of DNA damage. Nature 434: 605–611. [DOI] [PubMed] [Google Scholar]
- Forbes S. A., Beare D., Gunasekaran P., Leung K., Bindal N., et al. , 2015. COSMIC: exploring the world’s knowledge of somatic mutations in human cancer. Nucleic Acids Res. 43(Database Issue): D805–D811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Futreal P. A., Coin L., Marshall M., Down T., Hubbard T., et al. , 2004. A census of human cancer genes. Nat. Rev. Cancer 4: 177–183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giaever G., Chu A. M., Ni L., Connelly C., Riles L., et al. , 2002. Functional profiling of the Saccharomyces cerevisiae genome. Nature 418: 387–391. [DOI] [PubMed] [Google Scholar]
- Glickman M. S., Sawyers C. L., 2012. Converting cancer therapies into cures: lessons from infectious diseases. Cell 148: 1089–1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldmark J. P., Fazzio T. G., Estep P. W., Church G. M., Tsukiyama T., 2000. The Isw2 chromatin remodeling complex represses early meiotic genes upon recruitment by Ume6p. Cell 103: 423–433. [DOI] [PubMed] [Google Scholar]
- Harrison J. C., Haber J. E., 2006. Surviving the breakup: the DNA damage checkpoint. Annu. Rev. Genet. 40: 209–235. [DOI] [PubMed] [Google Scholar]
- Hartwell L. H., 1973. Three additional genes required for deoxyribonucleic acid synthesis in Saccharomyces cerevisiae. J. Bacteriol. 115: 966–974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoege C., Pfander B., Moldovan G. L., Pyrowolakis G., Jentsch S., 2002. RAD6-dependent DNA repair is linked to modification of PCNA by ubiquitin and SUMO. Nature 419: 135–141. [DOI] [PubMed] [Google Scholar]
- Hu Y., Rolfs A., Bhullar B., Murthy T. V., Zhu C., et al. , 2007. Approaching a complete repository of sequence-verified protein-encoding clones for Saccharomyces cerevisiae. Genome Res. 17: 536–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang M., Zhou Z., Elledge S. J., 1998. The DNA replication and damage checkpoint pathways induce transcription by inhibition of the Crt1 repressor. Cell 94: 595–605. [DOI] [PubMed] [Google Scholar]
- Huang M. E., Rio A. G., Nicolas A., Kolodner R. D., 2003. A genomewide screen in Saccharomyces cerevisiae for genes that suppress the accumulation of mutations. Proc. Natl. Acad. Sci. USA 100: 11529–11534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iost I., Dreyfus M., Linder P., 1999. Ded1p, a DEAD-box protein required for translation initiation in Saccharomyces cerevisiae, is an RNA helicase. J. Biol. Chem. 274: 17677–17683. [DOI] [PubMed] [Google Scholar]
- Jia X., Xiao W., 2003. Compromised DNA repair enhances sensitivity of the yeast RNR3-lacZ genotoxicity testing system. Toxicol. Sci. 75: 82–88. [DOI] [PubMed] [Google Scholar]
- Jong A. Y., Kuo C. L., Campbell J. L., 1984. The CDC8 gene of yeast encodes thymidylate kinase. J. Biol. Chem. 259: 11052–11059. [PubMed] [Google Scholar]
- Kainth P., Sassi H. E., Pena-Castillo L., Chua G., Hughes T. R., et al. , 2009. Comprehensive genetic analysis of transcription factor pathways using a dual reporter gene system in budding yeast. Methods 48: 258–264. [DOI] [PubMed] [Google Scholar]
- Klinkenberg L. G., Webb T., Zitomer R. S., 2006. Synergy among differentially regulated repressors of the ribonucleotide diphosphate reductase genes of Saccharomyces cerevisiae. Eukaryot. Cell 5: 1007–1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawley P. D., 1989. Mutagens as carcinogens: development of current concepts. Mutat. Res. 213: 3–25. [DOI] [PubMed] [Google Scholar]
- Li B., Reese J. C., 2001. Ssn6-Tup1 regulates RNR3 by positioning nucleosomes and affecting the chromatin structure at the upstream repression sequence. J. Biol. Chem. 276: 33788–33797. [DOI] [PubMed] [Google Scholar]
- Lopez-Mosqueda J., Maas N. L., Jonsson Z. O., Defazio-Eli L. G., Wohlschlegel J., et al. , 2010. Damage-induced phosphorylation of Sld3 is important to block late origin firing. Nature 467: 479–483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majka J., Niedziela-Majka A., Burgers P. M., 2006. The checkpoint clamp activates Mec1 kinase during initiation of the DNA damage checkpoint. Mol. Cell 24: 891–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naiki T., Wakayama T., Nakada D., Matsumoto K., Sugimoto K., 2004. Association of Rad9 with double-strand breaks through a Mec1-dependent mechanism. Mol. Cell. Biol. 24: 3277–3285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakada D., Matsumoto K., Sugimoto K., 2003. ATM-related Tel1 associates with double-strand breaks through an Xrs2-dependent mechanism. Genes Dev. 17: 1957–1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navadgi-Patil V. M., Burgers P. M., 2009. The unstructured C-terminal tail of the 9–1−1 clamp subunit Ddc1 activates Mec1/ATR via two distinct mechanisms. Mol. Cell 36: 743–753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negrini S., Gorgoulis V. G., Halazonetis T. D., 2010. Genomic instability—an evolving hallmark of cancer. Nat. Rev. Mol. Cell Biol. 11: 220–228. [DOI] [PubMed] [Google Scholar]
- Osman F., Whitby M. C., 2007. Exploring the roles of Mus81-Eme1/Mms4 at perturbed replication forks. DNA Repair (Amst.) 6: 1004–1017. [DOI] [PubMed] [Google Scholar]
- Paulsen R. D., Soni D. V., Wollman R., Hahn A. T., Yee M. C., et al. , 2009. A genome-wide siRNA screen reveals diverse cellular processes and pathways that mediate genome stability. Mol. Cell 3: 228–239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Putnam C. D., Jaehnig E. J., Kolodner R. D., 2009. Perspectives on the DNA damage and replication checkpoint responses in Saccharomyces cerevisiae. DNA Repair (Amst.) 8: 974–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rouse J., Jackson S. P., 2002. Lcd1p recruits Mec1p to DNA lesions in vitro and in vivo. Mol. Cell 9: 857–869. [DOI] [PubMed] [Google Scholar]
- Sanchez Y., Bachant J., Wang H., Hu F., Liu D., et al. , 1999. Control of the DNA damage checkpoint by chk1 and rad53 protein kinases through distinct mechanisms. Science 286: 1166–1171. [DOI] [PubMed] [Google Scholar]
- Sanvisens N., de Llanos R., Puig S., 2013. Function and regulation of yeast ribonucleotide reductase: cell cycle, genotoxic stress, and iron bioavailability. Biom. J. 36: 51–58. [DOI] [PubMed] [Google Scholar]
- Scholes D. T., Banerjee M., Bowen B., Curcio M. J., 2001. Multiple regulators of Ty1 transposition in Saccharomyces cerevisiae have conserved roles in genome maintenance. Genetics 159: 1449–1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz M. F., Duong J. K., Sun Z., Morrow J. S., Pradhan D., et al. , 2002. Rad9 Phosphorylation sites couple Rad53 to the Saccharomyces cerevisiae DNA damage checkpoint. Mol. Cell 9: 1055–1065. [DOI] [PubMed] [Google Scholar]
- Sclafani R. A., Fangman W. L., 1984. Yeast gene CDC8 encodes thymidylate kinase and is complemented by herpes thymidine kinase gene TK. Proc. Natl. Acad. Sci. USA 81: 5821–5825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma V. M., Tomar R. S., Dempsey A. E., Reese J. C., 2007. Histone deacetylases RPD3 and HOS2 regulate the transcriptional activation of DNA damage-inducible genes. Mol. Cell. Biol. 27: 3199–3210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S., Hwang J. Y., Banerjee S., Majeed A., Gupta A., et al. , 2004. Mutator genes for suppression of gross chromosomal rearrangements identified by a genome-wide screening in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 101: 9039–9044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonnhammer E. L., Ostlund G., 2014. InParanoid 8: orthology analysis between 273 proteomes, mostly eukaryotic. Nucleic Acids Res. 43(Database Issue): D234–D239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sopko R., Huang D., Preston N., Chua G., Papp B., et al. , 2006. Mapping pathways and phenotypes by systematic gene overexpression. Mol. Cell 21: 319–330. [DOI] [PubMed] [Google Scholar]
- Stirling P. C., Bloom M. S., Solanki-Patil T., Smith S., Sipahimalani P., et al. , 2011. The complete spectrum of yeast chromosome instability genes identifies candidate CIN cancer genes and functional roles for ASTRA complex components. PLoS Genet. 7: e1002057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Storchova Z., Pellman D., 2004. From polyploidy to aneuploidy, genome instability and cancer. Nat. Rev. Mol. Cell Biol. 5: 45–54. [DOI] [PubMed] [Google Scholar]
- Sun Z., Hsiao J., Fay D. S., Stern D. F., 1998. Rad53 FHA domain associated with phosphorylated Rad9 in the DNA damage checkpoint. Science 281: 272–274. [DOI] [PubMed] [Google Scholar]
- Sweeney F. D., Yang F., Chi A., Shabanowitz J., Hunt D. F., et al. , 2005. Saccharomyces cerevisiae Rad9 acts as a Mec1 adaptor to allow Rad53 activation. Curr. Biol. 15: 1364–1375. [DOI] [PubMed] [Google Scholar]
- Tang H. M., Siu K. L., Wong C. M., Jin D. Y., 2009. Loss of yeast peroxiredoxin Tsa1p induces genome instability through activation of the DNA damage checkpoint and elevation of dNTP levels. PLoS Genet. 5: e1000697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tkach J. M., Yimit A., Lee A. Y., Riffle M., Costanzo M., et al. , 2012. Dissecting DNA damage response pathways by analysing protein localization and abundance changes during DNA replication stress. Nat. Cell Biol. 14: 966–976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong A. H., Boone C., 2006. Synthetic genetic array analysis in Saccharomyces cerevisiae. Methods Mol. Biol. 313: 171–192. [DOI] [PubMed] [Google Scholar]
- Tsaponina O., Barsoum E., Astrom S. U., Chabes A., 2011. Ixr1 is required for the expression of the ribonucleotide reductase Rnr1 and maintenance of dNTP pools. PLoS Genet. 7: e1002061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogelstein B., Papadopoulos N., Velculescu V. E., Zhou S., Diaz L. A., Jr, et al. , 2013. Cancer genome landscapes. Science 339: 1546–1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao W., Chow B. L., Rathgeber L., 1996. The repair of DNA methylation damage in Saccharomyces cerevisiae. Curr. Genet. 30: 461–468. [DOI] [PubMed] [Google Scholar]
- Yu L., Pena Castillo L., Mnaimneh S., Hughes T. R., Brown G. W., 2006. A survey of essential gene function in the yeast cell division cycle. Mol. Biol. Cell 17: 4736–4747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zegerman P., Diffley J. F. X., 2010. Checkpoint-dependent inhibition of DNA replication initiation by Sld3 and Dbf4 phosphorylation. Nature 467: 474–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z., Reese J. C., 2004. Ssn6-Tup1 requires the ISW2 complex to position nucleosomes in Saccharomyces cerevisiae. EMBO J. 23: 2246–2257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z., Elledge S. J., 1992. Isolation of crt mutants constitutive for transcription of the DNA damage inducible gene RNR3 in Saccharomyces cerevisiae. Genetics 131: 851–866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou L., Elledge S. J., 2003. Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 300: 1542–1548. [DOI] [PubMed] [Google Scholar]