Abstract
Aims
Cardiovascular alterations contribute to a high mortality rate in patients with end-stage renal disease (ESRD). The aims of the present study are to evaluate left ventricular (LV) function and common carotid artery (CCA) parameters and to determine risk factors associated with these changes in patients undergoing peritoneal dialysis (PD).
Methods
This longitudinal prospective study was conducted in 50 ESRD patients in whom PD had been initiated and who were observed for 18 months after the commencement of dialysis treatment, with echocardiography and CCA ultrasound parameter evaluation.
Results
LV hypertrophy was observed in 78% of patients at baseline and in 60% after 18 months of PD treatment. LV systolic and diastolic function was found to be significantly better after 18 months of PD treatment. Examining predictors of LV systolic function, it was found that total cholesterol was an independent positive predictor and endothelin-1 (ET-1) an independent negative predictor of LV systolic function after 18 months of treatment with PD (p < 0.001). Independent negative predictors of diastolic LV function were hemoglobin and type 2 diabetes mellitus, and daily collection of urine was an independent positive predictor (p < 0.001). Female gender was an independent negative predictor of CCA intima-media thickness, whereas body mass index, ET-1 and C-reactive protein were independent positive predictors (p < 0.001).
Conclusions
The results suggest several novel modifiable mechanisms related to the short-term effects of dialysis that are potentially implicated in the development of uremic cardiomyopathy.
Key Words: End-stage renal disease, Peritoneal dialysis, Left ventricle, Atherosclerosis
Introduction
The mortality rate in patients with end-stage renal disease (ESRD) is much higher than in the general population despite advances in dialysis treatment. Cardiovascular structure and functional abnormalities, such as left ventricular hypertrophy (LVH), left ventricular (LV) systolic and diastolic dysfunction, accelerated atherosclerosis, arrhythmias and coronary artery calcification, contribute to a high cardiovascular mortality in patients with ESRD [1].
Patients with ESRD are a unique population harboring cardiovascular disease (CVD), while the analysis of risk factors in these patients may be obscured by comorbid conditions such as diabetes and preexisting atherosclerosis [2]. Until recently, the prevalence, severity and predictors of these cardiovascular abnormalities have not been investigated extensively in patients with ESRD from the start of dialysis treatment.
The aims of the present study were (1) to evaluate LV function and common carotid artery (CCA) parameters and (2) to determine risk factors associated with these cardiovascular changes in patients undergoing peritoneal dialysis (PD).
Methods
Study Population and Design
This longitudinal prospective study was conducted with 50 ESRD patients (type 2 diabetic and nondiabetic) in whom continuous ambulatory PD had been initiated and who were observed for 18 months after the commencement of dialysis treatment. All examined patients underwent 4-5 dialysis changes with 2 liters of dialysis solution.
Patients with a verified diagnosis of CCA stenosis (>70% internal CCA stenosis), cerebral vascular diseases (patients who had had a transient ischemic attack or stroke in the past 6 months) and patients with chronic rheumatic heart disease, congenital heart disease, underlying active malignancy or systemic lupus erythematosus, as well as patients with signs of peritonitis during the study period, were excluded from the study. In all patients, antihypertensive therapy as well as any therapy that can influence the values of monitored laboratory parameters (nitrites, sildenafil, captopril, NSAIDs, heparin and β2-agonists) were excluded 24 h before taking blood samples for determination of endothelin-1 (ET-1) and nitric oxide concentration. The patients did not receive diuretic therapy prior to diuresis level measurement and sampling. The substitution therapy with recombinant human erythropoietin (epoetin β 6,000 IU weekly) was administered to all patients.
The institutional ethics committee approved the study protocol, and all participants gave written informed consent prior to the study. Our examinations of the patients conformed to good medical and laboratory practices and the recommendations of the Declaration of Helsinki on Biomedical Research Involving Human Subjects. Ultrasound measurements were performed after drainage of peritoneal fluid in PD patients.
Echocardiographic Data
Comprehensive echocardiographic measurements were performed using an ultrasound machine (Toshiba 270SSA) with a 3.75-MHz sector probe by a single experienced cardiologist blinded to clinical information on patients at baseline and at the end of the study period. All images were obtained with standard techniques using M-mode, two-dimensional and Doppler measurements in accordance with the American Society of Echocardiography guidelines [3]. The LV mass was calculated using the modified formula proposed by Devereux et al. [4]. Echocardiographic evidence of LVH was defined as LV mass index divided by a body surface area >115 g/m2 in men and >95 g/m2 in women. LV systolic function was assessed by calculation of the ejection fraction (EF) using a modified Simpson's method [5], while fractional shortening (FS) was calculated according to the formula described by Lang et al. [6]. LV systolic weakness was defined as EF <50% and FS <30%. Pulsed Doppler echocardiography was used to evaluate transmitral LV filling velocities at the tips of the mitral valve. The peak early-diastolic flow velocity (E) and the peak late-diastolic velocity (A) shown as the E/A ratio were measured by analyzing the transmitral flow. LV diastolic dysfunction was defined as E/A ≤1. With these parameters, diastolic dysfunction was further categorized into four groups: normal, abnormal relaxation, pseudonormal and restrictive pattern [7].
CCA Ultrasound Data
Intima-media thickness (IMT) of the CCA and the presence of atherosclerotic plaques were measured by means of a high-resolution transducer probe of 7.5 MHz frequency in B mode (Wall Track System, Pie Medical, Maastricht, The Netherlands) by a single experienced angiologist blinded to all clinical details of patients during the follow-up period. The CCA, carotid bulbus and the first 2 cm of the internal and external CCA were scanned bilaterally. The measurements of IMT were done at a distance of 20 mm from bifurcation into the plaque-free area on the CCA. Three measurements were done on the left and right CCA, while the mean values of these measurements were utilized in the analysis. The plaque score in the CCA was calculated by summing the three IMT measurement values, bilaterally [8].
Parameters of PD
Adequacy of dialysis was calculated from weekly total removed urea mass by daily volume of dialysate and urine and divided by urea distribution volume. The distribution volume of urea was calculated using the Watson equation [9]. Residual renal function (RRF) was estimated as the mean value of renal creatinine clearance (ml/min). A simplified peritoneal equilibration test was performed using 4.25% glucose-based solution to obtain the dialysate to plasma creatinine concentration ratio at 4 h of dwell (D/P Cr) [10].
Statistical Analysis
Statistical analysis was performed using SPSS version 16.0 (SPSS, Inc.). Each value was expressed as the mean ± SD or as median and interquartile range where appropriate. The distribution of variables was tested by the Shapiro-Wilk test. Significant changes in the variables from baseline to 18 months after PD were tested by paired t test or by the Wilcoxon signed-rank test. The difference between two groups was analyzed by the Mann-Whitney test. Univariate correlation coefficients were determined by Pearson or Spearman analysis. A multiple regression analysis was applied to examine the relationship between ultrasound cardiovascular parameters and a set of clinical-biochemical parameters. The significant independent variables were ordered according to their standardized effect, defined as regression coefficient/standard error of the regression (β). A p value <0.05 was considered statistically significant.
Results
The main clinical and laboratory data are summarized in table 1. In our study, 52% of patients were diabetics. Significant reductions of blood pressure (BP) were observed after 18 months on PD. The median concentration of serum ET-1 decreased, while nitric oxide in- creased significantly after 18 months on PD. At the end of the follow-up period, dialysis adequacy was estimated by Kt/Vurea as satisfactory.
Table 1.
Baseline clinical characteristics
| Clinical characteristics | At baseline | After 18 months of PD | p value |
|---|---|---|---|
| Age, years | 60.5 (26–76) | ||
| Men | 25 (50) | ||
| Smoker | 18 (45) | ||
| Body mass index | 25.9 ± 3.7 | 25.7 ±2.2 | n.s. |
| Diabetes mellitus | 26 (52) | ||
| Mean arterial pressure, mm Hg | 109.2 ± 15.1 | 95.4 ±8.2 | <0.001 |
| Cause of ESRD | |||
| Diabetic nephropathy | 24 (48) | ||
| Hypertensive nephrosclerosis | 9 (18) | ||
| Glomerulonephritis | 7 (14) | ||
| Pyelonephritis | 7 (14) | ||
| Other | 3 (6) | ||
| Antihypertensives, n | |||
| On ARB or ACE inhibitor | 32 | 24 | |
| On diuretic | 36 | 18 | |
| On calcium channel blocker | 21 | 29 | |
| Other | 9 | 3 | |
| Laboratory measurements | |||
| Hemoglobin, g/l | 101.9 ± 10.3 | 118.6 ±11.1 | <0.001 |
| Albumin, g/l | 30.9 ± 2.6 | 31.5 ±2.0 | <0.01 |
| Urea nitrogen, mmol/l | 25.7 ± 6.7 | 17.5 ±2.5 | <0.001 |
| Creatinine, μmol/l | 912.3 ± 223.3 | 733.9 ±131.0 | <0.001 |
| Cholesterol, mmol/l | 6.5 ± 1.6 | 5.5 ±1.3 | <0.001 |
| Triglyceride, mmol/l | 2.4 ± 1.3 | 2.0 ±2.5 | <0.01 |
| Low-density lipoprotein, mmol/l | 4.7 ± 1.4 | 3.6 ±0.8 | <0.05 |
| C-reactive protein, mg/l | 11.1 (6.1–16.4) | 4.5 (2.8–7.7) | <0.001 |
| Fibrinogen, g/l | 6.2 ± 1.9 | 4.4 ±1.3 | <0.001 |
| Calcium, mmol/l | 2.2 ± 0.2 | 2.3 ±0.1 | n.s. |
| Phosphorous, mmol/l | 1.8 ± 0.3 | 1.6 ±0.2 | <0.05 |
| CaxP | 3.9 ± 0.6 | 3.6 ±0.5 | <0.001 |
| ß2-Microglobulin | 7.0 (2.9–11.2) | 4.2 (2.3–9.7) | <0.05 |
| Parathyroid hormone, pmol/l | 225.5 (97.8–387) | 200.0 (100.0–410) | n.s. |
| Plasma homocysteine, µmol/l | 25.2 (20.2–30.1) | 18.0 (14.0–20.9) | <0.001 |
| B-type natriuretic peptide, pg/ml | 183.9 (89.8–432.5) | 69.6 (50.2–98.8) | <0.001 |
| Troponine, ng/ml | 0.022 (0.001–0.12) | 0.001 (0.00–0.01) | <0.01 |
| Nitric oxide, μmol/l | 40.72 (19.4–56.7) | 48.0 (32.8–60.4) | <0.01 |
| ET-1, pg/ml | 6.32 (3.2–8.8) | 4.0 (2.27–6.3) | <0.001 |
| RRF, ml/min/1.73 m2 | 5.5 ± 3.8 | 7.0 ±5.0 | <0.05 |
| Urine volume, ml/day | 545.6 ± 378.5 | 584.8 ±489.7 | n.s. |
| nPNA, g/kg/day | 0.98 ± 0.13 | 1.11 ±0.1 | <0.05 |
| Weekly Kt/Vurea | 1.9 ± 0.8* | 2.1 ±0.6 | <0.05 |
Data are expressed as the mean ± SD, number (percentage) or median (range). n.s. = Not significant; ARB = angiotensin receptor blocker; ACE = angiotensin-converting enzyme; nPNA = protein equivalent of total nitrogen appearance.
After 6 months of PD.
LVH was observed in 78% of patients at baseline and in 60% after 18 months of PD treatment. A significant reduction in LV mass index, CCA diameter, IMT and plaque score was observed. LV function was determined as significantly better after 18 months of PD (table 2).
Table 2.
Clinical, echocardiographic and ultrasound measurements of CCA parameters
| Ultrasound parameters | At baseline | End of follow-up | p value |
|---|---|---|---|
| Echocardiographic parameter M-mode and 2D | |||
| LV end-diastolic diameter, mm | 52.9 ± 3.6 | 49.6 ± 6.5 | <0.05 |
| LV end-systolic diameter, mm | 35.0 ± 3.5 | 34.3 ± 3.9 | n.s. |
| LV mass, g | 280.7 ± 43.2 | 243.8 ± 40.8 | <0.05 |
| LV mass index, g/m2 | 162.2 ± 38.1 | 140.2 ± 37.8 | <0.001 |
| Left atrium diameter, mm | 43.3 ± 5.7 | 40.5 ± 6.7 | <0.05 |
| LV structure | |||
| Normal | 11 (22.0) | 19 (38) | <0.05 |
| LVH | 39 (78.0) | 30 (60) | <0.05 |
| Concentric hypertrophy | 22 (44) | 19 (38) | 0.056 |
| Eccentric hypertrophy | 15 (30) | 11 (22) | <0.05 |
| Concentric remodeling | 2 (4) | 1 (2) | n.s. |
| LV function | |||
| Normal | 10 (20.0) | 32 (64.0) | <0.01 |
| Systolic dysfunction | 0 | 5 (10.0) | <0.001 |
| LV EF, % | 50.1 ± 9.4 | 56.9 ± 10.0 | <0.05 |
| FS | 28.8 ± 5.1 | 30.8 ± 4.1 | <0.001 |
| Diastolic dysfunction | 20 (40.0) | 3 (6.0) | <0.01 |
| E/A ratio | 0.9 ± 0.1 | 1.1 ± 0.1 | <0.05 |
| Mixed systolic-diastolic dysfunction | 20 (40.0) | 10 (20.0) | <0.01 |
| Grading of diastolic dysfunction | |||
| Grade 1: impaired relaxation pattern | 26 (65) | 7 (53.85) | <0.01 |
| Grade 2: pseudonormal pattern | 13 (32.5) | 3 (23.08) | <0.01 |
| Grade 3: restrictive filling pattern | 1 (2.5) | 1 (7.69) | n.s. |
| Ultrasound measurements of carotid arteries | |||
| CCA IMT, mm | 0.76 (0.6–0.9) | 0.68 (0.5–0.8) | <0.05 |
| CCA diameter, mm | 5.8 (5.2–6.4) | 5.0 (4.9–5.4) | <0.05 |
| Plaque score | 4.15 (4.2–5.4) | 3.95 (2.9–5.1) | <0.001 |
Data are expressed as the mean ± SD, number (percentage) or median (range). n.s. = Not significant.
In our study, the EF and E/A ratios were significantly associated with the presented traditional and nontraditional risk factors as well as with the CCA diameter. CCA IMT did not show a significant correlation with protein equivalent of total nitrogen appearance, parathyroid hormone, serum calcium and daily collection of urine (table 3).
Table 3.
The factors correlated with LV function and CCA parameters after 18 months on PD
| Factors | EF | E/A ratio | CCA IMT | CCA diameter |
|---|---|---|---|---|
| Mean arterial pressure | −0.62** | −0.46** | 0.29* | 0.45* |
| Hemoglobin | 0.64** | 0.57** | −0.49** | −0.71** |
| Cholesterol | −0.65** | −0.62** | 0.42** | 0.74** |
| Low-density lipoprotein | −0.73** | −0.59** | 0.35* | 0.58** |
| C-reactive protein | −0.68** | −0.61** | 0.36* | 0.51** |
| Calcium | 0.08 | −0.02 | 0.01 | 0.02 |
| Phosphorous | −0.54** | −0.49** | 0.33* | 0.57** |
| CaxP | −0.49** | −0.46** | 0.31* | 0.54** |
| Parathyroid hormone | −0.5* | −0.39** | 0.2 | 0.48* |
| nPNA | 0.58** | 0.57** | −0.07 | −0.34* |
| Albumin | 0.68 | 0.62* | −0.34* | −0.58* |
| RRF | 0.43** | 0.55** | −0.18 | −0.37** |
| Urine volume, ml/day | 0.49** | 0.61** | −0.23 | −0.51** |
| Kt/Vurea | −0.65** | −0.61** | 0.38* | 0.59** |
| Nitric oxide | 0.745** | 0.766** | −0.824** | −0.648** |
| ET-1 | −0.708** | −0.695** | 0.807** | 0.698** |
| Homocysteine | −0.575** | −0.495** | 0.605* | 0.663* |
| Troponine | −0.697* | −0.76** | 0.788* | 0.629* |
| BNP | −0.578** | −0.409** | 0.485* | 0.317* |
nPNA = Protein equivalent of total nitrogen appearance; BNP = B-type natriuretic peptide.
p < 0.05;
p < 0.001.
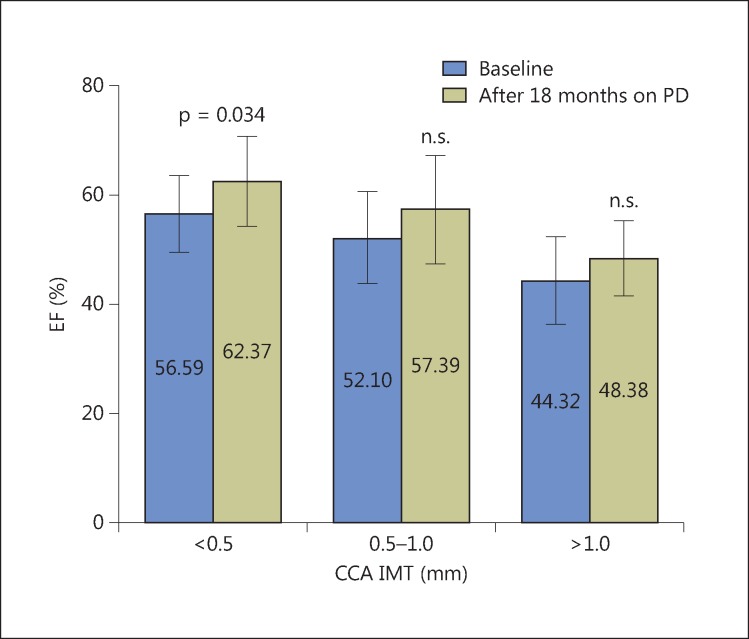
Average EF values were significantly higher after 18 months of PD in the group of patients without atherosclerosis compared to baseline values, whereas in the group of patients with mild/moderate and severe atherosclerosis, the CCA average value of EF was greater at the end of the observed period, but there was no statistically significant difference (fig. 1).
Fig. 1.
LV EF in patients during follow-up in relation to the level of CCA atherosclerosis. n.s. = Not significant.
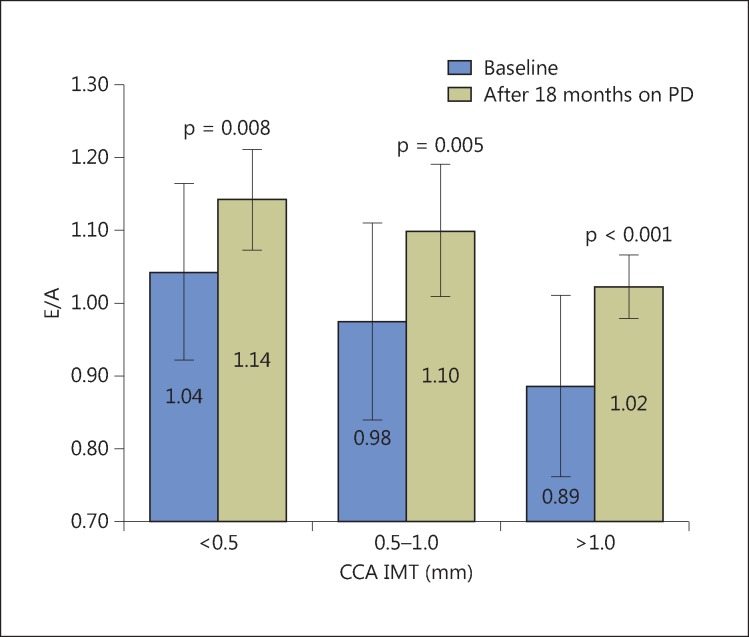
LV diastolic function, assessed by the average value of the E/A ratio, was statistically significantly better after 18 months of PD compared to baseline values within all groups of patients (fig. 2).
Fig. 2.
E/A ratio in patients during follow-up in relation to the degree of CCA atherosclerosis.
Examining predictors of LV EF using the model of logistic regression analysis, it was found that total cholesterol was an independent positive predictor and ET-1 an independent negative predictor of LV systolic function after 18 months on PD. Independent negative predictors of the E/A ratio were hemoglobin and diabetes mellitus, and daily collection of urine was an independent positive predictor. Examining predictors of CCA IMT after 18 months on PD, it was found that female gender was an independent negative predictor, whereas body mass index, ET-1 and C-reactive protein were independent positive predictors. Serum albumin and hemoglobin were independent negative predictors, while low-density lipoprotein and age were positive predictors of CCA diameter during dialysis treatment (table 4).
Table 4.
Significant independent predictors of LV function and carotid remodeling at the end of follow-up
| Predictors | 95% CI | p value |
|---|---|---|
| LV systolic function | ||
| Cholesterol | 2.647–45.433 | 0.001 |
| ET-1 | 0.002–0.778 | 0.007 |
| LV diastolic function | ||
| Urine volume (ml/day) | 0.401–1.004 | 0.001 |
| Diabetes mellitus type 2 | 0.346–0.997 | 0.007 |
| Hemoglobin | 0.485–0.893 | 0.010 |
| CCA IMT | ||
| Female gender | 0.002–0.898 | 0.042 |
| Body mass index | 1.035–2.304 | 0.033 |
| C-reactive protein | 1.124–2.200 | 0.008 |
| ET-1 | 1.874–3.007 | 0.024 |
| CCA diameter | ||
| Serum albumin | 0.134–0.809 | 0.015 |
| Hemoglobin | 0.741–0.991 | 0.037 |
| Low-density lipoprotein | 1.131–14.591 | 0.032 |
| Age | 1.018–1.383 | 0.029 |
Discussion
CVD accounts for half of the deaths in patients treated with renal replacement therapy, whilst mortality from cardiovascular causes is far higher than in the general population [11,12]. The influence of uremia and dialysis on cardiovascular structure and function was investigated as detailed by studies on patients with chronic renal failure [1,13,14].
There are two parallel processes involved in the development of CVD in ESRD patients. The first process involves cardiac changes including LVH and LV dysfunction as a response to mechanical or hemodynamic overload. The second process involves vascular changes, including atherosclerosis, arteriosclerosis and vascular calcification. Over the last decade, cardiac abnormalities such as LVH and LV dysfunction and vascular abnormalities, e.g. arterial stiffness, increased IMT and coronary calcification, have been accepted as early markers of cardiomyopathy and atherosclerosis [15].
LV diastolic dysfunction is frequently observed in dialysis patients and results from LVH, cardiac fibrosis and impaired cardiac relaxation [13]. Also, the presence of LVH and LV systolic dysfunction are well-recognized risk factors for cardiovascular mortality in this population [14,16].
Considering the large risk, our data may emphasize the importance of early recognition of LV dysfunction and may have significant therapeutic implications at the start of dialysis.
In this study, we determined traditional and nontraditional risk factors in patients on dialysis. Our results confirmed that ESRD patients have increased traditional and uremia-related risk factors. Important observations are that, before undergoing PD, ESRD patients have significant LVH, dominant atherosclerotic changes in the CCA and impaired LV function.
In patients with ESRD, assessment of cardiovascular structure and function is essential for planning appropriate management. Many reports have warned of the risks of LVH for future cardiac failure [17]. Many factors are associated with cardiac hypertrophy in patients on PD [18]. In our study, at the end of the follow-up, significantly fewer patients with LVH were recorded, suggesting a positive effect of PD on LV remodeling and a correction of observed cardiovascular risk factors.
It is known that LV systolic function usually remains normal despite LVH in patients with ESRD [19]. Many studies have shown that diastolic dysfunction may ultimately be a risk factor for LV systolic dysfunction and future congestive heart failure [20]. We also found that none of our patients had congestive heart failure and all had normal systolic function despite increased LV mass index, though with a significantly better LV systolic function after 18 months of PD treatment. Total cholesterol and ET-1 were identified as independent predictors of LV systolic function in PD patients.
Some studies suggested that abnormalities of diastolic function precede those of systolic function [21]. Doppler measurement of mitral inflow velocity is widely used to assess LV diastolic function. Using this method, our study suggested that LV relaxation (E/A ratio) was impaired in ESRD patients as compared with the values after 18 months of PD therapy. In the present study, the linear regression analysis revealed that hemoglobin levels, diuresis and diabetes mellitus type 2 were independent predictors of poor diastolic function (E/A ratio). Our results are similar to previous studies [22,23].
Unlike hemodialysis, which is associated with typically marked body water content fluctuations, PD is characterized by its almost steady state, which probably has a major impact on LV function. Also, the data of our study indicate that RRF is lower in ESRD than in PD. Wang [24] has found that RRF may play a role in limiting the increase in cardiovascular remodeling by improving the overall Kt/Vurea and removal of uremic toxins.
In our PD patients, there was a positive correlation between LV function parameters (EF, FS and E/A ratio), daily urinary output and RRF. This is a very important result, because it suggests the existence of some nondialyzable uremic toxins that may be important in the progression of pathological cardiovascular alteration in this population.
The second process playing a role in the development of CVD in ESRD patients is vascular injury. The measurement of CCA IMT using high-resolution ultrasonography has been suggested as an excellent marker of subclinical atherosclerosis. However, there are few studies about CCA IMT and atherosclerosis indicating that the CCA diameter represents an indirect indicator of the stiffness of the arterial wall [25]. In our study, female gender, body mass index, C-reactive protein and ET-1 were determined as independent predictors of an increased CCA IMT, whereas serum albumin, hemoglobin, low-density lipoprotein and age were identified as predictors of rigidity of the arterial wall. Also, our results suggest a high prevalence of atherosclerosis in the predialysis period, but also a significant positive effect of PD in the first year on the process of stopping accelerated atherogenesis.
Our results showed two phases of the cardiovascular effects of PD. Significant reductions in BP, LV mass and B-type natriuretic peptide were probably related to strict volume control, while in the later stage, better control of BP seemed to be related to changes in blood volume and possibly to sympathetic activity.
Our study had several shortcomings. First, a relatively small number of patients with diabetes mellitus were included in the trial which were not specifically separated from patients without diabetes mellitus; second, a longer period is needed for patient monitoring in order to be able to estimate a clear effect of PD, and third, there is a need to include patients after kidney transplantation.
The findings indicate that monitoring the traditional and nontraditional risk factors provides significant prognostic information for the estimation of alterations of LV structure and function and subclinical atherosclerosis, and that repeated measurements of such parameters are useful in the clinical practice for the management of PD patients.
Conclusion
In conclusion, LVH, increased CCA IMT and diameter, impaired diastolic function and normal systolic function are highly present in uremic patients before initiation of renal replacement therapy.
PD in the first 18 months of treatment has a positive effect on stopping or even on achieving partial regression of cardiovascular remodeling. The better control of BP and good volume control lead to significant improvements in cardiac function and arterial stiffness. The results suggest several novel modifiable mechanisms related to the short-term effects of dialysis that are potentially implicated in the development of uremic cardiomyopathy.
Disclosure Statement
The authors declare that they have no conflicts of interest.
References
- 1.Longenecker JC, Coresh J, Powe NR, Levey AS, Fink NE, Martin A, Klag MJ. Traditional cardiovascular disease risk factors in dialysis patients compared with the general population: the CHOICE study. J Am Soc Nephrol. 2002;13:1918–1927. doi: 10.1097/01.asn.0000019641.41496.1e. [DOI] [PubMed] [Google Scholar]
- 2.Middleton JP, Pun PH. Hypertension, chronic kidney disease, and the development of cardiovascular risk: a joint primacy. Kidney Int. 2010;77:753–755. doi: 10.1038/ki.2010.19. [DOI] [PubMed] [Google Scholar]
- 3.Oh JK, Appleton CP, Hatle LK, Nishimura RA, Seward JB, Tajik AJ. The noninvasive assessment of left ventricular diastolic function with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr. 1997;10:246–270. doi: 10.1016/s0894-7317(97)70062-2. [DOI] [PubMed] [Google Scholar]
- 4.Devereux RB, Alonso DR, Lutas EM, Gottlieb GJ, Campo E, Sachs I, Reichek N. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol. 1986;57:450–458. doi: 10.1016/0002-9149(86)90771-x. [DOI] [PubMed] [Google Scholar]
- 5.Otterstad JE, Froeland G, St John SM, Holme I. Accuracy and reproducibility of biplane two-dimensional echocardiographic measurements of left ventricular dimensions and function. Eur Heart J. 1997;18:507–513. doi: 10.1093/oxfordjournals.eurheartj.a015273. [DOI] [PubMed] [Google Scholar]
- 6.Lang RM, Bierig M, Devereux RB, Flaschskampf FA, Foster E, Pellikka PA, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18:1440–1463. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 7.Silbiger JJ. Doppler classification of diastolic dysfunction. J Am Soc Echocardiogr. 2011;24:930–932. doi: 10.1016/j.echo.2011.05.013. [DOI] [PubMed] [Google Scholar]
- 8.Coll B, Betriu A, Alonso MM, Borras M, Craver L, Amoedo LM, Marco MP, Sarro F, Junyent M, Valdivielso JM, Fernandez E. Cardiovascular risk factors underestimate atherosclerotic burden in chronic kidney disease: usefulness of non-invasive tests in cardiovascular assessment. Nephrol Dial Transplant. 2010;25:3017–3025. doi: 10.1093/ndt/gfq109. [DOI] [PubMed] [Google Scholar]
- 9.Watson PE, Watson ID, Batt RD. Total body water volumes for adult males and females estimated from simple anthropometric measurements. Am J Clin Nutr. 1980;33:27–39. doi: 10.1093/ajcn/33.1.27. [DOI] [PubMed] [Google Scholar]
- 10.Chung SH, Chu WS, Lee HA, Kim YH, Lee IS, Lindholm B, Lee HB. Peritoneal transport characteristics, comorbid diseases and survival in CAPD patients. Perit Dial Int. 2000;20:541–547. [PubMed] [Google Scholar]
- 11.Goicoechea M, Garcia de Vinuesa S, Gomez-Campdera F, Luno J. Predictive cardiovascular risk factors in patients with chronic kidney disease. Kidney Int. 2005;67((suppl 93)):S35–S38. doi: 10.1111/j.1523-1755.2005.09308.x. [DOI] [PubMed] [Google Scholar]
- 12.Turkmen F, Emre A, Ozdemir A, Sevinc C, Erisken E, Yesilcimen K. Relationship between aortic valve sclerosis and left ventricular hypertrophy in chronic haemodialysis patients. Int Urol Nephrol. 2008;40:497–502. doi: 10.1007/s11255-007-9317-4. [DOI] [PubMed] [Google Scholar]
- 13.Wang AY, Sanderson JE. Current perspectives on diagnosis of heart failure in long-term dialysis patients. Am J Kidney Dis. 2011;57:308–319. doi: 10.1053/j.ajkd.2010.07.019. [DOI] [PubMed] [Google Scholar]
- 14.De Mattos AM, Siedlecki A, Gaston RS, Perry GJ, Julian BA, Kew CE, Deierhoi MH, Young C, Curtis JJ, Iskandrian AE. Systolic dysfunction portends increased mortality among those waiting for renal transplant. J Am Soc Nephrol. 2008;19:1191–1196. doi: 10.1681/ASN.2007040503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moradi H, Sica DA, Kalantar-Zadeh K. Cardiovascular burden associated with uremic toxins in patients with chronic kidney disease. Am J Nephrol. 2013;38:136–148. doi: 10.1159/000351758. [DOI] [PubMed] [Google Scholar]
- 16.Kimura H, Takeda K, Tsuruya K, Mukai H, Muto Y, Okuda H, Furusho M, Ueno T, Nakashita S, Miura S, Maeda A, Kondo H. Left ventricular mass index is an independent determinant of diastolic dysfunction in patients on chronic hemodialysis: a tissue Doppler imaging study. Nephron Clin Pract. 2011;117:c67–c73. doi: 10.1159/000319649. [DOI] [PubMed] [Google Scholar]
- 17.Whalley GA, Marwick TH, Doughty RN, Cooper BA, Johnson DW, Pilmore A, Harris DC, Pollock CA, Collins JF. IDEAL Echo Substudy Investigators. Effect of early initiation of dialysis on cardiac structure and function: results from the echo substudy of the IDEAL trial. Am J Kidney Dis. 2013;61:262–270. doi: 10.1053/j.ajkd.2012.09.008. [DOI] [PubMed] [Google Scholar]
- 18.Chang JM, Chen SC, Huang JC, Su HM, Chen HC. Anemia and left ventricular hypertrophy with renal function decline and cardiovascular events in chronic kidney disease. Am J Med Sci. 2014;347:183–189. doi: 10.1097/MAJ.0b013e31827981be. [DOI] [PubMed] [Google Scholar]
- 19.Zoccali C. Left ventricular systolic dysfunction: a sudden killer in end-stage renal disease patients. Hypertension. 2010;56:187–188. doi: 10.1161/HYPERTENSIONAHA.110.151829. [DOI] [PubMed] [Google Scholar]
- 20.Wang AY, Wang M, Lam CW, Chan IH, Lui SF, Sanderson JE. Heart failure in long-term peritoneal dialysis patients: a 4-year prospective analysis. Clin J Am Soc Nephrol. 2011;6:805–812. doi: 10.2215/CJN.07130810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Han SH, Lee SC, Kang EW, Park JK, Yoon HS, Yoo TH, Choi KH, Han DS, Kang SW. Reduced residual renal function is associated with endothelial dysfunction in patients receiving peritoneal dialysis. Perit Dial Int. 2012;32:149–158. doi: 10.3747/pdi.2010.00111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu CK, Huang YT, Lin HH, Yang CY, Lien YC, Lee JK, Huang JW, Hung KY. Dissecting the mechanisms of left ventricular diastolic dysfunction and inflammation in peritoneal dialysis patients. PLoS One. 8;5:e62722. doi: 10.1371/journal.pone.0062722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lin HH, Lee JK, Yang CY, Lien YC, Huang JW, Wu CK. Accumulation of epicardial fat rather than visceral fat is an independent risk factor for left ventricular diastolic dysfunction in patients undergoing peritoneal dialysis. Cardiovasc Diabetol. 2013;12:127. doi: 10.1186/1475-2840-12-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang AY. The ‘heart’ of peritoneal dialysis. Perit Dial Int. 2007;27((suppl 2)):S228–S232. [PubMed] [Google Scholar]
- 25.Caliskan Y, Ozkok A, Akagun T, Alpay N, Guz G, Polat N, Tufan F, Ecder T, Bozfakioglu S. Cardiac biomarkers and noninvasive predictors of atherosclerosis in chronic peritoneal dialysis patients. Kidney Blood Press Res. 2012;35:340–348. doi: 10.1159/000332084. [DOI] [PubMed] [Google Scholar]