Abstract
Climate change poses direct or indirect influences on physiological mechanisms in plants. In particular, long living plants like trees have to cope with the predicted climate changes (i.e. drought and air warming) during their life span. The present study aimed to quantify the consequences of simulated climate change for foliar N metabolites over a drought-rewetting-drought course. Saplings of three Central European oak species (i.e. Quercus robur, Q. petraea, Q. pubescens) were tested on two different soil types (i.e. acidic and calcareous). Consecutive drought periods increased foliar amino acid-N and soluble protein-N concentrations at the expense of structural N in all three oak species. In addition, transient effects on foliar metabolite dynamics were observed over the drought-rewetting-drought course. The lowest levels of foliar soluble protein-N, amino acid-N and potassium cation with a minor response to drought and air warming were found in the oak species originating from the driest/warmest habitat (Q. pubescens) compared to Q. robur and Q. petraea. Higher foliar osmolyte-N and potassium under drought and air warming were observed in all oak species when grown on calcareous versus acidic soil. These results indicate that species-specific differences in physiological mechanisms to compensate drought and elevated temperature are modified by soil acidity.
Introduction
Current climate change scenarios suggest a significant increase in annual average air temperature (ca. 2.6°C) and significantly reduced summer precipitation by the end of the 21st century compared to 1986–2005 levels in Central Europe [1–4]. Whether woody plants could acclimate to these changes in climate strongly depends on their specific ecological requirements and genetic variability [5]. For example, oak species growing in regions of Europe with relatively dry and warm climate (e.g. Q. pubescens and Q. suber) are considered drought- and thermo-tolerant due to their deep penetrating root system, xeromorphic leaf structure and the capacity to interrupt CO2 assimilation due to drought and to resume it after rewatering [6–10]. Therefore, these species are expected to withstand or even benefit from future climate change [11–12], whereas other species with relevant shallow rooting systems such as spruce and beech are thought to suffer from impaired water balance [5, 13–15].
Drought and air warming, however, not only affect the water relations of trees, but also their nitrogen balance as indicated by foliar N composition, storage and remobilization [5]. Foliar N storage and remobilization processes respond to drought and enhanced air temperature with compensation mechanisms depending on leaf stomatal conductance and rates of net photosynthesis [5, 16–17]. For example, up-regulation of free amino acid levels in leaves of higher plants is frequently observed and thought to contribute to osmotic adjustment under various stress conditions [18–22]. The reduced cell damage operated by mechanisms of leaf turgor maintenance often occurs at the expense of other leaf N pools, in particular when whole plant N cycling is impaired as a consequence of drought and heat [5, 23]. However, differences among tree species in their capability to mobilize leaf internal N pools for osmotic adjustment by free amino acid accumulation have only recently been elucidated [24–26].
At the end of a “model-ecosystem” experiment with oak saplings exposed to drying-rewetting course for three consecutive years, drought but not air warming caused a strong decline in net photosynthesis and accumulation of amino acids in the leaves at the expense of structural N in the three most abundant European oak species (i.e. Quercus robur L., Q. petraea [Matt.] Libel. and Q. pubescens Willd.) [9, 25]. Air warming increased stomatal aperture [27] and caused seasonal up-regulation of net photosynthesis [9], but other leaf traits such as composition and contents of non-structural carbohydrates and nitrogen metabolites were not affected [25, 28]. However, physiological differences between oak species were not consistent with their drought tolerance. Q. pubescens, originating from dry habitats, showed higher drought tolerance compared to Q. robur regarding growth, but more Q. pubescens trees were counted with leaf drought injury symptoms in their foliage (leaf margin necrosis and yellowing) than Q. petraea, originating from wet and moderately dry habitats [10]. Net photosynthesis of the three oak species was similar under well-watered conditions [29].
In the present study, we focused on the development of leaf nitrogen metabolite levels in response to a soil drying-rewetting-drying course with and without air warming in leaves of the three most abundant European oak species (i.e. Quercus robur L., Q. petraea [Matt.] Libel. and Q. pubescens Willd.). Building on a previous studiy which reported other results of these treatments [25], we hypothesize that (і) effects of drought on foliar N composition at the end of a 1st drought period (1stD) were compensated by rewetting, whereas the 2nd period of drought (2ndD) would generate significant treatment effects on foliar N composition; (ii) drought and air warming would increase foliar concentrations of organic substances for osmotic adjustment, such as the selected osmolyte-N (reflected by free amino acids accumulation), and inorganic osmolyte cations such as potassium; (iii) foliar N dynamics during a drying-rewetting-drying course would be modified by the soil type.
Materials and Methods
Ethic statement
Field sampling in this study was permitted by Swiss Federal Research Institute for Forest, Snow and Landscape Research (WSL). The study did not involve endangered or protected species.
Experimental design and oak sapling establishment
The present study was part of the interdisciplinary oak research project “Querco” which was initiated in 2003 and experimentally carried out 2006 to 2009 in the research model ecosystem facility of the Swiss Federal Institute for Forest, Snow and Landscape Research (WSL) in Birmensdorf, Switzerland (545 m a.s.l., 47°21'48'' N, 8°27'23'' E). Experimental design and treatment details have previously been reported [8–9, 10, 30]. A total of 16 large (3m height, 6.7 m2 area) hexagonal ecosystem chambers were arranged in a Latin square with four repetition chambers for each treatment. The four climate treatments were (i) control (CO, with ambient conditions), (ii) increased air temperature = air warming (AW), (iii) reduced irrigation = drought (D), and (iv) air warming and drought (AWD). The four climate treatments were applied during the growing season from April to October each in 2007, 2008 and 2009. Natural rainfall was excluded from the chambers by automatic closure of the glass roofs at the beginning of rainfall events. During winter between the growing seasons, the roofs were kept open to allow for natural rainfall exposure. Drought periods were artificially applied by stopping irrigation for several consecutive weeks during selected periods. Air temperature was passively increased by reducing the opening angle of the chamber glass side walls. Each chamber was split into two 1.5 m height concrete-walled soil lysimeter compartments and filled in spring 2005 with either calcareous Fluvisol (sandy loam, pH 6.9) or acidic Haplic Alisol (loamy sand, pH 4.0) to 1 m depth with soil originating from mixed natural oak stands. Underneath the soil layers, a 0.5 m pure quartz gravel drainage stratum was established. The soils were selected for different acidity with otherwise similar properties (for details on soil physical and chemical properties see [30]).
The three oak species (i.e. Quercus robur L., Q. petraea [Matt.] Liebl. and Q. pubescens Willd.) were represented in the experiment by 4 provenances each. Acorns collected in 2003 originated from ‘natural’ oak stands located in the Swiss midland, western (Jura) and southern Switzerland, representing the Swiss distribution of the species (for details on plants and planting see [10], for genetic information see [30]). For the present study, the midland provenance was selected for Q. robur, the Jura provenance for Q. petraea and the southern Switzerland provenience for Q. pubescens, according to main territories of distribution (for details of the provenance sites see [8]).
The present study has been carried out during the drought treatments in the year 2009 with two prolonged water deprivation periods plus one short rewetting period in between for sapling recovery. This led to minimum soil water levels of less than 0.05 m3 m-3 in the entire calcareous soil profiles at the end of the first and during the second drought period in 2009 [30]; periods of drought < 0.06 m3.m-3 are shown in Table 1. The first prolonged period without irrigation (= 1stD) took place from 10th, March until 30th, June 2009 (113 days) and the second drought period (= 2ndD) from 7th, July to 20th, August 2009 (45 days) with a short rewetting period in between (= RW) from 1st to 6th, July (6 days) [30]. The irrigation of the control chambers remained 30% higher in 2009 compared to the long-term average of the local mean ambient precipitation (728 mm, from April to October, 1963 to 2009). Drought treatments had 43% lower irrigation than the mean local precipitation [30]. Mean predawn water potentials in the leaves of drought-exposed saplings decreased to a level between -1.9 and -2.6 MPa by the end of the 1stD period (minimum to maximum average over both soil types and D & AWD of the three species) and between -2.9 and -3.4 MPa at the end of the 2nd D period [10]. For the enhanced air temperature treatment, average daytime air temperature inside the chambers was increased by 1 to 2°C (Table 1), and average soil temperature at a depth of 12 cm increased by 0.5 to 1°C compared to the controls during the growing season of 2009 [30].
Table 1. Drought periods and air warming.
Leaf sampling
Leaf samples were collected at three different dates during the growing season of 2009, i.e. at the end of the 1stD period (30th, June), shortly (8 days) after the RW period (14th, July) and at the end of the 2ndD period (20th, August). For biochemical analyses, five to six leaves with approximately 1.5–2 g total fresh weight were taken from each tree, with four oak tree repetitions for each species, climate treatment, and soil type at each harvest. All leaf samples were immediately shock-frozen in liquid nitrogen and stored at -80°C until further analysis. Prior to biochemical analyses, all leaf samples were ground to a homogeneous fine powder in liquid nitrogen.
Biochemical analyses
Quantification of foliar total carbon and nitrogen concentrations
Foliar total carbon and nitrogen contents were determined as described by [31–32]. The dried leaf material (at 60°C for 48 h) with aliquots of 2.0–2.5 mg were weighed into tin capsules (IVA Analysentechnik, Meerbusch, Germany) and analyzed using a C/N elemental analyzer (NA 2500, CE Instruments, Milan, Italy) coupled by a Conflo II interface (Thermo Finnigan MAT, Bremen, Germany) to an isotope ratio mass spectrometer (Delta plus, Thermo Finnigan MAT, Bremen, Germany).
Fine grounded dry leaf powder (Retsch MM2000 zirkonoxid-bowl ultra-centrifuge mill), was digested in a high-pressure microwave system (UltraClav by Milstone: 240°C, 12MPa) and potassium was analyzed in duplicate (range < 10%) by ICP-OES (Optima 7300DV by Perkin Elmer) at WSL central laboratory, according to ISO 17025.
Quantification of foliar total soluble protein concentrations
Total soluble proteins were extracted from finely grounded frozen homogenized leaf powder and quantified using a modified version of the method described by [32–33]. To remove phenolic constituents, approximately 0.05 g were weighed and mixed with approximately 0.15 g polyvinylpolypyrrolidone (PVPP 6755, Sigma-Aldrich, Steinheim, Germany). For the extraction of soluble proteins, 1.5 mL buffer containing 50 mM Tris-HCl (pH 8.0), 1 mM ethylenediaminetetraacetic acid (EDTA), 15% glycerol (v/v), 100 mM phenylmethysulfonyl fluoride (PMSF), 1 M dithiothreitol (DTT) and 1‰ Trition X-100 were added. After incubation for 30 minutes at 4°C, samples were centrifuged at 14,000 g for 10 min at 4°C. A total of 500 μL 10% trichloroacetic acid was added to an aliquot of 500 μL of the supernatant followed by 10 min incubation at room temperature for protein precipitation. After 10 min centrifugation at 14,000 g and 4°C, the pellets were dissolved in 1 mL 1M KOH. Protein concentrations were quantified by adding 200 μL Bradford reagent (Amresco, Solon, Ohio, USA) to 5 μL aliquots of the extracts in 96-well micro-titer plates (Sarstedt, Newton, North Carolina, USA). All samples were measured in three technical replicates. Optical density was determined with a micro-plate spectrophotometer (Tecan, Groedig, Austria) at 595 nm wavelength. Bovine serum albumin (BSA A-7030, Sigma-Aldrich, Munich, Germany) was used as standard.
Quantification of total free amino acid concentrations in the leaves
Foliar amino acids were extracted using the method described by [34]. For this purpose, approximately 0.05 g finely homogenized frozen leaf powder were added to 1 mL methanol:chloroform (3.5:1.5, v:v) and 0.2 mL Hepes-buffer (containing 20 mM Hepes, 5 mM ethylene glycol tetra acetic acid (EGTA) and 10 mM NaF, pH 7.0). After 30 min incubation on ice, water-soluble amino acids were extracted twice by adding 600 μL distilled water each time; the suspension was vortex briefly and centrifuged for 5 min (14,000 g, 4°C). The combined supernatants were transferred to new tubes and stored on ice. Total amino acid concentrations were quantified from the combined supernatants using a modification of the method described by [32]. For this purpose, aliquots of 100 μL extract plus 100 μL ninhydrin reagent (1:1 mixture of solution A containing 4.2 g monohydrate citric acid and 0.134 g anhydrous stannous chloride in 40 ml 1M NaOH, made up to 100 ml with distilled water (pH 5.0) and solution B containing 4 g ninhydrin in 100 mL ethylene-glycol-monomethylether) were boiled at 100°C for 30 minutes. Isopropanol (1.25 ml, 50%) was added to the samples followed by 15 min incubation at room temperature in the dark. The optical density was determined using a UV-DU650 spectrophotometer (Beckman Coulter, Fullerton, CA, USA) at 570 nm wavelength. L-glutamine was used as standard.
Quantification of specific free amino acid compounds and ammonium concentrations in the leaves
Amino acids and ammonium were extracted as described above and the combined supernatants were freeze-dried and re-dissolved in 0.5 mL of 0.02 M HCl. Derivatization and quantification of specific amino acid compounds was performed as described by [35]. For derivatization, 5 μL of the re-dissolved extracts were mixed with 35 μL of AccQ-Tag Ultra Borate buffer and 10 μL of AccQ-Tag Reagent (both Waters, Milford, MA, USA). The samples were derivatized for 10 min at 55°C after incubation for 1 min at room temperature. An internal standard (Norvaline; 10 μM in reaction mixture) was added to the AccQ-Tag Ultra Borate buffer to account for differences in the efficiency of derivatization between samples. Determination of the composition and concentrations of specific amino acid compounds and ammonium were performed with an ultra-performance liquid chromatography system (Waters, Milford, MA, USA) equipped with an AccQ-Tag Ultra column (2.1 × 100 mm; Waters, Milford, MA, USA). Standard H of amino compounds (NCI0180, Pierce, Rockford, IL, USA) was used as analytical standard with amino compounds and ammonium contents added according to the composition of the leaf samples.
Quantification of foliar nitrate concentrations
Foliar nitrate contents were quantified using the method described by [32]. For extraction, approximately 0.05 g of frozen homogenized leaf material was added to 0.1 g polyvinylpyrrolidone (PVPP 6755, Sigma-Aldrich, Steinheim, Germany) soaked in 1 mL distilled water for 2 h. After shaking in the dark at 4°C for 1 h, extracts were boiled at 100°C for 10 min and centrifuged (14,000 g) at 4°C for 10 min. Nitrate in the supernatant was determined with an ion chromatograph (DX 120, Dionex, Idstein, Germany) coupled to an auto-sampler (AS 3500, Thermo Separation Products, Piscataway, NJ, USA). The ion chromatograph was equipped with a guard column (RFIC Ionpac AG9-SC, 4 × 50 mm, Dionex, Sunnyvale, CA, USA), an analytical column (IonPac AS9-SC, 4 × 250 mm, Dionex, Sunnyvale, CA, USA) and a self-regenerating suppressor (ASRS-ULTRA II, 4 mm, Dionex, Sunnyvale, CA, USA). Anion mixtures of NO3 -, PO4 3-, SO3 2-, and SO4 2- in distilled water were used as standards [25]. Foliar nitrate levels detected in the present study were extremely low (ca. 4.43 ± 0.55 μg NO3 - -N g-1 DW) compared to other N compounds as also observed by [36].
Calculation of foliar structural N concentrations
Foliar structural N contents were calculated by subtracting the N fractions in total soluble proteins and total amino acids from total nitrogen. Calculations were based on the dry weight (DW) of the samples. The ammonium-N contents in oak leaves determined by UPLC analyses and the nitrate concentrations determined by ion chromatography were found to be negligible; therefore, ammonium and nitrate contents were not considered for the calculation of foliar structural N in the present study.
Statistical analysis
All statistical analyses in the present study were performed using SAS 9.1 (Institute Inc., Cary NC, USA), SPSS 20.0 (SPSS, Chicago, IL, USA) and Sigmaplot 11.0 (Systat Software, Erkrath, Germany). First, either Kolmogorov-Smirnov or Shapiro-Wilk tests were performed to test for normal distribution. Analysis of variance (ANOVA) was performed to determine the main effects. For the treatment effects, the main factors AW, D and their interaction were tested against "chamber mean squares" (error term = interaction row*column*AW*D), according to the Latin square arrangement of the chambers. The effects of soil type with interaction with the treatments were tested against "subplot" mean squares (error term = interaction row*column* AW*D*soil) and the effects of the drought periods and their interactions were tested as nested within the subplot mean squares (error term = row*column*AW*D*soil*period). Significant differences (i) across climate treatments within each species, the drought-rewetting-drought course and soil type, (ii) across species within each climate treatment, the drought-rewetting-drought course and soil type, (iii) across the drought-rewetting-drought course within each climate treatment, species and soil type were tested using post-hoc Bonferroni multiple comparison tests (β = α/n, α = significance level of 0.05; n = number of tests). Pair-wise t-tests were used to detect significant differences between the two soil types within each climate treatment, species and the drought-rewetting-drought course. Differences were considered significant at P ≤ 0.050. All statistical tests were based on four independent repetitions of oak saplings (individual trees) for each climate treatment, species, time point within the drought-rewetting-drought course, and soil type.
Results
Responses of foliar total C and N concentrations and the C:N ratio to the treatments
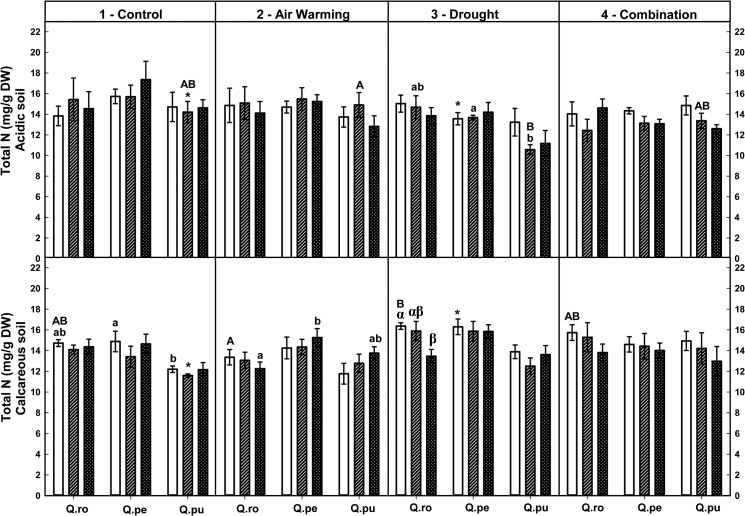
Overall, the strongest effects on foliar total C and the C:N ratio but not on foliar total N occurred by the drought-rewetting-drought course in all treatments (Table 2, Figs 1–3). Moreover, foliar total C concentrations of all oak species were on average increased after RW and the 2ndD period compared to the 1stD period regardless of treatment and soil type (P ≤ 0.035, Fig 3). Effects on foliar N concentrations as well as C:N ratio were not changed by the soil type at the end of the 2ndD period (Table 2, Figs 1 and 2).
Table 2. Main effects table during the experimental periods of 2009.
| Factor | Total N | Total C | C:N | Total protein-N | Total amino acid-N | Structural-N |
| Changes | ||||||
| Species(spec) | Qpu<Qro = Qpe *** | Qro<Qpe = Qpu ** | Qro = Qpe<Qpu *** | Qpu<Qpe<Qro *** | Qpu = Qpe<Qro *** | Qpu = Qro<Qpe *** |
| Period | ns | 1<2<3 *** | 1<2 = 3 *** | 2<1<3 *** | 2 = 1<3 *** | 3<2 = 1 *** |
| AW | ns | -1.4 ** | ns | ns | ns | ns |
| D | ns | ns | ns | +20.6 ** | +48.3 *** | -8.4 * |
| Soil | ns | -2.3 ** | ns | +9.6 ** | +15.2 *** | ns |
| Significant interactions | spec*AW,period*D,D*soil | spec*period | spec*AW,D*soil | spec*period,spec*D,spec*soil,period*AW,AW*D | spec*period,spec*D,period*D,period*soil,AW*D,D*soil | spec*period,spec*AW,period*D,D*soil |
| Factor | Potassium (K) | Gln-N | Gly-N | Glu-N | Asp-N | Ser-N |
| Changes | ||||||
| Species(spec) | Qpu = Qpe<Qro *** | Qpe = Qpu<Qro *** | ns | Qpu = Qpe<Qro ** | ns | ns |
| Period | na | 1 = 2<3 *** | 2 = 3<1 *** | ns | ns | ns |
| AW | ns | ns | +19.6 * | +18.6 (*) | +25.9 * | ns |
| D | -10.0 ** | +237.3 *** | +18.6 * | +53.7 ** | +33.6 * | +30.1 ** |
| Soil | +16.5 ** | ns | ns | ns | ns | ns |
| Significant interactions | spec*period,spec*D,period*D,period*soil | spec*soil,AW*soil | spec*period,period*soil,AW*D | spec*period,AW*D,D*soil | spec*D,spec*soil | |
Difference among species, drought-rewetting-drought periods (1 = at the end of 1stD period, 2 = after RW period and 3 = at the end of 2ndD period) or differences versus control (± %) or calcareous versus acidic soil (± %); significant difference:
(*) P ≤ 0.1,
* P ≤ 0.05,
** P ≤ 0.01,
*** P ≤ 0.001;
ns = not significant P > 0.05;
na = not available, because foliar K concentrations were determined only at the end of 2ndD period.
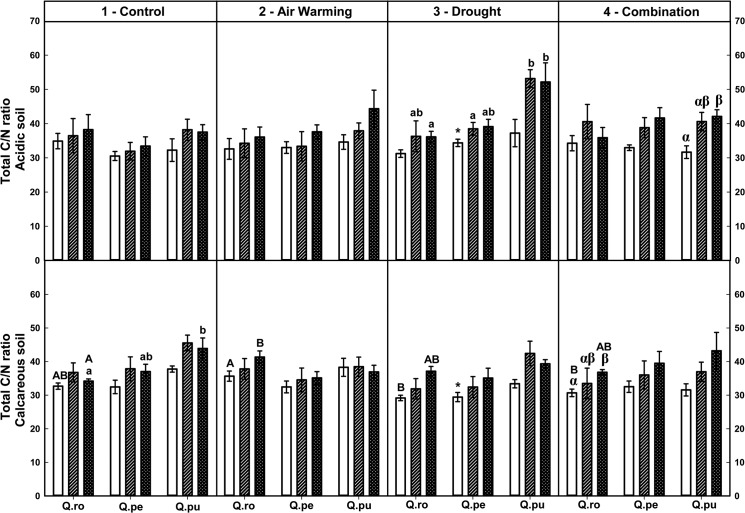
Fig 1. C:N ratio in leaves of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
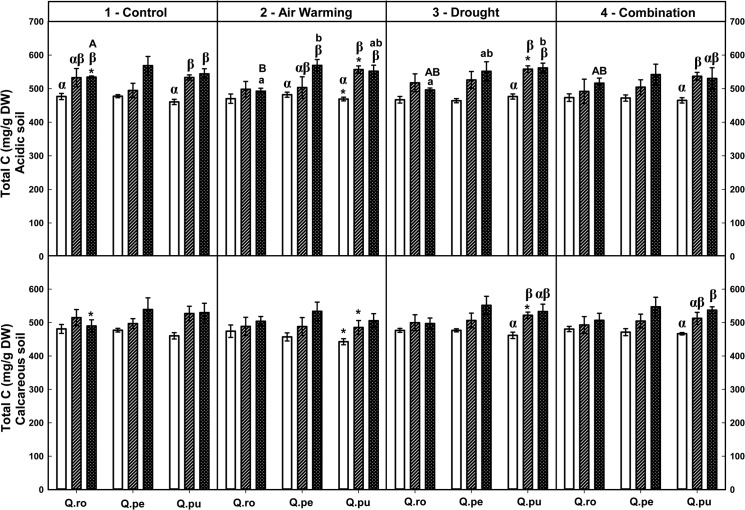
Fig 3. Foliar total C of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Fig 2. Foliar total N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Particular results show that at the end of the second drought period (2nd D), only Air warming (AW) led to increased foliar C:N ratios in Q. robur (Qro) on calcareous soil compared to the control (P = 0.009, Fig 1). At the end of the 2ndD period, the AWD treatment led to increased total C:N ratios in the leaves of Qro and Q. pubescens (Qpu compared to the 1stD period (Qro calcareous soil, Qpu acidic soil, P ≤ 0.025, Fig 1). Across species under D on acidic soil, Qpu had enhanced foliar total C:N ratios compared to Q. petraea (Qpe) (after RW) and Qro at the end of the 2ndD period (P = 0.013 and 0.034, respectively; Fig 1). In contrast, foliar total C concentration of Qro on acidic soil was significantly lower compared to Qpu (D) and Qpe (AW) at the end of the 2ndD period (P = 0.035 and 0.019, respectively; Fig 3).
Effects of drought and air warming on foliar N composition and potassium
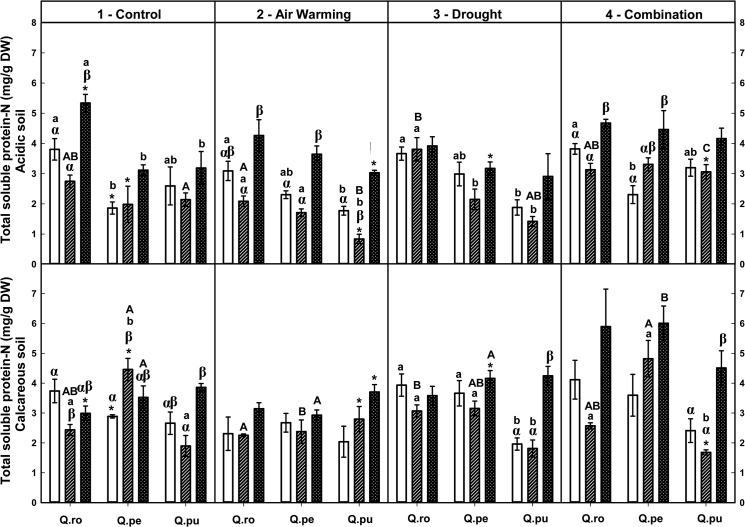
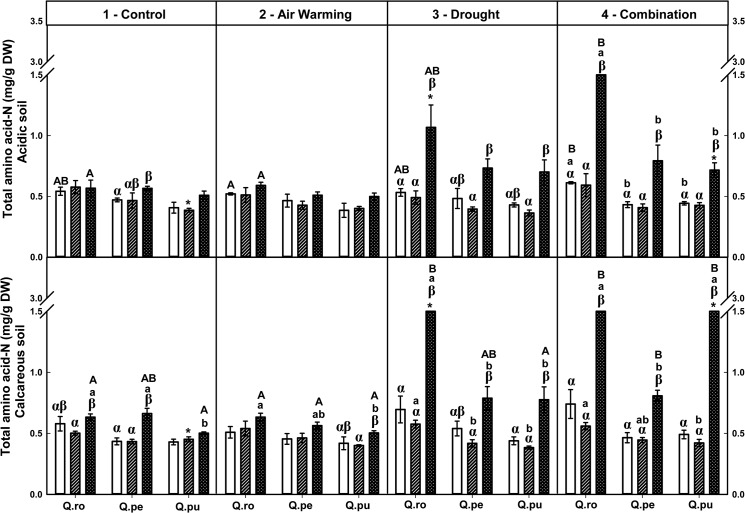
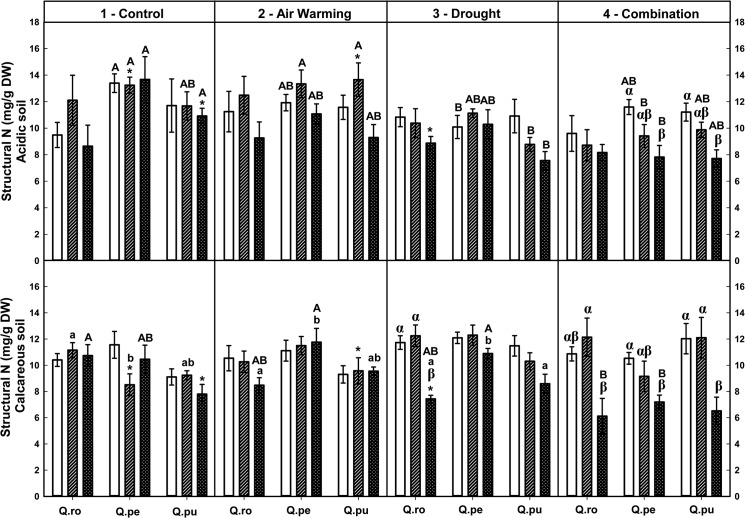
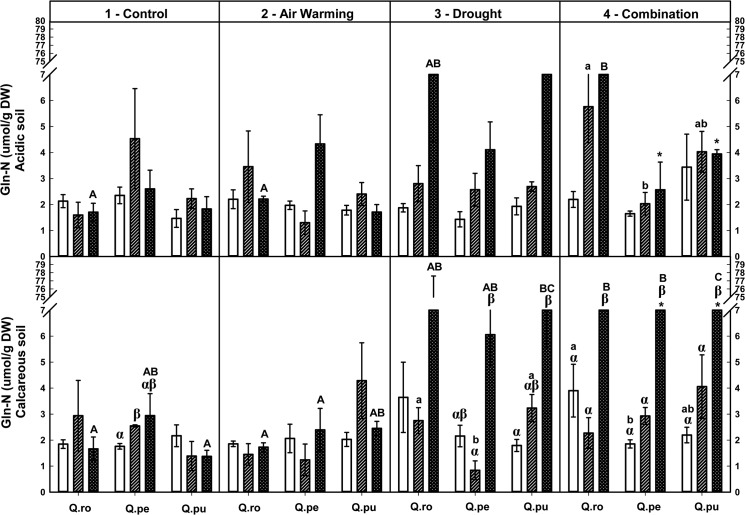
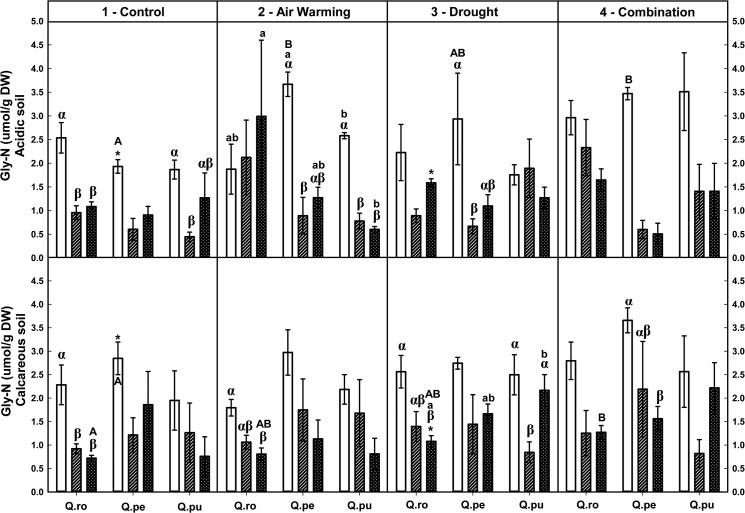
Foliar soluble protein-N and total amino acid-N concentrations were increased by the D or AWD treatment at the end of the 2ndD period on calcareous soil (Table 2, Figs 4 and 5), whereas potassium tended to decrease (Table 2, S4 Fig). Additionally, concentrations of both N fractions were increased by AWD compared to single D or AW treatment at the end of the 2ndD period (P ≤ 0.048, Figs 4 and 5). At the end of the 2ndD period, foliar structural N concentrations were decreased by drought compared to the controls (Table 2, Fig 6). All specific free amino acid-N concentrations in the leaves were increased in the D as well as AW (except AW for glutamine-N and serine-N) treatment over the drought-rewetting-drought course on both soil types (P ≤ 0.044, Table 2, Figs 7 and 8, S1–S3 Figs). In particular, glutamine (Gln)-N and serine (Ser)-N concentrations of Qro and Qpu leaves were increased with D and AWD compared to the control and AW treatment in both soil types at the end of the 2ndD period (P ≤ 0.044, Fig 7 and S3 Fig), whereas no effects were observed at the end of the 1stD period and after RW (Fig 7).
Fig 4. Foliar total soluble protein-N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Fig 5. Foliar total amino acid-N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Fig 6. Foliar total structural N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Fig 7. Foliar Glutamine (Gln)-N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Fig 8. Foliar Glycine (Gly)-N of three oak species exposed to different climate treatments and soil types during a drought-rewetting-drought course.
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
Variation in foliar N metabolites during the drought-rewetting-drought course
Foliar soluble protein-N and amino acid-N concentrations were generally increased at the end of the 2ndD period compared to the 1stD period and RW, particularly in the D and AWD treatment. As a tendency their concentrations were lower after rewatering (Table 2, Figs 4 and 5). Calculated structural N concentrations were decreased accordingly (Table 2, Fig 6). Also foliar Gln-N concentrations were significantly higher at the end of the 2ndD period than the 1stD period and RW (RW showed a tendency to lower values) in the D and AWD treatment on calcareous soil (P ≤ 0.025, Fig 7), but not Gly-N concentrations (Fig 8). Similar patterns were observed for Ser-N and Asp-N concentrations in Qro and Qpe leaves in response to D or AWD treatment compared to RW on calcareous soil (P < 0.025, S2 and S3 Figs).
Responses of foliar N metabolites and potassium as related to species and soil type
Over all climate treatments and both soil types foliar soluble protein-N concentration decreased in the order: Qro > Qpe > Qpu at the end of the 1stD period and RW (P ≤ 0.034), but at the end of the 2ndD period only on acidic soil (P ≤ 0.007, Fig 4, Table 2). Moreover, foliar total amino acid-N concentrations were highest in Qro compared to Qpe and Qpu in the D or AWD treatment across the drought-rewetting-drought time course (P ≤ 0.033, Fig 5); potassium was highest in Qro compared to Qpe and Qpu over all treatments (Table 2, S4 Fig). Also significant differences in particular amino acids among species (Gln, Glu, Table 2, Fig 7 and S1 Fig) depended on the treatments, soil types and periods.
Comparing the two soil types, levels of foliar soluble protein-N, amino acid-N and potassium in all oak species were significantly enhanced on calcareous soil compared to acidic soil after RW and at the end of the 2ndD period (P ≤ 0.042, Table 2, Figs 4 and 5). Foliar Gln-N in Qpe and Qpu showed higher levels on calcareous than on acidic soil at the end of the 2ndD period in the AWD treatment (P ≤ 0.029, Fig 7). Moreover, pooled over oak species, leaf Glu-N, Asp-N and Ser-N concentrations were enhanced on calcareous compared to acidic soil at the end of the 2ndD period (P ≤ 0.040, S1– S3 Figs).
Discussion
Delayed responses in the dynamics of foliar N metabolites and C:N ratio over the drought-rewetting-drought course
Drought led to significantly enhanced values of foliar δ13C abundance across the entire time course applied [26] indicating severe drought stress for all oak saplings investigated. The increased leaf total soluble protein-N and amino acid-N concentrations (particularly Gln-N, Asp-N and Ser-N) were observed at the expense of structural N only at the end of the 2nd D period. Thus, the effect of drought on N partitioning in oak leaves was not immediate, but delayed. It reflects the increased need for the synthesis of N containing osmolytic compounds against the drought stress along the consecutive drought-rewetting-drought course. Apparently, this enhanced need for N was not met by the mobilization of leaf external N sources, such as N stored in bark and wood [37], but only at the expense of internal structural N in the three oak species studied. Iincreased total soluble protein-N and amino acid-N concentrations were also found in the control treatment at the end of 2nd D period, but not for leaf total N, structural N concentrations and the C:N ratio. This indicated that the mobilization of an external N pool by well-watered oak trees was enough to counteract any possible phenological effect. Furthermore, an enhanced need for N containing osmolytic compounds may be due to a higher stress severity of the 2nd D period as indicated by the soil water contents, growth rate of shoots, predawn leaf water potential, stomata conductance and net photosynthesis of the leaves [9, 38]. Apparently, osmolytic compounds accumulated during the 1st D period and maintained during the short-recovery phase [26], were not sufficient to counteract drought stress during the 2nd D period. Similar effects along the drought-rewetting-drought course were found for the foliar C:N ratio that increased due to higher leaf C and lower N concentrations at the end of 2nd D period. From these results it appears that drought mediated changes in N partitioning of oak leaves were strongly related to the severity of drought stress.
Foliar N metabolite partitioning and total C, N and K concentrations response to climate treatments
Drought increased foliar soluble osmolyte-N concentrations as reflected by amino acid accumulation, confirming hypothesis (ii) whereas leaf structural N concentrations of oak saplings were reduced in the drought treatments (i.e. D and / or AWD) compared to well-irrigated plants (AW and controls). This suggests that drought-stressed leaves invested more N resources into organic N solutes as an acclimation strategy under impaired photosynthesis rates [9]. Moreover, accumulation of amino acids in the leaves is commonly considered as a cellular stress response to drought that plays a major role in the osmotic adjustment thereby promoting leaf turgor maintenance and reducing cell damage [39–42]. In many woody species, specific amino acids and cations such as proline (Pro) and/or γ-aminobutyric acid (GABA) and K+ Na+ are the principal compounds involved in osmotic adjustment under drought [18, 21, 43–44]. The increases in total amino acid-N as well as specific amino acid-N concentrations (i.e. glutamine (Gln)-N, serine (Ser)-N, aspartic acid (Asp)-N, glycine (Gly)-N) in D treated leaves over the drought-rewetting-drought course observed in the present study are consistent with these reports, but together with many other reports (e.g. [24–26,45–46]) show that the accumulation of foliar osmolyte-N compounds under drought is more general and not restricted to the amino acids mentioned above. Thus, the enhanced concentrations of amino acids-N corroborate a close relationship with active osmotic-protection to maintain a positive foliar cell turgor of the investigated oak saplings upon drought stress [47–50]. However, foliar potassium concentration tended to be decreased in the drought treatment contradicting hypothesis (ii). It was enriched on the calcareous versus the acidic soil and appeared to rather depend on the soil type than a foliar reaction. The acidic topsoil had a Kexch (mg kg-1) of 30.7, the acidic subsoil a Kexch (mg kg-1) of 19.0 compared to the calcareous soil with 24.8 over both layers [30]. Drought shifted root distribution to lower depths [51], which ensured an increased potassium nutrition. The mechanisms favoring either foliar amino acids for osmoprotection against drought (oaks, present study) over cations (K+, birch, [52]) may depend on nutrient availability in the soil and species properties.
The lack of air warming induced accumulation of foliar osmolyte-N concentrations in the tested oak saplings in contrast to hypothesis (ii), may be the result of the small increase in air temperature in the present experiment (increase in daily air temperature: approx. +1 to 2°C). A similar lack of effects of a minor increase of air temperature on the foliar N pools of trees was also found in other studies (reviewed by [5, 23, 53–54]). Still, the combination treatment AWD showed a synergistic amplified effect on leaf soluble N composition compared to the single D or AW treatments, comparable to the combined negative effect of this treatments on plant net primary production (NPP) observed in other studies [55–56]. This result clearly shows that the evaluation of potential consequences of climate change requires the combined analysis of different climate factors to identify synergistic, antagonistic, or additive effects [53, 56–57]. Similar combined effects of D and AW were reflected in the present experiment by decreased predawn leaf water potential and net photosynthesis rates under AWD compared to D or AW [9].
Surprisingly, AW caused an enhanced C:N ratios in the leaves of oaks at the end of 2nd D period. Several laboratory studies showed lower leaf N concentrations in plants growing at high compared to low temperatures [58–59]. For instance, for forb (i.e. Ambrosia psilostachyia, Aster ontarionis, and Aster ericoides) and oak species (i.e. Q. robur) studied in warmer regions, AW quickly inhibited N mineralization processes in the soil resulting in a progressive decrease in foliar N concentrations compared to ambient temperature controls [60–62]. Decreased leaf N concentration and increased C:N ratios were also found in a global survey of over 1000 plant species on the relationship between leaf C and N levels and local climate indices: leaf N levels decreased when mean annual air temperature increased [63]. This effect has been interpreted as evidence for an offsetting mechanism to reduced rates of biochemical reactions caused by the low efficiency of N-rich enzymes at low temperatures [58–59, 64–65], which was obviously in the present study with a relatively small temperature difference not the case.
Foliar N metabolite partitioning as related to species and soil type
In former studies, Qro showed higher water requirements and was more drought- and thermo-sensitive than Qpe and Qpu [66–67], the latter with a main distribution in dry and warm Mediterranean areas of Southern Europe [6, 68]. In the present study, Qro had highest foliar total protein-N, total amino acid-N (including in particular Gln-N, Gly-N, Glu-N) as well as potassium (K+) concentrations regardless of climate treatment and soil type. It also showed a significantly impaired photochemical efficiency of PSII (FV/FM) as a consequence of reduced pre-dawn leaf water potentials under D treatments compared to Qpe and Qpu [9]. Apparently, the requirements for the synthesis of N containing osmolytic compounds and other important osmolyte (i.e. K) in response to D and AW treatment increased from Qpu via Qpe to Qro. These results are in line with the most xeromorphic leaf structure (higher pilosity, leaf mass per area (LMA), chlorophyll concentration, C/N ratio, lower leaf size and water content) of Qpu compared to Qro and Qpe under D stress in the present experiment [10].
As hypothesized (iii), foliar N dynamics in the studied oak species during drying-rewetting-drying course depended on soil type, indicated by generally enhanced levels of total soluble protein-N, total amino acid-N and potassium in oak leaves on calcareous compared to acidic soil after the 2ndD period regardless of climate treatment. Soil type has been identified as an important factor that strongly modifies the effects of the experimental climate treatments [69–72]. Generally, oaks preferentially develop on acidic soil [12, 73], as also indicated by the foliar biomass of Qro and Qpe in the present experiment [10]. Considering the lower leaf chlorophyll concentrations and lower stand biomass of oaks growing on calcareous compared to acidic soil as well as the enhanced total N, phosphate and/or manganese contents in the leaves of oaks on acidic soil [10, 30, 74], oak saplings developing on calcareous soil in the present study exhibited a stronger foliar N acclimation capability in response to the D and AW treatments than on acidic soil by investing their internal structural N into the synthesis of N containing osmolytes only during the 2nd D period.
Conclusions
The present results on foliar N metabolites highlight the generally high drought tolerance of oak species, but also the significance of length and intensity of drought events and rewatering as well as species-specific differences. The first drought event was not intense enough to induce significant changes, but the second drought event of the drought-rewetting-drought course applied significantly induced the formation of more soluble protein and amino acids. Apparently, these compounds were increased for osmolytic adjustment in the leaves of all species at the expense of structural N by leaf-internal processes rather than changes in whole-plant distribution of N. The latter is indicated by the generally unchanged total leaf N content of the leaves. Surprisingly, the increase in soluble amino acids was not only attributed to compounds known to be drought responsive such as Pro and GABA, but to a whole set of major proteinogenic amino acids, including also Gln, Gly, Glu, Asp, and Ser. This results indicates general reprogramming of foliar N metabolism in response to drought and requires further analysis of the molecular changes responsible. All three oak species studied showed these responses, but differed considerably in response of intensity. Qpu showed a rather lower reaction to drought in the concentrations of osmotic N compounds, but was shown in another study of this experiment to be drought resistant by morphological and photosynthetic acclimation [9–10]. Reaction intensity of foliar N metabolism to the drought-rewetting-drought course was also highly dependent on soil substrate. The better nutrient availability in the calcareous soil induced stronger changes in soluble N compounds. On one hand, K+, known for its function for osmotic adjustment was not significantly changed by the D or AW treatment, irrespective of the different N-supply of the two soil types studied. Qpu, the species growing naturally under dryer conditions than Qpe and Qro, showed constitutively (in the control) higher K and C/N concentrations. Thus, for all biochemical parameters studied, interactions between soil and species were shown during the drought-rewetting-drought course and the D ± AW treatment, which have to be taken into account in future studies on the significance of climate change for the performance of oak trees.
Supporting Information
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Different small letters indicate significant differences across three oak species within each climate treatment and soil type. Different capital letters indicate significant differences across different climate treatments for each species and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment and species (P ≤ 0.050).
(EPS)
Acknowledgments
We thank Erika Fischer, Monika Eiblmeier and Peter Bleuler for the professional technical assistance.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
BH was financially supported by the fellowship of GRK 1305 - International Graduate School of Signal Systems in Plant Model Organisms, funded by the Deutsche Forschungsgemeinschaft (DFG), Germany, and the basic research funds for the Talents with Foreign Doctorate funded by central government, Shaanxi province and Northwest A&F University, China, respectively; JS was funded by the Ministry of Science, Research and the Arts Baden-Württemberg, Germany, and the European Social Fund. The authors are also gratefully indebted to the Velux foundation for funding (project number: 489, www.veluxstiftung.ch). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Ciais Ph, Reichstein M, Viovy N, Granier A, Ogee J, Allard V, et al. Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature. 2005; 437:529–533. [DOI] [PubMed] [Google Scholar]
- 2. Schmidli J, Frei Ch. Trends of heavy precipitation and wet and dry spells in Switzerland during the 20th century. Int J Climatol. 2005; 25:753–771. [Google Scholar]
- 3.CH2011. Swiss climate change scenarios CH2011. Zurich: C2SM, MeteoSwiss, ETH, NCCR Climate, and OcCC; 2011.
- 4. IPCC. Climate Change 2013: The Physical Science Basis In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM, editors. Contribution of working group I to the fifth assessment report of the intergovern-mental panel on climate change. United Cambridge: Cambridge University Press; 2013. pp. 1535. [Google Scholar]
- 5. Rennenberg H, Loreto F, Polle A, Brilli F, Fares S, Beniwal RS, et al. Physiological responses of forest trees to heat and drought. Plant Biol. 2006; 8:556–571. [DOI] [PubMed] [Google Scholar]
- 6. Ellenberg H. Vegetation ecology of central Europe 4th ed. New York: Cambridge University Press; 1998. [Google Scholar]
- 7. Abrams MD. Adaptations and responses to drought in Quercus species of North America. Tree Physiol. 1990; 7:227–238. [DOI] [PubMed] [Google Scholar]
- 8. Arend M, Kuster T, Günthardt-Goerg MS, Dobbertin M. Provenance-specific growth responses to drought and air warming in three European oak species (Quercus robur, Q. petraea and Q. pubescens). Tree Physiol. 2011; 31:287–297. 10.1093/treephys/tpr004 [DOI] [PubMed] [Google Scholar]
- 9. Arend M, Brem A, Kuster TM, Günthardt-Goerg MS. Seasonal photosynthetic responses of European oaks to drought and elevated daytime temperature. Plant Biol. 2013; 15 (Suppl.1):169–176. 10.1111/j.1438-8677.2012.00625.x [DOI] [PubMed] [Google Scholar]
- 10. Günthardt-Goerg MS, Kuster TM, Arend M, Vollenweider P. Foliage response of young central European oaks to air warming, drought and soil type. Plant Biol. 2013; 15 (Suppl.1): 185–197. 10.1111/j.1438-8677.2012.00665.x [DOI] [PubMed] [Google Scholar]
- 11. Zimmermann NE, Bolliger J, Gehrigfasel J, Guisan A, Kienast F, Lischke H, et al. Wo wachsen die Bäume in 100 Jahren? In: Wohlgemuth T, editor. Wald un klimawandel. Forum für wissen 2006, eidgenössische forschungsanstalt für wald, schnee und landschaft, WSL. Birmensdorf: WSL; 2006. pp. 63–71. [Google Scholar]
- 12. Landolt E, Bäumler B. Flora indicativa—ecological indicator values and biological attributes of the flora of Switzerland and the Alps Bern: Haupt; 2010. [Google Scholar]
- 13. Backes K, Leuschner C. Leaf water relations of competitive Fagus sylvatica and Quercus petraea trees during 4 years differing in soil drought. Can J Forest Res. 2000; 30:335–346. [Google Scholar]
- 14. Rennenberg H, Seiler W, Matyssek R, Geßler A, Kreuzwieser J. Die Buche (Fagus sylvatica L.)—ein Waldbaum ohne Zukunft im südlichen Mitteleuropa? AFJZ. 2004; 175:210–224. [Google Scholar]
- 15. Geßler A, Keitel C, Kreuzwieser J, Matyssek R, Seiler W, Rennenberg H. Potential risks for European beech (Fagus sylvatica L.) in a changing climate. Trees- Struct Funct. 2007; 21:1–11. [Google Scholar]
- 16. Quick WP, Chaves MM, Wendler R, David M, Rodrigues ML, Passaharinho JA, et al. The effects of water stress on photosynthetic carbon metabolism in four species grown under field conditions. Plant Cell Environ. 1992; 15:25–35. [Google Scholar]
- 17. Medrano H, Escalona JM, Bota J, Gulias J, Flexas J. Regulation of photosynthesis of C3 plants in response to progressive drought: stomatal conductance as a reference parameter. Ann Bot-London. 2002; 89:895–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Rai VK, Bapat CM. Water stress effects on amino acid metabolism during early vegetative growth in Phaseolus mungo L. Geobios-Lyon. 1997; 4:231–234. [Google Scholar]
- 19. Jones MM, Osmond CB, Turner NC. Accumulation of solutes in leaves of sorghum and sunflower in response to water deficits. Aust J Plant Physiol. 1980; 7:193–205. [Google Scholar]
- 20. Mansour MMF. Nitrogen containing compounds and adaptation of plants to salinity stress. Biol Plantarum. 2000; 43:491–500. [Google Scholar]
- 21. Rai VK. Role of amino acids in plant responses to stresses. Biol Plantarum. 2002; 45:481–487. [Google Scholar]
- 22. Ramachandra Reddy A, Viswanatha Chaitanya K, Vivekanandan M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J Plant Physiol. 2004; 161:1189–1202. [DOI] [PubMed] [Google Scholar]
- 23. Rennenberg H, Dannenmann M, Gessler A, Kreuzwieser J, Simon J, Papen H. Nitrogen balance in forest soils: nutritional limitation of plants under climate change stresses. Plant Biol. 2009; 11 (Suppl.1):4–23. 10.1111/j.1438-8677.2009.00241.x [DOI] [PubMed] [Google Scholar]
- 24. Warren CR, Aranda I, Cano FJ. Responses to water stress of gas exchange and metabolites in Eucalyptus and Acacia spp. Plant Cell Environ. 2011; 34:1609–1629. 10.1111/j.1365-3040.2011.02357.x [DOI] [PubMed] [Google Scholar]
- 25. Hu B, Simon J, Kuster TM, Arend M, Siegwolf R, Rennenberg H. Nitrogen partitioning in oak leaves depends on species, provenance, climate conditions and soil type. Plant Biol. 2013; 15 (Suppl. 1):198–209. 10.1111/j.1438-8677.2012.00658.x [DOI] [PubMed] [Google Scholar]
- 26. Hu B, Simon J, Rennenberg H. Drought and air warming affect the species-specific levels of stress related foliar metabolites of three oak species on acidic and calcareous soil. Tree Physiol. 2013; 33:489–504. 10.1093/treephys/tpt025 [DOI] [PubMed] [Google Scholar]
- 27. Contran N, Günthardt-Goerg MS, Kuster TM, Cerana R, Crosti P, Paoletti E. Physiological and biochemical responses of Quercus pubescens to air warming and drought on acidic and calcareous soils. Plant Biol. 2013; 15 (Suppl. 1):157–168. 10.1111/j.1438-8677.2012.00627.x [DOI] [PubMed] [Google Scholar]
- 28. Li MH, Cherubini P, Dobbertin M, Arend M, Xiao WF, Rigling A. Responses of leaf nitrogen and mobile carbohydrates in different Quercus species/provenances to moderate climate changes. Plant Biol. 2013; 15(Suppl. 1):177–184. 10.1111/j.1438-8677.2012.00579.x [DOI] [PubMed] [Google Scholar]
- 29. Steinbrecher R, Contran N, Gugerli F, Schnitzler J-P, Zimmer I, Menard T, et al. Inter- and intra-specific variability in isoprene production and photosynthesis of central European oak species. Plant Biol. 2013; 15 (Suppl. 1):148–156. 10.1111/j.1438-8677.2012.00688.x [DOI] [PubMed] [Google Scholar]
- 30. Kuster TM, Arend M, Bleuler P, Günthardt-Goerg MS, Schulin R. Water regime and growth of young oak stands subjected to air warming and drought on two different forest soils in a model ecosystem experiment. Plant Biol. 2013; 15 (Suppl. 1):138–147. 10.1111/j.1438-8677.2011.00552.x [DOI] [PubMed] [Google Scholar]
- 31. Gessler A, Duarte HM, Franco AC, Luettge U, Mattos EA, Nahm M, et al. Ecophysiology of selected tree species in different plant communities at the periphery of the Atlantic Forest of SE-Brazil II. Spatial and ontogenetic dynamics in Andira legalis, a deciduous legume tree. Trees- Struct Funct. 2005; 19:510–522. [Google Scholar]
- 32. Simon J, Waldhecker P, Brüggemann N, Rennenberg H. Competition for nitrogen resources between beech and sycamore maple seedlings. Plant Biol. 2010; 12:453–458. 10.1111/j.1438-8677.2009.00225.x [DOI] [PubMed] [Google Scholar]
- 33. Wildhagen H, Dürr J, Ehlting B, Rennenberg H. Seasonal nitrogen cycling in the bark of field-grown Grey poplar is correlated with meteorological factors and gene expression of bark storage proteins. Tree Physiol. 2010; 30:1096–1110. 10.1093/treephys/tpq018 [DOI] [PubMed] [Google Scholar]
- 34. Winter H, Lohaus G, Heldt W. Phloem transport of amino acids in relation to their cytosolic levels in barley leaves. Plant Physiol. 1992; 99:996–1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Luo ZB, Janz D, Jiang X, Göbel C, Wildhagen H, Tan YP, et al. Upgrading root physiology for stress tolerance by ectomycorrhizas: insights from metabolite and transcriptional profiling into reprogramming for stress anticipation. Plant Physiol. 2009; 151:1902–1917. 10.1104/pp.109.143735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Schneider S, Gessler A, Weber P, Sengbusch DV, Hanemann U, Rennenberg H. Soluble N compounds in trees exposed to high loads of N: a comparison of spruce (Picea abies) and beech (Fagus sylvatica) grown under field conditions. New Phytol. 1996; 134:103–114. [Google Scholar]
- 37. Rennenberg H, Wildhagen H, Ehlting B. Nitrogen nutrition of poplar trees. Plant Biol. 2010; 12:275–291. 10.1111/j.1438-8677.2009.00309.x [DOI] [PubMed] [Google Scholar]
- 38. Kuster TM, Dobbertin M, Günthardt-Goerg MS, Schaub M, Arend M. A Phenological Timetable of Oak Growth under Experimental Drought and Air Warming. PLoS ONE. 2014; 9(2): e89724 10.1371/journal.pone.0089724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Borowitzka LJ. Solute accumulation and regulation of cell water activity In: Paleg LG, Aspinall D, editors. The physiology and biochemistry of drought resistance in plants. Sydney: Academic Press; 1981. pp 97–130. [Google Scholar]
- 40. Zhu JK, Hasegawa PM, Bressan RA. Molecular aspects of osmotic stress in plants. Crit Rev Plant Sci. 1997; 16:253–277. [Google Scholar]
- 41. Cushman JC, Bohnert HJ. Genomic approaches to plant stress tolerance. Curr Opin Plant Biol. 2000; 3:117–124. [DOI] [PubMed] [Google Scholar]
- 42. Wang WX, Vinocur B, Altman A. Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta. 2003; 218:1–14. [DOI] [PubMed] [Google Scholar]
- 43. Clifford SC, Arndt SK, Corlett JE, Joshi S, Sankhla N, Popp M, et al. The role of solute accumulation, osmotic adjustment and changes in cell wall elasticity in drought tolerance in Ziziphus mauritiana (Lamk.). J Exp Bot. 1998; 49:967–977. [Google Scholar]
- 44. Patakas A, Nikolau N, Zioziou E, Radoglou KM, Noitsakis B. The role of organic solute and ion accumulation in osmotic adjustment in drought-stressed grapevines. Plant Sci. 2002; 163:361–367. [Google Scholar]
- 45. Fotelli MN, Rennenberg H, Gessler A. Effects of drought on the competitive interference of an early successional species (Rubus fruticosus) on Fagus sylvatica L. seedlings: 15N uptake and partitioning responses of amino acids and other N compounds. Plant Biol. 2002; 4:311–320. [Google Scholar]
- 46. De Diego N, Sampedro MC, Barrio RJ, Saiz-Fernandez I, Moncalean P, Lacuesta M. Solute accumulation and elastic modulus changes in six radiata pine breeds exposed to drought. Tree Physiol. 2013; 33:69–80. 10.1093/treephys/tps125 [DOI] [PubMed] [Google Scholar]
- 47. Turner NC, Jones MM. Turgor maintenance by osmotic adjustment: a review and evaluation In: Turner NC, Kramer PJ, editors. Adaption of plants to water and high temperature stress. New York: Wiley Interscience; 1980. pp 87–103. [Google Scholar]
- 48. Morgan JM. Osmoregulation and water-stress in higher plants. Annu Rev Plant Phys. 1984; 35:299–319. [Google Scholar]
- 49. Turner NC. Adaption to water deficits—a changing perspective. Aust J Plant Physiol. 1986; 13:175–190. [Google Scholar]
- 50. Kramer PJ, Boyer JS. Water relations of plants and soils San Diego: Academic Press; 1995. [Google Scholar]
- 51. Kuster TM, Arend M, Günthardt-Goerg MS, Schulin R. Root growth of different oak provenances in two soils under drought stress and air warming conditions. Plant Soil 2013; 369:61–71. [Google Scholar]
- 52. Gu M, Rom CR, Robbins JA, Oosterhuis DM. Effect of water deficit on gas exchange, osmotic solutes, leaf abscission, and growth of four birch genotypes (Betula L.) under a controlled environment. HortScience 2007; 42: 1383–1391. [Google Scholar]
- 53. Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, et al. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia. 2001; 126:543–562. [DOI] [PubMed] [Google Scholar]
- 54. Saxe H, Cannell MGR, Johnsen B, Ryan MG, Vourlitis G. Tree and forest functioning in response to global warming. New Phytol. 2001; 149:369–399. [DOI] [PubMed] [Google Scholar]
- 55. Luo YQ, Gerten D, Maire GL, Parton WJ, Weng ES, Zhou XH, et al. Modeled interactive effects of precipitation, temperature, and CO2 on ecosystem carbon and water dynamics in different climatic zones. Glob Change Biol. 2008; 14:1986–1999. [Google Scholar]
- 56. Larsen KS, Andresen LC, Beier C, Jonasson S, Albert KR, Ambus P, et al. Reduced N cycling in response to elevated CO2, warming, and drought in a Danish heath land: Synthesizing results of the CLIMAITE project after two years of treatment. Glob Change Biol. 2011; 17:1884–1899. [Google Scholar]
- 57. Borken W, Matzner E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob Change Biol. 2009; 15:808–824. [Google Scholar]
- 58. Tjoelker MG, Reich PB, Oleksyn J. Changes in leaf nitrogen and carbohydrates underlie temperature and CO2 acclimation of dark respiration in five boreal tree species. Plant Cell Environ. 1999; 22:767–778. [Google Scholar]
- 59. Woods HA, Makino W, Cotner JB, Hobbie SE, Harrison JF, Archarya K, et al. Temperature and the chemical composition of poikilothermic organisms. Funct Ecol. 2003; 17:237–245. [Google Scholar]
- 60. Dury SJ, Good JEG, Perrins CM, Buse A, Kaye T. The effects of increasing CO2 and temperature on oak leaf palatability and the implications for herbivorous insects. Glob Change Biol. 1998; 4:55–61. [Google Scholar]
- 61. An Y, Wan S, Zhou X, Subeda AA, Wallace LL, Luo Y. Plant nitrogen concentration, use efficiency, and contents in a tallgrass prairie ecosystem under experimental warming. Glob Change Biol. 2005; 11:1733–1744. [Google Scholar]
- 62. Way DA, Oren R. Differential responses to changes in growth temperature between trees from different functional groups and biomes: a review and synthesis of data. Tree Physiol. 2010; 30:669–688. 10.1093/treephys/tpq015 [DOI] [PubMed] [Google Scholar]
- 63. Reich PB, Oleksyn J. Global patterns of plant leaf N and P in relation to temperature and latitude. P Natl Acad Sci USA. 2004; 101:11001–11006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Körner C. Alpine plant life: functional plant ecology of high mountain ecosystems Berlin: Springer; 1999. [Google Scholar]
- 65. Weih M, Karlsson PS. Growth responses of mountain birch to air and soil temperature: is increasing leaf-nitrogen content an acclimation to lower air temperature? New Phytol. 2001; 150:147–155. [Google Scholar]
- 66. Vivin P, Aussenac G, Levy G. Differences in drought resistance among 3 deciduous oak species grown in large boxes. Ann For Sci. 1993; 50:221–233. [Google Scholar]
- 67. Gieger T, Thomas FM. Differential response of two Central-European oak species to single and combined stress factors. Trees-Struct Funct. 2005; 19:607–618. [Google Scholar]
- 68. Li C, Berninger F, Koskela J, Sonninen E. Drought responses of Eucalyptus microtheca provenances depends on seasonality of rainfall in their place of origin. Aust J Plant Physiol. 2000; 27:231–238. [Google Scholar]
- 69. Parker WC, Pallardy SG. Genotypic variation in tissue water relations of leaves and roots of black walnut (Juglans nigra) seedlings. Physiol Plantarum. 1985; 64:105–110. [Google Scholar]
- 70. Bongarten BC, Teskey RO. Water relations of loblolly pine seedlings from diverse geographic origins. Tree Physiol. 1986; 1:265–276. [DOI] [PubMed] [Google Scholar]
- 71. Abrams MD, Kubiske ME, Steiner KC. Drought and adaptations and responses in five genotypes of Fraxinus pennsylvanica Marsh: photosynthesis, water relations and leaf morphology. Tree Physiol. 1990; 6:305–315. [DOI] [PubMed] [Google Scholar]
- 72. Peuke AD, Rennenberg H. Carbon, nitrogen, phosphorus, and sulphur concentration and partitioning in beech ecotypes (Fagus sylvatica L.): phosphorus most affected by drought. Trees- Struct Funct. 2004; 18:639–648. [Google Scholar]
- 73. Wallnofer S, Hotter M. Syntaxonomy and site ecology of mixed oak forest communities in the Inner and Intermedial Alps of Tyrol (Austria). Bot Helv. 2008; 118:21–43. [Google Scholar]
- 74. Kuster TM, Schleppi P, Hu B, Schulin R, Günthardt-Goerg MS. Nitrogen dynamics in oak model ecosystems subjected to air warming and drought on two different soils. Plant Biol. 2013; 15 (Suppl. 1):220–229. 10.1111/j.1438-8677.2012.00686.x [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Vertical bar charts show mean and standard error and the bar charts were grouped for each species, in each treatment and soil type. Periods are indicated from left to right side: white bar = the first drought period (1stD), hedged bar = the rewetting period (RW), cyclone bar = the second drought period (2ndD). Different small letters indicate significant differences across three oak species within each climate treatment, period and soil type. Different capital letters indicate significant differences across different climate treatments for each species, period and soil type. Greek symbols indicate significant differences across the different drought-rewetting-drought periods for each species, climate treatment and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment, species and time course (P ≤ 0.050).
(EPS)
Different small letters indicate significant differences across three oak species within each climate treatment and soil type. Different capital letters indicate significant differences across different climate treatments for each species and soil type. Asterisks indicate significant differences between acidic and calcareous soil within each climate treatment and species (P ≤ 0.050).
(EPS)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.