Summary
Cell therapy is a potential approach for treatment of strokes. Mesenchymal stem cells (MSCs) are a potential cell source for clinical use because they are safe and easy to obtain. A peptide solution can promote neural regeneration. Previously, such a solution was stereotactically injected into the brain of rats with cerebral infarction, resulting in improvement in the animal's neurological function and reduction in the infarction volume, but the injury was relatively severe. The current study established a rat model of cerebral ischemia-reperfusion (I/R) injury. MSCs isolated from Wharton's jelly of human umbilical cords (HUMSCs) were injected intravenously immediately after cerebral I/R injury(3 × 106 cells per rat). Twenty-four h and 14 d after surgery, animal behavior was evaluated using the Rogers test and infarct lesion volume was evaluated by 2,3,5-triphenyltetrazolium chloride staining. Fourteen d after surgery, brain tissues were collected at 14 d to study migration/implantation of HUMSCs, cellular proliferation, neural regeneration and astrocyte activation. Compared to cerebral I/R injury alone, HUMSC treatment improved function at 14 d after surgery, with no reduction in infarct volume or migration or implantation of cells into the damaged brain areas. Nevertheless, 14 d after surgery, HUMSC administration increased cellular proliferation and the level of neurofilament 200 level and decreased the level of glial fibrillary acidic protein. After cerebral I/R injury, acute intravenous administration of HUMSCs could promote recovery by activating endogenous neural regeneration and inhibiting astrocyte activation, without migration and implantation directly into lesions.
Keywords: Cerebral ischemia-reperfusion, human umbilical cord mesenchymal stem cell, cellular proliferation, neural regeneration, astrocyte activation, neurofilament protein 200, glial fibrillary acidic protein
1. Introduction
Stroke is a major cause of death and disability in adults worldwide. Despite decades of intense research, current treatments for acute ischemia stroke are far from optimal (1). Ischemia stroke triggers a cascade of pathophysiological events, including excitotoxicity, oxidative stress, apoptosis and inflammation (2). In addition, neural regeneration, synaptogenesis, astrocyte activation and angiogenesis are stimulated within the brain post-stroke (3,4); however, neural regeneration is markedly reduced in the aged rodent brain (5). In recent years, experimental studies and clinical trials have indicated that cell-based therapiesoffer promise as a stroke treatment.
One of the main challenges is to identify the best source of stem cells for use after stroke. Transplantation of stem cells from fetal tissues and progenitor cells from bone marrow and human umbilical cord blood has promoted neuronal survival, tissue repair, and, most importantly, recovery after experimental stroke (6). However, limited sources of stem and progenitor cells as well as ethical and safety concerns associated with the use of embryonic stem cells have spurred a search for alternative stem cell sources for stroke treatment. Mesenchymal stem cells (MSCs) have emerged as a leading candidate source for stroke management; MSCs are easily obtained from a range of tissues (7), can be rapidly expanded in vitro while maintaining their potential to differentiate into multiple tissues, and are immune-privileged, with reduced risk of rejection (8). MSCs isolated from Wharton's jelly of human umbilical cords (HUMSCs) can escape immune surveillance possibly because of the absence of the major histocompatibility complex II antigen and co-stimulatory surface antigens CD40, CD80, and CD86 (9,10).
Originally, thinking was that stem cells had to be placed within the damaged sites of the brain after ischemia to promote recovery. In many studies, different routes have been tested, including intrastriatal (11), intracerebral (12), intraventricular (13), and intrathecal by lumbar puncture (14). However, these techniques raise the possibility of additional trauma from transplantation surgery and are often impractical for patients with clinically serious conditions. Recent studies have demonstrated that cell-based therapy is not intended to replace damaged cells but rather to remodel the central nervous system by promoting neuroplasticity, angiogenesis, and immunomodulation (15,16).
The optimal timing for the administration of stem cells to treat cerebral ischemia is another main challenge. Because of the time needed to prepare autologous stem cells after an unexpected stroke, such cells can only be administered several weeks later, and previous studies have focused on post-acute-phase intervention (16,17). Promising experimental animal data suggest that early stem cell administration can interrupt the initiation of the very beginning of the ischemic cascade (15). Despite several reports of the treatment of experimental cerebral ischemia with bone-marrow-derived MSCs (BM-MSCs) and adipose tissue-derived MSCs (AD-MSCs) in the acute phase (18,19), there are no reports of the acute intravenous administration of HUMSCs to treat cerebral ischemia.
Here, an established rat model of middle cerebral artery occlusion (MCAO) was used to investigate the potential therapeutic effects of acute intravenous administration of HUMSCs on cerebral ischemia-reperfusion (I/R) injury.
2. Materials and Methods
2.1. Animals
A randomized, controlled animal experiment was performed at the Experimental Animal Center and Neurobiology Laboratory of Taishan Medical College, China from September 2012 to June 2013. In total, 54 Sprague Dawley pathogen-free rats of both sexes weighing 250–300 g and 12–14 weeks old were provided by the Experimental Animal Center of Shandong Provincial University of Traditional Chinese Medicine (License No. SCXK [Lu] 2005–0015). Rats were housed at 24 ± 3°C in 40% to 70% relative humidity under a 12-h light-dark illumination cycle and had free access to food and water. Experimental protocols were in accordance with Guidance Suggestions for the Care and Use of Laboratory Animals, formulated by the Ministry of Science and Technology of the People's Republic of China.
2.2. Human umbilical cords
Human umbilical cords were obtained from full-term deliveries with informed consent from parents after caesarian section. The procedure for collecting tissues was approved by the ethics committee of the Second Hospital of Shandong University.
2.3. HUMSC isolation and in vitro culture
Isolation and culture of HUMSCs were as described. After arteries and veins were removed, the remaining tissue, Wharton's jelly, was cut into 0.5–1 cm3 pieces and suspended in Dulbecco's modified Eagle's low-glucose media (DMEM-LG; Gibco, USA) containing 10% fetal bovine serum (FBS; Gibco, USA), 100 mg/mL penicillin, and 100 mg/mL streptomycin. The tissue was left undisturbed for 7 days in a 37°C humidified incubator with 5% CO2 to allow cells to migrate from the explants. Culture medium was replaced every 3 days. The cells were passaged through use of a 0.25% trypsin-EDTA solution when cells reached 80% to 90% confluence. The identification of HUMSCs by flow cytometry was as previously described. The cells used in this study were positive for CD73, CD90, and CD105 but negative for CD34, CD45, and HLA-DR, which is consistent with the characteristics of HUMSCs.
2.4. Animal groups
The rats were randomly assigned to 3 groups for treatment (n = 18 animals each): sham-surgery without cerebral I/R injury and infusion of phosphate-buffered saline (PBS) via the tail vein; cerebral I/R injury induced by MCAO and reperfusion and infusion of PBS via the tail vein; and MCAO and reperfusion and infusion of HUMSC via the tail vein.
2.5. Preparation of a model of cerebral I/Rinjury
As previously described, rats were anesthetized with an intraperitoneal injection of 10% (w/v) chloral hydrate (350 mg/kg; Boster, Wuhan, China). A 2-cm incision was made in the middle of the neck line, separating the right carotid artery, as well as the internal and external carotid communicating arteries. Through a small incision to the external carotid artery, a previously selected plug thread was inserted from the right external carotid artery into the right internal carotid artery to a depth of 18.0 ± 0.5 mm (beginning with the common carotid artery bifurcation); the thread was maintained for 2 h and then removed to restore blood flow to the common carotid and internal carotid arteries. Body temperature was maintained at 37 ± 0.5°C with a heating lamp linked to a rectal thermometer during all phases of the surgery. Under anesthesia, rats in the sham surgery group only had the common carotid artery, internal carotid artery, and external carotid artery exposed.
2.6. Cell administration
A HUMSC suspension or PBS was injected via the tail vein at the onset of reperfusion. Before the injection, the cell suspension was washed with PBS 3 times to remove serum. Intravenous injections of 3 × 106 HUMSCs in 600 µL saline were administered via the tail vein over 4 min. Cerebral I/R injury was induced in animals with MCAO and reperfusion, but animals only received an equal volume of PBS at that time. Cerebral I/R injury was not induced in sham-operated animals, and animals received an injection of PBS via the tail vein. Sham-operated animals and animals with cerebral I/R injury both received an equal volume of PBS without HUMSCs over 4 min.
2.7. BrdU incorporation
All animals were given 50 mg/kg of daily intraperitoneal 5-bromo-2′-deoxyuridine (BrdU, Sigma-Aldrich) on days 4 to 7 after cerebral I/R injury . This administration protocol was based on previous reports of proliferation peaking 4 to 10 days after injury.
2.8. Functional evaluation
All animals were functionally evaluated 24 h and 14 d after surgery usingthe Rogers scale: 0, no deficit; 1, failure to extend left forepaw; 2, decreased grip of the left forelimb when the tail is pulled; 3, spontaneous movement in all directions, contralateral circling if pulled; 4, circling or walking to the left; 5, movement only when stimulated; 6, unresponsive to stimulation; and 7, dead.
2.9. Measurement of cerebral infarct volume
Twenty-four h and 14 d after surgery, cerebral infarct volume was measured in 6 animals from each group. Rats were decapitated, their brains were rapidly removed and cut into 6 2-mm-thick coronal sections, and sections were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC) solution at 37°C for 30 min. Stained slides were washed in PBS for 5 min and immersed overnight in 4% paraformaldehyde. Unstained areas were defined as infarcted tissue, and normal tissue was stained red. The total infarct volume was the sum of the infarcted areas in the 6 sections and was a percentage of cerebral ischemic volume in the hemisphere ipsilateralto the lesion.
2.10. Histological staining
Fourteen d after surgery, 6 animals from each group were used for histological staining. Rats were deeply anesthetized with chloral hydrate, then cardiac perfused immediately with ice-cold PBS and 4% paraformaldehyde in 0.1 M PBS (pH 7.4) after functional evaluation. Brains were excised and post-fixed overnight at 4°C and then incubated in 30% sucrose at 4°C until equilibration (5 mice per group). Coronal sections 10 µm thick were prepared using of a freezing microtome (LEICA CM1850; Germany) and were stored at −20°C.
Immunohistochemistry was performed as described. Brain sections were treated with 2 M HCL for 30 min at 37.0°C. Sections were washed in PBS, blocked in PBS containing 0.4% Triton X-100 (Sigma) and 5% goat serum (Jackson ImmunoResearch Lab) for 1 h, and then incubated with the primary antibody at 4°C: rabbit monoclonal anti-HuN (Chemicon, 1:300), overnight at 4°C to analyze the migration and implantation of HUMSCs in rat brains or rabbit anti-BrdU (1:500, Sigma) to detect cellular proliferation in brains. Sections were then exposed to biotinylated goat anti-rabbit IgG (1:200) at room temperature for 10 min, followed by exposure to biotin-streptavidin-alkaline phosphatase at room temperature for 10 min.
Immunofluorescence was performed as described to detect neural regeneration and astrocyte activation. Sections underwent double staining with mouse anti-neurofilament 200 (NF200) monoclonal antibody (1:100, Abnova; a marker of neural regeneration) followed by goat anti-mouse Alexa Fluor 488 (1: 750, Molecular Probes, Invitrogen) and rabbit anti-BrdU (1:500, Sigma) and then goat anti-rabbit Alexa Fluor 594 (1:750, Molecular Probes, Invitrogen). Sections were stained with mouse anti-glial fibrillary acid protein (GFAP) monoclonal antibody (1:400, Chemicon; a marker of astrocyte activation) followed by goat anti-mouse Alexa Fluor 488. Finally, sections were coverslipped with medium for fluorescence (Vector Labs) and viewed usingfluorescence microscopy (Olympus, Japan).
2.11. Statistical analysis
All data are expressed as mean ± S.D. Differences were analyzed using analysis variance (ANOVA) and the Student's t-test in SPSS 16.0 for Windows (SPSS Inc., Chicago, IL, USA). p value < 0.05 was considered statistically significant.
3. Results
3.1. Intravenous administration of HUMSCs improved functional recovery after cerebral I/R injury
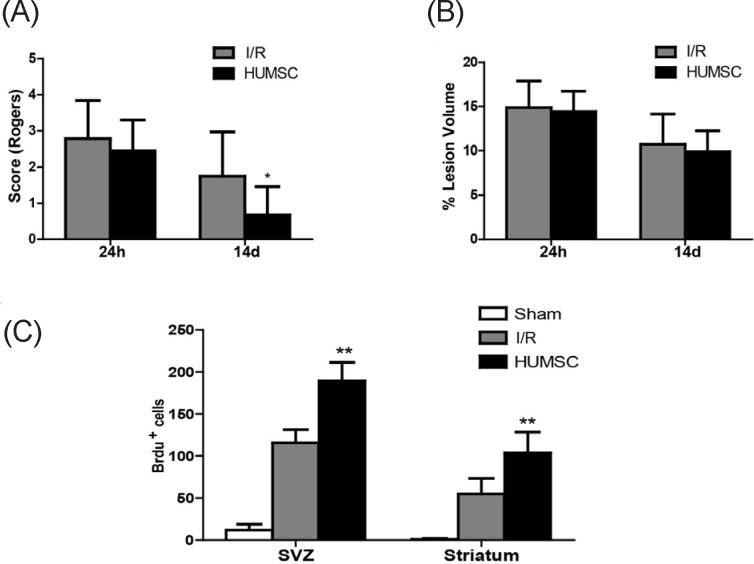
Animals were clinically evaluated 24 h and 14 d after surgery using the Rogers score. Sham-operated rats did not display a functional deficit 24 h or 14 d after surgery. Twenty-four h after surgery, the Rogers score did not differ for rats treated with HUMSCs and untreated rats with cerebral I/R injury (p = 0.307; Figure 1A), but 14 d after surgery, rats treated with HUMSCs had better functional recovery than did untrreated rats with cerebral I/R injury (p = 0.016).
Figure 1.
(A), Intravenous administration of HUMSCs improved functional recovery after cerebral I/R injury. Animals were clinically evaluated 24 h and 14 d after surgery using the Rogers score. Sham-operated rats did not display a functional deficit 24 h and 14 d after surgery. Twenty-four h after surgery, the Rogers score did not differ for rats treated with HUMSCs and untreated rats with cerebral I/R injury (p = 0.307), but 14d after surgery rats treated with HUMSCs had better functional recovery than did untreated rats with cerebral I/R injury(p = 0.016). (B), Infarct size after cerebral I/R injury did not change after intravenous HUMSC administration. TTC staining was used to evaluate infarct lesions24 h and 14 d after the experimental procedure, This staining revealed that brains from sham-operated rats had no lesions. Both rats treated with HUMSCs and untreated rats with cerebral I/R injury had fewer lesions 14d than 24h after the prodedure, and the number of lesions in the two groups did not differ at either time(p = 0.801 and p = 0.626, respectively). (C), Intravenous administration of HUMSC increased cellular proliferation after cerebral I/R injury. Cellular proliferation was detected with BrdU immunohistochemical staining of rat brain sections. Brain sections from sham-operated rats had only a few BrdU-positive cells. Fourteen d after surgery, brain sections from rats treated with HUMSCs had a greater number of BrdU-positive cells in both the subventricular zone (SVZ; p < 0.001) and striatum (p = 0.001) than did untreated rats with cerebral I/R injury.
3.2. Infarct size after cerebral I/R injury did not change after intravenous HUMSC administration
TTC staining was used to evaluate Infarct lesions 24 h and 14 d after the experimental procedure. by Staining revealed that brains from sham-operated rats had no lesions. Both rats treated with HUMSCs and untreated rats with cerebral I/R injury had fewer lesions 14 d after the procedure than they did 24 h afterwards. There were no differences in the number of lesions in the 2 groups at either time (p = 0.801 and p = 0.626, respectively, Figure 1B).
3.3. Intravenous administration of HUMSCs did not lead to migration or implantation of HUMSCs
Migration and implantation of HUMSCs was studied using immunohistochemistry with anti-human nuclear antigen (HuN) antibody. Staining was not evident in brain sections from sham-operated rats and untreated rats with cerebral I/R injury. However, brain sections from rats treated with HUMSCs displayed neither migration nor implantation at 14 d after treatment (data not shown).
3.4. Intravenous administration of HUMSC increased cellular proliferation after cerebral I/R injury
Cellular proliferation was detected with BrdU immunohistochemical staining of rat brain sections. Brain sections from sham-operated rats had only a few BrdU-positive cells (Figure 1C). Brain sections from rats treated with HUMSCs had a greater number of BrdU-positive cells in both the subventricular zone (SVZ; p < 0.001) and striatum (p = 0.001) than did untreated rats with cerebral I/R injury 14 d after the procedure.
3.5. Intravenous administration of HUMSCs increased endogenous neural regeneration after cerebral I/R injury
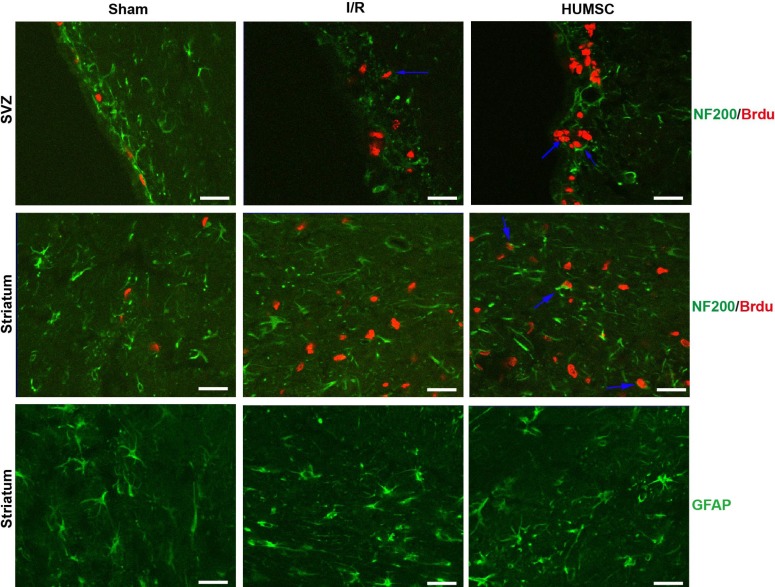
Since there was no migration and implantation of HUMSCs in rats treated with HUMSCs, endogenous neural regeneration was studied in brain sections 14 d after the procedure using immunofluorescence double staining with neurofilament 200 (NF200) and BrdU (Figure 2). Cells double-stained with NF200 and BrdU were not evident in brain sections from sham-operated rats. Cell double-stained with NF200 and BrdU were evident in the SVZ but not in the striatum of brain sections from untreated rats with cerebral I/R injury. As expected, cells double-stained with NF200 and BrdU were evident in both the SVZ and striatum of brains from rats treated with HUMSCs.
Figure 2.
Intravenous administration of HUMSCs increased endogenous neural regeneration after cerebral I/R injury. Since there was no migration and implantation of HUMSCs in rats treated with HUMSCs, endogenous neural regeneration was studied using immunofluorescence double staining with neurofilament 200 (NF200) and BrdU in brain sections 14 d after surgery. Cells double-stained with NF200 and BrdU were not evident in brains sections from sham-operated rats. Cells double-stained with NF200 and BrdU were evident in the SVZ but not in the striatum of brain sections from untreated rats with cerebral I/R injury. As expected, neuronal cells double-stained with NF200 and BrdU were evident in both the SVZ and striatum of brains from rats treated with HUMSCs. Intravenous administration of HUMSCs attenuated astrocyte activation after cerebral I/R injury. Glial fibrillary acidic protein (GFAP) was used as an immunofluorescence marker of astrocyte activation in the striatum of rats. Untreated rats with cerebral I/R injury had a greater number of GFAP-positive cells than did sham-operated rats and rats treated with HUMSCs had fewer GFAP-positive cells than did untreated rats with cerebral I/R injury.
3.6. Intravenous administration of HUMSCs attenuated astrocyte activation after cerebral I/R injury
GFAP was used as an immunofluorescence marker of astrocyte activation in the striatum of rats (Figure 2). Rats with cerebral I/R injury had a greater number of GFAP-positive cells than did sham-operated rats and rats treated with HUMSCs had fewer GFAP-positive cells than did untreated rats with cerebral I/R injury.
4. Discussion
This study investigated the effects of acute intravenous administration of HUMSCs in rats 24 h and 14 d after cerebral I/R injury. The treatment significantly improved functional recovery but did not reduce the infarct volume or cause migration or implantation of HUMSCs in damaged brain areas. Animals receiving HUMSCs had increased cell proliferation, a higher level of NF200 (a marker of neural regeneration), as well as a reduced level of GFAP (a marker of astrocyte activation) in the brain 14 d after cerebral I/R injury. A point worth noting is that acute intravenous administration of HUMSCs might have clinical implications for translation to human trials.
In a recent study of permanent MCAO in rats, the authors reported efficient functional recovery with intravenous administration of AD-MSCs and BM-MSCs (18). In the current study, the functional scores 14 d after cerebral I/R injury improved significantly with intravenous administration of HUMSCs. Twenty-four h and 14 d after cerebral I/R injury, there were no differences in TTC staining in rats treated with HUMSCs and untreated rats with cerebral I/R injury. HUMSCs administered intravenously did not migrate to or implant in the brain according to immunohistochemistry. Therefore, stem cells might not need to migrate and graft onto the lesion site for a positive functional result because of the paracrine effects of MSCs (20). This favorable outcome could be a consequence of the secretion of several growth factors that may act to enhance endogenous repair mechanisms normally activated in the brain after stroke. Such repair mechanisms include endogenous neural regeneration, synaptogenesis, astrocyte inactivation and immunomodulation (21,22).
Compared to cerebral I/R injury alone, intravenous administration of HUMSCs increased cellular proliferation in the SVZ and striatum of brains 14 d after cerebral I/R injury. The current results agree with those of previous studies reporting increases in cellular proliferation after AD-MSC and BM-MSC administration (18,23). In line with earlier studies, a later study found that, compared to infarction alone, administration of MSCs promoted cellular proliferation in SVZ and cell differentiation into neuroblasts in peri-infarct areas (24).
After a stroke, neuroblasts generated in the SVZ migrate to the ischemic boundary zone (IBZ) and actively interact with the microenvironment to reach the ischemic striatum individually or in chains. After migration, SVZ-derived neuroblasts differentiate into mature neurons in the IBZ (3). Levels of NF expression increase 7 d after focal cerebral ischemia in the IBZ (25). A small number of studies have documented increased neural regeneration in the SVZ or IBZ with transplantation of non-neuronal cells in rodents after stroke (26). The present study, noted significantly increased levels of NF200 in the SVZ after cerebral I/R injury and in both the SVZ and striatum following HUMSC administration 14 d after surgery, so neural regeneration after ischemia was enhanced by intravenous HUMSC administration.
Brain tissue repair is a dynamic ongoing process involving reactive astrocytes, microglia, and oligodendrocytes after stroke injury. In the early stage of cerebral ischemic injury, activated astrocytes have a protective effect on brain tissue by inactivating glutamate-mediated excitotoxicity and oxidative stress. However, astrocytes are then involved in hindering axonal regeneration because of glial scar formation. GFAP is commonly used as a marker of astrogliosis and astrocyte activation in several conditions involving cerebral ischemia (27). In the current study, significantly increased levels of GFAP were noted in the striatum after cerebral I/R injury, and these levels then decreased with intravenous administration of HUMSCs. This finding agrees with the results of previous studies using other types of MSCs (16,18).
Inthe current study, acute intravenous administration of HUMSCs following cerebral I/R injury promoted recovery with neural regeneration and astrocyte inactivation and no migration or implantation of cells into lesions. Future studies are needed to determine the cell fate after intravenous administration of HUMSCs, the mechanism of action for HUMSCs, and the optimal dose to avoid undesired adverse reactions.
Acknowledgements
The manuscript is original, has not been submitted to or is not under consideration by another publication, has not been previously published in any language or any form, including electronic, and contains no disclosure of confidential information or authorship/patent application/funding source disputations
References
- 1. Kunz A, Dirnagl U, Mergenthaler P. Acute pathophysiological processes after ischaemic and traumatic brain injury. Best Pract Res Clin Anaesthesiol. 2010; 24:495-509. [DOI] [PubMed] [Google Scholar]
- 2. Jauch EC, Saver JL, Adams HP, Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013; 44:870-947. [DOI] [PubMed] [Google Scholar]
- 3. Xiong Y, Mahmood A, Chopp M. Angiogenesis, neurogenesis and brain recovery of function following injury. Curr Opin Investig Drugs. 2010; 11:298-308. [PMC free article] [PubMed] [Google Scholar]
- 4. Namura S, Ooboshi H, Liu J, Yenari MA. Neuroprotection after cerebral ischemia. Ann N Y Acad Sci. 2013; 1278:25-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Jin K, Minami M, Xie L, Sun Y, Mao XO, Wang Y, Simon RP, Greenberg DA. Ischemia-induced neurogenesis is preserved but reduced in the aged rodent brain. Aging Cell. 2004; 3:373-377. [DOI] [PubMed] [Google Scholar]
- 6. Zhang J, Chopp M. Cell-based therapy for ischemic stroke. Expert Opin Biol Ther. 2013; 13:1229-1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physio. 2007; 213:341-347. [DOI] [PubMed] [Google Scholar]
- 8. Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, Granton J, Stewart DJ, Canadian Critical Care Trials Group. Safety of cell therapy with mesenchymal stromal cells (Safe Cell): A systematic review and meta-analysis of clinical trials. PLoS One. 2012; 7:e47559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Weiss ML, Anderson C, Medicetty S, Seshareddy KB, Weiss RJ, VanderWerff I, Troyer D, McIntosh KR. Immune properties of human umbilical cord Wharton's jelly-derived cells. Stem Cells. 2008; 26:2865-2874. [DOI] [PubMed] [Google Scholar]
- 10. Bongso A, Fong CY. The therapeutic potential, challenges and future clinical directions of stem cells from the Wharton's jelly of the human umbilical cord. Stem Cell Rev. 2013; 9:226-240. [DOI] [PubMed] [Google Scholar]
- 11. Denham M, Parish CL, Leaw B, Wright J, Reid CA, Petrou S, Dottori M, Thompson LH. Neurons derived from human embryonic stem cells extend long-distance axonal projections through growth along host white matter tracts after intra-cerebral transplantation. Front Cell Neurosci. 2012; 6:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Fan HJ, Yu WD, Wang ZL, Wang Q, He ZY. Ultra-early treatment of bone marrow-derived mesenchymal stem cells for focal cerebral ischemia/reperfusion injury. Neural Regeneration Res. 2010; 5:1296-1302. [Google Scholar]
- 13. Jin K, Sun Y, Xie L, Mao XO, Childs J, Peel A, Logvinova A, Banwait S, Greenberg DA. Comparison of ischemia-directed migration of neural precursor cells after intrastriatal, intraventricular, or intravenous transplantation in the rat. Neurobiol Dis. 2005; 18:366-374. [DOI] [PubMed] [Google Scholar]
- 14. Lim JY, Jeong CH, Jun JA, Kim SM, Ryu CH, Hou Y, Oh W, Chang JW, Jeun SS. Therapeutic effects of human umbilical cord blood-derived mesenchymal stem cells after intrathecal administration by lumbar puncture in a rat model of cerebral ischemia. Stem Cell Res Ther. 2011; 2:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Gutiérrez-Fernández M, Fuentes B, Rodríguez-Frutos B, Ramos-Cejudo J, Vallejo-Cremades MT, Díez-Tejedor E. Trophic factors and cell therapy to stimulate brain repair after ischaemic stroke. J Cell Mol Med. 2012; 16:2280-2290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. McGuckin CP, Jurga M, Miller AM, et al. Ischemic brain injury: A consortium analysis of key factors involved in mesenchymal stem cell-mediated inflammatory reduction. Arch Biochem Biophys. 2013; 534:88-97. [DOI] [PubMed] [Google Scholar]
- 17. van Velthoven CT, Kavelaars A, van Bel F, Heijnen CJ. Regeneration of the ischemic brain by engineered stem cells: Fuelling endogenous repair processes. Brain Res Rev. 2009; 61:1-13. [DOI] [PubMed] [Google Scholar]
- 18. Gutiérrez-Fernández M, Rodríguez-Frutos B, Ramos-Cejudo J, Teresa Vallejo-Cremades M, Fuentes B, Cerdán S, Díez-Tejedor E. Effects of intravenous administration of allogenic bone marrow- and adipose tissue-derived mesenchymal stem cells on functional recovery and brain repair markers in experimental ischemic stroke. Stem Cell Res Ther. 2013; 4:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Gutiérrez-Fernández M, Rodríguez-Frutos B, Alvarez-Grech J, Vallejo-Cremades MT, Expósito-Alcaide M, Merino J, Roda JM, Díez-Tejedor E. Functional recovery after hematic administration of allogenic mesenchymal stem cells in acute ischemic stroke in rats. Neuroscience. 2011; 175:394-405. [DOI] [PubMed] [Google Scholar]
- 20. Bunnell BA, Betancourt AM, Sullivan DE. New concepts on the immune modulation mediated by mesenchymal stem cells. Stem Cell Res Ther. 2010; 1:34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Li Y, Chopp M. Marrow stromal cell transplantation in stroke and traumatic brain injury. Neurosci Lett. 2009; 456:120-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Liew A, O'Brien T. Therapeutic potential for mesenchymal stem cell transplantation in critical limb ischemia. Stem Cell Res Ther. 2012; 3:28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Leu S, Lin YC, Yuen CM, Yen CH, Kao YH, Sun CK, Yip HK. Adipose-derived mesenchymal stem cells markedly attenuate brain infarct size and improve neurological function in rats. J Transl Med. 2010; 8:63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Yoo SW, Kim SS, Lee SY, Lee HS, Kim HS, Lee YD, Suh-Kim H. Mesenchymal stem cells promote proliferation of endogenous neural stem cells and survival of newborn cells in a rat stroke model. Exp Mol Med. 2008; 40:387-397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Schroeder E, Vogelgesang S, Popa-Wagner A, Kessler C. Neurofilament expression in the rat brain after cerebral infarction: Effect of age. Neurobiol Aging. 2003; 24:135-145. [DOI] [PubMed] [Google Scholar]
- 26. Jin K, Xie L, Mao X, Greenberg MB, Moore A, Peng B, Greenberg RB, Greenberg DA. Effect of human neural precursor cell transplantation on endogenous neurogenesis after focal cerebral ischemia in the rat. Brain Res. 2011; 1374:56-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ricci G, Volpi L, Pasquali L, Petrozzi L, Siciliano G. Astrocyte-neuron interactions in neurological disorders. J Biol Phys. 2009; 35:317-336. [DOI] [PMC free article] [PubMed] [Google Scholar]