Abstract
One of the tuberous sclerosis complex (TSC) gene products, tuberin is assumed to be the functional component, being involved in a wide variety of cellular processes. Here, we report for the first time that tuberin dysfunction may represent a mechanism for neuronal damage in Alzheimer’s disease (AD), Parkinson’s disease with dementia (PD/DLB), and a mouse model of PD. Tuberin was hyperphosphorylated at Thr1462 in postmortem frontal cortex tissue of both AD and PD/DLB patients and in mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride (MPTP). Both PTEN and Akt phosphoactivation corresponded to the hyperphosphorylation patterns of tuberin suggesting that the PTEN–Akt pathway might be the mechanism of tuberin phosphorylation. Our data provide new information regarding the possible role of tuberin dysfunction in major neurodegenerative disorders, such as AD and PD, whereby inhibition of tuberin function may trigger an onset of neuronal cell death.
Keywords: Tuberous sclerosis complex, Parkinson’s disease, Alzheimer’s disease, Tuberin, Akt, Neurodegeneration
Introduction
Tuberous sclerosis complex (TSC) is an autosomal dominant tumor syndrome that affects approximately 1 in 6,000 individuals. TSC is a genetic disease, but more than half of affected individuals have spontaneous rather than inherited mutations. The disease is associated with mutations in either TSC1 or TSC2, the genes that encode the proteins hamartin and tuberin. TSC is characterized by the development of tumors, named hamartomas, in the kidneys, heart, skin, and brain. Hallmarks of this disease are seizures, mental retardation, and a variety of developmental disorders, including autism [1].
In the brain, tuberin was found to be expressed in neurons, astrocytes, and glial cells. Tuberin is abundant in the cerebral cortex of analyzed control patients, but decreased or undetectable in cerebral cortex samples of TSC patients [2]. Tuberin protein levels are upregulated upon neuronal differentiation of neuroblastoma cells. Antisense inhibition of tuberin expression inhibits this neuronal differentiation process and ectopic overexpression of TSC2 accelerates it [3]. Tuberin protein levels have been reported to be decreased in the frontal cortex of patients with Alzheimer’s disease. In addition, tuberin levels have also been shown to be decreased in Down syndrome brain samples positive for ß-amyloid plaques and neurofibrillary tangles [4].
Our main goal was to study if tuberin hyperphosphorylation pathways might be active in common neurodegenerative disorders such as AD and PD. We studied this pathway in post-mortem frontal cortex tissues obtained from AD and PD/DLB patients and from mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride (MPTP), which represents an acute neurotoxic model for PD. Here, we report for the first time that tuberin phosphorylation, which impairs tuberin function represents a potential mechanism for neuronal damage in very common neurodegenerative disorders, such as Alzheimer’s disease (AD) and Parkinson’s disease with dementia (PD/DLB).
Methods
Post-mortem frontal cortex from AD and PD/DLB subjects and age-matched controls were obtained from the Department of Pathology, University of California, San Diego. For animal studies, 10-week-old male C57BL/J6 mice (Charles River Laboratories, Wilmington, MA) (n = 10 mice/time point) were treated four times with MPTP 20 mg/kg, i.p. at 2 h intervals. Animals were sacrificed by decapitation at different time points (0, 4, 12, 24, 48, 72, 96 h and 1 week) after the last dose of MPTP. Brains were removed rapidly on ice and frontal cortex was dissected and flash frozen until further analysis. All procedures involving animals were approved by and conformed to the guidelines of the Institutional Animal Care and Use Committee of Johns Hopkins University.
A 100-mg of tissue samples from post-mortem human brain or MPTP-treated mouse brain was homogenized by polytron in 1 ml of brain lysis buffer (10 mM Tris–HCl pH 7.4, 150 mM NaCl, 5 mM EDTA, 0.5% Triton X-100 (TX), 10 mM Na-β-glycerophosphate, phosphatase inhibitor cocktail 1 and 2 (Sigma), complete EDTA-free protease inhibitor mini tablet (Roche) in deionized water) by Polytron. The lysates were incubated on ice for 30 min and mixed twice for complete lysis. The samples were then centrifuged for 20 min at 4°C at 100,000×g. The supernatant, indicated as the TX-soluble fraction, was collected. The resulting pellet was washed 3× with brain lysis buffer without TX-100. 400 μl of DNase I buffer (10 mM MgCl2, 10 mM Tris–HCl, pH 7.5) was added and the pellet was sonicated six times for 30 s at 30% intensity. 1 μl DNase I (NEB) was then added and samples were incubated at room temperature for 10 min, followed by the addition of 75 μl of 10% SDS and incubation at room temperature for 1 h. The samples were then centrifuged at 13,000 rpm for 20 min at 4°C and the supernatant was collected as the TX-insoluble fraction. Both fractions were analyzed for protein concentration using the BCA protein assay (Bio-Rad). Equal amounts of protein were mixed from both soluble and insoluble fraction and the mixture designated as total cell lysates. A total of 60 μg of protein from each sample was loaded on 4–20% SDS-PAGE Tris–HCl Criterion gradients gels (Bio-Rad) and samples were analyzed by western blot with antibodies as indicated in the figures.
Results
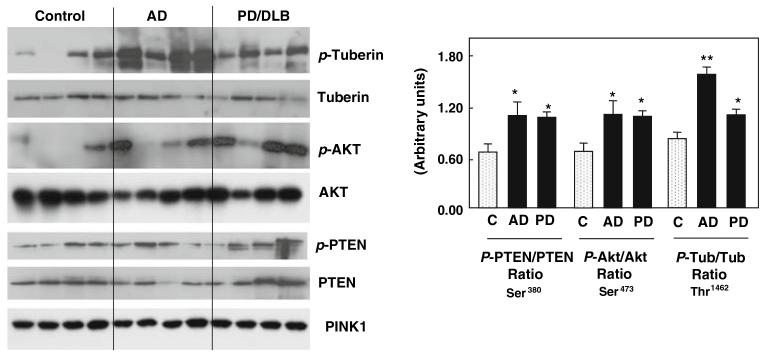
We first analyzed whole cell lysates of post-mortem frontal cortex of AD or PD/DLB patients and age-matched controls (Fig. 1). We found a significant upregulation of phosphotuberin (Thr1462) in samples of AD and PD/DLB patients (panel 1) as compared with age-matched controls. The expression level of tuberin itself was not significantly different among these samples (panel 2). We then looked for the active state of Akt, a kinase that phosphorylates tuberin, using phospho-Akt antibodies. Indeed, the phospho-Akt levels were higher in pathological samples as compared with controls, despite a fairly equal distribution of Akt protein in all the samples (panels 3 and 4). We further examined the activation pattern of PTEN kinase, an activator of Akt in this pathway. Our results indicated that PTEN was active in samples of AD and PD/DLB as compared with the controls as evidence by phospho-PTEN expression in these samples (panel 5). Surprisingly, PTEN expression was largely unchanged among these samples (panel 6). To normalize these observations to expression of a brain related protein that has been implicated as a cause of some familial forms of PD, we looked for the levels of PINK1 in our samples. PINK1 levels were unchanged and equally distributed throughout the samples (panel 7) suggesting an equal distribution of protein in all of our samples.
Fig. 1.
Tuberin was phosphorylated at threonine 1462 via the Akt–PTEN pathway in the frontal cortex of AD and PD/DLB patients. Total cell lysates were subjected to SDS-PAGE and then immunoblotted with antibodies to p-tuberin, tuberin, p-Akt, Akt, p-PTEN, and PTEN. The blots were also probed with antibodies to PINK1 a neuronal specific marker for cortex. Separate blots with 50 μg protein were run for each set such as for p-tuberin and tuberin, p-Akt and Akt, and p-PTEN and PTEN. The blots are representative of at least three different experiments. The panel on the right shows a quantitative analysis of the blots shown in the figures. Mean ± SEM and *,** denotes P < 0.05 as compared to controls and as compared to PD respectively
Second, we determined if the tuberin pathway is active in a mouse model of acute neuronal damage, which mimics some aspects of the biochemical, and neurochemical damage of PD. We performed a time-course analysis of effects of MPTP treatment of mice on tuberin phosphorylation and the related PTEN–Akt pathway (Fig. 2). There was a time-dependent increase in tuberin phosphorylation at Thr1462 in frontal cortex after MPTP treatment, which peaked at time points between 12 and 48 h and then subsequently declined (panel 1). Tuberin expression levels seem to be equal up to the time points as late as 96 h (panel 2). Both Akt and PTEN exhibited similar patterns of phosphoactivation after MPTP treatment (panels 3 and 5). Protein levels of Akt seem to be downregulated with time unlike the levels of PTEN expression, which seem to be upregulated with time after MPTP treatment (panels 4 and 6). Again, PINK1 levels were unchanged with this neuronal disease specific kinase distributed equally at all time points after MPTP treatment (panel 7).
Fig. 2.
Tuberin was phosphorylated at threonine 1462 via the Akt–PTEN pathway in the cortex of MPTP induced acute mouse model of PD. Total cell lysates were made from frontal cortex of mice (pooled from n = 4) treated with MPTP and sacrificed at various time points (0, 4, 12, 24, 48, 72, 96 h and 1 week). These lysates were subjected to SDS-PAGE and then immunoblotted with antibodies to p-tuberin, tuberin, p-Akt, Akt, p-PTEN and PTEN. The blots were also probed with antibodies to PINK1 a neuronal specific marker for cortex. Separate blots with 50 μg protein were run for each set such as for p-tuberin and tuberin, p-AKT and AKT, and p-PTEN and PTEN. The blots are representative of at least three different experiments
Discussion
In this communication, we report that tuberin is hyperphosphorylated in the post-mortem frontal cortex of AD and PD/DLB patients and in MPTP-treated mice. This hyperphosphorylation of tuberin seems to be mediated via the Akt–PTEN pathway. Our data provide new information regarding the possible role of tuberin dysfunction in major neurodegenerative disorders such as AD and PD whereby inhibition of tuberin function might trigger an onset of neuronal cell death. These findings provide rationale for new approaches to investigate the cellular mechanisms and to establish new therapeutic strategies for these neurodegenerative disorders.
Our data extend the understanding of the molecular functions of tuberin to another common disease, besides TSC. Although our report must be interpreted as preliminary data suggesting an association between tuberin and most common neurodegenerative disorders, it opens up an avenue to understand the role of this cell cycle regulatory protein in AD and PD/DLB. Tuberin is a multifunctional protein that interacts with a wide variety of different proteins [5-8]. At this time, it is purely speculative as to which of these functions could play a role in the molecular pathogenesis of AD or PD/DLB. However, our finding that hyperphosphorylation of tuberin correlates with PTEN/Akt pathway activation provides preliminary evidence of the major components of this pathway that might be of importance in order to elucidate the role of tuberin in more common neurodegeneration. Tuberin is also an inhibitor of mammalian target of rapamycin (mTOR) activity [9]. Rapamycin, an mTOR kinase inhibitor, is an approved drug that can normalize dysregulated mTOR signaling in cells that lack tuberin and is currently undergoing phase I/II clinical studies for TSC treatment [10]. Rapamycin could also be considered as a possible candidate in AD or PD/DLB, if the role of tuberin in these neurodegenerative disorders can be firmly established. Therefore, these studies open new avenues to clarify further the role of the tuberin–mTOR pathway in the pathogenesis of these neurodegenerative disorders.
Both AD and PD/DLB are multifactorial illnesses with both genetic and non-genetic causes. It will be of great interest to investigate the role of tuberin in AD associated with the ß-amyloid precursor protein gene, with presenilin 1 or 2, or with apolipoprotein E or for PD/DLB associated with α-synuclein. Therefore, future studies may reveal a broader role of tuberin in the cascade of molecular mechanisms of neurodegeneration during AD and PD/DLB and possibly may provide new avenues to develop targets for therapy to block the progression of neuronal loss.
Acknowledgments
This work was supported in part by NIH grants NS38377 and NS48206 and grants from American Diabetes Association (ADA), U.S. Dept of Veterans Affairs (VA), American Parkinson’s Disease Association (APDA), Parkinson’s Disease Foundation (PDF), San Antonio Area Foundation (SAAF), and Executive Research Council (ERC) of UTHSCSA. T.M.D. is the Leonard and Madlyn Abramson Professor in Neurodegenerative Diseases.
Contributor Information
Samy L. Habib, Department of Medicine, University of Texas, Health Science Center, San Antonio, TX, USA; South Texas Veterans Healthcare System, San Antonio, TX, USA
David Michel, Department of Medicine, University of Texas, Health Science Center, San Antonio, TX, USA.
Eliezer Masliah, Departments of Neurosciences and Pathology, University of California, San Diego, CA, USA.
Bobby Thomas, Departments of Neurology and Neurosciences, Institute for Cell Engineering, Johns Hopkins School of Medicine, Baltimore, MD, USA.
Han Seok Ko, Departments of Neurology and Neurosciences, Institute for Cell Engineering, Johns Hopkins School of Medicine, Baltimore, MD, USA.
Ted M. Dawson, Departments of Neurology and Neurosciences, Institute for Cell Engineering, Johns Hopkins School of Medicine, Baltimore, MD, USA
Hanna Abboud, Department of Medicine, University of Texas, Health Science Center, San Antonio, TX, USA.
Robert A. Clark, Department of Medicine, University of Texas, Health Science Center, San Antonio, TX, USA
Syed Z. Imam, Departments of Medicine & Pharmacology, University of Texas Health Science Center, San Antonio, TX, USA
References
- 1.Kwiatkowski DJ. Tuberous sclerosis: from tubers to mTOR. Ann Hum Genet. 2002;67:87–96. doi: 10.1046/j.1469-1809.2003.00012.x. [DOI] [PubMed] [Google Scholar]
- 2.Mizugichi M, Hino O. Neuropathology. In: Curatolo P, editor. Tuberous sclerosis complex: from basic science to clinical phenotypes. Mac Keith Press; London: 2003. [Google Scholar]
- 3.Soucek T, Holzl G, Bernaschek G, Hengstschläger M. A role of the tuberous sclerosis gene-2 product during neuronal differentiation. Oncogene. 1998;16:2197–2204. doi: 10.1038/sj.onc.1201743. [DOI] [PubMed] [Google Scholar]
- 4.Ferrando-Miguel R, Rosner M, Freilinger A, Lubec G, Hengstschläger M. Tuberin—A new molecular target in Alzheimer’s disease? Neurochem Res. 2005;30:1413–1419. doi: 10.1007/s11064-005-8511-y. [DOI] [PubMed] [Google Scholar]
- 5.Pan D, Dong J, Zhang Y, Gao X. Tuberous sclerosis complex: from Drosophila to human disease. Trends Cell Biol. 2004;14:78–85. doi: 10.1016/j.tcb.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 6.Xiao G-H, Shoarinejad F, Jin F, Golemis EA, Yeung RS. The tuberous sclerosis 2 gene product, tuberin, functions as a Rab5 GTPase activating protein (GAP) in modulating endocytosis. J Biol Chem. 1997;272:6097–6100. doi: 10.1074/jbc.272.10.6097. [DOI] [PubMed] [Google Scholar]
- 7.Henry KW, Yuan X, Koszewski NJ, Onda H, Kwiatkowski DJ, Noonan DJ. Tuberous sclerosis gene 2 product modulates transcription mediated by steroid hormone receptor family members. J Biol Chem. 1998;273:20535–20539. doi: 10.1074/jbc.273.32.20535. [DOI] [PubMed] [Google Scholar]
- 8.Soucek T, Pusch O, Wienecke R, DeClue JE, Hengstschläger M. Role of the tuberous sclerosis gene-2 product in cell cycle control. J Biol Chem. 1997;272:29301–29308. doi: 10.1074/jbc.272.46.29301. [DOI] [PubMed] [Google Scholar]
- 9.Corradetti YMN, Inoki K, Guan K-L. TSC2: filling the GAP in the mTOR signalling pathway. Trends Biochem Sci. 2003;29:32–38. doi: 10.1016/j.tibs.2003.11.007. [DOI] [PubMed] [Google Scholar]
- 10.Inoki K, Corradetti MN, Guan K-L. Dysegulation of the TSC–mTOR pathway in human disease. Nat Genet. 2004;37:19–24. doi: 10.1038/ng1494. [DOI] [PubMed] [Google Scholar]