Abstract
Q fever is a highly infectious disease with a worldwide distribution. Its causative agent, the intracellular bacterium Coxiella burnetii, infects a variety of vertebrate species, including humans. Its evolutionary origin remains almost entirely unknown and uncertainty persists regarding the identity and lifestyle of its ancestors. A few tick species were recently found to harbor maternally-inherited Coxiella-like organisms engaged in symbiotic interactions, but their relationships to the Q fever pathogen remain unclear. Here, we extensively sampled ticks, identifying new and atypical Coxiella strains from 40 of 58 examined species, and used this data to infer the evolutionary processes leading to the emergence of C. burnetii. Phylogenetic analyses of multi-locus typing and whole-genome sequencing data revealed that Coxiella-like organisms represent an ancient and monophyletic group allied to ticks. Remarkably, all known C. burnetii strains originate within this group and are the descendants of a Coxiella-like progenitor hosted by ticks. Using both colony-reared and field-collected gravid females, we further establish the presence of highly efficient maternal transmission of these Coxiella-like organisms in four examined tick species, a pattern coherent with an endosymbiotic lifestyle. Our laboratory culture assays also showed that these Coxiella-like organisms were not amenable to culture in the vertebrate cell environment, suggesting different metabolic requirements compared to C. burnetii. Altogether, this corpus of data demonstrates that C. burnetii recently evolved from an inherited symbiont of ticks which succeeded in infecting vertebrate cells, likely by the acquisition of novel virulence factors.
Author Summary
How virulent infectious diseases emerge from non-pathogenic organisms is a challenging question. Here, we address this evolutionary issue in the case of Q fever. Its causative agent, the intracellular bacterium Coxiella burnetii, is extremely infectious to humans and a variety of animals. However, uncertainty persists regarding its evolutionary origin, including the identity and lifestyle of its ancestors. In this article, we show that C. burnetii arose from a rare evolutionary transformation of a maternally-inherited endosymbiont of ticks into a specialized and virulent pathogen of vertebrates. While arthropod symbionts are typically transmitted maternally and thought not to be infectious to vertebrates, we establish here that one Coxiella symbiont has evolved the necessary adaptations to exploit the vertebrate cell, leading to the emergence of Q fever.
Introduction
‘Query fever’ (Q fever) is a highly infectious zoonotic disease first identified in 1937 [1,2,3,4,5]. The causative agent, the obligate intracellular bacterium Coxiella burnetii, infects a variety of vertebrate species, including humans. Sporadic cases in humans occur annually worldwide, but occasional outbreaks are also common [1,2,3,4]. For example, in the Netherlands more than 4,000 human cases were reported between 2007 and 2010 [6]. While most human cases are self-limiting with fever and fatigue, acute forms range from mild flu-like symptoms to pneumonia or hepatitis. The disease can also become chronic (mainly endocarditis), and, though rarely fatal, remains highly debilitating even when treated with antibiotics [1,2]. Most human cases are linked to contact with infected livestock, especially goats and sheep, which suffer abortion and reproductive disorders. Infection usually occurs by the inhalation of aerosolized resistant small cell variants that are present in the excretions of infected animals. Other modes of transmission including ingestion of unpasteurized milk or dairy products; human-to-human contact is also possible but considered rare [1,2,4,5]. One of the most virulent reference strains of C. burnetii (strain RSA 493 / Nine Mile I [7]) was isolated from a guinea pig on which field-collected Rocky Mountain wood ticks Dermacentor andersoni had fed, suggesting that transmission through tick bites may also occur [8]. The small cell variants of the bacterium can survive and remain highly infectious for long periods in the environment, leading to the classification of C. burnetii as potential bioterrorism agent [9].
The evolutionary origin of Q fever is unclear since the C. burnetii ancestor and its primary lifestyle remain entirely unknown. Historically, C. burnetii was assigned to the taxonomic order Rickettsiales (Alphaproteobacteria), but it has been recently considered more closely related to the Legionellales order (Gammaproteobacteria) because of its genetic proximity to the Legionnaires' disease agent, Legionella pneumophila [10]. The Legionellales order includes many other intracellular bacteria infecting non-vertebrate species, such as, for instance, Rickettsiella species that are both widespread and biologically diverse in arthropods [11,12,13]. Within the Coxiella genus, the only known relative of C. burnetii which has been formally identified is C. cheraxi, a pathogen of crayfishes [14]. Many past descriptions of Coxiella were likely biased toward the detection of pathogenic strains since most C. burnetii isolates were collected from humans or domestic ruminants during Q fever outbreaks [1,5,15]. However, the advent of 16S rRNA gene sequencing as a universal DNA barcoding marker in bacteria has led to the description of a few novel Coxiella-like organisms in non-vertebrate species (listed in [16]), and particularly in ticks [17,18,19,20,21,22,23,24,25]. All these Coxiella-like organisms are closely related, but genetically distinct to C. burnetii, suggesting that some diversity exists within the Coxiella genus. The highly conserved nature of the 16S rRNA gene sequences has prevented researchers from establishing the exact relationship between C. burnetii and Coxiella-like organisms, and a sister clade relationship is commonly assumed [16,18,21,22,23].
The Coxiella-like organisms differ from C. burnetii in their biological traits and some may behave as subtle symbionts engaged in intricate interactions with ticks. In ticks belonging to Ornithodoros, Amblyomma and Rhipicephalus genera, Coxiella-like organisms were found to massively infect ovaries and to be maternally inherited through the egg cytoplasm [18,20,21,26]. In these tick species, the presence of the bacteria in the Malpighian tubules further suggests a possible role in nutrition by potentially provisioning their hosts with essential nutriments [20,21,26]. Indeed, the elimination of these bacteria with an antibiotic treatment was shown to negatively impact the fitness of the lone star tick A. americanum [27]. Accordingly, when the Coxiella-like bacterium found in A. americanum was recently sequenced [16], no recognizable virulence genes were found, indicating that this bacterium is likely non-pathogenic. In contrast, its genome encodes major vitamin and cofactor biosynthesis pathways, suggesting that it may be a vitamin-provisioning endosymbiont. This interaction exhibits the typical hallmarks of maternally-inherited symbionts with essential roles in arthropod biology [28,29]. Such patterns have been found in other exclusive blood-feeding species like bedbugs [30] and tsetse flies [31], two insect groups which rely on a single food source throughout their developmental cycle and harbor beneficial microbes that provide nutrients absent from their restricted diets. The Coxiella-like organisms of ticks share obvious similarities with these beneficial endosymbionts.
Here, we examine the origin of the Q fever pathogen, C. burnetii, by inferring the evolutionary processes that have shaped diversity within the entire Coxiella genus. To this aim, we first sampled an extensive range of ticks, with 58 tick species examined, and developed a sensitive detection method that reveals a wider Coxiella diversity than recognized in past studies. Second, instead of relying solely on the 16S rRNA gene, a molecular marker that is notoriously inadequate for inferring reliable fine-scale phylogenies [32], we used a novel multilocus typing method, allied to Whole Genome Sequencing (WGS) data, and conducted phylogenetic analyses on a large amount of DNA sequence data. Third, we examined two major ecological features of Coxiella-like organisms, i.e. their ability to be maternally-inherited through the tick egg cytoplasm and to grow in a vertebrate cell environment suitable to C. burnetii. Altogether, this corpus of data has led to the characterization of a large genetic and ecological diversity within the Coxiella genus, far beyond the C. burnetii type species. The Coxiella-like organisms of ticks form an ancient lineage of maternally-inherited tick endosymbionts that do not lie as a sister-clade to C. burnetii but rather form a basal lineage illustrative of the ancestral Coxiella life style.
Results
Ticks commonly harbor Coxiella-like organisms
We performed an extensive screening for the presence of Coxiella in 916 tick specimens from 58 species belonging to the two main tick families, Ixodidae (hard ticks, 36 species) and Argasidae (soft ticks, 22 species) (Fig 1 and Table 1). Except for 37 specimens (6 species) derived from laboratory colonies, all other tick specimens were sampled from natural populations in Europe, Americas, Africa, Oceania and Asia (n = 112 localities). In these populations, ticks were collected either in the host habitat or directly on hosts (Table 1). To detect C. burnetii and its relatives, we developed a detection method based on a nested polymerase chain reaction (PCR) using total tick DNA extracts to amplify a 539–542 base-pair (bp) fragment of the Coxiella rpoB gene (Table A in S1 Text).
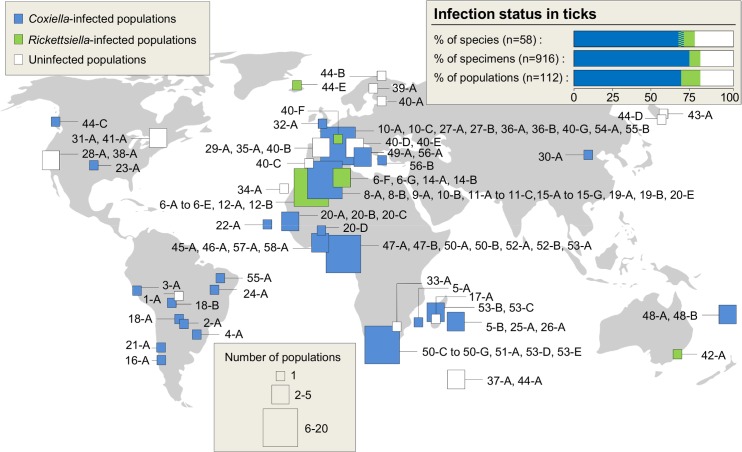
Fig 1. Geographic origin of the sampled ticks and distribution of Coxiella and Rickettsiella infections.
Square size indicates the number of populations sampled per geographic area. Numbers refer to the tick species whereas letters discriminate the different populations screened within a species; this nomenclature is detailed in Table 1. The colors within squares indicate the infection status of populations. Two tick species were infected by both Coxiella and Rickettsiella at the species level, but not at individual and population levels.
Table 1. List of tick species and populations included in the analysis, with details on their origin, the population sample size, and the prevalence of Coxiella spp. and Rickettsiella spp.
| Tick species | Sample sites | Tick host species or habitat | n | Coxiella prevalence (number of infected tick specimens) | Rickettsiella prevalence (number of infected tick specimens) | |
|---|---|---|---|---|---|---|
| Argasidae (soft ticks) | ||||||
| 1 - | Antricola guglielmonei Estrada-Peña, Barros-Battesti and Venzal, 2004 | 1-A, Porto Velho, Rondonia, Brazil | Bat guano in cave | 4 | 0.00 (0) | 0.00 (0) |
| 2 - | Argas monachus Keirans, Radovsky and Clifford, 1973 | 2-A, Chaco, Argentina | Unknown | 3 | 1.00 (3) | 0.00 (0) |
| 3 - | Ornithodoros amblus Chamberlin, 1920 | 3-A, Lobos de Tierra Island, Peru, 2009 | Peruvian Pelican (Pelecanus thagus), Peruvian Booby (Sula variegata) | 5 | 1.00 (5) | 0.00 (0) |
| 4 - | Ornithodoros brasiliensis Aragão, 1923 | 4-A, Sao Francisco de Paula,Brazil | Unknown | 3 | 1.00 (3) | 0.00 (0) |
| 5 - | Ornithodoros capensis Neumann, 1901 | 5-A, Juan de Nova Island, Mozambic Channel, 2011 | Sooty Tern (Onychoprion fuscatus) | 28 | 1.00 (28) | 0.00 (0) |
| 5-B, Réunion Island | Sea bird nests | 3 | 1.00 (3) | 0.00 (0) | ||
| 6 - | Ornithodoros costalis Diatta, Bouattour, Durand, Renaud and Trape, 2013 | 6-A, Kenitra, Morocco, 2006 | Rodent burrows | 7 | 0.00 (0) | 1.00 (7) |
| 6-B, Sidi Akhfennir, Morocco, 2006 | Rodent burrows | 18 | 0.00 (0) | 1.00 (18) | ||
| 6-C, Boujdour, Morocco, 2006 | Rodent burrows | 2 | 0.00 (0) | 1.00 (2) | ||
| 6-D, El Argoub, Morocco, 2006 | Rodent burrows | 2 | 0.00 (0) | 1.00 (2) | ||
| 6-E, Lahmiris, Morocco, 2010 | Rodent burrows | 1 | 0.00 (0) | 1.00 (1) | ||
| 6-F, Mostaganem, Algeria, 2012 | Rodent burrows | 4 | 0.00 (0) | 1.00 (4) | ||
| 6-G, Oudhna, Tunisia, 2010 | Rodent burrows | 4 | 0.00 (0) | 1.00 (4) | ||
| 7 - | Ornithodoros denmarki Kohls, Sonenshine and Clifford, 1965 | 7-H, unknown | Sea bird nests | 1 | 1.00 (1) | 0.00 (0) |
| 8 - | Ornithodoros erraticus Lucas, 1849 | 8-A, La Calle, Tunisia, 2009 | Rodent burrows | 1 | 1.00 (1) | 0.00 (0) |
| 8-B, Taher, Algeria, 2010 | Rodent burrows | 2 | 1.00 (2) | 0.00 (0) | ||
| 9 - | Ornithodoros kairouanensis Trape, Diatta, Bouattour, Durand and Renaud, 2013 | 9-A, Kairouan, Tunisia, 2010 | Rodent burrows | 3 | 1.00 (3) | 0.00 (0) |
| 10 - | Ornithodoros maritimus Vermeil and Marguet, 1967 | 10-A, Medes Island, Spain, 2009 | Yellow-legged Gull (Larus michahellis) | 20 | 1.00 (20) | 0.00 (0) |
| 10-B, Zembra Island, Tunisia, 2009 | Yellow-legged Gull (Larus michahellis) | 20 | 1.00 (20) | 0.00 (0) | ||
| 10-C, Carteau, France, 2014 | Yellow-legged Gull (Larus michahellis) | 8 | 1.00 (8) | 0.00 (0) | ||
| 11 - | Ornithodoros marocanus Velu, 1919 | 11-A, Izemmourèn, Morocco, 2009 | Rodent burrows | 1 | 1.00 (1) | 0.00 (0) |
| 11-B, Berkane Oued Kiss, Morocco, 2006 | Rodent burrows | 1 | 1.00 (1) | 0.00 (0) | ||
| 11-C, Bir-Jdid, Morocco, 2009 | Rodent burrows | 1 | 1.00 (1) | 0.00 (0) | ||
| 12 - | Ornithodoros merionesi Trape, Diatta, Belghyti, Sarih, Durand and Renaud, 2013 | 12-A, Guelmin, Morocco, 2006 | Rodent burrows | 1 | 0.00 (0) | 1.00 (1) |
| 12-B, Sidi Akhfennir, Morocco, 2006 | Rodent burrows | 2 | 0.00 (0) | 1.00 (2) | ||
| 13 - | Ornithodoros moubata Murray, 1877 | 13-A, Laboratory strain derived from field specimens of unknown origin | Unknown | 3 | 0.00 (0) | 0.00 (0) |
| 14 - | Ornithodoros normandi Larrousse, 1923 | 14-A, Bizerte, Tunisia, 2010 | Rodent burrows | 1 | 0.00 (0) | 1.00 (1) |
| 14-B, Oudhna, Tunisia, 2010 | Rodent burrows | 2 | 0.00 (0) | 1.00 (2) | ||
| 15 - | Ornithodoros occidentalis Trape, Diatta, Durand and Renaud, 2013 | 15-A, Fes, Morocco, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) |
| 15-B, Kenitra, Morocco, 2006 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 15-C, Beb-Lerba, Morocco, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 15-D, Oued Choufcherk, Morocco, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 15-E, Bouira, Algeria, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 15-F, Berrouaghia, Algeria, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 15-G, Chlef, Algeria, 2010 | Rodent burrows | 4 | 1.00 (4) | 0.00 (0) | ||
| 16 - | Ornithodoros peruvianus Kohls, Clifford and Jones, 1969 | 16-A, Chile | Common vampire Bat (Desmodus rotundus) | 3 | 1.00 (3) | 0.00 (0) |
| 17 - | Ornithodoros porcinus Walton, 1962 | 17-A, Laboratory strain derived from field specimens collected in Mahitsy, Madagascar, 2008–2010 | Unknown | 3 | 0.00 (0) | 0.00 (0) |
| 18 - | Ornithodoros rostratus Aragão, 1911 | 18-A, Salta, Argentina | Environement | 18 | 1.00 (18) | 0.00 (0) |
| 18-B, Laboratory strain derived from field specimens collected in Nhecolandi, Pantanal, Brazil | Environement | 4 | 1.00 (4) | 0.00 (0) | ||
| 19 - | Ornithodoros rupestris Trape, Bitam, Renaud and Durand, 2013 | 19-A, Saïda Mt Daïa, Algeria, 2012 | Rodent burrows | 1 | 1.00 (1) | 0.00 (0) |
| 19-B, Mostaganem, Algeria, 2012 | Rodent burrows | 2 | 1.00 (2) | 0.00 (0) | ||
| 20 - | Ornithodoros sonrai Sautet and Witkowski, 1943 | 20-A, Dielmo, Senegal, 2002 | Rodent burrows | 20 | 1.00 (20) | 0.00 (0) |
| 20-B, Kanène Khar, Senegal, 2003 | Rodent burrows | 17 | 1.00 (17) | 0.00 (0) | ||
| 20-C, Richard-Toll, Senegal, 2003 | Rodent burrows | 19 | 1.00 (19) | 0.00 (0) | ||
| 20-D, Sogoli, Mali, 2007 | Rodent burrows | 10 | 1.00 (10) | 0.00 (0) | ||
| 20-E, M’Chounèche, Algeria, 2009 | Rodent burrows | 37 | 1.00 (37) | 0.00 (0) | ||
| 21 - | Ornithodoros spheniscus Hoogstraal, Wassef, Hays and Keirans, 1985 | 21-A, Pan de Azucar, Chile, 2010–2013 | Humboldt penguin (Spheniscus humboldti) | 3 | 1.00 (3) | 0.00 (0) |
| 22- | Ornithodoros sp. (capensis species complex) | 22-A, Boa Vista Island, Cape Verde, 2008 | Cape Verde Shearwater (Calonectris edwardsii) and Brown Booby (Sula leucogaster) | 16 | 1.00 (16) | 0.00 (0) |
| Ixodidae (hard ticks) | ||||||
| 23 - | Amblyomma americanum (Linnaeus, 1758) | 23-A, Laboratory strain derived from field engorged females collected in Oklahoma, USA, 1976–2004 | Unknown | 20 | 1.00 (20) | 0.00 (0) |
| 24 - | Amblyomma cajennense (Fabricius, 1787) | 24-A, Chapada Gaucha, Brazil, 2013 | Unknown | 3 | 1.00 (3) | 0.00 (0) |
| 25 - | Amblyomma loculosum Neumann, 1907 | 25-A, Petite Ile, La Réunion, 2012 | Wedge-Tailed Shearwater (Puffinus pacificus) nests | 3 | 1.00 (3) | 0.00 (0) |
| 26 - | Amblyomma variegatum (Fabricius, 1794) | 26-A, La Réunion | Unknown | 2 | 1.00 (2) | 0.00 (0) |
| 27 - | Dermacentor marginatus (Sulzer, 1776) | 27-A, Cavaillon, France, 2011 | Vegetation | 1 | 1.00 (1) | 0.00 (0) |
| 27-B, Les Plantiers, France, 2013 | Vegetation | 1 | 1.00 (1) | 0.00 (0) | ||
| 28 - | Dermacentor occidentalis Marx, 1892 | 28-A, Hopland, Mendocino Co, California, 1985 | Environement | 6 | 0.00 (0) | 0.00 (0) |
| 29 - | Dermacentor reticulatus (Fabricius, 1794) | 29-A, Proveysieux, France, 2011 | Vegetation | 1 | 0.00 (0) | 0.00 (0) |
| 30 - | Dermacentor silvarum Olenev, 1931 | 30-A, Laboratory strain derived from field specimens collected in Xiaowutai National Natural Reserve Area, China | 2 | 1.00 (2) | 0.00 (0) | |
| 31 - | Dermacentor variabilis (Say, 1821) | 31-A, Queen's biology station, Ontario, Canada, 2013 | Vegetation | 2 | 0.00 (0) | 0.00 (0) |
| 32 - | Haemaphysalis punctata Canestrini and Fanzago, 1878 | 32-A, East Sussex, England, 2011 | Vegetation | 5 | 1.00 (5) | 0.00 (0) |
| 33 - | Hyalomma impeltatum Schulze and Schlottke, 1930 | 33-A, Zimbabwe, 1998 | Zebu (Bos indicus) | 3 | 0.00 (0) | 0.00 (0) |
| 34 - | Hyalomma lusitanicum Koch, 1844 | 34-A, Veneguera, Canaries, 2010 | Vegetation | 2 | 0.00 (0) | 0.00 (0) |
| 35 - | Ixodes frontalis (Panzer, 1798) | 35-A, Bretagne, France, 2008 | Eurasian Collared Dove (Streptopelia decaocto) | 1 | 0.00 (0) | 0.00 (0) |
| 36- | Ixodes hexagonus Leach, 1815 | 36-A, Bretagne, France, 2008 | European Red Fox (Vulpes vulpes) | 1 | 1.00 (1) | 0.00 (0) |
| 36-B, Beaumont-Monteux, France, 2008 | European Hedgehog (Erinaceus europaeus) | 2 | 1.00 (1) | 0.00 (0) | ||
| 37 - | Ixodes kerguelenensis André and Colas-Belcour, 1942 | 37-A, Crozet Archipelago, 2003 | Sea bird nests | 2 | 0.00 (0) | 0.00 (0) |
| 38 - | Ixodes pacificus Cooley and Kohls, 1943 | 38-A, Hopland, Mendocino Co, California, 1985 | Environement | 6 | 0.00 (0) | 0.00 (0) |
| 39 - | Ixodes persulcatus Schulze, 1930 | 39-A, Oulu, Finland, 2011 | Vegetation | 3 | 0.00 (0) | 0.00 (0) |
| 40 - | Ixodes ricinus (Linnaeus, 1758) | 40-A, Lehmäsaari, Finland, 2011 | Vegetation | 20 | 0.00 (0) | 0.00 (0) |
| 40-B, Rioja, Spain, 2011 | Vegetation | 20 | 0.00 (0) | 0.00 (0) | ||
| 40-C, Mafra, Portugal, 2013 | Vegetation | 16 | 0.00 (0) | 0.00 (0) | ||
| 40-D, Neuchâtel, Switzerland, 2012 | Vegetation | 16 | 0.00 (0) | 0.00 (0) | ||
| 40-E, Chur, Switzerland, 2012 | Hazel grouse (Tetrastes bonasia) | 4 | 0.00 (0) | 0.00 (0) | ||
| 40-F, Sénart, France, 2010 | European Roe Deer (Capreolus capreolus), rodents and passerines | 22 | 0.00 (0) | 0.05 (1) | ||
| 40-G, Ain, France, 2004 | Vegetation | 2 | 1.00 (1) | 0.00 (0) | ||
| 41 - | Ixodes scapularis Say, 1821 | 41-A, Queen's biology station, Ontario, Canada, 2013 | Vegetation | 3 | 0.00 (0) | 0.00 (0) |
| 42 - | Ixodes tasmani Neumann, 1899 | 42-A, Kilarney Circuit, Coolangubra, Australia, 1982 | Common Wombat (Vombatus ursinus) | 2 | 0.00 (0) | 1.00 (2) |
| 43 - | Ixodes unicavatus Neumann, 1908 | 43-A, Ariy Kamen Islet, Kamchatka, Russia, 2008 | Black-legged Kittiwake (Rissa tridactyla) and Red-legged Kittiwake (Rissa brevirostris) | 4 | 0.00 (0) | 0.00 (0) |
| 44 - | Ixodes uriae White, 1852 | 44-A, Possession Island, Crozet Archipelago, 2003 | King Penguin (Aptenodytes patagonicus) | 20 | 0.00 (0) | 0.00 (0) |
| 44-B, Hornoeya Island, Norway, 2010 | Brünnich's Guillemot (Uria lomvia) | 20 | 0.00 (0) | 0.00 (0) | ||
| 44-C, Triangle Island, Canada, 2010 | Rhinoceros Auklet (Cerorhinca monocerata) | 14 | 0.50 (7) | 0.00 (0) | ||
| 44-D, Pitchie2, Kamchatka, Russia, 2008 | Red-faced Cormorant (Phalacrocorax urile) | 20 | 0.00 (0) | 0.00 (0) | ||
| 44-E, Grimsey, Iceland, 2003 | Atlantic Puffin (Fratercula arctica), Common Guillemot (Uria aalge) and Black-legged Kittiwake (Rissa tridactyla) | 25 | 0.00 (0) | 0.20 (5) | ||
| 45 - | Ixodes sp.1 | 45-A, Guiglo, Ivory Coast, 1994 | Leopard (Panthera pardus) | 12 | 1.00 (12) | 0.00 (0) |
| 46 - | Ixodes sp.2 | 46-A, Guiglo, Ivory Coast, 1994 | Dog (Canis familiaris) | 12 | 1.00 (12) | 0.00 (0) |
| 47 - | Rhipicephalus annulatus (Say, 1821) | 47-A, Burkina-Faso, 2013 | Zebu (Bos indicus) | 17 | 1.00 (17) | 0.00 (0) |
| 47-B, Gogonou, Benin, 2012 | Zebu (Bos indicus) | 5 | 1.00 (5) | 0.00 (0) | ||
| 48 - | Rhipicephalus australis Fuller, 1899 | 48-A, CCA Farm, New Caledonia, 2003 | Cattle (Bos taurus) | 12 | 1.00 (12) | 0.00 (0) |
| 48-B, BMMMM Farm, New Caledonia, 2003 | Cattle (Bos taurus) | 12 | 1.00 (12) | 0.00 (0) | ||
| 49 - | Rhipicephalus bursa Canestrini and Fanzago, 1878 | 49-A, Italia | Cattle (Bos taurus) and Sheep (Ovis aries) | 2 | 1.00 (2) | 0.00 (0) |
| 50- | Rhipicephalus decoloratus Koch, 1844 | 50-A, Burkina-Faso, 2013 | Zebu (Bos indicus) | 20 | 0.95 (19) | 0.00 (0) |
| 50-B, Gogonou, Benin, 2012 | Zebu (Bos indicus) | 9 | 1.00 (9) | 0.00 (0) | ||
| 50-C, Sandvelt, South Africa, 2011 | Blue Wildebeest (Connochaetes taurinus), Greater Kudu (Tragelaphus strepsiceros) and Southern Eland (Taurotragus oryx) | 20 | 1.00 (20) | 0.00 (0) | ||
| 50-D, Queenstown, South Africa, 2011 | Zebu (Bos indicus) | 17 | 1.00 (17) | 0.00 (0) | ||
| 50-E, Vaalwater, South Africa, 2010 | Zebu (Bos indicus) | 19 | 1.00 (19) | 0.00 (0) | ||
| 50-F, Lephalale, South Africa, 2010 | South African Giraffe (Giraffa camelopardalis giraffa) | 9 | 1.00 (9) | 0.00 (0) | ||
| 50-G, Zimbabwe, 1998 | Impala (Aepyceros melampus) | 11 | 1.00 (11) | 0.00 (0) | ||
| 51 - | Rhipicephalus evertsi Neumann, 1897 | 51-A, Zimbabwe, 1998 | Zebu (Bos indicus) | 8 | 1.00 (8) | 0.00 (0) |
| 52 - | Rhipicephalus geigyi Aeschlimann and Morel, 1965 | 52-A, Burkina-Faso, 2013 | Zebu (Bos indicus) | 18 | 0.94 (17) | 0.00 (0) |
| 52-B, Gogonou, Benin, 2012 | Zebu (Bos indicus) | 3 | 1.00 (3) | 0.00 (0) | ||
| 53 - | Rhipicephalus microplus (Canestrini, 1888) | 53-A, Kpinnou, Benin, 2012 | Zebu (Bos indicus) | 20 | 1.00 (20) | 0.00 (0) |
| 53-B, Ambalanirana, Madagascar, 2013 | Zebu (Bos indicus) | 18 | 1.00 (18) | 0.00 (0) | ||
| 53-C, Imeritsiatosika, Madagascar, 2013 | Zebu (Bos indicus) | 14 | 1.00 (14) | 0.00 (0) | ||
| 53-D, Eglinton, South Africa, 2011 | Zebu (Bos indicus) | 20 | 1.00 (20) | 0.00 (0) | ||
| 53-E, Welverdiemda, South Africa, 2011 | Zebu (Bos indicus) | 11 | 1.00 (11) | 0.00 (0) | ||
| 53-F, Laboratory strain derived from field engorged females collected in Kpinnou, Benin, 2012 | Girolando (Cattle x Zebu hybrid) | 7 | 1.00 (7) | 0.00 (0) | ||
| 54 - | Rhipicephalus pusillus Gil Collado, 1936 | 54-A, Gard, France, 2006 | Vegetation | 2 | 1.00 (2) | 0.00 (0) |
| 55- | Rhipicephalus sanguineus (Latreille, 1806) | 55-A, Brazil | Dog (Canis familiaris) | 1 | 1.00 (1) | 0.00 (0) |
| 55-B, Montferrier-sur-Lez, France, 2013 | Dog (Canis familiaris) | 1 | 1.00 (1) | 0.00 (0) | ||
| 56 - | Rhipicephalus turanicus Pomerantzev, 1940 | 56-A, Italia | Vegetation | 2 | 1.00 (2) | 0.00 (0) |
| 56-B, Kerkyra, Greece, 2012 | Human (Homo sapiens) | 3 | 1.00 (3) | 0.00 (0) | ||
| 57- | Rhipicephalus sp.1 | 57-A, Guiglo area, Ivory Coast, 1994 | Leopard (Panthera pardus) | 3 | 1.00 (3) | 0.00 (0) |
| 58- | Rhipicephalus sp.2 | 58-A, Guiglo, Ivory Coast, 1994 | Dog (Canis familiaris) | 2 | 1.00 (2) | 0.00 (0) |
Using this procedure, all the tick-borne bacteria we detected belong to the Legionellales order and can be unambiguously assigned either to Coxiella or to its sister genus, Rickettsiella. Whole tick DNA extracts from more than two thirds of the specimens (637 out of 916, 69.6%) and the species (40 out of 58, 70.0%) were found to be positive for Coxiella (Fig 1 and Table 1). Coxiella was found in most tested genera of hard ticks (Rhipicephalus, Ixodes, Amblyomma, Dermacentor, Haemaphysalis) and soft ticks (Ornithodoros, Argas). In almost all infected species, Coxiella was detected in >90% of the examined specimens, indicating high Coxiella prevalence in diverse tick species. For example, infection was apparently fixed in populations of most Rhipicephalus and Ornithodoros species (Table 1). In contrast, Coxiella was frequently absent in Ixodes species and displayed highly variable prevalence in the five infected species (out of 12 screened).
Other Legionellales bacteria of the genus Rickettsiella were found in 52 specimens (5.7%) from six Ornithodoros species and three Ixodes species (Fig 1 and Table 1). In two of the three Rickettsiella-infected Ixodes species, i.e. I. ricinus and I. uriae, Coxiella was also found, but in different individuals and in distinct populations (i.e., no co-infection by Coxiella and Rickettsiella occurred at individual and population levels; Table 1). Adding the Rickettsiella-positive samples, we found that 689 of the 916 examined tick specimens (75.2%) and 44 of 58 screened species (76%) harbored either Coxiella or one of its relatives.
High genetic diversity among tick-borne Coxiella
To characterize Coxiella genetic diversity, we developed a multi-locus typing method based on five conserved bacterial genes including rpoB and four other housekeeping genes: 16S rRNA, 23S rRNA, GroEL and dnaK (Table A and Fig A in S1 Text). Multi-locus sequences were obtained from a subsample of 85 Coxiella- and 12 Rickettsiella-positive tick specimens (one to four specimens per infected species were examined). All five bacterial genes were successfully amplified from 71 Coxiella- and 12- Rickettsiella positive specimens representing 35 Coxiella- and six Rickettsiella-infected tick species. For five other Coxiella-infected species (i.e., 14 individual ticks), only three to four bacterial genes were successfully amplified. The sequences were easily readable without double peaks, indicating that there was no coinfection of Coxiella/Rickettsiella strains in any specimen.
The overall dataset included 33 to 40 alleles per bacterial gene (Table 2) and 51 new multi-locus genotypes (43 in Coxiella and eight in Rickettsiella). Within the Coxiella genus, all pairs of 16S rRNA gene sequences are at least 93% identical (Table 2) and range in threshold values typically used to delineate other Legionellales genera such as Legionella [33] and Rickettsiella [34]. Each of the infected tick species harbored a specific bacterial genotype or a set of closely related genotypes. None of the Coxiella multi-locus genotypes identified in ticks was identical to those of the 15 C. burnetii reference strains (Table B in S1 Text), although some showed moderate levels of nucleotide identity: pairwise identity between the two groups ranged from 77.8% to 97.7%. For each bacterial gene, the genetic diversity was significantly higher in the Coxiella strains of ticks than in C. burnetii as illustrated by the metrics on their respective genetic diversity (Table 2, paired t test, all P < 0.02).
Table 2. Genetic estimates for 85 Coxiella-like strains and for 15 Coxiella burnetii reference strains.
| Locus | Function | L | Strains | Ni | Pnsi | Na | Ps | Ad | π | D |
|---|---|---|---|---|---|---|---|---|---|---|
| 16S rRNA | Small ribosomal subunit | 1066 | Coxiella-like organisms | 85 | 93.0–100 | 40 | 262 | 0.985 | 0.038 | 39.960 |
| Coxiella burnetii | 15 | 99.7–100 | 4 | 4 | 0.552 | 0.001 | 1.224 | |||
| 23S rRNA | Large ribosomal subunit | 496 | Coxiella-like organisms | 82 | 84.2–100 | 34 | 177 | 0.978 | 0.079 | 39.393 |
| Coxiella burnetii | 15 | 99.4–100 | 2 | 2 | 0.133 | 0.001 | 0.267 | |||
| GroEL | Chaperone protein GROEL | 550 | Coxiella-like organisms | 82 | 68.7–100 | 37 | 292 | 0.982 | 0.179 | 98.527 |
| Coxiella burnetii | 15 | 99.4–100 | 5 | 5 | 0.562 | 0.002 | 0.895 | |||
| rpoB | DNA-directed RNA polymerase beta chain | 474 | Coxiella-like organisms | 85 | 68.7–100 | 38 | 250 | 0.981 | 0.205 | 97.136 |
| Coxiella burnetii | 15 | 99.3–100 | 4 | 4 | 0.467 | 0.001 | 0.648 | |||
| dnaK | Chaperone protein DNAK | 423 | Coxiella-like organisms | 74 | 69.6–100 | 33 | 227 | 0.979 | 0.177 | 75.789 |
| Coxiella burnetii | 15 | 99.8–100 | 3 | 1 | 0.362 | 0.001 | 0.381 | |||
| Full concatenated data set | 3009 | Coxiella-like organisms | 71 | 81.8–100 | 36 | 1139 | 0.984 | 0.115 | 346.990 | |
| Coxiella burnetii | 15 | 99.6–100 | 7 | 16 | 0.781 | 0.001 | 2.933 |
Analyses are based on nucleotide sequences of five housekeeping genes, excluding sites with alignment gaps and/or missing data. L, sequence fragment length in base pairs; Ni, Number of examined strains; Pnsi, Pairwise nucleotide sequence identity (%); Na, number of alleles; Ps, number of polymorphic sites; Ad, allelic diversity; π, nucleotide diversity; D, average number of nucleotide differences between sequences.
Coxiella burnetii originated from a tick-borne Coxiella ancestor
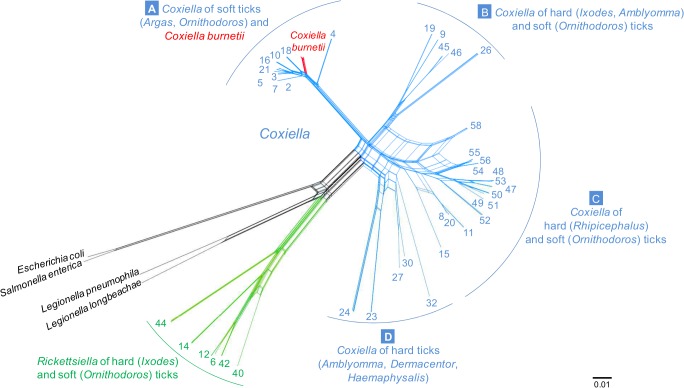
We constructed a multi-gene phylogeny of the entire Coxiella genus using a dataset that included the Coxiella and Rickettsiella sequences from ticks, the 15 C. burnetii reference genomes, as well as sequences from Legionella spp. and more distant outgroups that were available in GenBank (Table B in S1 Text). The concatenated sequences included 3009 unambiguously aligned base pairs (bp). Prior recombination tests showed that Coxiella and Rickettsiella strains did not exhibit a strictly clonal structure, but rather experienced significant genetic exchanges. We thus applied a sequence-based network approach that does not force relationships to be tree-like but rather incorporates recombination into the phylogenetic reconstruction. The network results (Fig 2), as well as the results from the Maximum Likelihood (ML) tree-based analysis (Fig B in S1 Text), consistently showed that the Coxiella genus can be split into four main clades (labeled A-to-D hereafter) with each clade clustering the Coxiella genotypes found in five to 15 tick species. The phylogenetic analyses also highlight that all C. burnetii isolates cluster into a unique subclade embedded within the A clade (Fig B and C in S1 Text and Fig 2). Notably, the closest relatives of C. burnetii are the Coxiella strains from soft ticks of the Ornithodoros and Argas genera, suggesting that the common ancestor of C. burnetii originated from a Coxiella hosted by soft ticks.
Fig 2. Phylogenetic network with concatenated 16S rRNA, 23S rRNA, GroEL, rpoB and dnaK sequences (3009 unambiguously aligned bp), including 71 Coxiella-like strains of ticks, 15 C. burnetii reference strains, and bacterial outgroups.
The four Coxiella clades are labeled A to D. A zoom on the A clade which contains C. burnetii isolates is shown in Supplementary Fig C in S1 Text. Each number corresponds to one tick species as detailed in Table 1. Blue—Coxiella-like organisms; red—C. burnetii; green—Rickettsiella; black- other bacteria. All multi-locus typing of Coxiella and Rickettsiella of ticks are new sequences from this study. The scale bar is in units of substitution/site.
The partitioning of Coxiella diversity among tick species revealed a complex structure, indicating a role for both co-divergence and horizontal transfer events in the evolution of this bacterial group. Closely related Coxiella-like organisms were frequently found in closely related tick species, a pattern suggestive of co-divergence between Coxiella and ticks (Fig B in S1 Text and Fig 2). For instance, all the Coxiella-like organisms found in the 12 examined Rhipicephalus tick species cluster together within the C clade, whereas all the Coxiella-like organisms in the Ixodes species cluster within the B clade (Fig B in S1 Text and Fig 2). Conversely, some Coxiella-like organisms found in related tick species are only distantly related and do not cluster together (e.g., the Coxiella-like organisms of Ornithodoros soft ticks are scattered among the A, B and C clades), a pattern suggestive of horizontal transfers among tick species.
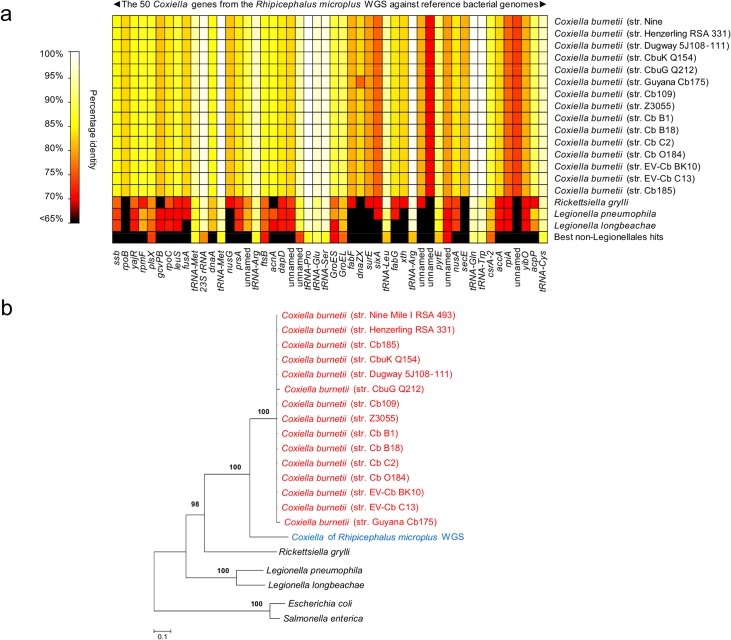
Further analyses were conducted by examining public repositories of DNA sequencing data generated by the whole genome sequencing (WGS) projects of the cattle tick R. microplus and the deer tick I. scapularis. Using the 1,995,281 bp C. burnetii (str. Nine Mile I RSA 493) genome as a probe, we found clear evidence of Coxiella infections in R. microplus, but not in I. scapularis. A total of 31 contigs (514–2,349 bp, totaling 34,990 bp) from R. microplus sequencing were uniquely attributable to Coxiella. They matched 50 genes of C. burnetii with 68-to-100% nucleotide identity (Fig A and Table C in S1 Text and Fig 3A). Alignment of the 31 Coxiella contigs to other bacterial genomes, including the 15 C. burnetii reference genomes (19,304 unambiguously aligned bp), corroborates the finding of our prior five loci-based analyses: the Coxiella strain identified in R. microplus is evolutionarily related, but distinct, to C. burnetii (Fig 3B).
Fig 3. Characterization of new Coxiella strains derived from whole-genome sequencing (WGS) of the cattle tick Rhipicephalus microplus.
(a) Percent identity of 50 genes uniquely attributable to Coxiella from R. microplus WGS versus 15 sequenced C. burnetii genomes and other reference genomes. (b) Bacterial phylogeny, comprising the Coxiella found in the R. microplus WGS data, reconstructed from the concatenated sequences of Fig 3A (19,304 unambiguously aligned bp) using maximum-likelihood (ML). Branch numbers indicate percent bootstrap support for major branches (1,000 replicates; only bootstrap values >90% are shown). The scale bar is in units of substitution/site.
Coxiella-like organisms are maternally inherited in ticks
It should be noted that the R. microplus WGS DNA examined above was extracted from eggs of an inbred strain of ticks (Deutsch strain), first derived from a few field specimens sampled in Texas in 2001, and reared for at least seven generations in the laboratory. The presence of Coxiella DNA in the WGS of R. microplus eggs thus raised the issue of their maternal inheritance in ticks. To address this question, 24 gravid females of four Coxiella-positive tick species were collected either from seabird nests (O. maritimus, n = 8 females), from a dog (R. sanguineus, n = 1) or from laboratory colonies (R. microplus, n = 7; A. americanum, n = 8) in order to test for the presence of Coxiella-infection in the cytoplasm of their progeny (8 to 14 surface-sterilized eggs per female were individually examined; i.e., 244 eggs in total). The occurrence of maternal transmission was detected in all four tick species and in almost all eggs: O. maritimus—79 Coxiella-positive eggs out of 80, R. sanguineus—14 out of 14, R. microplus—68 out of 70, and A. americanum—80 of 80. The mean transmission rate can thus be estimated at 0.988 (95% confidence interval, 0.965–0.994), demonstrating highly efficient maternal transmission of Coxiella in ticks. Maternal inheritance is thus widespread in the Coxiella genera, being found in three different clades (A: Coxiella-like organism of O. maritimus; C: R. sanguineus and R. microplus; D: A. americanum).
Differences in metabolic requirements of Coxiella-like organisms and C. burnetii
We next compared the metabolic requirements of Coxiella-like organisms with those of C. burnetii by assessing their ability to replicate in both an axenic medium ACCM2 (mimicking the environment of the acidified lysosome-like vacuoles of phagocytes typically colonized by C. burnetii; [35]) and directly inside vertebrate host cells. First, ACCM2 was inoculated with Coxiella-like organisms extracted from eggs of either O. maritimus, R. microplus or A. americanum. Inoculated media were incubated for 10 days under standard conditions used to amplify C. burnetii. Although our C. burnetii positive controls readily replicated in the media, the Coxiella obtained from eggs of the three tick species did not grow. We then incubated egg homogenates from ticks of O. maritimus and R. microplus with mammalian cell cultures for seven days. Similar to results under axenic conditions, the incubation of vertebrate cell lines with Coxiella-like organisms failed to produce Coxiella-containing vacuoles, whereas the same cell lines incubated with C. burnetii under the same conditions were readily infected. The apparent inability to amplify tick-borne Coxiella through standardized protocols, well-characterized for C. burnetii, suggests that, despite their phylogenetic proximity, the Coxiella-like bacteria are adapted to radically different environments.
Discussion
Since its original description, C. burnetii infections have been characterized in a wide variety of hosts. While only two species have been formally identified within the Coxiella genus, we show here that a far greater diversity of Coxiella exists in ticks. We detect the presence of Coxiella-like organisms in many more tick species than previously known [17,18,19,20,21,22,23,24,25] and describe a far wider genetic diversity among these bacteria than previously suspected. The incidence of Coxiella, as well as of its sister genus Rickettsiella, in ticks is exceptionally high, with approximately three quarters of tick species infected. Although possible tick-borne transmission of C. burnetii has been reported [1,2,8], none of the 43 new Coxiella genotypes identified here are identical to C. burnetii. We also demonstrate for genetically divergent Coxiella strains (i.e., members of the A, C and D clades) found in four tick species that infection is primarily transmitted maternally via the egg cytoplasm. These results converge to support the hypothesis that these Coxiella-like organisms are specific endosymbionts of ticks. Phylogenetic evidence further shows that one of the Coxiella-like organisms belonging to the A clade and primarily hosted by soft ticks has served as the progenitor of C. burnetii.
Three complementary lines of argument indicate a much longer evolutionary history for Coxiella-tick associations than for vertebrate-Coxiella associations. The first lies in the broad distribution of Coxiella and Rickettsiella bacteria across tick species, genera and families. The second concerns the extensive genetic diversity found in tick-borne Coxiella strains compared to C. burnetii strains, as illustrated by the clear subdivision of this genus into four highly divergent clades (A-D). Finally, the clustering of all C. burnetii strains within one of the clades of tick-borne Coxiella shows that the ancestor of C. burnetii was a tick-associated bacterium which succeeded in infecting vertebrate cells. The remarkably low genetic diversity of C. burnetii, previously noted in other studies [36,37], indicates a unique and recent emergence of this highly infectious vertebrate pathogen. Interestingly, this hypothesis was initially raised a decade ago from observations of the profound differences in genome architecture of C. burnetii relative to other pathogenic intracellular bacteria [38]. It was again emphasized from the genome sequencing of new C. burnetii strains [39]. Our data brings further support to this hypothesis by demonstrating that C. burnetii roots within the Coxiella phylogeny. Comparative genome sequences of C. burnetii [38, 39] and of the Coxiella-like organism from A. americanum [16] also suggest that Coxiella bacteria differ substantially in terms of genome size and gene content. The C. burnetii genome (A clade) has a size of ca 2Mb [38, 39], whereas the genome of the Coxiella-like organism isolated in A. americanum (D clade) is only about a 1/3 of this size (ie. 0.66 Mb) with a large percentage of missing genes [16]. This reduction in genome size may limit the transition to pathogenicity, and suggests that some Coxiella-like organisms may have evolved towards exclusive and irreversibly specialized interactions with their tick hosts. Overall, the diversity of genome sizes emphasizes that members of the different Coxiella clades may have retained a variety of evolutionary strategies to favour their spread and persistence in their hosts.
We identified Coxiella as a major emerging clade of bacterial endosymbionts allied to ticks. Coxiella-like organisms are maternally-transmitted through the egg cytoplasm at high frequency with 98–100% mother-to-offspring transmission, a pattern also reported in previous studies [20,21,26]. This transmission pattern is the rule for a variety of bacterial endosymbionts that live exclusively within arthropod cells [28,29,40]. While some, like Wolbachia, are globally common symbionts estimated to infect ca. 40% of insect species [41,42], others are globally rare, but common and important in particular arthropod groups [28]. This is precisely the case for Coxiella-like organisms; although they have not been found in other arthropod species, they are commonly associated with ticks. This leads to the obvious question of the phenotypic consequences of Coxiella-tick interactions. In some cases, Coxiella-like organisms of ticks likely act as obligate mutualistic symbionts required to support normal tick development, potentially provisioning their hosts with essential nutriments absent in vertebrate blood [16,20,21,26,27]. The ubiquity of Coxiella in some tick groups-such as in the Rhipicephalus genus in which infection is at fixation- corroborates the hypothesis of an obligate endosymbiont. This is not, however, the case for all tick species since some, such as I. ricinus and I. uriae, harbour Coxiella-like organisms at much lower frequencies. In these tick species, Coxiella is more likely to behave as a conditional mutualist-i.e., that confers advantages under certain environmental conditions- or as a reproductive parasite-i.e., that manipulates host reproduction toward the production of daughters (the transmitting sex), as commonly observed in arthropods with a variety of facultative symbionts [28,40]. It should also be noted that other endosymbionts also occur in ticks and may have evolved under complex multispecific interactions [17,24]. For instance, whereas the soft tick O. moubata was not found to be infected by a Coxiella-like organism in the present study, this tick species has been found to be infected by an endosymbiont belonging to the Francisella genus [17]. Endosymbionts other than Coxiella may thus interact with ticks, a pattern suggesting that endosymbiotic systems can be dynamic across tick lineages. These different hypotheses will now require specific testing.
Another question remains concerning the degree of vertebrate infection risk by the Coxiella-like organisms of ticks. Ticks are found worldwide and blood-feed on many different hosts; a combination of traits that may facilitate tick-to-vertebrate transfers of Coxiella. However, the bacteria observed in this study seem confined to ticks and, to our knowledge, none have ever been isolated from a vertebrate or associated with clinical symptoms. This suggests that these tick-associated bacteria currently pose a much lower infection risk to vertebrates than C. burnetii. As discussed above, the genome reduction of the Coxiella-like organism isolated in A. americanum, with the lack of nearly all the genes associated with pathogenicity [16], corroborates this view. Moreover, the inability to grow tick-borne bacteria in vertebrate cells highlights the significant barrier that must be overcome by the bacteria to successively achieve tick-to-vertebrate transmission. This type of transmission may, nonetheless, occasionally occur; an avian Coxiella-like organism was recently reported to induce fatal systematic infections in domestic birds [43,44,45]. A very similar infection pattern was found for another maternally inherited endosymbiont, Arsenophonus, a widespread bacterium in different insect groups [41,46,47,48,49]. In particular, some Arsenophonus strains were detected in the phloem of plants fed on by infected phytophagous insects and were assumed to be opportunistic plant pathogens [50]. In such cases, the plant host may act as an ecological arenas for the global exchange of endosymbionts like Arsenophonus, serving as a possible intermediate host for the horizontal transfer of bacteria among insect species [48]. In the case of Coxiella-like organisms, the extent of exchange between different tick species via the vertebrate host is yet to be established, but could be favoured by tick co-feeding (ticks feeding in close proximity on the host). The genetic similarity between Coxiella-like organisms found in unrelated tick species highlights the capacity to shift tick host species. Future research is now needed to assess the potential of different Coxiella-like organisms to infect vertebrates.
The reasons why C. burnetii is a highly virulent pathogen of vertebrates, but not Coxiella-like organisms (especially those from the A clade) remain unknown. As an intracellular pathogen with airborne transmission, C. burnetii has evolved specific mechanisms to survive in the abiotic environment, as well as to infect and exploit vertebrate cells [15]. Several evolutionary pathways may explain the acquisition of the genetic material necessary for this major lifestyle transition; this includes spontaneous genetic mutations in the genome of a tick-Coxiella ancestor, or the more likely transfer and integration of virulence genes from a co-infecting pathogen. The opportunity of gene transfer among bacteria, irrespective of their pathogenic or symbiotic properties, relies on their frequent co-occurrence within the same tick host [25,51,52]. The Coxiella-like organisms of the A clade may have dynamic genomes as observed in many arthropod symbionts: although they reside in confined intracellular environments, arthropod symbionts commonly experience variable degrees of recombination and gene transfer [53,54,55,56,57]. These gene transfers have served as immediate and powerful mechanisms of rapid adaptation in many endosymbionts, such as Wolbachia [56] and Hamiltonella [55,57]. This mechanism may explain the evolutionary transition from a Coxiella tick-symbiont of the A clade to the vertebrate pathogen C. burnetii. Other genetic connections are also possible; several C. burnetii genes that may contribute to major virulence traits, such as tissue tropism, are similar to eukaryotic genes and may have been acquired through lateral gene transfers from eukaryotes [38,39]. Detailed studies of virulence genes in C. burnetii and their homology with Coxiella-like organisms of the A clade will now be necessary to understand the remarkable emergence of the Q fever agent.
The evolutionary transition observed within the Coxiella genus is one of the rare cases reported to date of an arthropod-inherited symbiont evolving metabolic adaptations leading to the emergence of a vertebrate infectious disease. Another such transition occurred in the Rickettsia genus. The best-known members of this genus are transmitted by blood-feeding arthropods and are pathogenic in the vertebrate host. However, in recent years, many maternally-inherited Rickettsia endosymbionts found exclusively in arthropods have been discovered [58,59]. The examination of the evolutionary history of the Rickettsia genus revealed that this bacterium originated from endosymbionts of invertebrates and only secondarily became vertebrate pathogens [58,59]. Like Coxiella, some Rickettsia species of blood-feeding hosts have underwent a horizontal transmission through a vertebrate host, leading to pathogen emergence. Other bacteria, such as Arsenophonus [47,48] and Sodalis [60] may have had similar life cycle transitions, but the case of Coxiella is unique in that the arthropod host is no longer required to complete its life cycle.
In conclusion, we show that C. burnetii arose from a rare and recent event: the evolutionary transformation of a maternally inherited endosymbiont of ticks into a specialized and virulent pathogen of vertebrates. This raises a series of exciting questions related to both how Coxiella endosymbionts made the major evolutionary transition leading to the emergence of Q fever and their role in the population dynamics of ticks. Identifying the evolutionary processes that transform symbiotic bacteria into emerging pathogens will require further exploration into the biology of the entire Coxiella genus.
Methods
Tick collection
The examined specimens represent the two main tick families, nine genera, 58 species and 112 populations from around the world (Table 1). Field specimens were sampled on various host species belonging to major mammal and bird families or from their habitats. We also used specimens from laboratory colonies reared in captivity for at least three generations for six tick species (derived from field specimens collected in North America, South America, Africa and China). All samples were preserved in 70–90% ethanol at room temperature until use. Before storage, tick eggs collected under laboratory conditions were surface-sterilized with 2.6% sodium hypochlorite and 0.5% SDS for 1 min and washed with sterile water to avoid external bacterial contamination.
Coxiella screening and typing
Tick DNA was individually extracted using the DNeasy Blood & Tissue Kit (QIAGEN) following manufacturer instructions. DNA template quality was systematically verified by PCR amplification of the 18S ribosomal RNA (18S rRNA) or the cytochrome oxydase 1 (C01) arthropod primers (Table A in S1 Text). Tick DNA samples were then tested for Coxiella presence using a nested PCR assay and sequencing of the rpoB gene using Coxiella-specific primers. The use of nested PCR was efficient at decreasing the probability of contamination from unwanted amplification products. Additional PCR assays on the 16S rRNA, 23S rRNA, GroEL and dnaK genes were conducted on a subsample of Coxiella-positive tick DNA to obtain additional DNA sequences for phylogenetic analyses. We used 15 recently published genomes of C. burnetii (mainly isolated from humans and ruminants) and the genome of Rickettsiella grylli from woodlice (listed in Table B in S1 Text) as references to design PCR primers. The efficiency of our typing method was ascertained through positive PCR amplification and clear sequences for the five loci in four cultured reference strains of C. burnetii (Table B in S1 Text). Gene features, primers and PCR conditions are detailed in Table A in S1 Text. All PCR products were visualized through electrophoresis in a 1.5% agarose gel. Positive PCR products were purified and sequenced in both directions (EUROFINS). The chromatograms were manually inspected and cleaned with CHROMAS LITE (http://www.technelysium.com.au/chromas_lite.html) and sequence alignments were done using CLUSTALW [61], both implemented in MEGA [62].
Coxiella sequences were also searched for in the whole genome sequence (WGS) data of R. microplus and I. scapularis (GenBank accession numbers ADMZ02000000 and ABJB000000000, respectively) using the 1,995,281 bp C. burnetii genome (str. Nine Mile I RSA 493, GenBank accession number NC002971) as a probe and the Basic Local Alignment Search Tool (BLAST) with default parameters. Table C in S1 Text reports the number and content of Coxiella contigs that were detected in the R. microplus WGS data.
Molecular and phylogenetic analyses
The GBLOCKS program [63] with default parameters was used to remove poorly aligned positions and to obtain non-ambiguous sequence alignments. Sequences of individual genes that differed by one or more nucleotides were assigned distinct allele numbers using DNASP [64], with the option of excluding sites with alignment gaps and/or missing data. Tick-borne Coxiella strains are defined as each unique combination of alleles. The genetic diversity estimates (Ps, number of polymorphic sites; Ad, allelic diversity; π, nucleotide diversity; D, average number of nucleotide differences between sequences) were computed using DNASP. Other statistical analyses were carried out using the R statistical package. All sequence alignments were checked for putative recombinant regions using the GENECONV [65] and RDP [66] methods available in the RDP3 computer analysis package [67].
Phylogenetic analyses were based on single and concatenated sequences of the five bacterial genes used in the multi-locus typing scheme and on the 50 Coxiella genes found in the R. microplus WGS data. Sequence alignments included Coxiella and Rickettsiella sequences obtained in this study from tick DNA, as well as sequences available in GenBank from reference strains of C. burnetii, Rickettsiella grylli, Legionella pneumophila, L. longbeacheae, and two more distantly related bacteria, Escherichia coli and Salmonella enterica (Table B in S1 Text). The evolutionary models fitting the sequence data most closely were determined using the Akaike information criterion with the program MEGA. For each data set examined, the best-fit approximation was the general time reversible model with gamma distribution and invariant sites (GTR+G+I). Network-based phylogenetic analyses were done using SplitsTree, implementing the evolutionary model under the agglomerating NeighborNet algorithm [68]. Tree-based phylogenetic analyses were done using maximum-likelihood (ML) analyses. A ML heuristic search using a starting tree obtained by neighbor-joining was conducted in MEGA. Clade robustness was assessed by bootstrap analysis using 1,000 replicates.
Culture assays
We first assessed the ability of tick-borne Coxiella to replicate in an axenic medium as follows. Tick eggs were surface-sterilized as described above and homogenized by hand in sterile water. Eggs homogenates were used to inoculate 2ml of the axenic medium ACCM2 [35] and incubated three days in a humidified atmosphere of 5% CO2 and 2.5% O2 at 37°C. 50μl of each culture were then diluted in 2ml of fresh ACCM2 and further incubated under the same conditions for 10 days to assess bacterial growth. We then assessed the ability of tick-borne Coxiella to replicate inside vertebrate host cells as follows. Surface-sterilized tick eggs were homogenized by hand in 1 ml of 10% Fœtal Bovine Sérum (FBS) supplemented MEM medium (GIBCO). The homogenate (0.5 ml) was diluted in 25 ml of 10% SVF-MEM and centrifuged at 4000 rpm (2000g) at 4°C for 30 min. Ten ml of the supernatant was mixed with 10 ml of 10% FSB-MEM and again centrifuged at 2000g at 4°C for 30 min. Ten ml of the supernatant was harvested and filtered through a sterile 0.45 μm pore size filter (MILLIPORE). Two flasks containing confluent Sheep Fœtal Thymus cells (SFT) cells were inoculated with 5 ml of the obtained filtrate and incubated at 35°C and allowed to grow for 12 weeks. Cell culture flasks were observed daily for the presence of contamination or growth signs such as vacuoles containing Coxiella, during the first week then once a week. As a positive control, a homogenate of C. burnetii was used following the same protocol.
Accession codes
Nucleotide sequences of PCR-amplified fragments of tick-borne Coxiella and Rickettsiella genes have been deposited in the GenBank nucleotide database under accession codes KP994768-KP994862 (16S rRNA), KP994678-KP994767 (23S rRNA), KP985445-KP985537 (GroEL), KP985265-KP985357 (rpoB) and KP985358-KP985444 (dnaK).
Supporting Information
Genes and primers used in polymerase chain reaction (PCR) assays to detect Coxiella and relatives and to control tick DNA quality. The same primers were used for the Coxiella and Rickettsiella typing, with the exception of the 16S rRNA gene for which different primers were designed for the two bacteria. Nested PCR amplifications (16S rRNA, 23S rRNA, GroEL, rpoB and dnaK) were performed as follows: the first PCR run with the external primers was performed in a 10μLvolume containing 20–50 ng of genomic DNA, 3 mM of each dNTP (Thermo Scientific), 8 mM of MgCl2 (Roche Diagnostics), 3 μM of each primer, 1 μL of 10× PCR buffer (Roche Diagnostics), and 0.5 U of Taq DNA polymerase (Roche Diagnostics). A 1-μL aliquot of the PCR product from the first reaction was then used as a template for the second round of amplification. The second PCR was performed in a total volume of 25 μL and contained 8 mM of each dNTP (Thermo Scientific), 10 mM of MgCl2 (ThermoScientific), 7.5 μM of each of the internal primers, 2.5 μL of 10×PCR buffer (Thermo Scientific), and 1.25 U of Taq DNA polymerase (Thermo Scientific). Non-nested PCR amplifications (CO1 and 18S rRNA) were performed following conditions similar to the first PCR run used in the nested PCR assays. All PCR amplifications were performed under the following conditions: initial denaturation at 93°C for 3 min, 35 cycles of denaturation (93°C, 30 s), annealing (Tm = 50–56°C, depending on primers, 30 s), extension (72°C, 1–2 min), and a final extension at 72°C for 5 min. Table B. List, biological features and GenBank accession numbers of the bacterial strains used as references in molecular and phylogenetic analyses.* reference strains of C. burnetii used for primer testing. Table C. List, sequence accession numbers and features of the 31 Coxiella contigs from the whole-genome shotgun sequencing (WGS) of the cattle tick Rhipicephalus microplus. Fig A. Map of the Coxiella burnetii genome (strain Nine Mile I RSA 493) showing the position of the genetic markers (in blue) used in this study. The arrows indicate the position along the chromosome of the five housekeeping genes (16S rRNA, 23S rRNA, GroEL, rpoB and dnaK) used in the multi-locus typing of tick-borne Coxiella infections. The numbered boxes (1–31) indicate the position of the 31 Coxiella contigs (listed in Table C in S1 Text) detected from the whole genome sequencing of the hard tick Rhipicephalus microplus. Fig B. Coxiella and Rickettsiella phylogeny constructed using maximum-likelihood (ML) estimations based on 16S rRNA, 23S rRNA, GroEL, rpoB and dnaK concatenated sequences (3009bp), including 71 Coxiella-like strains of ticks, 15 C. burnetii reference strains and outgroups. The four Coxiella clades are labeled A to D. Each number corresponds to one tick species as detailed in Table 1. Blue, Coxiella-line organisms; red, C. burnetii; green, Rickettsiella; black, other bacteria. All multi-locus typing of tick-borne Coxiella and Rickettsiella of ticks are new sequences from this study. Branch numbers indicate percentage bootstrap support for major branches (1000 replicates; only bootstrap values >90% are shown). Fig C. Inset of Coxiella network from Fig 2 with focus on the A clade (Coxiella of soft ticks and C. burnetii). Each number corresponds to one tick species as detailed in Table 1. Blue, Coxiella-line organisms; red, C. burnetii. The scale bar is in units of substitution/site.
(DOCX)
Acknowledgments
We are grateful to X. Bailly, A. Barbour, L. Beati, T. Boulinier, J.L. Chapuis, F. Dantas-Torres, A. Estrada-Peña, M. Gauthier-Clerc, L. Gern, D. González Acuña, J. Gonzalez-Solis, U. Guidon, A. Guglielmone, K. Hansford, M. Hipfner, A. Jaeger, D. Jenny, M. Labruna, R. Lane, M.S. Latrofa, C. Lebarbenchon, J. Liu, J. Medlock, S. Nava, A.C. Norte, U.E. Schneppat, D. Spratt, P. Tortosa and M. Vittecoq for help at different stages of this work, and to three anonymous reviewers for helpful comments and suggestions.
Data Availability
Nucleotide sequences of PCR-amplified fragments of tick-borne Coxiella and Rickettsiella genes have been deposited in the GenBank nucleotide database under accession codes KP994768-KP994862 (16S rRNA), KP994678-KP994767 (23S rRNA), KP985445-KP985537 (GroEL), KP985265-KP985357 (rpoB) and KP985358-KP985444 (dnaK).
Funding Statement
Financial support was provided by the CNRS-INEE (Programme PEPS-Ecologie de la Santé 2014, ‘SYMPATTIQUES’: OD LZ; http://www.cnrs.fr/inee/outils/PEPS2014.htm), French Polar Institue (IPEV,programme no. 333: KDMcC; http://www.institut-polaire.fr/ipev/programmes_de_recherche), the CNRS–INEE/TAAF (Programme Iles Eparses “PathOrnithoTiques”: KDMcC; http://www.cnrs.fr/inee/recherche/programmes/iles_eparses.htm), and the Agence Nationale de la Recherche (ANR-11-BSV7-003-02: KDMcC, ANR-13-BSV7-0018-01: KDMcC, ANR-3215AO-3R077-STEV: FR, PD, CA, JFT, ANR-11-LABX-0048: FV, OM, LZ, EJ; http://www.agence-nationale-recherche.fr/). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Madariaga MG, Rezai K, Trenholme GM, Weinstein RA. Q fever: a biological weapon in your backyard. Lancet Infect Dis. 2003;3: 709–721. [DOI] [PubMed] [Google Scholar]
- 2. Raoult D, Marrie T, Mege J. Natural history and pathophysiology of Q fever. Lancet Infect Dis. 2005;5: 219–226. [DOI] [PubMed] [Google Scholar]
- 3. Vanderburg S, Rubach MP, Halliday JE, Cleaveland S, Reddy EA, Crump JA. Epidemiology of Coxiella burnetii infection in Africa: a OneHealth systematic review. PLoS Negl Trop Dis. 2014;8: e2787 10.1371/journal.pntd.0002787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Angelakis E, Raoult D. Q Fever. Vet Microbiol. 2010;140: 297–309. 10.1016/j.vetmic.2009.07.016 [DOI] [PubMed] [Google Scholar]
- 5. Bewley KR. Animal models of Q fever (Coxiella burnetii). Comp Med. 2013;63: 469–476. [PMC free article] [PubMed] [Google Scholar]
- 6. van der Hoek W, Dijkstra F, Schimmer B, Schneeberger PM, Vellema P, Wijkmans C, et al. Q fever in the Netherlands: an update on the epidemiology and control measures. Euro Surveill 2010;15. [PubMed] [Google Scholar]
- 7. Russell-Lodrigue KE, Andoh M, Poels MWJ, Shive HR, Weeks BR, Zhang GQ, et al. Coxiella burnetii isolates cause genogroup-specific virulence in mouse and guinea pig models of acute Q fever. Infect Immun. 2009;77: 5640–5650. 10.1128/IAI.00851-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. McDade JE. Historical aspects of Q fever In: Marie TJ, editor. Q Fever Volume 1: The Disease Boca Raton: CRC Press; 1990. pp. 5–21. [Google Scholar]
- 9. Oyston PC, Davies C. Q fever: the neglected biothreat agent. J Med Microbiol. 2011;60: 9–21. 10.1099/jmm.0.024778-0 [DOI] [PubMed] [Google Scholar]
- 10. Weisburg WG, Dobson ME, Samuel JE, Dasch GA, Mallavia LP, Baca O, et al. Phylogenetic diversity of the Rickettsiae. J Bacteriol. 1989;171: 4202–4206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Leclerque A, Kleespies RG. Type IV secretion system components as phylogenetic markers of entomopathogenic bacteria of the genus Rickettsiella . FEMS Microbiol Ecol. 2008;279: 167–173. 10.1111/j.1574-6968.2007.01025.x [DOI] [PubMed] [Google Scholar]
- 12. Bouchon D, Cordaux R, Grève P. Rickettsiella, intracellular pathogens of arthropods In: Zchori-Fein E, Bourtzis K, editors. Manipulative Tenants. Boca Raton: CRC Press; 2012. pp. 127–148. [Google Scholar]
- 13. Tsuchida T, Koga R, Horikawa M, Tsunoda T, Maoka T, Matsumoto S, et al. Symbiotic bacterium modifies aphid body color. Science 2010;330: 1102–1104. 10.1126/science.1195463 [DOI] [PubMed] [Google Scholar]
- 14. Tan CK, Owens L. Infectivity, transmission and 16S rRNA sequencing of a rickettsia, Coxiella cheraxi sp. nov., from the freshwater crayfish Cherax quadricarinatus . Dis Aquat Organ. 2000;41: 115–122. [DOI] [PubMed] [Google Scholar]
- 15. van Schaik EJ, Chen C, Mertens K, Weber MM, Samuel JE. Molecular pathogenesis of the obligate intracellular bacterium Coxiella burnetii . Nat Rev Microbiol. 2013;11: 561–573. 10.1038/nrmicro3049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Smith TA, Driscoll T, Gillespie JJ, Raghavan R. A Coxiella-like Endosymbiont is a potential vitamin source for the Lone Star Tick. Genome Biol Evol. 2015;7: 831–838. 10.1093/gbe/evv016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Noda H, Munderloh UG, Kurtti TJ. Endosymbionts of ticks and their relationship to Wolbachia spp. and tick-borne pathogens of humans and animals. Appl Environ Microbiol. 1997;63: 3926–3932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Almeida AP, Marcili A, Leite RC, Nieri-Bastos FA, Domingues LN, Ricardo Martins J, et al. Coxiella symbiont in the tick Ornithodoros rostratus (Acari: Argasidae). Ticks Tick Borne Dis. 2012;3: 203–206. 10.1016/j.ttbdis.2012.02.003 [DOI] [PubMed] [Google Scholar]
- 19. Duron O, Jourdain E, McCoy KD. Diversity and global distribution of the Coxiella intracellular bacterium in seabird ticks. Ticks Tick Borne Dis. 2014;5: 557–563. 10.1016/j.ttbdis.2014.04.003 [DOI] [PubMed] [Google Scholar]
- 20. Klyachko O, Stein BD, Grindle N, Clay K, Fuqua C. Localization and visualization of a Coxiella-type symbiont within the lone star tick, Amblyomma americanum . Appl Environ Microbiol. 2007;73: 6584–6594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Machado-Ferreira E, Dietrich G, Hojgaard A, Levin M, Piesman J, Zeidner NS, et al. Coxiella symbionts in the Cayenne tick Amblyomma cajennense . Microb Ecol. 2011;62: 134–142. 10.1007/s00248-011-9868-x [DOI] [PubMed] [Google Scholar]
- 22. Lalzar I, Harrus S, Mumcuoglu KY, Gottlieb Y. Composition and seasonal variation of Rhipicephalus turanicus and Rhipicephalus sanguineus bacterial Communities. Appl Environ Microbiol. 2012;78: 4110–4116. 10.1128/AEM.00323-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Jasinskas A, Zhong J, Barbour AG. Highly prevalent Coxiella sp. bacterium in the tick vector Amblyomma americanum . Appl Environ Microbiol. 2007;73: 334–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Clay K, Klyachko O, Grindle N, Civitello D, Oleske D, Fuqua C. Microbial communities and interactions in the lone star tick, Amblyomma americanum . Mol Ecol. 2008;17: 4371–4381. [DOI] [PubMed] [Google Scholar]
- 25. Wilkinson DA, Dietrich M, Lebarbenchon C, Jaeger A, Le Rouzic C, Lagadec E, et al. Massive infection of seabird ticks with bacterial species related to Coxiella burnetii . Appl Environ Microbiol. 2014;80: 3327–3333. 10.1128/AEM.00477-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lalzar I, Friedmann Y, Gottlieb Y. Tissue tropism and vertical transmission of Coxiella in Rhipicephalus sanguineus and Rhipicephalus turanicus ticks. Environ Microbiol; 2014;16: 3657–3668. 10.1111/1462-2920.12455 [DOI] [PubMed] [Google Scholar]
- 27. Zhong J, Jasinskas A, Barbour AG. Antibiotic treatment of the tick vector Amblyomma americanum reduced reproductive fitness. PLoS One. 2007;2: e405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Moran NA, McCutcheon JP, Nakabachi A. Genomics and evolution of heritable bacterial symbionts. Annu Rev Genet. 2008;42: 165–190. 10.1146/annurev.genet.41.110306.130119 [DOI] [PubMed] [Google Scholar]
- 29. Wernegreen JJ. Endosymbiosis. Curr Biol. 2012;22: R555–561. 10.1016/j.cub.2012.06.010 [DOI] [PubMed] [Google Scholar]
- 30. Hosokawa T, Koga R, Kikuchi Y, Meng XY, Fukatsu T. Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci USA. 2010;107: 769–774. 10.1073/pnas.0911476107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Akman L, Yamashita A, Watanabe H, Oshima K, Shiba T, Hattori M, et al. Genome sequence of the endocellular obligate symbiont of tsetse flies, Wigglesworthia glossinidia . Nat Genet. 2002;32: 402–407. [DOI] [PubMed] [Google Scholar]
- 32. Husnik F, Chrudimsky T, Hypsa V. Multiple origins of endosymbiosis within the Enterobacteriaceae (gamma-Proteobacteria): convergence of complex phylogenetic approaches. BMC Biol. 2011;9: 87 10.1186/1741-7007-9-87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Gomez-Valero L, Rusniok C, Buchrieser C. Legionella pneumophila: population genetics, phylogeny and genomics. Infect Genet Evol. 2009;9: 727–739. 10.1016/j.meegid.2009.05.004 [DOI] [PubMed] [Google Scholar]
- 34. Leclerque A, Kleespies RG. A Rickettsiella bacterium from the hard tick, Ixodes woodi: molecular taxonomy combining multilocus sequence typing (MLST) with significance testing. PLoS One. 2012;7: e38062 10.1371/journal.pone.0038062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Omsland A, Beare PA, Hill J, Cockrell DC, Howe D, Virtaneva K, et al. Isolation from animal tissue and genetic transformation of Coxiella burnetii are facilitated by an improved axenic growth medium. Appl Environ Microbiol. 2011;77: 3720–3725. 10.1128/AEM.02826-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Sekeyová Z, Roux V, Raoult D. Intraspecies diversity of Coxiella burnetii as revealed by com1 and mucZ sequence comparison. FEMS Microbiol Lett. 1999;180: 61–67. [DOI] [PubMed] [Google Scholar]
- 37. Pearson T, Hornstra HM, Sahl JW, Schaack S, Schupp JM, Beckstrom-Sternberg SM, et al. When outgroups fail; phylogenomics of rooting the emerging pathogen, Coxiella burnetii . Syst Biol. 2013;62: 752–762. 10.1093/sysbio/syt038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Seshadri R, Paulsen IT, Eisen JA, Read TD, Nelson KE, Nelson WC, et al. Complete genome sequence of the Q-fever pathogen Coxiella burnetii . Proc Natl Acad Sci USA. 2003;100: 5455–5460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Beare PA, Unsworth N, Andoh M, Voth DE, Omsland A, Gilk SD, et al. Comparative genomics reveal extensive transposon-mediated genomic plasticity and diversity among potential effector proteins within the genus Coxiella . Infect Immun. 2009;77: 642–656. 10.1128/IAI.01141-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Engelstadter J, Hurst GDD. The ecology and evolution of microbes that manipulate host reproduction. Annu Rev Ecol Evol Syst. 2009;40: 127–149. [Google Scholar]
- 41. Duron O, Bouchon D, Boutin S, Bellamy L, Zhou L, Engelstadter J, et al. The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol. 2008;6: 27 10.1186/1741-7007-6-27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Zug R, Hammerstein P. Still a host of hosts for Wolbachia: analysis of recent data suggests that 40% of terrestrial arthropod species are infected. PLoS One. 2012;7: e38544 10.1371/journal.pone.0038544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Shivaprasad HL, Cadenas MB, Diab SS, Nordhausen R, Bradway D, Crespo R, et al. Coxiella-like infection in psittacines and a toucan. Avian Dis. 2008;52: 426–432. [DOI] [PubMed] [Google Scholar]
- 44. Woc-Colburn AM, Garner MM, Bradway D, West G, D'Agostino J, Trupkiewicz J, et al. Fatal coxiellosis in Swainson's Blue Mountain Rainbow Lorikeets (Trichoglossus haematodus moluccanus). Vet Pathol. 2008;45: 247–254. 10.1354/vp.45-2-247 [DOI] [PubMed] [Google Scholar]
- 45. Vapniarsky N, Barr BC, Murphy B. Systemic Coxiella-like infection with myocarditis and hepatitis in an eclectus parrot (Eclectus roratus). Vet Pathol. 2012;49: 717–722. 10.1177/0300985811409251 [DOI] [PubMed] [Google Scholar]
- 46. Duron O, Wilkes TE, Hurst GDD. Interspecific transmission of a male-killing bacterium on an ecological timescale. Ecol Lett. 2010;13: 1139–1148. 10.1111/j.1461-0248.2010.01502.x [DOI] [PubMed] [Google Scholar]
- 47. Duron O, Schneppat UE, Berthomieu A, Goodman SE, Droz B, Paupy C, et al. Origin, acquisition and diversification of heritable bacterial endosymbionts in louse flies and bat flies. Mol Ecol. 2014;23: 2105–2017. 10.1111/mec.12704 [DOI] [PubMed] [Google Scholar]
- 48. Jousselin E, Coeur d'Acier A, Vanlerberghe-Masutti F, Duron O. Evolution and diversity of Arsenophonus endosymbionts in aphids. Mol Ecol. 2013;22: 260–270. 10.1111/mec.12092 [DOI] [PubMed] [Google Scholar]
- 49. Novakova E, Hypsa V, Moran NA. Arsenophonus, an emerging clade of intracellular symbionts with a broad host distribution. BMC Microbiol. 2009;9: 143 10.1186/1471-2180-9-143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Bressan A. Emergence and evolution of Arsenophonus bacteria as insect-vectored plant pathogens. Infect Genet Evol. 2014;22: 81–90. 10.1016/j.meegid.2014.01.004 [DOI] [PubMed] [Google Scholar]
- 51. Benson MJ, Gawronski JD, Eveleigh DE, Benson DR. Intracellular symbionts and other bacteria associated with deer ticks (Ixodes scapularis) from Nantucket and Wellfleet, Cape Cod, Massachusetts. Appl Environ Microbiol. 2004;70: 616–620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. van Overbeek L, Gassner F, van der Plas CL, Kastelein P, Nunes-da Rocha U, Takken W. Diversity of Ixodes ricinus tick-associated bacterial communities from different forests. FEMS Microbiol Ecol. 2008;66: 72–84. 10.1111/j.1574-6941.2008.00468.x [DOI] [PubMed] [Google Scholar]
- 53. Duron O. Lateral transfers of insertion sequences between Wolbachia, Cardinium and Rickettsia bacterial endosymbionts. Heredity 2013;111: 330–337. 10.1038/hdy.2013.56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Baldo L, Werren JH. Revisiting Wolbachia supergroup typing based on WSP: spurious lineages and discordance with MLST. Curr Microbiol. 2007;55: 81–87. [DOI] [PubMed] [Google Scholar]
- 55. Degnan PH, Moran NA. Evolutionary genetics of a defensive facultative symbiont of insects: exchange of toxin-encoding bacteriophage. Mol Ecol. 2008;17: 916–929. 10.1111/j.1365-294X.2007.03616.x [DOI] [PubMed] [Google Scholar]
- 56. Nikoh N, Hosokawa T, Moriyama M, Oshima K, Hattori M, Fukatsu T. Evolutionary origin of insect-Wolbachia nutritional mutualism. Proc Natl Acad Sci USA. 2014;111: 10257–10262. 10.1073/pnas.1409284111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Duron O. Arsenophonus insect symbionts are commonly infected with APSE, a bacteriophage involved in protective symbiosis. FEMS Microbiol Ecol. 2014;90: 184–194. 10.1111/1574-6941.12381 [DOI] [PubMed] [Google Scholar]
- 58. Perlman SJ, Hunter MS, Zchori-Fein E. The emerging diversity of Rickettsia . Proc R Soc Lond B Biol Sci. 2006;273: 2097–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Weinert LA, Werren JH, Aebi A, Stone GN, Jiggins FM. Evolution and diversity of Rickettsia bacteria. BMC Biol. 2009;7: 6 10.1186/1741-7007-7-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Clayton AL, Oakeson KF, Gutin M, Pontes A, Dunn DM, von Niederhausern AC, et al. A novel human-infection-derived bacterium provides insights into the evolutionary origins of mutualistic insect–bacterial symbioses. PLoS Genet. 2012;8: e1002990 10.1371/journal.pgen.1002990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Thompson JD, Gibson TJ, Higgins DG. Multiple sequence alignment using ClustalW and ClustalX. Curr Protoc Bioinformatics 2002; 2: 3 10.1002/0471250953.bi0203s00 [DOI] [PubMed] [Google Scholar]
- 62. Kumar S, Tamura K, Nei M. MEGA3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform. 2004;5: 150–163. [DOI] [PubMed] [Google Scholar]
- 63. Castresana J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol. 2000;17: 540–552. [DOI] [PubMed] [Google Scholar]
- 64. Librado P, Rozas J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009;25: 1451–1452. 10.1093/bioinformatics/btp187 [DOI] [PubMed] [Google Scholar]
- 65.Sawyer SA. GENECONV: A computer package for the statistical detection of gene conversion. http://www.math.wustl.edu/~sawy.
- 66. Martin D, Rybicki E. RDP: detection of recombination amongst aligned sequences. Bioinformatics. 2000;16: 562–563. [DOI] [PubMed] [Google Scholar]
- 67. Martin D, Lemey P, Lott M, Moulton V, Posada D, Lefeuvre P. RDP3: a flexible and fast computer program for analyzing recombination. Bioinformatics. 2010;26: 2462–2463. 10.1093/bioinformatics/btq467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Huson DH, Bryant D. Application of phylogenetic networks in evolutionary studies. Mol Biol Evol. 2006;23: 254–267. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Genes and primers used in polymerase chain reaction (PCR) assays to detect Coxiella and relatives and to control tick DNA quality. The same primers were used for the Coxiella and Rickettsiella typing, with the exception of the 16S rRNA gene for which different primers were designed for the two bacteria. Nested PCR amplifications (16S rRNA, 23S rRNA, GroEL, rpoB and dnaK) were performed as follows: the first PCR run with the external primers was performed in a 10μLvolume containing 20–50 ng of genomic DNA, 3 mM of each dNTP (Thermo Scientific), 8 mM of MgCl2 (Roche Diagnostics), 3 μM of each primer, 1 μL of 10× PCR buffer (Roche Diagnostics), and 0.5 U of Taq DNA polymerase (Roche Diagnostics). A 1-μL aliquot of the PCR product from the first reaction was then used as a template for the second round of amplification. The second PCR was performed in a total volume of 25 μL and contained 8 mM of each dNTP (Thermo Scientific), 10 mM of MgCl2 (ThermoScientific), 7.5 μM of each of the internal primers, 2.5 μL of 10×PCR buffer (Thermo Scientific), and 1.25 U of Taq DNA polymerase (Thermo Scientific). Non-nested PCR amplifications (CO1 and 18S rRNA) were performed following conditions similar to the first PCR run used in the nested PCR assays. All PCR amplifications were performed under the following conditions: initial denaturation at 93°C for 3 min, 35 cycles of denaturation (93°C, 30 s), annealing (Tm = 50–56°C, depending on primers, 30 s), extension (72°C, 1–2 min), and a final extension at 72°C for 5 min. Table B. List, biological features and GenBank accession numbers of the bacterial strains used as references in molecular and phylogenetic analyses.* reference strains of C. burnetii used for primer testing. Table C. List, sequence accession numbers and features of the 31 Coxiella contigs from the whole-genome shotgun sequencing (WGS) of the cattle tick Rhipicephalus microplus. Fig A. Map of the Coxiella burnetii genome (strain Nine Mile I RSA 493) showing the position of the genetic markers (in blue) used in this study. The arrows indicate the position along the chromosome of the five housekeeping genes (16S rRNA, 23S rRNA, GroEL, rpoB and dnaK) used in the multi-locus typing of tick-borne Coxiella infections. The numbered boxes (1–31) indicate the position of the 31 Coxiella contigs (listed in Table C in S1 Text) detected from the whole genome sequencing of the hard tick Rhipicephalus microplus. Fig B. Coxiella and Rickettsiella phylogeny constructed using maximum-likelihood (ML) estimations based on 16S rRNA, 23S rRNA, GroEL, rpoB and dnaK concatenated sequences (3009bp), including 71 Coxiella-like strains of ticks, 15 C. burnetii reference strains and outgroups. The four Coxiella clades are labeled A to D. Each number corresponds to one tick species as detailed in Table 1. Blue, Coxiella-line organisms; red, C. burnetii; green, Rickettsiella; black, other bacteria. All multi-locus typing of tick-borne Coxiella and Rickettsiella of ticks are new sequences from this study. Branch numbers indicate percentage bootstrap support for major branches (1000 replicates; only bootstrap values >90% are shown). Fig C. Inset of Coxiella network from Fig 2 with focus on the A clade (Coxiella of soft ticks and C. burnetii). Each number corresponds to one tick species as detailed in Table 1. Blue, Coxiella-line organisms; red, C. burnetii. The scale bar is in units of substitution/site.
(DOCX)
Data Availability Statement
Nucleotide sequences of PCR-amplified fragments of tick-borne Coxiella and Rickettsiella genes have been deposited in the GenBank nucleotide database under accession codes KP994768-KP994862 (16S rRNA), KP994678-KP994767 (23S rRNA), KP985445-KP985537 (GroEL), KP985265-KP985357 (rpoB) and KP985358-KP985444 (dnaK).