Abstract
Objective:
To study the evolution of the neuropathy and long-term disability in a large cohort of patients with POEMS (polyneuropathy, organomegaly, endocrinopathy, M protein, and skin changes) syndrome following autologous stem cell transplantation (ASCT).
Methods:
Retrospective chart review documenting the clinical, electrophysiologic, and laboratory characteristics of patients with POEMS syndrome undergoing ASCT at Mayo Clinic, Rochester.
Results:
Sixty patients with a median follow-up time of 61 months were studied. All patients had peripheral polyneuropathy and demonstrated neurologic improvement after ASCT (apart from one patient who died early). Before ASCT, 27 patients (45%) required a wheelchair and 17 (29%) required a walker or foot brace. At the end of the follow-up period, no patient was using a wheelchair and 23 patients (38%) were using a foot brace. The median Neuropathy Impairment Score improved from 66 to 48 points at 12 months and to 30 points at most recent follow-up (p < 0.0001). Median Rankin Scale score improved from 3 to 1.5 (p < 0.0001). Vascular endothelial growth factor levels decreased from a median of 452 to 63.5 pg/mL (p < 0.0001). The ulnar compound motor action potential amplitude (median) improved from 4.3 to 7.6 mV (p < 0.0001) and ulnar compound motor action potential conduction velocity (median) improved from 34 to 51 m/s (p < 0.0001). Predicted forced vital capacity improved from 81% to 88% (p < 0.0001). Periengraftment syndrome occurred in 24 patients. Fourteen patients required additional chemotherapy and/or radiation following ASCT, but there was no clinical deterioration in the neuropathy in any of these patients. Six patients died: 1 due to POEMS, 1 due to failed engraftment, and 4 due to other malignancies (2 myelodysplastic syndrome, 1 lymphoma, 1 metastatic lung cancer).
Conclusion:
Patients with POEMS syndrome who undergo ASCT have a significant and meaningful improvement of their neuropathy by multiple measurements during both short and long-term follow-up, which corresponds to reduction in morbidity and disability (none are in wheelchair long-term). Periengraftment syndrome was common but manageable. Fatal complications, although rare, did occur, usually in association with other malignancies.
Classification of evidence:
This study provides Class IV evidence that for patients with POEMS syndrome, ASCT improves neuropathy-related function.
In patients with POEMS (polyneuropathy, organomegaly, endocrinopathy, M protein, and skin changes) syndrome, clinical and neurologic improvement with immunosuppressive medications or radiation therapy remains low (<50%) and alternative treatments are needed.1,2 Autologous stem cell transplantation (ASCT) seems to be a more promising treatment strategy.3 Although randomized controlled trials are not available, experts seem to agree that currently, the preferred treatment for POEMS syndrome is ASCT.4,5 This reasoning is based in part on the efficacy of ASCT in multiple myeloma.
Our group has recently published the long-term outcomes after ASCT in 59 patients with POEMS syndrome.3 The report focused on the hematologic, radiographic, and general clinical response to ASCT. It did not assess the details of the polyneuropathy. Furthermore, a review of the neurology literature reveals very limited information regarding the evolution of the neuropathy in patients with POEMS syndrome in general, and more specifically following ASCT. To date, the largest study on the subject included 9 patients with POEMS treated with ASCT who were prospectively followed over a median of 20 months.6 The results were very promising, showing neurologic improvements on all measures. The authors concluded that ASCT could be considered as a first-line treatment for appropriate candidates. An editorial accompanying the article praised the encouraging results but had reservation because of the small number of patients.7
We hence sought to evaluate the evolution of the neuropathy and long-term disability of a large cohort of patients undergoing ASCT at a single institution.
METHODS
Standard protocol approvals, registrations, and patient consents.
The Mayo Clinic institutional review board approved the study. We queried our bone marrow transplantation database to identify the 61 patients with POEMS syndrome who underwent high-dose chemotherapy followed by ASCT at the Mayo Clinic (Rochester, MN) between March 1999 and June 2012. Information was abstracted from medical records of patients who had given consent, allowing their medical records to be used for research purposes.
All patients were seen by neurologists and had detailed neurologic examination. Other patients with POEMS who did not undergo ASCT were excluded. Those patients either had radiation for localized disease, i.e., 1 to 3 plasmacytomas, or other therapies because of perceived ASCT ineligibility (i.e., no active disease at the time of evaluation, age 70 years, coexisting solid cancers, and severe dysfunction of single or multiple organ systems).
Hematologic details.
Transplantation details and definitions for response and relapse were previously defined.3 In general, peripheral blood stem cells were collected using either cyclophosphamide with growth factors, growth factors alone, or growth factors with plerixafor mobilizing agents. Details of patient conditioning before ASCT can be found in the prior published study.3 All patients were conditioned with high-dose melphalan. Posttransplantation-associated periengraftment syndrome was defined based on the modified Spitzer criteria, as follows:
Major criteria: a temperature of 38.3°C without an identifiable infectious etiology, rash involving 25% body surface area, and noncardiogenic pulmonary edema.
Minor criteria: bilirubin greater than twice normal, doubling of transaminases, weight gain 2.5% of baseline, or transient encephalopathy.
Periengraftment syndrome was defined as the presence of all 3 major criteria or 1 major and 2 minor criteria. The requirement for a neutrophil count of 0.5 × 106/cubic mL around 96 hours of the criteria was relaxed.3
Data collection.
Patients' demographics and neuropathy characteristics were collected. Neuropathy Impairment Score (NIS), modified Rankin Scale (mRS) score, vascular endothelial growth factor (VEGF), nerve conduction study (NCS), forced vital capacity (FVC), and maximal inspiratory and expiratory pressure were collected at t0, t1, and t2. T0 was defined as the time of testing before ASCT, t1 as the time of the first visit after ASCT, and t2 as the time of the last visit after ASCT. Prior chemotherapy ± radiation therapy, organomegaly (hepatomegaly or splenomegaly), endocrinopathies, skin changes, fluid extravasation, M protein, thrombocytosis, CNS involvement, and engraftment syndrome were also recorded.
Neuropathy and disability evaluation.
The NIS was calculated from the Mayo Clinic standard neurologic examination sheet. In brief, the NIS is a summed score of a standard and representative list of motor, sensory, and muscle stretch reflex impairments. Scores range from 0 (normal) to 4 (paralyzed) for motor, and 0 to 2 (absent) for sensation and reflexes, for individual attributes.8 The higher the NIS, the worse the neuropathy. For example, a NIS of 188 meant the worst score possible with complete paralysis and loss of sensation and reflexes in all 4 limbs with facial and respiratory involvements. We recorded the NIS prior at t0, t1, and t2. We recorded the mRS score from the history and correspondence with patients. The mRS measures the degree of disability in daily living. The score ranges from 0 (no symptoms) to 6 (dead); 1 = some symptoms without disability; 2 = slight disability with inability to perform all previous activities; 3 = moderate disability requiring some help, but able to walk unassisted; 4 = moderately severe disability with inability to walk unassisted; and 5 = severe disability with need for constant nursing care and attention and bedridden. Because the mRS can be obtained without a detailed neurologic assessment, i.e., the patient not necessarily evaluated by a neurologist, these scores were available at a longer follow-up time than the NIS. We also reviewed the NCS and EMG. Compound motor action potentials (CMAPs) obtained from tibial nerve stimulation recording from the adductor hallucis, the fibular nerve stimulation recording from the extensor digitorum brevis, median nerve stimulation recording from the abductor pollicis brevis, and ulnar nerve stimulation recording from the adductor digiti minimi were noted at t0, t1, and t2. The ulnar CMAP was chosen for analysis because the median CMAP was not consistently recorded and the fibular and tibial CMAPs were frequently absent, even following treatment. The polyneuropathy was classified as demyelinating according to the American Academy of Neurology research criteria,9 as mixed when there was both demyelination and axonal loss, or as predominately axonal. Autonomic reflex studies were available on a subset of patients and were often not repeated following treatment.
Statistical analysis.
Descriptive summaries were presented as frequencies and percentages for categorical variables and median and ranges for continuous variables. Data for outcomes of interest were displayed using side-by-side box plots and/or scatterplots. Associations between continuous outcomes were assessed using Pearson correlation coefficient. Kruskal–Wallis test or Wilcoxon rank sum test was used to compare distribution of continuous outcomes (i.e., NIS at various time points) across categorical variables such as type of bone lesion and NIS or number of features, etc. All of the tests were 2-sided and p values <0.05 were considered statistically significant. All analyses were performed using SAS version 9.3 (SAS Inc., Cary, NC).
Research question.
The primary research question concerns the qualitative evaluation of the short- and long-term treatment response of neuropathy-related function following ASCT in patients with POEMS syndrome. In this design, this will result in Level IV evidence.
RESULTS
Sixty-one patients with POEMS syndrome were identified as having undergone ASCT. One patient who did not have a formal neurologic assessment by a neurologist was excluded. Therefore, 60 patients were included in the analysis. Hematologic but not neurologic outcome has been previously reported in 58 of these patients.3
Baseline characteristics.
Baseline characteristics are summarized in table 1. In 7 patients (12%), no monoclonal protein was detected on serum protein electrophoresis or serum or urine immunofixation (table 1). Six of those 7 patients had prior chemotherapy. Of those 7 patients, 3 had free light chain in the serum. The remaining 4 patients' light chain was determined on bone marrow biopsy. Neuropathy characteristics are summarized in table 2. Twenty-seven patients (45%) were using a wheelchair, 10 patients (17%) were using a walker, and 7 patients (12%) were using ankle foot orthoses. The severity of the neuropathy was independent of the use of prior radiation or chemotherapy (p = 0.1), the number of bone lesions (p = 0.6), bone lesion type (p = 0.6), the number of organs involved (features of POEMS) (p = 0.06), or VEGF level (figure e-1 on the Neurology® Web site at Neurology.org).
Table 1.
Patient demographics at baseline (t0)
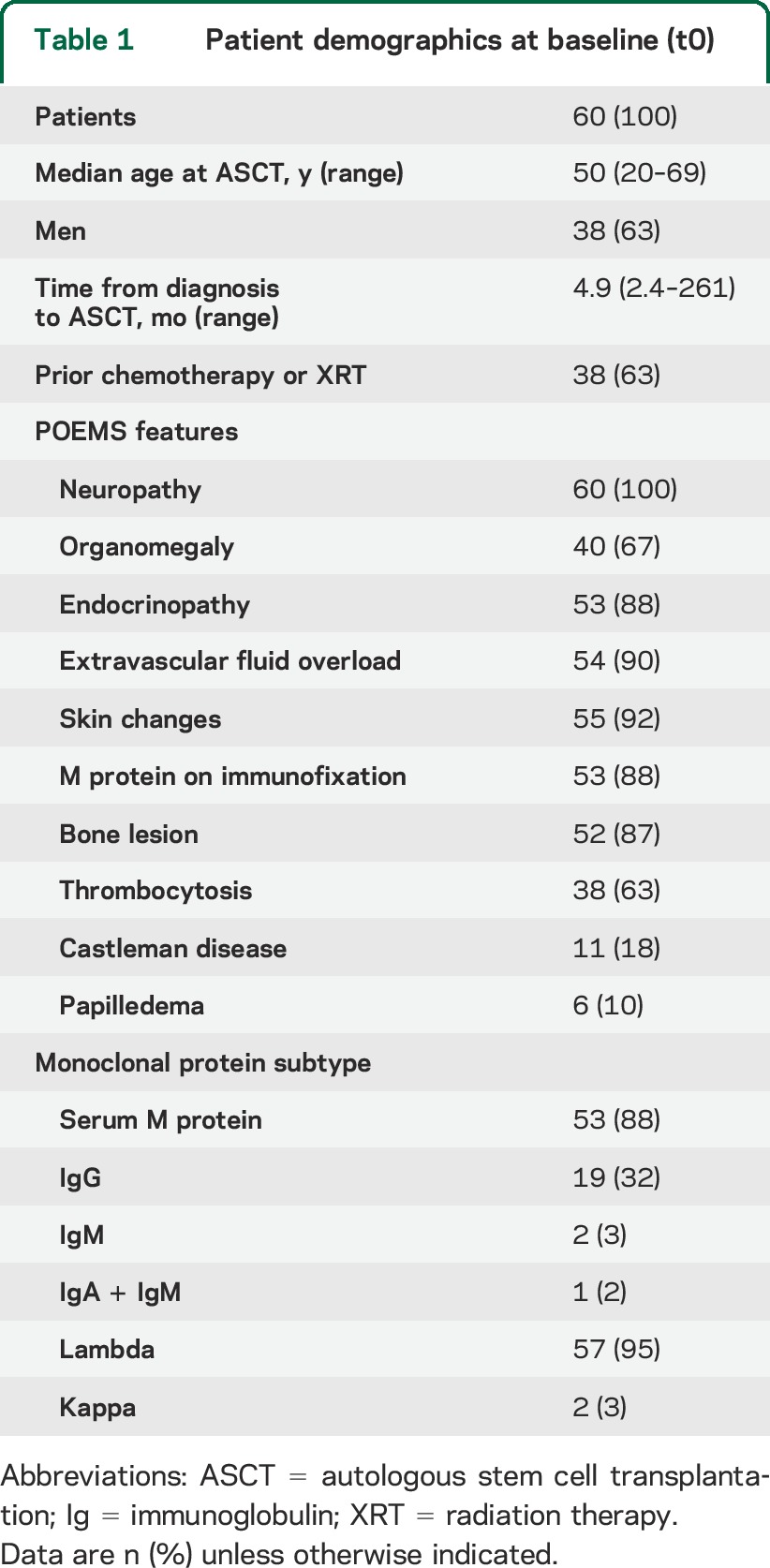
Table 2.
Neuropathy characteristics
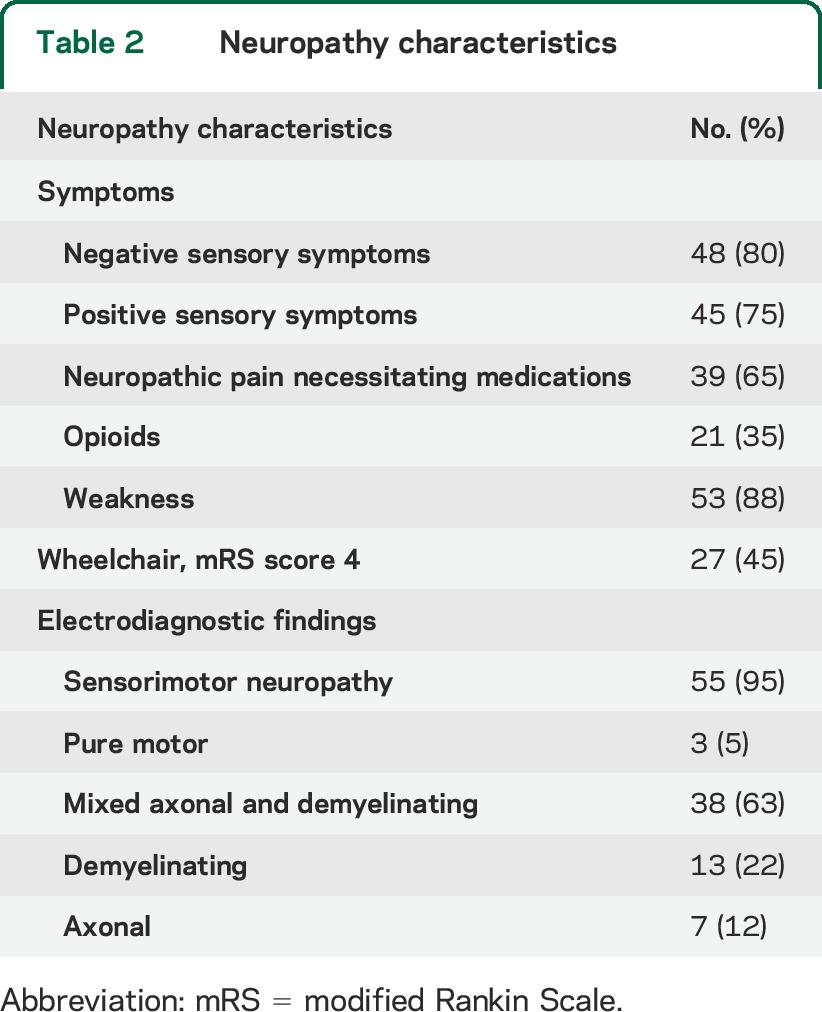
Fifty-eight patients underwent NCS and EMG (table 2). Those studies demonstrated sensory and motor involvement in 55 patients (95%). There were demyelinating changes in 51 patients (88%). Only 2 patients (3%) had autonomic symptoms (excluding erectile dysfunction). However, subclinical autonomic abnormalities were found in 11 of the 20 patients (55%) tested (mild to moderate abnormalities as follow: 9 patients had impaired cardiovagal function, 4 had impaired postganglionic cholinergic sudomotor function, and 4 had impaired cardioadrenergic function).
The cohort demonstrated marked disability. At t0, 27 patients (45%) were using a wheelchair (mRS score 4), 24 patients (40%) had an mRS score of 3, 7 patients (12%) had an mRS score of 2, and only 2 patients (3%) had an mRS score of 1. Eight patients had a prior stroke. Six patients had papilledema. Six patients had headache; 2 of those had a lumbar puncture with elevated opening pressure (26 and 43 cm Hg, respectively).
Outcomes.
Follow-up was available on all 60 patients. Median final follow-up after ASCT was 61 months (range 4–162 months). The median follow-up at t1 was 12 months (range 0–40 months) for both NIS and mRS. The median follow-up time at t2 for most recent NIS was 49 months (range 3–127 months) and the median follow-up time at t2 for most recent mRS was 61 months (range 9–162 months). The disability in all but one patient improved after treatment (figures 1 and 2). In the only patient who did not improve, the worsening of his NIS and mRS was related to a complicated posttransplant course, with sepsis necessitating intensive care unit admission. That patient died at 4 months posttransplant. After ASCT, there was a marked improvement in NIS score at first visit (t1) followed by a further improvement in NIS score at last visit (t2) (median 49 months) (figure 1). NIS improved from 66 points at t0 to 48 points at t1 and 30 points at t2 (p < 0.0001). There was significant correlation between the improvement in NIS scores and level of VEGF at t1 (p < 0.0001). The mRS score decreased from 3.0 at t0 to 2.0 at t1 (p < 0.0001) and from 2.0 to 1.5 at t2 (p = 0.25) (figure 2). At the end of the follow-up, no patient was using a wheelchair, 3 patients (5%) were using a walker, 7 patients (12%) were using a cane or crutch, and 23 patients (38%) were using ankle foot orthoses.
Figure 1. Improvement in NIS following autologous stem cell transplantation.
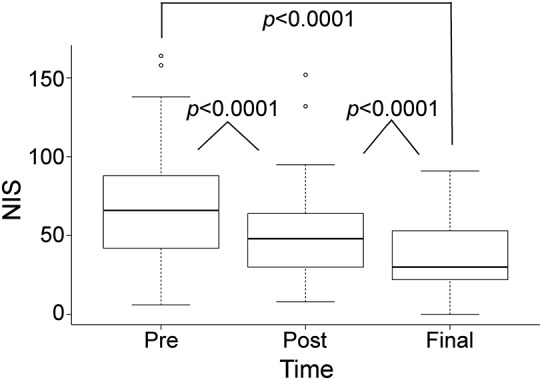
Median NIS improved from 66 points at t0 to 48 points at t1 and 30 points at t2 (p < 0.0001). Box = 25th and 75th percentiles; bars = minimum and maximum values. NIS = Neuropathy Impairment Score.
Figure 2. Improvement in mRS score following autologous stem cell transplantation.
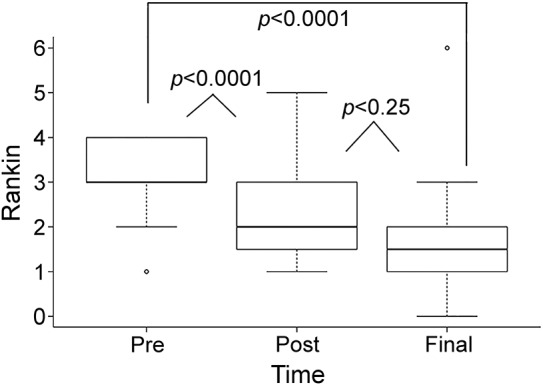
Median mRS score decreased from 3.0 at t0 to 2.0 at t1 (p < 0.0001) and from 2.0 to 1.5 at t2 (p = 0.25). Box = 25th and 75th percentiles; bars = minimum and maximum values. mRS = modified Rankin Scale.
There was no correlation between the duration of the disease before ASCT and the severity of the neuropathy or the response to treatment. However, when patients with a lag of more than 2 years to ASCT were excluded, there was positive correlation between time to treatment and neuropathy severity (NIS score) at baseline (Pearson correlation coefficients 0.32) as well as a negative correlation between time to treatment and neuropathy improvement (NIS score) following treatment (Pearson correlation coefficients −0.4 and −0.24 at t1 and t2, respectively). There was no difference in outcome between patients with predominantly axonal vs those with predominantly demyelinating neuropathy (p = 0.2), although the number of primarily axonal neuropathies was small (n = 7) and so power may be insufficient to see a difference. Disease duration was similar in patients with predominantly axonal vs those with predominantly demyelinating neuropathy (p = 0.5).
FVC increased from 81% of predicted value to 88% of predicted value (p < 0.0001) at t1 (median 12 months). Mean inspiratory pressure increased from 71 to 90 (p < 0.0001) and mean expiratory pressure increased from 59 to 72 (p = 0.004). VEGF levels decreased from a median of 452 pg/mL at t0 to 52 pg/mL at t1 (p < 0.0001) and from 74 to 63.5 pg/mL at t2 (p = 0.45) (figure e-2). Median ulnar CMAP amplitude improved from 4.3 mV at t0 to 6.5 mV at t1 (p < 0.0001) and from 6.5 to 7.6 mV at t2 (p < 0.0001) (figure 3). Median ulnar CMAP conduction velocity improved from 34 m/s at t0 to 43 m/s at t1 (p < 0.0001) and 51 m/s at t2 (p < 0.0001) (figure 3). Only one patient had a fibular–extensor digitorum brevis CMAP that could be recorded and only 3 patients had a tibial–abductor hallucis CMAP that could be recorded. After treatment, 5 fibular–extensor digitorum brevis and 6 tibial–abductor hallucis CMAPs were recordable. These CMAPs were not included in the statistical analysis because of small sample size. Autonomic symptoms improved in both patients at t1.
Figure 3. Improvement in ulnar CMAP amplitude and conduction velocity following autologous stem cell transplantation.
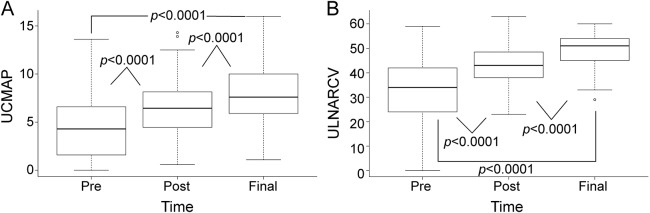
(A) Ulnar CMAP amplitude improved from a median of 4.3 mV at t0 to 6.5 mV at t1 and from 6.5 to 7.6 mV at t2 (p < 0.0001). (B) Ulnar CMAP conduction velocity improved from a median of 34 m/s at t0 to 43 m/s at t1 and 51 m/s at t2 (p < 0.0001). Box = 25th and 75th percentiles; bars = minimum and maximum values. CMAP = compound motor action potential; UCMAP = ulnar CMAP; ULNARCV = ulnar conduction velocity.
Morbidity and mortality.
Engraftment syndrome occurred in 24 patients (40%). Engraftment syndrome did not affect the NIS at t0, t1, or t2 (p = 0.6, 0.8, and 0.6, respectively). Six patients died during follow-up. One patient died of recurrent POEMS syndrome. One patient had graft failure and died early. The other 4 died of other malignancies (2 myelodysplastic syndrome, 1 lymphoma, and 1 metastatic lung cancer). All of these patients had improvement of their neuropathy apart from the one who died early. Eight other patients had progression or recurrence of their disease—either biochemical or radiographic, yet their NIS and mRS score continued to improve. Some of these patients were observed without intervention; others had radiation therapy and/or chemotherapy. These patients are discussed in detail in the prior report.3
DISCUSSION
Before the availability of ASCT, earlier treatments for POEMS syndrome with chemotherapy yielded a limited improvement of the neuropathy with <50% responding and a high mortality with 30% of patients dying within 5 years. With ASCT, there is a 5-year overall survival of 94% as well as good response to salvage chemotherapy in the case of radiologic or VEGF progression.3 Our study provides large-scale confirmation that ASCT improves the peripheral neuropathy, which is the major driver of the associated morbidity.
In general, patients with POEMS syndrome present with weakness, numbness, tingling, and pain in the lower extremities secondary to a length-dependent, typically mixed axonal and demyelinating polyneuropathy.10 These characteristics can cause frequent misdiagnoses with other demyelinating neuropathies, most frequently chronic inflammatory demyelinating polyradiculoneuropathy. The delay in diagnosis, as we have shown here, remains a significant issue. When patients with longer than 2 years between diagnosis and ASCT were excluded, those with longer times to diagnosis were more severely affected and response to treatment was less pronounced. Atypical features such as distal more than proximal lower limb weakness, pain, and dyspnea are more common in POEMS syndrome and help to differentiate these patients from those with chronic inflammatory demyelinating polyradiculoneuropathy. In patients with a demyelinating neuropathy, a monoclonal protein should be sought because 88% and 95% of patients with POEMS syndrome have a monoclonal protein and a lambda light chain, respectively, as shown here. The presence of an elevated VEGF and other systemic features confirm the diagnosis. Note that VEGF can be suppressed by prior immune treatment (e.g., steroids). Additional testing, such as screening for endocrinopathies, and skeletal survey and/or PET scan and eventually tissue analysis of a sclerotic lesion or bone marrow, confirms the diagnosis.
In patients with POEMS syndrome treated with ASCT, neurologic improvements often lag behind other aspects of the disease such as improved VEGF levels and extravascular volume overload reductions. However, we demonstrated here that the neuropathy and disability continued to improve with time after ASCT. A major finding of this study is that, while almost half of the patients needed a wheelchair for ambulation, within a year following ASCT all but one were able to walk and at the last follow-up, none were using a wheelchair. This illustrates the dramatic response to ASCT in patients with POEMS syndrome.
In the earlier study, VEGF levels correlated well with the clinical response.3 In our series, there was no correlation between the severity of the neuropathy and the VEGF level at baseline. However, the drop in VEGF levels correlated well with the improvement in neuropathy following ASCT. Of note, although an increasing VEGF level predicts radiographic progression as measured by fluorodeoxyglucose avidity on PET scan,3 patients may be stable neurologically or continue to improve despite POEMS recurrence.
Respiratory involvement is frequent in patients with POEMS syndrome.11 Respiratory muscle weakness and cough portend a poorer prognosis. Almost half of the patients with POEMS syndrome studied here had respiratory failure. A novel finding in our series is the improvement in FVC, maximal inspiratory pressure, and maximal expiratory pressure with ASCT.
ASCT can be performed safely in patients with POEMS syndrome. While ASCT was associated with a high incidence of periengraftment syndrome in this cohort, this was usually well managed with short-term steroid treatment. The presence of a periengraftment syndrome did not seem to affect the overall improvement of the neuropathy and disability. There are case reports that conditioning with lenalidomide decreases the rate of engraftment syndrome.12 This is a promising approach that needs further study. Mortality related to ASCT was low. Only one patient of 60 died directly as a result of ASCT and chemotherapy. This mortality rate is similar or even lower than what is observed in ASCT performed in other malignant conditions.13 Furthermore, this mortality rate is much lower than what is observed in patients with POEMS syndrome treated with steroids and melphalan alone.
The retrospective nature of this study did not allow comparison of neuropathy outcomes with a control group receiving a standard alkylator-based treatment or other immunomodulatory drugs. In addition, there was potential for selection bias in that younger and healthier patients may be selected to undergo transplantation over an alternative treatment. Hence, this study provided Class IV evidence that for patients with POEMS syndrome, ASCT improves neuropathy-related function.
ASCT universally improved neurologic symptoms, deficits, and necessity for gait aids in patients with POEMS syndrome. The degree of neurologic improvement was large and was sustained over the length of the study. Although periengraftment syndrome was common, mortality remains much lower than in patients treated with steroids and melphalan only. Newer chemotherapeutic agents such as lenalidomide are promising, and prospective randomized clinical trials with comparison to ASCT would determine the best approach for patients with POEMS syndrome.14 Such trials are difficult because of the rarity of the disease. Until then, management is mainly dictated by the overall performance status of the patient, response to prior treatments, and experience with other similar conditions such as multiple myeloma.
Supplementary Material
GLOSSARY
- ASCT
autologous stem cell transplantation
- CMAP
compound motor action potential
- FVC
forced vital capacity
- mRS
modified Rankin Scale
- NCS
nerve conduction study
- NIS
Neuropathy Impairment Score
- POEMS
polyneuropathy, organomegaly, endocrinopathy, M protein, and skin changes
- VEGF
vascular endothelial growth factor
Footnotes
Supplemental data at Neurology.org
AUTHOR CONTRIBUTIONS
Chafic Karam and Michelle L. Mauermann: conception and design of the study, acquisition, analysis, and interpretation of data, drafting of the paper, and are accountable for the study. Christopher J. Klein, Angela Dispenzieri, P. James B. Dyck, Jay Mandrekar, and Anita D'Souza: analysis and interpretation of the data, revising the paper critically for important intellectual content. All authors have given final approval of the version to be submitted.
STUDY FUNDING
This work was supported in part by the Robert A. Kyle Hematologic Malignancies Fund, the Predolin Foundation, the JABBS Foundation, the Andrew and Lillian A. Posey Foundation, and NIH K08 NS065007 (C.J.K.).
DISCLOSURE
C. Karam serves as deputy editor, WriteClick®: Neurology®. C. Klein, A. Dispenzieri, P. Dyck, J. Mandrekar, A. D'Souza, and M. Mauermann report no disclosures relevant to the manuscript. Go to Neurology.org for full disclosures.
REFERENCES
- 1.Dispenzieri A, Kyle RA, Lacy MQ, et al. POEMS syndrome: definitions and long-term outcome. Blood 2003;101:2496–2506. 10.1182/blood-2002-07-2299. [DOI] [PubMed] [Google Scholar]
- 2.Humeniuk MS, Gertz MA, Lacy MQ, et al. Outcomes of patients with POEMS syndrome treated initially with radiation. Blood 2013;122:68–73. 10.1182/blood-2013-03-487025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.D'Souza A, Lacy M, Gertz M, et al. Long-term outcomes after autologous stem cell transplantation for patients with POEMS syndrome (osteosclerotic myeloma): a single-center experience. Blood 2012;120:56–62. 10.1182/blood-2012-04-423178. [DOI] [PubMed] [Google Scholar]
- 4.Kuwabara S, Dispenzieri A, Arimura K, Misawa S, Nakaseko C. Treatment for POEMS (polyneuropathy, organomegaly, endocrinopathy, M-protein, and skin changes) syndrome. Cochrane Database Syst Rev 2012;6:CD006828. 10.1002/14651858.CD006828.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dispenzieri A. POEMS syndrome: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol 2014;89:214–223. 10.1002/ajh.23644. [DOI] [PubMed] [Google Scholar]
- 6.Kuwabara S, Misawa S, Kanai K, et al. Neurologic improvement after peripheral blood stem cell transplantation in POEMS syndrome. Neurology 2008;71:1691–1695. 10.1212/01.wnl.0000323811.42080.a4. [DOI] [PubMed] [Google Scholar]
- 7.Chalk C. Making the lame walk? Transplantation for POEMS. Neurology 2008;71:1658–1659. 10.1212/01.wnl.0000326574.37138.35. [DOI] [PubMed] [Google Scholar]
- 8.Dyck PJ, Hughes RAC, O'Brien PC. Quantitating overall neuropathic symptoms, impairments and outcomes. In: Dyck PJ, Thomas PK, editors. Peripheral Neuropathy, 4th ed Philadelphia: Elsevier Saunders; 2005:1031–1053. [Google Scholar]
- 9.Research criteria for diagnosis of chronic inflammatory demyelinating polyneuropathy (CIDP). Report from an Ad Hoc Subcommittee of the American Academy of Neurology AIDS Task Force. Neurology 1991;41:617–618. [PubMed] [Google Scholar]
- 10.Mauermann ML, Sorenson EJ, Dispenzieri A, et al. Uniform demyelination and more severe axonal loss distinguish POEMS syndrome from CIDP. J Neurol Neurosurg Psychiatry 2012;83:480–486. 10.1136/jnnp-2011-301472. [DOI] [PubMed] [Google Scholar]
- 11.Allam JS, Kennedy CC, Aksamit TR, Dispenzieri A. Pulmonary manifestations in patients with POEMS syndrome: a retrospective review of 137 patients. Chest 2008;133:969–974. 10.1378/chest.07-1800. [DOI] [PubMed] [Google Scholar]
- 12.Zagouri F, Kastritis E, Gavriatopoulou M, et al. Lenalidomide in patients with POEMS syndrome: a systematic review and pooled analysis. Leuk Lymphoma 2014;55:2018–2023. 10.3109/10428194.2013.869329. [DOI] [PubMed] [Google Scholar]
- 13.Jantunen E, Itala M, Lehtinen T, et al. Early treatment-related mortality in adult autologous stem cell transplant recipients: a nation-wide survey of 1,482 transplanted patients. Eur J Haematol 2006;76:245–250. 10.1111/j.1600-0609.2005.00605.x. [DOI] [PubMed] [Google Scholar]
- 14.Royer B, Merlusca L, Abraham J, et al. Efficacy of lenalidomide in POEMS syndrome: a retrospective study of 20 patients. Am J Hematol 2013;88:207–212. 10.1002/ajh.23374. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


