Abstract
The tumor microenvironment counters antitumor T cell responses in part by blunting their activation and infiltration. Ligands that engage Toll-like receptors (TLR) on T cells and antigen-presenting cells can act as potent immune adjuvants. In this study, we show how tumor-reactive T cells engineered to secrete bacterial flagellin, a TLR5 ligand (TLR5L), can engender a co-stimulatory signal that augments antitumor activity. Human T cells engineered to express TLR5L along with DMF5, a T cell receptor that recognizes the melanoma antigen MART-127-35 (DMF5TLR5L T cells), displayed increased proliferation, cytokine production and cytolytic activity against melanoma cells. In a xenogenetic model, adoptive transfer of DMF5TLR5L T cells reduced tumor growth kinetics and prolonged mouse survival. In a syngeneic model, similarly engineered melanoma-reactive T cells (pmelTLR5L) displayed a relative increase in antitumor activity against established tumors, compared to unmodified T cells. In this model, we documented increased T cell infiltration associated with increased levels of CCR1 and CXCR3 levels on T cells, a reduction in PD1+Lag3+ T cells and CD11+Gr1+ myeloid-derived suppressor cells, and changes in the chemokine/cytokine profile of tumors. Our findings show how T cell-mediated delivery of a TLR agonist to the tumor site can contribute to antitumor efficacy, in the context of adoptive T cell immunotherapy.
Keywords: TLR5 ligand, T cell immunotherapy, TCR transgenic T cells, melanoma
Introduction
Recent clinical trials highlight the potential of T cell–based immunotherapies in treating cancer patients, including individuals with melanoma(1, 2). Several approaches have been developed that harness the T cell's ability to destroy cancer, including vaccine–based strategies and adoptive T cell transfer (ACT) protocols using tumor-infiltrating lymphocytes or T cells engineered to express tumor-reactive T cell receptors (TCRs)(2). The use of antibodies (Ab) to block inhibitory receptors on T cells, such as Ipilimumab (anti-CTLA4) and anti-PD-L1 Abs have also demonstrated an ability to potentiate T cell responses against various cancer types including melanoma (1, 3-7). Despite encouraging results demonstrating objective responses in some cancer patients, only a subset of patients experience the medical benefits of this form of therapy, highlighting the need to optimize T cell cancer therapies.
The antitumor efficacy of T cell–based strategies is limited by various processes. One obstacle includes insufficient recruitment of T cells to the tumor due to the lack of specific chemokines or chemokine receptors on T cells. In fact, the introduction of CXCR2 into tumor-reactive T cells has been shown to increase T cell tumor infiltration and antitumor responses (8). Upon T cell infiltration, the expression of weakly immunogenic tumor antigens as well as the down-regulation of major histocompatibility complex (MHC) I expression on tumor cells poses another barrier in achieving maximal T cell activation. The presence of immunosuppressive cells such as CD4+ Tregs and myeloid-derived suppressor cells (MDSCs) can further restrain antitumor T cell responses (9, 10). Finally, the expression of a variety of inhibitory factors such as PD-L1, CD155, and HVEM on tumor cells or other cells in the tumor environment, also counter the generation of productive T cell responses (11, 12). Hence, the optimal activation of T cells in the tumor environment may require a continuous source of costimulatory signals, not typically found on tumors, that could reshape the environment toward one more supportive of T cell activation and survival.
TLR engagement on T cells has been demonstrated to function as a potent costimulatory signal and represents a promising strategy to enhance the efficacy of cancer immunotherapies. Studies from several groups including ours demonstrate that TLR engagement on CD4+ or CD8+ T cells enhances cell division, survival and cytotoxicity (13-18). The costimulatory effect of TLR signaling is partially associated with the ability to reduce the TCR activation threshold to weakly immunogenic tumor antigens (16, 19). However, because the costimulatory effects on T cells are dependent on concurrent TCR stimulation, effective antitumor responses generally occur when TLR ligand (TLRL) is administered peritumorally or intratumorally (16). Systemic administration of TLRL alone results in no or weak antitumor activity.
Given the lack of costimulatory signals in the tumor environment and the milieu of factors that counter effective antitumor T cell responses, we examined whether T cells engineered to secrete the TLR5L flagellin at the tumor site would generate a costimulatory tumor environment and restore antitumor activity. Flagellin, a bacterial protein, is the only known TLR5L and is one of the few TLR agonists that can be engineered to be expressed and secreted by T cells (the majority of known TLR agonists consist of lipids or nucleic acids) (20). Flagellin is well tolerated and relatively safe in preclinical models and is currently under investigation in Phase I studies (NCT01527136), however, it has been reported to have a short half-life of approximately 10 minutes (21, 22). The studies described here demonstrate that tumor-reactive T cells engineered to deliver the TLR5L to the tumor site show potent antitumor responses as compared with mice treated with T cells. Importantly, intratumoral delivery of TLR5L by T cells reshaped the tumor environment and included changes in the chemokine/cytokine profile, increased expression of chemokine receptors on T cells and increased tumor infiltration. Interestingly, TLR5L–secreting T cells reduced the numbers and phenotype of MDSCs. These findings provide the first characterization of the effects of intratumoral TLR5L delivery by T cells.
Materials and Methods
Mice and cell lines
NOD-Scid IL-2Rgammanull (NSG) and pmel mice were obtained from the Jackson Laboratory, and C57BL/6 mice were purchased from Charles River Laboratories. All experiments have been reviewed and approved by the Institutional Animal Care and Use Committee. HLA-A2+/MART-1+ Malme-3M cells were maintained in Iscove's modified Dulbecco's medium, and HLA-A2+/MART-1+ C32, HLA-A2+/MART-1- A375, B16, Phoenix Ampho and Eco packaging cell lines in Dulbecco's modified Eagle's medium, supplemented with fetal bovine serum and penicillin-streptomycin.
T-cell sorting and functional studies
Peripheral blood mononuclear cells (PBMCs) from healthy donors were purchased from Biological Specialty Corp, and isolated by Ficoll-Paque (GE Healthcare) density gradient centrifugation. CD4+ and CD8+ T cells were purified by negative selection using a Dynabeads Untouched Human CD4 or CD8 T Cells kit according to the manufacturer's protocol (Invitrogen). 2×105 T cells were cultured in 96-well round-bottom plates coated with 1 μg/mL OKT3 (eBioscience) in the presence or absence of TLR5L (100 ng/mL, InvivoGen) for 96 hours. Sixteen hours before harvesting, 0.5 μCi of 3H-thymidine was added to each well prior to measuring thymidine uptake using a 1450 LSC & luminescence counter (PerkinElmer). Cytokine and chemokine production levels were measured from culture supernatants collected 96 hours after stimulation using a Cytokine/Chemokine kit (Millipore) according to manufacturer's instructions. All tests were performed in triplicate wells and results are shown as mean ±SD.
Construction of retroviral vector
Figure 1B shows schematically the structure of our vectors used in this study, and the order of placement of components in-frame from the 5’ to the 3’ ends. The human IL-2 signal peptide sequence was linked to the cDNA coding sequence of Salmonella typhimurium flagellin (GenBank accession no. D13689). 6 changes were made in the flagellin coding sequence to eliminate predicted N-linked glycosylation sites as described by Applequist et al. (23). The sequence was synthesized by GenScript, confirmed by DNA sequencing and then cloned into retroviral vector pMSGV1-MART-1-TCR, downstream of MART-1 TCR sequence, using an IRES sequence upstream of flagellin. The sequence was also inserted into pMSGV1-IRES-GFP to yield pMSGV1-TLR5L-IRES-GFP. We used the TCR specific to MART-127-35 kindly provided by Dr. Laura Johnson at the University of Pennsylvania (24).
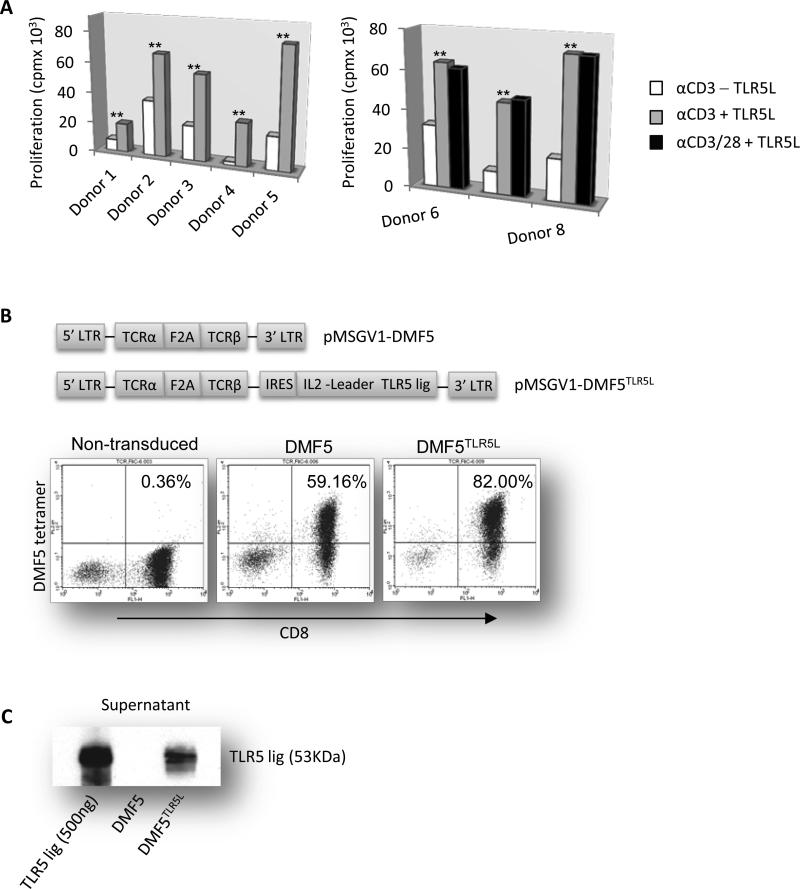
Figure 1. TLR5 stimulation augments the proliferation of human CD8+ T cells and engineered T cells express MART-1 TCR and TLR5L.
(A) Purified CD8+ T cells were activated with 1 μg/mL of plate-bound OKT3 with or without 100 ng/mL TLR5L. Four days later, proliferation was determined by 3H-thymidine uptake. PBMCs from eight healthy donors were examined. Data are presented as mean ±S.D; *p<0.05, **p<0.01, T-test. (B) Structure of retroviral vectors pMSGV1-DMF5 TCR, and pMSGV1-DMF5TLR5L using an intraribosomal entry site (IRES) to allow for dual gene expression. DMF5 TCR expression on the surface of transduced human T cells was determined by staining cells with CD8 and MART-1 tetramer and analyzed by flow cytometry. (C) TLR5L secretion from transduced T cells was detected by Western blot. Purified flagellin was used as a positive control. Data shown are representative of three or more independent experiments.
Retrovirus production and transduction of human and mouse T cells
Retroviral vector supernatants were produced from Phoenix Ampho and Eco packaging cell lines. Retrovirus production was initiated by transfecting the cells with plasmid DNA using the Lipofectamine 2000 transfection reagent (Invitrogen). 48 hours later, supernatants were collected and used to transduce human and mouse T cells. For transduction of human T cells, PBMCs were cultured at 3×106 per well in 24-well tissue culture plates in AIM V medium (GIBCO brand; Invitrogen) supplemented with 5% human AB serum (Sigma-Aldrich), 1% MEM non-essential amino acids, 1% penicillin-streptomycin and 100 U/ml recombinant human IL-2 (BioLegend), and activated with 50 ng/ml OKT3 for 48 hours. For transduction, 24-well non-tissue culture treated plates (BD Biosciences) were coated with 0.5 ml per well of 10 μg/ml recombinant human fibronectin fragment (RetroNectin, Takara Bio) overnight at 4°C. After incubation, 2 ml of retroviral supernatant was added to each coated well followed by centrifugation at 2000 × g for 2 hours at 32°C. 1.5 ml of viral supernatant was removed, and 1×106 (0.5 ml) activated PBMCs were added to each well in the presence of 100 U/ml IL-2. Plates were centrifuged at 1000 × g for 10 min, and then incubated overnight at 37 °C. Cells were washed and maintained in the presence of 100 U/ml IL-2. On day 8, cells were expanded with beads coated with HLA-A2/MART-1:27-35 and anti-CD28 antibody plus 100 U/ml IL-2 for 1 to 2 weeks. Transduction of mouse T cells was performed as previously described (16, 25).
Western blot
To determine TLR5L secretion from transduced T cells, culture supernatants were harvested, concentrated using Vivaspin 20 centrifugal concentrators (Sartorius Stedim Biotech), electrophoresed by SDS-PAGE and transferred to a PVDF membrane. Blots were incubated sequentially with rabbit anti-flagellin antibody (a kind gift from Dr. Jim Kaper at the University of Maryland, Baltimore) and horseradish peroxidase-conjugated anti-rabbit antibody, and detected using enhanced chemiluminescence (ECL Plus; Amersham Pharmacia Biotech).
Flow cytometry
The surface expression of MART-1 TCR on transduced human T cells was determined by flow cytometry. Cells were washed with FACS buffer (PBS plus 0.2% BSA and 0.1% sodium azide), stained with FITC-conjugated CD8 (BD Biosciences) and PE-conjugated HLA-A2/MART-1:27-35 tetramer (a gift from NIH Tetramer Facility at Emory University). Appropriate isotype controls were used for flow cytometry analysis. In some experiments 6- to 8-wk-old male NSG mice were injected s.c. with 5×106 Malme-3M tumor cells. Tumor was allowed to grow to 10mm2 and mice were injected intravenously (i.v.) with 2.5×106 non-transduced and 2.5×106 transduced human T cells. 3-5 days later, tumor samples were collected from 3 mice per group; single cell suspensions were prepared and stained with CD8-FITC/MART-1 tetramer-PE or HLA-DR, DP, DQ-FITC/CD11C-PE. Cells stained with relevant isotype control antibodies were used as isotype controls. In some experiments C57BL/6 mice were injected (s.c.) with 1×105 B16-F1 cells. On day ten post tumor cell injection, mice were exposed to irradiation (400 cGy), and injected (i.v.) with 2×106 pmel T cells. Mice were euthanized when tumors reached a size of approximately 50-100 mm2. Tumor and spleen were harvested. Briefly, organs were processed through a cell strainer and washed in PBS 2% FBS (Gemini) 1% PenStrep 1% NEAA (Gibco, Life Technologies). Spleen samples were treated with red blood cell lysis buffer (BioLegend) before washing. Cells were stained at 1×106 cells per well in 96-well plate format for 2 hours at 4°C in 100 μL FACS Buffer (1X PBS 5% FBS 0.1% NaN3). The following antibodies were used: CD90.1-APC, PD1-PE, CXCR3-APC (eBioscience), Lag3-PerCP, CD11b-PerCP, Gr1-APC (BioLegend), CD8-FITC, IAb-FITC, CD86-FITC, CD80-FITC (BD Pharmingen), H2Kb/H2Db-PE (Abcam), CCR1-PE (R&D Systems). Cells were washed twice in FACS Buffer and analyzed by flow cytometry. All flow cytometry was performed at the University of Maryland Greenebaum Cancer Center Flow Cytometry Shared Services on the BD LSR II and high throughput sampler (HTS). Flow cytometry acquisition was performed using a FACSCalibur or LSRII instrument (BD Biosciences) and FACS data were analyzed using FlowJo software (Tree Star Inc).
Transduced T cell proliferation, cytokine/chemokine production and cytotoxicity assays
For T cell specific reactivity against tumor cells, 1×105 transduced T cells were co-cultured with decreasing numbers of tumor cells (irradiated with 10,000 rads) at the indicated ratios in 200 μl of culture volume in 96-well round-bottom plates for 72 h, and proliferation was determined by measuring 3H-thymidine (0.5 μCi/well) uptake. Cytokine and chemokine production levels were measured from culture supernatants collected 48 hours after stimulation using a Cytokine/Chemokine kit (Millipore). Granzyme B production was detected by enzyme-linked immunosorbence assay (ELISA; BioLegend). Cytotoxicity was measured using a standard 51Cr release assay. 106 target cells were labeled with 200 μCi of 51Cr for 2 hours at 37°C and washed three times. 104 labeled target cells were then co-cultured with decreasing numbers of effector T cells at the indicated effector to target (E:T) ratios in 200 μl of culture volume in 96-well round-bottom plates. Target cells incubated in medium alone were used to determined spontaneous 51Cr release, and maximal release was determined by incubating labeled target cells in 10% Triton X-100. After 5 hours at 37°C, 50 μl of supernatant was collected and 51Cr radioactivity was measured in a 1450 LSC & luminescence counter. The mean percentage of specific lysis was calculated according to the following equation: % specific lysis = (test release – spontaneous release) / (maximal release – spontaneous release) × 100. All tests were performed in triplicate wells and results are shown as mean ±SD.
Tumor models and adoptive T cell immunotherapy
6- to 8-wk-old male NSG mice (n = 6-10 for each group) were injected subcutaneously (s.c.) in the rear leg flank with 5×106 Malme-3M tumor cells. Tumor was allowed to grow to approximately 50mm2 and mice were injected intravenously (i.v.) with 5×106 human T cells. One group of mice was injected with TLR5L (5 μg/mouse in 100 μL PBS) around the tumor site weekly. For the B16 tumor experiment, C57BL/6 mice were injected (s.c.) with 1×105B16-F1 cells. When the tumor grew to approximately 50mm2, the mice were exposed to irradiation (400 cGy), and injected (i.v.) with 2.5×106 transduced T cells. Mice in the pmel groups were injected (i.p.) 3-4 times with α4-1BB antibody (100 μg/mouse in 100 μL PBS) approximately once every 10 days. Mice in the pmel+TLR5L group were injected with flagellin (10 μg/mouse in 100 μL PBS) peritumorally on the same day as T cell injection. In one of the two experiments, mice received a second injection of TLR5L on day 38. Tumor area was measured with digital calipers and tumor sizes (mm2) were calculated by multiplying length by width of the tumor mass. Mice were euthanized when tumor sizes reached approximately 250 mm2 or if mice became moribund or had trouble ambulating. Experiments were performed independently twice each yielding similar trends. Tumor sizes (mm2) were analyzed using a mixed model approach for repeated measurements and mouse survival data were analyzed with the exact log-rank test.
Chemotaxis assay and PCR arrays
For chemotaxis assay in vitro, 1×105 Malme-3M or C32 cells were cultured in the lower chamber (Cell Biolabs) in the presence or absence of flagellin (100 ng/mL) for 24 h, 2×106 PBMC isolated by Ficoll-Paque and activated with 50 ng/mL OKT3 were placed in the upper chamber. Twenty-four hours after incubation at 37°C, cells migrated to the lower chamber through polycarbonate membranes (pore size, 3 μm) were collected and stained with CD3-FITC/CD4-PE, CD3-FITC/CD8-PE, CD3-FITC/CD16-PE or HLA-DR, DP, DQ-FITC/CD14-PE (BD Biosciences) for discriminating different leukocyte subpopulations. Cells stained with relevant isotype control antibodies were used as isotype controls. To normalize cell counts between samples, an equal volume of calibration beads was added to each sample and the instrument gates were set to count a constant number of beads and analyzed by flow cytometry. The level of cytokine/chemokine mRNA transcripts by human or mouse cells or tumor samples were determined using a SuperArray PCR Array. Total RNA was extracted from tumor samples using an RNeasy Mini kit (QIAGEN) and cytokine and chemokine mRNA levels were determined using a Cytokines & Chemokines PCR Array kit (QIAGEN) according to the manufacturer's instructions.
Results
TLR5 stimulation on human T cells augments proliferation
Costimulation via TLRs increases the proliferation of human and mouse T cells and enhances their in vivo cytolytic activity against established murine tumors (13, 16). However, the effects of different TLRLs vary depending upon which T cell subset is stimulated (17), therefore we first confirmed that human CD8+ (and CD4+) T cells responded to the TLR5L. Purified CD8+ T cells stimulated with plate-bound anti-CD3 antibody (Ab) and flagellin proliferated to a greater extent than T cells with anti-CD3 Ab alone (Fig. 1A, left panel). The costimulatory effects of flagellin were comparable to those of CD28 costimulation (Fig. 1A, right panel). Flagellin also enhanced CD4+ T cell proliferation (Supplementary Fig. S1).
Engineering flagellin–producing MART-1–specific T cells
Peripheral mononuclear cells (PBMCs) were transduced with DMF5, the TCR specific to the Melan-A antigen recognized by cytotoxic T cells (DMF5-125-37)(24) or engineered to express DMF5 TCR-IRES-TLR5L (referred to as DMF5TLR5L) (Figure 1B, top panels). DMF5 gene modified T cells have been shown to mediate tumor regression in melanoma patients (24). Transduction efficiencies varied between donor samples but DMF5+CD8+ T cells were expanded to between 59% and 82% using HLA-peptide-bound beads to stimulate MART1-reactive T cells (Fig. 1B, bottom panel). We observed a strong and specific band of flagellin protein in the supernatant from DMF5TLR5L T cells but not in control T cells (Fig. 1C). By comparing band intensity after accounting for the percentage of transduced T cells and supernatant dilution, we determined that DMF5TLR5L T cells secrete approximately 32-50ng of TLR5L per 1×106 T cells over 48 hours.
TLR5L–producing DMF5 T cells exhibit increased proliferation and enhanced effector responses
The proliferative capacity of DMF5 and DMF5TLR5L T cells was examined using beads coated with HLA-A2/MART127-35 at various bead-to-cell ratios. DMF5TLR5L T cells proliferated to a greater extent than DMF5 T cells in a bead-dependent manner (Fig. 2A, left panel). However, non-transduced T cells did not proliferate when co-cultured with HLA-A2/MART-127-35 beads, highlighting the antigen specificity of the DMF5 TCR. To confirm DMF5TLR5L T cell's specificity to recognize the MART-1+ antigen on melanoma cells, transduced T cells were co-cultured with the HLA-A2+MART-1+ melanoma line Malme-3M or with HLA-A2+MART-1– A375 cells. DMF5TLR5L T cells exhibited greater proliferation than did DMF5 T cells following stimulation with Malme-3M cells (Fig. 2A, middle panel). However, DMF5 and DMF5TLR5L T cells did not proliferate when co-cultured with A375 cells (Fig. 2A, right panel). The transduction efficiency varied between donors however an equal number of MART-1–reactive T cells were used in all experiments. In cases where we obtained similar transduction efficiencies of DMF5 and DMF5TLR5L, DMF5TLR5L continued to demonstrate enhanced effector function (Supplementary Fig. S2).
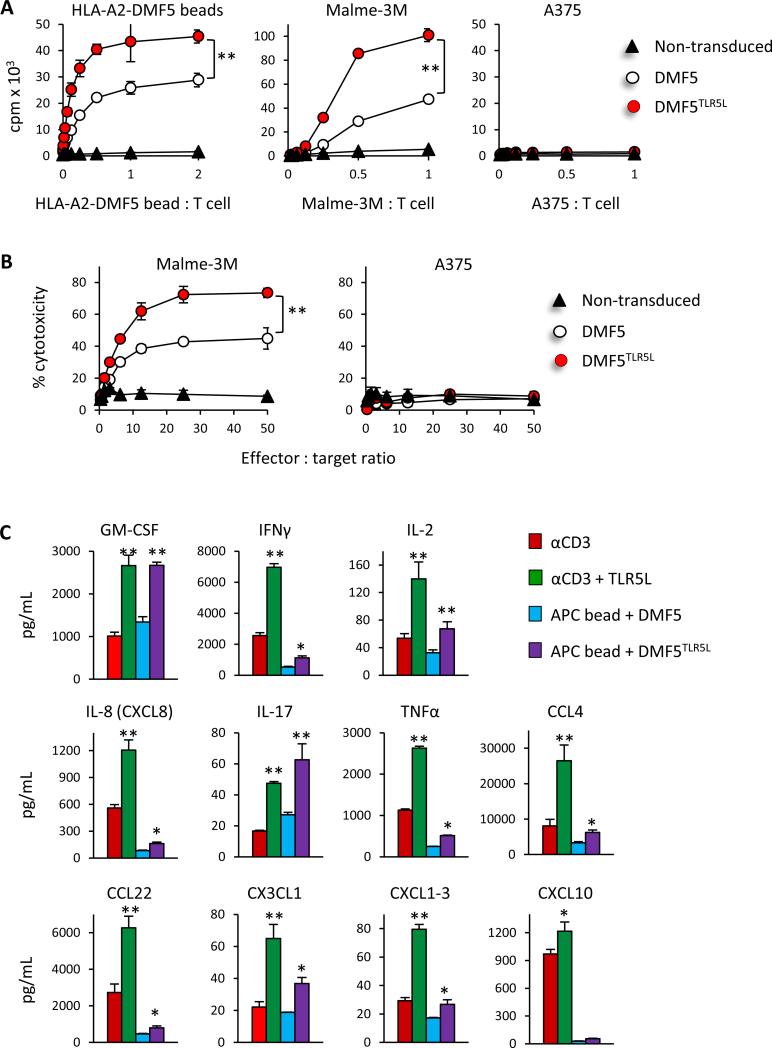
Figure 2. Transduced human T cells coexpressing DMF5 and the TLR5L show significantly greater proliferation, the production of cytokines and chemokines, and cytolytic activity than T cells expressing DMF5 alone.
(A) Control untransduced T cells or an equal number of DMF5 and DMF5TLR5L T cells were analyzed for proliferation in response to HLA-A2-MART-1 beads, MART-1+ Malme-3M or MART-1- A375 tumor cells at the indicated ratios determined by 3H-thymindine uptake 72 hours after stimulation. (B) Cytotoxicity against Malme-3M or A375 cells at varying effector-to-target ratios was determined in a 5-h 51Cr release assay. All data are shown as mean ±S.D. and representative of three independent experiments, each yielding identical trends. Data are presented as mean ±S.D; *p<0.05, **p<0.01, 2-way ANOVA. (C) The production of cytokines and chemokines in response to anti-CD3 stimulation or APC beads was evaluated with or without TLR5L stimulation using a Milliplex cytokine/chemokine array 48 hours after stimulation. Data are presented as mean ±S.D; *p<0.05, **p<0.01, T-test.
DMF5TLR5L T cells also exhibited significantly higher cytolytic activity towards Malme-3M melanoma tumor cells at different effector to target cell ratios than did DMF5 T cells (Fig. 2B, left panel). In contrast, non-transduced T cells did not lyse target cells. Neither MART-1 nor non-transduced T cells lysed A375 tumor cells (Fig. 2B, right panel). Moreover, TLR5L increased the expression of various cytokines and chemokines of anti-CD3–stimulated CD8 T cells (Fig. 2C) and in are agreement with previous reports demonstrating that TLR5L increased IFN-γ production (26, 27). Autocrine co-stimulation by DMF5TLR5L also increased the levels of various cytokines and chemokines as compared with DMF5 T cells (Fig. 2C). Collectively, these data demonstrate that TLR5 stimulation on T cells augments their proliferative and killing capacity and that TLR5L augments the expression of several cytokines and chemokines.
DMF5TLR5L T cells exhibit enhanced antitumor activity against an established melanoma tumor in vivo
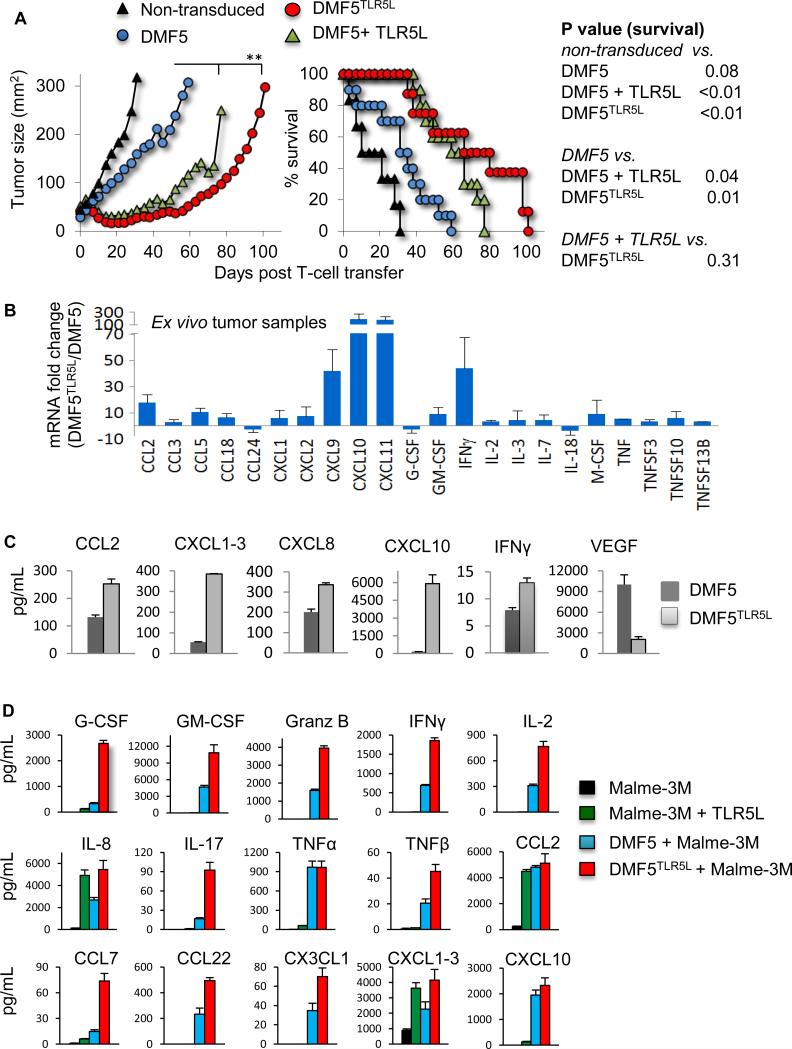
The antitumor activities of DMF5TLR5L T cells, DMF5, and DMF5 T cells co-injected with TLR5L were examined in NSG mice bearing established human melanoma tumors. Mice treated with either DMF5TLR5L or DMF5 T cells plus TLR5L exhibited tumor reduction during the first three weeks after T cell transfer (Fig. 3A, right panel). Tumors in mice treated with non-transduced T cells grew rapidly, whereas mice treated with DMF5 T cells exhibited moderately delayed tumor progression but did not demonstrate evidence of tumor regression. In contrast, tumors in animals treated with MART-1 plus TLR5L or DMF5TLR5L T cells initially regressed and remained stably controlled until approximately day 49 post T cell injection. The median survival for mice receiving DMF5
Figure 3. Human T cells engineered to express DMF5 and TLR5L enhance antitumor activity against an established human melanoma tumor and antitumor is also associated with the increased expression of various cytokines and chemokines.
(A) NSG mice were injected s.c. with Malme-3M cells and injected i.v. with transduced human T cells when tumors reached a size of ~50mm2. One group of mice received weekly peritumoral s.c. injections of TLR5L. Tumor sizes (mm2) were calculated by measuring perpendicular by longitudinal diameter. Survival curves were analyzed using the exact log-rank test and tumor growth using 2-way ANOVA. Data are compiled from two independent experiments, with each experiment yielding similar results. (B) Cytokine and chemokine mRNA levels in tumor cells were analyzed using a cytokines & chemokines PCR array. Representative data from two experiments are presented as mean ±S.D; *p<0.05, **p<0.01, T-test. (C) The expression levels of cytokines and chemokines in tumor cell lysate were determined using a Milliplex cytokine/chemokine array. (D) Malme-3M cells were cultured with or without T cells and 100 ng/mL TLR5L for 48 hours and cytokine and chemokine levels in the supernatant were examined. Shown are the mean ±S.D; *p<0.05, **p<0.01, T-test.
T cell alone was 16 days whereas mice receiving DMF5 T cells plus TLR5L or DMF5TLR5L T cell groups were 61 and 70 days, respectively. These findings are the first to highlight the efficacy of using tumor-reactive CD8+ T cells to deliver and produce a TLR ligand at the tumor site to enhance antitumor activity.
Increased influx and retention of tumor infiltrating lymphocytes has the potential to augment antitumor responses and is governed in large part by the levels of specific chemokines and cytokines produced within the tumor environment. We compared tumor chemokine/cytokine gene expression profiles between mice that received DMF5TLR5L and DMF5 T cells alone. The changes in gene expression levels shown in Fig. 3B reveal extensive alterations in the levels of various chemokines and cytokines, skewing the profile towards one that favors T cell infiltration and cell division. In particular, we detected increased levels of IL-2, IFN-γ, CCL2, CCL3, CCL5, CXCL9 and CXCL10. This set of chemokines has been associated with lymphocyte infiltration and antitumor responses in melanoma patients (17, 28). In agreement with the mRNA levels, protein concentrations of IFN-γ, IL-2, CCL2, CXCL1-3 and CXCL10 and several other not the gene array were elevated in DMF5TLR5L–treated mice, Fig. 3C.
Melanoma cells can express functional TLRs (17, 29). The Human Protein Atlas, an online immunohistochemistry (IHC) database evaluating the expression of various proteins and cancer types, shows TLR5 to be expressed at low levels on 30% of patients with melanoma. Additionally, the expression of TLR5 on human melanoma cell lines and biopsy specimens was confirmed by flow cytometry and immunohistochemistry (Supplementary Fig. S3). In light of these findings, we sought to determine whether one potential source of chemokines was from TLR5-stimulated melanoma cells. Addition of TLR5L to Malme-3M cells induced the expression of the various cytokines and chemokines shown in Figure 3D. We examined a broader list of cytokines/chemokines in melanoma that might have be impacted by TLR5 stimulation using a focused real-time PCR array and found significant increases in the levels of CCL1, CXCL2, CXCL8, CXCL10, CXCL11 and CXCL1-3 (Supplementary Fig. S4A). Notably, despite TLR5L's ability to stimulate cytokines/chemokines expression, it did not enhance melanoma proliferation (Supplementary Fig. S4B). The levels of cytokines and chemokines were also examined from the supernatants of DMF5TLR5L or DMF5 T cells co-cultured with Malme-3M cells. Various cytokines, including IL-2 and IL-17, and effector molecules Granzyme B, IFN-γ, and TNF-β were significantly increased in supernatants of DMF5TLR5L T cells as compared with DMF5 T cells (Fig. 3D).
TLR5 stimulation on CD8 T cells is a strong inducer of IFN-γ which can upregulate MHC I and potentiate tumor recognition by CD8+ T cells. We examined MHC I (pan HLA-A, B, C) and MHC II (HLA-DR, DP, DQ) expression on melanoma cells in response to supernatants from DMF5TLR5L T cells, DMF5 T cells or TLR5 ligand alone. All the supernatants from wells containing T cells increased MHC I expression 2 to 3-fold (Supplementary Fig. S5). Untreated Malme-3M cells did not appear to express MHC II but could be induced upon culture with T cell supernatants. We did not detect changes in the levels of MHC I or MHC II using supernatant from TLR5-stimulated T cells or from melanoma cells stimulated with TLR5L alone, indicating that the increased MHC expression levels were in response to factors produced by activated T cells and not dependent on TLR5L.
TLR5–stimulated melanoma cells promote lymphocyte migration
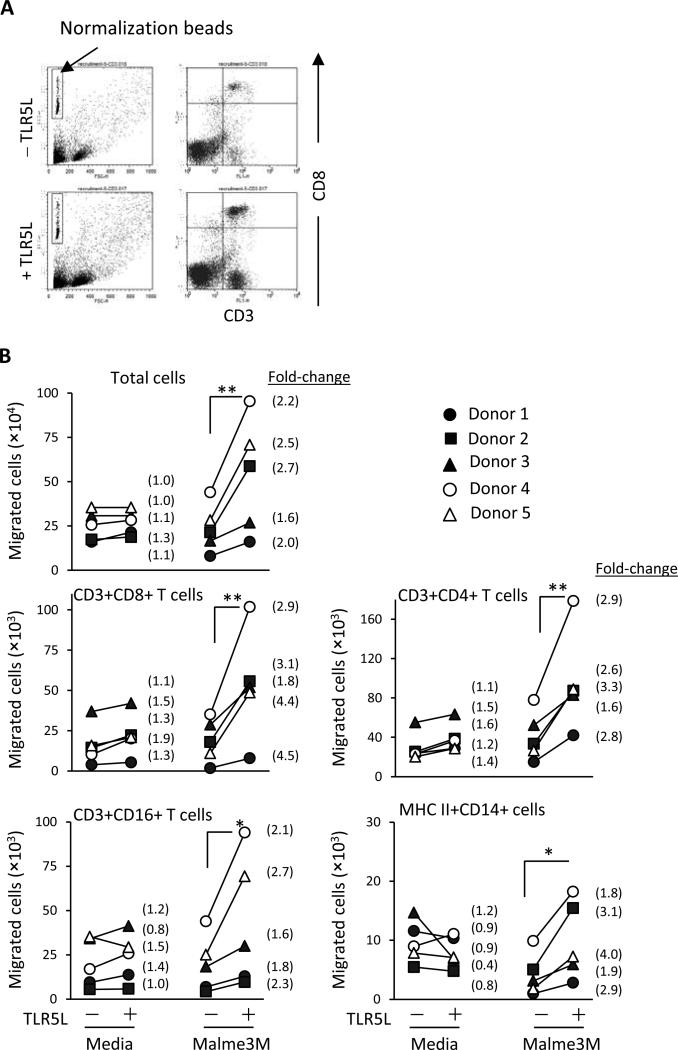
Specific chemokines in the tumor environment promote not only the infiltration of tumor-reactive T cells but also impact the subsets of lymphocytes that migrate to the tumor. The importance of certain chemokines is highlighted in studies demonstrating an association between specific chemokine profiles and the presence of T cells (17, 28). To determine whether TLR5-stimulated melanoma cells impact T cell migration, Malme-3M cells were or were not prestimulated with TLR5L for 48 hours. Cell migration across a transwell insert was examined by flow cytometry. Figure 4A shows a representative dot plot of CD3+CD8+ T cells that migrated towards TLR5-stimulated melanoma cells. Increased T cell migration toward TLR5-stimulated Malme-3M melanoma cells was observed when compared to unstimulated melanoma cells (Fig. 4B). Similar effects were also observed using the TLR5–stimulated C32 melanoma cells (Supplementary Fig. S6). These effects were a result of TLR5 stimulation on tumor cells and not the effects of residual TLR5L, as TLR5L alone did not alter lymphocyte migration (Fig. 4B). We also examined the expression of CCR2, CXCR2 and CXCR3 on T cells, receptors shown to be critical for tumor infiltration (8, 30, 31). CD8 T cells in general showed high levels of CXCR3 (Supplementary Fig. S7). However, DMF5TLR5L T cells and DMF5 T cells cultured with TLR5L showed a modest increase in CXCR3 expression levels above DMF5 T cells. CCR2 and CXCR2 were weakly expressed on CD8 T cells despite the presence of TLR5L.
Figure 4. Malme-3M cells prestimulated with TLR5L recruit different leukocyte subpopulations in vitro.
Malme-3M cells were stimulated with 100 ng/mL TLR5L for 24 hours and then washed off prior to adding PBMCs. PBMCs were activated using 50 ng/mL OKT3 for 24 hours and then placed in the upper transwell chamber. Twenty hours later, migratory cells were collected, stained with antibodies and analyzed by flow cytometry. (A) Representative FACS analysis. To normalize the cell count between groups, an equal volume of calibration beads was added to an equal volume of supernatant and the flow cytometry instrument gates were set to count a constant number of beads. (B) The number of cells that migrated toward TLR5-stimulated or unstimulated Malme-3M cells is shown. Medium with and without 100 ng/mL TLR5L was used as negative controls. PBMCs from five healthy donors were examined. The number in parentheses is the fold change of migratory human leukocyte number. Power values were calculated using the changes in fold migration between the indicated groups; *p<0.05, **p<0.01, T-test.
Altogether, these data demonstrate that TLR5-stimulated tumor cells promote T cell migration and suggest that TLR5-stimulated tumor cells in vivo might enhance T cell infiltration.
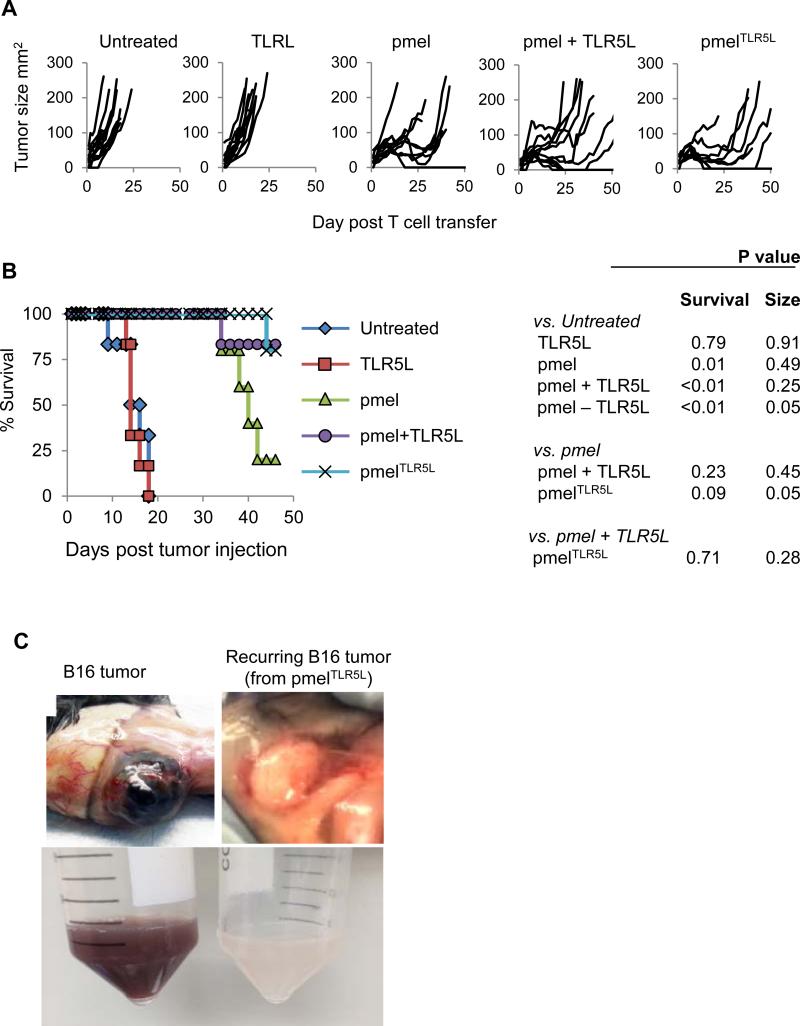
Intratumoral provision of TLR5L by CD8 T cells exhibits enhanced antitumor activity in wild type mice
We investigated the antitumor effects of TLR5L-producing T cells in a syngeneic murine model using B16-F1 melanoma cells and pmel CD8+ T cells specific to the gp10025-33 antigen. C57BL6 mice harboring established B16 tumors were injected with TLR5L, alone, pmel, pmel plus soluble TLR5L, or pmelTLR5L. Untreated and TLR5L-treated mice showed similar tumor growth kinetics. In contrast, in mice receiving pmelTLR5L T cells or pmel cells with or without TLR5L, tumors shrunk within 10 days (Fig. 5A). Mice treated with pmelTLR5L cells demonstrated the greatest tumor reduction and the longest period of tumor size stability and prolonged survival (Fig. 5A and 5B). Tumors resumed growth in all but one mouse in the group that received pmel T cells. Mice treated with pmelTLR5L T cells were equal to mice receiving pmel cells plus TLR5L. Noteworthy was that recurring tumors were white in color indicating the outgrowth of cancer cells that lost expression of the protein encoding the peptide targeted by pmel T cells (Fig. 5C) indicating that tumor recurrence was a consequence of antigen loss.
Figure 5. Tumor-specific mouse T cells engineered to secrete TLR5L show enhanced antitumor activity and improve mouse survival.
5×106 T cells were adoptively transferred to C57BL6 mice with established (35-50mm2) subcutaneous B16 tumors in combination with anti-4-1BB Ab. TLR5L was administered subcutaneously in indicated treatment groups. (A and B) Tumor growth and survival. (C) Recurring tumors were white in color compared to black primary tumors, indicating loss of melanin.
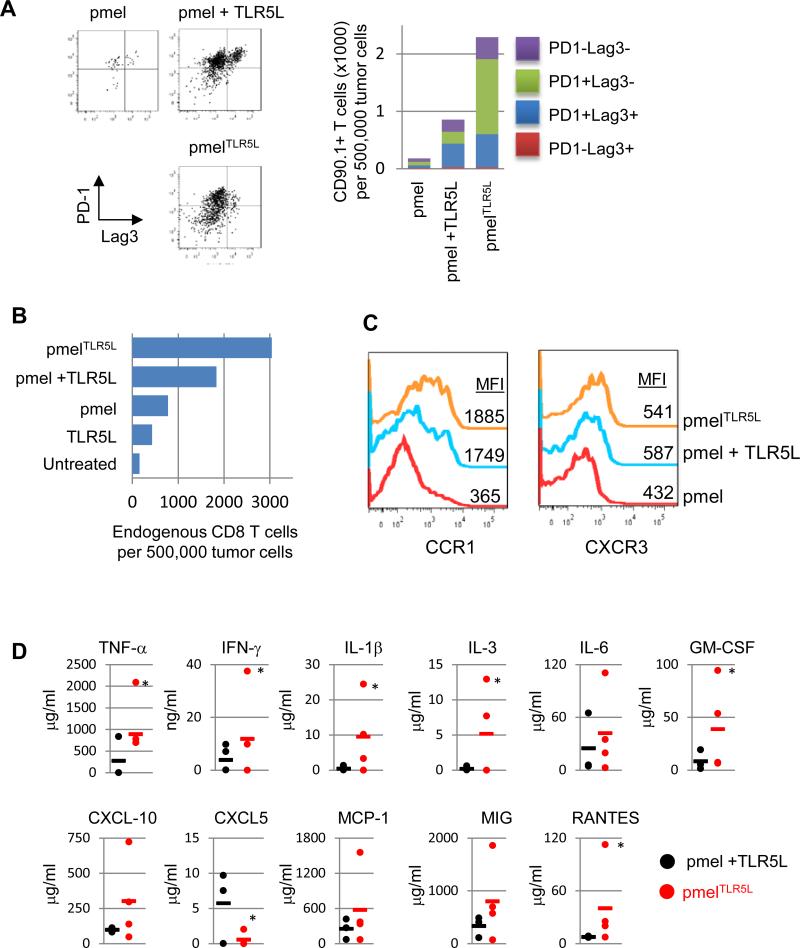
Enhanced antitumor responses by TLR5L-producing T cells is associated with increased tumor infiltration and fewer PD1+Lag3+ T cells
T cell exhaustion is associated with the expression of various markers including PD-1 and Lag3(12). The percentage of transferred T cells with an exhausted phenotype (PD-1+Lag3+) were highest in mice treated with pmel or pmel plus TLR5L as compared with pmelTLR5L (Fig. 6A). Moreover, there were ten and three times more pmelTLR5L T cells in the tumor than in tumors treated with pmel cells and pmel plus injected TLR5L, respectively. These results highlight a potential advantage of intratumoral delivery of TLR5L. PmelTLR5L T cells also increased the number of endogenous tumor infiltrating CD8+ T cells (Fig. 6B). Mice receiving pmelTLR5L cells or pmel cells plus TLR5L expressed three to five times higher levels of CCR1 as compared with mice receiving pmel cells alone (Fig. 6C). However, CXCR3 expression levels only increased slightly in the pmelTLR5L or pmel plus TLR5L groups relative to pmel treatment alone. Together with the improved antitumor activity, altered chemokine receptor expression and increased tumor infiltration of pmelTLR5L cells these data suggest that intratumoral delivery of TLR5L (by pmel cells) provides an advantage over mice injected with pmel cells and systemic TLR5L.
Figure 6. TLR5L-secreting T cells exhibit increased infiltration and decreased expression of exhaustion markers and changes to the cytokine/chemokine profile at the tumor site.
(A) Tumor-infiltrating T cells were stained for PD1 and Lag3 as markers of T cell exhaustion. Left panel shows representative flow plots of PD1+Lag3+ populations. Right panel shows summary of total CD90.1+ T cell numbers and proportions of PD-1+ and/or Lag3+ populations. (B) Tumor samples were collected and the total number of CD8 T cells was determined. (C) Tumor samples were examined for the expression of CCR1 and CXCR3 chemokine receptors presented as mean fluorescence intensity (MFI). (D) The levels of the indicated cytokines and chemokines in tumor samples were determined using a 38-cytokine/chemokines array. Each point represents an individual mouse; *p<0.05; T-test.
To determine whether intratumoral delivery of TLR5L by pmelTLR5L cells differentially altered the tumor environment than in mice treated with pmel cells and injected with TLR5L, we examined the levels of various cytokines and chemokines. We detected changes in the levels of various cytokines and chemokines within the tumor (Fig. 6D). Changes in the expression levels of these cytokines/chemokines correlated with increased T cell infiltration and antitumor activity and are in agreement with previous studies demonstrating a correlation between specific chemokines and CD8+ T cell infiltration (17, 28). In contrast, there was a trend towards higher levels of IL-10, IL-13 and MIG in mice treated systemically with TLR5L (Supplemental Fig. S8A). In vitro, the stimulation of pmel T cells in the presence or absence of B16 tumor cells with cocultured with TLR5L also augmented the production of a limited number of cytokines including IFN-γ and CXCL10 (Supplementary Fig. S8B). This is in contrast to broader array of factors induced in human T cells. Altogether, these results highlight that intratumoral delivery of TLR5L induces a distinct response that is associated with increased T cell priming and infiltration, and a reduced percentage of exhausted T cells.
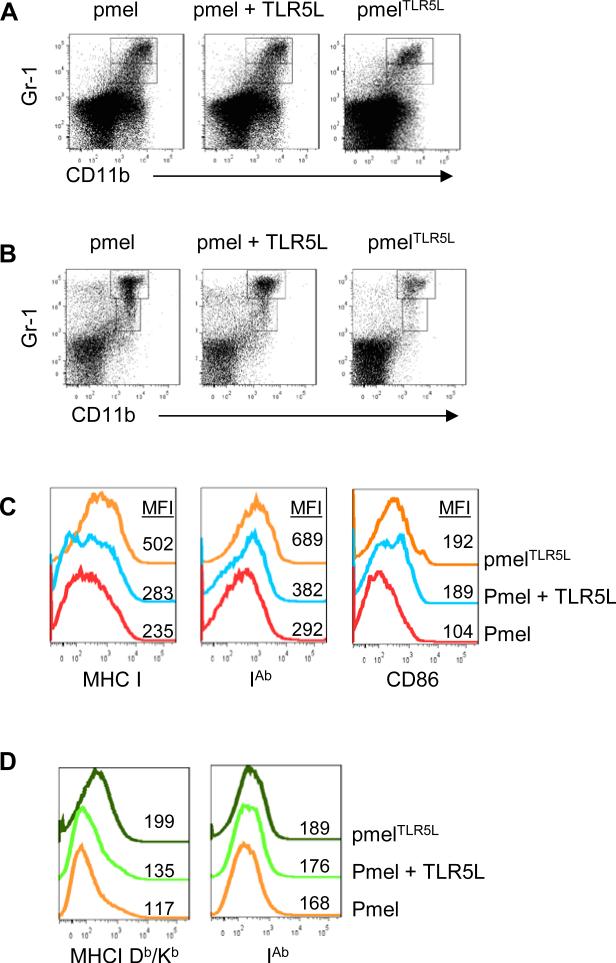
TLR5 ligand-producing T cells reduce the number of MDSCs and induce MHC I, MHC II and CD86 expression on MDSCs
To determine if intratumoral provision of TLR5L altered the intratumoral composition, we surveyed the presence of CD4+ TRegs, MDSC and PD-L1 expression. We found that PDL1 expression was weak and comparable between all treatment groups. Moreover, in all groups, CD4+ T cells were <0.1% and TReg (CD4+ Foxp3+CD25+) constituted less than 3% of the 0.1%. In sharp contrast, there was 39+6% reduction of CD11b+Gr-1med/hi cells in the tumors of pmelTLR5L–treated mice as compared with pmel–treated mice or mice treated with pmel cells and injected with TLR5L (Fig. 7A). However, we did not detect differences in the frequency of tumor-infiltrating MDSCs between mice treated with pmel T cells with or without TLR5L. The decreases in MDSC numbers were more obvious in the spleens of mice treated with pmelTLR5L cells and resulted in 4- and 2-fold fewer splenic MDSCs (CD11b+Gr1+) as compared with mice treated with pmel or pmel plus TLR5L, respectively (Fig. 7B). Most of the reduction in both spleen and tumor was due to fewer monocytic Gr-1highCD11b+ cells. Interestingly, the decrease of MDSCs correlated with a striking reduction in CXCL5 levels (Figure 6), the main chemokine involved in the migration of MDSCs into tissue including tumors (32, 33).
Figure 7. TLR5L-secreting T cells decrease myeloid derived suppressor cells and increase MHC and costimulatory expression.
Tumor (A) and spleen (B) samples were collected 7-10 days post-treatment and stained for analysis by flow cytometry. Myeloid derived suppressor cells were identified by Gr-1 and CD11b staining. (C) Representative histograms demonstrating the expression of MHC class I (H2-Db/H2-Kb), MHC class II (IA ), and CD86 displayed as mean fluorescence intensity (MFI) gated on MDSCs (D) or total tumor cells.
Based on TLR5L's immunostimulatory capacity, we examined the expression of MHC-I (H2-Db and Kb), MHC II (I-Ab) and the costimulatory molecule CD86. MDSCs from mice treated with pmelTLR5L demonstrated a slight increase in the expression of MHC I, MHC II and CD86 as compared with mice treated with pmel with or without TLR5L (Fig. 7C). There was also a trend towards increased MHC I and MHC II in mice treated with pmelTLR5L (Fig. 7D). Since MHC I expression on B16 melanoma cells is increased following co-culture with supernatants from pmel, pmelTLR5L or pmel plus TLR5L (Supplementary Fig. S9), in vivo increases in MHC I might also be due to increased IFN-γ production by pmelTLR5L. In agreement with these results, recent studies demonstrated that TLR5L induces CD80 and CD86 on wild type but not TLR5–/– murine CD11b+ and CD11c+ myeloid cells (34). We further examined whether TLR5 engagement on a purified population of MDSCs altered their ability to suppress T cells or to induce the expression of CD80, CD86 MHC I or II. However, TLR5L did not alter their suppressive ability and had minimal effects on CD80, MHC I or MHC II expression (Supplementary Fig. S10). The lack of response to TLR5L did not appear to be the lack of TLR5 on MDSCs from tumors and to some extent from spleen expressed TLR5, however TLR5 expression and blood derived MDSCs was weak (Supplementary Fig. S10). These data suggest that the changes observed in MDSCs in vivo might have occurred in response to factors produced by other cells in the tumor (or spleen) reacting to TLR5L. Collectively, these results suggest that T cell-mediated intratumoral delivery of flagellin helps overcome immune suppression in part by reducing the number of MDSCs and by altering their phenotype.
Discussion
We sought to determine whether T cell-mediated delivery of a potent immune adjuvant, TLR5L, to the tumor could restore T cell activity by providing a continuous source of costimulation. TLR5L-secreting T cells reduced tumor progression and prolonged host survival. Enhanced antitumor activity and tumor destruction was accompanied by increases in tumor infiltrating lymphocytes (TIL), a reduction in the number of exhausted tumor infiltrating lymphocytes (TIL), and changes in the expression of chemokines and receptors on T cells. Treatment with TLR5L-secreting T cells also reduced the number of MDSCs and increased the expression of costimulatory and stimulatory molecules on MDSCs and tumor cells. This study reveals for the first time the potential to use tumor-reactive T cells to transport and continuously secrete a TLRL at the tumor site to reshape the tumor environment and potentiate antitumor T cell responses.
TLR5L appears to be well-tolerated when administrated systemically and effective at augmenting immune responses in mice and nonhuman primates (21, 35). The TLR5 agonist is currently under investigation in Phase I trials in patients with advanced or metastatic solid tumors (NCT01527136) and in Phase II trials testing its immunogenicity in Influenza vaccines (NCT00966238). In agreement with preclinical models, early results from NCT00966238 indicate a low frequency of adverse events at amounts as high as 3 μg (injected intramuscularly). Although systemic administration of flagellin is relatively safe, it displays a short half-life in vitro and in vivo in mice likely limiting its efficacy (36, 37). Flagellin is undetectable in mouse serum 2 hours after injection of 50μg via i.p. administration. Therefore, an added advantage to intratumoral TLR5L delivery by T cells is its continuous secretion at the tumor site. It is important to note that, despite the continuous secretion of TLR5L by T cells, we did not detect grossly elevated levels of cytokines in the serum between all treatment groups as has been noted when using other TLRLs such as TLR4 (38). We detected a moderate increase in IL-12 and a reduction in IL-10, IL-13 and MIG in the serum (Supplemental Fig 8A). Furthermore, we did detect any differences in toxicities among mice treated with engineered T cells versus systemic administration of TLR5 ligand. TLR5L can also enhance DC maturation and increase antigen processing and presentation to T cells (39). TLR5 engagement has also been shown to convert tolerogenic DCs into activating antigen-presenting cells that promote Th1 responses by increasing CD83, CD80, CD86, MHC class II as well as several chemokines including CXCL1, CXCL8 and CCL2 (37, 40, 41).
The use of T cells to deliver TLR5L to the tumor offers several advantages over systemic administration. First, unlike drugs, T cells possess the ability to selectively home to tumor sites. This feature makes T cells ideal drug delivery vehicles for patients with widespread metastatic disease. Moreover, traditional systemic therapies can suffer from poor tumor penetration whereas T cells can penetrate deep into tumors. A second advantage to T cell-mediated drug delivery is that it allows for localized and concentrated production of therapeutics in the tumor environment. In contrast, most tissues are exposed to systemic therapies which can result in toxicity and non-specific targeting, consequently limiting the ability to supply an effective dose (42). Importantly, effective localized delivery of drugs such as TLRLs has the potential to prevent common adverse effects associated with systemic drug delivery, such as cytokine storms. T cell-released compounds are only exposed to the isolated metabolic pressures of the tumor environment. In contrast, traditionally administered therapeutics must be titrated to account for drug tissue distribution, biotransformation, degradation, and renal clearance. Finally, another benefit of the TLR5L-secreting T cell system is that intratumoral supply of TLR5L induces the production of chemokines and cytokines including CCL2, CXCL1-3, CXCL8 and CXCL10 as well IFN-γ and IL-2 that enhance tumor infiltration of other T cells.
TLR5 expression on immune cells differs between mice and humans. We observed that TLR5 engagement on human T cells robustly increased proliferation, cytoxicity and a broad array of cytokine and chemokines. However, responses by mouse T cells were not as vigorous and increased a limited number of cytokines and chemokines (Supplementary Fig. S8). These distinctions may be related to differences in TLR5 expression between mouse and human T cells. In humans, TLR5 is expressed on a broad range of cells including endothelial cells, dendritic cells, monocytes, CD4 and CD8 T cells and on various tumor cell types (17, 43-47). Studies by Galli and colleagues showed that human prostate tumor cells treated with TLR5L produced higher levels of chemokines which in turn enhanced the recruitment of various leukocyte subpopulations (48). The stimulation of TLR5 on other cells in the tumor environment could serve to potentiate antitumor responses by secreting factors that enhance T cell infiltration and/or by altering the phenotype of suppressive cells, resulting in reduced T cell suppression (17, 43-47).
Treating mice with TLR5–producing T cells (or co-administering TLR5L with tumor-reactive T cells) reduced MDSC numbers and induced the expression of costimulatory molecules. This reduction could be because TLR5L skewed the cytokine profile toward one less amenable to MDSC development and/or by driving the differentiation of MDSC to more mature cells. This idea is supported by our observations that injection of TLR5L or delivery by T cells increased MHC I, MHC II, and CD86 expression on MDSC. Taken together with a reduced number of MDSCs and an increase in the number of tumor infiltrating CD8+ T cells (Fig. 6B), we speculate that TLR5 L interferes with important immunoregulatory loops that might favor the cross-presentation of tumor antigens different from the one targeted (Fig. 7). Although, we cannot definitively attribute the changes in the numbers or phenotype of MDSCs with the final therapeutic outcome in mice that received TLR5 ligand–secreting T cells, the ability for TLRLs and cytokines to alter MDSC function or phenotype has been reported by others. For example, the TLR9L CpG-containing oligodeoxynucleotides, which, like TLR5 signaling, also activates the MyD88-dependent signaling pathway can induce high levels of IFN-g and cause MDSCs to lose their suppressive functions (49). Admittedly, in our studies the addition of TLR5L to purified MDSCs did not alter their suppressive activity which might have been due to low TLR5 expression. Another striking observation in pmelTLR5L–treated mice was a drastic reduction of CXCL5 which plays a critical in MDSC migration and perhaps development (32, 33). It is worth noting that mice treated with pmelTLR5L demonstrated a lower recurrence rate (1 of 4) than did mice treated with pmel plus TLR5L, in which 3 of 4 mice recurred after a prolonged period of being tumor-free. We speculate that changes in MDSC number and/or phenotype might have attributed to these outcomes. Clinical data highlighting that the recurrence-free survival is significantly shortened in patients with higher frequencies of MDSCs supports this idea (50, 51). Finally, the use of engineered T cells to deliver an immune adjuvant such as IL-12 to the tumor was recently shown to increase T cell responses in part by reprogramming and affecting MDSCs and macrophages within the tumor highlighting the potential to use T cells as delivery vehicles in order to localize the drug to the tumor site and to augment antitumor T cell responses (52, 53). In our model, there was a trend towards increased IL-12p40 levels in the serum of pmelTLR5L–treated mice as compared with mice receiving pmel T cells plus TLR5L highlighting the pleotropic effects of intratumoral TLR5L delivery (Supplemental Fig S8).
In conclusion, the delivery of a TLRL directly to the tumor site by tumor-reactive T cells represents a novel method to improve T cell cytotoxicity, infiltration and ultimately antitumor efficacy. A diagram depicting various features regarding how intratumoral TLR5L increases antitumor T cell activity is shown in Supplemental Fig. S11.
Supplementary Material
ACKNOWLEDGEMENTS
Research was supported by the National Institutes of Health (NIH) National Cancer Institute (NCI) R01CA140917, NIH Center for Biomedical Research Center Excellence grants 1P20RR021970, and the University of Maryland Marlene and Stewart Greenebaum Cancer Center. A rabbit anti-flagellin serum was kindly provided by Dr. Jim Kaper at the University of Maryland, Baltimore. The MART-1 TCR was kindly provided by Dr. Laura Johnson at the Translational Research Program, Abramson Family Cancer Research Institute, Perelman School of Medicine, University of Pennsylvania.
Footnotes
Conflict of Interest: The authors disclose no potential conflicts of interest.
Reference List
- 1.Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711–23. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Restifo NP, Dudley ME, Rosenberg SA. Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol. 2012;12:269–81. doi: 10.1038/nri3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, Hwu P, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366:2455–65. doi: 10.1056/NEJMoa1200694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.O'Day SJ, Maio M, Chiarion-Sileni V, Gajewski TF, Pehamberger H, Bondarenko IN, et al. Efficacy and safety of ipilimumab monotherapy in patients with pretreated advanced melanoma: a multicenter single-arm phase II study. Ann Oncol. 2010;21:1712–7. doi: 10.1093/annonc/mdq013. [DOI] [PubMed] [Google Scholar]
- 5.Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–54. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Weber J, Thompson JA, Hamid O, Minor D, Amin A, Ron I, et al. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin Cancer Res. 2009;15:5591–8. doi: 10.1158/1078-0432.CCR-09-1024. [DOI] [PubMed] [Google Scholar]
- 7.Wolchok JD, Neyns B, Linette G, Negrier S, Lutzky J, Thomas L, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 2010;11:155–64. doi: 10.1016/S1470-2045(09)70334-1. [DOI] [PubMed] [Google Scholar]
- 8.Peng W, Ye Y, Rabinovich BA, Liu C, Lou Y, Zhang M, et al. Transduction of tumor-specific T cells with CXCR2 chemokine receptor improves migration to tumor and antitumor immune responses. Clin Cancer Res. 2010;16:5458–68. doi: 10.1158/1078-0432.CCR-10-0712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gabrilovich DI, Ostrand-Rosenberg S, Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol. 2012;12:253–68. doi: 10.1038/nri3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zou W. Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol. 2006;6:295–307. doi: 10.1038/nri1806. [DOI] [PubMed] [Google Scholar]
- 11.Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol. 2013;13:227–42. doi: 10.1038/nri3405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Drake CG, Lipson EJ, Brahmer JR. Breathing new life into immunotherapy: review of melanoma, lung and kidney cancer. Nat Rev Clin Oncol. 2014;11:24–37. doi: 10.1038/nrclinonc.2013.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Asprodites N, Zheng L, Geng D, Velasco-Gonzalez C, Sanchez-Perez L, Davila E. Engagement of Toll-like receptor-2 on cytotoxic T-lymphocytes occurs in vivo and augments antitumor activity. FASEB J. 2008;22:3628–37. doi: 10.1096/fj.08-108274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Babu S, Blauvelt CP, Kumaraswami V, Nutman TB. Cutting edge: diminished T cell TLR expression and function modulates the immune response in human filarial infection. J Immunol. 2006;176:3885–9. doi: 10.4049/jimmunol.176.7.3885. [DOI] [PubMed] [Google Scholar]
- 15.Cottalorda A, Verschelde C, Marcais A, Tomkowiak M, Musette P, Uematsu S, et al. TLR2 engagement on CD8 T cells lowers the thresholdfor optimal antigen-induced T cell activation. Eur J Immunol. 2006;36:1684–93. doi: 10.1002/eji.200636181. [DOI] [PubMed] [Google Scholar]
- 16.Geng D, Zheng L, Srivastava R, Velasco-Gonzalez C, Riker A, Markovic SN, et al. Amplifying TLR-MyD88 signals within tumor-specific T cells enhances antitumor activity to suboptimal levels of weakly immunogenic tumor antigens. Cancer Res. 2010;70:7442–54. doi: 10.1158/0008-5472.CAN-10-0247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kaczanowska S, Joseph AM, Davila E. TLR agonists: our best frenemy in cancer immunotherapy. J Leukoc Biol. 2013;93:847–63. doi: 10.1189/jlb.1012501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Komai-Koma M, Jones L, Ogg GS, Xu D, Liew FY. TLR2 is expressed on activated T cells as a costimulatory receptor. Proc Natl Acad Sci U S A. 2004;101:3029–34. doi: 10.1073/pnas.0400171101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Geng D, Zheng L, Srivastava R, Asprodites N, Velasco-Gonzalez C, Davila E. When Toll-like receptor and T-cell receptor signals collide: a mechanism for enhanced CD8 T-cell effector function. Blood. 2010;116:3494–504. doi: 10.1182/blood-2010-02-268169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tallant T, Deb A, Kar N, Lupica J, de Veer MJ, DiDonato JA. Flagellin acting via TLR5 is the major activator of key signaling pathways leading to NF-kappa B and proinflammatory gene program activation in intestinal epithelial cells. BMC Microbiol. 2004;4:33. doi: 10.1186/1471-2180-4-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vijay-Kumar M, Aitken JD, Sanders CJ, Frias A, Sloane VM, Xu J, et al. Flagellin treatment protects against chemicals, bacteria, viruses, and radiation. J Immunol. 2008;180:8280–5. doi: 10.4049/jimmunol.180.12.8280. [DOI] [PubMed] [Google Scholar]
- 22.Llewellyn M, Dutton RJ, Easter J, O'donnol D, Gober JW. The conserved flaF gene has a critical role in coupling flagellin translation and assembly in Caulobacter crescentus. Mol Microbiol. 2005;57:1127–42. doi: 10.1111/j.1365-2958.2005.04745.x. [DOI] [PubMed] [Google Scholar]
- 23.Applequist SE, Rollman E, Wareing MD, Liden M, Rozell B, Hinkula J, et al. Activation of innate immunity, inflammation, and potentiation of DNA vaccination through mammalian expression of the TLR5 agonist flagellin. J Immunol. 2005;175:3882–91. doi: 10.4049/jimmunol.175.6.3882. [DOI] [PubMed] [Google Scholar]
- 24.Johnson LA, Heemskerk B, Powell DJ, Jr., Cohen CJ, Morgan RA, Dudley ME, et al. Gene transfer of tumor-reactive TCR confers both high avidity and tumor reactivity to nonreactive peripheral blood mononuclear cells and tumor-infiltrating lymphocytes. J Immunol. 2006;177:6548–59. doi: 10.4049/jimmunol.177.9.6548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tamada K, Geng D, Sakoda Y, Bansal N, Srivastava R, Li Z, et al. Redirecting gene-modified T cells toward various cancer types using tagged antibodies. Clin Cancer Res. 2012;18:6436–45. doi: 10.1158/1078-0432.CCR-12-1449. [DOI] [PubMed] [Google Scholar]
- 26.McCarron M, Reen DJ. Activated human neonatal CD8+ T cells are subject to immunomodulation by direct TLR2 or TLR5 stimulation. J Immunol. 2009;182:55–62. doi: 10.4049/jimmunol.182.1.55. [DOI] [PubMed] [Google Scholar]
- 27.Caron G, Duluc D, Fremaux I, Jeannin P, David C, Gascan H, et al. Direct stimulation of human T cells via TLR5 and TLR7/8: flagellin and R-848 up-regulate proliferation and IFN-gamma production by memory CD4+ T cells. J Immunol. 2005;175:1551–7. doi: 10.4049/jimmunol.175.3.1551. [DOI] [PubMed] [Google Scholar]
- 28.Harlin H, Meng Y, Peterson AC, Zha Y, Tretiakova M, Slingluff C, et al. Chemokine expression in melanoma metastases associated with CD8+ T-cell recruitment. Cancer Res. 2009;69:3077–85. doi: 10.1158/0008-5472.CAN-08-2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Srivastava R, Geng D, Liu Y, Zheng L, Li Z, Joseph MA, et al. Augmentation of therapeutic responses in melanoma by inhibition of IRAK-1,-4. Cancer Res. 2012;72:6209–16. doi: 10.1158/0008-5472.CAN-12-0337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Johrer K, Pleyer L, Olivier A, Maizner E, Zelle-Rieser C, Greil R. Tumour-immune cell interactions modulated by chemokines. Expert Opin Biol Ther. 2008;8:269–90. doi: 10.1517/14712598.8.3.269. [DOI] [PubMed] [Google Scholar]
- 31.Moon EK, Carpenito C, Sun J, Wang LC, Kapoor V, Predina J, et al. Expression of a functional CCR2 receptor enhances tumor localization and tumor eradication by retargeted human T cells expressing a mesothelin-specific chimeric antibody receptor. Clin Cancer Res. 2011;17:4719–30. doi: 10.1158/1078-0432.CCR-11-0351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Toh B, Wang X, Keeble J, Sim WJ, Khoo K, Wong WC, et al. Mesenchymal transition and dissemination of cancer cells is driven by myeloid-derived suppressor cells infiltrating the primary tumor. PLoS Biol. 2011;9:e1001162. doi: 10.1371/journal.pbio.1001162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Srivastava MK, Zhu L, Harris-White M, Kar UK, Huang M, Johnson MF, et al. Myeloid suppressor cell depletion augments antitumor activity in lung cancer. PLoS ONE. 2012;7:e40677. doi: 10.1371/journal.pone.0040677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Leigh ND, Bian G, Ding X, Liu H, Aygun-Sunar S, Burdelya LG, et al. A flagellin-derived toll-like receptor 5 agonist stimulates cytotoxic lymphocyte-mediated tumor immunity. PLoS ONE. 2014;9:e85587. doi: 10.1371/journal.pone.0085587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Weimer ET, Ervin SE, Wozniak DJ, Mizel SB. Immunization of young African green monkeys with OprF epitope 8-OprI-type A- and B-flagellin fusion proteins promotes the production of protective antibodies against nonmucoid Pseudomonas aeruginosa. Vaccine. 2009;27:6762–9. doi: 10.1016/j.vaccine.2009.08.080. [DOI] [PubMed] [Google Scholar]
- 36.Llewellyn M, Dutton RJ, Easter J, O'donnol D, Gober JW. The conserved flaF gene has a critical role in coupling flagellin translation and assembly in Caulobacter crescentus. Mol Microbiol. 2005;57:1127–42. doi: 10.1111/j.1365-2958.2005.04745.x. [DOI] [PubMed] [Google Scholar]
- 37.Jones RM, Sloane VM, Wu H, Luo L, Kumar A, Kumar MV, et al. Flagellin administration protects gut mucosal tissue from irradiation-induced apoptosis via MKP-7 activity. Gut. 2011;60:648–57. doi: 10.1136/gut.2010.223891. [DOI] [PubMed] [Google Scholar]
- 38.Lin WJ, Yeh WC. Implication of Toll-like receptor and tumor necrosis factor alpha signaling in septic shock. Shock. 2005;24:206–9. doi: 10.1097/01.shk.0000180074.69143.77. [DOI] [PubMed] [Google Scholar]
- 39.Honko AN, Mizel SB. Effects of flagellin on innate and adaptive immunity. Immunol Res. 2005;33:83–101. doi: 10.1385/IR:33:1:083. [DOI] [PubMed] [Google Scholar]
- 40.Vicente-Suarez I, Brayer J, Villagra A, Cheng F, Sotomayor EM. TLR5 ligation by flagellin converts tolerogenic dendritic cells into activating antigen-presenting cells that preferentially induce T-helper 1 responses. Immunol Lett. 2009;125:114–8. doi: 10.1016/j.imlet.2009.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vicente-Suarez I, Takahashi Y, Cheng F, Horna P, Wang HW, Wang HG, et al. Identification of a novel negative role of flagellin in regulating IL-10 production. Eur J Immunol. 2007;37:3164–75. doi: 10.1002/eji.200737306. [DOI] [PubMed] [Google Scholar]
- 42.Kratz F, Warnecke A. Finding the optimal balance: challenges of improving conventional cancer chemotherapy using suitable combinations with nano-sized drug delivery systems. J Control Release. 2012;164:221–35. doi: 10.1016/j.jconrel.2012.05.045. [DOI] [PubMed] [Google Scholar]
- 43.Feuillet V, Medjane S, Mondor I, Demaria O, Pagni PP, Galan JE, et al. Involvement of Toll-like receptor 5 in the recognition of flagellated bacteria. Proc Natl Acad Sci U S A. 2006;103:12487–92. doi: 10.1073/pnas.0605200103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Maaser C, Heidemann J, von EC, Lugering A, Spahn TW, Binion DG, et al. Human intestinal microvascular endothelial cells express Toll-like receptor 5: a binding partner for bacterial flagellin. J Immunol. 2004;172:5056–62. doi: 10.4049/jimmunol.172.8.5056. [DOI] [PubMed] [Google Scholar]
- 45.Means TK, Hayashi F, Smith KD, Aderem A, Luster AD. The Toll-like receptor 5 stimulus bacterial flagellin induces maturation and chemokine production in human dendritic cells. J Immunol. 2003;170:5165–75. doi: 10.4049/jimmunol.170.10.5165. [DOI] [PubMed] [Google Scholar]
- 46.Hayashi F, Smith KD, Ozinsky A, Hawn TR, Yi EC, Goodlett DR, et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature. 2001;410:1099–103. doi: 10.1038/35074106. [DOI] [PubMed] [Google Scholar]
- 47.Gewirtz AT, Navas TA, Lyons S, Godowski PJ, Madara JL. Cutting edge: bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J Immunol. 2001;167:1882–5. doi: 10.4049/jimmunol.167.4.1882. [DOI] [PubMed] [Google Scholar]
- 48.Galli R, Starace D, Busa R, Angelini DF, Paone A, De CP, et al. TLR stimulation of prostate tumor cells induces chemokine-mediated recruitment of specific immune cell types. J Immunol. 2010;184:6658–69. doi: 10.4049/jimmunol.0902401. [DOI] [PubMed] [Google Scholar]
- 49.Zoglmeier C, Bauer H, Norenberg D, Wedekind G, Bittner P, Sandholzer N, et al. CpG blocks immunosuppression by myeloid-derived suppressor cells in tumor-bearing mice. Clin Cancer Res. 2011;17:1765–75. doi: 10.1158/1078-0432.CCR-10-2672. [DOI] [PubMed] [Google Scholar]
- 50.Arihara F, Mizukoshi E, Kitahara M, Takata Y, Arai K, Yamashita T, et al. Increase in CD14+HLA-DR -/low myeloid-derived suppressor cells in hepatocellular carcinoma patients and its impact on prognosis. Cancer Immunol Immunother. 2013;62:1421–30. doi: 10.1007/s00262-013-1447-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Diaz-Montero CM, Salem ML, Nishimura MI, Garrett-Mayer E, Cole DJ, Montero AJ. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol Immunother. 2009;58:49–59. doi: 10.1007/s00262-008-0523-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kerkar SP, Goldszmid RS, Muranski P, Chinnasamy D, Yu Z, Reger RN, et al. IL-12 triggers a programmatic change in dysfunctional myeloid-derived cells within mouse tumors. J Clin Invest. 2011;121:4746–57. doi: 10.1172/JCI58814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang L, Kerkar SP, Yu Z, Zheng Z, Yang S, Restifo NP, et al. Improving adoptive T cell therapy by targeting and controlling IL-12 expression to the tumor environment. Mol Ther. 2011;19:751–9. doi: 10.1038/mt.2010.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.