Abstract
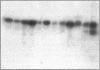
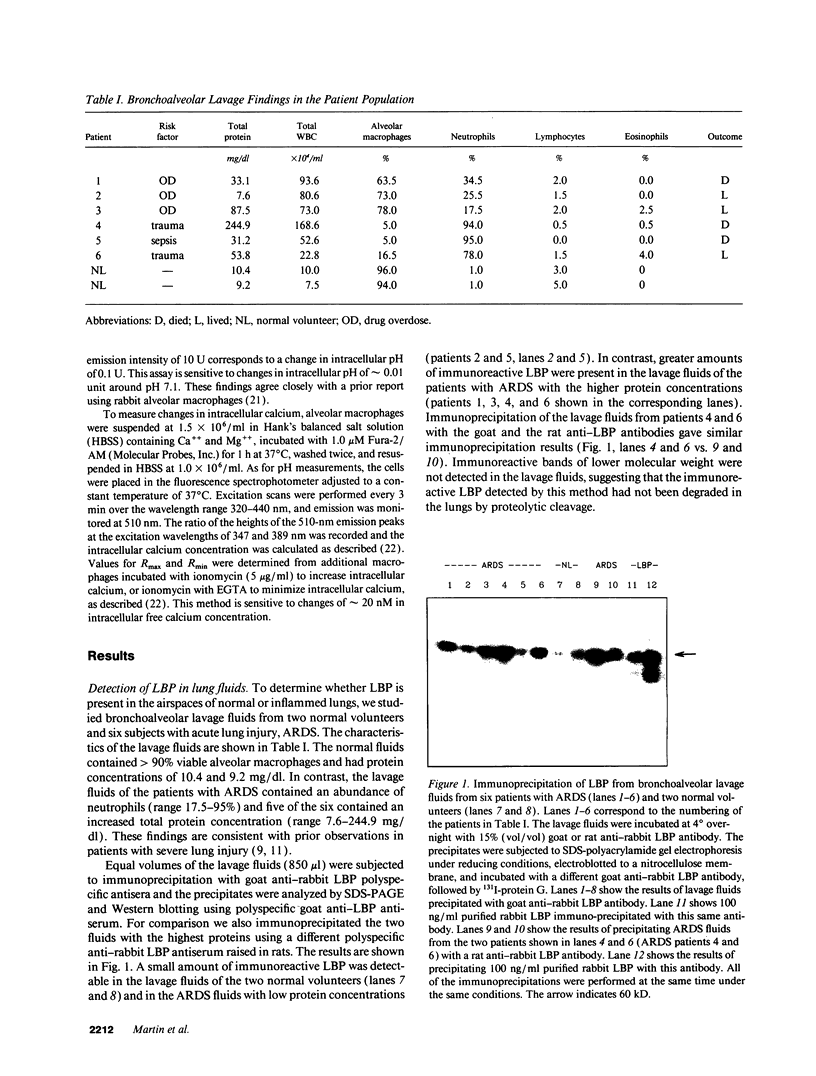
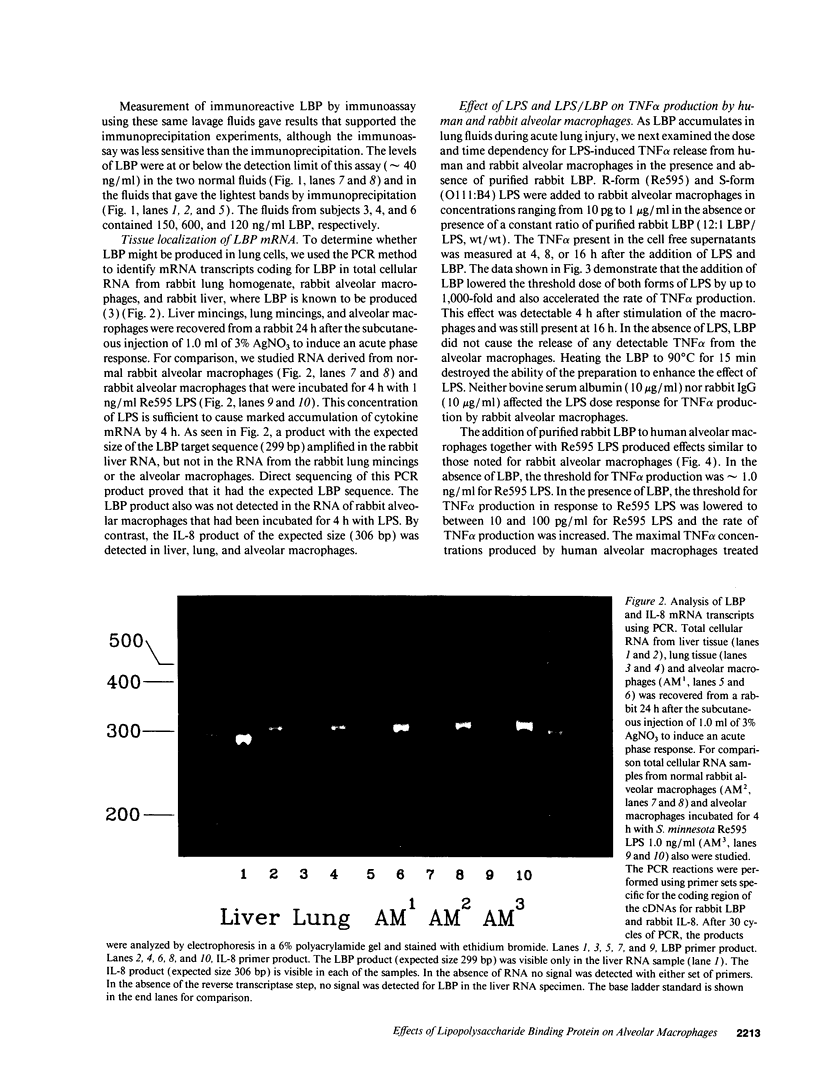
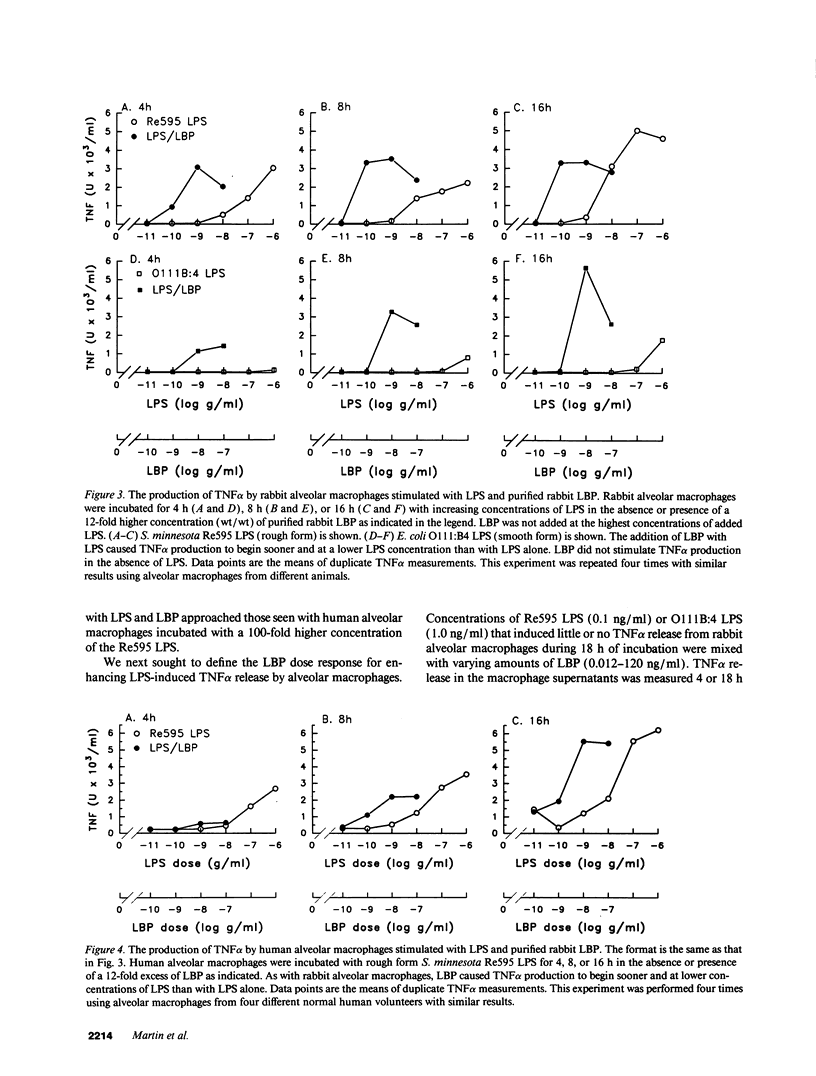
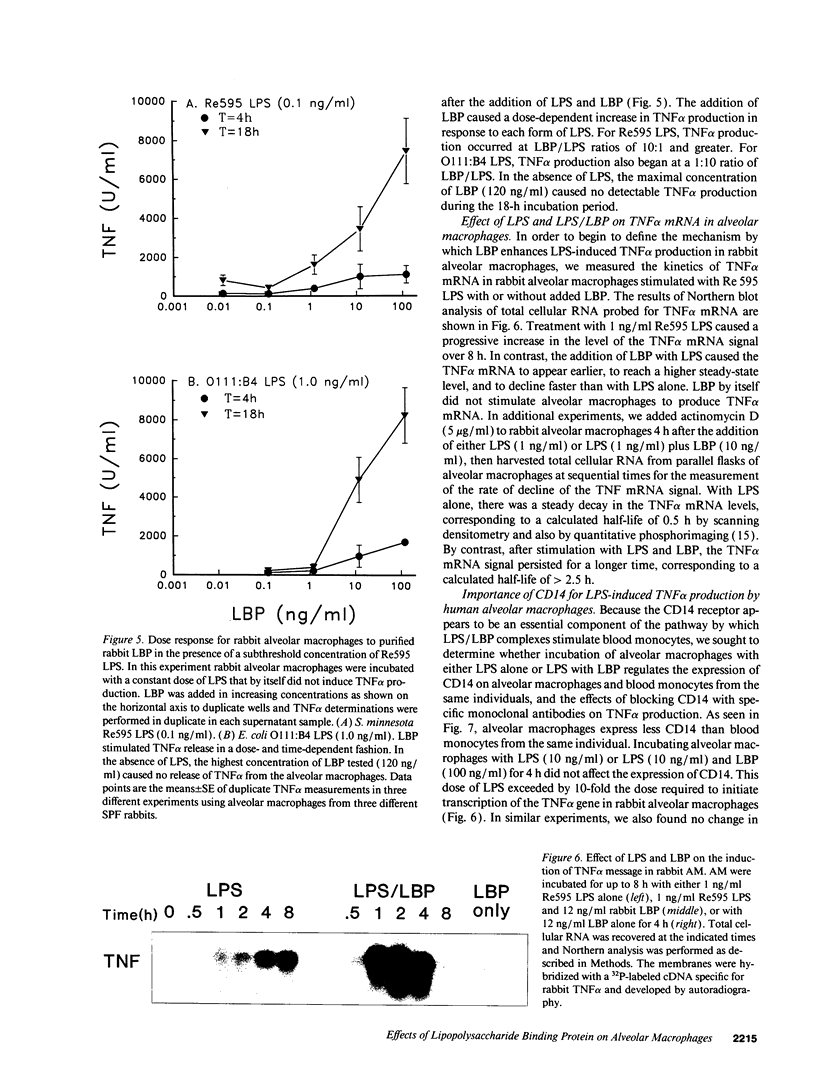
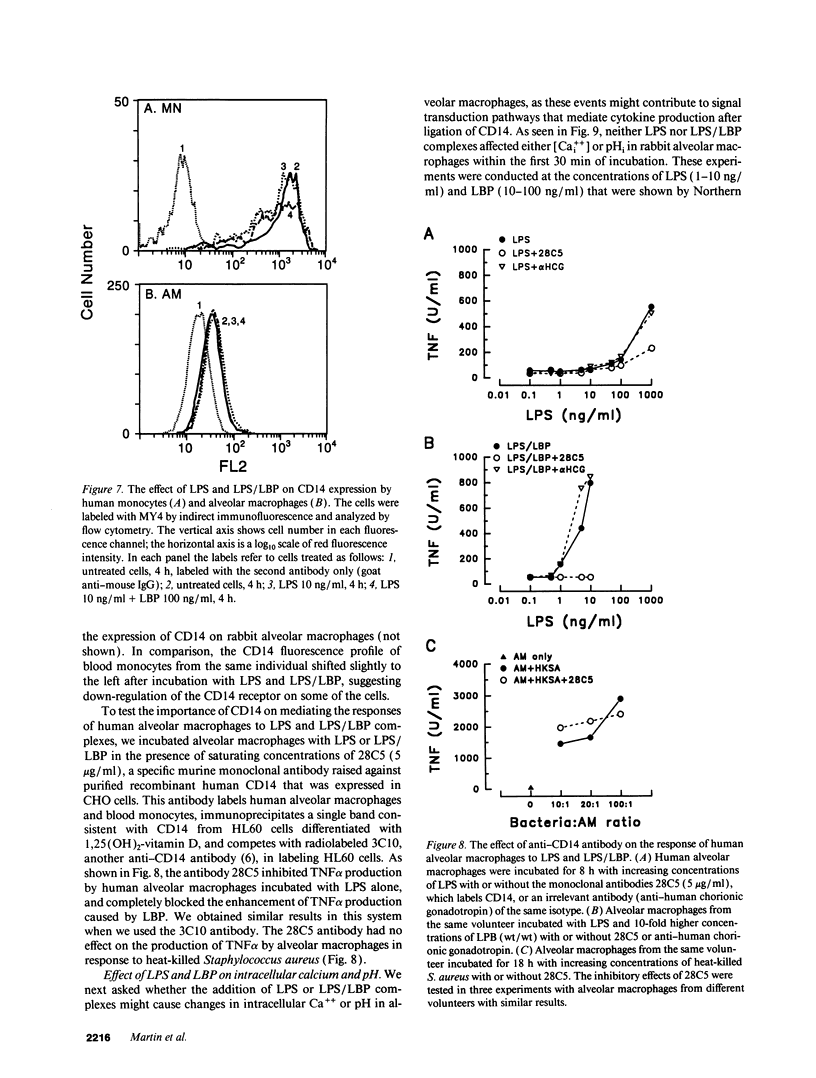
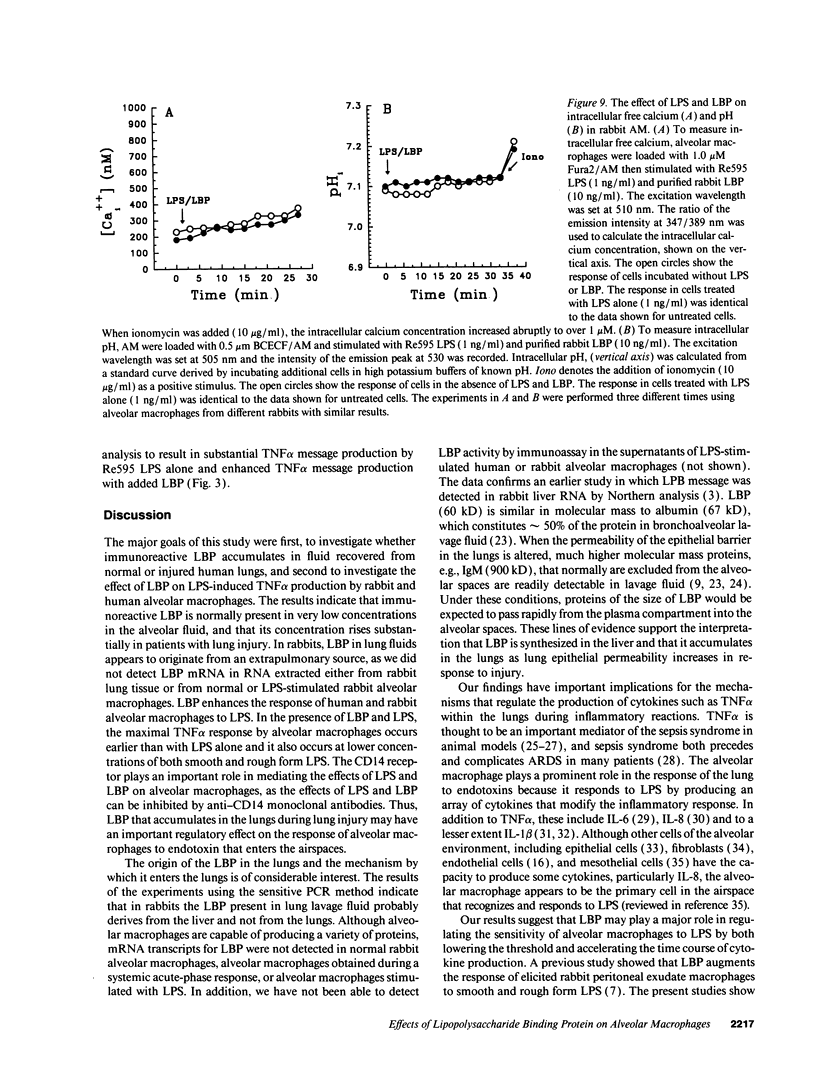
A plasma lipopolysaccharide (LPS)-binding protein (LBP) has been shown to regulate the response of rabbit peritoneal macrophages and human blood monocytes to endotoxin (LPS). We investigated whether LBP is present in lung fluids and the effects of LBP on the response of lung macrophages to LPS. Immunoreactive LBP was detectable in the lavage fluids of patients with the adult respiratory distress syndrome by immunoprecipitation followed by Western blotting, and also by specific immunoassay. In rabbits, the LBP appeared to originate outside of the lungs, inasmuch as mRNA transcripts for LBP were identified in total cellular RNA from liver, but not from lung homogenates or alveolar macrophages. Purified LBP enhanced the response of human and rabbit alveolar macrophages to both smooth form LPS (Escherichia coli O111B:4) and rough form LPS (Salmonella minnesota Re595). In the presence of LBP and LPS, the onset of tumor necrosis factor-alpha (TNF alpha) production occurred earlier and at an LPS threshold dose that was as much as 1,000-fold lower for both types of LPS. In rabbit alveolar macrophages treated with LBP and LPS, TNF alpha mRNA appeared earlier, reached higher levels, and had a prolonged half-life as compared with LPS treatment alone. Neither LPS nor LPS and LBP affected pHi or [Cai++] in alveolar macrophages. Specific monoclonal antibodies to CD14, a receptor that binds LPS/LBP complexes, inhibited TNF alpha production by human alveolar macrophages stimulated with LPS alone or with LPS/LBP complexes, indicating the importance of CD14 in mediating the effects of LPS on alveolar macrophages. Thus, immunoreactive LBP accumulates in lung lavage fluids in patients with lung injury and enhances LPS-stimulated TNF alpha gene expression in alveolar macrophages by a pathway that depends on the CD14 receptor. LBP may play an important role in augmenting TNF alpha expression by alveolar macrophages within the lungs.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beatty P. G., Ledbetter J. A., Martin P. J., Price T. H., Hansen J. A. Definition of a common leukocyte cell-surface antigen (Lp95-150) associated with diverse cell-mediated immune functions. J Immunol. 1983 Dec;131(6):2913–2918. [PubMed] [Google Scholar]
- Bernaudin J. F., Yamauchi K., Wewers M. D., Tocci M. J., Ferrans V. J., Crystal R. G. Demonstration by in situ hybridization of dissimilar IL-1 beta gene expression in human alveolar macrophages and blood monocytes in response to lipopolysaccharide. J Immunol. 1988 Jun 1;140(11):3822–3829. [PubMed] [Google Scholar]
- Beutler B., Milsark I. W., Cerami A. C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science. 1985 Aug 30;229(4716):869–871. doi: 10.1126/science.3895437. [DOI] [PubMed] [Google Scholar]
- Beutler B., Thompson P., Keyes J., Hagerty K., Crawford D. Assay of a ribonuclease that preferentially hydrolyses mRNAs containing cytokine-derived UA-rich instability sequences. Biochem Biophys Res Commun. 1988 May 16;152(3):973–980. doi: 10.1016/s0006-291x(88)80379-6. [DOI] [PubMed] [Google Scholar]
- Bidani A., Brown S. E., Heming T. A., Gurich R., Dubose T. D., Jr Cytoplasmic pH in pulmonary macrophages: recovery from acid load is Na+ independent and NEM sensitive. Am J Physiol. 1989 Jul;257(1 Pt 1):C65–C76. doi: 10.1152/ajpcell.1989.257.1.C65. [DOI] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Couturier C., Haeffner-Cavaillon N., Caroff M., Kazatchkine M. D. Binding sites for endotoxins (lipopolysaccharides) on human monocytes. J Immunol. 1991 Sep 15;147(6):1899–1904. [PubMed] [Google Scholar]
- Golenbock D. T., Hampton R. Y., Raetz C. R., Wright S. D. Human phagocytes have multiple lipid A-binding sites. Infect Immun. 1990 Dec;58(12):4069–4075. doi: 10.1128/iai.58.12.4069-4075.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman R. B., Wood R. G., Martin T. R., Hanson-Painton O., Kinasewitz G. T. Cytokine-stimulated human mesothelial cells produce chemotactic activity for neutrophils including NAP-1/IL-8. J Immunol. 1992 Jan 15;148(2):457–465. [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Halling J. L., Hamill D. R., Lei M. G., Morrison D. C. Identification and characterization of lipopolysaccharide-binding proteins on human peripheral blood cell populations. Infect Immun. 1992 Mar;60(3):845–852. doi: 10.1128/iai.60.3.845-852.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han J., Brown T., Beutler B. Endotoxin-responsive sequences control cachectin/tumor necrosis factor biosynthesis at the translational level. J Exp Med. 1990 Feb 1;171(2):465–475. doi: 10.1084/jem.171.2.465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han J., Huez G., Beutler B. Interactive effects of the tumor necrosis factor promoter and 3'-untranslated regions. J Immunol. 1991 Mar 15;146(6):1843–1848. [PubMed] [Google Scholar]
- Hance A. J., Douches S., Winchester R. J., Ferrans V. J., Crystal R. G. Characterization of mononuclear phagocyte subpopulations in the human lung by using monoclonal antibodies: changes in alveolar macrophage phenotype associated with pulmonary sarcoidosis. J Immunol. 1985 Jan;134(1):284–292. [PubMed] [Google Scholar]
- Haziot A., Chen S., Ferrero E., Low M. G., Silber R., Goyert S. M. The monocyte differentiation antigen, CD14, is anchored to the cell membrane by a phosphatidylinositol linkage. J Immunol. 1988 Jul 15;141(2):547–552. [PubMed] [Google Scholar]
- Holter J. F., Weiland J. E., Pacht E. R., Gadek J. E., Davis W. B. Protein permeability in the adult respiratory distress syndrome. Loss of size selectivity of the alveolar epithelium. J Clin Invest. 1986 Dec;78(6):1513–1522. doi: 10.1172/JCI112743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoogsteden H. C., van Dongen J. J., van Hal P. T., Delahaye M., Hop W., Hilvering C. Phenotype of blood monocytes and alveolar macrophages in interstitial lung disease. Chest. 1989 Mar;95(3):574–577. doi: 10.1378/chest.95.3.574. [DOI] [PubMed] [Google Scholar]
- Johnston R. F., Pickett S. C., Barker D. L. Autoradiography using storage phosphor technology. Electrophoresis. 1990 May;11(5):355–360. doi: 10.1002/elps.1150110503. [DOI] [PubMed] [Google Scholar]
- Kotloff R. M., Little J., Elias J. A. Human alveolar macrophage and blood monocyte interleukin-6 production. Am J Respir Cell Mol Biol. 1990 Nov;3(5):497–505. doi: 10.1165/ajrcmb/3.5.497. [DOI] [PubMed] [Google Scholar]
- Lauener R. P., Geha R. S., Vercelli D. Engagement of the monocyte surface antigen CD14 induces lymphocyte function-associated antigen-1/intercellular adhesion molecule-1-dependent homotypic adhesion. J Immunol. 1990 Sep 1;145(5):1390–1394. [PubMed] [Google Scholar]
- Lee J. D., Kato K., Tobias P. S., Kirkland T. N., Ulevitch R. J. Transfection of CD14 into 70Z/3 cells dramatically enhances the sensitivity to complexes of lipopolysaccharide (LPS) and LPS binding protein. J Exp Med. 1992 Jun 1;175(6):1697–1705. doi: 10.1084/jem.175.6.1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low M. G., Saltiel A. R. Structural and functional roles of glycosyl-phosphatidylinositol in membranes. Science. 1988 Jan 15;239(4837):268–275. doi: 10.1126/science.3276003. [DOI] [PubMed] [Google Scholar]
- Martin T. R., Pistorese B. P., Chi E. Y., Goodman R. B., Matthay M. A. Effects of leukotriene B4 in the human lung. Recruitment of neutrophils into the alveolar spaces without a change in protein permeability. J Clin Invest. 1989 Nov;84(5):1609–1619. doi: 10.1172/JCI114338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin T. R., Pistorese B. P., Hudson L. D., Maunder R. J. The function of lung and blood neutrophils in patients with the adult respiratory distress syndrome. Implications for the pathogenesis of lung infections. Am Rev Respir Dis. 1991 Aug;144(2):254–262. doi: 10.1164/ajrccm/144.2.254. [DOI] [PubMed] [Google Scholar]
- Martin T. R., Raugi G., Merritt T. L., Henderson W. R., Jr Relative contribution of leukotriene B4 to the neutrophil chemotactic activity produced by the resident human alveolar macrophage. J Clin Invest. 1987 Oct;80(4):1114–1124. doi: 10.1172/JCI113168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathison J. C., Tobias P. S., Wolfson E., Ulevitch R. J. Plasma lipopolysaccharide (LPS)-binding protein. A key component in macrophage recognition of gram-negative LPS. J Immunol. 1992 Jul 1;149(1):200–206. [PubMed] [Google Scholar]
- Mathison J. C., Virca G. D., Wolfson E., Tobias P. S., Glaser K., Ulevitch R. J. Adaptation to bacterial lipopolysaccharide controls lipopolysaccharide-induced tumor necrosis factor production in rabbit macrophages. J Clin Invest. 1990 Apr;85(4):1108–1118. doi: 10.1172/JCI114542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathison J. C., Wolfson E., Ulevitch R. J. Participation of tumor necrosis factor in the mediation of gram negative bacterial lipopolysaccharide-induced injury in rabbits. J Clin Invest. 1988 Jun;81(6):1925–1937. doi: 10.1172/JCI113540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montgomery A. B., Stager M. A., Carrico C. J., Hudson L. D. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis. 1985 Sep;132(3):485–489. doi: 10.1164/arrd.1985.132.3.485. [DOI] [PubMed] [Google Scholar]
- Ramadori G., Meyer zum Buschenfelde K. H., Tobias P. S., Mathison J. C., Ulevitch R. J. Biosynthesis of lipopolysaccharide-binding protein in rabbit hepatocytes. Pathobiology. 1990;58(2):89–94. doi: 10.1159/000163569. [DOI] [PubMed] [Google Scholar]
- Rankin J. A., Sylvester I., Smith S., Yoshimura T., Leonard E. J. Macrophages cultured in vitro release leukotriene B4 and neutrophil attractant/activation protein (interleukin 8) sequentially in response to stimulation with lipopolysaccharide and zymosan. J Clin Invest. 1990 Nov;86(5):1556–1564. doi: 10.1172/JCI114875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen A., Nairn A. C., Greengard P., Cohn Z. A., Aderem A. Bacterial lipopolysaccharide regulates the phosphorylation of the 68K protein kinase C substrate in macrophages. J Biol Chem. 1989 Jun 5;264(16):9118–9121. [PubMed] [Google Scholar]
- Schoene R. B., Swenson E. R., Pizzo C. J., Hackett P. H., Roach R. C., Mills W. J., Jr, Henderson W. R., Jr, Martin T. R. The lung at high altitude: bronchoalveolar lavage in acute mountain sickness and pulmonary edema. J Appl Physiol (1985) 1988 Jun;64(6):2605–2613. doi: 10.1152/jappl.1988.64.6.2605. [DOI] [PubMed] [Google Scholar]
- Schumann R. R., Leong S. R., Flaggs G. W., Gray P. W., Wright S. D., Mathison J. C., Tobias P. S., Ulevitch R. J. Structure and function of lipopolysaccharide binding protein. Science. 1990 Sep 21;249(4975):1429–1431. doi: 10.1126/science.2402637. [DOI] [PubMed] [Google Scholar]
- Standiford T. J., Kunkel S. L., Basha M. A., Chensue S. W., Lynch J. P., 3rd, Toews G. B., Westwick J., Strieter R. M. Interleukin-8 gene expression by a pulmonary epithelial cell line. A model for cytokine networks in the lung. J Clin Invest. 1990 Dec;86(6):1945–1953. doi: 10.1172/JCI114928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefanová I., Horejsí V., Ansotegui I. J., Knapp W., Stockinger H. GPI-anchored cell-surface molecules complexed to protein tyrosine kinases. Science. 1991 Nov 15;254(5034):1016–1019. doi: 10.1126/science.1719635. [DOI] [PubMed] [Google Scholar]
- Strieter R. M., Kunkel S. L., Showell H. J., Remick D. G., Phan S. H., Ward P. A., Marks R. M. Endothelial cell gene expression of a neutrophil chemotactic factor by TNF-alpha, LPS, and IL-1 beta. Science. 1989 Mar 17;243(4897):1467–1469. doi: 10.1126/science.2648570. [DOI] [PubMed] [Google Scholar]
- Strieter R. M., Phan S. H., Showell H. J., Remick D. G., Lynch J. P., Genord M., Raiford C., Eskandari M., Marks R. M., Kunkel S. L. Monokine-induced neutrophil chemotactic factor gene expression in human fibroblasts. J Biol Chem. 1989 Jun 25;264(18):10621–10626. [PubMed] [Google Scholar]
- Thornton A. J., Strieter R. M., Lindley I., Baggiolini M., Kunkel S. L. Cytokine-induced gene expression of a neutrophil chemotactic factor/IL-8 in human hepatocytes. J Immunol. 1990 Apr 1;144(7):2609–2613. [PubMed] [Google Scholar]
- Tobias P. S., Mathison J. C., Ulevitch R. J. A family of lipopolysaccharide binding proteins involved in responses to gram-negative sepsis. J Biol Chem. 1988 Sep 25;263(27):13479–13481. [PubMed] [Google Scholar]
- Tobias P. S., Soldau K., Ulevitch R. J. Identification of a lipid A binding site in the acute phase reactant lipopolysaccharide binding protein. J Biol Chem. 1989 Jun 25;264(18):10867–10871. [PubMed] [Google Scholar]
- Tobias P. S., Soldau K., Ulevitch R. J. Isolation of a lipopolysaccharide-binding acute phase reactant from rabbit serum. J Exp Med. 1986 Sep 1;164(3):777–793. doi: 10.1084/jem.164.3.777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tracey K. J., Fong Y., Hesse D. G., Manogue K. R., Lee A. T., Kuo G. C., Lowry S. F., Cerami A. Anti-cachectin/TNF monoclonal antibodies prevent septic shock during lethal bacteraemia. Nature. 1987 Dec 17;330(6149):662–664. doi: 10.1038/330662a0. [DOI] [PubMed] [Google Scholar]
- Weiner I. D., Hamm L. L. Use of fluorescent dye BCECF to measure intracellular pH in cortical collecting tubule. Am J Physiol. 1989 May;256(5 Pt 2):F957–F964. doi: 10.1152/ajprenal.1989.256.5.F957. [DOI] [PubMed] [Google Scholar]
- Weinstein S. L., Gold M. R., DeFranco A. L. Bacterial lipopolysaccharide stimulates protein tyrosine phosphorylation in macrophages. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4148–4152. doi: 10.1073/pnas.88.10.4148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wewers M. D., Rennard S. I., Hance A. J., Bitterman P. B., Crystal R. G. Normal human alveolar macrophages obtained by bronchoalveolar lavage have a limited capacity to release interleukin-1. J Clin Invest. 1984 Dec;74(6):2208–2218. doi: 10.1172/JCI111647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wightman P. D., Raetz C. R. The activation of protein kinase C by biologically active lipid moieties of lipopolysaccharide. J Biol Chem. 1984 Aug 25;259(16):10048–10052. [PubMed] [Google Scholar]
- Wright S. D., Ramos R. A., Tobias P. S., Ulevitch R. J., Mathison J. C. CD14, a receptor for complexes of lipopolysaccharide (LPS) and LPS binding protein. Science. 1990 Sep 21;249(4975):1431–1433. doi: 10.1126/science.1698311. [DOI] [PubMed] [Google Scholar]
- Wright S. D., Tobias P. S., Ulevitch R. J., Ramos R. A. Lipopolysaccharide (LPS) binding protein opsonizes LPS-bearing particles for recognition by a novel receptor on macrophages. J Exp Med. 1989 Oct 1;170(4):1231–1241. doi: 10.1084/jem.170.4.1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimura T., Yuhki N. Neutrophil attractant/activation protein-1 and monocyte chemoattractant protein-1 in rabbit. cDNA cloning and their expression in spleen cells. J Immunol. 1991 May 15;146(10):3483–3488. [PubMed] [Google Scholar]