Abstract
The yellow tetrazolium salt 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) is widely used to determine cell viability in cell proliferation and cytotoxic assays. MTT is reduced by metabolically active cells to form an insoluble purple formazan product that is quantifiable by spectrophotometry. It is the most common and direct assay for cell viability. However, in this present study, we demonstrated that the vitamin E isomers α-β-γ-δ-tocotrienols and α-tocopherol were able to reduce MTT into a formazan product, despite the absence of living cells. For comparison, a second method for determining cell viability, which is the neutral red uptake assay, was used in parallel with the MTT assay. The results showed that neutral red did not interact with the vitamin E isomers. Our findings suggest that the MTT assay is not suitable for studying the proliferative effects of vitamin E isomers on cell growth.
Keywords: MTT, Neutral Red Uptake, Vitamin E Isomers, Cell Viability
Abstract
Garam tetrazolium kuning 3-(4, 5-dimetilltiazol-2-il)-2, 5-difenilltetrazolium bromida (MTT) digunakan secara meluas untuk menentukan daya maju sel dalam cerakin-cerakin percambahan sel dan sitotoksik. MTT dikurangkan oleh sel-sel yang aktif secara metabolik untuk membentuk produk formazan ungu yang tidak larut yang boleh dikira secara spektrofotometrik. Ini adalah teknik cerakin yang paling biasa dan terus bagi ujian daya maju sel. Walau bagaimanapun, dalam kajian ini, kami menunjukkan bahawa isomer-isomer vitamin E, iaitu α-β-y-δ-tokotrienol dan α-tokoferol mampu mengurangkan MTT kepada produk formazan tanpa kehadiran sel-sel hidup. Untuk perbandingan, kaedah kedua untuk menentukan daya maju sel, iaitu teknik penyerapan neutral merah telah digunakan secara selari dengan MTT. Hasil kajian menunjukkan bahawa neutral merah tidak berinteraksi dengan isomer-isomer vitamin E. Penemuan kami mencadangkan bahawa teknik cerakin MTT adalah tidak sesuai untuk mengkaji kesan isomer-isomer vitamin E pada pertumbuhan sel-sel.
Keywords: MTT, Penyerapan Neutral Merah, Isomer-isomer Vitamin E, Daya Maju Sel
3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) is a yellow water-soluble tetrazolium salt. MTT is reduced in the mitochondria of metabolically active cells by succinate dehydrogenase to yield a water-insoluble purple formazan crystal (Garn et al. 1994; Thom et al. 1993; Shoemaker et al. 2004; Peng et al. 2005), which can be spectrophotometrically measured upon solubilization. The total amount of formazan produced is directly proportional to the number of viable cells in the culture; thus, the MTT assay has become a standard method used to evaluate cell viability (Kim et al. 2002, 2003; Lin et al. 2003; Shoemaker et al. 2004; Peng et al. 2005).
Another assay that has been used to determine cell viability is the neutral red uptake (NRU) assay. The principle of this assay is based on the ability of viable cells to incorporate and bind the supravital dye neutral red. It is a weak cationic dye that is able to penetrate the cell membranes by non-ionic passive diffusion and concentrates in lysosomes, where it binds anionic and/or phosphate groups of the lysosomal matrix via electrostatic hydrophobic bonds (Winckler 1974; Nemes et al. 1979; Repetto et al. 2008). In fact, the ability of cells to maintain a pH gradient through the production of adenosine triphosphate (ATP) plays a major role in neutral red uptake. Normal physiological pH levels balance the net charge of the dye close to zero so the dye can manage to penetrate through the membranes of the cell. Inside the lysosomes, the proton gradient maintains a lower pH level within the organelles than that of the cytoplasm; therefore, the dye is charged and retained in the lysosomes (Repetto et al. 2008). However, when the cell dies, the pH in the lysosomal matrix becomes imbalanced, and the dye can no longer be retained (Filman et al. 1975; Repetto et al. 2008). Thus, the amount of retained dye is proportional to the number of viable cells.
Vitamin E is found naturally in palm oil, rice bran oil, barley, corn, oats, rye and wheat (McLaughlin & Weihrauch 1979). The vitamin E family is composed of two main subgroups, i.e., tocopherols and tocotrienols; both exist naturally in their alpha (α), beta (β), gamma (γ) and delta (δ) forms. In particular, the tocotrienols of the γ isoform have been studied extensively and are reported to possess antiproliferative effects against several types of cancers, including breast cancer (Takahashi & Loo 2004, Nikolic & Agababa 2009), colon cancer (Xu et al. 2009), gastric adenocarcinoma (Sun et al. 2009), liver cancer (Sakai et al. 2006) and prostate cancer (Yap et al. 2008). In addition, α-tocopherol showed potent growth inhibitory effects against cancerous brain cells following the adjunct treatment of plant extracts (Lim et al. 2013). These previous studies have stimulated our interest in studying both tocopherols and tocotrienols as potential cancer treatments. For instance, we have initiated research to investigate the growth inhibition of these isomers in cancerous cell lines using common cell viability assays, as mentioned above.
This paper describes the effects of MTT and NRU assays on cancerous cells treated with vitamin E isomers and demonstrates a better choice of cell viability assessment, which is very important in producing reliable findings for anticancer potential of a vitamin E resource.
Palm-based pure vitamin E isomers (α-β-γ-δ-tocotrienols and α-tocopherol) were used as working solutions and prepared in dimethyl sulfoxide (DMSO). DMSO, vinblastine sulfate (assay validation control), MTT and neutral red powder were purchased from Sigma (Missouri, USA). RPMI-1640 medium, Eagle’s Minimum Essential Medium (EMEM), and fetal calf serum were purchased from GIBCO (New York). The attempted cancerous cell lines, i.e., A549 (human lung adenocarcinoma) and U87MG (grade IV human glioblastoma), were cultured and maintained at the conditions described by Lim et al. (2011).
For the MTT test with cells, a total of 5 × 103 cells per well were seeded in a 96-well plate (Orange Scientific, Braine-l’Alleud, Belgium) and incubated for 24 hours. Cells were then treated with vitamin E isomers at different concentrations ranging from 1–100 µM and with vinblastine at concentrations ranging from 0.011–11 µM (0.01 µg/ml–1 µg/ml) for 72 hours. A similar concentration range of vitamin E isomers and incubation period was applied to a 96-well plate containing no cells. MTT was dissolved in phosphate buffered saline (PBS, pH 7.2) to obtain a concentration of 5 mg/ml; 20 µl was added to each well, followed by a 4-hour incubation period. The supernatant in each well was then carefully removed, and 200 µl of DMSO was added to each well. The amount of formazan formed was determined by measuring the absorbance at 570 nm using a Varioskan Flash 96-well microplate reader (Thermo Fisher Scientific, Massachusetts, USA).
The NRU assay was carried out in accordance with the protocols reported by Repetto et al. (2008). Similar to the MTT assay, a total of 5 × 103 cells were seeded per well in the 96-well plates and incubated for 24 hours. Plates with and without cells were then treated with vitamin E isomers and vinblastine at a similar concentration range and incubation period as that used in the MTT assay. Neutral red working solution at a 40 µg/ml concentration was freshly prepared by overnight incubation at 37°C a day before running the assay.
The neutral red medium was centrifuged for 10 minutes at 1800 rpm to remove any precipitated dye crystals. The medium was carefully aspirated off, and a washing step with 150 µl of PBS was carried out. Then, 100 µl of neutral red medium was added to each well, followed by incubation for 2 hours. The neutral red medium was removed, and a washing step with 150 µl of PBS was carried out. Neutral red destaining solution (150 µl) was added to each well. The plate was shaken for at least 10 minutes or until the neutral red dye had been extracted from the cells to form a homogeneous solution. The neutral red extract was then measured at 540 nm using the same microplate reader (Thermo Fisher Scientific, USA).
All cell viability assays were performed in triplicate in three separate experiments with statistical analysis performed. Graphs of percent viability against the concentrations for the vitamin E isomer treated cell lines were plotted. The absorbance (optical density, OD) of the treated samples was compared with the untreated ones, considering the vehicle control (DMSO), to obtain the percent viability curve (ODtreated / ODuntreated × 100%). Data were presented as the mean ± standard deviation. Unpaired or independent Student’s t test and ANOVA were used to compare the treated and untreated groups, and the level of statistical significance was set at p<0.05. Cells were examined daily under a light microscope (Nikon, Tokyo) for growth performance before and after treatment of vitamin E isomers. Following the MTT and NRU procedures, images of the cells were captured.
It is undeniable that both MTT and NRU assays are commonly used in the screening of anticancer drugs in cell lines. Principally, the absorbance value of both assays is proportionally related to cell viability and number.
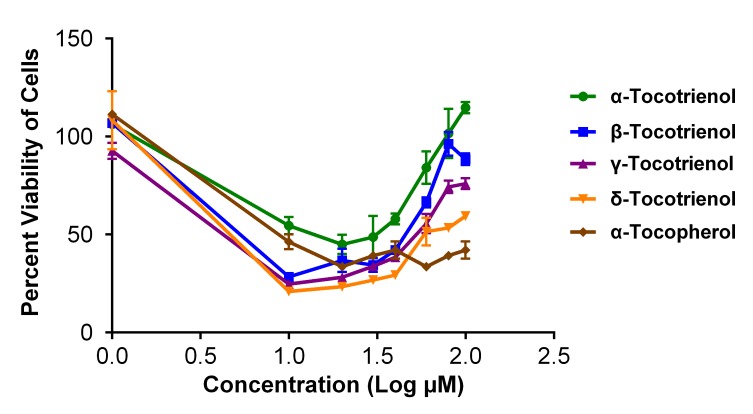
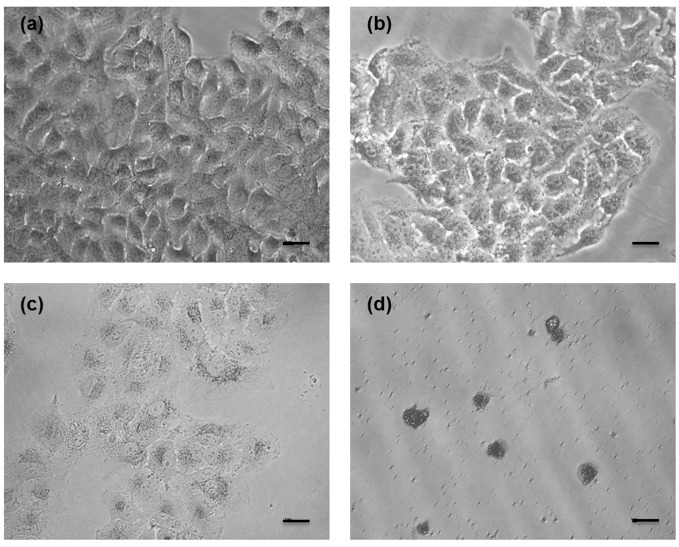
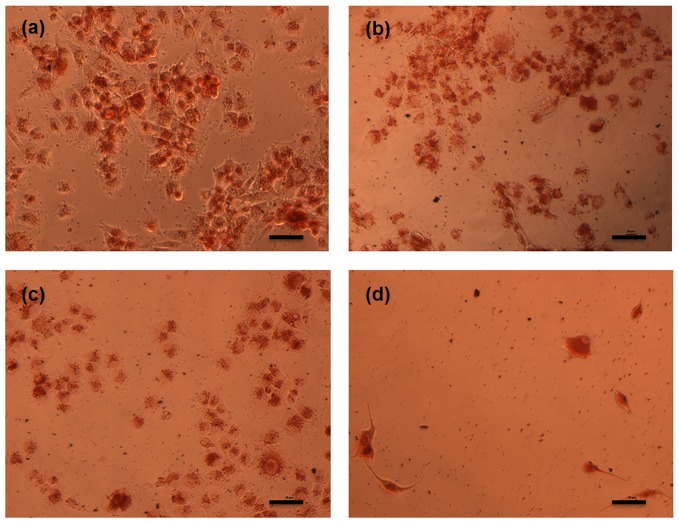
In this study, the vehicle control, i.e., DMSO, did not have any noticeable effects on the cell lines. As shown in Figure 1, the percent viability of cells receiving all vitamin E isomers improved with increasing concentrations. In other words, α-β-γ-δ-tocotrienols had less antiproliferative effects against cancerous cells when a higher concentration was used (Fig. 1). However, these findings did not correspond to the microscopic observations of the cancerous cells prior to the MTT procedure (see the example slides in Fig. 2); in fact, more cell death was evidenced at higher concentrations of vitamin E isomers, as observed in Figure 2.
Figure 1:
MTT results showing the viability percentage of cancerous cells treated with vitamin E isomers, i.e., α-tocotrienol, β-tocotrienol, γ-tocotrienol, δ-tocotrienol and α-tocopherol. All cells treated with vitamin E isomers are observed to exhibit reversed cell viability profiles starting at a concentration of 20 µM. Values are presented as the mean percentage ± SD (n = 3).
Figure 2:
Microscopic observations of unstained cancerous cells receiving different treatments: (a) untreated; (b) treated with 10 µM of δ-tocotrienol; (c) treated with 30 µM of δ-tocotrienol; (d) treated with 50 µM of δ-tocotrienol. An increased concentration of δ-tocotrienol inhibits more cancerous cell growth. Bar: 50 µm.
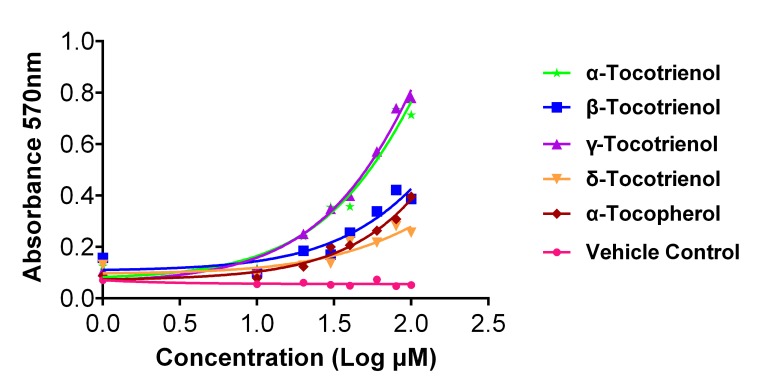
This finding suggests that vitamin E isomers may interfere with MTT directly. To confirm this hypothesis, vitamin E isomers were incubated with MTT in the absence of cells, and the production of the colored formazan product was then measured, as presented in Figure 3. Plots of absorbance values against concentrations (Fig. 3) were used in this case because of the lack of involvement of living cells, which made the generation of percent viability graphs impractical. It can be observed that MTT was markedly reduced into purple formazan with all vitamin E isomers (i.e., α-β-γ-δ-tocotrienol isomers and α-tocopherol) when tested under a cell-free condition. Essentially, the absorbance value increased in a dose-dependent manner for vitamin E isomers. The MTT reduction levels can be ranked in descending order of the vitamin E isomers as follows: γ-tocotrienol > α-tocotrienol > β-tocotrienol > α-tocopherol > δ-tocotrienol.
Figure 3:
Reduction of MTT to purple formazans by α-tocotrienol, β-tocotrienol, γ-tocotrienol, δ-tocotrienol and α-tocopherol in the absence of cells. A descending ranking order of MTT reduction level can be given as following: γ-tocotrienol > α-tocotrienol > β-tocotrienol > α-tocopherol > δ-tocotrienol. Values are expressed as the mean absorbance ± SD (n = 3).
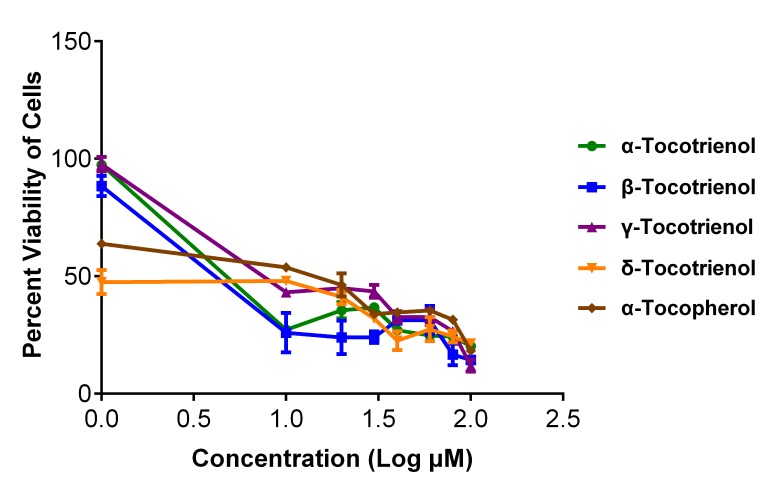
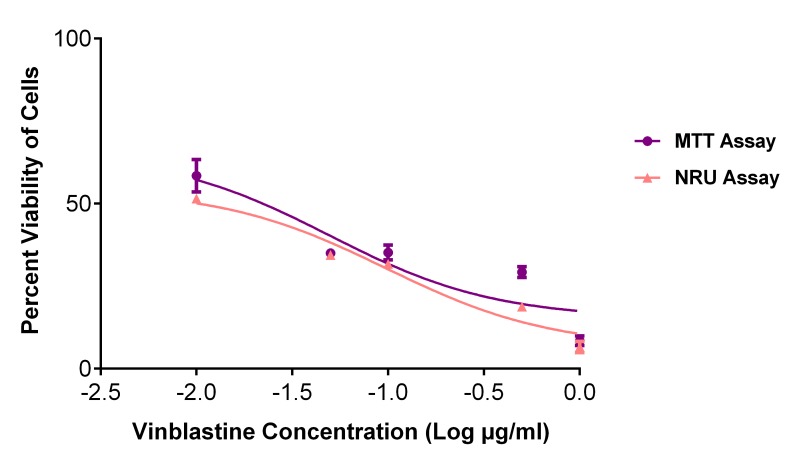
Figure 4 shows the percent viability graphs plotted using the results obtained from the NRU assay. It can be observed that these vitamin E isomers actually exhibited significant antiproliferative activity on cancerous cells when the NRU assay was used to assess cell viability. This finding conformed to the microscopic examination indicating that vitamin E isomers inhibited the growth of cancerous cells in a concentration-dependent manner (see example slides in Figure 5). However, because MTT degradation can also lead to the faulty quantification of absorbance, a validation control assay, i.e., one that used vinblastine, was employed in this study. As shown in Figure 6, the similar cell killing patterns tested by both the MTT and NRU assays imply that MTT degradation is not present. These observations are solely attributable to the capability of vitamin E isomers to reduce MTT into its purple formazan in a cell-free condition.
Figure 4:
Neutral red uptake results showing the viability percentage of cancerous cells treated with α-tocotrienol, β-tocotrienol, γ-tocotrienol, δ-tocotrienol and α-tocopherol. Treatment of vitamin E isomers inhibits cancerous cell growth in a concentration-dependent manner. Values are presented as the mean percentage ± SD (n = 3).
Figure 5:
Microscopic observations of neutral red-stained cancerous cells: (a) untreated; (b) treated with 10 µM of δ-tocotrienol; (c) treated with 30 µM of δ-tocotrienol; (d) treated with 50 µM of δ-tocotrienol. Increased concentrations of δ-tocotrienol inhibit more cancerous cell growth. Bar: 10 µm.
Figure 6:
MTT and neutral red uptake results showing the viability percentage of cancerous cells treated with vinblastine serving as a validation control. Treatment of vinblastine inhibits cancerous cell growth in a concentration-dependent manner. Values are presented as the mean percentage ± SD (n = 3).
Based on these contradicting facts (Fig. 1 versus Fig. 4), the reduction of MTT into purple formazan by vitamin E isomers would definitely jeopardize the accuracy of an experiment. Hence, it is suggested that the MTT assay is not an appropriate option for cell viability determination involving vitamin E isomers. Compared with the MTT assay, the NRU assay is known to present less interference and is generally more sensitive. The NRU assay does not require unstable reagents, such as the tetrazolium salts that are required for the MTT, MTS (3-(4, 5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium), and XTT (2, 3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5[(phenylamino)carbonyl]-2H-tetrazolium hydroxide) viability assays to quantify dehydrogenase activity by the chemical reduction of the salts to formazans (Repetto et al. 2008). In fact, our test of the NRU assay in the absence of cells showed that neutral red did not interact with vitamin E isomers; the absorbance reading of all concentrations of vitamin E isomers remained the same as that of the blank. Thus, this assay would be a more favorable option for measuring cell viability when treated by certain natural products such as vitamin E isomers.
In fact, a direct reaction of the MTT reagent with vitamin E isomers has been reported in previous studies; several different antioxidants in addition to vitamin E, such as ascorbic acid, N-acetylcysteine, kaempferol, and any other thiol-containing compounds that contain free thiol-groups (strong reducing chemical side chain group) or other reducing equivalents, could reduce MTT to produce purple formazan, irrespective of the viability of the cells present (Natarajan et al. 2000, Shoemaker et al. 2004). Because vitamin E isomers belong to a family with great antioxidant properties (Watson & Preedy 2008), this finding might explain their ability to reduce MTT into purple formazan.
In addition, as reported by Peng et al. (2005) and Shoemaker et al. (2004), MTT was found to be reduced by flavonoid compounds and several other aqueous herbal extracts into purple formazans, resulting in a false interpretation of the data. However, not all plant extracts possess the capability to reduce MTT. Our previous study involving the screening of cancerous cell viability treated with plant extracts using the MTT assay was found to be unaffected; the results obtained from the MTT and NRU assays were similar (Lim et al. 2011). Nonetheless, special precautions should always be taken when MTT is used for cell viability tests that involve natural products including plant extracts and vitamins.
Because vitamin E isomers have been discovered to be potential anticancer therapeutic candidates, an appropriate cell viability assay should be conducted because the antiproliferative effects are the first criterion used to determine cancer treatment potential prior to any investigation of the mechanism of action. Because the MTT assay is always used to screen for anticancer drugs, this report can serve as a kind reminder to researchers involved in drug discovery in the area of vitamin E sample testing (not excluding other natural products with high antioxidant levels), where using the MTT assay might not be accurate. An alternative NRU assay is highly suggested to overcome this misinterpretation of cell viability results.
In conclusion, we have shown that vitamin E isomers interact with MTT directly. It is suggested that controls without cells should always be included in experiments studying unknown samples or natural products to validate the data and prevent the false interpretation of a research conclusion.
Acknowledgments
The authors would like to thank the Ministry of Science, Technology and Innovation (MOSTI), Malaysia for financially supporting the first author’s study.
REFERENCES
- Filman DJ, Brawn RJ, Dankler WB. Intracellular supravital stain delocalization as an assay for antibody-dependent complement-mediated cell damage. Journal of Immunological Methods. 1975;6(3):189–207. doi: 10.1016/0022-1759(75)90064-2. [DOI] [PubMed] [Google Scholar]
- Garn H, Krause H, Enzmann V, Drossler K. An improved MTT assay using the electron-coupling agent menadione. Journal of Immunological Methods. 1994;168(2):253–256. doi: 10.1016/0022-1759(94)90062-0. [DOI] [PubMed] [Google Scholar]
- Kim SR, Park MJ, Lee MK, Sung SH, Park EJ. Flavonoids of Inula Britannica protect cultured cortical cells from necrotic cell death by glutamate. Free Radical Biology and Medicine. 2002;32(7):596–604. doi: 10.1016/s0891-5849(02)00751-7. [DOI] [PubMed] [Google Scholar]
- Kim SH, Zo JH, Kim MA, Hwang KK, Chae IH. Naringin suppresses the mitogenic effect of lysophosphatidylcholine on vascular smooth muscle cells. Nutrition Research. 2003;23(12):1671–1683. [Google Scholar]
- Lim SW, Loh HS, Ting KN, Bradshaw TD, Zeenathul NA. Acalypha wilkesiana ethyl acetate extract enhances the in vitro cytotoxic effects of α-tocopherol in human brain and lung cancer cells. International Journal of Bioscience, Biochemistry and Bioinformatics. 2013;3(4):335–340. [Google Scholar]
- Lim SW, Ting KN, Bradshaw TD, Zeenathul NA, Wiart C, Khoo TJ, Lim KH, Loh HS. Acalypha wilkesiana extracts induce apoptosis by causing single strand and double strand DNA breaks. Journal of Ethnopharmacology. 2011;138(2):616–623. doi: 10.1016/j.jep.2011.10.005. [DOI] [PubMed] [Google Scholar]
- Lin HY, Juan SH, Shen SC, Hsu FL, Chen YC. Inhibition of lipopolysaccharide-induced nitric oxide production by flavonoids in RAW264.7 macrophages involved heme oxygenase-1. Biochemical Pharmacology. 2003;66(9):1821–1832. doi: 10.1016/s0006-2952(03)00422-2. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Weihrauch JL. Vitamin E content of foods. Journal of the American Dietetic Association. 1979;75(6):647–665. [PubMed] [Google Scholar]
- Natarajan M, Mohan S, Martinez BR, Meltz ML, Herman TS. Antioxidant compounds interfere with the 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide cytotoxic assay. Cancer Detection and Prevention. 2000;24(5):405–414. [PubMed] [Google Scholar]
- Nemes Z, Dietz R, Luth JB. The pharmacological relevance of vital staining with neutral red. Experientia. 1979;35(11):1475–1476. doi: 10.1007/BF01962793. [DOI] [PubMed] [Google Scholar]
- Nikolic K, Agababa D. Design and QSAR study of analogs of γ-tocotrienol with enhanced antiproliferative activity against human breast cancer cells. Journal of Molecular Graphics and Modelling. 2009;27(7):777–783. doi: 10.1016/j.jmgm.2008.11.007. [DOI] [PubMed] [Google Scholar]
- Peng L, Wang B, Ren P. Reduction of MTT by flavonoids in the absence of cells. Colloids and Surfaces B: Biointerfaces. 2005;45(2):108–111. doi: 10.1016/j.colsurfb.2005.07.014. [DOI] [PubMed] [Google Scholar]
- Repetto G, del Peso A, Zurita JL. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nature Protocols. 2008;3(7):1125–1131. doi: 10.1038/nprot.2008.75. [DOI] [PubMed] [Google Scholar]
- Sakai M, Okabe M, Tachibana H, Yamada K. Apoptosis induction by γ-tocotrienol in human hepatoma Hep3B cells. Journal of Nutritional Biochemistry. 2006;17(10):672–676. doi: 10.1016/j.jnutbio.2005.11.001. [DOI] [PubMed] [Google Scholar]
- Shoemaker M, Cohen I, Campbell M. Reduction of MTT by aqueous herbal extracts in the absence of cells. Journal of Ethnopharmacology. 2004;93(2–3):381–384. doi: 10.1016/j.jep.2004.04.011. [DOI] [PubMed] [Google Scholar]
- Sun WG, Xu W, Liu H, Liu J, Wang Q, Zhou J, Dong F, Chen B. γ-tocotrienol induces mitochondria-mediated apoptosis in human gastric adenocarcinoma SGC-7901 cells. Journal of Nutritional Biochemistry. 2009;20(4):276–284. doi: 10.1016/j.jnutbio.2008.03.003. [DOI] [PubMed] [Google Scholar]
- Takahashi K, Loo G. Disruption of mitochondria during tocotrienol-induced apoptosis in MDA-MB-231 human breast cancer cells. Biochemical Pharmacology. 2004;67(2):315–324. doi: 10.1016/j.bcp.2003.07.015. [DOI] [PubMed] [Google Scholar]
- Thom SM, Horobin RW, Seidler E, Barer MR. Factors affecting the selection and use of tetrazolium salts as cytochemical indicators of microbial viability and activity. Journal of Applied Bacteriology. 1993;74(4):433–443. doi: 10.1111/j.1365-2672.1993.tb05151.x. [DOI] [PubMed] [Google Scholar]
- Watson RR, Preedy VR. Tocotrienols: Vitamin E beyond tocopherols. New York: CRC Press; 2008. p. 6. [Google Scholar]
- Winckler J. Vital staining of lysosomes and other cell organelles of the rat with neutral red. Progress in Histochemistry and Cytochemistry. 1974;6(3):1–91. [PubMed] [Google Scholar]
- Xu WL, Liu JR, Liu HK, Qi GY, Sun XR, Sun WG, Chen BQ. Inhibition of proliferation and induction of apoptosis by γ-tocotrienol in human colon carcinoma HT-29 cells. Nutrition. 2009;25(5):555–566. doi: 10.1016/j.nut.2008.10.019. [DOI] [PubMed] [Google Scholar]
- Yap WN, Chang PN, Han HY, Lee DT, Ling MT, Wong YC, Yap YL. γ-tocotrienol suppresses prostate cancer cell proliferation and invasion through multiple-signalling pathways. British Journal of Cancer. 2008;99(11):1832–1841. doi: 10.1038/sj.bjc.6604763. [DOI] [PMC free article] [PubMed] [Google Scholar]