Abstract
While the isolated responses of marine phytoplankton to climate warming and to ocean acidification have been studied intensively, studies on the combined effect of both aspects of Global Change are still scarce. Therefore, we performed a mesocosm experiment with a factorial combination of temperature (9 and 15°C) and pCO2 (means: 439 ppm and 1040 ppm) with a natural autumn plankton community from the western Baltic Sea. Temporal trajectories of total biomass and of the biomass of the most important higher taxa followed similar patterns in all treatments. When averaging over the entire time course, phytoplankton biomass decreased with warming and increased with CO2 under warm conditions. The contribution of the two dominant higher phytoplankton taxa (diatoms and cryptophytes) and of the 4 most important species (3 diatoms, 1 cryptophyte) did not respond to the experimental treatments. Taxonomic composition of phytoplankton showed only responses at the level of subdominant and rare species. Phytoplankton cell sizes increased with CO2 addition and decreased with warming. Both effects were stronger for larger species. Warming effects were stronger than CO2 effects and tended to counteract each other. Phytoplankton communities without calcifying species and exposed to short-term variation of CO2 seem to be rather resistant to ocean acidification.
Introduction
The well known increase of atmospheric CO2 does not only lead to climate warming because of the greenhouse effect but also to “ocean acidification”, i.e. an increase of dissolved CO2, a decrease of water pH and a decrease in the saturation state of calcium carbonates in the ocean. Current predictions for atmospheric CO2 assume an increase from approximately 390 ppm to 700 ppm by the end of 21st Century (RCP8.5 scenario of the IPCC report 2013 [1]). The same scenario also predicts temperature increases of up to 6°C. The predicted increase in CO2 will lead to a further pH decrease by 0.3–0.4 units [2], while until today, ocean pH has declined by 0.1 units from pre-industrial level and carbonate ion concentration have decreased by 30% [3]. The shift in carbonate chemistry is primarily a stressor for organisms with skeletal calcium carbonate structures [4], i.e. among phytoplankton coccolithophores should be affected most strongly [5,6,7].
For non-calcifying phytoplankton, however, CO2 might be a limiting resource because of the low pCO2 under ocean pH and the low affinity of the enzyme RubisCO for dissolved CO2 [8]. While phytoplankton have evolved carbon-concentrating mechanisms (CCM) to overcome this problem [9,10,11], increased CO2 concentration might still be beneficial because they could help to save metabolic costs for CCMs. Indeed, Riebesell [12] found enhanced carbon assimilation of phytoplankton in mesocosms receiving enhanced CO2 concentrations. There is some evidence, that CCM efficiency and regulation differs between taxa [13,14] which should lead to the prediction, that phytoplankton composition is sensitive to CO2-enrichment if CO2 is a limiting factor. In a review article, [15] it was proposed that coccolithophores should be negatively affected by increasing CO2, for diatoms it should be neutral or slightly beneficial, and for N2-fixing cyanobacteria strongly beneficial. Tortell [16] found a shift from small pennate diatoms (Pseudo-nitzschia subcurvata G.A. Fryxell) to large centric ones (Chaetoceros spp.) under enhanced CO2. Similarly, Eggers [17] found that CO2 enrichment could favour large diatoms (Chaetoceros sp., Thalassiosira constricta Gaardner) depending on initial species composition. In contrast, only subtle changes in species composition were reported in [18]. A recent review of ocean acidification effects on marine pelagic microbes [19] emphasizes an insufficient state of knowledge, conflicting results between different studies and a tendency of effects to be rather subtle. The latter is inter alia explained by the fact, that present day seawater pCO2 and pH undergo short-term and seasonal fluctuations which often exceed the expected increase of the atmospheric input.
The previously mentioned review [19] also emphasizes the need to understand the joint effects of ocean acidification with other aspect of Global Change, in particular climate warming. We have accepted this challenge and designed one of the first large volume (1400 L) mesocosm experiments with a crossed factorial design of temperature and CO2 enrichment (but see [20] for a smaller scale experiment). The experiment was performed in area where phytoplankton contains no coccolithophores and where plankton is already now subject to strong short term and seasonal variations in pCO2 (summer and autumn maxima ca. 2300 ppm, summer and autumn means ca. 700 ppm [21]). Therefore, we expected no direct detrimental effect of CO2 on phytoplankton, while indirect food-web effects could not be excluded a priori. The predictions for the effects of the temperature treatment were derived from previous experiments performed at the same site, but in a different season (summarized in [22]). We extended the analysis from species shifts to size shifts within species, because several experiments have indicated a shrinkage of cell size with warming [23,24] and nutrient stress [25]. We chose autumn as the season of our experiment, because it is usually the period of maximal phytoplankton diversity in our region, in particular because of the mixture between diatoms and flagellates of various phylogenetic origin. Our working hypotheses are:
Warming will lead to decreased phytoplankton biomass, in agreement with previous experimental results and generally explained by a stronger effect of warming on loss terms than on photosynthesis, as summarized in [22].
Addition of CO2 will lead to increased phytoplankton biomass, because acidification sensitive coccolithophores [5,6] are lacking in the Baltic Sea and CO2 might be a limiting resource [6,12].
Warming will lead to shifts in taxonomic composition of phytoplankton as observed in previous experiments [summarized in 22].
Addition of CO2 will lead to taxonomic shifts in phytoplankton because of taxonomic differences in CCM [13,14] and of previous experimental results [15,17, but see 18].
Warming will lead to smaller cell sizes of phytoplankton and heterotrophic nanoflagellates in agreement with previous experimental findings [23,24]
Addition of CO2 will lead to bigger cell sizes of phytoplankton, but not of heterotrophic protists. We expect this response because of the analogy to the limiting nutrient effect on phytoplankton cell size [25] while CO2 is not a source of nutrient for heterotrophic nanoflagellates.
Material and Methods
The field samples taken for our study did not involve protected species and were not taken from a protected area. No permissions were required.
Experimental design
The experiment consisted of 12 mesocosms in temperature controlled rooms of GEOMAR-Kiel. We employed a fully factorial combination of two temperature levels (9 and 15°C, in situ temperature was 12°C) and two CO2 levels (target values 560 ppm and 1400 ppm CO2). Target values of CO2 were chosen to represent present days annual minima of the Kiel Bight and the mean value expected for 2100 assuming the more pessimistic IPCC-scenarios for 2100 [1]. Each treatment was replicated three times. In the following text, the experimental treatments will be characterized by a two-letter code (CL: cold, low CO2, CH: cold, high CO2, WL: warm, low CO2, WH: warm, high CO2). The mesocosms had a volume of 1400 L. In order to assure a homogenous distribution of plankton and to minimize sedimentation, mesocosms were gently and continuously stirred by a propeller. A series of previous experiments with the same mesocosms [22,26,27] has shown that this treatment did not harm our experimental organisms.
The mesocosms were illuminated by computer controlled light units (GHL Groß Hard- und Softwarelösungen, Lampunit HL3700 and ProfiluxII) consisting of 5 HIBay-LED spotlights (purpose build item of Econlux, each 100 W) above each mesocosm. The lamps cover the entire PAR spectrum and have emission peaks close to the blue absorption peak of chlorophyll a and to the absorption peak of xanthophylls. Daily irradiance patterns were set to follow the pattern for a cloudless 21 October at Kiel (calculated according [28]) reduced to 50% to account for moderate under water light attenuation. The day-night cycle was 11h50 min: 12h10 min and noon irradiance in the middle of the water column was 252.3 μmol quanta m-2 s-1 PAR at the start of the experiment. The impact of temporal changes in underwater light attenuation due to growth and decline of phytoplankton biomass was minor, because the mesocosms were only 1 m deep.
The mesocosms were filled with unfiltered natural seawater (salinity: 19.7, in situ SST 12°C) from Kiel Bight, Western Baltic Sea (54°19’45.99”N, 10°08’58.19”E) near the dock of the GEOMAR West Shore Building on 19 October 2012. The water contained the natural autumn community of phytoplankton, bacteria, and protozoa. Mesozooplankton were added from net catches with a target density of 20 individuals L-1, mimicking seasonal maximum levels. CO2 was manipulated by a flow-through of air-CO2-mixtures with 560 and 2400 ppm CO2 through the head-space between the water level and the cover of the mesocosms at a rate of 30–60 L h-1. Because of incomplete equilibration between the headspace and the water body and because of photosynthetic CO2-consumption mean values of pCO2 in the water were 439 ppm (sd = 187) and for high CO2 1040 ppm (sd = 210) with peak values of 686 ppm and 1400 ppm. In the high CO2-treatments biological CO2-drawdown was counterbalanced by adding the required amount of CO2-saturated filtered mesocosm water at days 7, 12, and 19. The low CO2-mesocosms received the same amount of filtered water, but without CO2 addition. Full divergence of temperature and CO2 between the treatments was reached on 22 October, henceforth called day 0, while the filling day will be called day -3. The experiment was terminated on 12 November.
Samples
Water temperature, salinity and pH were monitored daily. DIC and total alkalinity were measured and pCO2 was calculated 3 times per week. Details of the methodology and of the temporal change of pH and the carbonate system are available from the data archive and will be published by C. Paul et al. (submitted).
Sampling for phytoplankton took place on three times per week on Mondays, Wednesdays, and Fridays. Samples were taken with a bucket after mixing the mesocosms. Samples for phytoplankton >5 μm were fixed with Lugol’s iodine for subsequent identification, counting (>100 individuals for common taxa), and sizing by inverted microscopy [29]. For phytoplankton <5 μm, 20 ml water samples pre-filtered with a 64 μm mesh were fixed with formaldehyde, incubated, filtered onto black Nuclepore filters (0.8 μm pore size) and stained with DAPI [30] within a few hours of sampling and stored at -80°C until counting and sizing. Filters were examined using fluorescence microscopy under blue and green excitation at 1000X. Dimensions were measured on freeze-frame micrographs of individuals using the Nikon DS-L1 digital camera tools (Nikon, Tokyo, Japan). An unfixed aliquot was immediately analysed by a flow cytometer (FACScalibur, Becton Dickinson) and distinguished on the basis of pigment fluorescence (chlorophyll a, phycoerythrine) and size (equivalent spherical diameter). Flow-cytometric categories were matched to taxa identified by fluorescence microscopy on the basis of size and correlations between abundances. In addition, small phytoplankton not identifiable by microscopy were identified by means of 18S rRNA gene tag pyrosequencing according to [31].
Sizing of phytoplankton was performed at three levels of effort. The flow cytometer delivered size data for each sample individually. Common phytoplankton taxa >5 μm were sized individually for each mesocosm at the time of their abundance peak. Twenty random cells were measured per sample. Taxa which were too rare for this kind of measurement were measured in a composed sample from all mesocosms taken at 2 November (day 12) and, therefore, not used for the analysis of the size response of individual species. Their size data were only used for calculating biomass and together contributed <5% to total biomass. Appropriate geometric standard figures were used to calculate cell volumes from linear measurements [32]. We also measured the sizes of three sufficiently abundant heterotrophic nanoflagellates in order to see, whether the heterotrophic nanoflagellates would respond differently to the experimental treatments than phytoplankton.
Statistical analysis
The responses to temperature and CO2 were analyzed by two-factor ANOVA. In each ANOVA, each mesocosms was represented by a single value (usually the mean over time) in order to avoid pseudoreplication. Biovolume (B) and individual cell volume (V) data were log-transformed and relative biomass data of taxa (pi = Bi/Btot) were arcsine-square root transformed. We also calculated effect sizes for temperature (ET) and CO2 (Ec) on cell volumes by calculating the log-ratios between the mean values for the different treatment levels, i.e. the mean cell volumes of a given species for the mesocosms receiving the same temperarure (ET) or the same CO2-treatement (EC):
Results
Phytoplankton biovolume
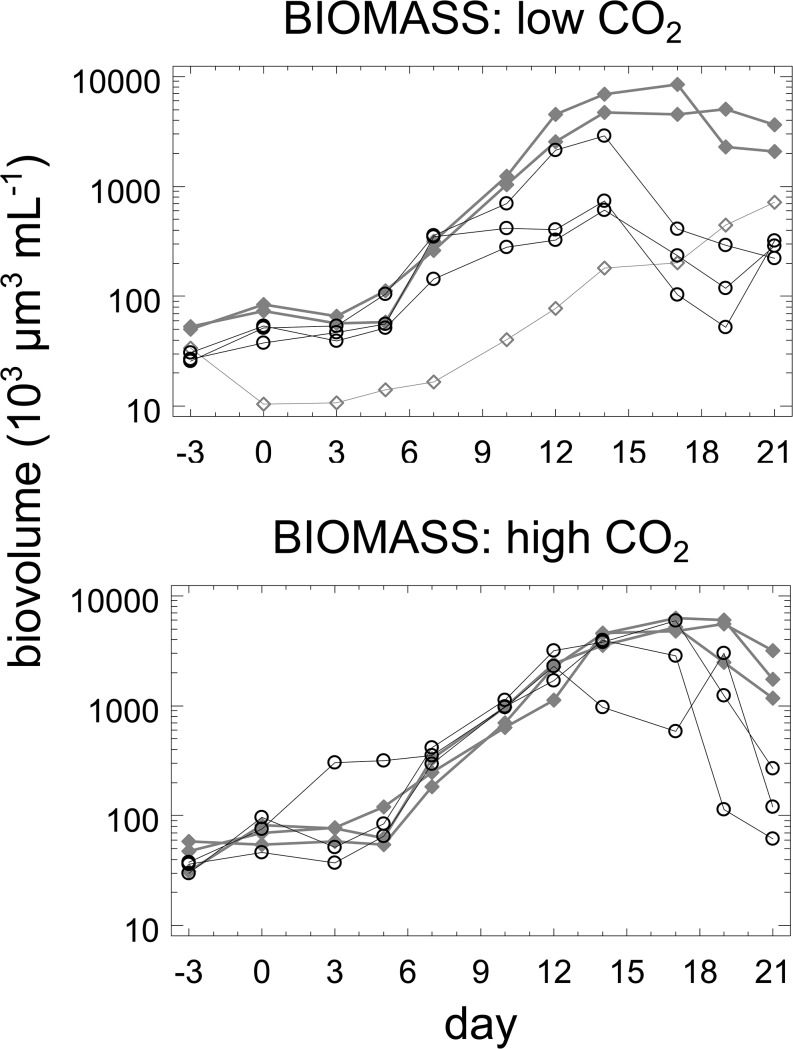
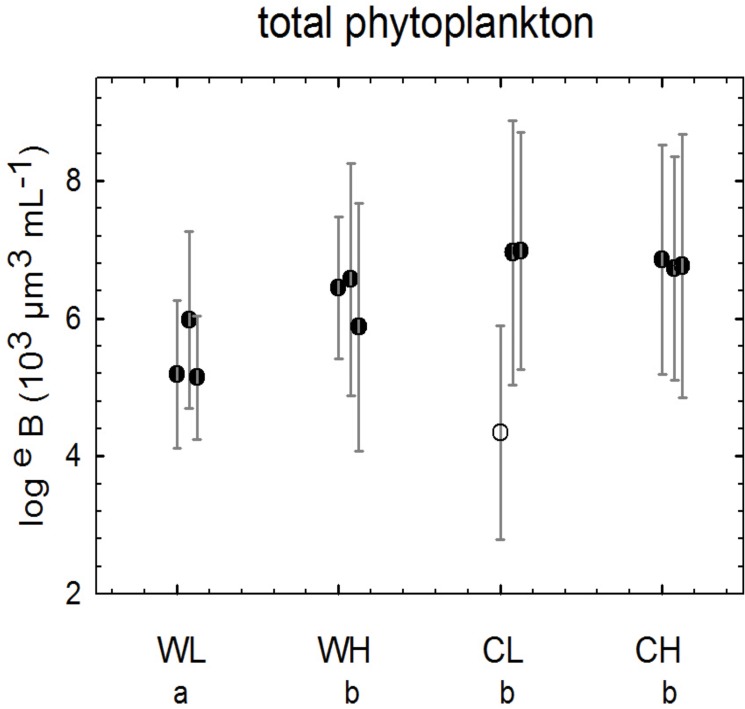
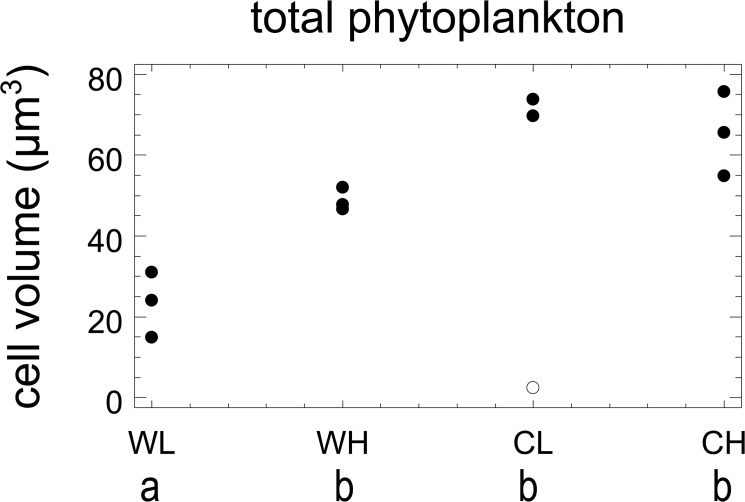
In most mesocosms, appreciable growth of biomass began in the interval from day 3 on, except for mesocosm 9, one of the CL mesocosms (Fig 1). In this mesocosm, there was a 72 hrs failure of the light supply at the beginning of the experiment which initiated a completely aberrant trajectory of plankton dynamics. This mesocosm was excluded from further analysis. At low CO2, warm and cold mesocosms began to diverge during the period from day 10 to 14when growth was maintained in the cold mesocosms but began to slow down in the warm ones. From day 14 on, biomass began to decline in the warm mesocosm to levels almost one order lower than maximal ones for the warm treatments. In the cold mesocosms higher maximal levels were reached, decline started on day 16 and was less pronounced. In the high CO2 mesocosm divergence between cold and warm ones was observed towards the end of the experiment (days 19 to 21) and the differences between peak values of warm and cold mesocosms were less pronounced than in the low CO2 treatments. A two-factor ANOVA based on mean values over the entire experiment (Fig 2) showed a significant effect of temperature (p = 0.0014), no significant effect of CO2, and a significant interaction effect (p = 0.0027). A multiple range test showed two homogenous groups, one consisting of CL, CH, and WH treatments and one of the WL treatments with significantly lower biovolumes.
Fig 1. Time course of phytoplankton biovolumes.
Open circles: warm mesocosms; grey and filled diamonds: cold mesocosms; open diamonds: mesocosms 9.
Fig 2. Means of total phytoplankton biomass.
Means over entire experiment calculated after loge-transformation and standard deviations (vertical lines). Mesocosm is 9 shown by an open symbol. Treatment codes: WL: warm, low CO2, WH: warm, high CO2, CL: cold, low CO2, CH: cold, high CO2; codes for homogenous groups according to Tukey’s HSD: a, b.
Species composition
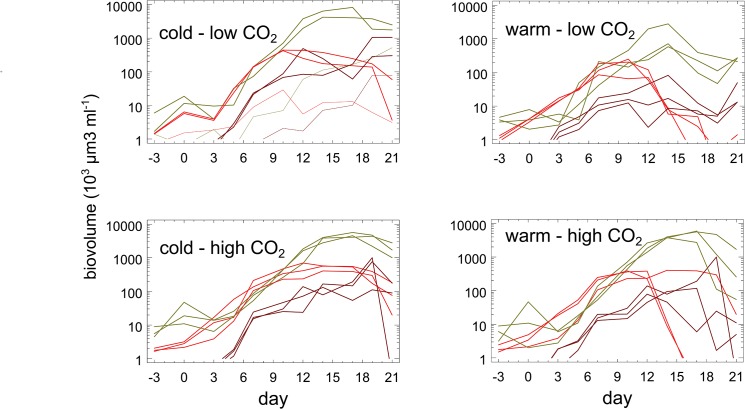
The succession patterns of higher phytoplankton taxa did not differ strongly between the different treatments (shown for diatoms, cryptophytes, and prymnesiophytes in Fig 3). Initially, large dinoflagellates (Ceratium spp.) together with large diatoms (Ditylum brightwelli Grunow, Proboscia alata Sundström, Rhizosolenia setigera Brightwell) dominated biomass. The large dinoflagellates stagnated until day 3 to 5 and then began to decline slowly, possibly because of the mixing of the mesocosms. The large diatoms grew slowly, but were quickly surpassed by the small celled Skeletonema marinoi Sarno & Zingone which together with extremely large Coscinodiscus wailesii Gran & Angst made up for most of the diatoms growth observed in the mesocosms. Until ca. day 9, cryptophyte growth (mainly Teleaulax acuta D.R.A. Hill) kept pace with the diatoms and in some cases even surpassed them, but thereafter cryptophytes either stagnated or declined. They were replaced by abundant prymnesiophytes (2 spp. of Chrysochromulina), but prymnesiophyte biomass never surpassed diatom biomass. At the level of higher taxa, the biomass trajectories in the aberrant mesocosm 9 looked almost like a delayed and miniaturized copy of the other mesocosms.
Fig 3. Time course of the biovolumes of dominant higher taxa.
Diatoms (olive), cryptophytes (red), and prymnesiophytes (brown), the trajectories from the aberrant mesocosm 9 are shown by dotted lines.
The relative biomass of the two dominant higher taxa, diatoms (grand mean 83% of total biomass) and cryptophytes (grand mean 10.5%) did not respond to warming and acidification. The relative biomass of prymnesiophytes (grand mean 4.7%) responded negatively to warming, while the relative biomass of dinophytes (1.5%), pico-chlorophytes (0.6%) and pico-cyanobacteria (0.14%) responded positively to warming. Dinophytes responded also negatively to acidification and showed a significant interaction effect between temperature and CO2. The other higher taxa did not show any significant response to CO2 (Table 1).
Table 1. Relative biomass of higher taxa and species.
| Grand mean pi | sign-temp | sign-CO2 | F-temp | F-CO2 | F-int | |
|---|---|---|---|---|---|---|
| BACILLARIOPHYCEAE | 0.826 | 0.68 ns | 0.97 ns | 3.22 ns | ||
| Skeletonema marinoi Sarno & Zingone | 0.627 | 0.87 ns | 1.32 ns | 0.52 ns | ||
| Coscinodiscus wailesii Gran & Angst | 0.061 | 2.15 ns | 0.97 ns | 0.65 ns | ||
| Rhizosolenia setigera Brightwell | 0.051 | 1.99 | 0.10 | 0.17 | ||
| Cerataulina pelagica Hendey | 0.019 | - | 11.8 * | 5.47 ns | 0.00 ns | |
| Cylindrotheca closterium Reimann & J. Lewis | 0.016 | + | 8.73 * | 3.52 ns | 2.74 ns | |
| Proboscia alata Sundström | 0.015 | - | - | 9.53 * | 16.6 ** | 1.32 ns |
| Thalassiosira nordenskioeldii Cleve | 0.013 | + | 7.15 * | 0.00 ns | 1.25 ns | |
| Thalassionema nitzschioides Mereschkowski | 0.012 | - | 0.07 ns | 20.4 ** | 2.53 ns | |
| Dactyliosolen fragilissimus Hasle | 0.004 | - | 19.9 ** | 0.03 ns | 0.75 ns | |
| Ditylum brightwellii Grunow | 0.003 | + | 7.21 * | 2.17 ns | 0.50 ns | |
| Guinarida flaccida H. Peragallo | 0.002 | - | - | 6.93 * | 11.5 * | 0.00 ns |
| Chaetoceros spp. | 0.0006 | - | 0.41 ns | 10.1 * | 0.01 ns | |
| Pseudontzschia sp. | 0.0002 | - | 1.21 ns | 6.84 * | 0.67 ns | |
| DINOPHYTA | 0.015 | + | - | 8.10 * | 7.40 * | 6.65 * |
| Ceratium fusus Dujardin | 0.010 | - | 4.77 ns | 8.14 ** | 6.34 ** | |
| Ceratium tripos Nitzsch | 0.004 | + | 18.3 ** | 3.94 ns | 6.93 * | |
| Ceratium lineatum Cleve | 0.0002 | - | 0.93 ns | 8.74 * | 1.34 ns | |
| Prorocentrum micans Ehrenberg | 0.0001 | 5.15 ns | 3.69 ns | 0.71 ns | ||
| CRYPTOPHYTA | 0.105 | 0.20 ns | 0.04 ns | 5.11 ns | ||
| Teleaulax acuta D.R.A.Hill | 0.096 | 0.05 ns | 0.14 ns | 5.20 ns | ||
| R5 = Plagioselmis prolonga Butcher | 0.010 | 4.96 ns | 1.52 ns | 3.40 ns | ||
| PRYMNESIOPHYTA | ||||||
| R6 = Chrysochromulina spp. 1 | 0.047 | - | 11.5 * | 2.48 ns | 0.41 ns | |
| CHLOROPHYTA | 0.006 | + | 11.9 * | 1.49 ns | 5.70 * | |
| R1 = Ostreococcus sp. | 0.004 | + | 8.65 * | 1.16 ns | 4.58 ns | |
| R2 = Bathycoccus sp. | 0.0017 | + | 40.0 *** | 3.74 ns | 11.97 * | |
| CYANOBACTERIA | 0.0014 | + | 6.55 * | 2.26 ns | 2.81 ns | |
| R4 = Synechococcus—type | 0.0008 | 5.35 ns | 4.11 ns | 4.26 ns | ||
| R7 = Synechocystis—type | 0.0004 | 3.49 ns | 0.00 ns | 0.13 ns | ||
| R3 = Synechococcus—type | 0.0002 | + | 10.74 * | 4.34 ns | 6.10 * | |
| UNKNOWN | ||||||
| R8 | 0.0007 | - | 9.65 * | 1.52 ns | 0.05 ns |
2-factor ANOVA; independent variables temperature and CO2, dependent variable asin√pi; species sorted within higher taxa according to the grand mean (without mesocosm 9) of biomass; R1, … R8 denote taxa whose abundance and biomass data were obtained by flow cytometry. Besides F-values also the sign of significant temperature and CO2 effects are shown; d.f. in all cases 1,7; Fp-int: F-ratio for interaction term; significance levels
ns:p>0.05
*: p<0.05
**: p<0.01
***: p<0.001.
1Most probably, in the flow cytometric analysis there were two, albeit overlapping clusters of Chrysochromulina, both were uncultured according to the genetic analysis and had overlapping sizes according to microscopy analysis.
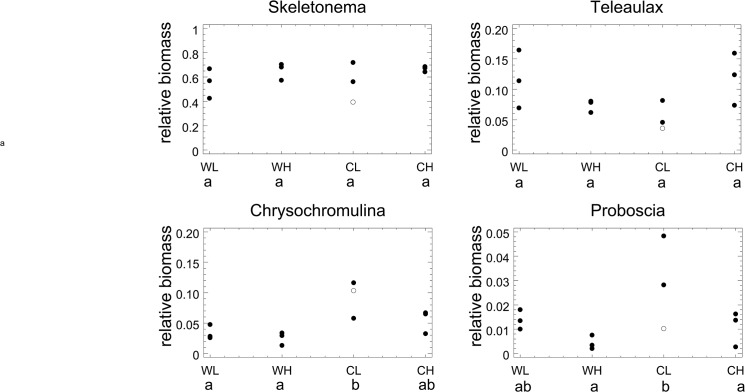
The relative biomass (Table 1, Fig 4) of 8 species did neither respond to temperature nor to CO2. Among them were the 4 most important taxa in terms of biomass, the small-celled diatom Skeletonema marinoi, the cryptophyte Teleaulax acuta, and the large celled-diatoms Coscinodiscus wailesii and Rhizosolenia setigera. Together, they made up for 83.5% of total biomass (grand mean). Six species responded negatively to temperature, among them Chrysochromulina spp., the 5th taxon in the ranking of biomass. 10 species responded positively to temperature. A significant negative response to CO2 was found in 7 species, while no significant positive response was found. Together, the species responsive to CO2 formed on average 4% of biomass.
Fig 4. Relative biomass of selected species.
Relative biomass (mean over entire duration of experiment) of the two most abundant species (Skeletonema marinoi, Teleaulax acuta) and of the most abundant species responsive to temperature (Chrysochromulina sp.) and the most abundant species responsive to CO2-enrichment (Proboscia alata). Treatment codes: WL: warm, low CO2, WH: warm, high CO2, CL: cold, low CO2, CH: cold, high CO2; codes for homogenous groups according to Tukey’s HSD: a, b.
Cell size
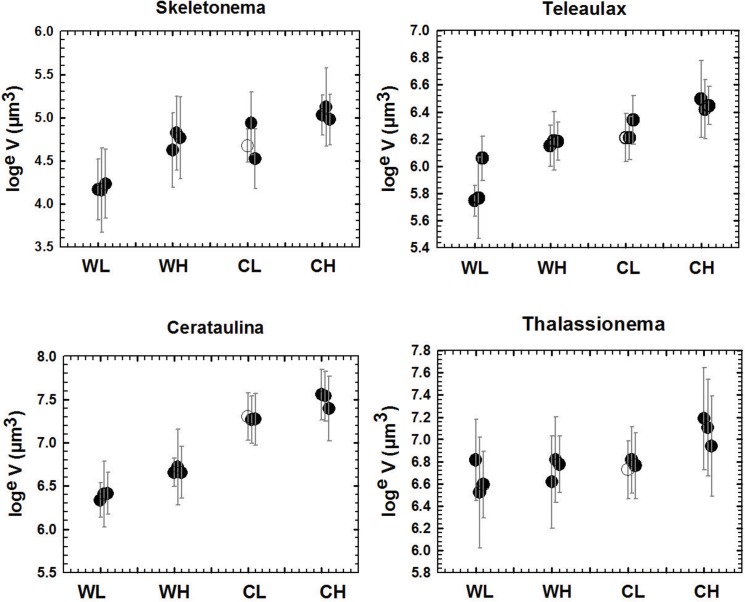
The mean cell size of total phytoplankton decreased significantly with warming and increased with CO2-enrichment (Fig 5, Table 2). Also the interaction term was significant. Fourteen phytoplankton species and 3 heterotrophic flagellates (the pico-sized Bolidomonas- like, and the nano-sized Telonema subtilis Griessmann and Cryothecomonas cf. longipes Schnepf & Kühn) were abundant enough for reliable size measurements (Fig 6, Table 2). Eleven phytoplankton species showed a significant, negative response to warming. There was no case of a significant positive response. Responses to CO2 were significantly positive for 8 phytoplankton taxa, significantly negative for the small phytoflagellate Plagioselmis prolonga Butcher and non-significant for 5 taxa. The two larger heterotrophic flagellate taxa showed a significant negative response to temperature and none responded to CO2 while the smallest heterotrophic flagellate showed no significant response to the experimental treatments.
Fig 5. Community mean cell size.
Response of community mean cell size (calculated from mean total biovolumes divided by mean cell numbers for each mesocosm) to the experimental treatments, mesocosm 9 shown by an open symbol. Treatment codes: WL: warm, low CO2, WH: warm, high CO2, CL: cold, low CO2, CH: cold, high CO2; codes for homogenous groups according to Tukey’s HSD: a, b.
Table 2. Cell size.
| Vmeanμm3 | ES-temp | ES-CO2 | F-temp | F-temp | F-int | |
|---|---|---|---|---|---|---|
| Total phytoplankton | ||||||
| logBVmean | -0.725 | 0.339 | 29.2 *** | 6.38 * | 10.8 * | |
| indidual taxa, algae (V; log μm3 cell-1) | ||||||
| Skeletonema marinoi | 121 | -0.392 | 0.444 | 20.8 ** | 26.8 ** | 2.32 ns |
| Cerataulina pelagica | 1191 | -0.851 | 0.276 | 442.4 *** | 46.9 *** | 0.42 ns |
| Thalassiosira nordenskioeldii. | 1402 | -0.451 | 0.382 | 45.9 *** | 32.7 *** | 0.47 ns |
| Dactyliosolen fragilissimus | 4865 | -0.715 | 0.409 | 237.2 *** | 77.5 *** | 0.24 ns |
| Rhizosolenia setigera | 160175 | -0.403 | 0.398 | 17.2 ** | 13.0 ** | 0.91 ns |
| Guinardia flaccida | 5225 | -0.140 | 0.188 | 7.86 * | 13.0 ** | 0.34 ns |
| Thalassionema nitzschioides | 991 | -0.294 | 0.238 | 25.1 ** | 19.6 ** | 3.26 ns |
| Teleaulax acuta | 507 | -0.333 | 0.256 | 31.4 *** | 16.0 ** | 0.94 ns |
| R6 = Chrysochromulina spp. 1 | 4.51 | -0.751 | -0.17 | 54.5 *** | 3.09 ns | 1.07 ns |
| R5 = Plagioselmis prolonga | 20.63 | -0.049 | -0.323 | 0.31 ns | 7.38 * | 2.49 ns |
| R1 = Ostreococcus sp. | 0.373 | -0.020 | 0.003 | 6.79 * | 0.15 ns | 0.05 ns |
| R2 = Bathycoccus sp. | 0.456 | -0.004 | -0.001 | 0.01 ns | 0.00 ns | 0.21 ns |
| R3 = Synechococcus-type | 0.357 | -0.016 | -0.004 | 2.73 ns | 0.18 ns | 0.00 ns |
| R4 = Synechococcus-type | 0.356 | -0.045 | -0.019 | 27.7 ** | 5.16 ns | 1.15 ns |
| indidual taxa, heterotrophic flagellates (V; log μm3 cell-1) | ||||||
| Bolidomonas—like | 2.074 | -0.021 | 0.031 | 0.84 ns | 1.96 ns | 0.58 ns |
| Telonema subtilis | 60.65 | -0.624 | 0.0013 | 200.6 *** | 0.10 ns | 0.72 ns |
| Cryothecomonas cf. longipes | 182.4 | -0.729 | -0.027 | 57.3 *** | 0.08 ns | 0.012 ns |
2-factor ANOVA; independent variables temperature and CO2, dependent variable log cell volume (μm3); species sorted within higher taxa according to the grand mean (without mesocosm 9) of biomass; besides F-values also effect sizes (ES) are shown; d.f. in all cases 1,7; significance levels
ns: p>0.05
*: p<0.05
**: p<0.01
***: p<0.001.
1Because of the appearance of a larger, but overlapping Chrysochromulina-cluster towards the end of the experiment Chrysochromolina-sizes were taken already from day 12.
Fig 6. Cells size of selected species.
Size response of 4 selected phytoplankton species to the experimental treatments, means and standard deviations (vertical lines) based on measurements of 20 cells per mesocosms. Mesocosm 9 shown by an open symbol. Treatment codes: WL: warm, low CO2, WH: warm, high CO2, CL: cold, low CO2, CH: cold, high CO2.
An analysis of the effect sizes of temperature and CO2 on the cell volumes of phytoplankton showed that ET and EC on size are themselves size dependent (expressed by the grand mean of cell volume, VGM):
This means, that both the negative size effect of temperature and the positive size effect of CO2 are more pronounced for larger species. The larger heterotrophic flagellates also showed a negative size effect of temperature, but no size effect of CO2. The ET-cell size relationship for heterotrophic flagellates was marginally insignificant because of the low number of taxa included:
Discussion
Phytoplankton biomass
The phytoplankton succession during the experiment took the typical course of transient autumn blooms in Kiel Bight, when after extended vertical mixing in Kiel Bight surface concentrations of nitrate, phosphate and silicate are saturating for algal growth and calm and sunny conditions permit a growth pulse of phytoplankton. Hypothesis 1 (lower biomass under warmer conditions) was confirmed while hypothesis 2 (higher biomass at higher CO2) was only partially confirmed, in our case for the warm mesocosms. The temperature effect agrees with a suite of spring experiments performed in the same mesocosm system [22,27,33] and a similar experiment in coastal waters of North Carolina [34,35]. However, one summer experiment using the Kiel mesocosm produced the opposite result [36]. The usual explanation for the temperature effect is a stronger increase of top-down factors (zooplankton grazing) with warming than of algal growth, because of the temperature-insensitivity of light limited photosynthesis [27,34,35]. The CO2-effect is consistent with the assumption that CO2 might be occasionally limiting for phytoplankton growth, while it is inconsistent with the assumption that CO2 is a stressor for the local phytoplankton community.
Taxonomic shifts
Hypotheses 3 and 4 predicting taxonomic shifts in response to warming and CO2 addition were supported only to a small extent. Neither warming nor CO2 addition changed the relative biomass of the two major higher taxa (diatoms, cryptophytes) and of the 4 most important species. Shifts were only found for subdominant or rare species. The weakness of temperature effects was a surprise for us, because a 6°C temperature difference had strong effects on species composition in the spring bloom experiments in the same experimental systems in 5 successive years [22]. However, a shift from 9 to 15°C might have a smaller selection effect between species than a shift from 2 to 8°C. Interestingly, two summer experiments in the same facility [36,37] also revealed weak temperature effects on taxonomic composition. It is also possible that the different responses of subdominant/rare species could have been the precursor of dominance shifts if phytoplankton succession could have proceeded further.
The weakness of a CO2 effect on the dominant species agrees with an experiment at the Azores where differences in the relative biomass of dominant between the acidified and non-acidified treatments was usually <10% of total biomass. [17]. Contrary to a previous study [20] found a slight negative effect of CO2 on Chrysochromulina sp., this effect was not found for mean values in our experiment, but the decline of Chrsochromulina spp. during the final sampling intervals in the low CO2 treatments might hint in the same direction. Overall weak effects indicate similar adaptations to CO2 among the species coming from an environment with high natural CO2-fluctuations [21]. We cannot exclude that a stronger CO2 manipulation could have led to stronger effects, but it would have been beyond the atmospheric CO2-enrichment predicted for the end of the 21st century even in the pessimistic IPCC-scenarios.
Cell size
Hypothesis 5 predicted reduced cell sizes of phytoplankton and heterotrophic nanoflagellates under warmer condition while hypothesis 6 predicted an increase of cell sizes under CO2-enrichment only for phytoplankton. The general trend shrinkage of cell size with warming is in agreement with the majority of recent literature, which have analyzed cell size shifts within species [23], at the community level [38,39] or at both levels simultaneously [24,25]. However, the relative importance of shifts between and shifts within species differs from the previous papers, where shifts between species were by far the dominant factor. This can be shown if actual cell sizes in the individual mesocosms are replaced by the grand mean of each species, calculated community mean cell size still declines, but the effect size shrinks from -0.728 to -0.34, i.e. to less than the half. This is due to exceptional insensitivity of taxonomic composition to temperature in our experiment, while most of the previous studies have reported strong taxonomic effects of warming [22]. The CO2-effect on cell size bears some resemblance to the nitrogen effect in a recent study [25] where cell sizes strongly declined with the intensity of N-limitation. This is in agreement with the assumption of CO2-limitation and the well known effect that smaller cells suffer a smaller disadvantage, if diffusion of a limiting resource is the rate limiting effect (for CO2: [11]). However, we cannot exclude the alternative explanation that a better supply of CO2 led to an increased synthesis of non-protein components of biomass and thereby lead to an inflation of cell sizes. An increase share of non-protein biomass components would agree with previous findings of increased C: N consumption ratios under elevated CO2 [12]. The explanation by CO2-limitation would be supported if the positive size effect of CO2 would be absent under depletion by mineral nutrients precluding CO2-limitation. This was indeed the case in a small-scale (9 L) bottle experiment in the Okhostk Sea [40] where CO2-addition lead to a shift towards pico-cyanobacteria and picoplankton eukaryotes.
Implications for the ocean acidification problem
We admit, that the global applicability of our findings is limited by the fact, that the Western Baltic Sea is an environment of high CO2 variability [21] and, therefore, phytoplankton already now have been selected for wide CO2 tolerance limits. However, wide fluctuations of CO2 are normal for productive ocean regions where the actual pCO2 in a water body is more strongly influenced by the balance between photosynthesis and respiration than by changes in the atmospheric pCO2 [19]. Because of the outstanding importance of the productive zones of the oceans for global primary production, we suggest that at least outside the zones of coccolithophore dominance phytoplankton is not the most sensitive component of the global ocean’s pelagic ecosystems. On the contrary, CO2 enrichment might have a mildly eutrophicating function at restricted regional and seasonal scales, such as in our experiment. However, we expect that the increased strength of thermal stratification, the spatial extension of the subtropical gyres and the resulting decreased supply of nutrients to the surface ocean in a warming world [41,42,43] will exceed any eutrophication effect of CO2, thus making climate warming the much stronger impact on the global pelagic system than ocean acidification.
In order to evaluate potential changes of the pelagic ecosystem in response to warming and ocean acidification it is also necessary to ask how phytoplankton effects will be transmitted to higher trophic levels, in particular to mesozooplankton which forms the direct link between primary production and fish production. A recent laboratory study with one phytoplankton species (the diatom Thalsassiosira pseudonana) and one mesozooplankton species (the copepod Acartia tonnsa) gave reason for concern, because it indicated an amplification of effects [44]. Neither of the two species suffered from a fitness loss if cultivated at elevated CO2, but Acartia showed a lowered egg production rate when fed with Thalssiosira cultured at elevated CO2 because of a reduced content of polyunsaturated fatty acids (PUFAs) in the food. In a follow-up study with a natural protists of phytoplankton and heterotrophic /protozoan assemblage as food base this amplification of effects could not be repeated [45]. Similarly, in this mesocosms experiment there was no indication of an adverse effect of CO2 on copepod feeding conditions [46]. Basically, the abundance of copepodids and adults at the end of the experiment showed the same pattern as phytoplankton biovolume: highest in the cold—high CO2 treatments (30–35 ind l-1), intermediate in the cold—low CO2 and warm—high CO2 (20–25 ind l-1) and lowest in the warm—low CO2 treatments (15–20 ind L-1). Also an analysis of fatty acids showed no indication of a nutritional inadequacy of phytoplankton under high CO2 [46].Thus, it seems that increasing complexity of the experimental community dampens adverse effects, as anticipated in the insurance hypothesis about biodiversity—ecosystem effects [47]. However, it will take more experiments with different plankton communities to decide whether food web interactions act as “amplifiers” or “shock absorbers” of external forcing.
Acknowledgments
This project was funded by the German Federal Ministry of Education and Research (program “BIOACID”). Technical assistance by T. Hansen, C. Meyer and L. Berghoff is gratefully acknowledged.
Data Availability
Related data have been deposited in the archive Pangaea and are available at http://doi.pangaea.de/10.1594/PANGAEA.840845.
Funding Statement
Bundesministerium für Bildung und Forschung (Federal Ministry of Eductation and Pesearch, www.bmbf.de) through the compound project BIOACID (www.bioacid.de), grant number 03F0655A.
References
- 1. Intergovernmental Panel on Climate Change (2013) Climate Change 2013: The Physical Science Basis Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, et al. , editors Cambridge, United Kingdom and New York, NY, USA: Cambridge University Press; Available: http://www.climatechange2013.org/images/uploads/WGI_AR5_SPM_brochure.pdf. [Google Scholar]
- 2. Feely RA, Sabine CL, Lee K, Berelson W, Kleypas J, Fabry VJ, et al. (2004) Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science 305: 362–366. [DOI] [PubMed] [Google Scholar]
- 3. Hoegh-Guldberg O, Bruno JF (2010) The impact of climate change on the world`s marine ecosystems. Science 328: 1523–1528. 10.1126/science.1189930 [DOI] [PubMed] [Google Scholar]
- 4. Kroeker KJ, Kordas RL, Crim RN, Hendriks IE, Ramajo L, Singh GS, et al. (2013) Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Global Change Biol 19: 1884–1896. 10.1111/gcb.12179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Berry L, Taylor AR, Lucken U, Ryan KP, Brownlee C (2002) Calcification and inorganic carbon acquisition in coccolithophores. Funct Plant Biol 29: 289–299. [DOI] [PubMed] [Google Scholar]
- 6. Riebesell U, Zondervan I, Rost B, Tortell BT, Zeebe RE, Morel FMM (2000) Reduced calcification in marine plankton in response to increased atmospheric CO2 . Nature 407: 634–637. [DOI] [PubMed] [Google Scholar]
- 7. Engel A, Zondervan I, Aerts K, Beaufort L, Benthien A, Chou L, et al. (2005) Testing the direct effect of CO2 concentration on a bloom of the coccolithophorid Emiliania huxleyi in a mesocsm experiment. Limnol Oceanogr 50:493–507. [Google Scholar]
- 8. Falkowski PG, Raven JA (2007) Aquatic photosynthesis Princenton Univ Press, Princeton NJ: [Google Scholar]
- 9. Badger MR, Andrews TJ, Whitney SM, Ludwig M, Yellowlees DC, Leggat W, et al. (1998) The diversity and coevolution of RubisCO, plastids, pyrenoids and chloroplast-based CO2-concentrating mechanisms. Can J Bot 76: 1052–1071. [Google Scholar]
- 10. Thoms S, Pahlow M, Wolf-Gladrow DA (2001) Model of the carbon concentrating mechanisms in chloroplast of eukaryotic algae. J Theor Biol 208: 295–313. [DOI] [PubMed] [Google Scholar]
- 11. Reinfelder JR (2011) Carbon concnetrating mechnaisms in eukaryotic marine phytoplankton. Ann Rev Mar Sci 3: 291–325. [DOI] [PubMed] [Google Scholar]
- 12. Riebesell U, Schulz KG, Bellerby RGJ, Botros M, Fritsche P, Meyerhöfer M, et al. (2007) Enhanced biological carbon consumption in a high CO2 ocean. Nature 450: 545–548. [DOI] [PubMed] [Google Scholar]
- 13. Burkhardt S, Amoroso G, Riebesell U, Sültemeyer D (2001) CO2 and HCO3- uptake in marine diatoms acclimated to different CO2 concentrations. Limnol Oceanog 46: 1378–1391. [Google Scholar]
- 14. Rost B, Riebesell U, Burkhardt S, Sültemeyer D (2003) Carbon acquisition of bloom- forming marine phytoplankton. Limnol Oceanogr 48: 55–67. [Google Scholar]
- 15. Rost B, Zondervan D, Wolf-Gladrow D (2008) Sensitivity of phytoplankton to future changes in ocean carbonate chemistry: current knowledge, contradictions and research directions. Mar Ecol Progr Ser 373: 227–237. [Google Scholar]
- 16. Tortell PD, Payne CD, Li Y, Trimborn S, Rost B, Smith WO, et al. (2008) CO2 sensitivity of Southern Ocean phytoplankton. Geophys Res Lett 35: L04605, 10.1029/2007GL032583 [DOI] [Google Scholar]
- 17. Eggers SL, Lewandowska AM, Barcelos Ramos J, Blanco-Ameijeiras S, Gallo F, Matthiessen B (2014) Community composition has greater impact on the functioning of marine phytoplankton communities than ocean acidification. Glob Change Biol 20: 713–723. 10.1111/gcb.12421 [DOI] [PubMed] [Google Scholar]
- 18. Kim JM, Lee K, Shin K, Kong JH, Lee HW, Kim M, et al. (2006) The effect of seawater CO2 concentrations on growth of a natural phytoplankton assemblage in a controlled mesocosm experiment. Limnol Oceanogr 51: 1629–1636 [Google Scholar]
- 19. Joint I, Doney SC, Karl DM (2011) Will ocean acidification affect marine microbes? ISME J 5: 1–7. 10.1038/ismej.2010.79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Lindh MV, Riemann L, Baltar F, Romero-Oliva C, Salomon PS, Graneli E, et al. (2013) Consequences of increased temperature and acidification on bacterioplankton community composition during a mesocosms spring bloom in the Baltic Sea. Env Microbiol Rep 5: 252–262. [DOI] [PubMed] [Google Scholar]
- 21. Thomsen J, Gutowska MA, Saphorster J, Heinemann A, Trübenbach K, Fietzke J, et al. (2010) Calcifying invertebrates succeed in a naturally CO2-rich coastal habitat but are threatened by high levels of future acidification. Biogeosciences 7: 3879–3891. [Google Scholar]
- 22. Sommer U, Adrian R, Bauer B, Winder M (2012) The response of temperate aquatic ecosystems to global warming: novel insights from a multidisciplinary project. Mar Biol 159: 2367–2377. [Google Scholar]
- 23. Atkinson D, Ciotti B, Montagnes DJS (2003) Protists decrease in size linearly with temperature: ca. 2.5% C−1. Proc Royal Soc London Ser B: Biol Sci 270: 2605–2611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Peter KH, Sommer U (2012) Phytoplankton cell size, inter- and intraspecific effects of warming and grazing. PLoS One 7: e49632 10.1371/journal.pone.0049632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Peter KH, Sommer U (2013) Phytoplankton cell size reduction in response to warming mediated by nutrient limitation. PLoS One 8: e71528 10.1371/journal.pone.0071528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Sommer U, Aberle N, Engel A, Hansen T, Lengfellner K, Sandow M, et al. (2007) An indoor mesocosm system to study the effect of climate change on the late winter and spring succession of Baltic Sea phyto- and zooplankton. Oecologia 150: 655–667. [DOI] [PubMed] [Google Scholar]
- 27. Sommer U, Lewandowska A (2011) Climate change and the phytoplankton spring bloom: warming and overwintering zooplankton have similar effects on phytoplankton. Glob Change Biol 17: 154–162. [Google Scholar]
- 28. Brock TD (1981) Calculating solar radiation for ecological models. Ecol Model 14: 1–19. [Google Scholar]
- 29. Utermöhl H (1958) Zur Vervollkommnung der quantitativen Phytoplankton Methodik. Mitt Internat Verein Theor Angew Limnol 9: 263–272. [Google Scholar]
- 30. Porter KG, Feig YS (1980) The use of DAPI for identifying and counting aquatic microflora. Limn Oceanogr 25: 943–948. [Google Scholar]
- 31. Christaki U, Kormas K, Genitsaris S, Georges C, Sime-Ngando T, Viscogliosi E, et al. (2014) Winter-Summer succession of unicellular eukaryotes in a meso-eutrophic coastal system. Microb Ecol 67: 13–23. 10.1007/s00248-013-0290-4 [DOI] [PubMed] [Google Scholar]
- 32. Hillebrand H, Duerselen CD, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J of Phycol 35: 403–424. [Google Scholar]
- 33. Sommer U, Lengfellner K (2008) Climate change and the timing, magnitude and composition of the phytoplankton spring bloom. Glob Change Biol 14:1199–1208 [Google Scholar]
- 34. O'Connor MI (2009) Warming strengthens an herbivore–plant interaction. Ecology 90: 388–398. [DOI] [PubMed] [Google Scholar]
- 35. O'Connor MI, Piehler MF, Leech DM, Anton A, Bruno JF (2009) Warming and resource availability shift food web structure and metabolism. PLoS Biol 7: e1000178 10.1371/journal.pbio.1000178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Taucher J, Schulz KG, Dittmar T, Sommer U, Oschlies A, Riebesell U (2012) Enhanced carbon overconsumption in response to increasing temperatures during a mesocosm experiment. Biogeosciences 9: 3531–3545. [Google Scholar]
- 37. Lewandowska AM, Boyce DG, Hofmann M, Matthiessen B, Sommer U, Worm B (2014) Effects of sea surface warming on marine plankton. Ecol Lett 17: 614–623 10.1111/ele.12265 [DOI] [PubMed] [Google Scholar]
- 38. Daufresne M, Lengfellner K, Sommer U (2009) Global warming benefits the small in aquatic ecosystems. Proc Nat Acad Sci 106: 12788–12793 10.1073/pnas.0902080106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Yvon‐Durocher G, Montoya JM, Trimmer M, Woodward G (2011) Warming alters the size spectrum and shifts the distribution of biomass in freshwater ecosystems. Glob Change Biol 17: 1681–1694. [Google Scholar]
- 40. Yoshimura T, Nishioka J, Suzuki K, Hattori H, Kiyosawa H, Watanabe YW, et al. (2010) Impact of elevated CO2 on organic carbon dynamics in the nutrient depleted Okhotsk Sea surface waters. L Exp Mar Biol Ecol 395: 191–198. [Google Scholar]
- 41. Behrenfeld MJ, O’Malley RT, Siegel DA, McClain CR, Sarmiento JL, Feldman JC, et al. (2006) Climate-driven trends in contemporary ocean productivity. Nature 444: 752–755. [DOI] [PubMed] [Google Scholar]
- 42. Boyce DG, Lewis MR, Worm B (2010) Global phytoplankton decline over the past century. Nature 466: 591–596. 10.1038/nature09268 [DOI] [PubMed] [Google Scholar]
- 43. Hofmann M, Worm B, Rahmstorf S, Schellnhuber HJ (2011) Declining ocean chlorophyll under unabated anthropogenic CO2 emmissions. Environm Res Lett 6: 1–8. [Google Scholar]
- 44. Rossoll D., Bermudez R, Hauss H, Schulz KG, Riebesell U, Sommer U, et al. (2012) Ocean acidification-induced food quality deterioration constrains trophic transfer. PLoS One 7: e34737 10.1371/journal.pone.0034737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Rossoll D, Sommer U, Winder M (2013) Community interactions dampen acidification effects in a coastal plankton system. Mar Ecol Progr Se. 486: 37–46. [Google Scholar]
- 46.Garzke, J. (2014) Global change effects on zooplankton body size: a range of experimental approaches. PhD-thesis, University of Kiel, Germany
- 47. Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: The insurance hypothesis. Proc Nat Acad Sci USA 96: 1463–1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Related data have been deposited in the archive Pangaea and are available at http://doi.pangaea.de/10.1594/PANGAEA.840845.