Abstract
Purpose
To evaluate the efficacy of photodynamic antimicrobial chemotherapy (PACT) with the new porphyrin derivative TONS 504 and a light-emitting diode (LED) against acyclovir (ACV)-sensitive and -resistant herpes simplex virus type 1 (HSV-1).
Methods
Human FL cells infected with the viral strains were subjected to PACT with TONS 504 at various concentrations (0.01 to 10 mg/l) and irradiation at various light energies (10 to 30 J/cm2) and were then incubated for 24 h before analysis.
Results
Immunocytofluorescence analysis with antibodies to HSV-1 revealed that PACT eliminated HSV-1 and ACV-resistant HSV-1 in a manner dependent on the TONS 504 concentration and light energy. Complete eradication of both viruses was apparent at a TONS 504 concentration of 10 mg/l and light energy of 10 to 30 J/cm2 as well as at a TONS 504 concentration of 1 mg/l and light energy of 20 or 30 J/cm2. No antiviral effect was apparent with TONS 504 in the absence of irradiation or with irradiation in the absence of TONS 504. Staining of cell nuclei with 4′, 6-diamidino-2-phenylindole revealed no apparent cytotoxicity of the PACT system, a finding that was confirmed by the system’s failure to induce the release of lactate dehydrogenase from the host cells.
Conclusions
We conclude that our PACT system based on TONS 504 and an LED is effective for eliminating HSV-1 and ACV-resistant HSV-1 without a harmful effect on host cells.
Introduction
Herpes simplex virus (HSV), a member of the Herpesviridae family, is an enveloped virus with a double-stranded DNA genome. HSV infection is prevalent in humans worldwide and is characterized by an acute phase in which the virus replicates rapidly at the site of infection followed by movement of the virus to regional ganglia and the establishment of a latent infection. Humans are thought to be the sole reservoir for transmission of the virus to other humans [1]. The attachment of HSV to host cells and its subsequent penetration of the cells are supported by viral surface glycoproteins, and the virus then mobilizes cellular proteins to promote the efficient synthesis of viral DNA and protein. HSV also blocks the host response to infection as well as programmed cell death of the infected host cells, thus ensuring the virus’s survival [2]. Most humans show serological evidence of HSV infection by middle age, with one-third of the global population thought to experience recurrent HSV infection. HSV-1 is responsible for herpes labialis, whereas HSV-2 gives rise to herpes genitalis. Although HSV-1 and HSV-2 are usually transmitted by different routes and affect different parts of the body, the epidemiology and clinical manifestations of infection with these two viruses overlap. Primary HSV-1 infection generally results in mucocutaneous symptoms of the skin, buccal, and gingival mucosa. Infection can also occur as a neonatal, central nervous system, or ocular infection. Specific ocular manifestations include blepharoconjunctivitis, epithelial and stromal keratitis (known as herpetic keratitis), uveitis, and retinitis. Herpetic keratitis is the most common cause of infectious corneal blindness in the United States and is often difficult to treat [1,3].
Antiviral treatment of HSV infection relies on nucleoside analogs such as acyclovir (ACV) and penciclovir. ACV is phosphorylated by the viral thymidine kinase and then competes with dGTP as a precursor in the synthesis of viral DNA by the viral DNA polymerase. Resistance to ACV has become a problem, however, with mutations of the viral thymidine kinase gene having been found to be responsible for such resistance in about 95% of cases. Mutations of the viral DNA polymerase have also been associated with ACV resistance in a small proportion of HSV isolates [4]. Given the increasing prevalence of such resistance, the development of new strategies to combat HSV infection is warranted.
Photodynamic therapy is based on the deleterious effect on cells and living organisms of the combination of light and highly sensitive molecules that interact with light (photosensitizers) and has long been known to inhibit the activity of viruses including herpes viruses [5-7]. The development of photodynamic antimicrobial chemotherapy (PACT) has focused not only on the synthesis of new photosensitizers but also on potential side effects on normal human cells. Human cells (keratinocytes) have been found to survive PACT under conditions that are lethal to microorganisms, with the difference in susceptibility likely due to differences in cell size and structure. Human cells are 25 to 50 times as large as bacterial cells and thus require more damage for the induction of cell death [8]. PACT with the combination of heterotricyclic dyes and irradiation with an ordinary fluorescent light was found to be effective for treating localized recurrent herpes simplex lesions of the skin and mucous membranes [9]. PACT with porphyrin photosensitizers such as hematoporphyrin derivatives and dihematoporphyrin ether has also been tested and found to be efficacious for treating the clinical manifestations of HSV infection [10].
With the goal of developing new treatments for corneal infection, we recently showed that PACT with a new porphyrin derivative, TONS 504, as the photosensitizer and a light-emitting diode (LED) as the light source was effective in eliminating methicillin-sensitive Staphylococcus aureus (MSSA) and methicillin-resistant S. aureus (MRSA) in vitro [11]. We investigated the effectiveness of PACT with this system against HSV-1 and ACV-resistant HSV-1 as well as evaluated the possible side effects of this treatment on host cells.
Methods
Cells and viruses
The human FL cell (ATCC®CCL-62™: American Type Culture Collection, Manassas, VA) was kindly provided by S. Takao (Hiroshima Prefectural Technology Research Institute, Hiroshima, Japan). The cells were maintained under 5% CO2 at 37 °C in Dulbecco’s minimum essential medium (DMEM; Life technologies Japan, Tokyo, Japan) supplemented with 10% fetal bovine serum, penicillin G (100 U/ml), and streptomycin (100 mg/l). HSV-1 strain VR-3 [12] and two strains of ACV-resistant HSV-1 (Takiguchi strain and Inoue strain) kindly provided by Y. Inoue (Tottori University, Yonago, Japan) [13] were described previously and were propagated in FL cells. Uninfected cells were incubated with DMEM instead of the virus inoculum. Viral infectivity was measured as the 50% tissue culture infective dose (TCID50) as described previously [12].
The photosensitizer
TONS 504 [13,17-bis (1-carboxyethyl) carbamoyl (3-methylpyridine)-3-(1,3-dioxane-2-yl) methylidene-8-ethenyl-2-hydroxy-2,7,12,18-tetramethyl chlorin, diN-methyl iodide (C51H58O5I2)], a hydrophilic and cationic porphyrin derivative with a greenish color and molecular weight of 1116.9, was obtained from Porphyrin Laboratory (Okayama, Japan) [11]. It was dissolved in sterile double-distilled water and serially diluted with DMEM to the desired concentrations (0.01 to 10 mg/l).
LED system
An LED system that provides a single light beam with a specific wavelength of 660 nm was obtained from CCS (Kyoto, Japan). The LED power was measured with an optical power meter (Hioki, Nagano, Japan) during each experiment. The increase in temperature conferred by the LED device was measured with a wire thermometer placed inside the culture dish during irradiation. We found that irradiation at a distance of 5 cm from the light source to the bottom of the dish yielded light power of 0.055 W and light energy of 10 J/cm2 over 3 min. Based on the temperature measurements, we included a 1-min rest period between each 3-min light exposure to avoid a problematic increase in the temperature of the incubation medium (data not shown) [11].
PACT for HSV-1 or ACV-resistant HSV-1
Confluent FL cells grown on a coverslip (9 mm x 28 mm) in a 30-mm culture dish were inoculated with multiple infections of 0.02 (moi) for 1 h, washed with serum-free medium to remove excess virus, and then incubated in serum-free DMEM for 3 h. The medium was then replaced with DMEM containing TONS 504 (0.01, 0.1, 0.5, 1, or 10 mg/l), and the cells were incubated for 5 min before exposure to the LED at 10 J/cm2 (single 3-min exposure), 20 J/cm2 (two 3-min exposures separated by a 1-min rest period), or 30 J/cm2 (three 3-min exposures separated by two 1-min rest periods) followed by culture under 5% CO2 at 37 °C for 24 h. The selected TONS 504 and LED doses were chosen to be similar to a previous study [11].
Immunocytofluorescence analysis
Cells were fixed with 3.7% formaldehyde and stained with rabbit antibodies to HSV-1 (Diagnostic Biosystems, Pleasanton, CA) and Alexa Fluor 488–conjugated goat antibodies to rabbit immunoglobulin G (Molecular Probes, Eugene, OR). Nuclei were also stained with 4′,6-diamidino-2-phenylindolse (DAPI; Vector Laboratories, Burlingame, CA). The stained cells were examined with a BZ-X700 fluorescence microscope (Keyence, Osaka, Japan). The efficacy of PACT was determined based on immunocytofluorescence detection of HSV-1. The safety of PACT was determined by evaluation of host cell viability based on manual counting of normal nuclei stained with DAPI [14] in each of ten fields of 100 μm2 at a magnification of 400X.
Measurement of cell lysis
The possible cytotoxic effect of PACT on FL cells was also evaluated by measurement of the release of lactate dehydrogenase (LDH). The culture supernatants of cells subjected to PACT were transferred to the wells of a 96-well plate and assayed for LDH with the use of a Cytotoxicity Detection Kit Plus (Roche, Basel, Switzerland). Absorbance at 595 and 490 nm was measured with a microplate reader (Model 550: Bio-Rad, Richmond, CA).
Results
Efficacy of TONS 504-PACT for HSV-1 and ACV-resistant HSV-1
Immunocytofluorescence analysis revealed that PACT with TONS 504 as the photosensitizer and an LED as the light source (TONS 504-PACT) resulted in eradication of HSV-1 and two strains (the Takiguchi strain and the Inoue strain) of ACV-resistant HSV-1 from infected FL cells within 24 h in a manner dependent on LED energy and TONS 504 concentration (Table 1, Figure 1). The infected cells manifested the cell–cell fusion and giant cell formation characteristic of HSV infection [15], with inactivation of HSV-1 apparent from the loss of immunofluorescence at the host cell membrane. For cells infected with either HSV-1 or ACV-resistant HSV-1 (the Takiguchi strain or the Inoue strain) and irradiated at 10 J/cm2, the antiviral effect of TONS 504-PACT was complete at a TONS 504 concentration of 10 mg/l, whereas for infected cells irradiated at 20 or 30 J/cm2 the antiviral effect was complete at a TONS 504 concentration of 1 mg/l. For cells infected with either virus strain, exposure to TONS 504 without irradiation or irradiation in the absence of TONS 504 had no apparent antiviral effect.
Table 1. Efficacy of TONS 504–PACT against HSV-1 and two-strains of ACV-resistant HSV-1 (Takiguchi strain and Inoue strain).
| LED energy (J/cm2) | TONS 504 concentration (mg/l) |
|||||
|---|---|---|---|---|---|---|
| 0 | 0.01 | 0.1 | 0.5 | 1 | 10 | |
| 0 |
+ |
+ |
+ |
+ |
+ |
+ |
| 10 |
+ |
+ |
+ |
+ |
+ |
– |
| 20 |
+ |
+ |
+ |
+ |
– |
– |
| 30 | + | + | + | + | – | – |
Complete (–) or incomplete (+) eradication of virus from infected FL cells was judged on the basis of immunocytofluorescence analysis. Identical results were obtained with cells infected with HSV-1 or ACV-resistant HSV-1 (Takiguchi strain or Inoue strain). Data represent a summary of 3 independent experiments performed with each virus strain.
Figure 1.
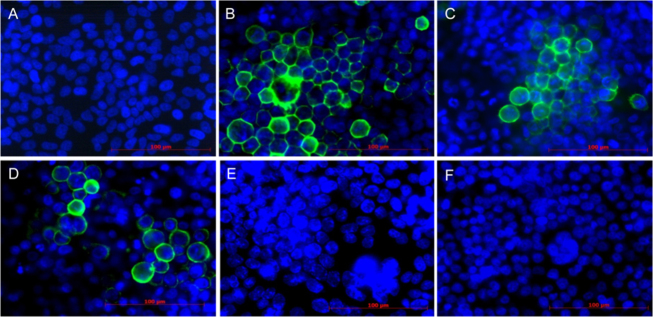
Immunocytofluorescence analysis of the efficacy of TONS 504–PACT against HSV-1 and ACV-resistant HSV-1. Uninfected FL cells (A) as well as FL cells infected with herpes simplex virus type 1 (HSV-1) and either left untreated (B) or subjected to TONS 504–photodynamic antimicrobial chemotherapy (PACT) at a light-emitting diode (LED) energy of 20 J/cm2 and TONS 504 concentrations of 0.1 mg/l (C), 0.5 mg/l (D), 1 mg/l (E), or 10 mg/l (F) were subjected to immunocytofluorescence staining with antibodies to HSV-1 (green fluorescence). Cell nuclei were also stained with 4′,6-diamidino-2-phenylindolse (DAPI; blue fluorescence). Similar results were obtained for cells infected with two strains of acyclovir (ACV)-resistant HSV-1. Scale bars, 100 μm.
Evaluation of the cytotoxicity of TONS 504-PACT
We first examined the possible effect of TONS 504-PACT on host cell viability by manually counting the number of apparently normal nuclei. Data for infected cells from three independent experiments revealed the lack of effect of TONS 504-PACT on host cell viability as evaluated based on nuclear morphology compared with the corresponding value for uninfected cells not subjected to TONS 504-PACT. There are no significant differences at all conditions of treatment (data not shown). PACT did not exhibit cytotoxicity at any of the TONS 504 concentrations and light energies examined. We then determined the possible effect of TONS 504-PACT on the integrity of the host cells by measuring the release of LDH into the incubation medium (Figure 2 A,B). Whereas the extent of LDH release was increased slightly after exposure of infected FL cells to the higher concentrations of TONS 504 or higher light energies, these effects were not statistically significant (p>0.05, Student t test).
Figure 2.
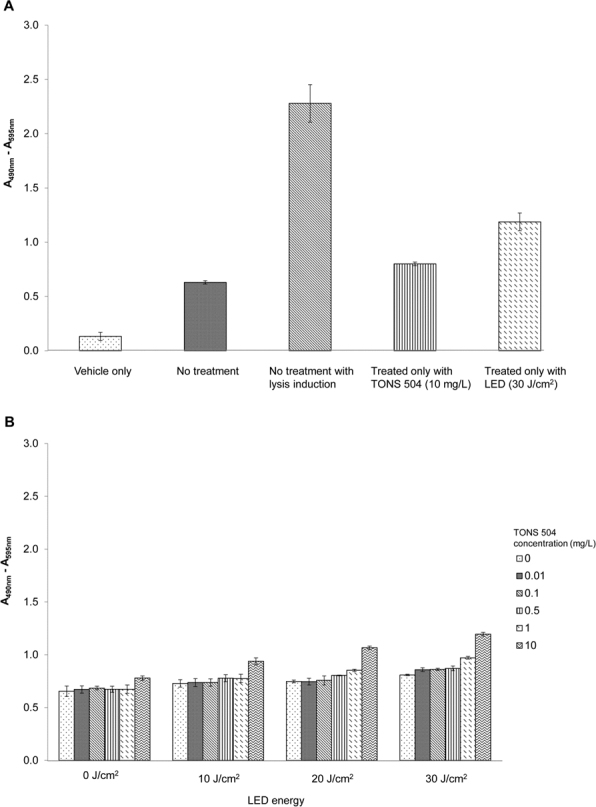
Lack of cytotoxicity of TONS 504–PACT for host cells as evaluated with measurement of LDH release. A: Control conditions. Serum-free medium alone (vehicle only) or culture supernatants of uninfected FL cells that were either not subjected to TONS–504 photodynamic antimicrobial chemotherapy (PACT), not subjected to TONS 504–PACT but exposed to a cell lysis reagent, exposed to TONS 504 (10 mg/l) without irradiation, or subjected to irradiation (30 J/cm2) in the absence or TONS 504 were assayed for lactate dehydrogenase (LDH). B: Experimental conditions. FL cells infected with herpes simplex virus type 1 (HSV-1) or acyclovir (ACV)-resistant HSV-1 were subjected to TONS 504–PACT under the indicated conditions, after which culture supernatants were assayed for LDH. All data are presented as absorbance at 490 nm (A490) minus A595 and are means ± SD for three independent experiments.
Discussion
Our results have demonstrated TONS 504-PACT has an antiviral effect on HSV-1 and two-strains of ACV-resistant HSV-1 in vitro. The combination of TONS 504 as the photosensitizer and an LED as the light source was required to achieve this effect. Furthermore, we found that TONS 504-PACT was safe for host cells.
PACT with hematoporphyrin and other dyes has been shown to inactivate HSV in vitro [16]. Porphyrins form radicals that may give rise to lipid peroxidation as well as damage DNA and protein molecules. The antiviral action of PACT is thought to result largely from damage to DNA and cytoplasmic membranes. Photosensitizers produce free radicals by two types of reaction. In the type I reaction, the photosensitizer in the excited triplet state reacts directly with and transfers an atom to a target molecule to form a radical anion or cation, which then reacts with oxygen to produce reactive oxygen species. In the type II reaction, the photosensitizer transfers its energy directly to molecular oxygen to generate reactive oxygen species [17].
We previously showed that TONS 504-PACT is effective for eliminating MSSA and MRSA in vitro [11]. In the present study, we applied immunocytofluorescence analysis to show that TONS 504-PACT is also effective against HSV-1 and ACV-resistant HSV-1. This approach allowed us not only to detect the elimination of HSV-1 immunostaining from infected cells subjected to TONS 504-PACT but also to demonstrate that PACT appeared to have no detrimental effect on the host cells. This apparent lack of cytotoxicity of TONS 504-PACT was confirmed by measurement of LDH release from the treated cells, which revealed no induction of cell lysis.
We found that TONS 504-PACT was equally effective against HSV-1 and two strains of ACV-resistant HSV-1 (the Takiguchi strain and the Inoue strain). This finding highlights an advantage of PACT over antiviral drugs in that the reactive oxygen species generated by PACT are not influenced by the mutations of thymidine kinase that give rise to ACV resistance. This advantage is in addition to the lack of side effects of TONS 504-PACT on host cells.
There are several limitations in this study. First, the comparison between TONS 504 with other hematoporphyrin derivatives should be addressed. Second, additional experiments should measure infectious progeny virus production with standard plaque assay, real-time RT–PCR, or western blot. However, the results in this study were in an all-or-nothing manner: The cells were either brightly stained or not stained at all.
In conclusion, we have shown that PACT with the novel porphyrin derivative TONS 504 as the photosensitizer and an LED as the light source inactivates HSV-1 and ACV-resistant HSV-1 in vitro without a deleterious effect on host cells. Together with the results of our previous study that demonstrated the antibacterial efficacy of TONS 504-PACT against MSSA and MRSA [11], our present data suggest that this system may prove effective against a wide range of pathogens. Both studies were performed to provide a basis for the development of new treatments for corneal infection. Further investigations are now warranted to pursue TONS 504-PACT as a new mode of treatment for bacterial and viral infections of the cornea.
Acknowledgments
We thank Yoshitsugu Inoue (Tottori University, Yonago, Japan) for providing two-different strains of ACV-resistant HSV-1 (Takiguchi strain and Inoue strain), Isao Sakata (Porphyrin Laboratory, Okayama, Japan) for providing information on TONS 504 as well as Akira Ichikawa (CCS Inc., Kyoto, Japan) for producing the LED device according to our experimental design. Funding: This work is supported by Adaptable and Seamless Technology transfer Program through target-driven R&D (A-STEP: AS232Z01652F to TC) of the Japan Science and Technology Agency (JST).
References
- 1.Whitley RJ, Kimberlin DW, Roizman B. Herpes simplex viruses. Clin Infect Dis. 1998;26:541–553. doi: 10.1086/514600. [DOI] [PubMed] [Google Scholar]
- 2.Whitley RJ, Roizman B. Herpes simplex virus infections. Lancet. 2001;357:1513–8. doi: 10.1016/S0140-6736(00)04638-9. [DOI] [PubMed] [Google Scholar]
- 3.Guess S, Stone DU, Chodosh J. Evidence-based treatment of Herpes simplex virus keratitis: a systemic review. Ocul Surf. 2007;5:240–50. doi: 10.1016/s1542-0124(12)70614-6. [DOI] [PubMed] [Google Scholar]
- 4.Bacon TH, Levin MJ, Leary JL, Sarisky RT, Sutton D. Herpes simplex virus resistance to acyclovir and penciclovir after two decades of antiviral therapy. Clin Microbiol Rev. 2003;16:114–28. doi: 10.1128/CMR.16.1.114-128.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wallis C, Melnick JL. Irreversible photosensitization of viruses. Virology. 1964;23:520–7. doi: 10.1016/0042-6822(64)90236-3. [DOI] [PubMed] [Google Scholar]
- 6.Macdonald IJ, Dougherty TJ. Basic principles of photodynamic therapy. J Porphyr Phthalocyanines. 2001;5:105–29. [Google Scholar]
- 7.Ackroyd R, Kelty C, Brown N, Reed M. The history of photodetection and photodynamic therapy. Photochem Photobiol. 2001;74:656–69. doi: 10.1562/0031-8655(2001)074<0656:thopap>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 8.Zeina B, Greenman J, Corry D, Purcell WM. Antimicrobial photodynamic therapy: assessment of genotoxic effects on keratinocytes in vitro. Br J Dermatol. 2003;148:229–32. doi: 10.1046/j.1365-2133.2003.05091.x. [DOI] [PubMed] [Google Scholar]
- 9.Felber TD, Smith EB, Knox JM, Wallis C, Melnick JL. Photodynamic inactivation of Herpes Simplex; report of a clinical trial. JAMA. 1973;223:289–92. [Google Scholar]
- 10.Wainwright M. Photoinactivation of viruses. Photochem Photobiol Sci. 2004;3:406–11. doi: 10.1039/b311903n. [DOI] [PubMed] [Google Scholar]
- 11.Latief MA, Chikama T, Shibasaki M, Sasaki T, Ko JA, Kiuchi Y, Sakaguchi T, Obana A. Antimicrobial action from a novel porphyrin derivative in photodynamic antimicrobial chemotherapy in vitro. Lasers Med Sci. 2015;30:383–387. doi: 10.1007/s10103-014-1681-6. [DOI] [PubMed] [Google Scholar]
- 12.Ueda K, Kawabata R, Irie T, Nakai Y, Tohya Y, Sakaguchi T. Inactivation of pathogenic viruses by plant-derived Tannins: strong effects of extracts from Persimmon (Diospyros kaki) on a broad range of viruses. PLoS ONE. 2013;8:e55343. doi: 10.1371/journal.pone.0055343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yao YF, Inoue Y, Kase T, Uchihori Y, Mori Y, Ohashi Y. Clinical characteristics of acyclovir-resistant herpetic keratitis and experimental studies of isolates. Graefes Arch Clin Exp Ophthalmol. 1996;234:S126–32. doi: 10.1007/BF02343061. [DOI] [PubMed] [Google Scholar]
- 14.Bridger JM, Kill IR. Aging of Hutchinson-Gilford progeria syndrome fibroblasts is characterised by hyperproliferation and increased apoptosis. Exp Gerontol. 2004;39:717–724. doi: 10.1016/j.exger.2004.02.002. [DOI] [PubMed] [Google Scholar]
- 15.Muggeridge MI. Characterization of cell-cell fusion mediated by herpes simplex virus 2 glycoproteins gB, gD, gH and gL in transfected cells. J Gen Virol. 2000;81:2017–27. doi: 10.1099/0022-1317-81-8-2017. [DOI] [PubMed] [Google Scholar]
- 16.Schnipper LE, Lewin AA, Swartz M, Crumpacker CS. Mechanisms of photodynamic inactivation of Herpes Simplex viruses; comparison between methylene blue, light plus electricity, and hematoporphyrin plus light. J Clin Invest. 1980;65:432–8. doi: 10.1172/JCI109686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Smetana Z, Ben-Hur E, Mendelson E, Salzberg S, Wagner P, Malik Z. Herpes Simplex virus proteins are damaged following photodynamic inactivation with Phthalocyanines. J Photochem Photobiol B. 1998;44:77–83. doi: 10.1016/S1011-1344(98)00124-9. [DOI] [PubMed] [Google Scholar]


