Abstract
Unique intercellular junctional complexes between the central nervous system (CNS) microvascular endothelial cells and the choroid plexus epithelial cells form the endothelial blood–brain barrier (BBB) and the epithelial blood–cerebrospinal fluid barrier (BCSFB), respectively. These barriers inhibit paracellular diffusion, thereby protecting the CNS from fluctuations in the blood. Studies of brain barrier integrity during development, normal physiology, and disease have focused on BBB and BCSFB tight junctions but not the corresponding endothelial and epithelial adherens junctions. The crosstalk between adherens junctions and tight junctions in maintaining barrier integrity is an understudied area that may represent a promising target for influencing brain barrier function.
Introduction
The endothelial blood–brain barrier (BBB), composed of the highly specialized central nervous system (CNS) microvascular endothelial cells, and the epithelial blood–cerebrospinal fluid barrier (BCSFB), composed of the choroid plexus epithelial cells, protect the CNS from the constantly changing milieu in the bloodstream as well as infections and toxins, and are thus essential for maintaining CNS homeostasis (Fig. 1). Work on how these barriers prevent paracellular diffusion of harmful elements into the CNS usually focuses on characterization of the highly complex tight junctions (TJs) of the BBB, which resemble epithelial TJs (Kniesel et al., 1994; Liebner et al., 2000b), and the unique parallel running TJ strands of the BCSFB (Engelhardt et al., 2001; Wolburg and Paulus, 2010). Yet, formation of TJs generally requires the existence of adherens junctions (AJs), and central to the organization of dynamic junctions is the continuous crosstalk between components of AJs and TJs, a fact often neglected by researchers studying the BBB and the BCSFB. AJs do certainly exist in these barriers and are established between neighboring cells by the homophilic interaction between the transmembrane proteins vascular endothelial cadherin (VE-cadherin) and epithelial cadherin (E-cadherin) in CNS endothelial and choroid plexus epithelial cells, respectively (Vorbrodt and Dobrogowska, 2003).
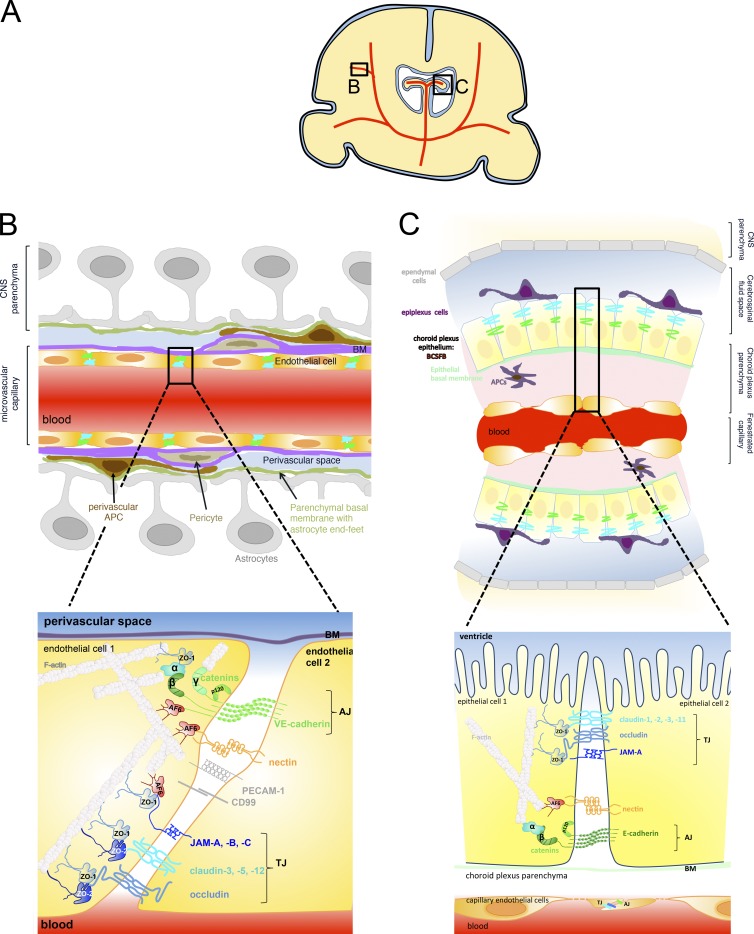
Figure 1.
Intercellular junctions between CNS microvascular endothelial cells forming the BBB and between choroid plexus epithelial cells forming the BCSFB. (A) Schematic of the localization of the brain barriers in parenchymal microvessels (B) and the choroid plexus in the ventricles (C) in a coronal brain section. (B) Localization (top) and molecular composition (bottom) of endothelial BBB AJs and TJs. AJs at the BBB are established by the homophilic interaction of cis dimers of transmembrane VE-cadherin in between adjacent endothelial cells. The cytoplasmic tail of VE-cadherin binds the armadillo family proteins p120- and β-catenin, which, via interaction with α-catenin and afadin (AF-6), mediate the link to the actin cytoskeleton. Nectins are transmembrane proteins belonging to the immunoglobulin (Ig) superfamily that form homodimers in cis and contribute to AJ formation by adhering to nectins on the adjacent cell and binding AF-6 via their cytoplasmic tail, which connects nectin with the actin cytoskeleton. VE-cadherin can also bind to plakoglobin (γ-catenin) instead of to β-catenin and thus connect endothelial AJs to the intermediate filaments in addition to the actin cytoskeleton. Although drawn separately in this figure, TJ strands that encircle the entire circumference of the CNS endothelial cells are intermingled with AJs. The transmembrane TJ proteins found to be localized in CNS endothelium are occludin, claudin-3, claudin-5, and claudin-12 as well as the junctional adhesion molecules (JAM) A, B, and C. Both claudins and JAMs mediate homophilic and heterophilic cis and trans interactions within their family, thus sealing the paracellular cleft between the adjacent endothelial cells. With the exception of claudin-12, the transmembrane TJ proteins carry a PDZ-binding motif in their carboxy terminus, through which they bind the scaffolding proteins ZO-1 and ZO-2 and, in the case of JAMs, also AF-6. These scaffolding proteins mediate the link to the actin cytoskeleton. ZO-1 has also been shown to bind to α-catenin and F-actin and thus regulate AJs. Additional molecules found to be present in BBB cell-to-cell contacts are the Ig supergene family members ESAM, PECAM-1, and CD99. Linkage of TJ proteins to cell polarity complexes are omitted from this figure for reasons of simplicity. (C) Localization (top) and molecular composition (bottom) of the epithelial BCSFB established by choroid plexus epithelial cells. The microvessels within the highly vascularized choroid plexus are fenestrated capillaries that allow the free diffusion of water-soluble molecules across the vascular wall. Nevertheless, these vascular endothelial cells form AJs and TJs resembling those of peripheral vascular beds and are thus not drawn in detail here. AJs of the BCSFB are formed by the homophilic interaction of cis homodimers of the transmembrane protein E-cadherin. Via its cytoplasmic tail, E-cadherin binds the armadillo family members p120- and β-catenin, which, in turn bind α-catenin, linking E-cadherin to the epithelial actin cytoskeleton. Oriented apically relative to the AJs, the TJ strands of the BCSFB run in parallel around the entire circumference of the choroid plexus epithelial cells, with characteristic gaps that have been suggested to be important in the context of the transport functions of the choroid plexus. The transmembrane proteins that have been characterized as localized to the BCSFB TJs include occludin, claudin-1, claudin-2, claudin-3, and claudin-11 as well as JAM-A and JAM-C. Via their carboxy terminus, which harbors a PDZ domain binding motif, these proteins bind the scaffolding proteins ZO-1, ZO-2, and probably ZO-3, which link to the actin cytoskeleton. BM, basement membrane.
TJs and AJs are considered to have distinct functions. TJs regulate the diffusion of solutes and ions through the paracellular route, which is referred to as their “gate” function. TJs also establish a “fence” function by limiting the free movements of lipids and proteins from the apical and basolateral cell surfaces, thus contributing to cell polarity (Dejana et al., 2009). In addition to the TJs between two adjacent cells, a special molecularly distinct TJ has been described at tricellular contacts, where the corners of three endothelial or epithelial cells meet. Interestingly, these tricellular TJs exist in BBB endothelial cells and BCSFB epithelial cells (Iwamoto et al., 2014), suggesting that tricellular TJs may be required for full barrier function of both types of brain barriers. Before TJ formation, AJs initiate cell-to-cell contacts and promote their maturation, maintenance, and plasticity, and regulate tensile forces. Yet, far less is known about the underlying role of AJs in regulating dynamics of cell–contacts and the establishment of cell polarity at the brain barriers.
In epithelial cells, including the choroid plexus epithelium, TJs are the most apical component of the junctional complex and thus clearly distinguishable from AJs. In endothelial cells, however, localization of TJs and AJs is more variable, and both junctional complexes appear intermingled, especially in brain endothelial cell-to-cell junctions (Schulze and Firth, 1993; Vorbrodt and Dobrogowska, 2003; Dejana et al., 2009). Given that endothelial AJs in peripheral vascular beds play an important role in the control of vascular permeability (Giannotta et al., 2013), the contribution of these junctions to the integrity of BBB junctions during development, normal physiology, and disease should be elucidated. This is underscored by recent evidence demonstrating that the TJ protein zonula occludens 1 (ZO-1) is also a central regulator of endothelial AJs (Tornavaca et al., 2015).
CNS endothelial and epithelial AJs and TJs: Components and dynamics
At first glance the basic molecular makeup of BBB and BCSFB AJs and TJs resembles that of general endothelial and epithelial barriers (Fig. 1 and Table 1), and most work on the function of junctional molecules in regulating brain barriers has been extrapolated from studies on their non-CNS counterparts. Yet, their high complexity and continuity distinguishes brain barrier TJs from peripheral TJs, and there are also important differences in their molecular makeup. In addition to the unique combinations of claudins observed in BBB and BCSFB TJs (Fig. 1 and Table 1), high expression levels of the integral TJ proteins occludin and claudin-5 and low expression levels of the integral AJ protein VE-cadherin characterize BBB endothelial cells (Table 1). This influences spatial arrangement of the cytoplasmic junctional scaffolding proteins, and thus affects characteristics of the anchorage of brain barrier junctional complexes to the cytoskeleton and, ultimately, the downstream signaling events regulating junctional dynamics (Table 1; Goddard and Iruela-Arispe, 2013; Citi et al., 2014). Such events include junctional protein phosphorylation, the continuous recycling of junctional molecules, and regulation of cytoskeletal dynamics by Rho family GTPases (summarized in Citi et al. [2014] and Goddard and Iruela-Arispe [2013]). Unfortunately, few of these processes have been analyzed specifically in brain endothelial or choroid plexus epithelial cells. In particular, junction dynamics in brain endothelial cells could be very different, as pinocytotic activity is very low in brain endothelial cells. If endocytic activity was also low, this would slow down the recycling of junctional components such as cadherins. Furthermore, despite the generation of elegant mouse mutants to interrogate the specific role of individual junctional components in regulating AJ or TJ dynamics, careful analysis of brain barriers has yet to be performed in these models (Schulte et al., 2011; Wessel et al., 2014).
Table 1.
Expression and known function of junctional molecules at brain barriers
| Junctional molecule | General characteristics | Brain barrier expression/localization | Function in cell-to-cell contacts |
| Transmembrane AJ proteins | |||
| E-cadherin (uvomorulin, L-CAM) | Main integral membrane protein of epithelial AJs; member of classical cadherin family (Type I) mediating homophilic adhesions in cis and trans; links to actin cytoskeleton via catenin complex (Vestweber, 2015) | Basolateral localization in rat and chicken choroid plexus epithelium (Marrs et al., 1993) and epithelial cells of benign human choroid plexus tumors (Figarella-Branger et al., 1995) | Core homophilic cell adhesion molecule of epithelial AJs, controls epithelial cell contact formation, barrier integrity, junctional plasticity, and cytoskeletal tension |
| VE-cadherin (cadherin-5) | Main integral membrane protein of endothelial AJs; member of the classical cadherin family (Type II) mediating homophilic adhesions in cis and trans; cytoplasmic domain links AJs via catenin complex to actin cytoskeleton and intermediate filaments (for review see Dejana and Vestweber, 2013) | At junctions in mouse CNS blood vessels even during embryonic development (Breier et al., 1996); localizes to junctions in rat and human brain microvessels (Vorbrodt and Dobrogowska, 2003) | Core homophilic cell adhesion molecule of endothelial AJs; controls endothelial cell survival, stabilization of blood vessel assembly, and vascular permeability (Carmeliet et al., 1999; Crosby et al., 2005) |
| N-cadherin (cadherin-2) | Transmembrane protein of the classical cadherin family (Type I) | Protein detected during early angiogenesis at the abluminal surface of eye and brain endothelial cells (Gerhardt et al., 1999) | Mediates pericyte–endothelial interactions during brain angiogenesis (Gerhardt et al., 2000) |
| VE-PTP | Endothelial-specific phospho tyrosine phosphatase (VE-PTP) | VE-PTP-LacZ reporter expression observed in brain vessels during embryonic development (Bäumer et al., 2006) | Controls VE-cadherin phosphorylation and thus vascular junctional integrity and leukocyte diapedesis (Küppers et al., 2014) |
| Nectin | Member of the nectin family of the Ig superfamily; mediates homophilic or heterophilic interactions with other nectins (Indra et al., 2013); highly conserved cytoplasmic tail binds to the adaptor protein afadin (AF-6), linking nectins to actin cytoskeleton | Afadin observed in choroid plexus epithelium, suggesting presence of nectin (Lagaraine et al., 2011) | Nectin–afadin complex involved in the formation of AJs (Ikeda et al., 1999) and TJs (Takai et al., 2003) |
| Transmembrane TJ proteins | |||
| Occludin | Integral membrane protein localized exclusively to TJs (Furuse et al., 1993); Type II transmembrane protein with a tetraspanning Marvel (MAL and related proteins for vesicle trafficking and membrane link) motif; member of the TAMP family (like Tricellulin and MarvelD3) sharing the Marvel domain (Raleigh et al., 2010) | High expression and strong junctional localization in chicken brain microvessels (Furuse et al., 1993; Hirase et al., 1997) and in mouse BCSFB epithelial cells (Wolburg et al., 2001; Diez-Roux et al., 2011; Kratzer et al., 2012) | Links TJs to the actin cytoskeleton by recruiting scaffolding proteins of the MAGUK (membrane associated with a guanylyl kinase-like domain) family, such as zonula occludens 1 (ZO-1), ZO-2, and ZO-3 (Morita et al., 1999; Nasdala et al., 2002); regulates Ca2+ transport across BBB (Saitou et al., 2000); localizes tricellulin to tricellular TJs (Ikenouchi et al., 2008); regulates paracellular permeability via its phosphorylation status (Raleigh et al., 2011) |
| Tricellulin | TAMP family member (like occludin; Raleigh et al., 2010) | Restricted localization to tricellular TJs of BBB, recruited by the angulin/LSR-family (Furuse et al., 2014); absent in fenestrated endothelial cells of choroid plexus (Iwamoto et al., 2014) | Required for full barrier formation of in vitro cultured epithelial cells (Iwamoto et al., 2014) |
| Claudin-1 | Member of the claudin family of tetraspanning TJ-specific membrane proteins (Furuse et al., 1998); form the backbone of TJs by establishing homophilic and heterophilic interactions in cis and trans via their extracellular loops (Piontek et al., 2008); essential and sufficient to induce TJs in fibroblasts (Furuse et al., 1998); PDZ-binding motif at the claudin carboxy terminus mediates interaction with the scaffolding proteins ZO-1, ZO-2, and ZO-3, which contributes to TJ strand organization (for review see Van Itallie and Anderson, 2014) | mRNA detected in mouse BCSFB (Kratzer et al., 2012) and protein localized to mouse BCSFB junctions (Wolburg et al., 2001); discrepant observations of expression in BBB: there is an absence of mRNA and protein in mouse brain parenchymal brain microvessels but a presence of protein in meningeal brain microvessels (Pfeiffer et al., 2011); protein detected in brain endothelial TJs (Haseloff et al., 2015) | Induces and seals TJs; regulates paracellular movement of water (skin) and macromolecules (Furuse et al., 2002); claudin-1 overexpression in brain endothelial cells of mouse models of neuroinflammation reduces BBB leakiness and ameliorates clinical disease (Pfeiffer et al., 2011). |
| Claudin-2 | See general comments on claudin family above for claudin-1 | mRNA and protein localizes to BCSFB (Wolburg et al., 2001; Kratzer et al., 2012) | This pore forming claudin (Furuse et al., 2001) regulates paracellular ion and water flow; claudin-2–deficient mice are viable, BCSFB function has not yet been analyzed (Muto et al., 2010) |
| Claudin-3 | See general comments on claudin family above for claudin-1 | Detected in TJs in brain endothelial cells in some studies (Wolburg et al., 2003) but not in others (Ohtsuki et al., 2008; Daneman et al., 2010a); induced during brain angiogenesis by canonical Wnt/β-catenin signaling (Liebner et al., 2008); expressed in choroid plexus epithelial cells and localizes to TJs (Wolburg et al., 2003; Kratzer et al., 2012; Kooij et al., 2014) | Expression is induced during brain angiogenesis and correlates with barriergenesis (Liebner et al., 2008); sealing of BCSFB but not BBB TJs under neuroinflammatory conditions as detected in claudin-3–deficient mice (Kooij et al., 2014) |
| Claudin-5 | Endothelial cell–specific component of TJ strands (Morita et al., 1999) | Highly expressed in BBB TJs in zebrafish, rodents, nonhuman primates, and humans (Nitta et al., 2003; Jeong et al., 2008; Hoshi et al., 2013); junctional localization in zebrafish BCSFB (Henson et al., 2014) | Claudin-5–deficient mice do not survive due to BBB leakiness to molecules <800 D (Nitta et al., 2003); focal loss of claudin-5 correlates with BBB dysfunction (Zhou et al., 2014) |
| Claudin-11 | Induces parallel-array TJ strands in myelin sheaths of oligodendrocytes (Gow et al., 1999); claudin-11–deficient mice develop neurological deficits including deafness (Gow et al., 1999, 2004) | Localizes to TJs of BCSFB in mice (Wolburg et al., 2001) | TJs of the BCSFB are characterized by parallel running particle strands (Wolburg and Paulus, 2010) probably induced by claudin-11 |
| Claudin-12 | See general comments on the claudin family above for claudin-1; claudin-12 lacks a C-terminal PDZ-binding motif | Shown to be specifically expressed in and to localize to TJs of brain endothelial cells by some laboratories (Nitta et al., 2003; Schrade et al., 2012) but not by others (Ohtsuki et al., 2008; Daneman et al., 2010a) | Function at BBB unknown |
| Additional claudins | See general comments on claudin family above | mRNA and protein of additional claudins detected in mouse choroid plexus (Kratzer et al., 2012); mRNA of additional claudins detected in purified brain microvessels (Hoshi et al., 2013); additional claudin proteins detected in rat and marmoset brain microvessels (Ohtsuki et al., 2008) | Functions in brain barriers unknown |
| JAM-A (Junctional adhesion molecule A, JAM-1) | Type I transmembrane protein and member of the classical JAM family of the Ig superfamily; two extracellular Ig-like domains, cytoplasmic tail with a PDZ-binding motif; JAMs engage in homophilic and heterophilic interactions among their family members and bind integrins (Martìn-Padura et al., 1998; for review see Garrido-Urbani et al., 2014) | Expressed in and localized to mouse and human BBB endothelial TJs (Aurrand-Lions et al., 2001b; Vorbrodt and Dobrogowska, 2004; Padden et al., 2007) | Regulates cell polarity by intracellular association with the PAR-3/atypical protein kinase C (aPKC)/PAR-6 complex (Ebnet et al., 2001) and leukocyte trafficking across endothelial barriers including the BBB (Martìn-Padura et al., 1998; Williams et al., 2013); loss of vascular JAM-A immunostaining in human brain tissue correlates with BBB leakiness (Padden et al., 2007) |
| JAM-B (human JAM-2; human and mouse VE-JAM; mouse JAM-3) | See explanation for JAM-A | Expressed in and localized to TJs at mouse BBB (Aurrand-Lions et al., 2001a,b) | Leukocyte trafficking across skin endothelium (Ludwig et al., 2009); function at brain barriers is unknown |
| JAM-C (human JAM-3; mouse JAM-2) | See explanation for JAM-A | Localizes to TJs in mouse BBB and BCSFB (Arrate et al., 2001; Wyss et al., 2012) | Might contribute to human BBB integrity (Mochida et al., 2010); JAM-C−/− C57BL/6 mice develop a hydrocephalus independent of endothelial JAM-C, suggesting that JAM-C at the BCSFB might influence brain fluid homeostasis (Wyss et al., 2012) |
| Transmembrane proteins outside of organized AJs and TJs | |||
| PECAM-1 | Type I integral membrane protein of the Ig superfamily with six extracellular Ig domains, a short transmembrane, and a long cytoplasmic domain that can be serine and tyrosine phosphorylated upon cellular activation; highly expressed in all endothelial cells (summarized in Privratsky and Newman, 2014) | Highly expressed in endothelial cell-to-cell junctions outside of organized AJs and TJs, including the BBB (Graesser et al., 2002; Lyck et al., 2009) | Contributes to steady-state barrier function of endothelial cells; functions as a mechanosensor and accelerates restoration of barrier integrity following perturbations, including the BBB (Graesser et al., 2002; Privratsky and Newman, 2014) |
| CD99 and CD99L | Heavily O-glycosylated type I transmembrane proteins (Schenkel et al., 2002) | Localizes to endothelial cell-to-cell contacts including those of brain endothelial cells (Bixel et al., 2004). | Immune cell trafficking across brain endothelium in vitro (Bixel et al., 2004) |
| Intracellular scaffolding proteins of AJs | |||
| p120-catenin | Armadillo-repeat protein (Harris, 2012); binds to the juxtamembrane region of the cytoplasmic domain of both VE- and E-cadherin | Detected at ultrastructural level in AJs in human brain microvessels (Vorbrodt and Dobrogowska, 2004) | Stabilizes AJs by inhibiting constitutive endocytosis of cadherins; regulates activity of Rho family GTPases and thus actin cytoskeleton dynamics; recruits microtubules to the cadherin complex (Vestweber, 2015) |
| β-Catenin | Armadillo repeat protein (Harris, 2012); binds to the distal region of the cytoplasmic domain of VE- and E-cadherin and mediates interaction with α-catenin that engages F-actin, thus linking the AJ complex with the actin cytoskeleton (Buckley et al., 2014) | Localizes to AJs of BBB in chicken and mouse (Liebner et al., 2000a) and to AJs of BCSFB in rats (Lippoldt et al., 2000); early association with endothelial N-cadherin during brain angiogenesis followed by junctional localization (Liebner et al., 2000a); localizes to endothelial nuclei during brain angiogenesis to regulate transcription (summarized in Engelhardt and Liebner, 2014) | Stabilizes AJs by inhibiting proteolysis of cadherins (Vestweber, 2015); regulates BBB differentiation during brain angiogenesis by inducing expression of claudin-3 via the canonical Wnt/β-catenin signaling pathway (summarized in Engelhardt and Liebner, 2014) |
| γ-Catenin (plakoglobin) | Binds to the cytoplasmic tail of VE-cadherin | Shown to localize to AJ of BBB in chickens (Liebner et al., 2000a) | Links cadherin complex to intermediate filaments |
| α-Catenin | α-Catenin binds directly or indirectly to β-catenin and to the actin cytoskeleton | Localizes at the ultrastructural level to interendothelial junctions of vessels in human or mouse brain tissue sections (Vorbrodt and Dobrogowska, 2003); localizes to AJs at rat BCSFB (Lippoldt et al., 2000) | Anchors AJs to the actin cytoskeleton; regulates the adhesive function of cadherins (summarized in Vestweber, 2015) |
| Intracellular scaffolding protein TJs | |||
| ZO-1 (Zonnula occludens -1) | TJ scaffolding protein of the MAGUK (membrane associated with a guanylyl kinase-like domain) family; binds to integral TJ proteins, ZO-2, and F-actin (for review see Van Itallie and Anderson, 2014). | Localizes to BBB junctions in mouse (Nico et al., 1999) and human brain tissue (Kirk et al., 2003; Vorbrodt and Dobrogowska, 2003); localizes to TJs in mouse BCSFB (Wolburg et al., 2001; Kratzer et al., 2012); also localizes to AJs by binding to α-catenin and F-actin (Itoh et al., 1999) | ZO-1 organizes components of TJs in epithelial cells as well as both TJs and AJs in endothelial cells, and links them to the cortical actin cytoskeleton (Fanning et al., 1998; Itoh et al., 1999); central regulator of VE-cadherin–dependent AJs (Tornavaca et al., 2015) |
| ZO-2 | TJ scaffolding protein of the MAGUK family | Associates with junctions in primary bovine and human brain microvascular endothelial cells in vitro (Mark and Davis, 2002; Lee et al., 2009); ZO-2 mRNA detected in choroid plexus of mice (Kratzer et al., 2012) | Can replace ZO-1 in TJs |
| ZO-3 | TJ scaffolding protein of the MAGUK family | Low mRNA expression levels in mouse choroid plexus and brain microvascular endothelial cells (Kratzer et al., 2012) and mRNA and protein detected in choroid plexus of ewes (Lagaraine et al., 2011) | Can replace ZO-1 and ZO-2 in TJs |
Maturation of brain barrier junctional complexes during development
Vascular junctions at the developing BBB.
In mice, brain angiogenesis starts at 9.5 d postcoitum, when vascular sprouts invade from the perineural vascular plexus into the developing neuroectoderm, leading to the formation of an undifferentiated vascular network. The molecular mechanisms driving brain angiogenesis are fairly well understood and have been summarized elsewhere (Engelhardt, 2003; Engelhardt and Liebner, 2014). How the barrier characteristics of CNS endothelial cells, including their unique junctional complexes, mature during brain angiogenesis directed by factors from the developing neuroectoderm is less well understood.
The AJ component VE-cadherin is one of the first endothelial cell–specific molecules expressed and required for endothelial survival, blood vessel assembly, and stabilization (Carmeliet et al., 1999; Crosby et al., 2005). Interestingly, expression of VE-cadherin during brain angiogenesis has been observed to be relatively low (Breier et al., 1996), and instead cadherin-10 was abundantly present in AJs of CNS endothelial cells (Williams et al., 2005). In addition to VE-cadherin, N-cadherin is expressed in CNS endothelial cells during brain angiogenesis (Gerhardt et al., 2000). It accumulates at contact zones between endothelial cells and the surrounding pericytes, where it regulates expression of VE-cadherin at the cell membrane (Luo and Radice, 2005) and thus contributes to AJ maturation (Gerhardt et al., 2000; Liebner et al., 2000a).
Stable AJs are considered to be required for the formation of TJs. A first hint to how endothelial AJs might induce TJ formation at the molecular level is provided by the observation that VE-cadherin, through Akt activation and by inhibition of β-catenin translocation to the nucleus, causes phosphorylation of the transcription factor forkhead box factor 1 (FoxO1), allowing it to activate expression of the TJ protein claudin-5 (Taddei et al., 2008). This molecular link between AJs and TJs may also contribute to the formation of BBB TJs and thus barrier maturation. This is supported by studies in zebrafish, where expression of claudin-5 and ZO-1 has been described in CNS microvessels starting at day 3 postfertilization, which was concomitant with the maturation of the BBB as determined by restricted permeability to small and large size tracers (Jeong et al., 2008).
In contrast to our limited knowledge about the molecular mechanisms leading to AJ maturation at the BBB, the mechanism of induction of expression of TJ proteins such as claudin-3, which are unique to CNS microvascular TJs, is much better understood. Wnt ligands produced by immature neuroectodermal cells bind to Frizzled receptors (Fzd) on CNS vascular endothelial cells, inducing canonical Wnt signaling. This leads to translocation of β-catenin into the nucleus of brain endothelial cells resulting in the induction of BBB-specific gene transcription, including the TJ protein claudin-3 (Liebner et al., 2008; Stenman et al., 2008; Daneman et al., 2009). In addition, the death receptors TROY (TNFRSF19) and DR6 (TNFRS21), which are downstream targets of the Wnt/β-catenin signaling pathway and thus involved in brain angiogenesis in mice and zebrafish (Tam et al., 2012), have been linked to regulating expression of ZO-1 in brain endothelial cells. Other factors such as the alternative Frizzled-4 ligand Norrin or GPR 124, an orphan member of the G protein–coupled receptor family, which are coactivators of the canonical Wnt/β-catenin signaling pathway in nonoverlapping CNS regions, also contribute to the induction of BBB-specific junctional molecules such as claudin-5 (Zhou and Nathans, 2014; Zhou et al., 2014).
The Sonic hedgehog (Shh) pathway has also been implicated in brain angiogenesis and BBB maturation (Alvarez et al., 2011). This study demonstrated that, during brain angiogenesis, Shh is secreted by immature astrocytes, and that lack of the Shh signal transducer Smoothened (Smo) in CNS endothelial cells leads to reduced expression of the TJ proteins occludin, claudin-3, and claudin-5, but also of ZO-1 and p120-catenin, which was accompanied by increased leakage of plasma tracers in the developing BBB. By regulating expression of p120-catenin, the Shh pathway might influence maturation of both CNS endothelial AJs and TJs. At the same time, this study provided in vitro evidence that Shh increases the expression of the TJ proteins occludin, claudin-3, claudin-5, and JAM-A in human brain endothelial cells (Alvarez et al., 2011). Another single study that awaits confirmation showed that astrocyte-derived SSeCKS (Src-suppressed C-kinase substrate) contributes to CNS endothelial junctional maturation by regulating the release of astrocyte-derived Ang-1, which, by binding to its receptor Tie-2 on the CNS endothelial cells, increases expression of the TJ proteins occludin, claudin-1, ZO-1, and ZO-2 in human endothelial cells (Lee et al., 2003).
A further step in the maturation of the BBB is the interaction of endothelial cells from nascent vessels with pericytes. Pericytes are recruited to the sprouting CNS vessels during brain angiogenesis and contribute to BBB maturity including the regulation of astrocyte polarity (Armulik et al., 2010; Daneman et al., 2010b). In addition to the homophilic interaction between endothelial and pericyte N-cadherins, platelet-derived growth factor-B (PDGF-B) secreted by endothelial cells and its receptor PDGFR-β on the pericytes regulates pericyte/endothelial interaction (summarized in Siegenthaler et al., 2013). In contrast to astrocyte-derived factors, pericytes seem to have no influence on the maturation of BBB AJs and TJs, as pericyte-deficient mice develop a normal BBB TJ architecture (Armulik et al., 2010), while showing increased BBB leakiness due to, surprisingly, increased vesicular trafficking across CNS endothelial cells. Thus, pericytes seem to inhibit vesicular transport across the BBB endothelium. This has recently been underlined by the finding that Mfsd2a (major facilitator super family domain containing 2a) is specifically induced in CNS endothelial cells by endothelial/pericyte interactions and suppresses transcytotic activity in CNS endothelial cells, thus contributing to BBB maturation (Ben-Zvi et al., 2014).
Interepithelial junctions at the developing BCSFB.
Although there are numerous studies dedicated to the investigation of barrier integrity of the BCSFB during embryonic development, few studies have specifically addressed maturation of cell-to-cell junctions in choroid plexus epithelial cells and whether this correlates with the barrier properties of the BCSFB. Complex TJs have been visualized by freeze-fracture and transmission electron microscopy during very early human and sheep fetal development, at a time when peripheral tracers were still found to diffuse across BCSFB obviously via a transcellular route (Møllgård and Saunders, 1975). These observations indirectly imply that BCSFB AJs connect choroid plexus epithelial cells early during development before barrier maturation. More recent studies that focused on investigating expression of TJ proteins during embryonic development showed expression of occludin and MarvelD3 in the choroid plexus of mouse embryos (Diez-Roux et al., 2011), while in rats early and high expression levels of up to 10 different claudins as well as of occludin and ZO-1, ZO-2, and ZO-3 were observed and thus suggest the early development of mature TJ complexes at the BCSFB (Kratzer et al., 2012; Liddelow et al., 2013). Specifically, in addition to claudin-1, claudin-2, claudin-3, and claudin-11, these studies discovered expression of claudin-9, claudin-19, and claudin-22 in choroid plexus epithelial cells.
Investigation of the maturation of the BCSFB in zebrafish demonstrated that this barrier becomes functional starting at day 2 postfertilization as shown by reduced leakage of different sized tracers (Henson et al., 2014). Interestingly, this study also describes immunostaining for claudin-5, which in rodents has been found to be specifically expressed in endothelial cells, in the choroid plexus epithelium (Henson et al., 2014). Considering its proposed role in BBB maturation, one of the signaling pathways involved in regulating junctional maturation in the BCSFB might be the Shh pathway, which was found to be critical in the continual growth and expansion of the hindbrain choroid plexus throughout development (Huang et al., 2009).
Maintenance of junctional complexes at mature brain barriers
A combination of transcriptome and proteome analyses of the BBB and the BCSFB (Huntley et al., 2014), the development of antibodies to detect the subcellular localization of the individual molecular components of junctions, as well as high-resolution imaging have improved our knowledge of the junctional architecture of BBB endothelial cells and the choroid plexus epithelial cells (Fig. 1 and Table 1). Choroid plexus epithelial cells seem to maintain their unique barrier characteristics, including proper junctional complexes, when removed from the CNS and put in culture (Gath et al., 1997; Strazielle and Ghersi-Egea, 1999; Schroten et al., 2012). However, endothelial cells forming the BBB need pericytes and astrocytes as well as the extracellular matrix surrounding the BBB endothelium to maintain their unique barrier characteristics (Wolburg et al., 1994; Liebner et al., 2000b). Adhesive contacts between pericytes and endothelial cells are provided by N-cadherin, which in this case is not restricted to AJs (Gerhardt et al., 2000). Studies in viable pericyte-deficient mouse mutants have shown that pericytes do not directly affect BBB junctional architecture but rather regulate barrier characteristics of CNS endothelial cells by inhibiting transcellular vesicular trafficking and inducing astrocyte polarity (Armulik et al., 2010; Daneman et al., 2010b). However, astrocyte-derived factors seem to maintain junctional integrity of the BBB endothelium. Indeed, toxin-induced deletion of GFAP+ astrocytes in certain brain regions in mice and rats was found to induce a reversible focal dysfunction of the BBB (Bush et al., 1999; Willis et al., 2013) that is accompanied by the loss of junctional localization of claudin-5 and altered phosphorylation of occludin in brain endothelium (Willis et al., 2013). Interestingly, although junctional localization of VE-cadherin and β-catenin was found unchanged in the latter study, expression of VE-cadherin was up-regulated, underlining the tight connection between regulating BBB AJs and TJs. The molecular cues provided by astrocytes to maintain junctional integrity of the BBB are not well understood.
In addition to its role in BBB development, recent studies support a role of the canonical Wnt/β-catenin signaling pathway in BBB maintenance and thus probably in sustaining junctional integrity. Using novel transgenic reporter lines to visualize Wnt/β-catenin signaling in zebrafish allowed for the demonstration of Wnt/β-catenin signaling activity in CNS vessels of adult zebrafish (Moro et al., 2012). Furthermore, elegant gain- and loss-of-function mouse mutants for Norrin/Fz4 signaling show a cell-autonomous gain or loss of BBB function accompanied by the presence or absence of claudin-5 expression, respectively, in the cerebellum of these mice, which supports the continuous requirement of Frizzled signaling in preserving the mature BBB junctional structure (Wang et al., 2012; Zhou et al., 2014).
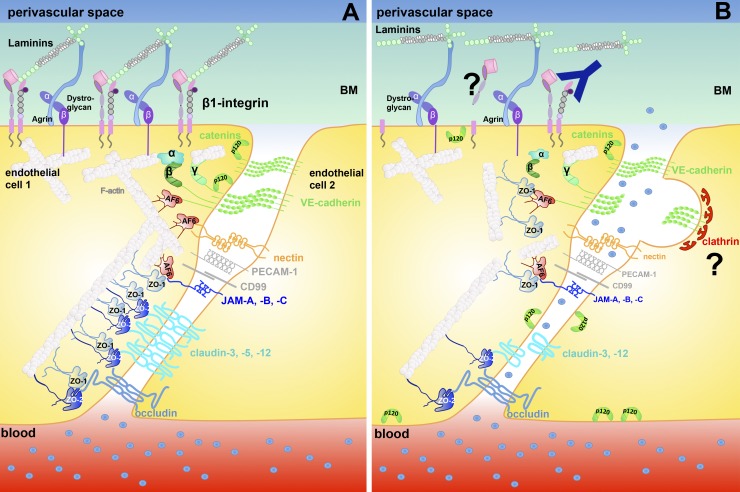
Finally, components of the basement membranes have recently been shown to contribute to junctional integrity of brain endothelial cells (Fig. 2). The heparan sulfate proteoglycan agrin contributes to barrier properties of brain endothelial cells by stabilizing junctional localization of molecules forming AJs rather than TJs (Steiner et al., 2014). Using mouse models in which β1-integrins are specifically inactivated in endothelial cells, Yamamoto et al. (2015) showed that the absence of this integrin reduced the interaction between VE-cadherin and p120-catenin, causing an increase in the internalization of VE-cadherin. Thus, β1-integrin–mediated binding of CNS endothelial cells to the extracellular matrix is critical for stabilizing junctional localization of VE-cadherin and vascular integrity (Yamamoto et al., 2015; Fig. 2). The molecular processes identified to be downstream of endothelial β1-integrin involved myosin light chain phosphorylation by the GTPase Rap1 and the kinase MRCK in addition to Rho/Rho-kinase signaling. In light of the previous observations that junctional localization of VE-cadherin regulates expression and junctional localization of claudin-5 (Taddei et al., 2008), it is interesting to note that endothelial cell matrix interactions via endothelial β1-integrins were also found to increase expression and junctional localization of claudin-5 to BBB TJs (Osada et al., 2011; Fig. 2). In addition, laminin α2, which is part of the astrocyte-derived parenchymal basement membrane and thus critical for astrocyte polarity, influences BBB TJ morphology and barrier function, as recently observed in laminin α2–deficient mice (Menezes et al., 2014).
Figure 2.
Loss of endothelial–matrix interactions in neuroinflammation affects junctional integrity of the BBB. (A) Under physiological conditions, BBB endothelial cells are anchored via β1-integrins to the ECM proteins laminin, collagen, and perlecan in the endothelial basement membrane. β1-integrin–mediated adhesive contacts contribute by not-yet-defined downstream signaling events to junctional stabilization. (B) After ischemic stroke, edema formation and BBB breakdown is associated with loss of vascular β1-integrin, probably by shedding or degradation (indicated by a question mark). Genetic or functional inactivation of β1-integrin at the BBB induces BBB leakiness by reducing the association of p120 and β-catenin with VE-cadherin. This leads to increased internalization of VE-cadherin via clathrin-coated pits and ultimately loss of junctional claudin-5. BM, basement membrane.
In contrast to the BBB TJs, not much is known about factors regulating junctional integrity at the BCSFB. In general, junctional complexes of epithelial cells including those of the BCSFB are intrinsically more stable, as indicated by the formation of mature AJs and TJs in cultured choroid plexus epithelial cells (Gath et al., 1997; Strazielle and Ghersi-Egea, 1999). However, besides providing the BCSFB, the choroid plexus has been suggested to regulate access of hormones to the CSF. In this context, a study performed in sheep has demonstrated photoperiod-dependent regulation of the expression levels of occludin, ZO-1, ZO-2, afadin, and E-cadherin in the choroid plexus in correlation with the regulation of the passage of hormones into the brains of the sheep (Lagaraine et al., 2011). This study therefore provides evidence for a physiological and dynamic regulation of junctional complexes at the BCSFB.
Neuroinflammation: junctional complexes at dysfunctional brain barriers
Neuroinflammation is generally accompanied by impaired BBB and BCSFB function, which includes alterations in their junctional complexes. For example, in the vascular dysplasia referred to as cerebral cavernous malformation (CCM), loss-of-function mutations in CCM-1, CCM-2, or CCM-3, which localize generally to AJs, primarily affect brain vessels. CCMs form a tripartite complex that associates with β-catenin and afadin at AJs. By regulating the small GTPase Rap-1 (CCM-1) and the Rho GTPase pathway (CCM-2), they contribute to junction stabilization and endothelial cell polarity (Dejana and Orsenigo, 2013). Mutations in CCM proteins lead to endothelial–mesenchymal transition, which is characterized by loss of VE-cadherin, up-regulated expression of N-cadherin, and the accompanying loss of endothelial cell polarity (Maddaluno et al., 2013). Thus, even though the CCM genes are mutated in endothelial cells throughout the body, barrier function of brain endothelial cells seems to more critically rely on the adhesive properties of VE-cadherin to stabilize cell polarity (Dejana and Orsenigo, 2013).
Disruption of BBB and BCSFB function also contributes to neurological disorders such as stroke or multiple sclerosis (MS) and its animal model experimental autoimmune encephalomyelitis (EAE). MS is an inflammatory demyelinating disease of the CNS, in which immune cells cross the BBB and the BCSFB causing CNS inflammation, edema formation, and brain barrier dysfunction. In fact, early lesion formation in MS is associated with focal BBB dysfunction as visualized by gadolinium-enhanced magnetic resonance imaging (Kermode et al., 1990), which is used as a diagnostic criterion for this disease. A series of immunofluorescence studies of postmortem brain samples from MS patients have convincingly demonstrated that an abnormal distribution of the TJ proteins occludin, JAM-A, and ZO-1 but not of the AJ protein β-catenin correlates with the premortem pattern of perivascular serum protein leakage in active MS lesions (Plumb et al., 2002; Kirk et al., 2003; Padden et al., 2007). These data therefore strongly support a contribution of TJ disruption to BBB dysfunction in MS. Based on immunostainings for ZO-1, BBB junction alterations as observed in MS are quite reliably modeled in brain tissues of mice after induction of EAE (Bennett et al., 2010). Using the EAE model, a critical function for BBB TJs in maintaining barrier functions of the BBB in neuroinflammation has been further supported by the observation that inducible endothelial cell–specific ectopic expression of claudin-1 ameliorated clinical symptoms of EAE in mice by specifically blocking BBB leakiness without affecting immune cell migration into the CNS (Pfeiffer et al., 2011). Immune cell infiltration into the CNS during EAE might, however, add to BBB TJ pathology, as further studies in the EAE model showed the selective loss of claudin-3 or claudin-5 immunostaining in brain and spinal cord microvessels surrounded by inflammatory infiltrates (Wolburg et al., 2003; Paul et al., 2013). Loss of immunostaining for an individual TJ protein is, however, not predictive for a dysfunctional BBB, as recent studies on EAE pathogenesis in claudin-3–deficient mice failed to define a role for claudin-3 in maintaining BBB integrity or regulating leukocyte infiltration into the CNS during EAE (Kooij et al., 2014). Also, considering the known function of claudin-5 in regulating the diffusion of small molecules (<800 D) across the BBB, specific loss of claudin-5 immunostaining in BBB TJs can hardly be the equivalent of BBB leakiness for large serum proteins such as fibrinogen into the CNS parenchyma. Rather, these observations suggest that the loss of additional junctional components is mandatory for an impairment of BBB function as observed in MS and EAE. Indeed, a recent study has found severe loss of both the TJ protein claudin-5 and the AJ protein VE-cadherin in microvessels in preactive and active inflammatory lesions in postmortem brain tissues of MS patients, which supports the notion that both BBB AJs and TJs are impaired in MS and that alterations in the molecular composition of TJs are early events in BBB dysfunction during MS (Alvarez et al., 2015). A possible molecular mechanism behind these junctional changes might be the loss of β1-integrin–mediated anchorage of brain endothelial cells to the basement membrane, as observed in stroke (Osada et al., 2011), leading to the internalization of VE-cadherin and claudin-5 as outlined earlier (Fig. 2). In addition, loss of claudin-5 and occludin at the BBB during EAE was found to correlate with increased expression of caveolin-1 and increased leakage of FITC-dextran across the BBB, which suggests that caveolae-mediated endocytosis of claudin-5 and occludin contributes to BBB TJ breakdown in EAE (Errede et al., 2012). Another molecule that has been shown to induce disruption of BBB TJs during neuroinflammation by down-regulating claudin-5 and occludin is VEGF, expression of which is significantly up-regulated in astrocytes during EAE and MS (Argaw et al., 2009, 2012). In peripheral vascular beds, VEGF induces tyrosine phosphorylation of VE-cadherin, leading to disruption of p120-catenin and β-catenin binding, subsequent internalization and degradation of VE-cadherin, and thus increased vascular permeability (summarized in Goddard and Iruela-Arispe, 2013). In contrast to peripheral vascular beds, the BBB endothelium responds to VEGF-A in a highly polarized fashion—e.g., only abluminal from the CNS, but not luminal VEGF-A, induces increased BBB permeability by activation of p38 MAPK and loss of junctional localization of claudin-5 (Hudson et al., 2014). Thus, in neuroinflammation, astrocyte-derived factors seem to be instrumental in regulating BBB junctional integrity. This is further supported by the observations that BBB dysfunction is associated with loss of astrocyte polarity. This is visualized by the decrease of the polarized localization of the water channel aquaporin 4 (AQP4) from astrocyte end-feet in EAE and other neurological disorders such as stroke and glioblastoma multiforme (Wolburg-Buchholz et al., 2009). Loss of astrocyte polarity suggests loss of polarized secretion of other astrocyte-derived factors such as Shh, Wnts, and Norrin that support appropriate expression and localization of BBB junctional molecules. Indeed, up-regulated expression of Shh in hypertrophic astrocytes in active demyelinating MS lesions was accompanied by increased expression of the Shh receptor Patched-1 and nuclear translocation of the Shh pathway transcription factor Gli-1 in BBB endothelial cells (Alvarez et al., 2011). These observations suggest that neuroinflammation activates the Shh pathway at the level of the BBB, thereby possibly promoting BBB repair.
The concept that neuroinflammation might reactivate pathways operative during the maturation of BBB junctional complexes in embryonic development is further supported by the recent observations that retinoic acid (RA) contributes to BBB maturation. Radial glial cells in fetal human brain tissue were found to express high levels of the RA-producing enzyme retinaldehyde dehydrogenase 2 (RALDH2) and to tightly associate with the developing brain vasculature expressing the RA receptor β during embryogenesis (Mizee et al., 2013). In vitro studies on brain endothelial cells confirmed the ability of RA to increase barrier characteristics, including enhancing expression of AJ and TJ proteins. Interestingly, expression of RALDH2 is found to be significantly up-regulated in reactive astrocytes in active and chronic MS lesions (Mizee et al., 2014). These observations further support the notion that neuroinflammation induces increased expression of Shh and enhanced release of RA from reactive astrocytes, with the aim of providing a protective response allowing restoration of BBB junctional integrity.
Finally, the proinflammatory cytokine interleukin-1β (IL-1β) is up-regulated in the CNS during neuroinflammation and critically contributes to brain barrier dysfunction. It was shown to mediate transcriptional repression of claudin-5 by inducing nuclear translocation of β-catenin and FoxO1 in brain endothelial cells (Beard et al., 2014). These observations suggest that neuroinflammation reverts some processes described for junctional maturation, e.g., induction of claudin-5 expression (Taddei et al., 2008) during development. However, as IL-1β–induced nuclear translocation of β-catenin was associated with a decrease in claudin-5 and occludin, but Wnt3a-mediated nuclear translocation of β-catenin rather increased expression of claudin-1 in an in vitro BBB model, β-catenin–mediated transcriptional activity in brain endothelial cells seems to be dependent on the respective stimulus (Beard et al., 2014).
Neuroinflammation induces expression of additional inflammatory mediators such as the chemokine CCL2, especially in astrocytes. At least in vitro, CCL2 has been shown to induce Src-dependent tyrosine phosphorylation of VE-cadherin and β-catenin, causing their transient dissociation from AJs (Roberts et al., 2012). Concurrently, β-catenin is recruited to PECAM-1, thereby remaining sequestered at the cell membrane. Upon CCL2-dependent tyrosine phosphorylation of PECAM-1, SHP-2 is engaged in inducing the release of β-catenin and its reassociation with the AJ. This study assigns an important role for PECAM-1 in transient sequestration of β-catenin: allowing for rapid reorganization of AJ complexes in brain endothelium. In fact, the observation that PECAM-1−/− C57BL/6 mice develop aggravated EAE that is accompanied by increased and prolonged BBB permeability (Graesser et al., 2002) highlights this function of endothelial PECAM-1 in restoring BBB AJs and further underlines the fundamental role of mature AJs for properly formed junctional complexes at the BBB.
Less still is known about possible changes at the level of BCSFB junctions during neuroinflammation. Although several studies have shown dramatic changes of the choroid plexus during MS and EAE (Engelhardt et al., 2001; Murugesan et al., 2012), only a few specifically addressed alterations in the junctional architecture of the choroid plexus epithelial cells (Wolburg et al., 2001; Kooij et al., 2014) and reported interrupted immunoreactivities for occludin, claudin-1, claudin-2, and claudin-11 as well as for ZO-1 and complete loss of claudin-3 immunostaining at the level of the BCSFB during EAE and MS. With the presently limited knowledge of the specific function of each of these junctional molecules, the implications of these findings for BCSFB function during neuroinflammation are of course limited.
Concluding remarks
Despite great progress in our understanding of the molecular composition of brain barrier junctions, research in the field has mostly focused on studying brain barrier TJs. There is now strong experimental evidence for crosstalk between AJs and TJs influencing their stability and dynamics at a structural and transcriptional level. Beyond their structural functions, AJs and TJs have emerged as central regulators of intracellular signaling pathways that regulate cytoskeletal dynamics. Thus, their cooperation is central to the control of morphogenesis and homeostasis. Perturbation of these junctional complexes at the level of the brain barriers thus disrupts CNS homeostasis and is associated with neurological disorders. There is evidence that protecting the brain barriers by specifically improving junctional integrity is beneficial in animal models for MS or stroke (Pfeiffer et al., 2011; Wacker et al., 2012). Considering the recent findings that circulating sphingosine-1-phosphate (S1P) stabilizes vascular junctions via downstream signaling through its G-protein–coupled receptor S1P1, which influences the cortical actin cytoskeleton (summarized in Goddard and Iruela-Arispe, 2013), it is tempting to speculate that therapeutic efficacy of the S1P1 agonist fingolimod in MS patients also relies on stabilizing the BBB. Therefore, future research needs to go beyond studying the role of individual TJ proteins at the brain barriers. A more integrated cell biological view on the dynamic collaboration of AJs and TJs and how they regulate junctional dynamics and downstream signaling events is required to improve our understanding of how these junctional complexes establish cell polarity and maintain barrier properties of the BBB and the BCSFB.
Acknowledgments
We thank Dr. Urban Deutsch for his critical comments for this review. We apologize that space restrictions did not allow for citation of all original work of our colleagues.
The B. Engelhardt laboratory has been supported by the Swiss National Science Foundation, the Swiss Multiple Sclerosis Society and the European Union’s Seventh Framework Program (FP7/2007-2013) under grant agreements no. 241861 (JUSTBRAIN) and no. 607962 (nEUROinflammation). S. Tietz is funded by a fellowship of the German Research Foundation.
The authors declare no competing financial interests.
Footnotes
Abbreviations used in this paper:
- AJ
- adherens junction
- BBB
- blood–brain barrier
- BCSFB
- blood–cerebrospinal fluid barrier
- CCM
- cerebral cavernous malformation
- CNS
- central nervous system
- E-cadherin
- epithelial cadherin
- EAE
- experimental autoimmune encephalomyelitis
- Ig
- immunoglobulin
- MS
- multiple sclerosis
- RA
- retinoic acid
- Shh
- Sonic hedgehog
- TJ
- tight junction
- VE-cadherin
- vascular endothelial cadherin
References
- Alvarez J.I., Dodelet-Devillers A., Kebir H., Ifergan I., Fabre P.J., Terouz S., Sabbagh M., Wosik K., Bourbonnière L., Bernard M., et al. 2011. The Hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science. 334:1727–1731. 10.1126/science.1206936 [DOI] [PubMed] [Google Scholar]
- Alvarez J.I., Saint-Laurent O., Godschalk A., Terouz S., Briels C., Larouche S., Bourbonnière L., Larochelle C., and Prat A.. 2015. Focal disturbances in the blood-brain barrier are associated with formation of neuroinflammatory lesions. Neurobiol. Dis. 74:14–24. 10.1016/j.nbd.2014.09.016 [DOI] [PubMed] [Google Scholar]
- Argaw A.T., Gurfein B.T., Zhang Y., Zameer A., and John G.R.. 2009. VEGF-mediated disruption of endothelial CLN-5 promotes blood-brain barrier breakdown. Proc. Natl. Acad. Sci. USA. 106:1977–1982. 10.1073/pnas.0808698106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Argaw A.T., Asp L., Zhang J., Navrazhina K., Pham T., Mariani J.N., Mahase S., Dutta D.J., Seto J., Kramer E.G., et al. 2012. Astrocyte-derived VEGF-A drives blood-brain barrier disruption in CNS inflammatory disease. J. Clin. Invest. 122:2454–2468. 10.1172/JCI60842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armulik A., Genové G., Mäe M., Nisancioglu M.H., Wallgard E., Niaudet C., He L., Norlin J., Lindblom P., Strittmatter K., et al. 2010. Pericytes regulate the blood-brain barrier. Nature. 468:557–561. 10.1038/nature09522 [DOI] [PubMed] [Google Scholar]
- Arrate M.P., Rodriguez J.M., Tran T.M., Brock T.A., and Cunningham S.A.. 2001. Cloning of human junctional adhesion molecule 3 (JAM3) and its identification as the JAM2 counter-receptor. J. Biol. Chem. 276:45826–45832. 10.1074/jbc.M105972200 [DOI] [PubMed] [Google Scholar]
- Aurrand-Lions M., Duncan L., Ballestrem C., and Imhof B.A.. 2001a. JAM-2, a novel immunoglobulin superfamily molecule, expressed by endothelial and lymphatic cells. J. Biol. Chem. 276:2733–2741. 10.1074/jbc.M005458200 [DOI] [PubMed] [Google Scholar]
- Aurrand-Lions M., Johnson-Leger C., Wong C., Du Pasquier L., and Imhof B.A.. 2001b. Heterogeneity of endothelial junctions is reflected by differential expression and specific subcellular localization of the three JAM family members. Blood. 98:3699–3707. 10.1182/blood.V98.13.3699 [DOI] [PubMed] [Google Scholar]
- Bäumer S., Keller L., Holtmann A., Funke R., August B., Gamp A., Wolburg H., Wolburg-Buchholz K., Deutsch U., and Vestweber D.. 2006. Vascular endothelial cell-specific phosphotyrosine phosphatase (VE-PTP) activity is required for blood vessel development. Blood. 107:4754–4762. 10.1182/blood-2006-01-0141 [DOI] [PubMed] [Google Scholar]
- Beard R.S. Jr, Haines R.J., Wu K.Y., Reynolds J.J., Davis S.M., Elliott J.E., Malinin N.L., Chatterjee V., Cha B.J., Wu M.H., and Yuan S.Y.. 2014. Non-muscle Mlck is required for β-catenin- and FoxO1-dependent downregulation of Cldn5 in IL-1β-mediated barrier dysfunction in brain endothelial cells. J. Cell Sci. 127:1840–1853. 10.1242/jcs.144550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett J., Basivireddy J., Kollar A., Biron K.E., Reickmann P., Jefferies W.A., and McQuaid S.. 2010. Blood-brain barrier disruption and enhanced vascular permeability in the multiple sclerosis model EAE. J. Neuroimmunol. 229:180–191. 10.1016/j.jneuroim.2010.08.011 [DOI] [PubMed] [Google Scholar]
- Ben-Zvi A., Lacoste B., Kur E., Andreone B.J., Mayshar Y., Yan H., and Gu C.. 2014. Mfsd2a is critical for the formation and function of the blood-brain barrier. Nature. 509:507–511. 10.1038/nature13324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bixel G., Kloep S., Butz S., Petri B., Engelhardt B., and Vestweber D.. 2004. Mouse CD99 participates in T-cell recruitment into inflamed skin. Blood. 104:3205–3213. 10.1182/blood-2004-03-1184 [DOI] [PubMed] [Google Scholar]
- Breier G., Breviario F., Caveda L., Berthier R., Schnürch H., Gotsch U., Vestweber D., Risau W., and Dejana E.. 1996. Molecular cloning and expression of murine vascular endothelial-cadherin in early stage development of cardiovascular system. Blood. 87:630–641. [PubMed] [Google Scholar]
- Buckley C.D., Tan J., Anderson K.L., Hanein D., Volkmann N., Weis W.I., Nelson W.J., and Dunn A.R.. 2014. The minimal cadherin-catenin complex binds to actin filaments under force. Science. 346:1254211 10.1126/science.1254211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bush T.G., Puvanachandra N., Horner C.H., Polito A., Ostenfeld T., Svendsen C.N., Mucke L., Johnson M.H., and Sofroniew M.V.. 1999. Leukocyte infiltration, neuronal degeneration, and neurite outgrowth after ablation of scar-forming, reactive astrocytes in adult transgenic mice. Neuron. 23:297–308. 10.1016/S0896-6273(00)80781-3 [DOI] [PubMed] [Google Scholar]
- Carmeliet P., Lampugnani M.G., Moons L., Breviario F., Compernolle V., Bono F., Balconi G., Spagnuolo R., Oosthuyse B., Dewerchin M., et al. 1999. Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell. 98:147–157. 10.1016/S0092-8674(00)81010-7 [DOI] [PubMed] [Google Scholar]
- Citi S., Guerrera D., Spadaro D., and Shah J.. 2014. Epithelial junctions and Rho family GTPases: the zonular signalosome. Small GTPases. 5:1–15. 10.4161/21541248.2014.973760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crosby C.V., Fleming P.A., Argraves W.S., Corada M., Zanetta L., Dejana E., and Drake C.J.. 2005. VE-cadherin is not required for the formation of nascent blood vessels but acts to prevent their disassembly. Blood. 105:2771–2776. 10.1182/blood-2004-06-2244 [DOI] [PubMed] [Google Scholar]
- Daneman R., Agalliu D., Zhou L., Kuhnert F., Kuo C.J., and Barres B.A.. 2009. Wnt/β-catenin signaling is required for CNS, but not non-CNS, angiogenesis. Proc. Natl. Acad. Sci. USA. 106:641–646. 10.1073/pnas.0805165106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daneman R., Zhou L., Agalliu D., Cahoy J.D., Kaushal A., and Barres B.A.. 2010a. The mouse blood-brain barrier transcriptome: a new resource for understanding the development and function of brain endothelial cells. PLoS ONE. 5:e13741 10.1371/journal.pone.0013741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daneman R., Zhou L., Kebede A.A., and Barres B.A.. 2010b. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature. 468:562–566. 10.1038/nature09513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dejana E., and Orsenigo F.. 2013. Endothelial adherens junctions at a glance. J. Cell Sci. 126:2545–2549. 10.1242/jcs.124529 [DOI] [PubMed] [Google Scholar]
- Dejana E., and Vestweber D.. 2013. The role of VE-cadherin in vascular morphogenesis and permeability control. Prog. Mol. Biol. Transl. Sci. 116:119–144. 10.1016/B978-0-12-394311-8.00006-6 [DOI] [PubMed] [Google Scholar]
- Dejana E., Tournier-Lasserve E., and Weinstein B.M.. 2009. The control of vascular integrity by endothelial cell junctions: molecular basis and pathological implications. Dev. Cell. 16:209–221. 10.1016/j.devcel.2009.01.004 [DOI] [PubMed] [Google Scholar]
- Diez-Roux G., Banfi S., Sultan M., Geffers L., Anand S., Rozado D., Magen A., Canidio E., Pagani M., Peluso I., et al. 2011. A high-resolution anatomical atlas of the transcriptome in the mouse embryo. PLoS Biol. 9:e1000582 10.1371/journal.pbio.1000582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebnet K., Suzuki A., Horikoshi Y., Hirose T., Meyer Zu Brickwedde M.K., Ohno S., and Vestweber D.. 2001. The cell polarity protein ASIP/PAR-3 directly associates with junctional adhesion molecule (JAM). EMBO J. 20:3738–3748. 10.1093/emboj/20.14.3738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelhardt B. 2003. Development of the blood-brain barrier. Cell Tissue Res. 314:119–129. 10.1007/s00441-003-0751-z [DOI] [PubMed] [Google Scholar]
- Engelhardt B., and Liebner S.. 2014. Novel insights into the development and maintenance of the blood-brain barrier. Cell Tissue Res. 355:687–699. 10.1007/s00441-014-1811-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelhardt B., Wolburg-Buchholz K., and Wolburg H.. 2001. Involvement of the choroid plexus in central nervous system inflammation. Microsc. Res. Tech. 52:112–129. [DOI] [PubMed] [Google Scholar]
- Errede M., Girolamo F., Ferrara G., Strippoli M., Morando S., Boldrin V., Rizzi M., Uccelli A., Perris R., Bendotti C., et al. 2012. Blood-brain barrier alterations in the cerebral cortex in experimental autoimmune encephalomyelitis. J. Neuropathol. Exp. Neurol. 71:840–854. 10.1097/NEN.0b013e31826ac110 [DOI] [PubMed] [Google Scholar]
- Fanning A.S., Jameson B.J., Jesaitis L.A., and Anderson J.M.. 1998. The tight junction protein ZO-1 establishes a link between the transmembrane protein occludin and the actin cytoskeleton. J. Biol. Chem. 273:29745–29753. 10.1074/jbc.273.45.29745 [DOI] [PubMed] [Google Scholar]
- Figarella-Branger D., Lepidi H., Poncet C., Gambarelli D., Bianco N., Rougon G., and Pellissier J.F.. 1995. Differential expression of cell adhesion molecules (CAM), neural CAM and epithelial cadherin in ependymomas and choroid plexus tumors. Acta Neuropathol. 89:248–257. 10.1007/BF00309340 [DOI] [PubMed] [Google Scholar]
- Furuse M., Hirase T., Itoh M., Nagafuchi A., Yonemura S., Tsukita S., and Tsukita S.. 1993. Occludin: a novel integral membrane protein localizing at tight junctions. J. Cell Biol. 123:1777–1788. 10.1083/jcb.123.6.1777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuse M., Fujita K., Hiiragi T., Fujimoto K., and Tsukita S.. 1998. Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J. Cell Biol. 141:1539–1550. 10.1083/jcb.141.7.1539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuse M., Furuse K., Sasaki H., and Tsukita S.. 2001. Conversion of zonulae occludentes from tight to leaky strand type by introducing claudin-2 into Madin-Darby canine kidney I cells. J. Cell Biol. 153:263–272. 10.1083/jcb.153.2.263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuse M., Hata M., Furuse K., Yoshida Y., Haratake A., Sugitani Y., Noda T., Kubo A., and Tsukita S.. 2002. Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1-deficient mice. J. Cell Biol. 156:1099–1111. 10.1083/jcb.200110122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuse M., Izumi Y., Oda Y., Higashi T., and Iwamoto N.. 2014. Molecular organization of tricellular tight junctions. Tissue Barriers. 2:e28960 10.4161/tisb.28960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrido-Urbani S., Bradfield P.F., and Imhof B.A.. 2014. Tight junction dynamics: the role of junctional adhesion molecules (JAMs). Cell Tissue Res. 355:701–715. 10.1007/s00441-014-1820-1 [DOI] [PubMed] [Google Scholar]
- Gath U., Hakvoort A., Wegener J., Decker S., and Galla H.J.. 1997. Porcine choroid plexus cells in culture: expression of polarized phenotype, maintenance of barrier properties and apical secretion of CSF-components. Eur. J. Cell Biol. 74:68–78. [PubMed] [Google Scholar]
- Gerhardt H., Liebner S., Redies C., and Wolburg H.. 1999. N-cadherin expression in endothelial cells during early angiogenesis in the eye and brain of the chicken: relation to blood-retina and blood-brain barrier development. Eur. J. Neurosci. 11:1191–1201. 10.1046/j.1460-9568.1999.00526.x [DOI] [PubMed] [Google Scholar]
- Gerhardt H., Wolburg H., and Redies C.. 2000. N-cadherin mediates pericytic-endothelial interaction during brain angiogenesis in the chicken. Dev. Dyn. 218:472–479. [DOI] [PubMed] [Google Scholar]
- Giannotta M., Trani M., and Dejana E.. 2013. VE-cadherin and endothelial adherens junctions: active guardians of vascular integrity. Dev. Cell. 26:441–454. 10.1016/j.devcel.2013.08.020 [DOI] [PubMed] [Google Scholar]
- Goddard L.M., and Iruela-Arispe M.L.. 2013. Cellular and molecular regulation of vascular permeability. Thromb. Haemost. 109:407–415. 10.1160/TH12-09-0678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gow A., Southwood C.M., Li J.S., Pariali M., Riordan G.P., Brodie S.E., Danias J., Bronstein J.M., Kachar B., and Lazzarini R.A.. 1999. CNS myelin and sertoli cell tight junction strands are absent in Osp/claudin-11 null mice. Cell. 99:649–659. 10.1016/S0092-8674(00)81553-6 [DOI] [PubMed] [Google Scholar]
- Gow A., Davies C., Southwood C.M., Frolenkov G., Chrustowski M., Ng L., Yamauchi D., Marcus D.C., and Kachar B.. 2004. Deafness in Claudin 11-null mice reveals the critical contribution of basal cell tight junctions to stria vascularis function. J. Neurosci. 24:7051–7062. 10.1523/JNEUROSCI.1640-04.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graesser D., Solowiej A., Bruckner M., Osterweil E., Juedes A., Davis S., Ruddle N.H., Engelhardt B., and Madri J.A.. 2002. Altered vascular permeability and early onset of experimental autoimmune encephalomyelitis in PECAM-1-deficient mice. J. Clin. Invest. 109:383–392. 10.1172/JCI0213595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris T.J. 2012. Adherens junction assembly and function in the Drosophila embryo. Int. Rev. Cell Mol. Biol. 293:45–83. 10.1016/B978-0-12-394304-0.00007-5 [DOI] [PubMed] [Google Scholar]
- Haseloff R.F., Dithmer S., Winkler L., Wolburg H., and Blasig I.E.. 2015. Transmembrane proteins of the tight junctions at the blood-brain barrier: Structural and functional aspects. Semin. Cell Dev. Biol. 38:16–25. 10.1016/j.semcdb.2014.11.004 [DOI] [PubMed] [Google Scholar]
- Henson H.E., Parupalli C., Ju B., and Taylor M.R.. 2014. Functional and genetic analysis of choroid plexus development in zebrafish. Front Neurosci. 8:364 10.3389/fnins.2014.00364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirase T., Staddon J.M., Saitou M., Ando-Akatsuka Y., Itoh M., Furuse M., Fujimoto K., Tsukita S., and Rubin L.L.. 1997. Occludin as a possible determinant of tight junction permeability in endothelial cells. J. Cell Sci. 110:1603–1613. [DOI] [PubMed] [Google Scholar]
- Hoshi Y., Uchida Y., Tachikawa M., Inoue T., Ohtsuki S., and Terasaki T.. 2013. Quantitative atlas of blood-brain barrier transporters, receptors, and tight junction proteins in rats and common marmoset. J. Pharm. Sci. 102:3343–3355. 10.1002/jps.23575 [DOI] [PubMed] [Google Scholar]
- Huang X., Ketova T., Fleming J.T., Wang H., Dey S.K., Litingtung Y., and Chiang C.. 2009. Sonic hedgehog signaling regulates a novel epithelial progenitor domain of the hindbrain choroid plexus. Development. 136:2535–2543. 10.1242/dev.033795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson N., Powner M.B., Sarker M.H., Burgoyne T., Campbell M., Ockrim Z.K., Martinelli R., Futter C.E., Grant M.B., Fraser P.A., et al. 2014. Differential apicobasal VEGF signaling at vascular blood-neural barriers. Dev. Cell. 30:541–552. 10.1016/j.devcel.2014.06.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huntley M.A., Bien-Ly N., Daneman R., and Watts R.J.. 2014. Dissecting gene expression at the blood-brain barrier. Front Neurosci. 8:355 10.3389/fnins.2014.00355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda W., Nakanishi H., Miyoshi J., Mandai K., Ishizaki H., Tanaka M., Togawa A., Takahashi K., Nishioka H., Yoshida H., et al. 1999. Afadin: A key molecule essential for structural organization of cell-cell junctions of polarized epithelia during embryogenesis. J. Cell Biol. 146:1117–1132. 10.1083/jcb.146.5.1117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikenouchi J., Sasaki H., Tsukita S., Furuse M., and Tsukita S.. 2008. Loss of occludin affects tricellular localization of tricellulin. Mol. Biol. Cell. 19:4687–4693. 10.1091/mbc.E08-05-0530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Indra I., Hong S., Troyanovsky R., Kormos B., and Troyanovsky S.. 2013. The adherens junction: a mosaic of cadherin and nectin clusters bundled by actin filaments. J. Invest. Dermatol. 133:2546–2554. 10.1038/jid.2013.200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh M., Furuse M., Morita K., Kubota K., Saitou M., and Tsukita S.. 1999. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J. Cell Biol. 147:1351–1363. 10.1083/jcb.147.6.1351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwamoto N., Higashi T., and Furuse M.. 2014. Localization of angulin-1/LSR and tricellulin at tricellular contacts of brain and retinal endothelial cells in vivo. Cell Struct. Funct. 39:1–8. 10.1247/csf.13015 [DOI] [PubMed] [Google Scholar]
- Jeong J.Y., Kwon H.B., Ahn J.C., Kang D., Kwon S.H., Park J.A., and Kim K.W.. 2008. Functional and developmental analysis of the blood-brain barrier in zebrafish. Brain Res. Bull. 75:619–628. 10.1016/j.brainresbull.2007.10.043 [DOI] [PubMed] [Google Scholar]
- Kermode A.G., Thompson A.J., Tofts P., MacManus D.G., Kendall B.E., Kingsley D.P., Moseley I.F., Rudge P., and McDonald W.I.. 1990. Breakdown of the blood-brain barrier precedes symptoms and other MRI signs of new lesions in multiple sclerosis. Pathogenetic and clinical implications. Brain. 113:1477–1489. 10.1093/brain/113.5.1477 [DOI] [PubMed] [Google Scholar]
- Kirk J., Plumb J., Mirakhur M., and McQuaid S.. 2003. Tight junctional abnormality in multiple sclerosis white matter affects all calibres of vessel and is associated with blood-brain barrier leakage and active demyelination. J. Pathol. 201:319–327. 10.1002/path.1434 [DOI] [PubMed] [Google Scholar]
- Kniesel U., Reichenbach A., Risau W., and Wolburg H.. 1994. Quantification of tight junction complexity by means of fractal analysis. Tissue Cell. 26:901–912. 10.1016/0040-8166(94)90039-6 [DOI] [PubMed] [Google Scholar]
- Kooij G., Kopplin K., Blasig R., Stuiver M., Koning N., Goverse G., van der Pol S.M., van Het Hof B., Gollasch M., Drexhage J.A., et al. 2014. Disturbed function of the blood-cerebrospinal fluid barrier aggravates neuro-inflammation. Acta Neuropathol. 128:267–277. 10.1007/s00401-013-1227-1 [DOI] [PubMed] [Google Scholar]
- Kratzer I., Vasiljevic A., Rey C., Fevre-Montange M., Saunders N., Strazielle N., and Ghersi-Egea J.F.. 2012. Complexity and developmental changes in the expression pattern of claudins at the blood-CSF barrier. Histochem. Cell Biol. 138:861–879. 10.1007/s00418-012-1001-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Küppers V., Vockel M., Nottebaum A.F., and Vestweber D.. 2014. Phosphatases and kinases as regulators of the endothelial barrier function. Cell Tissue Res. 355:577–586. 10.1007/s00441-014-1812-1 [DOI] [PubMed] [Google Scholar]
- Lagaraine C., Skipor J., Szczepkowska A., Dufourny L., and Thiery J.C.. 2011. Tight junction proteins vary in the choroid plexus of ewes according to photoperiod. Brain Res. 1393:44–51. 10.1016/j.brainres.2011.04.009 [DOI] [PubMed] [Google Scholar]
- Lee S.W., Kim W.J., Choi Y.K., Song H.S., Son M.J., Gelman I.H., Kim Y.J., and Kim K.W.. 2003. SSeCKS regulates angiogenesis and tight junction formation in blood-brain barrier. Nat. Med. 9:900–906. 10.1038/nm889 [DOI] [PubMed] [Google Scholar]
- Lee S.W., Kim W.J., Jun H.O., Choi Y.K., and Kim K.W.. 2009. Angiopoietin-1 reduces vascular endothelial growth factor-induced brain endothelial permeability via upregulation of ZO-2. Int. J. Mol. Med. 23:279–284. [PubMed] [Google Scholar]
- Liddelow S.A., Dziegielewska K.M., Ek C.J., Habgood M.D., Bauer H., Bauer H.C., Lindsay H., Wakefield M.J., Strazielle N., Kratzer I., et al. 2013. Mechanisms that determine the internal environment of the developing brain: a transcriptomic, functional and ultrastructural approach. PLoS ONE. 8:e65629 10.1371/journal.pone.0065629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liebner S., Gerhardt H., and Wolburg H.. 2000a. Differential expression of endothelial beta-catenin and plakoglobin during development and maturation of the blood-brain and blood-retina barrier in the chicken. Dev. Dyn. 217:86–98. [DOI] [PubMed] [Google Scholar]
- Liebner S., Kniesel U., Kalbacher H., and Wolburg H.. 2000b. Correlation of tight junction morphology with the expression of tight junction proteins in blood-brain barrier endothelial cells. Eur. J. Cell Biol. 79:707–717. 10.1078/0171-9335-00101 [DOI] [PubMed] [Google Scholar]
- Liebner S., Corada M., Bangsow T., Babbage J., Taddei A., Czupalla C.J., Reis M., Felici A., Wolburg H., Fruttiger M., et al. 2008. Wnt/β-catenin signaling controls development of the blood-brain barrier. J. Cell Biol. 183:409–417. 10.1083/jcb.200806024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lippoldt A., Jansson A., Kniesel U., Andbjer B., Andersson A., Wolburg H., Fuxe K., and Haller H.. 2000. Phorbol ester induced changes in tight and adherens junctions in the choroid plexus epithelium and in the ependyma. Brain Res. 854:197–206. 10.1016/S0006-8993(99)02355-0 [DOI] [PubMed] [Google Scholar]
- Ludwig R.J., Hardt K., Hatting M., Bistrian R., Diehl S., Radeke H.H., Podda M., Schön M.P., Kaufmann R., Henschler R., et al. 2009. Junctional adhesion molecule (JAM)-B supports lymphocyte rolling and adhesion through interaction with α4β1 integrin. Immunology. 128:196–205. 10.1111/j.1365-2567.2009.03100.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Y., and Radice G.L.. 2005. N-cadherin acts upstream of VE-cadherin in controlling vascular morphogenesis. J. Cell Biol. 169:29–34. 10.1083/jcb.200411127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyck R., Ruderisch N., Moll A.G., Steiner O., Cohen C.D., Engelhardt B., Makrides V., and Verrey F.. 2009. Culture-induced changes in blood-brain barrier transcriptome: implications for amino-acid transporters in vivo. J. Cereb. Blood Flow Metab. 29:1491–1502. 10.1038/jcbfm.2009.72 [DOI] [PubMed] [Google Scholar]
- Maddaluno L., Rudini N., Cuttano R., Bravi L., Giampietro C., Corada M., Ferrarini L., Orsenigo F., Papa E., Boulday G., et al. 2013. EndMT contributes to the onset and progression of cerebral cavernous malformations. Nature. 498:492–496. 10.1038/nature12207 [DOI] [PubMed] [Google Scholar]
- Mark K.S., and Davis T.P.. 2002. Cerebral microvascular changes in permeability and tight junctions induced by hypoxia-reoxygenation. Am. J. Physiol. Heart Circ. Physiol. 282:H1485–H1494. 10.1152/ajpheart.00645.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marrs J.A., Napolitano E.W., Murphy-Erdosh C., Mays R.W., Reichardt L.F., and Nelson W.J.. 1993. Distinguishing roles of the membrane-cytoskeleton and cadherin mediated cell-cell adhesion in generating different Na+,K+-ATPase distributions in polarized epithelia. J. Cell Biol. 123:149–164. 10.1083/jcb.123.1.149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martìn-Padura I., Lostaglio S., Schneemann M., Williams L., Romano M., Fruscella P., Panzeri C., Stoppacciaro A., Ruco L., Villa A., et al. 1998. Junctional adhesion molecule, a novel member of the immunoglobulin superfamily that distributes at intercellular junctions and modulates monocyte transmigration. J. Cell Biol. 142:117–127. 10.1083/jcb.142.1.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menezes M.J., McClenahan F.K., Leiton C.V., Aranmolate A., Shan X., and Colognato H.. 2014. The extracellular matrix protein laminin α2 regulates the maturation and function of the blood-brain barrier. J. Neurosci. 34:15260–15280. 10.1523/JNEUROSCI.3678-13.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizee M.R., Wooldrik D., Lakeman K.A., van het Hof B., Drexhage J.A., Geerts D., Bugiani M., Aronica E., Mebius R.E., Prat A., et al. 2013. Retinoic acid induces blood-brain barrier development. J. Neurosci. 33:1660–1671. 10.1523/JNEUROSCI.1338-12.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizee M.R., Nijland P.G., van der Pol S.M., Drexhage J.A., van Het Hof B., Mebius R., van der Valk P., van Horssen J., Reijerkerk A., and de Vries H.E.. 2014. Astrocyte-derived retinoic acid: a novel regulator of blood-brain barrier function in multiple sclerosis. Acta Neuropathol. 128:691–703. 10.1007/s00401-014-1335-6 [DOI] [PubMed] [Google Scholar]
- Mochida G.H., Ganesh V.S., Felie J.M., Gleason D., Hill R.S., Clapham K.R., Rakiec D., Tan W.H., Akawi N., Al-Saffar M., et al. 2010. A homozygous mutation in the tight-junction protein JAM3 causes hemorrhagic destruction of the brain, subependymal calcification, and congenital cataracts. Am. J. Hum. Genet. 87:882–889. 10.1016/j.ajhg.2010.10.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Møllgård K., and Saunders N.R.. 1975. Complex tight junctions of epithelial and of endothelial cells in early foetal brain. J. Neurocytol. 4:453–468. 10.1007/BF01261375 [DOI] [PubMed] [Google Scholar]
- Morita K., Furuse M., Fujimoto K., and Tsukita S.. 1999. Claudin multigene family encoding four-transmembrane domain protein components of tight junction strands. Proc. Natl. Acad. Sci. USA. 96:511–516. 10.1073/pnas.96.2.511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moro E., Ozhan-Kizil G., Mongera A., Beis D., Wierzbicki C., Young R.M., Bournele D., Domenichini A., Valdivia L.E., Lum L., et al. 2012. In vivo Wnt signaling tracing through a transgenic biosensor fish reveals novel activity domains. Dev. Biol. 366:327–340. 10.1016/j.ydbio.2012.03.023 [DOI] [PubMed] [Google Scholar]
- Murugesan N., Paul D., Lemire Y., Shrestha B., Ge S., and Pachter J.S.. 2012. Active induction of experimental autoimmune encephalomyelitis by MOG35-55 peptide immunization is associated with differential responses in separate compartments of the choroid plexus. Fluids Barriers CNS. 9:15 10.1186/2045-8118-9-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muto S., Hata M., Taniguchi J., Tsuruoka S., Moriwaki K., Saitou M., Furuse K., Sasaki H., Fujimura A., Imai M., et al. 2010. Claudin-2-deficient mice are defective in the leaky and cation-selective paracellular permeability properties of renal proximal tubules. Proc. Natl. Acad. Sci. USA. 107:8011–8016. 10.1073/pnas.0912901107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasdala I., Wolburg-Buchholz K., Wolburg H., Kuhn A., Ebnet K., Brachtendorf G., Samulowitz U., Kuster B., Engelhardt B., Vestweber D., and Butz S.. 2002. A transmembrane tight junction protein selectively expressed on endothelial cells and platelets. J. Biol. Chem. 277:16294–16303. 10.1074/jbc.M111999200 [DOI] [PubMed] [Google Scholar]
- Nico B., Quondamatteo F., Herken R., Marzullo A., Corsi P., Bertossi M., Russo G., Ribatti D., and Roncali L.. 1999. Developmental expression of ZO-1 antigen in the mouse blood-brain barrier. Brain Res. Dev. Brain Res. 114:161–169. 10.1016/S0165-3806(99)00008-5 [DOI] [PubMed] [Google Scholar]
- Nitta T., Hata M., Gotoh S., Seo Y., Sasaki H., Hashimoto N., Furuse M., and Tsukita S.. 2003. Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J. Cell Biol. 161:653–660. 10.1083/jcb.200302070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohtsuki S., Yamaguchi H., Katsukura Y., Asashima T., and Terasaki T.. 2008. mRNA expression levels of tight junction protein genes in mouse brain capillary endothelial cells highly purified by magnetic cell sorting. J. Neurochem. 104:147–154. [DOI] [PubMed] [Google Scholar]
- Osada T., Gu Y.H., Kanazawa M., Tsubota Y., Hawkins B.T., Spatz M., Milner R., and del Zoppo G.J.. 2011. Interendothelial claudin-5 expression depends on cerebral endothelial cell-matrix adhesion by β(1)-integrins. J. Cereb. Blood Flow Metab. 31:1972–1985. 10.1038/jcbfm.2011.99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padden M., Leech S., Craig B., Kirk J., Brankin B., and McQuaid S.. 2007. Differences in expression of junctional adhesion molecule-A and β-catenin in multiple sclerosis brain tissue: increasing evidence for the role of tight junction pathology. Acta Neuropathol. 113:177–186. 10.1007/s00401-006-0145-x [DOI] [PubMed] [Google Scholar]
- Paul D., Cowan A.E., Ge S., and Pachter J.S.. 2013. Novel 3D analysis of Claudin-5 reveals significant endothelial heterogeneity among CNS microvessels. Microvasc. Res. 86:1–10. 10.1016/j.mvr.2012.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeiffer F., Schäfer J., Lyck R., Makrides V., Brunner S., Schaeren-Wiemers N., Deutsch U., and Engelhardt B.. 2011. Claudin-1 induced sealing of blood-brain barrier tight junctions ameliorates chronic experimental autoimmune encephalomyelitis. Acta Neuropathol. 122:601–614. 10.1007/s00401-011-0883-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piontek J., Winkler L., Wolburg H., Müller S.L., Zuleger N., Piehl C., Wiesner B., Krause G., and Blasig I.E.. 2008. Formation of tight junction: determinants of homophilic interaction between classic claudins. FASEB J. 22:146–158. 10.1096/fj.07-8319com [DOI] [PubMed] [Google Scholar]
- Plumb J., McQuaid S., Mirakhur M., and Kirk J.. 2002. Abnormal endothelial tight junctions in active lesions and normal-appearing white matter in multiple sclerosis. Brain Pathol. 12:154–169. 10.1111/j.1750-3639.2002.tb00430.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Privratsky J.R., and Newman P.J.. 2014. PECAM-1: regulator of endothelial junctional integrity. Cell Tissue Res. 355:607–619. 10.1007/s00441-013-1779-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raleigh D.R., Marchiando A.M., Zhang Y., Shen L., Sasaki H., Wang Y., Long M., and Turner J.R.. 2010. Tight junction-associated MARVEL proteins marveld3, tricellulin, and occludin have distinct but overlapping functions. Mol. Biol. Cell. 21:1200–1213. 10.1091/mbc.E09-08-0734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raleigh D.R., Boe D.M., Yu D., Weber C.R., Marchiando A.M., Bradford E.M., Wang Y., Wu L., Schneeberger E.E., Shen L., and Turner J.R.. 2011. Occludin S408 phosphorylation regulates tight junction protein interactions and barrier function. J. Cell Biol. 193:565–582. 10.1083/jcb.201010065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts T.K., Eugenin E.A., Lopez L., Romero I.A., Weksler B.B., Couraud P.O., and Berman J.W.. 2012. CCL2 disrupts the adherens junction: implications for neuroinflammation. Lab. Invest. 92:1213–1233. 10.1038/labinvest.2012.80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saitou M., Furuse M., Sasaki H., Schulzke J.D., Fromm M., Takano H., Noda T., and Tsukita S.. 2000. Complex phenotype of mice lacking occludin, a component of tight junction strands. Mol. Biol. Cell. 11:4131–4142. 10.1091/mbc.11.12.4131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schenkel A.R., Mamdouh Z., Chen X., Liebman R.M., and Muller W.A.. 2002. CD99 plays a major role in the migration of monocytes through endothelial junctions. Nat. Immunol. 3:143–150. 10.1038/ni749 [DOI] [PubMed] [Google Scholar]
- Schrade A., Sade H., Couraud P.O., Romero I.A., Weksler B.B., and Niewoehner J.. 2012. Expression and localization of claudins-3 and -12 in transformed human brain endothelium. Fluids Barriers CNS. 9:6 10.1186/2045-8118-9-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroten M., Hanisch F.G., Quednau N., Stump C., Riebe R., Lenk M., Wolburg H., Tenenbaum T., and Schwerk C.. 2012. A novel porcine in vitro model of the blood-cerebrospinal fluid barrier with strong barrier function. PLoS ONE. 7:e39835 10.1371/journal.pone.0039835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulte D., Küppers V., Dartsch N., Broermann A., Li H., Zarbock A., Kamenyeva O., Kiefer F., Khandoga A., Massberg S., and Vestweber D.. 2011. Stabilizing the VE-cadherin-catenin complex blocks leukocyte extravasation and vascular permeability. EMBO J. 30:4157–4170. 10.1038/emboj.2011.304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulze C., and Firth J.A.. 1993. Immunohistochemical localization of adherens junction components in blood-brain barrier microvessels of the rat. J. Cell Sci. 104:773–782. [DOI] [PubMed] [Google Scholar]
- Siegenthaler J.A., Sohet F., and Daneman R.. 2013. ‘Sealing off the CNS’: cellular and molecular regulation of blood-brain barriergenesis. Curr. Opin. Neurobiol. 23:1057–1064. 10.1016/j.conb.2013.06.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steiner E., Enzmann G.U., Lyck R., Lin S., Rüegg M.A., Kröger S., and Engelhardt B.. 2014. The heparan sulfate proteoglycan agrin contributes to barrier properties of mouse brain endothelial cells by stabilizing adherens junctions. Cell Tissue Res. 358:465–479. 10.1007/s00441-014-1969-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenman J.M., Rajagopal J., Carroll T.J., Ishibashi M., McMahon J., and McMahon A.P.. 2008. Canonical Wnt signaling regulates organ-specific assembly and differentiation of CNS vasculature. Science. 322:1247–1250. 10.1126/science.1164594 [DOI] [PubMed] [Google Scholar]
- Strazielle N., and Ghersi-Egea J.F.. 1999. Demonstration of a coupled metabolism-efflux process at the choroid plexus as a mechanism of brain protection toward xenobiotics. J. Neurosci. 19:6275–6289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taddei A., Giampietro C., Conti A., Orsenigo F., Breviario F., Pirazzoli V., Potente M., Daly C., Dimmeler S., and Dejana E.. 2008. Endothelial adherens junctions control tight junctions by VE-cadherin-mediated upregulation of claudin-5. Nat. Cell Biol. 10:923–934. 10.1038/ncb1752 [DOI] [PubMed] [Google Scholar]
- Takai Y., Irie K., Shimizu K., Sakisaka T., and Ikeda W.. 2003. Nectins and nectin-like molecules: roles in cell adhesion, migration, and polarization. Cancer Sci. 94:655–667. 10.1111/j.1349-7006.2003.tb01499.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tam S.J., Richmond D.L., Kaminker J.S., Modrusan Z., Martin-McNulty B., Cao T.C., Weimer R.M., Carano R.A., van Bruggen N., and Watts R.J.. 2012. Death receptors DR6 and TROY regulate brain vascular development. Dev. Cell. 22:403–417. 10.1016/j.devcel.2011.11.018 [DOI] [PubMed] [Google Scholar]
- Tornavaca O., Chia M., Dufton N., Almagro L.O., Conway D.E., Randi A.M., Schwartz M.A., Matter K., and Balda M.S.. 2015. ZO-1 controls endothelial adherens junctions, cell-cell tension, angiogenesis, and barrier formation. J. Cell Biol. 208:821–838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Itallie C.M., and Anderson J.M.. 2014. Architecture of tight junctions and principles of molecular composition. Semin. Cell Dev. Biol. 36:157–165. 10.1016/j.semcdb.2014.08.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vestweber D. 2015. Cadherins in tissue architecture and disease. J. Mol. Med. 93:5–11. 10.1007/s00109-014-1231-5 [DOI] [PubMed] [Google Scholar]
- Vorbrodt A.W., and Dobrogowska D.H.. 2003. Molecular anatomy of intercellular junctions in brain endothelial and epithelial barriers: electron microscopist’s view. Brain Res. Brain Res. Rev. 42:221–242. 10.1016/S0165-0173(03)00177-2 [DOI] [PubMed] [Google Scholar]
- Vorbrodt A.W., and Dobrogowska D.H.. 2004. Molecular anatomy of interendothelial junctions in human blood-brain barrier microvessels. Folia Histochem. Cytobiol. 42:67–75. [PubMed] [Google Scholar]
- Wacker B.K., Freie A.B., Perfater J.L., and Gidday J.M.. 2012. Junctional protein regulation by sphingosine kinase 2 contributes to blood-brain barrier protection in hypoxic preconditioning-induced cerebral ischemic tolerance. J. Cereb. Blood Flow Metab. 32:1014–1023. 10.1038/jcbfm.2012.3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Rattner A., Zhou Y., Williams J., Smallwood P.M., and Nathans J.. 2012. Norrin/Frizzled4 signaling in retinal vascular development and blood brain barrier plasticity. Cell. 151:1332–1344. 10.1016/j.cell.2012.10.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessel F., Winderlich M., Holm M., Frye M., Rivera-Galdos R., Vockel M., Linnepe R., Ipe U., Stadtmann A., Zarbock A., et al. 2014. Leukocyte extravasation and vascular permeability are each controlled in vivo by different tyrosine residues of VE-cadherin. Nat. Immunol. 15:223–230. 10.1038/ni.2824 [DOI] [PubMed] [Google Scholar]
- Williams D.W., Calderon T.M., Lopez L., Carvallo-Torres L., Gaskill P.J., Eugenin E.A., Morgello S., and Berman J.W.. 2013. Mechanisms of HIV entry into the CNS: increased sensitivity of HIV infected CD14+CD16+ monocytes to CCL2 and key roles of CCR2, JAM-A, and ALCAM in diapedesis. PLoS ONE. 8:e69270 10.1371/journal.pone.0069270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams M.J., Lowrie M.B., Bennett J.P., Firth J.A., and Clark P.. 2005. Cadherin-10 is a novel blood-brain barrier adhesion molecule in human and mouse. Brain Res. 1058:62–72. 10.1016/j.brainres.2005.07.078 [DOI] [PubMed] [Google Scholar]
- Willis C.L., Camire R.B., Brule S.A., and Ray D.E.. 2013. Partial recovery of the damaged rat blood-brain barrier is mediated by adherens junction complexes, extracellular matrix remodeling and macrophage infiltration following focal astrocyte loss. Neuroscience. 250:773–785. 10.1016/j.neuroscience.2013.06.061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolburg H., and Paulus W.. 2010. Choroid plexus: biology and pathology. Acta Neuropathol. 119:75–88. 10.1007/s00401-009-0627-8 [DOI] [PubMed] [Google Scholar]
- Wolburg H., Neuhaus J., Kniesel U., Krauss B., Schmid E.M., Ocalan M., Farrell C., and Risau W.. 1994. Modulation of tight junction structure in blood-brain barrier endothelial cells. Effects of tissue culture, second messengers and cocultured astrocytes. J. Cell Sci. 107:1347–1357. [DOI] [PubMed] [Google Scholar]
- Wolburg H., Wolburg-Buchholz K., Liebner S., and Engelhardt B.. 2001. Claudin-1, claudin-2 and claudin-11 are present in tight junctions of choroid plexus epithelium of the mouse. Neurosci. Lett. 307:77–80. 10.1016/S0304-3940(01)01927-9 [DOI] [PubMed] [Google Scholar]
- Wolburg H., Wolburg-Buchholz K., Kraus J., Rascher-Eggstein G., Liebner S., Hamm S., Duffner F., Grote E.H., Risau W., and Engelhardt B.. 2003. Localization of claudin-3 in tight junctions of the blood-brain barrier is selectively lost during experimental autoimmune encephalomyelitis and human glioblastoma multiforme. Acta Neuropathol. 105:586–592. [DOI] [PubMed] [Google Scholar]
- Wolburg-Buchholz K., Mack A.F., Steiner E., Pfeiffer F., Engelhardt B., and Wolburg H.. 2009. Loss of astrocyte polarity marks blood-brain barrier impairment during experimental autoimmune encephalomyelitis. Acta Neuropathol. 118:219–233. 10.1007/s00401-009-0558-4 [DOI] [PubMed] [Google Scholar]
- Wyss L., Schäfer J., Liebner S., Mittelbronn M., Deutsch U., Enzmann G., Adams R.H., Aurrand-Lions M., Plate K.H., Imhof B.A., and Engelhardt B.. 2012. Junctional adhesion molecule (JAM)-C deficient C57BL/6 mice develop a severe hydrocephalus. PLoS ONE. 7:e45619 10.1371/journal.pone.0045619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto H., Ehling M., Kato K., Kanai K., van Lessen M., Frye M., Zeuschner D., Nakayama M., Vestweber D., and Adams R.H.. 2015. Integrin β1 controls VE-cadherin localization and blood vessel stability. Nat. Commun. 6:6429 10.1038/ncomms7429 [DOI] [PubMed] [Google Scholar]
- Zhou Y., and Nathans J.. 2014. Gpr124 controls CNS angiogenesis and blood-brain barrier integrity by promoting ligand-specific canonical wnt signaling. Dev. Cell. 31:248–256. 10.1016/j.devcel.2014.08.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Wang Y., Tischfield M., Williams J., Smallwood P.M., Rattner A., Taketo M.M., and Nathans J.. 2014. Canonical WNT signaling components in vascular development and barrier formation. J. Clin. Invest. 124:3825–3846. 10.1172/JCI76431 [DOI] [PMC free article] [PubMed] [Google Scholar]