Abstract
Environmental stress causes membrane damage in plants. Lipid studies are required to understand the adaptation of plants to climate change. Here, LC-MS-based lipidomic and microarray transcriptome analyses were carried out to elucidate the effect of short-term heat stress on the Arabidopsis thaliana leaf membrane. Vegetative plants were subjected to high temperatures for one day, and then grown under normal conditions. Sixty-six detected glycerolipid species were classified according to patterns of compositional change by Spearman’s correlation coefficient. Triacylglycerols, 36:4- and 36:5-monogalactosyldiacylglycerol, 34:2- and 36:2-digalactosyldiacylglycerol, 34:1-, 36:1- and 36:6-phosphatidylcholine, and 34:1-phosphatidylethanolamine increased by the stress and immediately decreased during recovery. The relative amount of one triacylglycerol species (54:9) containing α-linolenic acid (18:3) increased under heat stress. These results suggest that heat stress in Arabidopsis leaves induces an increase in triacylglycerol levels, which functions as an intermediate of lipid turnover, and results in a decrease in membrane polyunsaturated fatty acids. Microarray data revealed candidate genes responsible for the observed metabolic changes.
Due to climate change, land plants may suffer heat stress, which causes an increase in molecular disorder and disintegration of lipid bilayers1,2. Heat stress-induced reactive oxygen species promote peroxidation of unsaturated fatty acids3. Plastidic fatty acid desaturase (FAD) mutants, in which the number of glycerolipid fatty acid double bonds is reduced, are known to be heat tolerant4,5,6. Another mutation resulting in impaired biosynthesis of digalactosyldiacylglycerol (DGDG) in chloroplasts has been shown to lead to heat sensitivity7. These mutant studies suggest that changes in the proportion of glycerolipid composition in chloroplasts are required for plant acclimatization to heat stress.
One study on Arabidopsis plants under various high temperatures suggested that the heat-induced decrease in unsaturation of membrane glycerolipids is slow and consistent with the rate of de novo fatty acid synthesis8. Therefore, a decrease in glycerolipid unsaturation is considered unsuitable for a rapid response (increase) to environmental temperature, but is advantageous for plants for long-term acclimatization to gradual temperature increases. A time-course study on Arabidopsis plants grown at 33 °C showed that α-linolenic acid (18:3) decreased within 3 days, whereas hexadecatrienoic (roughanic) acid (16:3) gradually decreased after an additional 13 days6. Metabolic labelling experiments have shown that incorporation of 1-[14C]-acetate into DGDG was slower than that into monogalactosyldiacylglycerol (MGDG) and phosphatidylcholine (PC)9. Because the rate of biosynthesis and degree of unsaturation of glycerolipids are different among lipid classes, it is useful to compare compositions of various lipid species under different temperatures and correlate metabolite patterns with metabolic pathways.
Plant leaves synthesise small amounts of triacylglycerol (TAG) in the endoplasmic reticulum (ER) and accumulate it as lipid droplets in the cytosol10. TAG is also possibly synthesised in chloroplasts and accumulated as plastoglobules in chloroplasts11. The accumulation of plastoglobules was observed after a short-term moderate heat treatment in Arabidopsis12, suggesting an increase in the accumulation of TAG under heat stress. Recent LC-MS-based lipidomic studies revealed that long-term cold stress and a 2-week drought stress resulted in increases in TAG species in Arabidopsis13,14. According to these data, the composition of the detected TAG species seems to be different among the stresses even in the same ecotype plants. During recovery from drought stress in Craterostigma plantagineum, the extent of the decrease differed among individual stress-induced TAG species14. These results suggest that each TAG species behaves differently during the environmental stress, possibly reflecting the change (decrease) in membrane glycerolipid composition.
The utility of the integration of metabolic changes with transcriptome data in gene discovery has often been reported in studies on plant specialized (secondary) metabolism15. Published microarray transcriptome data suggest the coexpression of genes involved in de novo fatty acid synthesis16, glycolipid synthesis17, and the β-oxidation/glyoxylate cycle18. Senescent leaves coordinately reduce and induce the expression of genes involved in de novo fatty acid synthesis and β-oxidation, respectively19. An integrated lipidomic and transcriptomic study in Arabidopsis under environmental stress was recently conducted20. Their results suggest that Arabidopsis transcriptionally coordinates the regulation of glycerolipid metabolism, and the metabolic changes under heat stress may be explained by induced and reduced expression of genes encoding enzymes, proteins involved in lipid trafficking, and transcription factors.
Plant membrane properties should be determined and explained as resulting from the complex mixture of lipid species having different headgroup structures, fatty acid lengths, and the degree of unsaturation. In this study, we simultaneously analysed the changes in glycerolipids including glycolipids, phospholipids, and storage lipids in Arabidopsis leaves under heat stress and during recovery by an LC-MS-based lipidomics platform. Although plant leaf lipidomic studies under heat stress have been reported7,21,22, a thorough determination of TAG species, changes during recovery, and integration with gene expression are required for a better understanding of heat responses in lipid remodelling. In the present study, lipid species were classified according to the pattern of compositional changes, and these changes were represented on a metabolic pathway map with possible lipid trafficking among cellular organelles. A microarray transcriptome analysis was performed to determine the molecular regulation of lipid metabolism under heat stress. We describe plant acclimatization responses to heat stress based on the changes in glycerolipid properties, and suggest an important role for TAG formation.
Results and Discussion
Lipidomics reveals a diversity of lipids accumulated under heat stress
Arabidopsis ecotypes Col-0 and Nossen were grown on agar plates and treated with one day of heat stress at 14 days of age (Supplementary Fig. S1). Four temperatures, 22 °C (control), 30 °C, 34 °C and 38 °C were examined. Nossen plants grown at 22 °C had a greater fresh weight than Col-0 plants grown at 22 °C. As compared to the plants grown at 22 °C, both ecotype plants at 30 °C and 34 °C showed more petiole length (Supplementary Fig. S2). Indeed, hyponastic growth is known to be positively regulated by the plant hormones auxin and abscisic acid under heat stress23,24. The 38 °C treatment was not lethal, but no considerable elongation was observed (Supplementary Fig. S2). Aerial parts were harvested and analysed using a lipidomics method. Lipid composition during recovery from heat stress was also examined. Col-0 plants at 18 days and Nossen plants at 14 days were subjected to 38 °C heat stress for one day. Aerial parts were harvested at one and two days after the stress treatment (Supplementary Fig. S1).
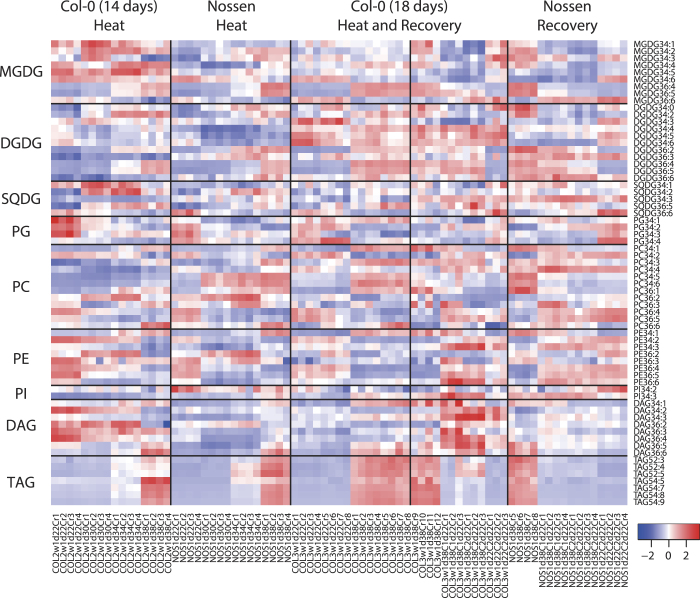
Lipidomic analysis detected a number of glycerolipid species including (1) the glycolipids MGDG, DGDG, and sulfoquinovosyldiacylglycerol (SQDG); (2) the phospholipids phosphatidylglycerol (PG), PC, phosphatidylethanolamine (PE), and phosphatidylinositol (PI); (3) diacylglycerol (DAG), a lipid intermediate; and (4) the storage lipid TAG (Fig. 1, Supplementary Table S1).
Figure 1. Overview of glycerolipid levels in Arabidopsis leaves under heat stress and during recovery.
The 66 lipid species observed from the all environmental conditions (heat and recovery) and ecotypes (Col-0 and Nossen) were shown. Values were normalised to 1d38C data in each experiment, and scaled among measurements as described in Methods. Each column represents an LC-MS measurement. Biological replicates N = 3, 4, 8, or 12. MGDG, monogalactosyldiacylglycerol; DGDG, digalactosyldiacylglycerol; SQDG, sulfoquinovosyldiacylglycerol; PG, phosphatidylglycerol; PC, phosphatidylcholine; PE, phosphatidylethanolamine; PI, phosphatidylinositol; DAG, diacylglycerol; TAG, triacylglycerol. Experimental condition can be found in Supplementary Fig. S1. The normalised data was available in Supplementary Table S1.
Mass spectrometry was used to determine the total number of carbon atoms and double bonds of two fatty acids within a glycerolipid species. Arabidopsis leaves mostly accumulate glycerolipids containing the fatty acids 34:1, 34:2, 34:3, 34:6, 36:1, 36:2, 36:4, 36:5, and 36:6, which are likely composed of two fatty acid combinations: palmitic acid (16:0)/ oleic acid (18:1), 16:0/ linoleic acid (18:2), 16:0/18:3, 16:3/18:3, 18:0/18:1, 18:1/18:1, 18:2/18:2, 18:2/18:3 and 18:3/18:3, respectively9,25,26. Seventy lipid species ranging in number from 34:0 to 34:6 and 36:0 to 36:6 were observed in at least one environmental condition (Supplementary Table S1). Among them, 59 lipid species were observed in all conditions (Fig. 1).
TAG is composed of 3 fatty acids. Twenty-eight TAG species were observed in at least one environmental condition. The major TAG species in Arabidopsis leaves were TAGs consisting of 52:x and 54:x, which contain 52 and 54 carbon atoms in the 3 fatty acids in total. TAGs consisting of 50:x and 56:x, which contain 50 and 56 carbon atoms, respectively, were also detected, but occupied less than 3% of the total amount of TAG species in Arabidopsis leaves. Seven TAG species were detected in all conditions (Fig. 1).
Lipid species can be classified by their patterns of change under heat stress
To classify the detected lipid species according to trends in accumulation changes, Spearman’s correlation coefficients were calculated among 12 variables of conditions and the result is shown in matrix form (Supplementary Fig. S3). Hierarchical clustering analysis using the complete linkage method based on Euclidean distance classified the 66 lipid species (59 diacyl glycerolipids and 7 TAG) into 10 groups. This result indicated that there were 3 pairs having strong negative correlations, i.e. the lipid species belonging to clusters 1 and 10, clusters 2 and 6, and clusters 6 and 7. The groups exhibiting a negative correlation are possibly interconverted during the stress treatment.
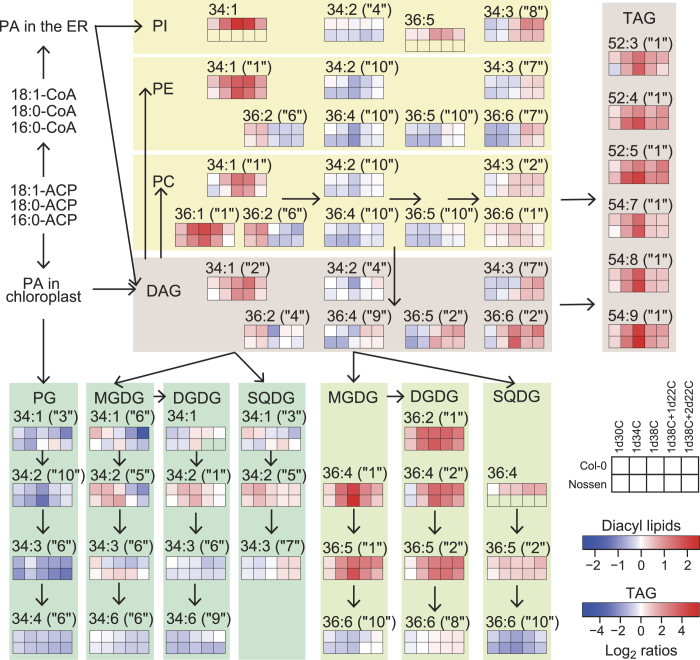
The obtained metabolic changes were represented as a heat map superimposed on a metabolic pathway map (Fig. 2). Fifty-seven lipid species were selected based on their relatively high accumulation in Col-0 plants and their detectability in all conditions. Characteristic lipid species belonged to 5 clusters (clusters 1, 2, 6, 7 and 10). Cluster 1 represents the lipid species that were increased by heat stress and returned to normal levels during recovery. Cluster 2 represents the lipid species that were increased by heat stress and gradually returned to normal levels (more slowly than cluster 1) during recovery. Cluster 7 represents the lipid species that increased to above normal levels during recovery. Cluster 10 represents the lipid species that were decreased by heat stress and returned to normal levels during recovery. Cluster 6 represents the lipid species that decreased at 38 °C and further decreased or remained constant during recovery. Lipid species belonging to the same cluster tended to be closely situated in the metabolic pathway (Fig. 2). The reported clusters of Arabidopsis leaves after wounding27 were not reproduced well in our clusters after heat stress, suggesting the different response occurred under the different abiotic stresses in Arabidopsis leaves.
Figure 2. Heat map representation of lipid species in Arabidopsis leaves under heat stress superimposed on a glycerolipid metabolic pathway map.
The average changes in lipid species are shown by 10 boxes (2 rows and 5 columns), which contain 2 genotypes: Col-0 (upper row) and Nossen (lower row); and 5 environments: 1d30C, 1d34C, 1d38C, 1d38C + 1d22C, 1d38C + 2d22C (left-to-right). Heat map colours show the average log2 ratios of fold-changes in each stress condition to the normal 22 °C condition. Due to different induction levels, values of diacyl membrane lipids and TAG are shown by different scales. Samples depicted by boxes with no colour in the Nossen data were not detected. Lipid species belonging to the 10 clusters (Spearman’s correlation coefficients, see Supplementary Fig. S3 for details) are shown with the numbers in parentheses (e.g. “1”, “2”, “6”, “7”, and “10”). Individual experimental data can be found in Supplementary Table S1.
TAG increased under heat stress and likely enhanced the changes in membrane composition
Although most of the detected TAG species increased under heat stress, the relative amounts of TAG species were also altered (Supplementary Fig. S4). The relative amount of 54:9 TAG increased along with increased temperature. The relative amount of 52:5 TAG peaked at 34 °C and decreased at 38 °C. During recovery, the relative amount of 52:5 TAG increased and that of 54:9 TAG decreased. Because 54:9 TAG is likely composed of three 18:3 chains, these data suggest that TAG synthesis under heat stress favours 18:3 as a substrate. Increase in the relative amount of 52:5 TAG has been reported in studies of Arabidopsis under long-term cold stress and during leaf senescence13,28. A slower decrease in 52:x TAGs during recovery from drought stress as compared to that of 54:x TAG in C. plantagineum has been reported14. Taken together, these results suggest that the increase in 54:9 TAG is more specific to short-term heat stress and the increase in 52:5 TAG represents a comprehensive and slow response to environmental stress.
It has been suggested that the ER glycerolipid pathway and TAG synthesis in leaves, rather than β-oxidation, function in storage of excess fatty acids, a process that has been referred to as a cellular vent10 or a transient buffer19. Lipid turnover in leaves was reported to be slow, about 4% per day29. With regard to the metabolic flux of TAG synthesis in the ER, acyltransferase has been suggested to have adequate capacity10. Therefore, Arabidopsis plants likely accumulate TAG under heat stress to remove excess 18:3 from membranes.
Microarray transcriptome analysis revealed heat-responsive genes possibly involved in glycerolipid metabolism under heat stress
To explain the changes in lipid composition from the perspective of gene expression, microarray transcriptome analysis was performed. Arabidopsis ecotype Col-0 plants at 18 days of age were subjected to heat stress at 38 °C for one day, and then returned to 22 °C (Supplementary Fig. S1). Aerial parts of the plants were harvested at 2 time points for heat treatment (Heat08 hr, Heat24 hr) and 2 time points for recovery (Recovery08 hr, Recovery24 hr). Because the transcriptome data from normally grown plants were similar when they were harvested at the same clock time, mean values for the 22 °C control condition (Control08 hr) were calculated from the transcriptome data of both 18-day-old and 19-day-old plants harvested at the same clock time of Heat08 hr and Recovery08 hr. Similarly, the other control condition (Control24 hr) was averaged from the data among 18-day-old, 19-day-old, and 20-day-old plants harvested at the same clock time of Heat24 hr and Recovery24 hr.
Overview of changes in glycerolipid metabolism
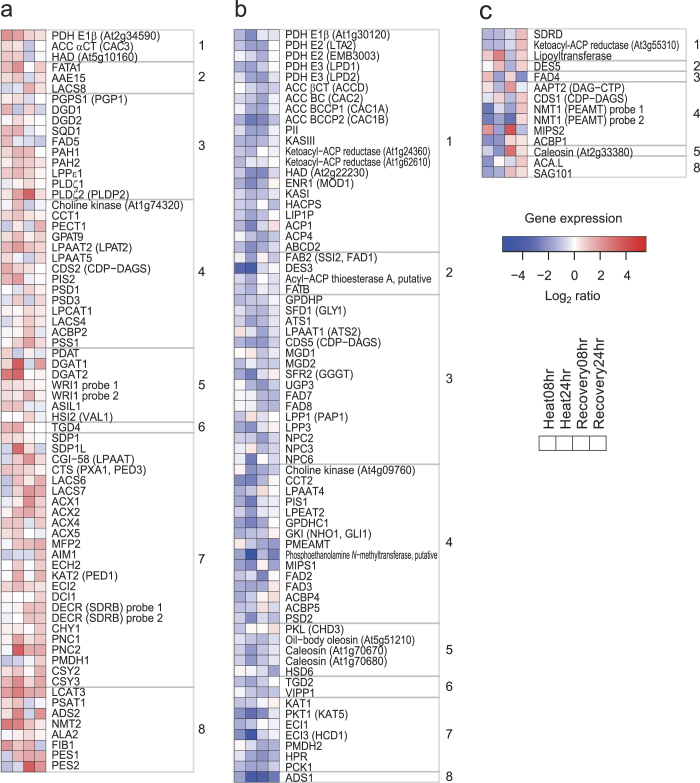
From the literature, 244 genes were selected as possibly being involved in glycerolipid metabolism11,30,31,32 (Supplementary Table S2). Among them, the expression levels of 233 genes were detectable as signal intensities from 228 probe sets; the genes exhibiting significantly increased (>1.5-fold) and decreased (<0.5-fold) expression were analysed. Because the number of genes with increased expression was smaller, we analysed the >1.5-fold, but not the >2-fold data. The samples at Heat08 hr, Heat24 hr, Recovery08 hr, and Recovery24 hr contained 31, 48, 38, and 38 increased probe sets, respectively (Supplementary Fig. S5). On the other hand, they also contained 41, 54, 44, and 13 decreased probe sets, respectively (Supplementary Fig. S5). Recovery24 hr contained the smallest number of genes (13) with decreased expression, suggesting that the plants quickly recovered from the stress at least at the transcriptional level. In total, there were 68 genes (70 probe sets) with increased expression under at least one condition and no decrease, 71 genes (69 probe sets) with decreased expression under at least one condition and no increase, 13 genes (13 probe sets) with both increased and decreased expression under at least one condition, and 81 genes (76 probe sets) with neither increased nor decreased expression (Fig. 3, Supplementary Table S2).
Figure 3. The expression of genes annotated to be involved in glycerolipid metabolism in Arabidopsis leaves under heat stress and during recovery.
The expression of 233 genes (228 probe sets) selected from the literature was analysed by microarray. Four sets of fold-changes were calculated as Heat08 hr/Control08 hr, Heat24 hr/Control24 hr, Recovery08 hr/Control08 hr and Recovery24 hr/Control24 hr and are shown as a heat map from left-to-right. (a) Probe sets increased >1.5-fold under at least one condition and not decreased, (b) probe sets decreased <0.5-fold under at least one condition and not increased, (c) probe sets both increased >1.5-fold under at least one condition and decreased <0.5-fold under at least one condition. Genes were selected from the literature, and classified into 8 groups of metabolic pathways: (1) plastidial fatty acid synthesis; (2) fatty acid elongation, desaturation and export from plastid; (3) plastidial glycerolipid, galactolipid and sulfolipid synthesis; (4) eukaryotic phospholipid synthesis; (5) TAG synthesis; (6) lipid trafficking; (7) TAG degradation, β-oxidation, the TCA/glyoxylate cycles; (8) miscellaneous lipid-related genes, and phytyl ester synthases. Genes that changed significantly (t-test with Benjamini-Hochberg FDR, p < 0.05, N = 3, 6, or 9) among the selected genes are shown. Further information is available in Supplementary Table S2.
Stress-responsive transcription factors
The expression of transcription factor genes involved in abiotic stress in Arabidopsis was examined (Supplementary Fig. S6, Supplementary Table S3). DREB1A, which was reported to be involved in cold stress33, decreased <0.5-fold at Heat08 hr and Recovery08 hr. DREB2A, which was reported to be involved in drought and heat stress34,35, increased >2-fold at Heat08 hr, Heat24 hr, Recovery08 hr, and Recovery24 hr. HSFA2, which was reported to be involved in heat stress36, increased >2-fold at Heat08 hr, Heat24 hr, and Recovery08 hr. These results indicate that appropriate heat-stress responses of Arabidopsis leaves were detectable in our experimental conditions.
Circadian changes in glycerolipid metabolism-relate gene expression
The observation that some genes displayed different expression levels between Control08 hr and Control24 hr suggests an effect of circadian rhythm on their expression. There were 17 probe sets that increased >1.5-fold at Control24 hr (i.e., in the morning), whereas 24 sets increased >1.5-fold at Control08 hr (i.e., in the afternoon) at 22 °C (Supplementary Table S2). Among them, 40 probe sets changed under heat stress or during recovery. Therefore, increase in the expression of DGD1 (digalactosyldiacylglycerol synthase 1), FAD5 (fatty acid desaturase 5), PDAT (phospholipid: diacylglycerol acyltransferase), FIB1 (fibrillin 1), FAD4, and MIPS2 (myo-inositol-1-phosphate synthase 2) at Heat08 hr may reflect a heat stress-induced decrease in circadian-influenced expression of genes related to glycerolipid metabolism.
Prokaryotic glycerolipid pathway in chloroplasts
The expression of 21 genes, which are likely involved in de novo fatty acid synthesis in chloroplasts, was decreased by heat stress and/or during recovery (group 1 of Fig. 3b). ATS1 (glycerol-3-phosphate acyltransferase 1), LPAAT1 (1-acylglycerol-3-phosphate acyltransferase 1), and CDS5 (CDP-diacylglycerol synthase 5) are involved in the so-called prokaryotic pathway, which refers to the plastidic biosynthesis of 16:3-containing glycolipids and PG30. The expression of these genes decreased at Heat08 hr, Heat24 hr, and/or Recovery08 hr (Fig. 3b). These results suggest a decrease in chloroplast glycerolipid synthesis, which is consistent with the decrease observed in 34:x glycolipids and PG under heat stress and during recovery (Fig. 2). Decrease in the expression of MGD1 (monogalactosyldiacylglycerol synthase 1) and MGD2 at Recovery08 hr can explain the lipidomics results, in which the total amount of MGDG decreased during recovery (Supplementary Fig. S7). MGDG and PG may be also converted to TAG and other lipids, which have been detected after wounding27. FAB2 (also referred to as SSI2 and FAD1) converts 18:0-ACP to 18:1-ACP in chloroplasts37. Its expression decreased at Heat24 hr, which may explain the increased accumulation of 18:0-containing 36:1 PC under heat stress (Fig. 2).
Phospholipid synthesis in the ER
FATA1 prefers to hydrolyse 18:1-ACP rather than 16:0-ACP in chloroplasts38. Mutations resulting in reduced FATA1 gene expression were reported to affect fatty acid composition in Arabidopsis seeds39. The expression of FATA1, PAH1 (phosphatidate phosphohydrolase 1), PAH2, CCT1 (choline-phosphate cytidylyltransferase 1) and LPAAT2 increased at Heat08 hr and/or Heat24 hr (Fig. 3a). These observations could explain the increase in 18:1-containing 34:1 PC, 36:1 PC, 36:2 PC, 34:1 PE, 36:2 PE, and 34:1 PI in extrachloroplasts under heat stress (Fig. 2, pathway ‘a’ of Fig. 4).
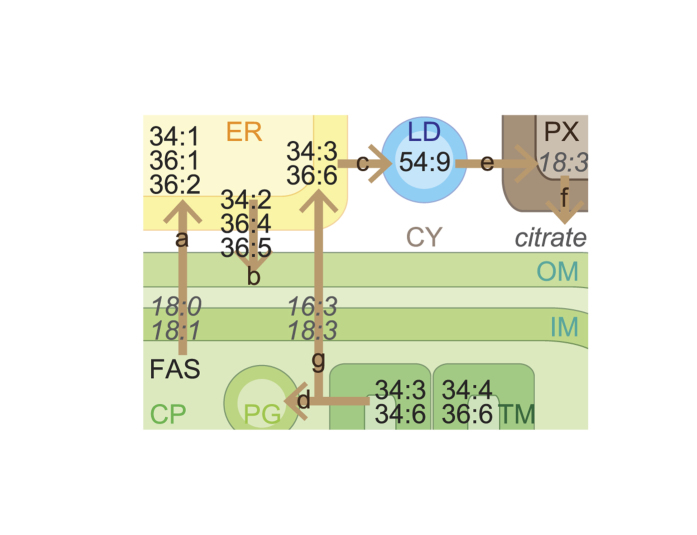
Figure 4. Predicted intracellular lipid trafficking. Integrated lipidome and transcriptome investigations suggest an enhanced lipid movement among organelles under heat stress in Arabidopsis leaves.
Seven arrows (‘a’ through ‘g’) show metabolic pathways suggested to be induced by heat stress. FAS, fatty acid synthesis; CP, chloroplasts; TM, thylakoid membrane; PG, plastoglobules; IM, inner envelope membrane; OM, outer envelope membrane; ER, endoplasmic reticulum; LD, lipid droplets; PX, peroxisome; CY, cytosol.
Eukaryotic glycolipid synthesis
PAH1 and PAH2 were reported to be involved in the transport of diacyl glycerolipids from the ER to chloroplasts under phosphorus-depleted stress40. LPPε1 encodes one of the chloroplast-localised phosphatidic acid (PA) phosphatases41. PLDζ2 was reported to be induced by phosphorus deficiency and be involved in phospholipid metabolism42. TGD4 was reported to bind to PA and be involved in the transport of glycerolipids from the ER to chloroplasts43,44. The expression of these genes increased at Heat08 hr and/or Heat24 hr (Fig. 3a). This result could explain the decrease in 18:2-containing 34:2 PC, 36:4 PC, 36:5 PC, 34:2 PE, 36:4 PE, 36:5 PE, and 34:2 PI accumulated in extrachloroplasts, as well as the increase in 18:2-containing glycolipids including 36:4 MGDG, 36:5 MGDG, 36:4 DGDG, 36:5 DGDG, 36:4 SQDG, and 36:5 SQDG, which are synthesised via the so-called eukaryotic pathway, in which the substrates of glycolipid fatty acids are derived from the ER (Fig. 2, pathway ‘b’ of Fig. 4). These data reveal drastic changes in the flow of DAG in glycolipid metabolism accompanied by changes in the expression of genes involved in phospholipid degradation and lipid transport from the ER to chloroplasts. In the case of SQDG, UDP-sulfoquinovose may be newly synthesised under heat stress because the expression of SQD1 (UDP-sulfoquinovose synthase) increased (Fig. 3a).
TAG synthesis
The expression of PAH1, PAH2, PLDζ2, LPAAT2, PDAT, DGAT1 (acyl-CoA: diacylglycerol acyltransferase 1), and DGAT2 increased at Heat08 hr and/or Heat24 hr (Fig. 3a). These genes may be involved in increases in TAG synthesis in the ER via the activation of the Kennedy pathway under heat stress (Fig. 2, pathway ‘c’ of Fig. 4). FIB1a and/or FIB1b, which are recognised by the same probe set, were reported to be involved in plastoglobule formation in chloroplasts under abiotic stress45. Their expression increased at Heat08 hr, Heat24 hr, and Recovery08 hr, suggesting the involvement of these genes in plastoglobule formation under heat stress (Fig. 3a). The expression of PES111 and DGAT146, which were reported to be involved in TAG synthesis in chloroplasts, increased under heat stress and during recovery (Fig. 3a). TAG might also accumulate in chloroplasts as plastoglobules (pathway ‘d’ of Fig. 4).
Genes involved in lipid turnover
The expression of SDP1 and SDP1L, which hydrolyse TAG47, increased at Heat08 hr and Heat24 hr, respectively (Fig. 3a). These results suggest that, although TAG may accumulate, TAG degradation is also enhanced under heat stress (pathway ‘e’ of Fig. 4). The expression of 21 genes, which are likely involved in β-oxidation and the tricarboxylic acid (TCA) and glyoxylate cycles, increased at Heat24 hr, Recovery08 hr, and Recovery24 hr (group 7 of Fig. 3a). These results suggest that Arabidopsis increases turnover of membrane glycerolipids under heat stress and during recovery at the transcriptional level (pathway ‘f’ of Fig. 4).
In germinating Arabidopsis, the first step of the TCA cycle is catalysed by CSY2 and CSY3, which were reported to be localised in the peroxisome48. The authors showed that these proteins are essential for exporting citrate derived from acetyl-CoA from the peroxisome to the cytosol for fatty acid respiration. The expression of these genes increased under heat stress and during recovery (Fig. 3a). Conversely, the expression of other genes (PMDH2, HPR, and PCK1), which also belong to the glyoxylate cycle and have been suggested to be involved in lipid catabolism32, did not increase under heat stress (Fig. 3b). These results suggest that Arabidopsis produces citrate in the peroxisome to effect changes in membrane composition, rather than use lipids as energy sources under heat stress (pathway ‘f’ of Fig. 4).
Conclusion
The lipidomics data indicate that the composition of membrane lipids in leaves was dynamic under heat stress within one day (Fig. 2). Most of these changes can be understood by a decrease in unsaturated fatty acids from chloroplast membranes. This decrease was likely derived from (1) lipid transfer between chloroplasts and the ER, and (2) decrease in chloroplast glycolipids and PG containing 18:3 and 16:3 by lipid turnover (pathways ‘a’ through ‘f’ of Fig. 4). This process was likely enhanced by increases in 18:3-containing TAG species. In most cases, microarray results suggest the regulation of glycerolipid metabolism at the transcriptional level. However, for lack of gene annotations, the process of degradation and export of chloroplastic 16:3 and 18:3 cannot be explained (pathway ‘g’ of Fig. 4). The integration of the lipidomics data and transcriptome data will be useful for elucidation of the unknown pathways in the future.
Methods
Lipidomic analysis
Arabidopsis thaliana ecotype Columbia (Col-0) and Nossen were used. Arabidopsis seeds were surface-sterilised and sown on an agar-solidified Murashige and Skoog medium containing 0.5% (w/v) sucrose. Plants were grown at 22 °C under a 16-h-light/8-h-dark cycle. Fourteen-day-old Col-0 and Nossen plants at about 3 h after the onset of the light phase were subjected to heat stress at 22 °C (control), 30 °C, 34 °C or 38 °C for one day under a continuous light condition (biological replicate N = 4) (Supplementary Fig. S1). For recovery experiments, 18-day-old Col-0 and 14-day-old Nossen plants were subjected to heat stress at 38 °C for one day under the 16/8 h light-dark cycle, and then returned to 22 °C and grown for one day and 2 days longer (biological replicate N = 3, 4, 8, or 12) (Supplementary Fig. S1).
Aerial parts were harvested at about 3 h after the onset of light phase. Crude lipid was extracted from the aerial parts according to the method of Okazaki et al.49. The dried chloroform extract was dissolved in 160 μL ethanol for LC-MS analysis. The conditions for LC-MS analysis were described in the same paper49. Lipid data were obtained from 77 distinct plant samples that included 6 environmental conditions and 2 ecotypes, in which the plant samples were prepared from 6 individual growth periods.
Levels of individual lipid species in every sample preparation were normalised to the sum of peak areas of lipid species within the same lipid class in samples grown at 38 °C for one day (1d38C). The scaled levels of the 66 lipid species, which were detected under the all conditions and ecotypes, were calculated to show overview as a heat map, where levels of individual lipid species were subtracted by the mean of values of the lipid species among the 77 samples, then divided by the standard deviations for values of the lipid species among the 77 samples as described in Vu et al.27. Spearman’s correlation coefficients were calculated among 12 variables, which were expressed as the average of each condition (22 °C control (1d22C and 1d22C+2d22C), 30 °C for one day (1d30C), 34 °C for one day (1d34C), 1d38C, 38 °C for one day then 22 °C for one day (1d38C+1d22C), and 38 °C for one day then 22 °C for 2 days (1d38C+2d22C)) for each ecotype (Col-0 (C) and Nossen (N)). Hierarchical clustering of the lipid species was conducted by the complete linkage method based on Euclidean distance (k = 10) by the software R. A heat map was constructed to depict the log2 of the ratio of the average of each stress condition to that of the 22 °C control. Significant differences in individual values were investigated by Welch’s t test (two-sided).
Transcriptome analysis
Arabidopsis ecotype Col-0 seeds were surface-sterilised and sown on an agar-solidified medium, which was identical to that used for lipidomic analysis as described above. Plants were grown at 22 °C under a 16-h-light/8-h-dark cycle. Eighteen-day-old plants were subjected to heat stress at 38 °C for 24 h (Supplementary Fig. S1). The stress treatment was started at 3 h after the onset of the light phase. After the heat stress treatment, the plants were grown at 22 °C for 24 h.
Aerial parts were harvested at 8 h after the start of heat stress (Heat08 hr, biological replicate N = 3), 24 h after the start of heat stress (Heat24 hr, biological replicate N = 3), 8 h after the start of recovery (Recovery08 hr, biological replicate N = 3), and 24 h after the start of recovery (Recovery24 hr, biological replicate N = 3) (Supplementary Fig. S1). The samples named Control08 hr (N = 6, including 3 biological replicates of 18-day-old and 19-day-old plants) and Control24 hr (N = 9, including 3 biological replicates of 18-day-old, 19-day-old and 20-day-old plants) were harvested from plants grown under the 22 °C condition at 11 and 3 h after the onset of light phase, respectively.
Total RNA was extracted using an RNeasy Plant Mini Kit (Qiagen, Valencia, CA). The Arabidopsis Genome ATH1 DNA array (Affymetrix, Santa Clara, CA) was hybridised according to the manufacturer’s protocol. Raw data were deposited in NCBI-GEO under accession numbers GSE63128. The obtained data were summarised by the method of MAS5, and normalised as baseline to median of all samples using GeneSpring version 12 (Agilent Technologies, Palo Alto, CA).
Genes possibly involved in glycerolipid metabolism were selected from the literature (Supplementary Table S2)11,30,31,32. There were 244 genes annotated as being involved in the metabolic pathways: plastidial fatty acid synthesis; fatty acid elongation, desaturation and export from the plastid; plastidial glycerolipid, galactolipid and sulfolipid synthesis; eukaryotic phospholipid synthesis; triacylglycerol (TAG) synthesis; lipid trafficking; TAG degradation; β-oxidation; the tricarboxylic acid cycle and glyoxylate cycle (selected only the genes possibly related to fatty acid catabolism32); miscellaneous lipid-related (selected only the genes having gene symbols31); and phytyl ester synthases11 (Supplementary Table S2). The genes showing significant changes in expression were chosen by volcano plots of fold-changes and the moderated t-test with the Benjamini-Hochberg FDR method (corrected p-values) among the selected genes (or the whole probe sets for analysis of transcription factors) by the software GeneSpring.
Additional Information
How to cite this article: Higashi, Y. et al. Landscape of the lipidome and transcriptome under heat stress in Arabidopsis thaliana. Sci. Rep. 5, 10533; doi: 10.1038/srep10533 (2015).
Supplementary Material
Acknowledgments
We thank K. Takano and T. Nishizawa from RIKEN CSRS for technical support. This research was supported in part by JST, Strategic International Collaborative Research Program, SICORP.
Footnotes
The authors declare no competing financial interests.
Author Contributions K.Sa. conceived the project. Y.H., Y.O., F.M., K.Sh. and K.Sa. designed the experiments. Y.H. performed plant sample preparation, lipid and RNA extraction, and data analysis. Y.O. carried out lipidomic measurements. Y.H. and K.Sa. wrote the manuscript. All authors commented on the manuscript.
References
- Los D. A., Mironov K. S. & Allakhverdiev S. I. Regulatory role of membrane fluidity in gene expression and physiological functions. Photosynth. Res. 116, 489–509 (2013). [DOI] [PubMed] [Google Scholar]
- Webb M. S. & Green B. R. Biochemical and biophysical properties of thylakoid acyl lipids. Biochim. Biophys. Acta 1060, 133–158 (1991). [Google Scholar]
- Boca S. et al. Arabidopsis lipocalins AtCHL and AtTIL have distinct but overlapping functions essential for lipid protection and seed longevity. Plant Cell Environ. 37, 368–381 (2014). [DOI] [PubMed] [Google Scholar]
- Kunst L., Browse J. & Somerville C. Enhanced thermal tolerance in a mutant of Arabidopsis deficient in palmitic acid unsaturation. Plant Physiol. 91, 401–408 (1989). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murakami Y., Tsuyama M., Kobayashi Y., Kodama H. & Iba K. Trienoic fatty acids and plant tolerance of high temperature. Science 287, 476–479 (2000). [DOI] [PubMed] [Google Scholar]
- Routaboul J. M., Skidmore C., Wallis J. G. & Browse J. Arabidopsis mutants reveal that short- and long-term thermotolerance have different requirements for trienoic fatty acids. J. Exp. Bot. 63, 1435–1443 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J., Burke J. J., Xin Z., Xu C. & Velten J. Characterization of the Arabidopsis thermosensitive mutant atts02 reveals an important role for galactolipids in thermotolerance. Plant Cell Environ. 29, 1437–1448 (2006). [DOI] [PubMed] [Google Scholar]
- Falcone D. L., Ogas J. P. & Somerville C. R. Regulation of membrane fatty acid composition by temperature in mutants of Arabidopsis with alterations in membrane lipid composition. BMC Plant Biol. 4, 17 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miquel M. & Browse J. Arabidopsis mutants deficient in polyunsaturated fatty acid synthesis. Biochemical and genetic characterization of a plant oleoyl-phosphatidylcholine desaturase. J. Biol. Chem. 267, 1502–1509 (1992). [PubMed] [Google Scholar]
- Chapman K. D., Dyer J. M. & Mullen R. T. Commentary: why don’t plant leaves get fat? Plant Sci. 207, 128–134 (2013). [DOI] [PubMed] [Google Scholar]
- Lippold F. et al. Fatty acid phytyl ester synthesis in chloroplasts of Arabidopsis. Plant Cell 24, 2001–2014 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang R., Wise R. R., Struck K. R. & Sharkey T. D. Moderate heat stress of Arabidopsis thaliana leaves causes chloroplast swelling and plastoglobule formation. Photosynth. Res. 105, 123–134 (2010). [DOI] [PubMed] [Google Scholar]
- Degenkolbe T. et al. Differential remodeling of the lipidome during cold acclimation in natural accessions of Arabidopsis thaliana. Plant J. 72, 972–982 (2012). [DOI] [PubMed] [Google Scholar]
- Gasulla F. et al. The role of lipid metabolism in the acquisition of desiccation tolerance in Craterostigma plantagineum: a comparative approach. Plant J. 75, 726–741 (2013). [DOI] [PubMed] [Google Scholar]
- Saito K., Hirai M. Y. & Yonekura-Sakakibara K. Decoding genes with coexpression networks and metabolomics - ‘majority report by precogs’. Trends Plant Sci. 13, 36–43 (2008). [DOI] [PubMed] [Google Scholar]
- Higashi Y. & Saito K. Network analysis for gene discovery in plant-specialized metabolism. Plant Cell Environ. 36, 1597–1606 (2013). [DOI] [PubMed] [Google Scholar]
- Okazaki Y. et al. A chloroplastic UDP-glucose pyrophosphorylase from Arabidopsis is the committed enzyme for the first step of sulfolipid biosynthesis. Plant Cell 21, 892–909 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linka N. et al. Peroxisomal ATP import is essential for seedling development in Arabidopsis thaliana. Plant Cell 20, 3241–3257 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troncoso-Ponce M. A., Cao X., Yang Z. & Ohlrogge J. B. Lipid turnover during senescence. Plant Sci. 205-206, 13–19 (2013). [DOI] [PubMed] [Google Scholar]
- Szymanski J., Brotman Y., Willmitzer L. & Cuadros-Inostroza A. Linking gene expression and membrane lipid composition of Arabidopsis. Plant Cell 26, 915–928 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su K. M., Bremer D. J., Jeannotte R., Welti R. & Yang C. Membrane lipid composition and heat tolerance in cool-season turfgrasses, including a hybrid bluegrass. J. Am. Soc. Hortic. Sci. 134, 511–520 (2009). [Google Scholar]
- Burgos A. et al. Analysis of short-term changes in the Arabidopsis thaliana glycerolipidome in response to temperature and light. Plant J. 66, 656–668 (2011). [DOI] [PubMed] [Google Scholar]
- Gray W. M., Ostin A., Sandberg G., Romano C. P. & Estelle M. High temperature promotes auxin-mediated hypocotyl elongation in Arabidopsis. Proc. Natl. Acad. Sci. USA 95, 7197–7202 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Zanten M., Voesenek L. A., Peeters A. J. & Millenaar F. F. Hormone- and light-mediated regulation of heat-induced differential petiole growth in Arabidopsis. Plant Physiol. 151, 1446–1458 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunst L., Browse J. & Somerville C. A mutant of Arabidopsis deficient in desaturation of palmitic Acid in leaf lipids. Plant Physiol. 90, 943–947 (1989). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaiah S. P. et al. Quantitative profiling of polar glycerolipid species from organs of wild-type Arabidopsis and a phospholipase Dalpha1 knockout mutant. Phytochemistry 67, 1907–1924 (2006). [DOI] [PubMed] [Google Scholar]
- Vu H. S. et al. Lipid changes after leaf wounding in Arabidopsis thaliana: expanded lipidomic data form the basis for lipid co-occurrence analysis. Plant J. 80, 728–743 (2014). [DOI] [PubMed] [Google Scholar]
- Watanabe M. et al. Comprehensive dissection of spatiotemporal metabolic shifts in primary, secondary, and lipid metabolism during developmental senescence in Arabidopsis. Plant Physiol. 162, 1290–1310 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao X., Focke M., Pollard M. & Ohlrogge J. Understanding in vivo carbon precursor supply for fatty acid synthesis in leaf tissue. Plant J. 22, 39–50 (2000). [DOI] [PubMed] [Google Scholar]
- Li-Beisson Y. et al. Acyl-lipid metabolism. Arabidopsis Book 11, e0161 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bates P. D. et al. Fatty acid synthesis is inhibited by inefficient utilization of unusual fatty acids for glycerolipid assembly. Proc. Natl. Acad. Sci. USA 111, 1204–1209 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theodoulou F. L. & Eastmond P. J. Seed storage oil catabolism: a story of give and take. Curr. Opin. Plant Biol. 15, 322–328 (2012). [DOI] [PubMed] [Google Scholar]
- Maruyama K. et al. Metabolic pathways involved in cold acclimation identified by integrated analysis of metabolites and transcripts regulated by DREB1A and DREB2A. Plant Physiol. 150, 1972–1980 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakuma Y. et al. Functional analysis of an Arabidopsis transcription factor, DREB2A, involved in drought-responsive gene expression. Plant Cell 18, 1292–1309 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakuma Y. et al. Dual function of an Arabidopsis transcription factor DREB2A in water-stress-responsive and heat-stress-responsive gene expression. Proc. Natl. Acad. Sci. USA 103, 18822–18827 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu H. C. & Charng Y. Y. Common and distinct functions of Arabidopsis class A1 and A2 heat shock factors in diverse abiotic stress responses and development. Plant Physiol. 163, 276–290 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lightner J., James D. W., Dooner H. K. & Browse J. Altered body morphology is caused by increased stearate levels in a mutant of Arabidopsis. Plant J. 6, 401–412 (1994). [Google Scholar]
- Salas J. J. & Ohlrogge J. B. Characterization of substrate specificity of plant FatA and FatB acyl-ACP thioesterases. Arch. Biochem. Biophys. 403, 25–34 (2002). [DOI] [PubMed] [Google Scholar]
- Moreno-Perez A. J. et al. Reduced expression of FatA thioesterases in Arabidopsis affects the oil content and fatty acid composition of the seeds. Planta 235, 629–639 (2012). [DOI] [PubMed] [Google Scholar]
- Nakamura Y. et al. Arabidopsis lipins mediate eukaryotic pathway of lipid metabolism and cope critically with phosphate starvation. Proc. Natl. Acad. Sci. USA 106, 20978–20983 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura Y., Tsuchiya M. & Ohta H. Plastidic phosphatidic acid phosphatases identified in a distinct subfamily of lipid phosphate phosphatases with prokaryotic origin. J. Biol. Chem. 282, 29013–29021 (2007). [DOI] [PubMed] [Google Scholar]
- Li M., Qin C., Welti R. & Wang X. Double knockouts of phospholipases Dzeta1 and Dzeta2 in Arabidopsis affect root elongation during phosphate-limited growth but do not affect root hair patterning. Plant Physiol. 140, 761–770 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu C., Fan J., Cornish A. J. & Benning C. Lipid trafficking between the endoplasmic reticulum and the plastid in Arabidopsis requires the extraplastidic TGD4 protein. Plant Cell 20, 2190–2204 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z., Xu C. & Benning C. TGD4 involved in endoplasmic reticulum-to-chloroplast lipid trafficking is a phosphatidic acid binding protein. Plant J. 70, 614–623 (2012). [DOI] [PubMed] [Google Scholar]
- Youssef A. et al. Plant lipid-associated fibrillin proteins condition jasmonate production under photosynthetic stress. Plant J. 61, 436–445 (2010). [DOI] [PubMed] [Google Scholar]
- Kaup M. T., Froese C. D. & Thompson J. E. A role for diacylglycerol acyltransferase during leaf senescence. Plant Physiol. 129, 1616–1626 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly A. A. et al. The sugar-dependent1 lipase limits triacylglycerol accumulation in vegetative tissues of Arabidopsis. Plant Physiol. 162, 1282–1289 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pracharoenwattana I., Cornah J. E. & Smith S. M. Arabidopsis peroxisomal citrate synthase is required for fatty acid respiration and seed germination. Plant Cell 17, 2037–2048 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okazaki Y., Kamide Y., Hirai M. Y. & Saito K. Plant lipidomics based on hydrophilic interaction chromatography coupled to ion trap time-of-flight mass spectrometry. Metabolomics 9, 121–131 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.